Abstract
Delayed but prolonged hypothermia persistently decreases cell death and functional deficits after global cerebral ischemia in rodents. Postischemic hypothermia also reduces infarction after middle cerebral artery occlusion (MCAO) in rat. Because initial neuroprotection is sometimes transient and may not subserve functional recovery, especially on demanding tasks, the authors examined whether postischemic cooling would persistently reduce infarction and forelimb reaching deficits after MCAO. Male spontaneously hypertensive rats were trained to retrieve food pellets in a staircase test that measures independent forelimb reaching ability. Later, rats underwent 90 minutes of normothermic MCAO, through a microclip, or sham operation. In some rats, prolonged cooling (33°C for 24 hours and then 35°C for 24 hours) began 2.5 hours after the onset of ischemia (60 minutes after the start of reperfusion; n = 17 with subsequently 1 death) or sham procedures (n = 4), whereas untreated sham (n = 4) and ischemic (n = 16 with subsequently 1 death) rats maintained normothermia. An indwelling abdominal probe continually measured core temperature, and an automated fan and water spray system was used to produce hypothermia. One month later rats were reassessed in the staircase test over five days and then killed. The contralateral limb impairment in food pellet retrieval was completely prevented by hypothermia (P = 0.0001). Hypothermia reduced an infarct volume of 67.5 mm3 after untreated ischemia to 35.8 mm3 (P < 0.0001). These findings of persistent benefit encourage the clinical assessment of hypothermia.
Numerous drugs have shown promising effects in rodent models of global and focal cerebral ischemia; however, none have proven efficacious in clinical trials (De Keyser et al., 1999). These failures are at least partially because of an incomplete preclinical evaluation, notably no histologic evaluation using long survival times and frequently an absence of functional assessment (Corbett and Nurse, 1998). Examining histology after a brief recovery (for example, 1 to 7 days) does not necessarily predict the final outcome. For example, brief global (Colbourne et al., 1999a) and focal (Du et al., 1996) ischemic insults can mature more than once thought. Likewise, interventions such as brief postischemic hypothermia (Dietrich et al., 1993; Colbourne et al., 1997), ischemic preconditioning (Corbett and Crooks, 1997), NBQX (Colbourne et al., 1999a), SNX-111 (Colbourne et al., 1999a), and MK-801 (Valtysson et al., 1994) all result in transient neuroprotection. Methodologic limitations have compounded this problem. For instance, standard infarct size measurements and cell counting procedures cannot tell whether “normal looking” cells function properly (that is, covert injury) (Hori and Carpenter, 1994), nor do such measurements take into account structural damage outside that area of interest (that is, collateral damage). Conversely, deficits on simple behavioral measures (for example, Bederson scale; Bederson et al., 1986) can recover quickly in spite of massive brain injury (Cregan et al., 1997), whereas performance on more demanding tasks (for example, Montoya staircase test; Montoya et al., 1991) can overestimate true protection because of compensation (Whishaw et al., 1997). Thus, the possibility of contradictory behavioral and histologic outcomes in preclinical studies necessitates the use of both endpoints in an effort to better predict clinical effectiveness.
Prolonged postischemic hypothermia is perhaps the most promising candidate therapy (Colbourne et al., 1997). Some controversy existed regarding whether delayed cooling could permanently salvage hippocampal CA1 neurons after forebrain ischemia. Fortunately, recent studies show that whereas brief hypothermia (for example, 3 hours) is only transiently beneficial (Dietrich et al., 1993), prolonged hypothermia (for example, 2 days) protects most CA1 sector cells indefatigably with comparable levels of functional savings (working memory, habituation) (Colbourne et al., 1997, 1999b). The fact that cooling is beneficial even when initiated many hours after global ischemia offers hope for effective clinical intervention, such as after resuscitation from cardiac arrest (Zeiner et al., 2000). At the current time, the true efficacy of hypothermia after adult focal ischemia (stroke) is not known because brief (Chen et al., 1992; Xue et al., 1992; Markarian et al., 1996; Maier et al., and prolonged postischemic cooling (Yanamoto et al., 1996; Corbett et al., 2000) has typically been shown only to reduce infarct size and only one study examined long-term outcome. In that study (Corbett et al., 2000), rats were cooled to 34°C for 2 days starting 1 hour after onset of ischemia (30 minutes of MCA intraluminal suture occlusion plus systemic hypotension of 60 mm Hg) and infarct size was assessed at 2 months. Although these data are encouraging, hypothermia may not reduce infarction in a more standard stroke model, and equally important, hypothermia may not lessen functional deficits. Another limitation of previous studies is that they often use clinically unrealistic (early) intervention delays that would grossly overestimate true benefit.
In this study, the authors used the standard cerebral artery occlusion (MCAO) model (90-minute clip occlusion) with some rats receiving hypothermia treatment starting one hour after reperfusion (2.5 hours after onset of ischemia). Functional outcome was assessed in a staircase test starting 30 days after ischemia and infarct size was quantified after a survival of ≈ 40 days. The staircase test is sensitive to motor cortex injury and can reveal persistent deficits (Montoya et al., 1991; Grabowski et al., 1993), unlike the simpler measures (for example, Bederson scale) in which deficits are quite transient in spite of massive brain injury (Markgraf et al., 1992; Cregan et al., 1997). Thus, the current study was designed to clearly determine whether delayed cooling provides effective and lasting neuroprotection in a well-characterized MCAO model of stroke.
MATERIALS AND METHODS
Subjects
Spontaneously hypertensive male rats (weighing ≈ 280 g) were obtained from Charles River (Montreal, Quebec, Canada). All procedures were approved by the University of Calgary's animal care committee and were in accordance with the Canadian Council on Animal Care guidelines.
Staircase training
Rats were starved to 85% of their free-feeding weight over 3 days. Rats then were placed in the staircase boxes (Montoya et al., 1991) in a quiet room for 2 sessions per day (15-minute sessions separated by 4 to 5 hours). Rats were considered to have acquired the task when they retrieved an average of 9 pellets per side (maximum per side of 21) each day over the last 3 days with a minimum training time of 8 days (16 trials). Three rats were excluded because they did not attain this performance level.
Core temperature telemetry probe implantation
Sterilized telemetry probes (model TA10TA-F40; Data Sciences, St. Paul, MN, U.S.A.) were implanted in the peritoneal space using 1.5% to 2% halothane anesthesia. This was done after staircase training and at least 5 days before ischemia surgery. Rats were individually housed in cages placed upon receivers (RLA-1020; Data Sciences) several days before ischemia. Temperature was sampled every 30 seconds. The data collected the day before ischemia and sham occlusion surgery served as a baseline.
Middle cerebral artery occlusion model of focal ischemia
Middle cerebral artery occlusion surgery or operation was performed as previously described (Li et al., 2000). Briefly, rats were anesthetized with a mixture of 2% halothane in 70% air and 28% O2. After cannulation of the tail artery for blood pressure measurement, the right common carotid artery was isolated and a ligature placed around it. A craniotomy (2 to 3 mm) then was over the right MCA (2 to 3 mm anterior to the union of the zygomatic arch and the temporal bone). Some rats were made ischemic through permanent right carotid artery occlusion combined with the temporary placement of a microclip (#1, Codman) on the MCA for a period of 90 minutes. Other rats were sham occluded (no ischemia) over a similar time period. Regional cerebral blood flow (estimated by a laser—Doppler flowmetry probe situated over the core and penumbral regions; sampled in series), blood pressure, blood gases, plasma glucose, and hematocrit were all measured at the onset and end of ischemia or sham ischemia. Wound clips were used on the skin and anesthesia discontinued soon after the onset and end of ischemia and sham procedures. Group assignment was random. Three ischemic rats were excluded—two rats had subarachnoid hemorrhage and the MCA was ruptured on the other rat.
A feedback-controlled infrared lamp maintained core temperature (target of 37.3°C) during ischemia and sham occlusion surgery until 1 hour postischemia. Animals randomly received either postischemic hypothermia or freely regulated their own temperature. Precise temperature control (often within O.5°C of desired temperature) in the awake rat was made possible by a servo-controlled system that used fans and fine water spray to cool while infrared lamps warmed when necessary (Colbourne et al., 1996). Cooling began l hour after onset of reperfusion from ischemia or sham occlusion. Rats were cooled slowly at a rate of 1 °C per 30 minutes to 33°C and were maintained near this level for 24 hours before slow warming to 35°C (1°C per 30 minutes), which was maintained for a further 24 hours. At that time rats were warmed to 36°C and maintained between 36°C and 37°C for a further 24 hours. The slow rate of cooling and the stepwise rewarming were chosen to mimic the clinical situation. One cooled rat was excluded because of a technical problem. All rats had access to food and water throughout the experiment and did eat and drink in the postischemic period (that is, during hypothermia).
Groups
There were two sham-operated groups, one that was normothermic (SHAM+NORMO; n = 4) after sham ischemia and one that was cooled after surgery (SHAM+HYPO; n = 4). The authors combined these SHAM groups because there were only differences on all measures. Rats made ischemic were either normothermic (ISCH+NORMO; n = 16) or subjected to delayed cooling (ISCH+HYPO; n = 17) in the postischemic period. Groups (stated n) did not include rats excluded for technical reasons (for example, for failing to attain criterion in the staircase test or surgical problems).
Staircase testing
At 30 to 35 days after ischemia and sham operation, rats were starved to 85% of their previous weight. Rats then were tested in the staircase test over five days (two trials per day). Data were analyzed with mixed factor analysis of variance (ANOVA; group and trial factors) with simple effects on each trial (the latter performed in an overall ANOVA with ISCH+NORMO and ISCH+HYPO rats).
Histology
Rats were killed 35 to 42 days after ischemia or sham operation with 65 mg/kg sodium pentobarbital anesthesia and perfusion-fixed (saline flush followed by 4% formaldehyde). Brains were embedded in paraffin, coronally cut at 6 μm and stained with hematoxylin and eosin. Sections were analyzed with a Macintosh computer using the NIH Image program by a research assistant unaware of treatment groups. With the aid of a microscope, the areas of cortex in the intact and ischemic hemispheres were determined as well as the area of infarction in the hemisphere. These measurements were taken from 10 standardized sections spaced 1.0 mm apart that extended from the prefrontal cortex to the rostral pons. The volume of injury in the ischemic hemisphere was calculated as follows:
Volume = area of cortex in contralateral hemisphere – (area of cortex in ischemic hemisphere - area of infarction) × interval between sections.
These data were analyzed with one-way ANOVA.
RESULTS
Baseline core temperature (± SD) averaged over the full day before ischemia and sham occlusion was 37.3°C ± 0.1°C, 37.2°C ± 0.1°C, and 37.3°C ± 0.1°C in SHAM, ISCH+NORMO, and ISCH+HYPO groups, respectively. Average temperatures during ischemia were as follows: 36.8°C ± 0.6°C in SHAM, 36.9°C ± 0.6°C in ISCH+NORMO, and 36.4°C ± O.8°C in the ISCH+HYPO group. The ISCH+HYPO group was only slightly less than the ISCH+NORMO group but not significantly (one-way ANOVA; F2,35 = 2.73, P = 0.079). Postischemic temperature was regulated as desired (Fig. 1). Blood gases at onset of ischemia and reperfusion were not significantly different between ischemic groups (Table 1). Likewise, cerebral blood flow estimated by laser—Doppler flowmetry was not notably (significantly) different between ischemic groups in either the core or penumbral zones before or after placement or removal of the microclip (data not shown). Hypothermia transiently caused a greater loss of weight after sham and ischemia surgery (Table 2). This had recovered such that there were no significant group differences at the time of staircase retesting.
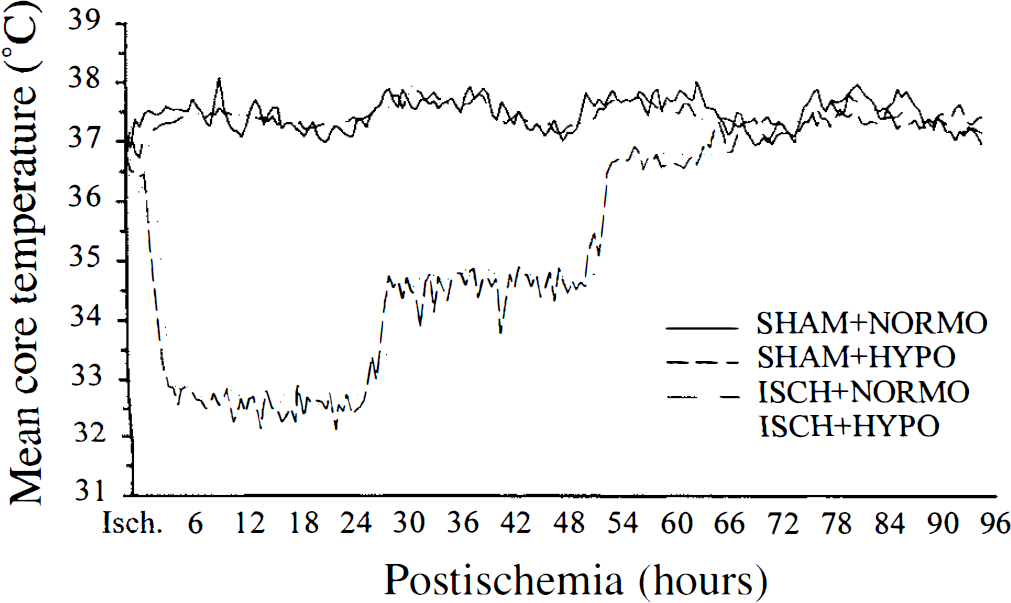
Intra- and postischemic core temperature (°C) in SHAM+NORMO (n = 4), SHAM+HYPO (n = 4), ISCH+NORMO (n = 15), and ISCH+HYPO groups (n = 16). Temperature was sampled twice each minute and is presented as 30-minute averages. Temperature was maintained during ischemia and for 1 hour postischemia (see Results). In some groups hypothermia was induced slowly starting at 1 hour after the onset of reperfusion and was maintained for over 48 hours by a servo-controlled system (see Methods).
Physiologic variable (mean ± SD) taken immediately after onset of ischemia and reperfusion
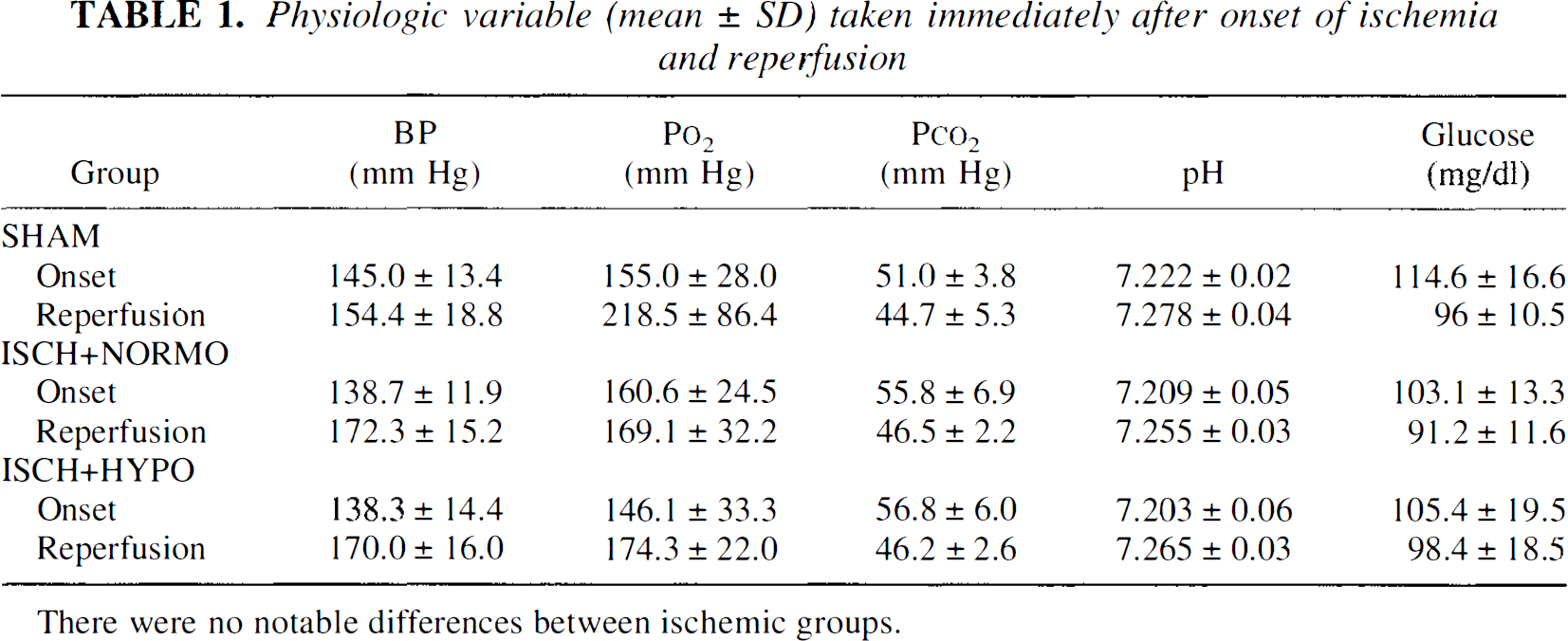
There were no notable differences between ischemic groups.
Mean (±SD) body weight (g) at various times
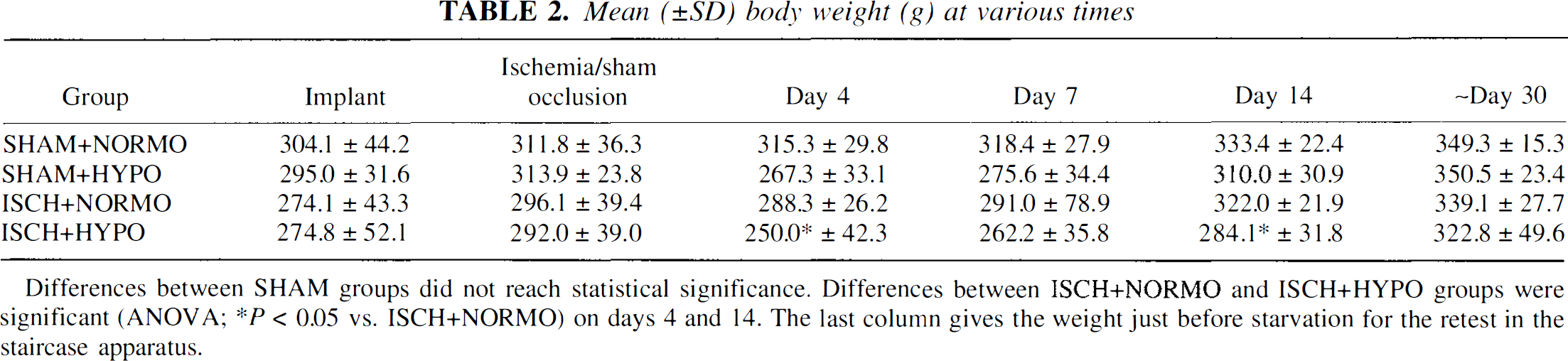
Differences between SHAM groups did not reach statistical significance. Differences between ISCH+NORMO and ISCH+HYPO groups were significant (ANOVA; *p < 0.05 vs. ISCH+NORMO) on days 4 and 14. The last column gives the weight just before starvation for the retest in the staircase apparatus.
All animals were trained to criterion in the staircase before ischemia. Testing, which started at approximately 30 days after ischemia and sham occlusion, showed that SHAM, ISCH+NORMO, and ISCH+HYPO groups performed similarly (number of pellets eaten) with the ipsilateral (right) forelimb (Fig. 2, main effect F2,36 = 1.51, P = 0.2354). On the contralateral (left) side (Fig. 2), there was a significant group effect (F2,36 = 10.42, P = 0.0003). The ISCH+NORMO group ate less than both SHAM (F1,36 = 10.78, P = 0.0023) and ISCH+HYPO rats (F1,36 = 18.19, P = 0.0001) who ate similar amounts (F1,36 < 1). Simple effects (from ANOVA with ISCH+NORMO and ISCH+HYPO rats) for each trial revealed that the ISCH+HYPO rats were more successful than untreated ISCH+NORMO rats (P = 0.0268 to 0.0001).
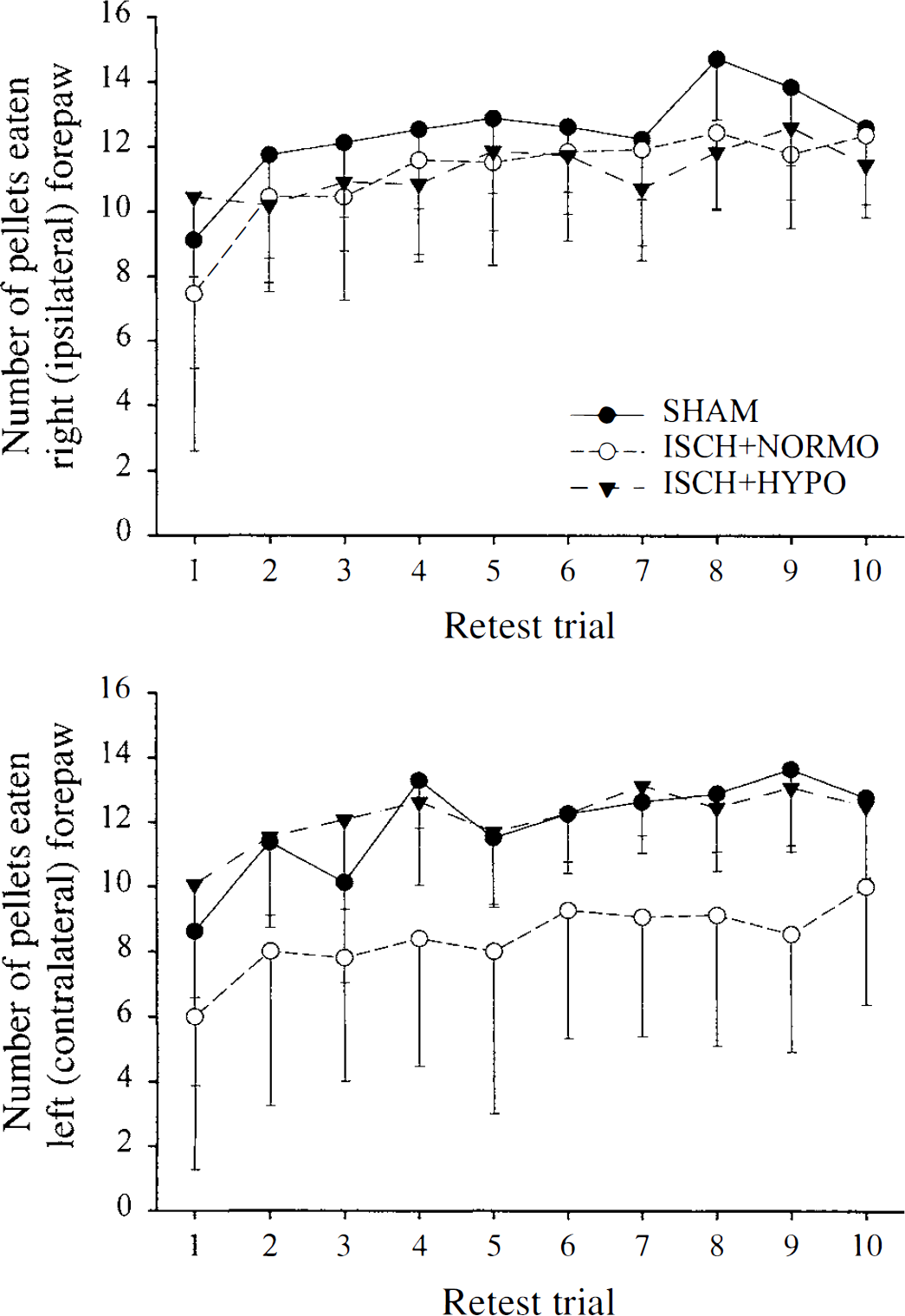
Number of pellets eaten per side in the staircase test starting at one month after ischemia (ISCH+NORMO, n = 15; ISCH+HYPO, n = 16) or sham occlusion surgery (SHAM, n = 8). There were 2 trials per day for 5 days.
The total volume (including atrophy; ± SD) of cortical tissue loss was 67.5 mm3 ± 17.2 in the ISCH+NORMO group. Hypothermia significantly (F1,29 = 23.89, P < 0.0001) reduced this damage to 35.8 mm3 ± 18.8 (Figs. 3 and 4). The remaining noninfarcted cortical tissue on the ischemic side was expressed as a percentage of the contralateral (normal) side:
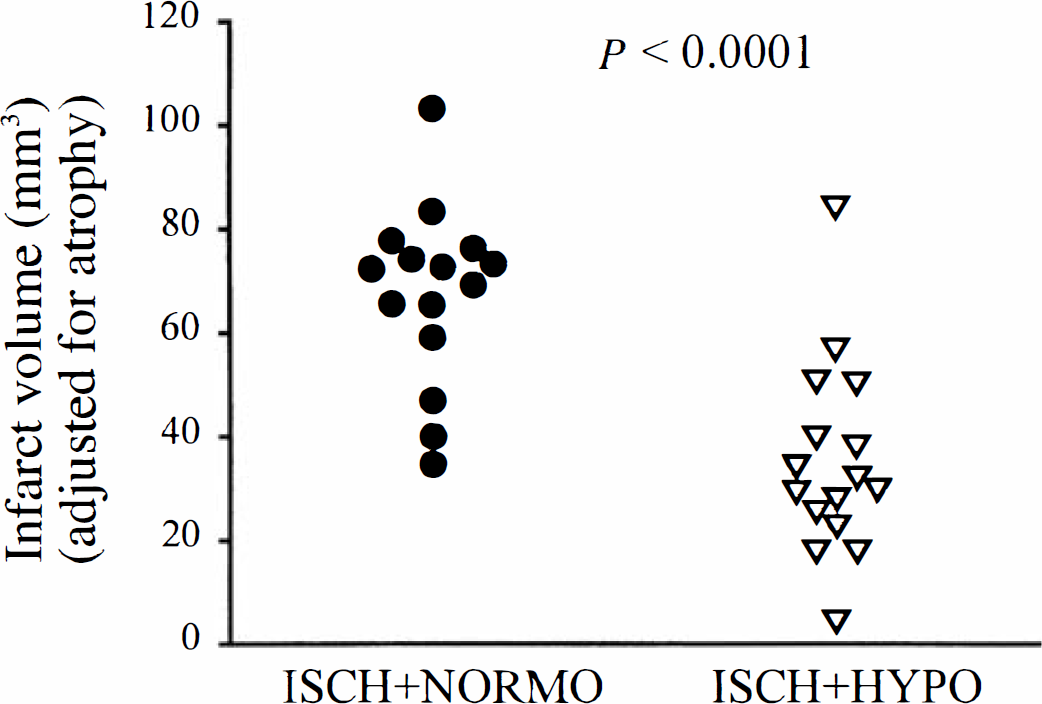
Volume of cortical infarction (corrected for atrophy) in ISCH+NORMO and ISCH+HYPO rats. Hypothermia significantly reduced injury. See Fig. 4 for representative photomicrographs.
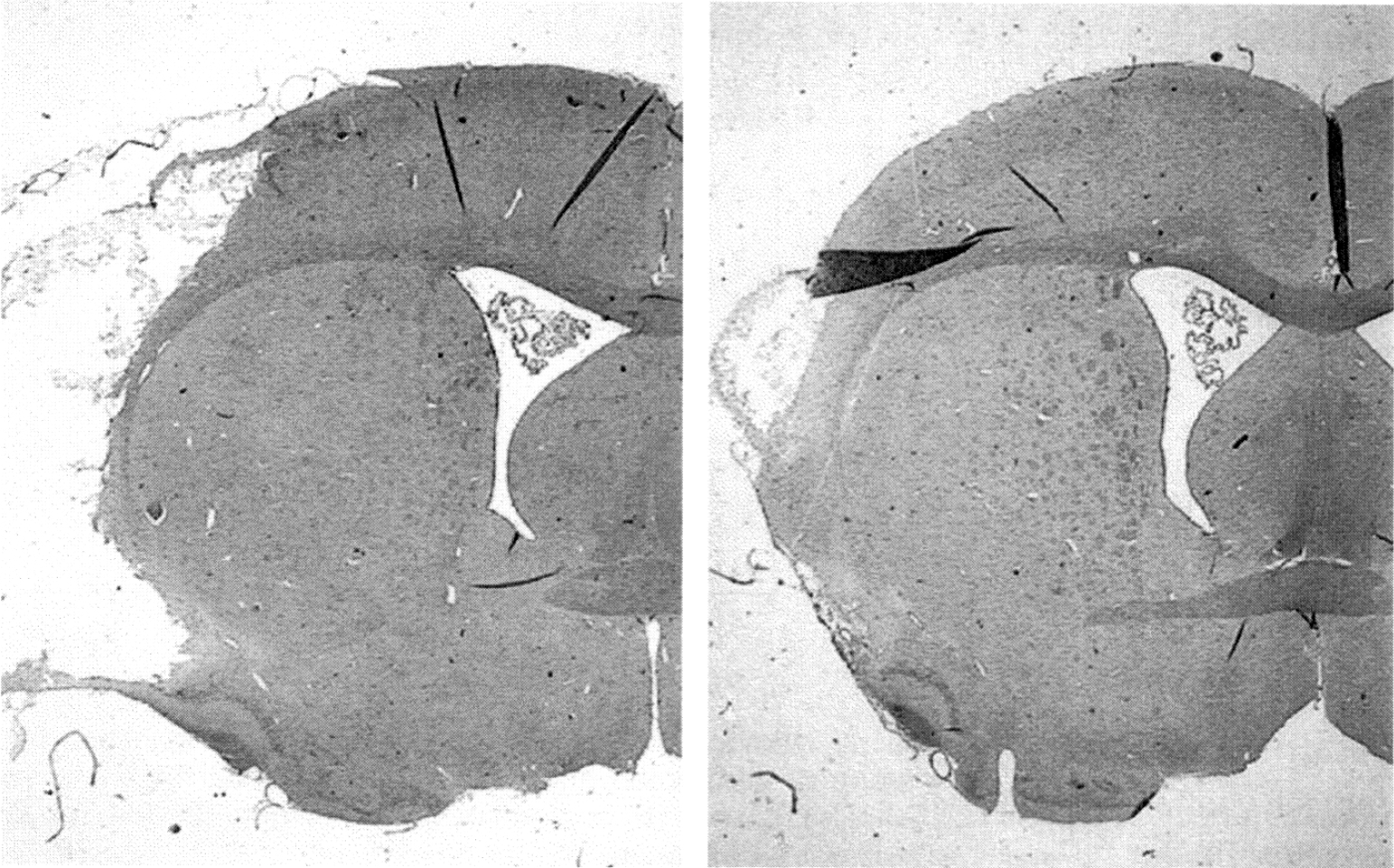
Representative examples of cortical infarction in ISCH+NORMO (left) and ISCH+HYPO (right) rats. Sections displayed illustrate the maximal extent of the injury and approximate the mean infarct for each group.
Percentage of remaining intact cortex = (volume of intact cortex in ischemic hemisphere/volume of cortex in contralateral, normal hemisphere) × 100%.
Hypothermia reduced total cortical injury from 39.3% (untreated ISCH+NORMO group) to 23.7%. The infarct volumes corrected for atrophy in ISCH+NORMO and ISC H+HYPO rats combined significantly predicted (r = −0.574, P = 0.0007) the mean number of pellets retrieved on the retest. The correlation for only ISCH+HYPO rats is −0.507 (P = 0.045).
Two rats died, 1 on day 6 (ISCH+HYPO) and the other on day 11 postischemia (ISCH+NORMO). The first rat sustained a seizure whereas the second lost significant weight. These rats were not included in the data analysis.
DISCUSSION
Temporary MCAO in the rat caused substantial cortical infarction and significant impairments in the rat's ability to retrieve food pellets in the Montoya staircase test a month after ischemia. Prolonged cooling, which began 2.5 after the onset of ischemia, significantly and persistently reduced the volume of brain injury and completely blocked the reaching deficits in the staircase apparatus.
Other focal ischemia studies have found protection with postischemic hypothermia (for example, Chen et al., 1992; Yanamoto et al., 1996; Maier et al., 1998; Corbett et al., 2000). However, these studies are by the use of short survival times (however, see Corbett et al., 2000), or the lack of functional assessment, or both. The use of short survival times is problematic because cell death has been shown to continue for days or weeks after mild ischemic insults (Du et al., 1996). This condition is analogous to that created by a partially effective neuroprotective intervention. For example, brief duration (for example, 2 to 3 hours) postischemic hypothermia may appear quite effective with short survival times (that is, <1 week), but in reality may only postpone cell death as has been noted in global ischemia (Dietrich et al., 1993). The authors' previous study (Corbett et al., 2000) addressed the issue of long-term benefit after prolonged (that is, >24 hours) postischemic hypothermia because a persistent reduction in cortical, and to a lesser extent striatal, infarct size was seen at a survival time of 60 days. However, a hybrid stroke model (intraluminal suture occlusion + systemic hypotension) was used in that experiment, thus the result might not apply to pure focal ischemia. Furthermore, it is clear that functional outcome measures are of paramount importance in assessing new neuroprotective therapies (Corbett and Nurse, 1998) because improved behavioral outcome is the most important goal of stroke treatment. Thus, the current findings of persistent histologic and functional protection in a standardized stroke model is an important advance over previous efficacy studies, especially given that hypothermia was delayed 2.5 hours after the onset of ischemia. The preservation of forelimb dexterity in the staircase task is especially significant because stroke patients with upper limb dysfunction frequently regain coarse motor control of the shoulder, elbow, and hand, but do not regain fine sensorimotor control of the fingers (Gowland, 1987). Likewise, rats with MCAO tend not to have appreciable recovery of skilled forelimb use (Grabowski et al., 1993).
The ideal hypothermic therapy is unknown. In global ischemia, brief cooling is either ineffective or only transiently beneficial (Dietrich et al., 1993), whereas longer bouts of hypothermia convey substantial protection (Colbourne et al., 1997, 1999b). Furthermore, longer durations of hypothermia not only increase protection but also extend the therapeutic window to at least 12 hours postischemia. Because no study has compared diverse periods of hypothermia in focal ischemia (for example, 12 vs. 24 vs. 48 hours), it is unknown whether the current period of hypothermia was necessary or sufficient and whether this therapy would universally be maximally efficacious. Studies to address these issues, especially the therapeutic window, are planned.
Importantly, hypothermia has been safely induced for protracted durations in trauma (Clifton, 1995; Marion et al. 1997; Shiozaki et al., 1998), stroke (Schwab et al., 1998), and cardiac arrest survivors (Zeiner et al., 2000). Others urge caution because of the increased risk of complications such as infection with prolonged hypothermia (>24 hours) (Schubert, 1995). However, many of these complications are more at temperatures below 32°C (Schubert, 1995; Colbourne et al., 1997) and progress is being made in reducing infections after hypothermic treatment of patients with head injuries by administration of agents (for example, granulocyte colony stimulating factor) that elevate neutrophil function (Ishikawa et al., 1999). Nonetheless, until management concerns are thoroughly resolved, an alternative approach is to combine briefer durations of mild hypothermia with neuroprotective drug therapy, thereby minimizing any untoward effects of hypothermia per se. In this regard, it is important to note that several studies (Dietrich et al., 1999) show that combination therapy of brief hypothermia and antiinflammatory and antipyretic drugs (for example, IL-10 antagonist) can chronically protect hippocampal CA1 neurons. The authors suspect that, at present, prolonged cooling alone provides much better protection (Colbourne et al., 1997). Nonetheless, combination therapy of prolonged cooling and neuroprotective drugs might prove optimal. Further studies must address these possibilities in global and focal cerebral ischemia.
The Montoya staircase test clearly distinguished between untreated and hypothermia-treated ischemic rats. Performance with the contralateral limb significantly predicted volume of infarction in ischemic animals and within the hypothermia-treated ischemic rats. However, contrary to some previous studies (Montoya et al., 1991; Cregan et al., 1997), the authors did not see bilateral deficits, only contralateral deficits. The reasons for this are not clear but may relate to the size and type of lesion made as well as pretraining schedules and time of assessment. For example, ipsilateral reaching deficits after MCAO are much smaller in rats with staircase training than in rats without staircase training before ischemia (Grabowski et al., 1993). A more important and more difficult question is whether benefit in the staircase test from hypothermia treatment originates from true preservation of function, or facilitation of compensatory movement strategies, or both. There is potential for compensatory strategies to mask deficits over the course of recovery. Thus, although the ideal assessment time or times with the staircase test in this MCAO model are unknown, an intermediate survival time of one month is at least useful in detecting group differences. These issues will be addressed in future studies.
In summary, the current study demonstrates for the first time long-term functional and histologic protection in a model of focal ischemia. These findings along with other recent studies in global and focal ischemia, an epidemiologic study in human stroke victims that suggests that spontaneous hypothermia is beneficial (Wang et al., 2000), and data indicating that fever is detrimental (Ginsberg and Busto, 1998), all strongly support the clinical investigation of hypothermia in stroke.
Footnotes
Acknowledgments
The authors thank Kathy McKay and Carol Jocelyn for technical assistance.
