Abstract
To assess if magnetic resonance spectroscopy (MRS)-measured Glutamate (Glu) and GABA reflect excitatory and inhibitory neural activities, respectively, we conducted MRS measurements along with two-photon mesoscopic imaging of calcium signals in excitatory and inhibitory neurons of living, unanesthetized mice. For monitoring stimulus-driven activations of a brain region, MRS signals and mesoscopic neural activities were measured during two consecutive sessions of 15-min prolonged sensory stimulations. In the first session, putative excitatory neuronal activities were increased, while inhibitory neuronal activities remained at the baseline level. In the second half, while excitatory neuronal activities remained elevated, inhibitory neuronal activities were significantly enhanced. We assessed regional neurochemical statuses by measuring MRS signals, which were overall in accordance with the neural activities, and neuronal activities and neurochemical statuses in a mouse model of Dravet syndrome under resting condition. Mesoscopic assessments showed that activities of inhibitory neurons in the cortex were diminished relative to wild-type mice in contrast to spared activities of excitatory neurons. Consistent with these observations, the Dravet model exhibited lower concentrations of GABA than wild-type controls. Collectively, the current investigations demonstrate that MRS-measured Glu and GABA can reflect spontaneous and stimulated activities of neurons producing and releasing these neurotransmitters in an awake condition.
Introduction
Proton magnetic resonance spectroscopy (MRS) is a noninvasive technique that can measure the concentrations of glutamate (Glu; chief excitatory neurotransmitter) and gamma-aminobutyric acid (GABA; chief inhibitory neurotransmitter) within a region of interest in the brain. Since the first human application in the 1980s, there has been a growing interest in the use of MRS to correlate cognition and behavior with the concentrations of Glu and GABA in specific brain regions.1–5 For a better understanding of the underlying metabolic processes of brain activity, many studies were conducted both in humans and rodents to tackle this question: do MRS-measured Glu and GABA in a brain region reflect activities of excitatory and inhibitory neurons, respectively, in the region? This question has been explored in human imaging studies. There have been many studies in which brain activity, as measured by non-invasive function measures such as functional MRI (fMRI)6,7 or magnetoencephalography (MEG), 8 has been related to MRS neurotransmitter measures. 9 Those studies are rationalized based on the notion that proton MRS allows measurements of Glu and GABA pools that reflect neural activities in humans, and that synaptic concentrations of MRS-measured Glu and GABA are not distinguishable. Here, one open question remains unsolved: how MRS can distinguish Glu and GABA during brain activation? This uncertainty has been one of the main limitations to understanding the relationships between behavior and brain processing including metabolism that underly MRS signals. To address those questions, linking the activity of subtypes of neurons to MRS-measured Glu and GABA could be accomplished using two-photon calcium imaging and functional MRS (fMRS).
Owing to developments in two-photon imaging and genetic tools, it is now possible to monitor the activity of many neurons in the brain of awake behaving animals. Two-photon imaging can monitor neural activities, since fluorescent calcium indicators are used as a readout of spiking activity, which is attributed to the association between action potentials and large/fast increase of intracellular free calcium concentration. 10 Moreover, using genetically encoded calcium indicators, the same population of neurons can be monitored, allowing the investigation of activities in excitatory and inhibitory neurons. Thus, by using two-photon calcium imaging, we can link the activity of excitatory and inhibitory neurons with MRS-measured Glu and GABA, respectively. To our knowledge, there has been no neuroscientific study that aimed to link between neural activities at the cellular level and MRS-measured Glu and GABA. In rodents under anesthetic condition, several proton fMRS studies have been conducted, resulting in the Glu increase during activation.11–13 On the other hand, the experiments identifying GABA changes during fMRS in rodents have been scarce. 13 In the fMRS studies, since most of them were conducted with anesthesia, 14 GABA measurements are generally poorly reliable.
Numerous evidence has shown significant influence of anesthesia on neural activity.15,16 Despite the importance of the awake condition for investigating neural activity and brain function, to our knowledge, no MRS work in rodents has been performed in an awake condition. Thus, to satisfactorily answer the above question, it is essential to establish a link between neural activities and MRS signals in awake and behaving conditions.
This study aims to test the hypothesis that MRS-measured Glu and GABA concentrations reflect activities of excitatory and inhibitory neurons, respectively, in an awake condition. We specifically focused on two types of awake conditions: task and quiet rest. Several studies in cognitive neuroscience have measured MRS signals in association with certain tasks.1–4,17,18 On the other hand, clinical studies have measured MRS signals mainly at quiet rest.19–21 If the hypothesis is correct, MRS-measured Glu and GABA should reflect both task-evoked and spontaneous activities of excitatory and inhibitory neurons, respectively.
To test this hypothesis, we utilized the following two cutting-edge modalities: a MRS compatible awake mouse restraint device and a large-field-of-view two-photon mesoscope with subcellular resolution, which has the advantage for measuring neural activities from a large number of neurons simultaneously. 22 In a task condition (Experiment 1), we measured time-courses of calcium signals and MRS during a 30-min whisker stimulation, since MRS-measured Glu and GABA in brain areas were found to be changed over the course of 20–30-min task loads.1,23 To examine the relationships between MRS signals and neural activities at quiet rest (Experiment 2), we used a mouse model of Dravet syndrome, which is a severe epileptic encephalopathy from de novo mutations of SCN1A. 24 It is known that activities of inhibitory neurons are specifically reduced in these mice compared to wild-type mice, 24 allowing us to test whether the GABA concentration, but not the Glu concentration, is specifically decreased in these model mice.
Methods
Animals
C57BL/6
All mice were housed individually in separate cages with water and food ad libitum (n = 27 (20 male, 7 female, one was excluded due to high Glutamine concentration 25 ) body weight 27.7 ± 1.97 g, 2.9 ± 0.2 months of age (mean ± standard deviation)). Mouse cages were kept at 25 °C in a 12-h light/dark cycle. Prior to two-photon imaging and MRI/MRS scanning, all mice used for each experiment were habituated daily to the two-photon microscope or the scanner for five days (5–10 min/day) to minimize handling stress. The levels of stress during the two-photon microscopy and MR experiments were monitored by moving frequency. Different mice were used for two-photon imaging and MRI/MRS scanning in order to avoid influence of the prior experiment on the next experiment.
A mouse model of dravet syndrome
Balb/c-Scn1a<+/−> mouse (Dravet mouse) was provided by RIKEN BRC through the National Bio Resource Project of MEXT, Japan (BRC No.: RBRC06422) 26 (n = 18 (9 male, 9 female), body weight 26.7 ± 1.1 g, 4.4 ± 0.5 months of age (mean ± standard deviation)). For in vivo two-photon imaging, heterozygous mutant and wild-type control mice (6–7 months old) were generated by crossing once with the C57BL/6 background (n = 13 (7 male, 6 female), body weight 27.4 ± 0.78 g, 6.5 ± 0.4 months of age).
All animal experiments were approved by the Institutional Animal Care and Use Committee of the National Institute of Radiological Sciences (Chiba, Japan) and were performed in accordance with the institutional guidelines on human care and use of laboratory animals approved by the Institutional Committee for Animal Experimentation. All the animal experiments were reported following the ARRIVE guidelines 2.0 (Animal Research: Reporting of In Vivo Experiments). 27
MRI/MRS-compatible awake mouse restraint device and whisker stimulation
To measure the levels of neurochemicals from an awake mouse by MRS, we developed an MRI/MRS compatible awake mouse restraint device to hold the skull of the mouse tightly on the holder (Figure 1(a)). The fixation method of a mouse head is in principle identical to the one for two-photon microscopy. 28 The surgery was performed two weeks before the MRI and MRS measurements. Then after recovery from surgery, the mice were habituated to the mouse restraint device for five days. The homemade holder for the MRI scanner was optimized for adjusting the space in the bore of the 7 T magnet with cryoprobe for mice. Whisker stimulation with air-puffs is available on this holder (Figure 1(b)). Almost all whiskers were moved by the air-puffs from this tool. To give sufficient stimulation to the mouse brain, multiple whiskers on the right side were stimulated together using the air-puff for 1 sec at a rate of once every 3 sec.
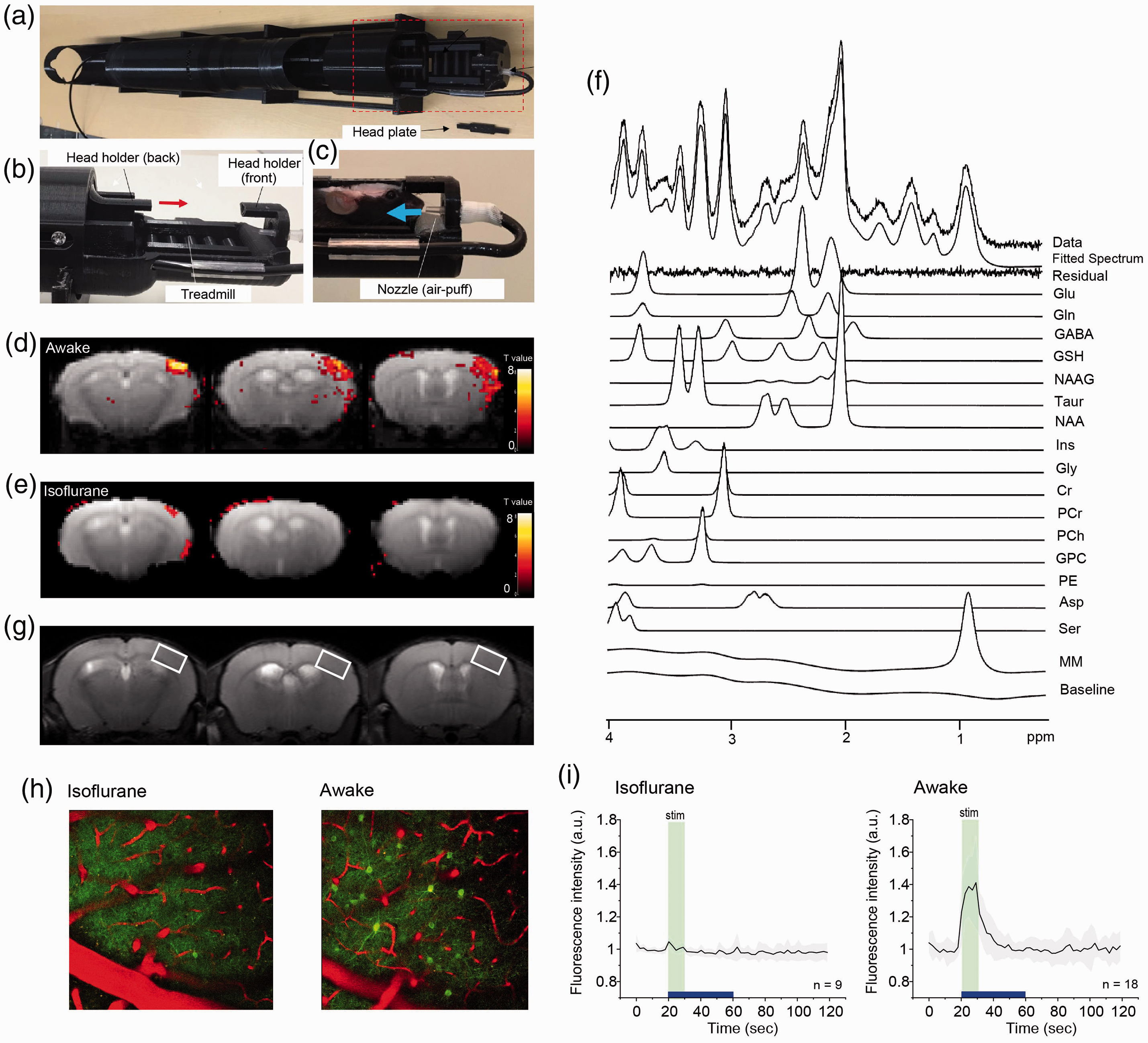
Pictorial features of the awake mouse restraint device and representative spectrum (a) An overview of the MRI/MRS-compatible awake mouse restraint device mouse holder is shown. The region within the red-dotted rectangle is magnified in B. (b, c) Two parts (front and back) of the head plate, which is fixed on the mouse head, are inserted into the mouse holder to prevent movement of the mouse. (c) Whisker stimulation by air puff is possible for a mouse on the holder. (d) BOLD signals during whisker stimulation are shown on the barrel cortex. (e) Under aneasthetic condition, BOLD signals were less apparent than in the awake condition. (f) A representative spectrum at the awake condition, the corresponding LCModel spectral fit, fit residual, macromolecules (MM), baseline and individual metabolite fits were demonstrated. (g) A representative voxel placement on the barrel cortex for MRS is shown. (h, i) Whisker stimulation on mice was performed for 10 sec by air-puff at 1 Hz. While no significant alterations of calcium signal intensities were found under anesthetic condition (h, i left), alterations of calcium signal intensities during the awake condition were evident (h, i right).
MRI and MRS measurements
C57BL/6
MRI and MRS were performed on a 7.0 T MRI scanner (20 cm bore, Biospec, Avance-III system; Bruker Biospin, Ettlingen, Germany) with a 2-channel phased-array cooled surface coil for transmission/reception (cryoprobe for mouse brain, Bruker Biospin). For awake-mouse experiments, mice were lightly anesthetized (1–2% isoflurane in air; 50: 50 at 1 L/min) for <3 min while being placed in the MRI/MRS-compatible awake-mouse restraint device. After moving the mouse on the holder, anesthesia was turned off. Axial and sagittal multi-slice TurboRARE T2-weighted images (Axial: TR = 3000 ms, TE = 12 ms (effective TE = 36 ms), Rare Factor = 8, number of averages = 1, number of slices = 20, FOV = 19.2 mm × 19.2 mm, matrix = 256 × 256; Sagittal: TR = 3000 ms, TE 10 ms (effective TE = 30 ms), Rare Factor = 8, number of averages = 1, number of slices = 13, FOV = 19.2 mm × 12.8 mm, matrix = 128) were acquired. To confirm that the VOI of MRS in the barrel cortex is appropriately localized, functional MRI (fMRI) was performed under whisker stimulation at a rate of 2 Hz with 1-sec stimulation using a gradient-echo echo-planar sequence (TR = 500 ms, TE = 15 ms, BW = 250 kHz, number of averages = 2, number of slices = 13, spatial resolution = 200 μm × 200 μm × 1000 μm, FOV = 19.2 × 19.2 mm, matrix = 96 × 96). The stimulus was delivered using a block design paradigm of 100-sec rest and 20-sec activation alternately repeated five times. Once we confirmed the barrel cortex on the brain of C57BL/6J mice with the exploratory fMRI experiment, we chose the VOI anatomically by following the representative fMRI results (Figure 1(d) and (g)). Magnetic resonance spectra were acquired from a VOI centered in the left barrel cortex. The size of the voxel was 1.175 mm × 2.0 mm × 3.5 mm. Localized MRS was applied using Point Resolved Spectroscopy (PRESS) with TE/TR = 11/3000 ms, 292 averages (saved in a block). Water signal was suppressed using a VAPOR module. MRS measurements were repeated three times in a row. At first, MRS scan was performed without whisker stimulation. Then, two more MRS scans (1st half and 2nd half) were performed with whisker stimulation at 0.333 Hz with 1-s pulse duration and 2-sec interval. The MRS protocol is shown in Figure 3(a). For experiments under isoflurane anesthesia, induction was performed with 3% anesthesia, and it was then maintained with 1.5 ± 0.3% isoflurane during the experiments. The rest of the manipulations were the same between awake mouse and anesthetized mouse experiments.
A mouse model of Dravet syndrome
Heterozygous mutant and wild-type mice (2-6 months old) were used for the MRS measurements in the awake condition. Magnetic resonance spectra were acquired from a VOI centered in the middle frontal cortex. The voxel size was 1.2 mm × 2.5 mm × 3.0 mm. Localized MRS was applied using PRESS with TE/TR = 11/4000 ms, 192 averages.
fMRI data analysis
fMRI images were created using a statistical parametric mapping package (SPM8; www.fil.ion.ucl.ac.uk/spm). Image processing steps, including (1) realigning and reslicing all EPI images within a time-series, based on their averaged image, to minimize movement artifacts, and (2) smoothing the images by using a Gaussian kernel with full width at half maximum (FWHM) of 0.5 mm, were applied to the raw data. Statistical analysis was conducted using general linear modeling with a hemodynamic response function. To control the probability of false positive clusters below 0.05, we determined the significant BOLD response areas with an individual voxel threshold of P < 0.05. This analysis was performed after the fMRI experiment.
MRS data analysis
Acquired spectra were analyzed using LCModel (Stephen Provencher Inc, Oakville, Ontario, Canada). The unsuppressed water signal measured from the same VOI was used as an internal reference for the absolute quantification of metabolites. Data were fitted to a linear combination of 21 metabolites, without preprocessing, in a simulated basis set containing: alanine, aspartate (Asp), phosphorylcholine (PCh), creatine (Cre), phosphocreatine (PCr), GABA, glutamine (Gln), Glu, glutathione (GSH), glycine (Gly), myo-inositol (mI), lactate (Lac), N-acetylaspartate (NAA), Scyllo-inositol (Scyllo), taurine (Taur), glucose (Glc), N-acetylaspartylglutamate (NAAG), glycerophosphorylcholine (GPC), phosphorylethanolamine (PE), serine (Ser) and β-hydroxybutyrate (bHB).
Plasmid construction and AAV preparation
cDNA encoding GCaMP6s, an ultra-sensitive genetically encoded fluorescent calcium indicator, 29 was amplified by polymerase chain reaction from pGP-CMV-GCaMP6s plasmid (a gift from Douglas Kim, Addgene plasmid # 40753) and inserted into the multi-cloning site of Adeno Associated Virus (AAV) transfer plasmid with rat synapsin promoter (pAAV-Syn), woodchuck posttranscriptional regulatory element (WPRE), and polyA signal flanked with ITRs. To generate AAV plasmid drive mCherry expression in inhibitory neurons, the promoter was substituted by mDlx enhancer sequence from pAAV-mDlx-GFP-Fishell-1 (a gift from Gordon Fishell, Addgene plasmid # 83900) and cDNA encoding mCherry was further inserted into the multi-cloning site. 30 For large scale preparation of recombinant AAV, each AAV transfer plasmid and AAV serotype DJ packaging plasmids (pHelper and pRC-DJ) were introduced into HEK293T cells with polyethyleneimine transfection. 48 h after transfection, cells were harvested, lysed, and AAV particles were purified with HiTrap heparin column (GE Healthcare) as described previously. 31
Wide-field two-photon imaging in mice
AAV vector driving GCaMP6s gene (AAV-Synapsin) and DLX (AAV-DLX) were co-injected into the barrel cortex two months before the experiment with two-photon imaging. For implantation of the cranial window, the animals were anesthetized with a mixture of air, oxygen, and isoflurane (3–4% for induction and 2% for surgery) via a facemask, and a cranial window (3–4 mm in diameter) was attached over the left somatosensory cortex, centered at 1.8 mm caudal and 2.5 mm lateral from the bregma, according to the ‘Seylaz-Tomita method’. 32 All two-photon imaging experiments were performed four weeks after the cranial window surgery. The method for preparing the chronic cranial window was previously reported elsewhere.28,33 The awake animal was placed on a custom-made apparatus, and real-time imaging was conducted by wild-field two-photon laser scanning microscopy (Multiphoton Mesoscope, THORLABS, NJ, USA). In this study, two laser oscillators were used to efficiently separate red and green fluorescence (Supplementary Figure 1). One is a commercially available laser oscillator (Mai Tai, Spectra-Physics, CA, USA) and the other, a Yb-doped fiber laser system, was used as a pump laser for the two-photon microscope. The latter laser system was home-made and consisted of a mode-locked fiber oscillator, multi-stage fiber amplifiers and a pulse compressor. We used a nonlinear amplification method in the amplifiers.34–36 The laser had a pulse repetition frequency of 73 MHz, a central wavelength of 1036 nm, a pulse length (full width at half maximum) of 130 fs and an output power of 5 W. Excitation wavelength of 900 nm (commercial laser oscillator) and 1036 nm (home-made laser system) were used for simultaneous measurements of GCaMP and DLX, respectively. An emission signal was separated by a beam splitter (560/10 nm) and simultaneously detected with a band-pass filter for GCaMP (525/50 nm) and mCherry (610/75 nm). The visual field sizes of the image were 500 μm × 500 μm and 3000 μm × 3000 μm, and in-plane pixel size was 1 µm. Temporal resolution was 1–4 Hz for 60 sec (60–240 frames/trial) depending on the size of the field of view. Images were acquired at a depth of about 150 μm from the brain surface.
All the other experiments by two-photon imaging were performed in awake mice, since the suppression of neural activities measured by calcium signals was apparent by 1.5% isoflurane. The experimental protocol for measurements by two-photon imaging was reported previously.15,33 Briefly, the handmade fixing device on the animal's head was screwed to a custom-made stereotactic apparatus. The animal was then placed on a styrofoam ball that was floating using a stream of air. This allowed the animal to exercise freely on the ball while its head was fixed to the apparatus.
Image analysis of two-photon microscopy
Image analysis of two-photon calcium imaging was performed using Matlab (MathWorks, MA, USA). For analysis of sensory stimulation experiments, data on the timing when the mouse moved were first removed. Next, motion correction was performed using NoRMCorre. The images for all time points were divided by the first image, and the images of signal change rate were obtained. Region of interests (ROIs) were manually drawn on the image obtained by maximum intensity projection (MIP) processing for images of the sensory stimulation period. The time-dependent change in average luminance value was then obtained for each ROI.
Statistical analyses
Results are presented as mean ± standard deviation. Normality testing was performed, all data passed normality (Shapiro-Wilk test), and then parametric statistical analysis was performed. Data were analyzed in OriginPro 2019 (OriginLab, Northampton, MA, USA) with two-sample, paired t-test or one-way repeated-measures ANOVA with Bonferroni post-hoc test, when appropriate. P < 0.05 was considered significant, except where otherwise indicated.
Results
Differential neural activities at awake and anesthetic conditions
We first confirmed this assumption in our study: neural activities in the awake condition are different from those in the anesthetic condition. 1-Hz whisker stimulation, administered via air-puffs, was performed for 10 sec on a mouse while its skull was tightly fixed by a homemade awake mouse restraint device (Figure 1(a) to (c)). Two-photon calcium imaging of neurons in the barrel cortex was performed on the head-fixed mice during awake condition and under anesthesia (isoflurane) 15 (Figure 1(h)). Next, we identified the area of the barrel cortex that responds to whisker stimulation using fMRI. 37 As shown in Figure 1(d) and (e), the region in the somatosensory cortex was activated by whisker stimulation. Based on this data, we localized a voxel of interest (VOI) that was used for the following MRS measurements (Figure 1(g) and (f)). While no significant increase of calcium signal intensities in neurons was found in the anesthetic condition (Figure 1(h) and (i), left), we found an increase in calcium signal intensities in the awake condition in response to whisker stimulation (Figure 1(h) and (i), right). These differences clearly indicate that neural activities are different between the awake and anesthetic conditions. In the case of Figure 1(i), ten trials were successively performed with an inter-trial interval of 60 sec both at anesthetic and awake conditions. After 30 sec from the start of the scan, activation of neurons was induced by sensory stimulation with an air puff for 10 sec.
Quality of MRS measurements and metabolites information
The average Cramér-Rao lower bounds (CRLB) values of the following 10 metabolites were <15%: Asp, GABA, Gln, Glu, GSH, mI, Taur, total choline (tCho; the sum of GPC and PCh), total NAA (tNAA; the sum of NAA and NAAG) and total Cre (tCre; the sum of Cre and PCre). Since we focused on excitatory and inhibitory neuronal activities, Glu and GABA were utilized for the further analysis. The other information from measured metabolites is shown in a Tables 1 to 3. Mean correlation matrices between metabolite concentrations for Glu and GABA, and other metabolites are shown in Supplementary Table 1. The comparison of metabolite concentrations of the baseline at anesthetic and awake conditions is demonstrated in Supplemental Table 2. There was a significant difference of tNAA concentrations between the anesthetic and awake conditions at the baseline (p < 0.001).
Metabolite concentrations (μmol/g) of the pre-stimulation (pre), the first half and second half stimulations are demonstrated.
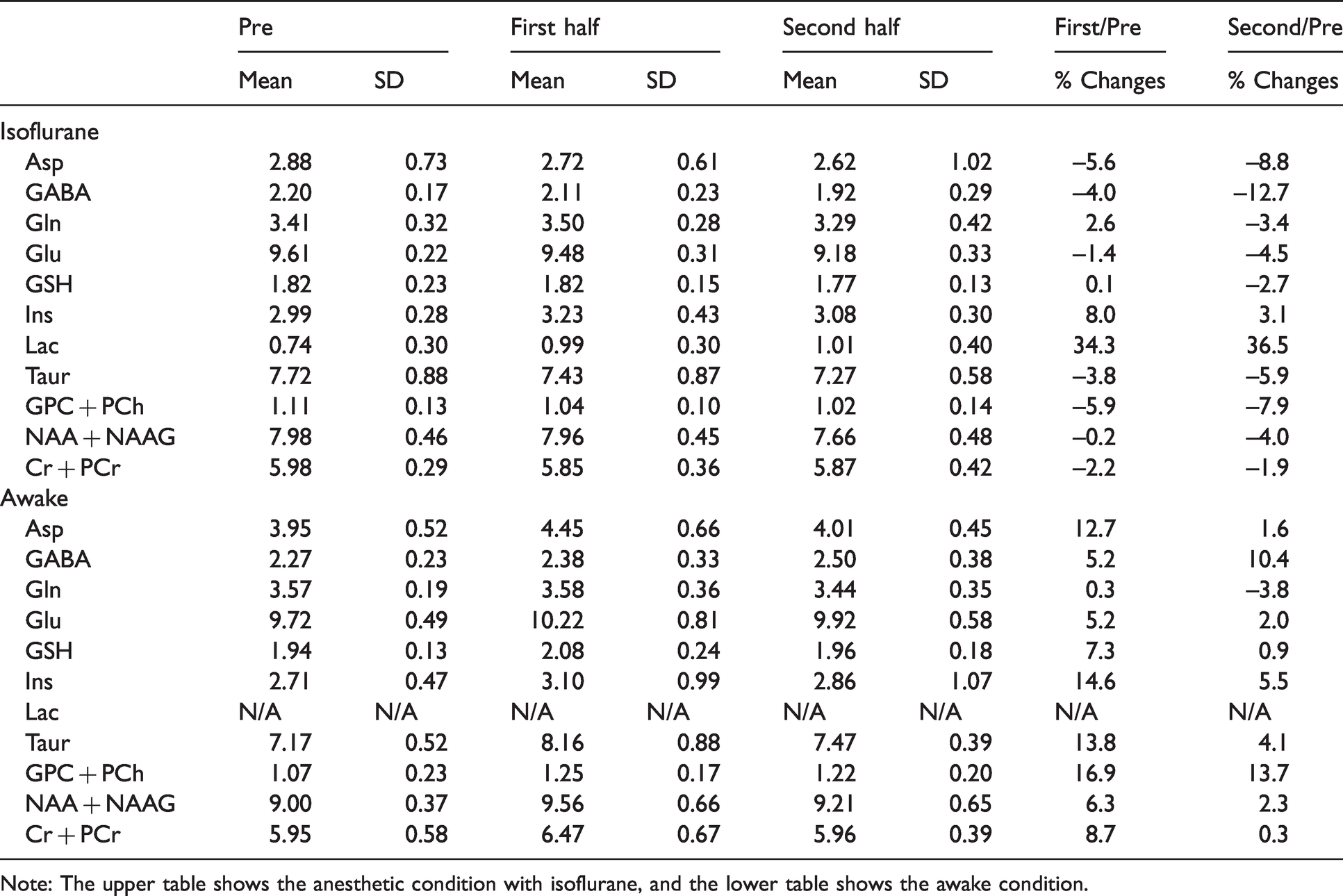
Note: The upper table shows the anesthetic condition with isoflurane, and the lower table shows the awake condition.
Cramér-Rao lower bound (CRLB) values of the metabolites are shown.
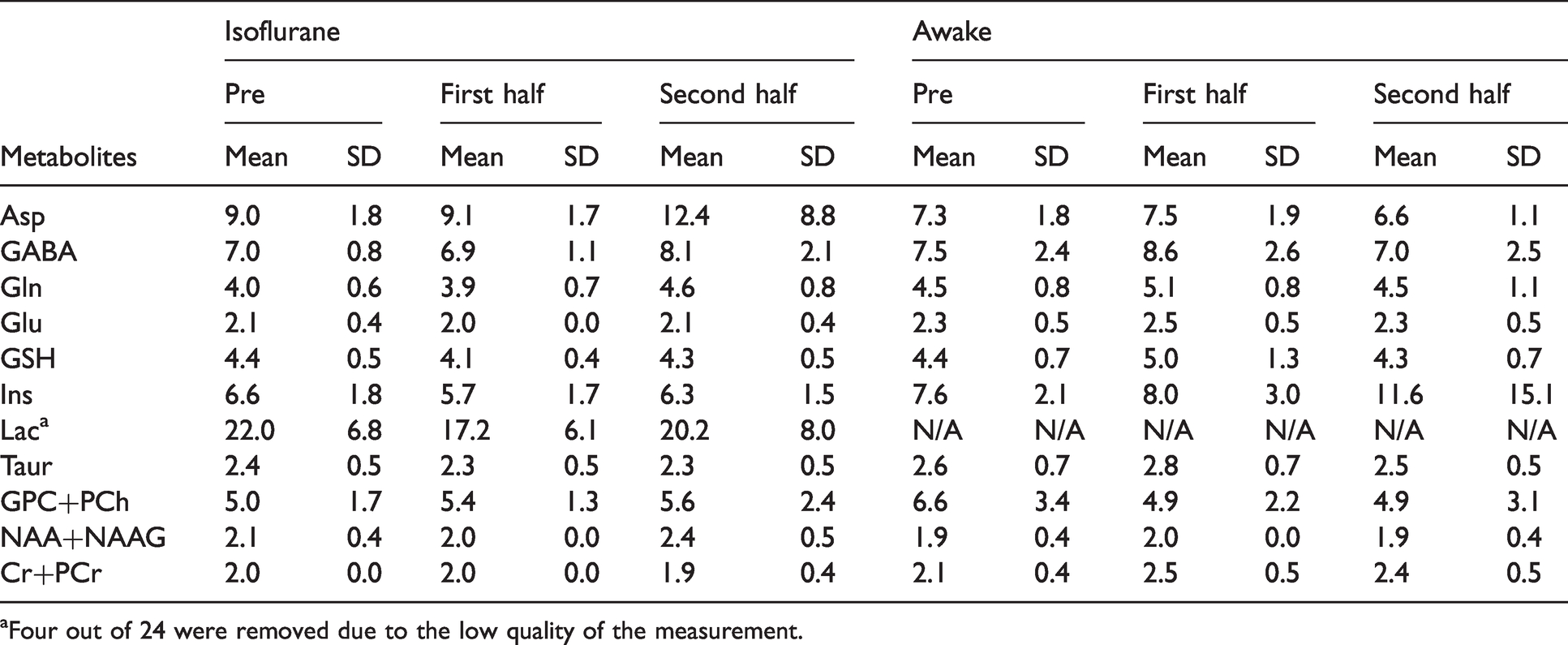
aFour out of 24 were removed due to the low quality of the measurement.
Signal to noise ratio (SNR) and full width at half maximum (FWHM) values are shown.
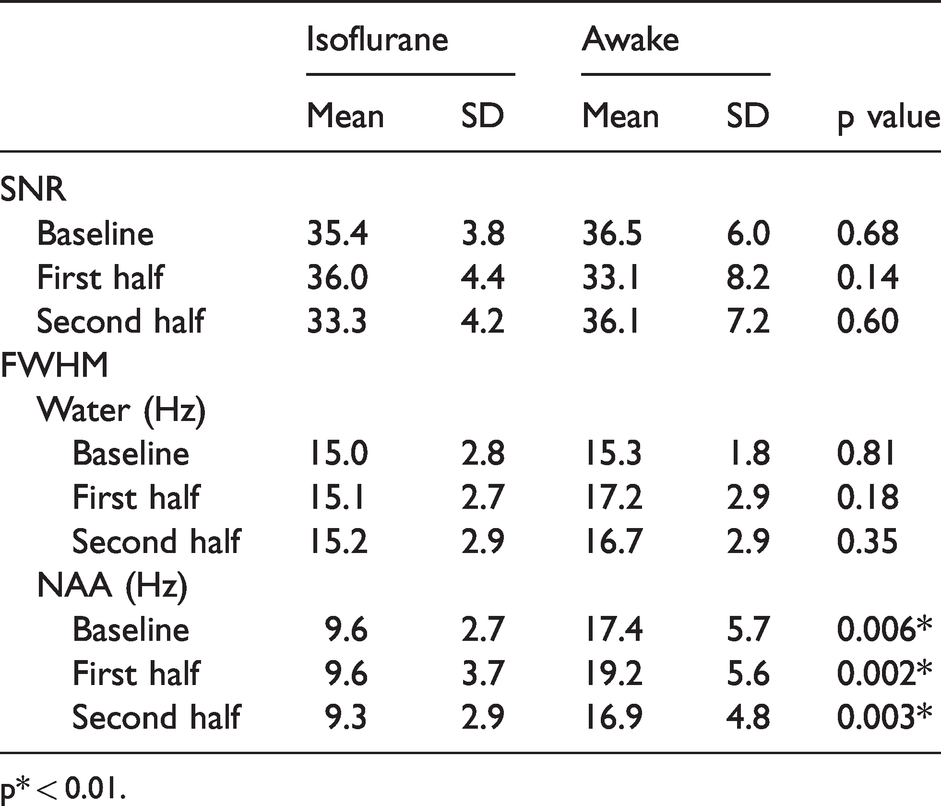
p* < 0.01.
Activities of excitatory and inhibitory neurons changed in accordance with changes in MRS-measured Glu and GABA concentrations, respectively, during prolonged whisker stimulation
To test whether MRS-measured Glu and GABA reflect excitatory and inhibitory neural activities, respectively, we applied the identical whisker stimulation protocol to both two-photon imaging and MRS experiments. To provoke changes of excitatory and inhibitory neural activities in the barrel cortex (Figure 2(a)), we applied whisker stimulation (1-sec air-puff at 0.333 Hz frequency) for 30-min. The time duration was designed based on previous reports in which MRS-measured Glu and GABA in the cortex could be differentially altered over the course of 20–30-min task loads.1,23 Before the stimulation, 10- to 15-min baseline measurements were performed in both two-photon imaging and MRS measurements. Data obtained from this pre-stimulation period served as a baseline.
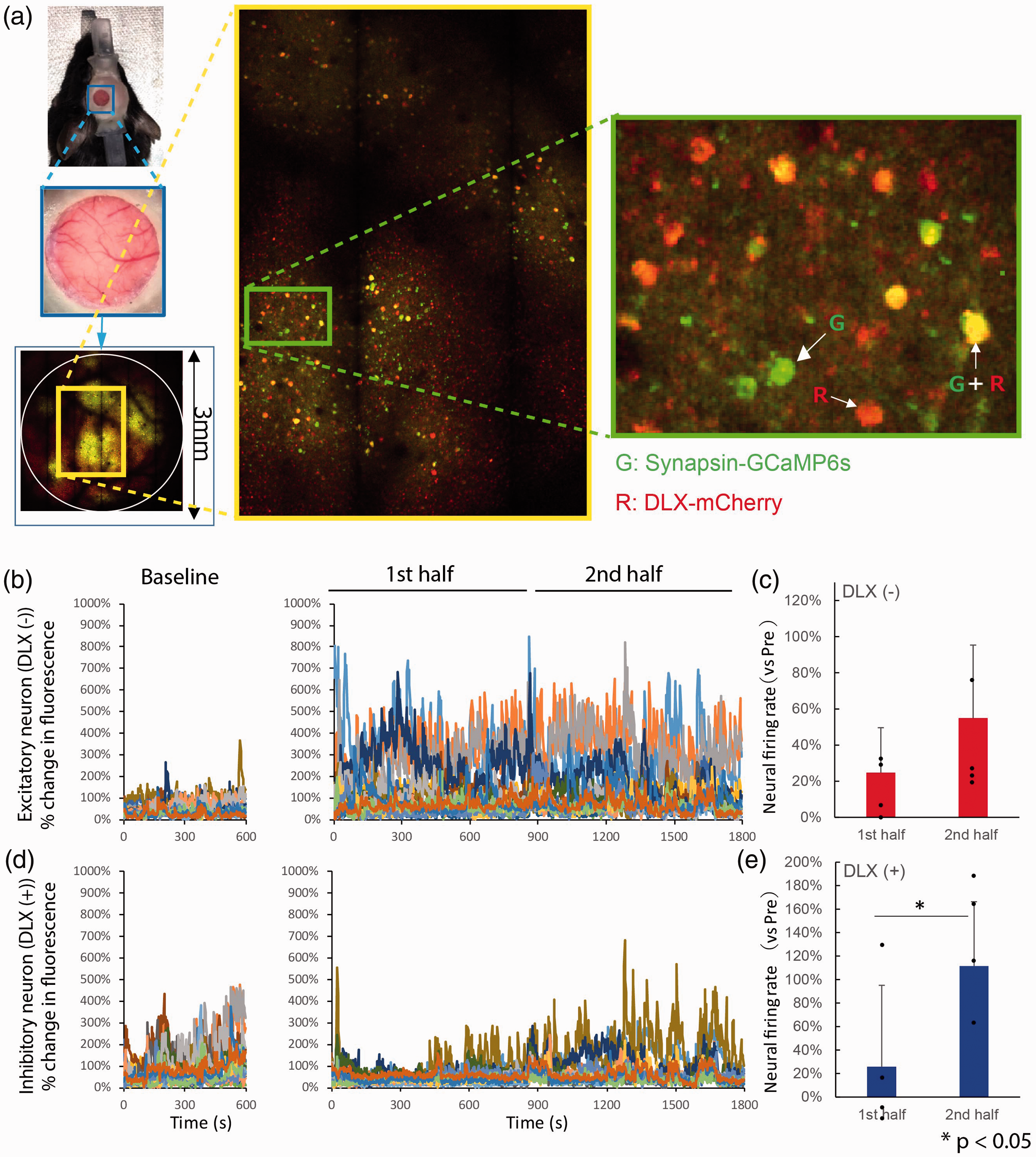
Neural activities measured by two-photon laser microscopy at the awake condition.
As shown in Figure 2 (3000 μm × 3000 μm image), 60-sec images were made 15 times (total 30 min), once per min. Immediately after the start of the scan, air puff stimulation was given continuously for 30 min, once every 3 sec. The patterns of whisker stimulation were the same condition as for the MRS experiments. During the first half of whisker stimulation (15 min), activities of putative excitatory neurons were increased from baseline (Figure 2(b) and (c)). The activities stayed at an increased level in the second half, resulting in an insignificant difference between the first and second half sessions (paired t-test, n = 4 mice, t = −1.971, p = 0.143). On the other hand, the activities of inhibitory neurons became elevated from baseline only during the second half (paired t-test, n = 4 mice, t = −3.354, p = 0.044) (Figure 2(d) and (e)). These results indicate that the prolonged whisker stimulation protocol successfully led to changes in the activities of excitatory and inhibitory neurons at the different time points.
Using the same stimulation protocol, we performed MRS to confirm the link between changes in excitatory and inhibitory neural activities and changes in the concentrations of MRS-measured Glu and GABA, respectively. Three 15-min MRS sessions were conducted consecutively (Figure 3(a)) ⎼ the baseline acquisition without stimulation, and the first half and second half acquisitions with whisker stimulation. Representative spectra in the awake condition were shown (Figures 1(f) and 3(b)). All of the fitted spectra by LCModel 38 are presented in Supplementary Figure 2.
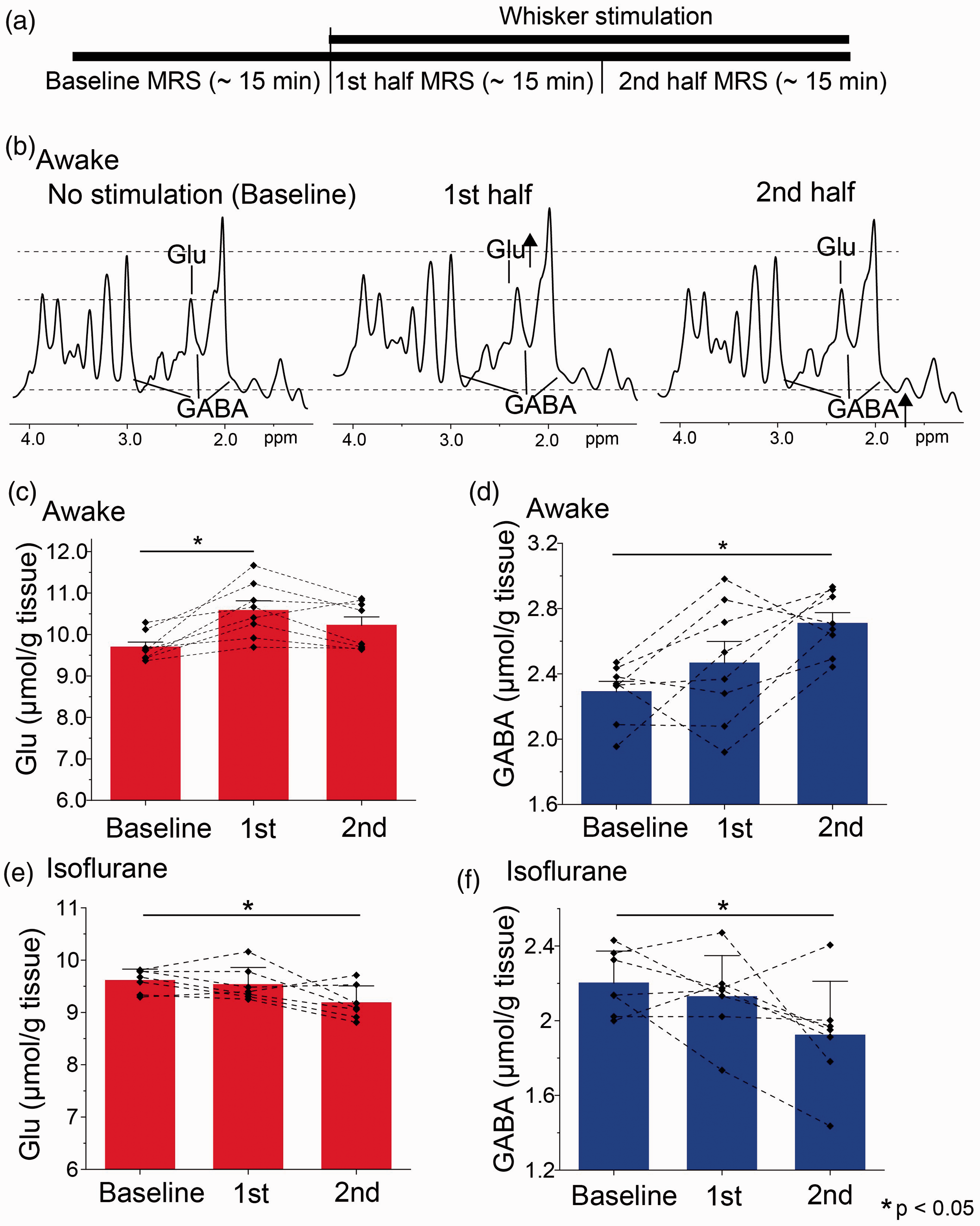
Alterations of Glu and GABA levels during whisker stimulation measured by MRS.
In the first half of the stimulation (15 min), a significant increase of Glu levels was observed (one-way repeated-measures ANOVA with Bonferroni post-hoc test (rm-ANOVA), n = 8, t = 4.288, p = 0.002) without significant alteration of the GABA levels (rm-ANOVA, n = 8, t = 1.505, p = 0.155). In the second half, the GABA levels were increased significantly compared to baseline (rm-ANOVA, n = 8, t = 3.589, p = 0.003), while the Glu levels were slightly reduced versus the 1st half and did not different from baseline (Figure 3(c)). While there were slight differences regarding excitatory neural activities and MRS-measured Glu at the 2nd half, these results indicate that the changes in MRS-measured Glu and GABA concentrations were overall in accordance with the changes in excitatory and inhibitory neural activities, respectively, in the awake condition. In contrast to those findings, the Glu and GABA levels under anesthetic condition did not increase by whisker stimulation, and in fact they rather decreased in the second half (Glu; rm-ANOVA, n = 8, t = 3.055, p = 0.030, GABA; rm-ANOVA, n = 8, t = 2.737, p = 0.018) (Figure 3).
Weaker activities of inhibitory neurons are associated with lower concentrations of MRS-measured GABA during quiet rest
Next, we tested whether MRS-measured Glu and GABA during quiet rests reflect spontaneous activities of excitatory and inhibitory neurons, respectively. We employed an epilepsy mouse model of Dravet syndrome, which is known to have dysfunction of inhibitory neurons due to a gene mutation of SCN1A. 24 We found no significant changes in the spontaneous activities of excitatory neurons as measured by two-photon microscopy (two-sample t-test, n = 219–356 cells, t = 0.9315, p = 0.3519; Figure 4(a) and (b)) or MRS-measured Glu levels (two-sample t-test, n = 8, t = 0.066, p = 0.948; Figure 4(c)) between the model and wild-type mice. On the other hand, we found that spontaneous activities of inhibitory neurons in the model mice were significantly lower than those in wild-type mice (Two-sample t-test, n = 41–61 cells, t = −4.168, p = 6.628E-5; Figure 4(d) and (e)). Consistent with these decreased activities, the model mice also showed lower GABA levels in gray matter than wild-type mice at the resting state (two-sample t-test, n = 9, t = 2.9935, p = 0.008; Figure 4(f)). These results indicate that MRS-measured Glu and GABA reflect excitatory and inhibitory neural activities, respectively, under quiet rest condition. The other metabolites information is demonstrated in the Supplemental Table 3.
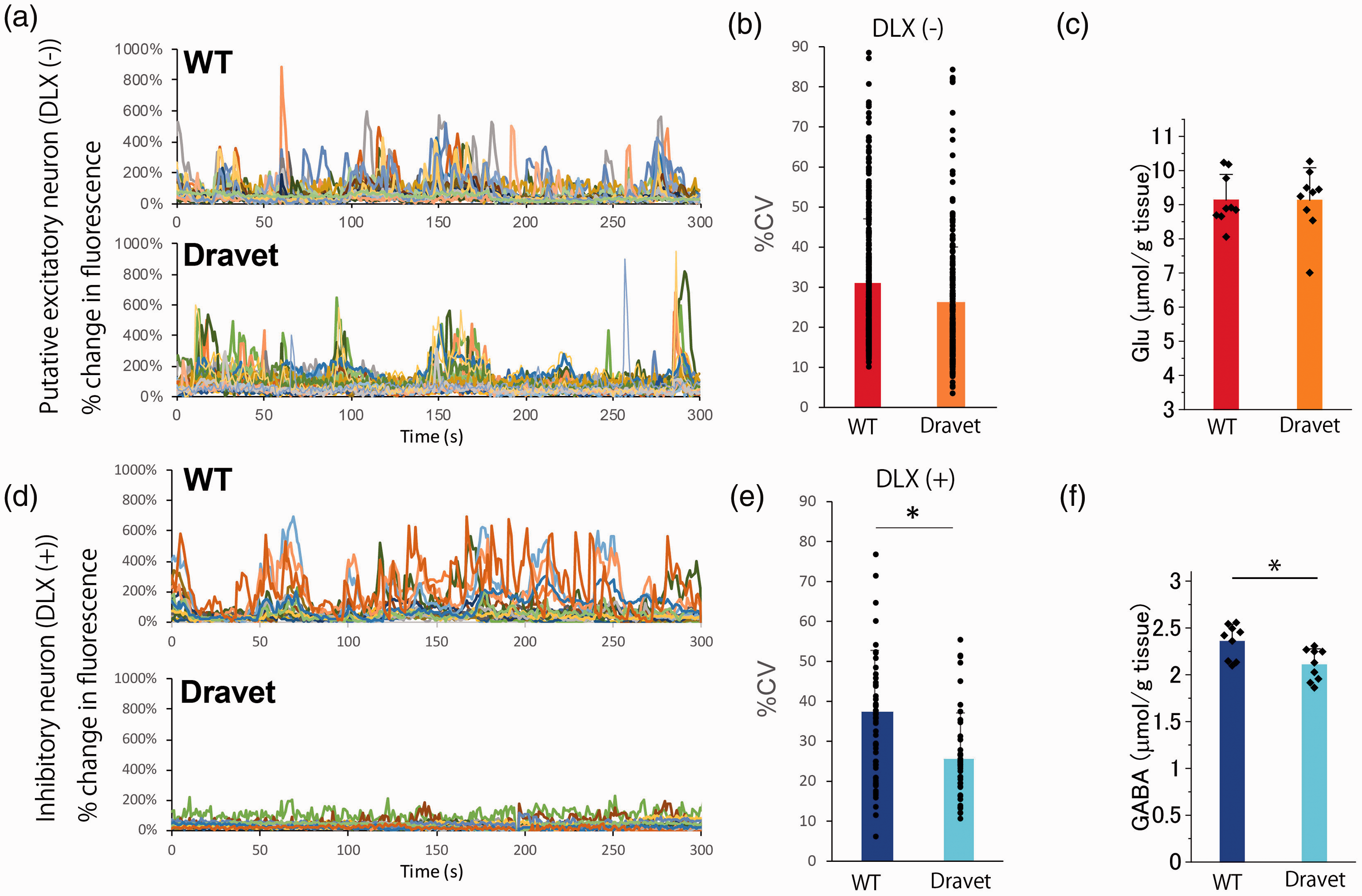
MRS-measured Glu and GABA and neural activities in a mouse model of Dravet syndrome with spontaneous brain activities (A, B, C) Calcium signals of putative excitatory neurons imaged by two-photon microscopy showed no significant difference between the two groups (paired t-test, n = 4 mice, t = −1.971, p = 0.143), which was in accordance with the unchanged MRS-measured Glu levels (two-sample t-test, n = 9, t = 0.01984, p = 0.984; Figure 4(c)). (D, E, F) Calcium signals of inhibitory neurons decreased significantly in a mouse model of Dravet syndrome (two-sample t-test, n = 41–62 cells, t = −4.168, p = 6.628e-5), which was in accordance with the results of MRS-measured GABA (two-sample t-test, n = 9, t = 2.9935, p = 0.008; (F)). In the Figure 4(b) and (e), the percent coefficient of variance (%CV), which is defined as the ratio of standard deviation (SD) to the mean of fluorescence intensities, is shown. Error bars indicate standard deviation.
Discussion
Despite the increasing number of findings based on MRS, one open question remains unresolved: how can MRS distinguish between Glu and GABA during brain activation? This uncertainty has been one of the main limitations to understanding the relationship between behavior and brain processing that underlies MRS signals. Using MRS and two-photon microscopy in the mouse brain, the present work collectively demonstrated that MRS-measured Glu and GABA levels can reflect excitatory and inhibitory neural activities, respectively, in the awake and behaving conditions.
MRS in rodents in awake/anesthetized conditions
A recent work by Sonnay et al. 39 using proton MRS and 13 C MRS measured the glutamate-glutamine cycle under stimulation and rest. They did not find major Glu changes, but they observed marked changes in this cycle, suggesting that MRS-measured Glu concentration is not related to glutamatergic neurotransmission per se. We believe that there are a few possible reasons for the controversy between the previous work by Sonnay et al and our current work. First, there could be the influence of anesthesia, while Sonnay et al minimized the effect by utilizing a constant dose. As we found by two-photon microscopy, neural activities were significantly suppressed under anesthetic condition. Thus, the different conditions between under anesthesia and awake could lead to the different results. Consistent with our studies, awake MRS studies in humans could detect Glu increases in target VOIs.40–42 Second, the targeted regions for MRS in rodent brains between Sonnay et al and ours were different. While Sonnay et al investigated the visual cortex, we used a barrel cortex in this work, which is consistent with the work by Just et al., 11 showing increase of Glu levels in the barrel cortex caused by trigeminal nerve stimulation.
Consistent with the current work using MRS, experiments in awake animals have significant advantages for detecting brain activities compared to the anesthetic condition. The advantages of awake condition are especially noticeable by two-photon microscopy imaging, which can detect neural activities at the single cell level. 43 The impact of general anesthesia on neural activities has been reported,44,45 although the mechanism of anesthesia remains largely unknown. 46 For monitoring brain activities, given the influence of general anesthesia on brain activities, the awake condition has became the golden standard for two-photon microscopy, 47 which was also applied in our work. Nevertheless, due to its technical difficulties, awake fMRS experiments in mice have not been previously performed. 14 The technical requirement including the mouse holder and head-restricting tools may affect the quality of the spectra, such as FWHM and CRLB of each metabolite, which was also the case in this work. When focusing on the quality of the spectra, the anesthetized condition has advantages over the awake condition. Moreover, there are not many differences regarding the concentrations of metabolites at the static conditions between awake and anesthetized animals. In the current study, the concentrations of tNAA at the baseline were different between the anesthetic and awake conditions, which is consistent with the different levels of neural activities measured by two-photon calcium imaging at the anesthetic and awake conditions. The difference of cerebral metabolite concentrations between the anesthetic and awake conditions would be in agreement with the previous work showing the different functional connectivity between rats under anesthesia by isoflurane and the awake condition. 48 Nevertheless, given the technical difficulties of awake MRS, the importance and usefulness of the anesthetic condition for MRS in preclinical researches remain, 49 and the awake condition would be preferable specifically for fMRS experiments for rodents. 14
Awake MRS for exploring the neural basis of brain activities in human and rodents
An increasing number of human MR studies for exploring brain function using MRS have been performed;1,3,50,51 however, no human studies demonstrated the underlying neural basis of altered neurochemicals measured by MRS. Thus, we utilized a custom-made awake mouse restraint device to perform whisker stimulation for causing neural activities, which was confirmed by an awake two-photon imaging system, in the barrel cortex of a mouse brain. Using those devices, we successfully detected neural activities as well as Glu and GABA level alterations, both of which were significantly suppressed by isoflurane anesthesia (Figure 1, Table 1). Collectively, this work using awake mouse MRS provides the rationale for human MRS studies, most of which are performed in the awake condition, for exploring the neurochemical basis of cognition and learning,1,5 which has not been possible by other modalities.
Metabolic aspects of MRS-measured glu and GABA
Metabolic aspects of MRS-measured Glu and GABA are essential when interpreting the results of MRS data, which are deducted from the slight difference in the time courses between the MRS-measured Glu and excitatory neural activities at the 2nd half stimulation. This controversy could be attributed to the metabolism process in neurons and glial cells, in addition to neurotransmitter release. Synaptically-released Glu is uptaken by astrocytes, then converted to GABA in GABAergic neurons. These metabolic processes may cause a discrepancy between MRS-measured Glu and excitatory neural activities measured by two-photon microscopy. Thus, while MRS-measured Glu and GABA can reflect excitatory and inhibitory tones in a VOI, respectively, it is presumable that those tones could be composed of not only neurotransmitter release but also metabolism. Indeed, awareness of the importance of metabolism in brain function and the pathomechanism of neurological diseases is increasing, 52 leading to more attention being given to MRS-measured Glu and GABA in the neuroscience research community in the future.
The relationship between BOLD signals and the release of GABA is also an important topic, which was previously investigated using optical intrinsic signal imaging. 53 While this work did not focus on BOLD signals, our tool will be useful for investigating unsolved questions in relation to brain function.
Spontaneous neural activities are associated with MRS-measured glu and GABA levels
The results obtained from the Dravet mouse model in this work indicated that MRS-measured Glu and GABA at quiet rest reflect neural excitatory and inhibitory activities, respectively. This may also imply that awake MRS is sensitive enough to detect neuronal activities via Glu and GABA level alterations at quiet rest, using persistent neuronal activities during a resting condition. This notion is supported by previous works that demonstrated the relationship between Glu and GABA levels and altered signals of the resting state fMRI.3,54 Other previous work, which detected different Glu levels according to the open/closed eye status, 51 also suggested the sensitivity of the MRS measurement during the awake condition. This line of evidence indicates that awake MRS is capable of investigating brain function using Glu and GABA alterations even during quiet rest.
Alterations of MRS-measured Glu and GABA levels under various conditions
MRS-measured Glu and GABA reflect the total Glu and GABA levels in the brain region, and they are derived from all compartments, including cytosol, synaptic vesicles, and extracellular space in the VOI. It can be assumed that respective compartments contribute differently to MRS-measured Glu and GABA depending on the brain conditions such as resting/active, and also pathological/physiological conditions. While a previous report of Alzheimer’s disease model mice demonstrated that synaptic loss was associated with decreased MRS-measured Glu, 55 it can also be considered that reduced cerebral metabolism in Alzheimer’s disease 56 contributes to the reduced MRS-measured Glu. Since most Glu measured by MRS is thought to be associated with metabolism, 57 small changes in Glu levels were detectable by MRS. Given the localization of Glu in soma or/and synapses, MRS-measured Glu can reflect neural loss and synaptic loss in the pathological condition at quiet rest. Since metabolic status can be changed in the diseased brain compared to the physiological status55,58 the behavior of Glu levels may be different in the diseased brain, as was shown in such fMRI studies. 59 This notion is supported by the fact that Glu can be consumed as brain fuel when there is not enough glucose, 60 requiring caution for interpretation of the results in the diseased brain. Nevertheless, awake MRS in the diseased brain, such as in Alzheimer’s disease model mice, would be intriguing, since excitation and inhibition balance (E/I balance) disruption exists in Alzheimer’s disease 61 as an important potential pathological mechanism.
In contrast to the complexity of Glu localization, a large portion of GABA in the brain under basal conditions is reported to localize in the cytoplasm, 62 suggesting that MRS-measured GABA may reflect the GABA pool in the cytoplasm. While the functional significance of the GABA pool has remained unknown, 63 several previous studies reported that baseline GABA levels in humans were associated with brain functions.64–66 The association between MRS-measured GABA and inhibitory neural activities at quiet rest in a Dravet mouse model 67 in the present work thus highlighted the importance of baseline GABA levels, which may be attributed to the cytoplasmic GABA and reflect inhibitory neural activities. While the mouse model of Dravet syndrome is known to have dysfunction of inhibitory neurons, it is also known that this mouse model is sensitive to body temperature elevation. 68 It would be interesting to perform whisker stimulation on this mouse, although there may be another factor such as body temperature change when interpreting the results of metabolite changes. Nevertheless, it would be intriguing to perform awake MRS studies with body temperature changes, which may lead to an understanding of febrile seizure.
Under the anesthetic condition with 1.5 ± 0.3% isoflurane, we observed high Lac levels even at baseline, which is known as an effect of isoflurane. 69 Changes in Lac concentrations were not observed under anesthetic condition. No apparent changes may be attributed to the large voxel size in the small mouse brains. Given the relatively high dose of isoflurane in this work, more studies will be necessary to reach conclusions about the changes of Lac levels at anesthetic conditions. In the awake state, we did not detect Lac under any conditions, which is in consistent with a human fMRS study. 40 This also may be attributed to the large voxel size compared to the target regions. Given the importance of Lac for brain function, 70 more investigations will be necessary.
Limitations of the study
This study has potential limitations. First, regarding the whisker stimulation frequencies, we did not investigate variations in the stimulation frequencies. Since we successfully observed neural activities at this exploratory value (0.33 Hz), we did not try other stimulation frequencies. While this 0.33 Hz rate did not create any issues regarding our aims with this work, the influence of different frequencies would be important as was demonstrated by Sonnay et al. 71 Secondly, information about the relationship between fMRI and MRS was limited in this work. Since we focused on the relationship between MRS-measured metabolites and neural activities measured by two-photon imaging, fMRI experiments were exploratory and were not fully involved in this study. Given the significance of the physiological importance of BOLD signals to the neuroscience research community, this aspect should be investigated in future studies. Third, there may be some stress to the mice during the awake MRS experiments. While we confirmed that mice stayed calm during the MRS experiments, we did not conduct precise stress level measurements. In future studies, measuring corticosterone levels would be useful for a more accurate understanding of the stress levels during awake experiments. Fourth, the number of animals was relatively small, given the challenging study design including awake experiments. Nevertheless, since these animal numbers are comparable with previous studies using cryoprobes and increased magnetic field strength,39,72 the number of animals might have been reasonable. Fifth, the MRS sequence could be altered to improve the spectra quality. As recommended in the other review of fMRS in rodents, 14 the usage of sequences such as sLASER 73 or SPECIAL 74 is highly recommended, especially regarding the lower impact of chemical shift displacement and motion with these sequences. These may improve the quality of the spectra, resulting in a smaller voxel size. The large voxel size may encompass other regions besides the barrel cortex, leading to a lack of BOLD effects on the spectra in this work.
Conclusion
Our work indicates that MRS-measured Glu and GABA could be an index of excitatory and inhibitory activities, respectively, in both spontaneous and task-evoked neural activities. By using MRS in an awake condition, we can monitor the E/I balance in various conditions, such as epilepsy and neuropsychiatric/neurological disorders, which provide us with important information in clinical studies. Also, using awake MRS in mice, we may address physiological and/or pathological questions by combining with other modalities such as positron emission tomography.75–77 Moreover, this method can be combined with optogenetics 12,78,79 and chemogenetics 80 for further validating the mechanistic link between MRS findings and neural activities. Lastly, given that glial cells, such as astrocytes, are also known to contribute to Glu metabolism, 81 there are likely other factors, particularly in pathological conditions, that contribute to MRS neurotransmitter measures besides neuronal activity, which should be addressed in future studies.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211045449 - Supplemental material for MRS-measured glutamate versus GABA reflects excitatory versus inhibitory neural activities in awake mice
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211045449 for MRS-measured glutamate versus GABA reflects excitatory versus inhibitory neural activities in awake mice by Yuhei Takado, Hiroyuki Takuwa, Kazuaki Sampei, Takuya Urushihata, Manami Takahashi, Masafumi Shimojo, Shoko Uchida, Nobuhiro Nitta, Sayaka Shibata, Keisuke Nagashima, Yoshihiro Ochi, Maiko Ono, Jun Maeda, Yutaka Tomita, Naruhiko Sahara, Jamie Near, Ichio Aoki, Kazuhisa Shibata and Makoto Higuchi in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by Kakenhi Grant Number 19K08005. Y Takado and KS are partially supported by Kakenhi Grant Number 19H01041. This research is also supported by AMED under Grant Number 17dm0107066h (IA) and partly supported by COI program (JPMJCE1305, IA) by JST.
Acknowledgements
We would like to express our appreciation to Ms Kanami Ebata, Mr Takeharu Minamihisamatsu, Mr Takahiro Shimizu and other colleagues who helped our experiments. We also appreciate the valuable comments about this manuscript by Dr. Takafumi Minamimoto.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Y Takado, HT and MH came up with the concept of the paper and designed the experimental procedures. Y Takado, HT, K Sampei, TU, SU, NN, SS performed the MRS experiments. JN made a basis set for the MRS analysis. HT, TU, MT, MS, MO, JM, Y Tomita designed and performed two-photon imaging experiments. KN and YO designed a homemade laser system for mesoscopic imaging. Y Takado and K Shibata wrote the original draft of the paper. All authors gave comments on the manuscript and approved the final version of the paper.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
