Abstract
Stroke remains a significant unmet need in the clinic with few therapeutic options. We, and others, have implicated the role of inflammatory microbiota in stroke secondary cell death. Elucidating this inflammation microbiome as a biomarker may improve stroke diagnosis and treatment. Here, adult Sprague-Dawley rats performed 30 minutes of exercise on a motorized treadmill for 3 consecutive days prior to transient middle cerebral artery occlusion (MCAO). Stroke animals that underwent exercise showed 1) robust behavioral improvements, 2) significantly smaller infarct sizes and increased peri-infarct cell survival and 3) decreasing trends of inflammatory microbiota BAC303, EREC482, and LAB158 coupled with significantly reduced levels of inflammatory markers ionized calcium binding adaptor molecule 1, tumor necrosis factor alpha, and mouse monoclonal MHC Class II RT1B in the brain, gut, spleen, and thymus compared to non-exercised stroke rats. These results suggest that a specific set of inflammatory microbiota exists in central and peripheral organs and can serve as a disease biomarker and a therapeutic target for stroke.
Introduction
Stroke stands as a major cause of death and disability in the U.S and around the world, leaving surviving patients with significant locomotor and cognitive deficits. 1 , 2 Treatment for stroke remains limited to thrombolysis options such as tissue-type plasminogen activator (tPA) and endovascular interventions with limited therapeutic window and the risk of time-dependent hemorrhagic transformation. 3 Finding innovative treatments that will complement tPA and mechanical thrombectomy will likely improve stroke outcomes.
Physical activity or exercise may counteract motor and memory impairment in healthy, elderly individuals, 4 , 5 thereby representing as a potent stroke prevention and treatment. 6 The protective effects of physical activity in stroke animal models include enhanced cerebral blood flow during reperfusion and nitric oxide synthase-dependent vascular reactivity of cerebral arterioles, and increased insulin-like growth factor (IGF), vascular endothelial growth factor (VEGF), and hippocampal neurogenesis.7–12 Despite these implicated therapeutic pathways, the mechanisms underlying the beneficial effects of exercise are complex and not fully understood but with direct clinical applications. Indeed, clinical trials reveal that exercise is associated with less severe stroke and better functional outcomes.13–15
The secondary inflammatory response exacerbates the brain damage post-stroke and induces further neurodegeneration.16–18 A plethora of pro-inflammatory cytokines precipitate from the periphery, such as the gut, the spleen and the thymus.19–21 A dynamic pathological link may exist between brain, gut, spleen, and thymus through a crosstalk of dysbiosis and inflammation. The gut-brain axis contributes to the maintainance of a homeostatic environment for cell, tissue, and organ survival. 22 Indeed, following a CNS insult like stroke, the gut-brain axis is recruited to attenuate dysbiosis via inflammatory pathways. 22 That a signature microbiome characteristic of the inflammatory state provides a robust disease biomarker and a sensitive therapeutic index may reveal a novel understanding of stroke and its treatment. Tandem fluctuations of inflammatory microbiota in central and peripheral organs suggest a key interaction of the brain with the periphery.22–24 Accordingly, to test the hypothesis of microbiome “disease” biomarker profiling that peripheral dysbiosis approximates central inflammation, the present study investigated the inflammatory microbiota signature of the brain, gut, spleen and thymus in adult rats exposed to experimental stroke. Additionally, to test the hypothesis of microbiome “therapeutic” biomarker profiling, we similarly profiled for inflammatory signals in the same organs in stroke animals subjected to exercise. The overarching hypothesis is that a stroke microbiome signature would be profiled in the periphery that closely parallels and likely precedes the brain inflammatory response to stroke pathology and treatment.
Materials and methods
Animals
All experiments were conducted in accordance with the National Institutes of Health Guide and Use of Laboratory Animals, and were approved by the Institutional Animal Care and Use committee of the University of South Florida, Morsani College of Medicine. 25 Male Sprague‐Dawley rats (8 weeks old, n = 24) were housed under normal conditions (20 °C, 50% relative humidity, and a 12‐hour light/dark cycle). All necessary precautions were taken to reduce pain and suffering of animals throughout the study. Animals were closely checked daily throughout the 7-day study period. All procedures were performed by personnel blinded to the treatment condition. Another person not involved in the study randomly assigned the animals to each treatment condition and coded the animals, with decoding done after all analyses were completed. Furthermore, all experiments followed the ARRIVE guidelines 2.0 (Animal Research: Reporting in Vivo Experiments), 26 which included blinding and randomization among other quality control procedures to ensure reproducibility, transparency, and rigor of the experiments.
Surgical procedures
Adult Sprague–Dawley rats were subjected to the middle cerebral artery occlusion (MCAO) model of stroke (n = 6), MCAO + exercise (n = 6), sham (n = 6), or sham + exercise (n = 6) and anesthetized by a mixture of 1–2% isoflurane in nitrous oxide/oxygen (69%/30%) via face mask. 25 To closely resemble the clinical scenario whereby stroke patients are routinely provided rehabilitation therapy, the sham + exercise group was included as a reference control condition to compare against the other treatment groups. 25 Body temperature was maintained at 37 ± 0.3 °C during the surgical procedures. 25 A midline skin incision was made in the neck with subsequent exploration of the right common carotid artery (CCA), the external carotid artery, and internal carotid artery. 25 A 4-0 monofilament nylon suture (27.0–28.0 mm) was advanced from the CCA bifurcation until it blocked the origin of the middle cerebral artery (MCA). 25 Animals were allowed to recover from anesthesia during MCAO. After 60 min of transient MCAO, animals were reanesthetized and reperfused by withdrawal of the nylon thread. 25 For the sham surgery, animals were anesthetized, a midline incision was made in the neck, and the right CCA was isolated. 25 The incision was then closed and the animal was allowed to recover from anesthesia. 25 Physiological outcome parameters of MCAO were maintained constant across all animals throughout the experiment. 25 Only animals that showed at least 75% behavioral deficits were included in this study.
Treadmill exercise
The animals in the exercise groups were forced to run on a motorized treadmill (Lafayette Instrument; Model 80805 A) for 30 min on day -3, -2, and -1 before surgery. The exercise groups ran at a speed of 2 m/min for the first 5 min, 5 m/min for the next 5 min, and 8 m/min for the remaining 20 min. 27 Animals in the non-exercise groups were left on the treadmill for the same period of time without running. The treadmill was designed with no electrical shock or aversive stimulus and prevented the animals from falling off.
Behavioral tests
Each rat was subjected to a series of behavioral tests to reveal motor and neurological performances of animals before (Baseline) and after MCAO (day 1, 3, and 7). The tests included the elevated body swing test (EBST) and cylinder test. The behavioral tests were performed by two co-authors blind to the group of rats.
Elevated Body Swing Test
EBST is a measure of asymmetrical motor behavior that does not require animal training or drug injection. 28 The rat was held, in the vertical axis, approximately 1 in from the base of its tail and then elevated to an inch above the surface on which it had been resting. The frequency and direction of the swing behavior were recorded for over 20 tail elevations. A swing was counted when the head of the rat moved more than 10° from the vertical axis to either side. Normally, intact rats display a 50% swing bias; that is, the same number of swings to the left and to the right. A 75% swing bias towards one direction was used as criterion of motor deficit. 28 The total number of swings made to the biased side was added per group and divided by the n, giving us the average number of swings per treatment group.
Cylinder test
The cylinder test was performed to assess the degree of forepaw asymmetry. Rats were placed in a transparent cylinder (diameter: 20 cm, height: 30 cm) for 3 minutes and the number of forepaw contacts to the cylinder wall was counted. The score of cylinder test in this study was calculated as a contralateral bias, that is, [(the number of contacts with the non-paretic limb)-(the number of contacts with the paretic limb)/(the number of total contacts) ×100]. 29
Perfusion
Under deep anesthesia, rats were euthanized on day 7 for immunofluorescent and protein analysis. 25 For immunofluorescent analysis, animals were perfused through the ascending aorta with 200 mL of cold PBS, followed by 200 mL of 4% paraformaldehyde in phosphate buffer (PB). 25 Brains were harvested and post-fixed in the same fixative for 72 h, followed by 30% sucrose in PB until completely sunk. 25 Series of coronal sections were cut at a thickness of 40 µm using a cryostat and stored at −20°C. 25
Nissl staining
Nissl staining was performed with 0.1% cresyl violet solution (Sigma-Aldrich) using a standard protocol to evaluate the peri-infarct injury of our MCAO model. 25 From each perfused brain, six coronal sections between the anterior edge and posterior edge of the MCAO infarct area were collected and processed for Nissl staining. 25 Every sixth coronal tissue section was chosen at random to quantify cell survival in the peri-infarct area. 25 Brain sections were examined using a light microscope (Keyence). 25 Neuronal survival in the peri-infarct area of the brain was quantified using a computer-assisted image analysis system (NIH Image Software, USA) and was expressed as a percentage of the ipsilateral hemisphere compared to the contralateral hemisphere. 25
Immunohistochemistry protocol
Brain, thymus, spleen, and gut sections were processed for mechanism-based immunohistochemical analyses of tissue samples focusing on inflammation. 25 Immunofluorescence labeling was conducted on every third coronal section of the brain, thymus, and gut and every sixth coronal section of the spleen. 25 Briefly, sections were washed three times for 10 min in 0.1 M phosphate-buffered saline (PBS). 25 Six sections were incubated with saline sodium citrate (SSC) solution at pH 6.0 for 40 min at 80 °C for antigen retrieval purposes. 25 Then, samples were blocked for 60 min at room temperature with 5% normal goat serum (Invitrogen, CA) in 0.1 M PBS containing 0.1% Tween 20 (PBST) (Sigma). 25 Sections were then incubated overnight at 4 °C with their corresponding primary antibody with 5% normal goat serum. 25 Established sensitive inflammatory markers, including Iba1, OX-6, and TNF-α, 21 , 30 , 31 were employed here to assess the inflammatory response to stroke and exercise. The primary antibodies used for brain tissue were rabbit polyclonal ionized calcium binding adaptor molecule 1 (Iba1; 1:500; Wako; 019-19741), 30 rabbit polyclonal anti-tumor necrosis factor alpha (TNF-α; 1:500; ab6671) 21 and mouse monoclonal MHC Class II RT1B (OX-6; 1:500; Novus; NB100-65541). 25 , 31 Then, sections were washed five times for 10 min in 0.1 M PBST and soaked in 5% normal goat serum in 0.1 M PBST containing corresponding secondary antibodies, goat anti-mouse IgG-Alexa 488 (green) (1:500; Invitrogen) or goat anti-rabbit IgG-Alexa 594 (red) (1:500; Invitrogen) for 2 h. 25 Finally, coronal sections were washed five times for 10 min in 0.1 M PBST and three times for 5 min in 0.1 M PBS, and cover-slipped with Vectashield Hardset Antifade Mounting Medium with DAPI (Vector; H-1500). 25 Coronal sections were examined using a confocal microscope (Zeiss). 21 Control studies included exclusion of primary antibody substituted with 5% normal goat serum in 0.1 M TBS. No immunoreactivity was observed in these controls. 25
Microbiome analysis
The presence and concentration of specific inflammatory, potentially harmful, commensal bacteria residing within the small intestine of the gut, brain, spleen, and thymus was determined. 32 We focused on the inflammatory microbiota that were found elevated in our previous studies on Parkinson’s disease animal models. 32 , 33 Fluorescent in‐situ hybridization (FISH) analysis was used to identify the specific microbiota within the feces of the rats in this study. 34 Brain infarct area tissue, thymus tissue, spleen tissue, and fecal matter (each tissue: 1 g) collected during tissue processing was suspended in 2 mL of PBS and homogenized by repeated pipetting with a 1 mL micropipettor and mixed using a vortex. 32 Four per cent paraformaldehyde (6 mL) was added to the homogenized feces and stored overnight at 4 °C. 32 The tubes were then centrifuged at 10 g for 5 minutes to sediment undigested material. 32 The bacterial laden supernatant was transferred a new tube and the centrifugation step was repeated. 32 Following the final centrifugation the supernatant was transferred to a new tube and centrifuged at 60 g for 5 minutes to pellet any bacteria present. 32 The supernatant was discarded and the pellet was resuspended in 500 µL of hybridization solution comprised of 50% (v/v) formamide, 100 µg/mL salmon sperm DNA, 5× saline‐sodium citrate buffer, and 0.1% (v/v) Tween 20. 32 After a 30 minutes incubation at 37 °C, 50 µL volumes of each sample were combined with 2.7 µL of fluorescently labelled oligonucleotides in a new tube and incubated in the dark for 2.5 hours at 45 °C. 32 The tubes were then centrifuged at 60 g for 5 minutes, the supernatant discarded, and the pellet was washed by resuspending in 20 µL of 0.1 SSC. 32 This wash step was repeated an additional two times. 30 After the final wash, the samples were resuspended in 20 µL of PBS, and centrifuged at 13000 g for 5 minutes. 32 Next, 10 µL was transferred to a new slide that had been cleaned with ethanol (EtOH) and 5 µL of Vectashield mounting medium with 2‐(4‐Amidinophenyl)‐6‐indolecarbamidine dihydrochloride, 4′,6‐Diamidino‐2‐phenylindole (DAPI) was added. 32 The slides were carefully cover‐slipped and images were collected at 40× using an Olympus FV1000 laser scanning confocal microscope equipped with Fluoview SV1000 imaging software. 32 During the duration of the FISH analysis, researchers worked in sterile conditions.
Statistical analysis
The data were evaluated statistically using one-way analysis of variance (ANOVA) and subsequent post hoc Bonferroni’s test and two-way ANOVA for behavior. 25 Statistical significance was preset at p < 0.05 (GraphPad version 5.01). 25 Data are presented as mean ± SD from quintuplicates of each treatment condition. The Kolmogorov–Smirnov test was performed to assess normality and the resulting values were <5% of the critical values. 25 Pearson’s correlational analyses were performed to examine possible correlations between the inflammatory microbiomes and stroke functional outcomes.
Results
Exercise improves motor behavior and attenuates forepaw asymmetry
Two behavioral tests, EBST and Cylinder test, were performed at baseline, day 1, 3, and 7 after stroke (Figure 1(a)). The MCAO + Ex group demonstrated significant improvement in motor activity (p < 0.0001, p < 0.01) as seen by attenuation of swing bias in EBST compared to the MCAO group on days 1, 3, and 7 (Figure 1(c)). Similarly, the Cylinder test revealed that the MCAO + Ex group exhibited significantly less forepaw asymmetry (p < 0.0001) on days 1, 3, and 7 compared to the MCAO group (Figure 1(d)). Sham and Sham + Ex did not demonstrate any significant changes in motor behavior or asymmetry and were not significantly different from each other.
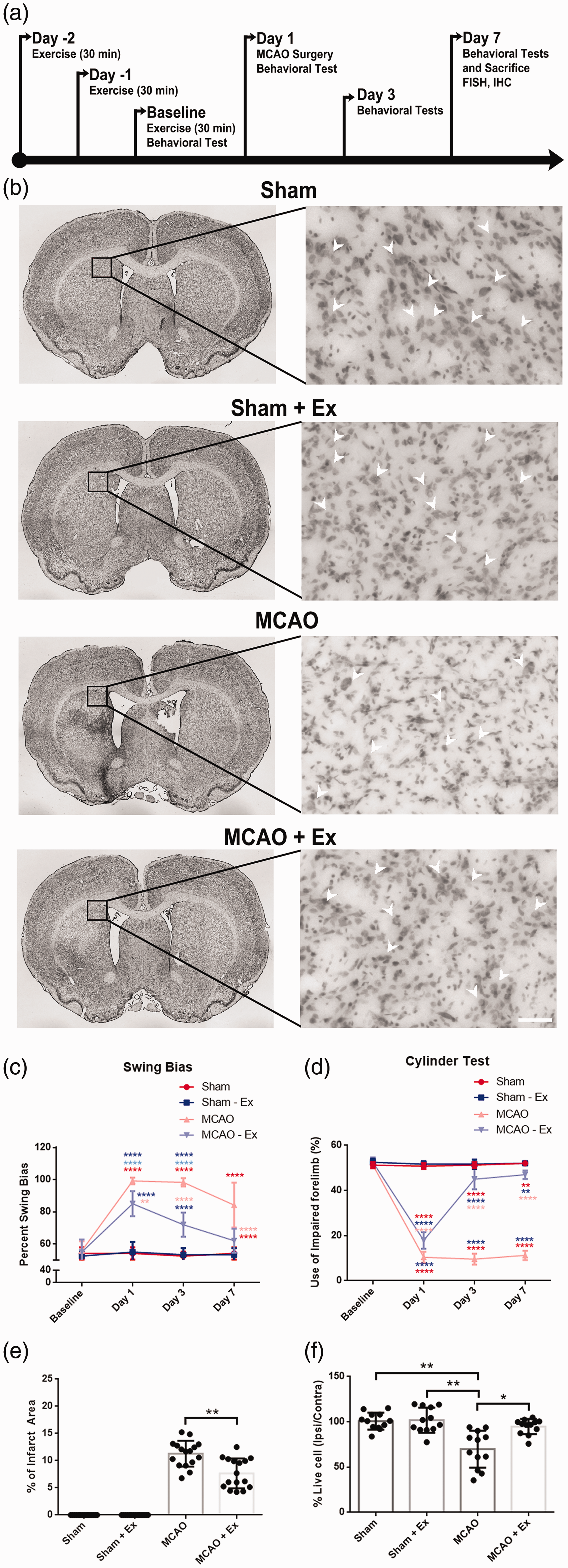
A timeline of the experimental procedures, results from behavioral testing, and Infarct areas and quantitative analysis of cell survival in the peri-infarct. (a) Prophylactic exercise was performed on days -2, -1, and baseline. Behavioral testing was performed at (baseline), days 1, 3, and 7. Middle Cerebral Arterial Occlusion (MCAO) was performed on day 1. All animals were sacrificed on day 7. (b) Nissl staining for coronal brain sections showing infarct areas (black outline) of sham, sham + Ex, MCAO, MCAO + Ex. (c) Motor activity revealed by EBST. MCAO-Ex group displayed significantly less asymmetry on days 1, 3, and 7 (**p < 0.01, ****p < 0.0001, ****p < 0.0001 respectively). (d) Motor activity revealed by the Cylinder Test. MCAO-Ex group demonstrated significantly more use of impaired forelimb (****p < 0.0001). (e) The MCAO + Ex group displayed significantly smaller infarcts (**p < 0.01). (f) Quantitative analysis of live cells (arrow head) in the peri-infarct area for sham, sham + Ex, MCAO, MCAO + Ex. MCAO + Ex showed significantly more living cells in the peri-infarct (*p < 0.05). Scale bar = 100 µm.
Exercise reduces infarct volumes and increases cell survival in the peri-infarct area
Animals in the MCAO + Ex group showed significantly reduced infarct volumes (p < 0.01) compared to animals in the MCAO group (Figure 1(e)). In a quantitative analysis of the peri-infarct area cell count, MCAO + Ex animals displayed a significantly greater percentage of live cells (p < 0.05) as compared to the MCAO group (Figure 1(f)). Sham and Sham + Ex did not display any significant changes in infarct area or peri-infarct cell survival and were not significantly different from each other.
Exercise may dampen inflammatory makers and microbiota in the brain
Immunohistochemical analyses of microglia/macrophage marker Iba1 (Figure 2(a)), activated microglia marker OX-6 (Figure 2(b)), and inflammatory cytokine marker TNF-α (Figure 2(c)) in the peri-infarct area revealed significantly decreased expression of Iba1 and OX-6 (p < 0.01, p < 0.001, respectively) in the MCAO + Ex group compared to the MCAO group (Figure 2(a′) and (b′)). TNF-α showed a decreasing trend (Figure 2(c′)). Sham and Sham + Ex did not express any significant differences in inflammatory markers in the brain and were not significantly different from each other. Next, the prevalence of potentially harmful microbiota was analyzed using FISH. BAC303, EREC482, and LAB158 showed significantly reduced levels (p < 0.0001, p < 0.0001, p < 0.001, respectively) in the MCAO + Ex group compared to the MCAO group (Figure 2(d)).
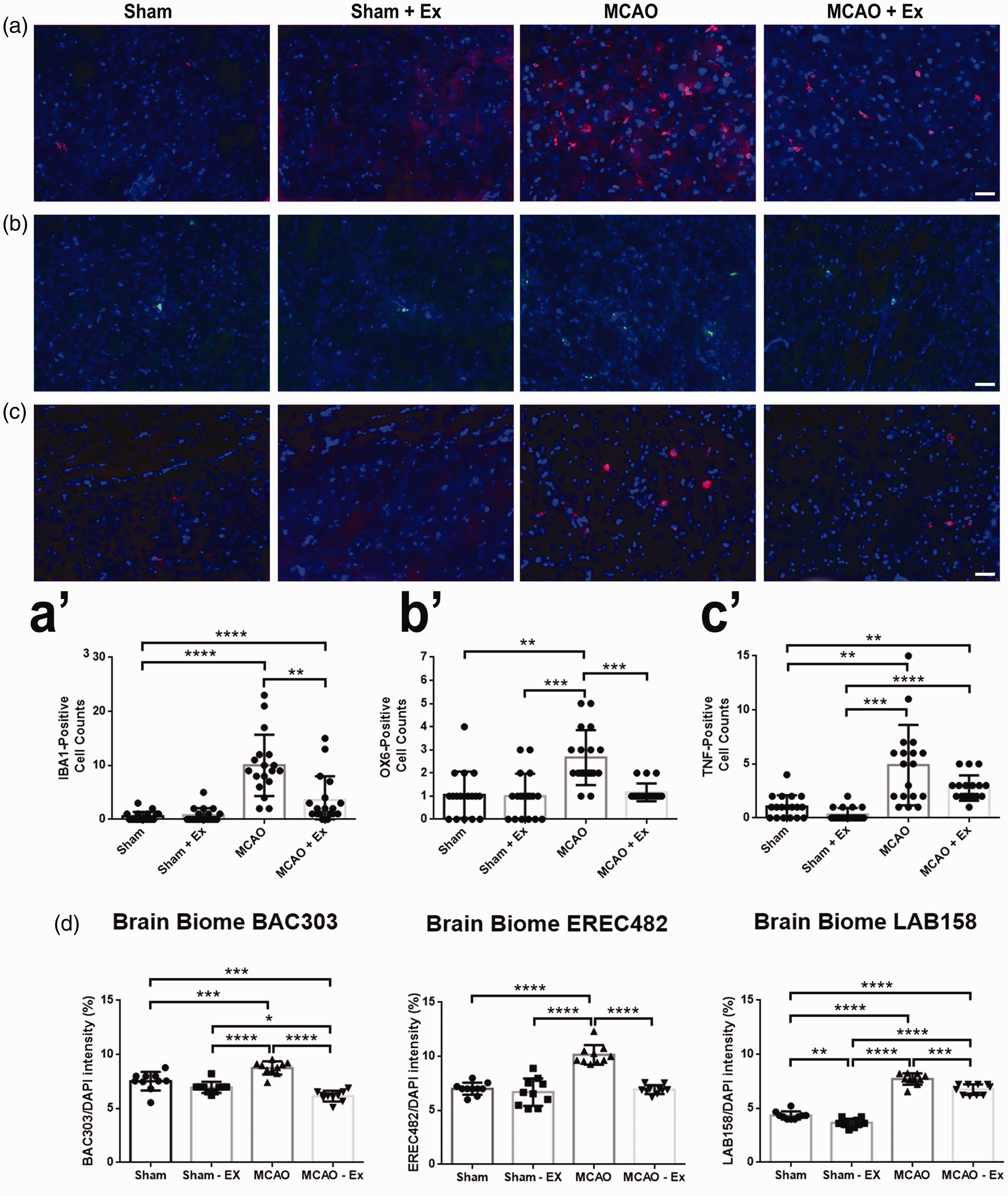
Immunohistochemical analysis of the brain for inflammation markers. (a) Staining for Iba1 positive cells (red) in the peri-infarct. (a’) Represents the cell count observed in figure A. MCAO + Ex showed significantly reduced amounts of positive cells (**p < 0.01) than MCAO alone. (b) Staining for OX-6 positive cells (green) in the peri-infarct. (b’) MCAO + Ex showed significantly reduced amounts of positive cells (***p < 0.001) than MCAO alone. (c) Staining for TNF-α positive cells (red) in the peri-infarct. (c’) MCAO + Ex showed decreasing amounts of positive cells than MCAO alone. (d) Quantitative analysis of pro-inflammatory microbiota present in the brain. BAC303, EREC482, and LAB158 showed significantly reduced expression (****p < 0.0001, ****p < 0.0001, ***p < 0.001, respectively) in the MCAO + Ex group compared to the MCAO group. Scale bar = 100 µm.
Exercise may attenuate inflammatory makers and microbiota in the thymus
Immunohistochemical markers Iba1 (Figure 3(a)), OX-6 (Figure 3(b)), and TNF-α (Figure 3(c)) were used to analyze the thymus. OX-6 and TNF-α markers showed a significant decrease in expression (p < 0.05) in the MCAO + Ex group compared to the MCAO group (Figure 3(a’) and (b’)) while Iba1 showed a decreasing trend (Figure 3(a’)). Sham and Sham + Ex did not show any significant differences in inflammatory markers in the thymus and were not significantly different from each other. BAC303, EREC482, and LAB158 expression was significantly reduced (p < 0.0001) between the MCAO + Ex group and MCAO group (Figure 3(d)).
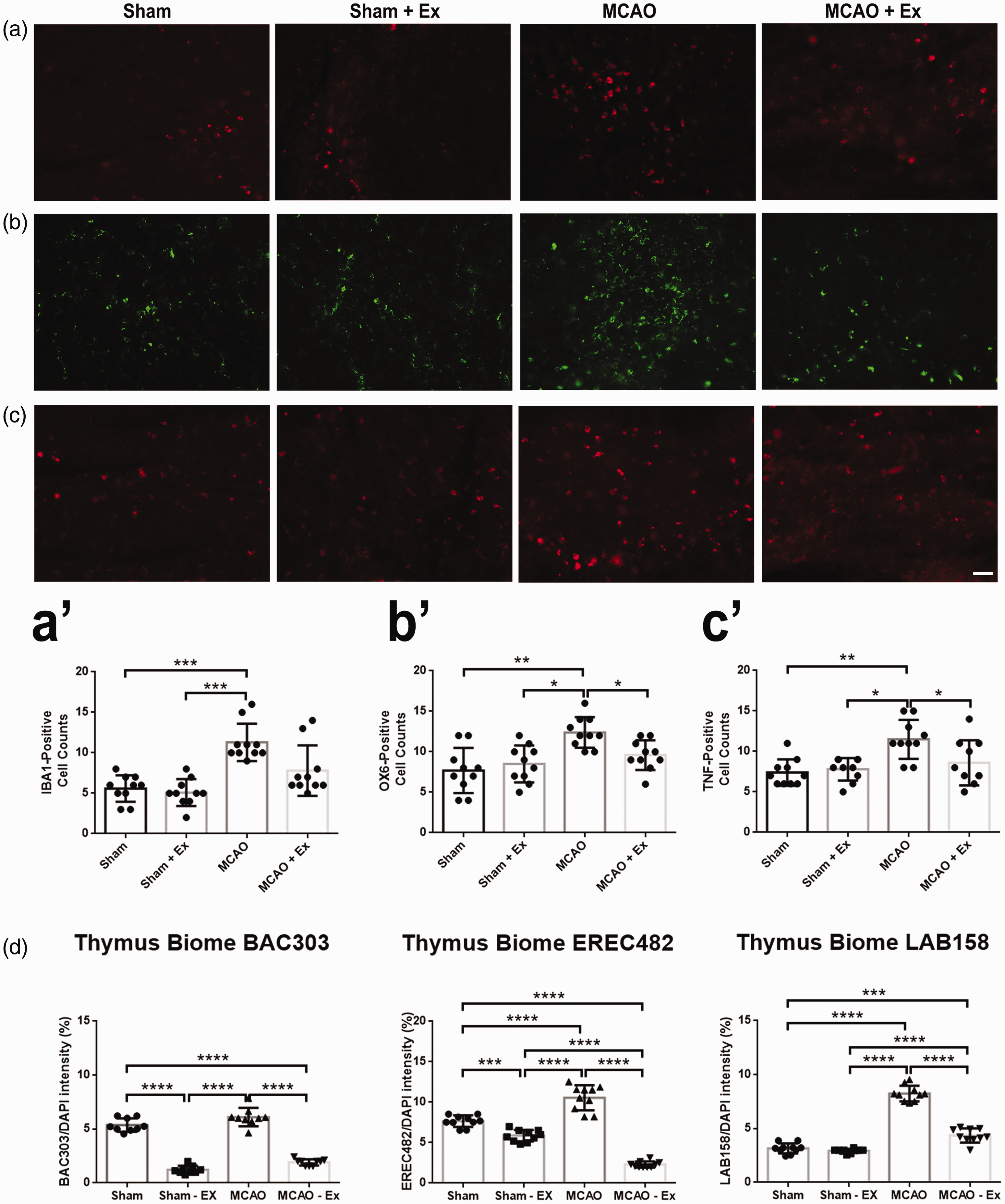
Immunohistochemical analysis of the thymus for inflammation markers. (a) Staining for Iba1 positive cells (red) in the thymus. (a’) MCAO + Ex showed reduced amounts of positive cells than MCAO alone. (b) Staining for OX-6 positive cells (green) in the thymus. (b’) MCAO + Ex showed significantly reduced amounts of positive cells (*p < 0.05) than MCAO alone. (c) Staining for TNF-α positive cells (red) in the thymus. (c’) MCAO + Ex showed significantly reduced amounts of positive cells (*p < 0.05) than MCAO alone. (d) Quantitative analysis of pro-inflammatory microbiota present in the thymus. BAC303, EREC482, and LAB158 expression was significantly reduced (****p < 0.0001) between the MCAO + Ex group and MCAO group. Scale bar = 100 µm.
Exercise may diminish inflammatory makers and microbiota in the spleen
Immunohistochemical markers Iba1 (Figure 4(a)), OX-6 (Figure 4(b)), and TNF-α (Figure 4(c)) were used to analyze the spleen. The MCAO + Ex group showed a significant decrease in Iba1 positive cells (p < 0.0001) compared to the MCAO group (Figure 4(a’)). TNF-α and OX-6 markers revealed decreasing trends between the MCAO and MCAO + Ex groups (Figure 4(b’) and (c’)). Sham and Sham + Ex did not indicate any significant changes in inflammatory markers in the spleen and were not significantly different from each other. BAC303 and LAB158 expression was significantly reduced (p < 0.0001, p < 0.0001, respectively) between the MCAO + Ex group and MCAO group (Figure 4(d)).
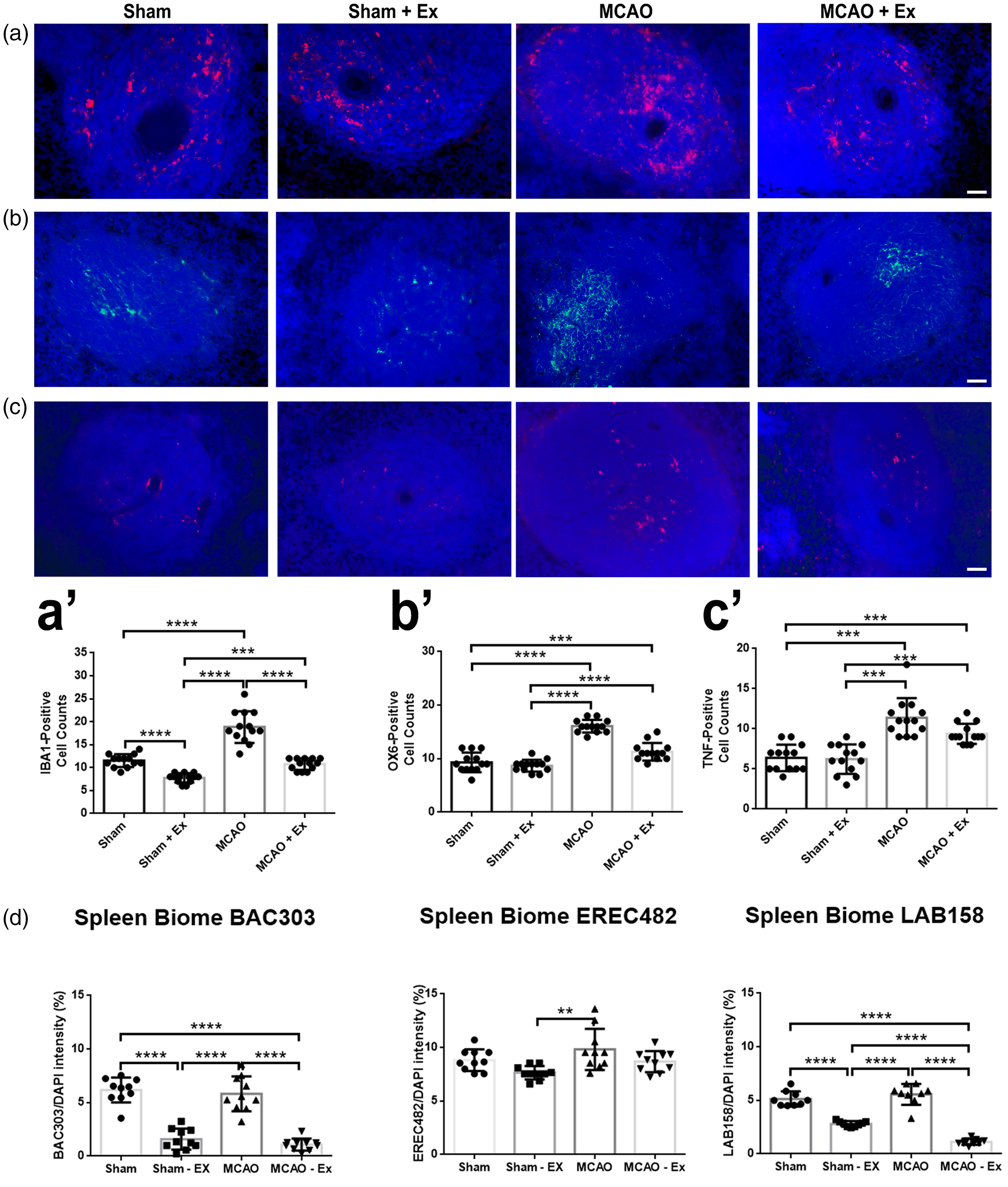
Immunohistochemical analysis of the spleen for inflammation markers. (a) Staining for Iba1 positive cells (red) in the spleen. (a’) MCAO + Ex showed significantly reduced amounts of positive cells (****p < 0.0001) than MCAO alone. (b) Staining for OX-6 positive cells (green) in the spleen. (b’) MCAO + Ex showed a decreasing trend of positive cells compared to MCAO alone. (c) Staining for TNF-α positive cells (red) in the spleen. (c’) MCAO + Ex showed a decreasing trend of positive cells compared to MCAO alone. (d) Quantitative analysis of pro-inflammatory microbiota present in the spleen. BAC303 and LAB158 expression was significantly reduced (****p < 0.0001, ****p < 0.0001, respectively) between the MCAO + Ex group and MCAO group. Scale bar = 100 µm.
Exercise may reduce inflammatory makers and microbiota in the gut
Immunohistochemical markers Iba1 (Figure 5(a)), OX-6 (Figure 5(b)), and TNF-α (Figure 5(c)) were used to analyze the gut. Each marker showed a significant decrease in expression (p < 0.05, p < 0.05, p < 0.01 respectively) between MCAO and MCAO + Ex groups (Figure 5(a’) to (c’)). Sham and Sham + Ex did not reveal any significant changes in inflammatory markers in the gut and were not significantly different from each other. Microbiota analysis revealed a significant decrease in BAC303 and LAB158 (p < 0.05 and p < 0.01 respectively) and a non-significant decrease in EREC482 between MCAO and MCAO + Ex groups (Figure 5(d)).
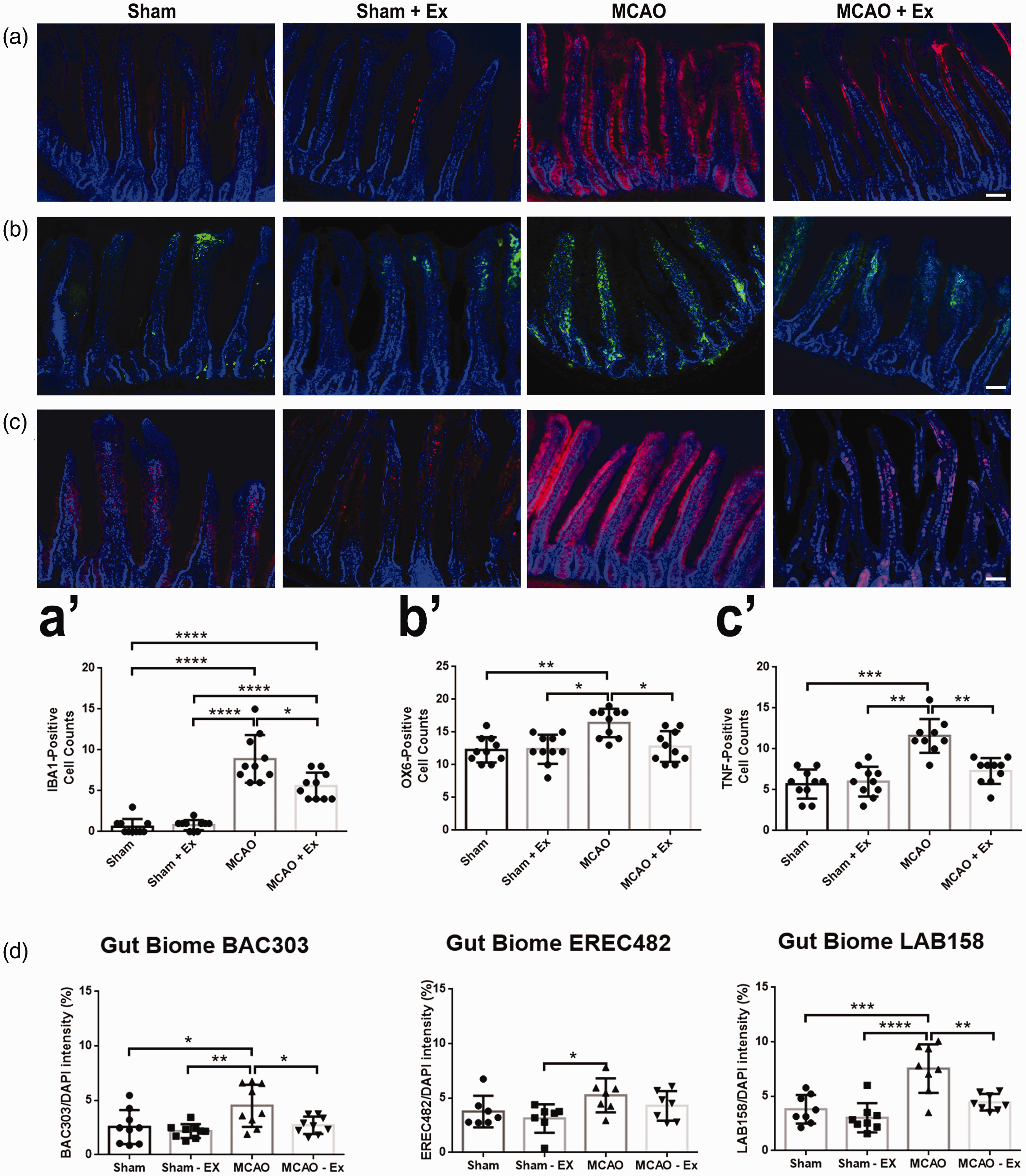
Immunohistochemical analysis of the gut for inflammation markers. (a) Staining for Iba1 positive cells (red) in the small intestines of the gut. (a’) MCAO + Ex showed significantly reduced amounts of positive cells (*p < 0.05) than MCAO alone. (b) Staining for OX-6 positive cells (green) in the small intestines of the gut. (b’) MCAO + Ex showed significantly reduced amounts of positive cells (*p < 0.05) than MCAO alone. (c) Staining for TNF-α positive cells (red) in the small intestines of the gut. (c′) MCAO + Ex showed significantly reduced amounts of positive cells (**p < 0.01) than MCAO alone. (d) Quantitative analysis of pro-inflammatory microbiota present in the small intestines of the gut. BAC303 and LAB158 showed a significant reduction in pro-inflammatory microbiota (*p < 0.05 and **p < 0.01 respectively) in the MCAO + Ex group compared to the MCAO group. EREC482 showed a decreasing trend between MCAO +Ex and MCAO alone. Scale bar = 200 µm.
Correlation analysis of inflammatory markers, microbiota, and functional outcomes
In order to further test our hypothesis that inflammation-relevant microbiota might have contributed to stroke behavioral and histological deficits, we conducted correlational analyses between these endpoint assays. A positive correlation was observed between the functional outcomes and the inflammatory microbiome, including EBST (Supplemental Figure 1, Supplemental Table 1), cylinder test (Supplemental Figure 2, Supplemental Table 2), and cerebral infarct (Supplemental Figure 3, Supplemental Table 3), in that an increased expression of the inflammatory microbiome in the brain, thymus, spleen and gut accompanied worsened behavioral and histological dysfunctions in animals that were subjected to MCAO alone. Conversely, a decreased expression of the inflammatory microbiome in the same organs corresponded with reduced behavioral and histological deficits in animals that were subjected to MCAO + Ex.
Discussion
For the first time, we report that a microbiome signature closely approximated the inflammation that predominated in brain, gut, spleen, and thymus after stroke. Such inflammation-plagued microbiota profile reveals a novel biomarker for stroke. In tandem, we provide evidence that such microbiome was sensitive to the neuroprotective effects of exercise, supporting the additional use of microbiome profiling as a sensitive index of stroke therapeutics (Figure 6).
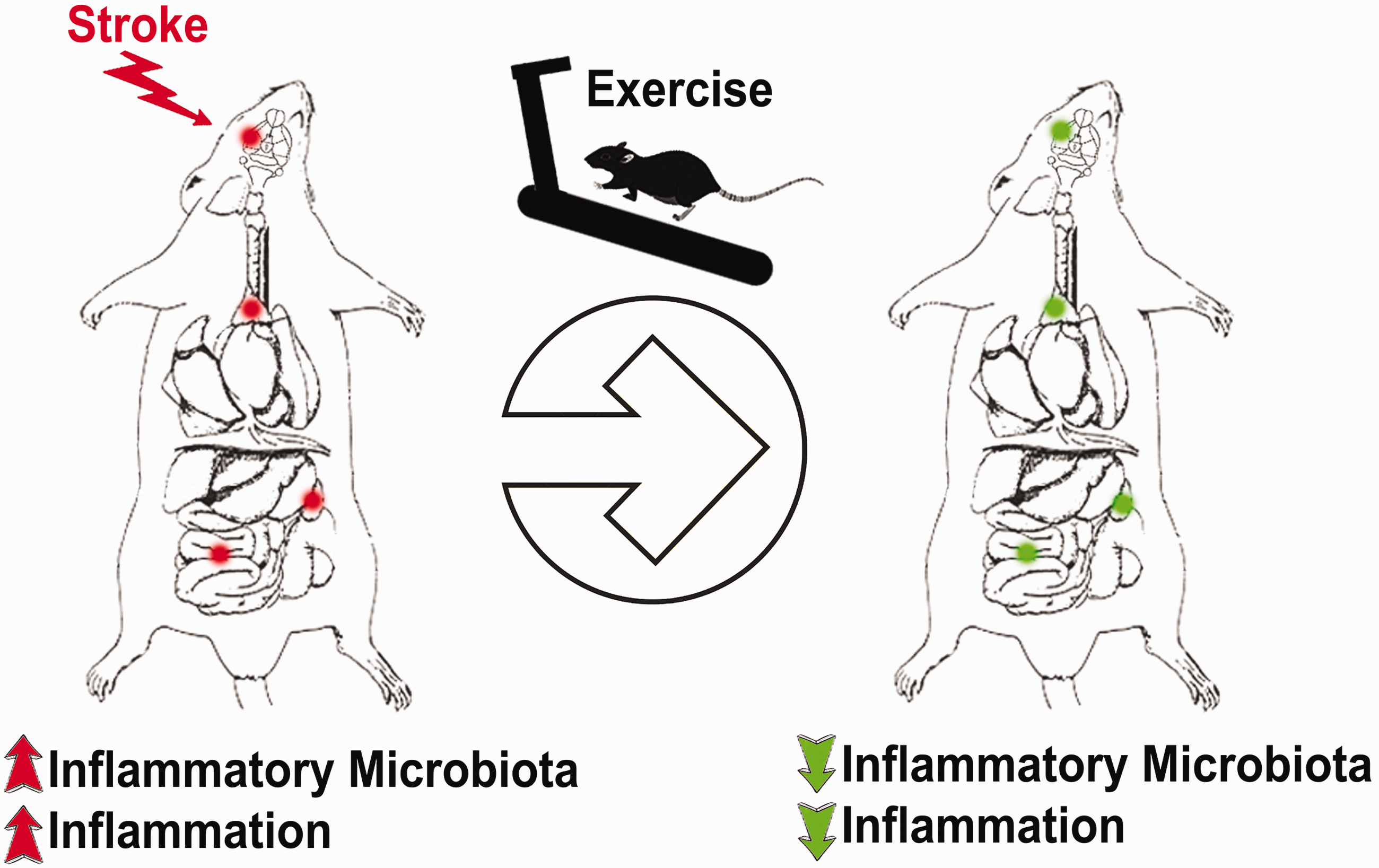
Stroke microbiome. Following stroke, inflammatory microbiota are upregulated in different parts of the body, including the gut, brain, thymus, and spleen. Rehabilitation therapy or exercise may dampen these inflammatory microbiota and thus reduce stroke symptoms.
A significant increase in gut dysbiosis ensues after ischemic insult. 35 Such inflammatory microbiota has been characterized in the gut, and its influence within the gut-brain axis.36–39 Our results parallel this significant increase in inflammatory microbiota (BAC303, EREC482, LAB158) in the gut of stroke rats, but also show a dramatic upregulation of inflammatory microbiota that occurs in the brain, spleen, and thymus. That a dysbiotic and inflamed gut accompanies stroke and extends to the brain, spleen, and thymus, supports a microbiome-mediated inflammatory response to stroke.40–42
After stroke, a secondary cellular death involving inflammation-related cascade exacerbates brain damage 23 , 24 , 43 with coincident upregulation of inflammatory signals in the gut, spleen, and thymus 22 , 41 , 44 , 45 suggesting the contribution of peripheral organs to the central inflammatory response to stroke. The markers Iba1, OX-6, and TNF-α have all been shown to accompany inflammation in ischemic stroke models. 21 , 30 , 31 Interestingly, pre-exposure of animals to exercise attenuates stroke-induced inflammation, 34 implicating that preventative exercise may protect against stroke-induced secondary inflammation. To maximize the potential of translating an experimental treatment from the lab to the clinic, Sham and MCAO models each contained an exercise group to capture the clinical scenario of rehabilitation therapy. The present prophylactic bouts of forced exercise on a motorized treadmill restored the healthy microbiome profile in central and peripheral organs that corresponded with improved behavioral performance and reduced histopathological deficits. Our correlational analyses revealed that the higher the level of each microbiota, there was a proportional increase in the inflammatory cytokine in each organ. On the other hand, exercise decreased microbiota expression in each organ, which was accompanied by a lower level of inflammatory cytokines. Nonetheless, the mechanism behind this apparent inflammatory cytokine-microbiota relationship remains to be determined. Each organ may consist of an innate population of microbiota that can be activated following a stressor like stroke or dampened by a treatment like exercise. The distribution of microbiota between organs may also influence inflammatory signaling pathways spatiotemporally after stroke and exercise. These speculative mechanisms warrant further investigations to fully understand the crosstalk between inflammatory cytokines and microbiota.
To date, the mechanism of action by which inflammation is upregulated is not well understood; however, that the gut and brain communicate via the gut-brain axis has been recently advanced. 46 Inflammation in the brain may be either upregulated or downregulated by the over- or under-expression, or dysbiosis, of certain microbiota in the gut. 46 Specific microbiota, such as BAC303, EREC482, and LAB158, are believed to be closely associated with increased central and peripheral inflammation in animal models of Parkinson’s disease. 32 , 33 The detection of these microbiota centrally and peripherally may indicate a microbiome-initiated inflammatory response that is active not only in the gut, but the brain, spleen, and thymus as well. Therefore, we chose to probe for these same three microbiota in stroke as candidate inflammatory signals to filter through the plethora of bacteria in the microbiome. Our present results align with our previous studies demonstrating increased levels of inflammation-associated microbiota in the brain and gut, 32 , 33 but also implicate the exacerbating roles of the spleen and thymus in stroke inflammation. 47 On the contrary, when these microbiota are downregulated, as produced by exercise in the present study, inflammation decreases which signals functional improvement. 32 , 33 Our present observations extend the concept of CNS-periphery crosstalk in stroke, from the reported gut-brain axis to the spleen and thymus serving as equally sensitive inflammation-plagued organs for detecting a signature microbiome biomarker of stroke inflammation. The current observation of varying levels of inflammatory microbiota across all organs may suggest that the distribution of microbiota may be organ-specific and can be triggered by stroke or exercise. Equally innovative is that profiling these same organs for their inflammation status following exercise treatment may represent a sensitive therapeutic index of recovery from stroke. Interestingly, correlational analysis revealed that the severity, as well as the improvement of stroke outcomes are closely associated with the upregulation and downregulation of inflammatory markers and microbiota in the brain, gut, spleen, and thymus (Supplemental Figures 1 to 3, Supplemental Tables 1-3), further supporting our hypothesis of a pathological link between the microbiome, inflammation, and stroke outcomes. The application of this multi-organ dysbiosis as a microbiome biomarker may be similarly applied to other diseases characterized by inflammation. Moreover, that exercise normalizes the microbiome in our stroke animals, may be utilized as a microbiome-based therapeutic index for other diseases that respond to exercise or metabolic activity-directed treatments such as Parkinson’s disease and traumatic brain injury. 48 , 49
Further testing of this microbiome biomarker and therapeutic index by incorporating stroke comorbidities, such as aging and hypertension, under standard treatments, like tPA and mechanical thrombectomy, may further reveal the crosstalk between dysbiosis and inflammation in central and peripheral systems. Additionally, we acknowledge that other microbiota may be contributing to the systemic inflammatory response and a broader approach to investigating the microbiome network should be utilized in future studies. Lastly, additional experimentation is warranted to uncover the mechanism by which microbiota are released and mobilized in the circulation.
Altogether, the current results provide evidence of a signature microbiome of the brain, thymus, spleen, and gut that closely accompanies inflammation after stroke. Furthermore, this study highlights the microbiome signature of the neuroprotective effects of exercise in central and peripheral organs. In the clinic, microbiome testing may serve as a biomarker to evaluate the patient’s condition following ischemic stroke and as a potential therapeutic index to monitor stroke recovery.
Availability of data and material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211039598 - Supplemental material for Inflammation-relevant microbiome signature of the stroke brain, gut, spleen, and thymus and the impact of exercise
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211039598 for Inflammation-relevant microbiome signature of the stroke brain, gut, spleen, and thymus and the impact of exercise by Chase Kingsbury, Alex Shear, Matt Heyck, Nadia Sadanandan, Henry Zhang, Bella Gonzales-Portillo, Blaise Cozene, Michael Sheyner, Lisset Navarro-Torres, Julián García-Sánchez, Jea-Young Lee and Cesario V Borlongan in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Center of Excellence for Aging and Brain Repair at the University of South Florida Morsani College of Medicine, Tampa, Florida, USA.
Acknowledgements
All research materials, including data, reported in this study can be easily accessed by contacting Dr. Cesario V. Borlongan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
The authors thank the entire staff of the Borlongan Neural Transplant Lab for the preparation of this manuscript. CK, AS, MH, NS, HZ, BG-P, BC, MS, LN-T, JG-S, J‐YL, CVB: experimental work, collection and/or assembly of data, data analysis and interpretation, manuscript writing; CVB: conceptualization and experimental design, project supervision, final editing and manuscript oversight.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
