Abstract
Nicotinamide phosphoribosyltransferase (NAMPT) is the key enzyme for the synthesis of nicotinamide adenine dinucleotide (NAD) in the salvaging pathway. Though NAMPT inhibitors such as FK866 were originally developed as anti-cancer drugs, they also display neuroprotective effects. Here we show that the administration of FK866 at 0.5 mg/kg (ip, qod) for four weeks, i.e., ∼1% of the dose used for the treatment of cancer, significantly alleviates the aging-induced impairment of cognition and locomotor activity. Mechanistically, FK866 enhanced autophagy, reduced protein aggregation, and inhibited neuroinflammation indicated by decreasing TNFα, IL-6, GFAP, and Iba1 levels in the aged mouse brain. Though FK866 did not affect the total NAD and nicotinamide mononucleotide (NMN) levels in the mouse brain at the dose we used, FK866 increased nicotinamide (NAM) level in the young mouse brain and decreased NAM level in the aged mouse brain. On the other hand, FK866 did not affect the serum glucose, cholesterol, and triglyceride of young and aged mice and exhibited no effects on the various indices of young mice. Thus, the NAMPT inhibitor can be repurpose to counteract the cognitive impairment upon aging. We also envision that NAMPT inhibitor can be used for the treatment of age-related neurodegenerative diseases.
Introduction
Aging is a significant risk for neurodegeneration, which impairs learning and memory. 1 There have been many proposed mechanisms for aging. Among them, the chronic neuroinflammation and the aggregation of toxic and misfolded proteins in the central nervous system are primary mechanisms for aging-induced cognitive impairment.2,3 Glia cell-mediated chronic neuroinflammation, including microglia and astrocyte activation, has already become a target for the prevention and treatment of neurocognitive disorders in the aging population. 4
Toxic and misfolded proteins increase upon aging. Such proteins can be ubiquitinated and degraded via ubiquitin-proteasome-system (UPS). However, the UPS activity declines progressively upon aging.3,5 As a result, the ubiquitinated proteins can aggregate and form aggresome. The aggregated proteins can be further directed to autophagy-lysosome pathway for clearance. However, the autophagy turnover also decreases with aging, which would cause further accumulation of protein aggregate. The accumulation of protein aggregate has been shown to induce neuronal death and sustained neuroinflammation.3,5 Thus, both proteasome enhancers and autophagy inducers exemplify new strategies for the treatment of age-related brain diseases.6,7
At another side, metabolism dysfunction is a hallmark of brain aging. For example, nicotinamide adenine dinucleotide (NAD) is a critical metabolite for the oxidative phosphorylation and citric acid cycle. NAD is also an essential co-factor for NAD-consuming enzymes including sirtuins and poly(ADP-ribose) polymerases (PARPs). 8 NAD level declines continuously upon aging owing to the increased consumption and decreased synthesis.9–11 In the aged brain, PARPs, especially PARP1, become over-active due to sustained DNA damage and reactive oxygen stress, and consume a large amount of NAD to generate poly(ADP-ribose) (PAR). Though PAR formation is important for DNA repair. Nevertheless, excessive PAR may cause neuronal death.12–14 NAD is mainly synthesized via the salvaging pathway in neurons. In this pathway, nicotinamide (NAM), derived from the NAD-consuming enzymes, is converted to nicotinamide mononucleotide (NMN) by nicotinamide phosphoribosyltransferase (NAMPT), which is further converted to NAD by adenylyl transferase (NMNAT). 15 NAMPT is the key enzyme in the NAD salvaging synthesis pathway, and the decline of NAMPT protein expression and NAMPT activity can be an important cause for the decrease of NAD in aging.9–11 Thus, NAD precursors can increase intracellular NAD level and be beneficial to the organism,16–18 whereas the inhibition of NAMPT or knockout of Nampt gene can impair the cognitive behaviors of mice.19,20
FK866 is a potent inhibitor of NAMPT with an IC50 value of several nanomolar.21,22 Cancer cells have high demand for NAD and the NAMPT expression level is usually high. Therefore, NAMPT inhibitors including FK866 were originally developed as anti-cancer drugs.23–25 Yet, it has been shown that NAMPT inhibitors also exert potent anti-inflammatory effects in the peripheral system.26,27 We and others have shown that NAMPT inhibitors have neuroprotective effects, mainly through their anti-neuroinflammation effects. For instance, FK866 protects the mouse brain against cryo-injury of the mouse brain, 28 spinal injury, 29 and ischemic injury. 30
NAMPT is absent in microglia in the young mouse brain but expresses in microglia in the aged mouse brain, and FK866 can inhibit the activation of microglia.11,31 We have also shown that FK866 at both cellular toxic (10−8-10−7 M) and non-toxic (10−9 M) concentrations induces autophagy in C6 glioma cells. FK866 at 10−7 M and 10−9 M increases the degrading autophagic vacuoles and the initial autophagic vacuoles, respectively. 32 FK866-induced autophagy can be protective. For example, FK866 attenuates acute hepatic failure by inducing autophagy. 33 As illustrated in Figure 1(a), both glia-mediated neuroinflammation and protein aggregation can cause cognitive impairment in the aged brain. Thus, it is interesting to know whether FK866 can decrease protein aggregation by enhancing autophagy and can inhibit glia-mediated neuroinflammation, thus beneficial to the aged brain.
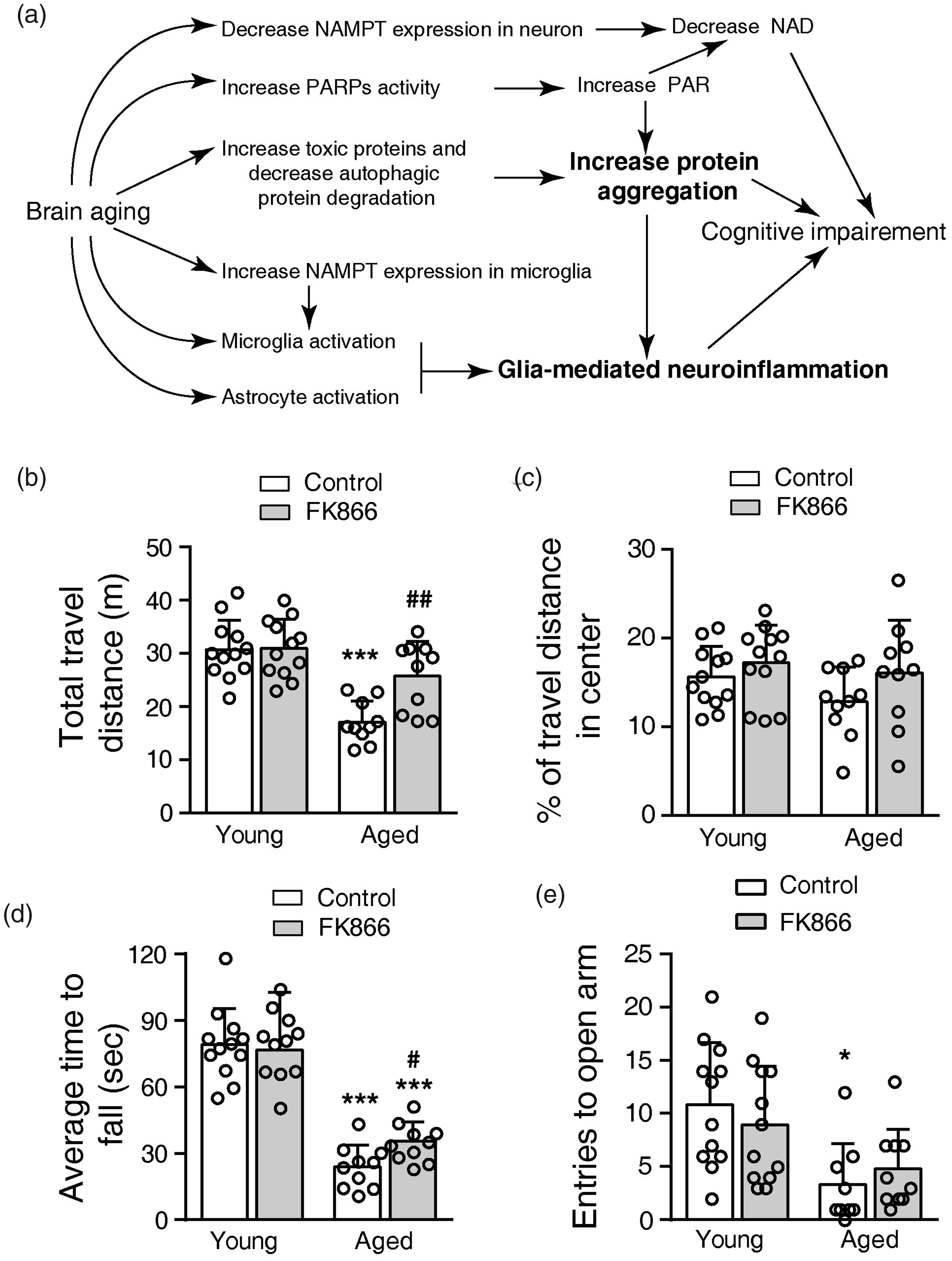
Effects of FK866 (0.5 mg/kg ip qod for six weeks) on the motor activity, motor skill learning, and anxiety behaviors of the mice. (a) Illustration of several changes in the aging brain that induces cognitive deficiency in mice. NAD, Nicotinamide adenine dinucleotide; NAMPT, Nicotinamide phosphoribosyltransferase; PAR, Poly(ADP-ribose); PARPs, Poly ADP-ribose polymerases. (b) Total distance traveled in the open field. ***P < 0.001, compared with young control mice, ##P < 0.01, compared with aged control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. (c) Percentage of distance traveled in the center of the open field. (d) Average time to fall from the Rotarod. ***P < 0.001, compared with young control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. (e) Entries to open arm in the elevated cross maze. *P < 0.05, compared with young control mice, one-way ANOVA with Kruskal-Wallis test. N = 12 for young groups, and N = 10 for aged groups.
Here we show that the administration of FK866 at a low and non-toxic dose, far below the dose for cancer treatment, leads to cognitive improvements in the aged mice. We show that FK866 enhances autophagy, decreases protein aggregates, and inhibits glial-responses in the aged mouse brain. Thus, our finding indicates that FK866, a classical NAMPT inhibitor, can be repurposed for the treatment of aging-induced cognitive impairment.
Materials and methods
Animals
The mice were purchased from Zhejiang Academy of Medical Science. All mice were kept in the Laboratory Animal Center, Zhejiang University School of Medicine. And they had free access to water and food in air-conditioned rooms (20∼26°C, relative humidity ∼50%) on a 12-h light/dark cycle. Mice were handled following the Guide for the Care and Use of the Laboratory Animals of the National Institutes of Health. The experimental protocols were approved by the Ethics Committee of Laboratory Animal Care and Welfare, Zhejiang University School of Medicine, with the proven number ZJU2015-012-02. The data reporting were conducted in compliance with the ARRIVE guidelines (PMID: 3,26,63,096). 34
Mice information were illustrated in Figure S1. Thirty-four C57BL/6J female mice at 20 months old (aged mice) and 36 C57BL/6J female mice at 3 months old (young mice) were used for the formal experiments. Another 12 C57BL/6J female mice at 16 months old and 6 C57BL/6J female mice at 3 months old were used for the pilot experiments. In this batch, one aged mouse was sacrificed due to the dramatic decrease in body weight.
Pilot experiments to determine the dose of FK866
To determine the proper dose of FK866, five aged mice were intraperitoneal (ip) injected with 1 mg/kg FK866 twice a week (biw). FK866 was purchased from Shanghai Fuqi Pharmaceutical Technology Co., Ltd. (Shanghai, China, CAS: 658084-64-1). Sequential behavioral tests were performed to evaluate the cognitive functions of mice. The serum glucose, cholesterol and triglyceride were determined to assess the side effects of FK866.
Based on pilot experiment results, FK866 was adjusted to ip injection with 0.5 mg/kg every other day (qod) in the formal experiments. In the day with behavioral tests, FK866 was injected in the evening after behavioral test has been finished. Normal saline was injected as a control.
Behavioral tests
As indicated in Figure S1(a) and (b), the indicated mouse received six behavioral tests according to our previous report. 16 Before the first test, all mice were gently handled for four days (5 min/day) to minimize the stress of mice to the handle. Before each test, mice were transferred to the experimental room 30 min in advance to adapt to the environment. Experiments were started at 9:30 in the morning. Between each test, the equipment was thoroughly cleaned to minimize the effect of odor from the previous mouse.
Open-field test
This test was used to evaluate the locomotor activity and the anxious behavior of mice. The open-field test was performed in the field test system (ViewPoint Behavior Technology, France), with the box size 45 cm × 45 cm × 45 cm. Each mouse was gently put into the center of the box and was free to move. The movement was recorded for 10 min. The total traveling distance and the percentage of the distance in the center area (1/4 of the total area) were calculated.
Y-maze test
Two trial strategy was used for Y-maze test to evaluate the recognition memory of mice. Three arms of Y-maze (30 cm ×10 cm ×15 cm for each arm, Any-Maze test system) were labeled as 1st arm, 2nd arm, and closed or novel arm. In the first trial (for the training), the novel arm was closed. The mouse was put into the 1st arm and allowed to explore the two open arms freely. After this training trial, the mouse was returned to the home cage for 2 hrs. Then, in the second trial (for the testing), all three arms were accessible. The mouse was put into the 1st arm again for 10 min testing. The movement of the mouse was recorded. The entry numbers to each arm and the time spent in each arm were analyzed. The recognition memory index (%) = (time spent in the novel arm/total time spent in all arms during the first minute of the second trial) × 100.
Novel object recognition
NOR was used to evaluate the object recognition memory of mice using the open-field test system in a 45 cm × 45 cm × 45 cm box. During training, two identical objects (blue cones, 5 cm diameter in the bottom and 5 cm in tall) were placed diagonally. The mouse was put at the center of the box. The movement of the mouse was recorded for 10 min. Then the mouse was returned to the home cage. Two hours later, the mouse was put in the same box and was recorded for 10 min as testing. Before testing, one object was replaced by an object in distinct color and shape (yellow cub, 5 cm × 5 cm in the bottom, and 10 cm in tall), and the replaced object was identified as a novel object. The number of visits (sniff number) and time spent around the old and novel objects was recorded and analyzed.
Elevated plus-maze
EPM was used to evaluate the anxious behavior of mice. The Any-Maze test system (Global Biotech Inc. Shanghai, China) has two open arms and two close arms (30 cm × 5 cm × 15 cm for each arm), and was 45 cm elevated above the floor. The mouse was put in the center of the plus-maze with its nose towards an open arm. The movement of the mouse in the maze was recorded for 5 min. The entries into the open arms were analyzed—the more entries into the open arms, the lower the anxiety.
Rotarod test
Rotarod test was used to evaluate the motor skill learning of mice using an accelerating Rotarod (LE8205; Panlab, Barcelona, Spain). During training, the mouse was subjected to five trials until fall with 10 min interval between each test. For testing, the mouse was placed on the rod with an accelerating rotating speed from 4 to 40 rpm in 300 sec. A total of 8 experiments was performed, the first three trials were used as training, and the last five tests were used for the analysis of average time to fall. The average time to fall was used to evaluate the performance of mice.
Fear conditioning test
Fear conditioning test was performed in the apparatus (ACT-100A, Coulbourn Instruments Inc., Lehigh Valley, PA, USA). Contextual memory was used to determine the hippocampus-dependent fear memory. Cued memory was used to determine the amygdala-dependent fear memory. During training, the mouse was put into the context for 2min (indicated by “training in boxA”), then an 85 dB, 3 kHz tone was activated for 30 sec. Two seconds before the end of the tone, a 2 seconds foot shock was delivered (1 mA). Thirty seconds after foot shock, the mouse was returned to the home cage. Twenty-four hours after training, mice were put in the same apparatus receiving foot shock, and allowed to explore freely in 5 min (indicated by “testing in boxA”). The contextual memory was evaluated by measure the freezing time (lack of motion) in boxA. Two hours after the contextual memory test, mouse was placed in a context containing distinct visual and olfactory cues. After 2min in the novel context (indicated by “without tone in boxB”), the tone accompanied with foot shock was applied for 3 min (indicated by “with tone in boxB”). The cue memory was evaluated by measure the freezing time in boxB.
The measurement of serum glucose, cholesterol and triglyceride
Mice were euthanized by intraperitoneal injection of pentobarbital sodium (250 mg/kg) before sacrifice. Around 0.5 ml of blood was taken transcardially. The blood was placed under room temperature for 40 min and under 4°C for 4–5 hours. After centrifuged at 3000 rpm for 10 min, the supernatant was carefully removed for the measurement of glucose, cholesterol, and triglyceride.
A triglyceride detection kit (Beckman Coulter, CA, USA), total cholesterol detection kit ((Beckman Coulter, CA, USA), and glucose detection kit (Beckman Coulter, CA, USA) was used. The serum concentrations of glucose, triglyceride, and cholesterol were measured according to the manuscript.
Immunofluorescence staining
After sacrifice, mice were transcardially perfused with 4°C saline and fresh prepared 4% paraformaldehyde. The brains were removed and fixed in 4% paraformaldehyde at 4°C for one day, and transferred to 30% sucrose 3–5 days for dehydration.
The fixed brains were sliced into 25 µm thick slices using cryomicrotomy (CM1900, Leica, Wezlar, Germany). Brain slices were incubated in 0.1% Triton-X PBS for 30 min, followed by 5% donkey serum for 1 hr. Then the slices were incubated with mouse anti-GFAP (glial fibrillary acidic protein) antibody (1:600, Millipore, MAB360, Billerica, MA, USA), rabbit anti-Iba1 (ionized calcium-binding adapter molecule 1) antibody (1:500, Wako, 019-19741, Osaka, Japan), rabbit anti-NeuN (neuronal nuclei) antibody (1:500, Cell Signaling Technology, #12943S, Boston, MA, USA) and mouse anti-ubiquitin antibody (1:300, Santa Cruz Biotechnology, sc-8017, Santa Cruz, CA, USA) at 4°C overnight. After washed with PBS (10 min × 3 times), the slices were incubated with Cy3-conjugated donkey anti-rabbit IgG antibody (1:200, Millipore, AP182C) or FITC-conjugated goat anti-mouse IgG antibody (1:200, Millipore, AP124F) for 2 hr. After washing with PBS (10 min × 3 times), the slices were mounted using an anti-fade medium containing DAPI (Invitrogen Corp., Carlsbad, CA, USA). Images were taken under an Olympus FV100 confocal microscope (Olympus, Japan). For each staining, three separate mice were used.
Preparation of soluble and insoluble protein samples
After sacrifice, mice were transcardially perfused with 4°C saline. The brains were quickly removed, and were separated as cortex and subcortex. Total protein from brain tissues was extracted using RIPA Lysis Buffer (P0013B, Beyotime Biotechnology Research Institute, Jiangsu, China). After adding lysis buffer (0.5 ml/100 mg tissues), the brain was thoroughly homogenized with a Heidolph Diax 900 homogenizer (Heidolph Instruments GmbH & CO., Schwabach, Germany) for 1 min at 4°C. The supernatant of the mixture was collected by centrifugation at 14,000 g for 30 min. The supernatant was identified as a soluble protein sample, which was blotted for measuring the level of GFAP, CD11b, LC3, TNFα, and IL-6.
After the centrifugation at 14,000 g for 30 min, the precipitation was resuspended in PBS. After centrifuged at 13000 rpm at 4°C for 15min, the precipitation was resuspended in SDS buffer (2% SDS, 50 mM Tris-HCl, pH 7.5), and followed by ultrasonication (Diagenode, Belgium) for eight cycles at 4°C (10 sec on and 30 sec off for one cycle). After centrifuged at 13000 rpm at 4°C for 30 min, the supernatant was collected as an insoluble protein sample, which were blotted for measuring the level of ubiquitin representing protein aggregation.
The protein concentration of soluble and insoluble part was determined by using BCA Protein Assay Kit (Beyotime Biotechnology Research Institute, Jiangsu, China. P0009).
Western blotting analysis
Protein samples (100 µg) were used for western blotting, and the following antibodies were used: rabbit anti-GFAP (1:1000), rabbit anti-CD11b (1:1000, Abcam, ab133357, Cambridge, UK), mouse anti-GAPDH (1:5000, Proteintech, 60004-1-Ig, Wuhan, China), rabbit anti-LC3 (1:1000, Sigma, L7543, USA) and mouse anti-ubiquitin antibody (1:300). The secondary antibody was IRDye™ 800-conjugated affinity-purified anti-mouse (1:12000, LI-COR, Bioscience, Lincoln, NE, USA) or anti-mouse IgG (1:3000, CST,7076S, USA) or anti-Rabbit IgG (1:10000, Jackson, 111-035-003, USA). The immunoblots were then measured using an Odyssey fluorescent Scanner (LI-COR) and G:BOX Chemi XRQ gel doc system (Chemi XR 5, Syngene, UK).
After blotting of ubiquitin, the PVDF membrane was stained by Ponceau (P0022 Beyotime Biotechnology Research Institute, Jiangsu, China) for 5min, and washed with distilled water 3 times × 5min. Images were taken by Huawei cell phone (P30, Shenzhen, China), and the intensity was analyzed by using Image J (ImageJ 1.47v, National Institutes of Health, USA).
The results for soluble protein samples were normalized to GAPDH (as a loading reference), and the results for insoluble protein samples were normalized to Ponceau staining (as a loading reference). Then the expression was normalized to the young control mouse samples on the same immunoblot membrane.
The measurement of TNFα and IL-6 expression in mouse brains
The level of TNFα and IL-6 was measured in the soluble protein samples by using commercial enzyme-linked immunosorbent assay (ELISA) kits according to the manual. The TNF-α ELISA kit (EK0527) and IL-6 ELISA kit (EK0411) were purchased from BOSTER Biological Technology Co.Itd (CA, USA). The ELISA results were calculated as pg/mg protein.
The measurement of total NAD, NAM and NMN level
The total NAD, NAM, and NMN levels in subcortex were measured using commercial kits. The NAD+/NADH Quantification Colorimetric Kit was purchased from BioVision (#K337-100). A mouse NAM ELISA kit was purchased from Ruixin Biotech (Quanzhou, Fujian, China, SU-B28743). A mouse NMN ELISA kit was purchased from Lunchangshuo (Biotech, Xiamen, Fujian, China, #23400). Briefly, 5 µl lysis buffer was used for 1 mg brain tissues. The result of total NAD level was calculated as pmol/mg brain tissue. The level of NAM and NMN was calculated as ng/mg brain tissue.
Statistical analysis
Data are presented as mean ± SD. The GraphPad Prism Software (version 9.0, GraphPad Software Inc., San Diego, CA, USA) was used for statistical analysis. “N” indicates the mice used in each group. ROUT test was used to identify outliers. Agonstino & Pearson omnibus normality test was performed to test the normality of the data when the data have the N ≥ 8. The Brown-Forsythe test was performed to assess the equal variances of the data. If the data pass the normality test and equal variance test, we used parametric one-way ANOVA (Newman-Keuls multiple comparisons test) or two-side paired t-test to assess the difference between means. If not, then nonparametric statistics (Kruskal-Wallis test or Wilcoxon matched-pairs signed rank test) was used to evaluate the difference among medians. A value of P < 0.05 was considered statistically significant.
Results
The pilot experiments showed FK866 at 1 mg/kg (biw, ip) improved learning and memory of aged mice
In the pilot experiment (Figure S1(a)), five aged mice were injected with 1 mg/kg FK866 (ip, biw) for four weeks before the sequential behavioral tests. In the open-field test, the control aged mice traveled 37.9% less distance than the young mice. FK866 treated aged mice traveled a longer distance than the control aged mice but with no significant difference (Figure S2(a)). The three groups of mice traveled a similar percentage of distance in the center of the open field (Figure S2(b)). In the Rotarod test, the aged control mice stayed 38.4% shorter than the young mice on the rotarod. FK866 treatment significantly improves this task performance for aged mice, and the FK866 treated aged mice performed similarly as the young mice (Figure S2(c)). On the elevated cross maze test, the three groups of mice had a similar number of entries to the open arms (Figure S2(d)). Thus, FK866 at 1 mg/kg (biw, ip) improved the motor activity and the motor skill learning of aged mice but did not affect the anxiety activity of the aged mice.
In the novel object recognition test, the three groups of mice had comparable sniff numbers to two objects during training (Figure S3(a)). During testing, the young mice and the FK866 treated aged mice showed a preference for the novel object, while the aged control mice did not (Figure S3(b)). In the Y-maze, the three groups of mice had similar entries to two arms during training (Figure S3(c)). During testing in the Y-maze, the young mice and the FK866 treated aged mice had more entries to the novel arm, and the aged control mice had similar entries to all three arms (Figure S3(d)). The aged control mice had a significantly lower recognition memory index than the young mice, and the FK866 treated aged mice (Figure S3(e)).
In the fear-conditioning test, all mice had a similar low freezing percentage in the box before foot shock (training). Twenty-four hours after the foot shock, all mice had significantly higher freezing percentages in the same boxA. The aged control mice had a significantly lower freezing percentage than the young mice, and the FK866 treated aged mice (Figure S3(f)). For the cue memory test, all mice had a similar low freezing percentage in boxB without the tone accompanied with food shock. After turn on the tone, both young mice and FK866 treated aged mice had a higher freezing percentage, and the freezing percentage of aged control mice did not increase (Figures S3(g)).
The serum cholesterol increased in aged control mice when compared with young mice. The administration of FK866 slightly decreased the serum cholesterol level of aged mice (Figure S4(a)). The serum triglyceride level was similar among the three groups of mice (Figure S4(b)). Surprisingly, the administration of FK866 significantly increased the serum glucose level by 47.7% compared with the aged control mice (Figure S4(c)).
The administration of FK866 at 1 mg/kg (biw, ip) significantly improved the recognition function of aged mice. However, the increase of the serum glucose by FK866 may also improve the recognition function of the aged mice, which may perturb our evaluation of the neurological effects of FK866. To minimize the effect on serum glucose, we decreased the single FK866 dose from 1 mg/kg to 0.5 mg/kg in the subsequent experiments. The injection frequency was increased from twice a week (biw) to every other day (qod). In addition, we applied FK866 to both young and aged mice.
FK866 at 0.5 mg/kg (qod, ip) had no effects on blood index and anxiety behaviors
The administration of FK866 at 0.5 mg/kg (qod, ip) did not affect the level of serum glucose, cholesterol, and triglyceride, for either young or aged mice (Figure S5(a) to (c)).
In open-field, the aged control mice traveled 44.3% less distance than the young control mice. FK866 increased the travel distance of the aged mice by 50.5% but did not affect that of young mice (Figure 1(b)). The mice in the four groups had a similar percentage of travel distance in the center (Figure 1(c)). In the Rotarod task, the aged control mice stayed 69.8% less time than the young control mice. FK866 increased the stay duration for 37.7% of the aged mice, but it did not affect the young mice (Figure 1(d)). On the elevated cross maze, the aged control mice spent less time in the open arm than the young control mice. The administration of FK866 did not affect the performance of either young or aged mice (Figure 1(e)). Thus, FK866 at 0.5 mg/kg (qod, ip) also improved the motor activity and motor skill learning of aged mice but did not change the anxiety behaviors of young and aged mice.
FK866 at 0.5 mg/kg (qod, ip) improved the recognition and fear memory of aged mice
During training in the novel object recognition test, all mice in the four groups spent a similar amount of time and had comparable number sniffs to the two identical objects (Figure 2(a) and (c)). During testing, both control and FK866 treated young mice preferred the novel object. They spent more time and had more sniff number to the novel object (Figure 2(b) and (d)). The aged control mice spent similar time around the two objects, and had identical sniff number to the familiar and the novel object. FK866 treatment significantly increases the preference of aged mice to the novel object, as indicated by increasing time and sniff number to the novel object (Figure 2(b) and (d)).
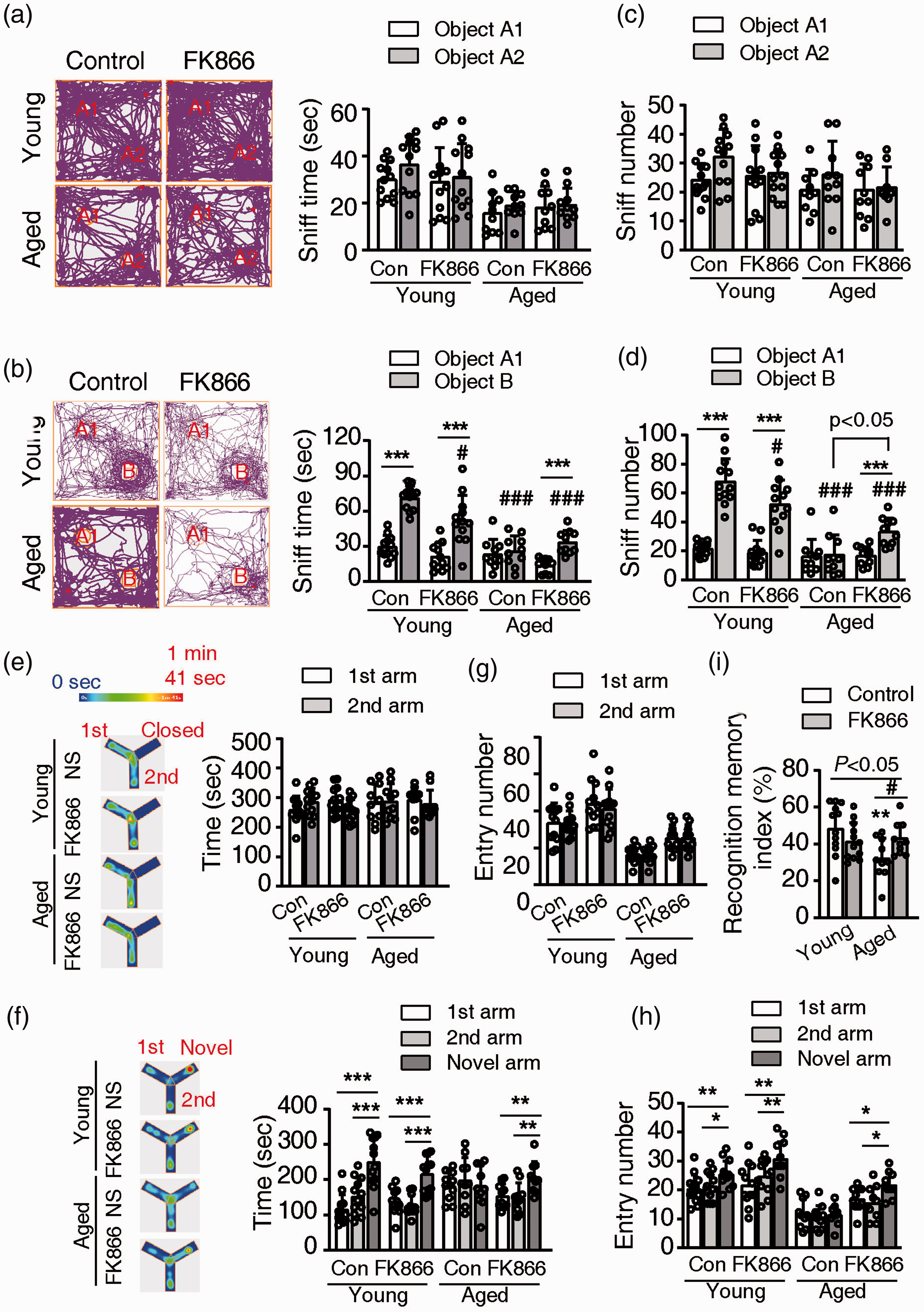
Effects of FK866 (0.5 mg/kg ip qod for six weeks) on recognition memory of the mice. (a and b). The sniff time to the two novel objects during training (a) and to the old/novel objects during testing (b) in the open field. Left panel, representative tracing images of mice traveled. Right panel, statistical analysis of the sniff time to the two objects. ***P < 0.001, two tail paired t-test. #P < 0.05, ###P < 0.001, compared with the sniff time to the novel object (object B) of young control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. (c and d). The sniff number to the two novel objects during training (c) and to the old or novel objects during testing (d) in the open field. ***P < 0.001, two tail paired t-test. #P < 0.05, ###P < 0.001, compared with the sniff number to the novel object (object b) of young control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. (e and f). Entry time to the two open arms during training (e) and to the three arms (two arms were old, and one arm is novel) during testing (f) in the Y-maze. Left panel, representative heat maps of mice traveled in the Y-maze. Right panel, statistical analysis of the entry time to the arms. **P < 0.01, ***P < 0.001, one-way ANOVA with Newman-Keuls multiple comparisons test. (g and h). Entry numbers to the two open arms during training (g) and to the three arms (two arms were old, and one arm is novel) during testing (h) in the Y-maze. *P < 0.05, **P < 0.01, ***P < 0.001, one-way ANOVA with Newman-Keuls multiple comparisons test. (i). The recognition memory index during testing in the Y-maze. P < 0.05 among four groups, one-way ANOVA. *P < 0.05, compared with young control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. #P < 0.05, one-way ANOVA with Newman-Keuls multiple comparisons test. N = 12 for young groups and N = 10 for aged groups.
During Y-maze training, all mice in the four groups spent about the same amount of time and had a similar number of entries to the two open arms (Figure 2(e) and (g)). During testing, both control and FK866-treated young mice spent more time in the novel arm and had an increased number of entries to the novel arm than the two old arms (Figure 2(f) and (h)). The aged control mice showed no preference to the novel arm, they spent similar time and had an identical entry number to the three arms. The FK866-treated aged mice showed their preference to the novel arm indicated by the increasing time and entry number to the novel arm (Figure 2(f) and (h)). The aged control mice had a significantly lower recognition memory index when compared with the young control mice. The administration of FK866 significantly increased the recognition memory index of the aged mice but did not affect the young mice (Figure 2(i)).
In the fear conditioning experiment, mice in all groups showed a significantly higher freezing percentage when returned to the box receiving foot shock, indicative of the contextual memory. However, the freezing percentage of aged control mice was 24.5% lower than the young control mice. FK866 increased the freezing percentage of aged mice by 67.2% but did not affect the young mice (Figure 3(a)). In the test of cue memory, the tone significantly increased the freezing percentage of control and FK866-treated young mice, but did not increase the freezing percentage of the aged mice (Figure 3(b)). The aged control mice had 34.1% lower freezing percentage than the young control mice. The treatment of FK866 did not increase the freezing percentage of aged mice (Figure 3(b)). Thus, FK866 treatment improved the contextual memory of aged mice but did not affect their cue memory. For the young mice, FK866 did not affect the contextual and cue memory.
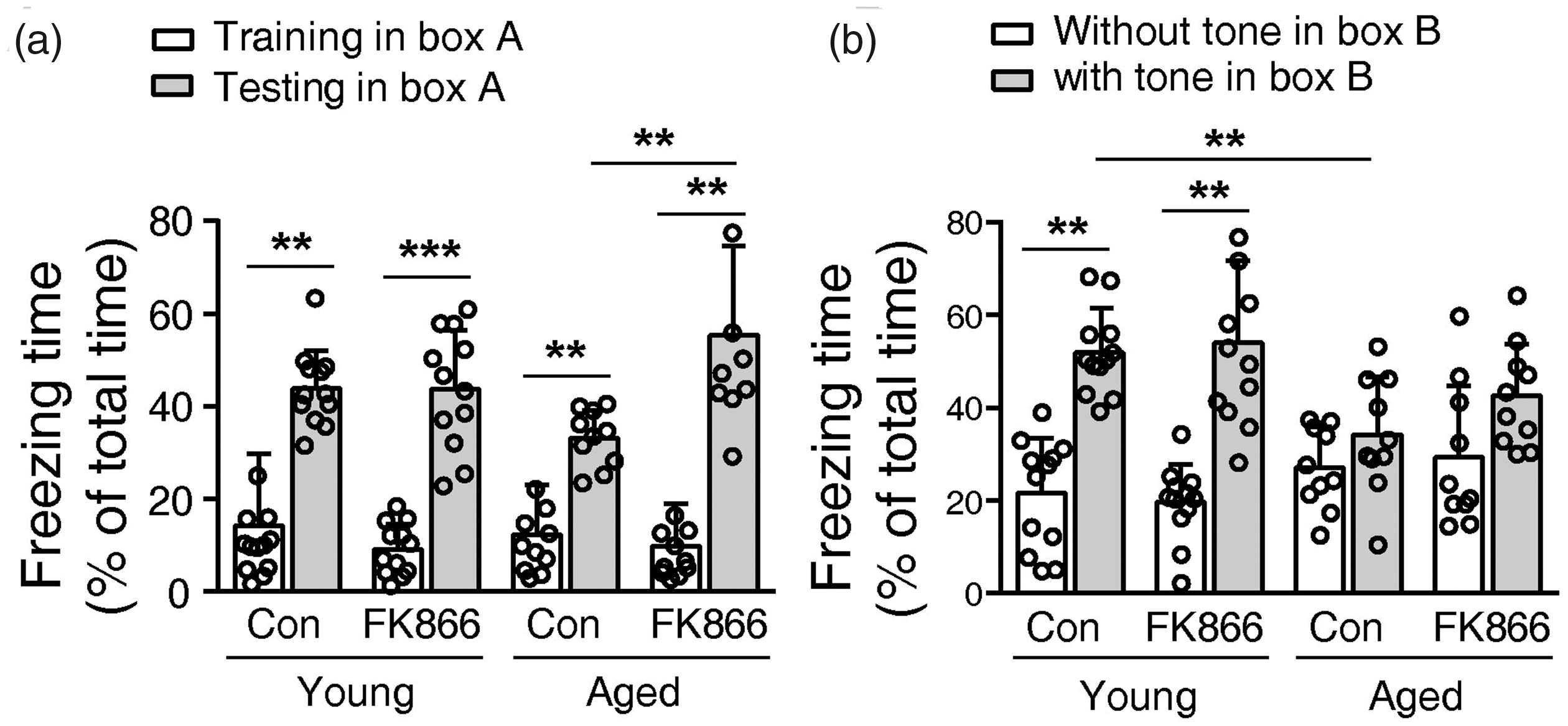
Effects of FK866 (0.5 mg/kg ip qod for six weeks) on short-term fear memory of the mice. (a) The percentage of freezing time of mice during training in boxA and testing in the same box. BoxA is the context in which the mouse received a shock. (b) The percentage of freeze time of mice tested in the boxB with or without the tone during the foot shock. N = 12 for young groups and N = 10 for aged groups. Con means control. **P < 0.01, ***P < 0.01, one-way ANOVA with Newman-Keuls multiple comparisons test.
FK866 at 0.5 mg/kg (qod, ip.) significantly inhibited glial-responses of aged mice
Glial-response indicated by the activation of astrocytes and microglia is common in brain aging. Using immunofluorescence staining, we found that the intensity of both Iba1 and GFAP increased obviously in the hippocampus of the aged control mouse brain compared with the young mouse brain. FK866 had no obvious effect on the young mouse brain but appeared to decrease the intensity of Iba1 and GFAP in the aged mouse brain (Figure 4(a) and (c)).
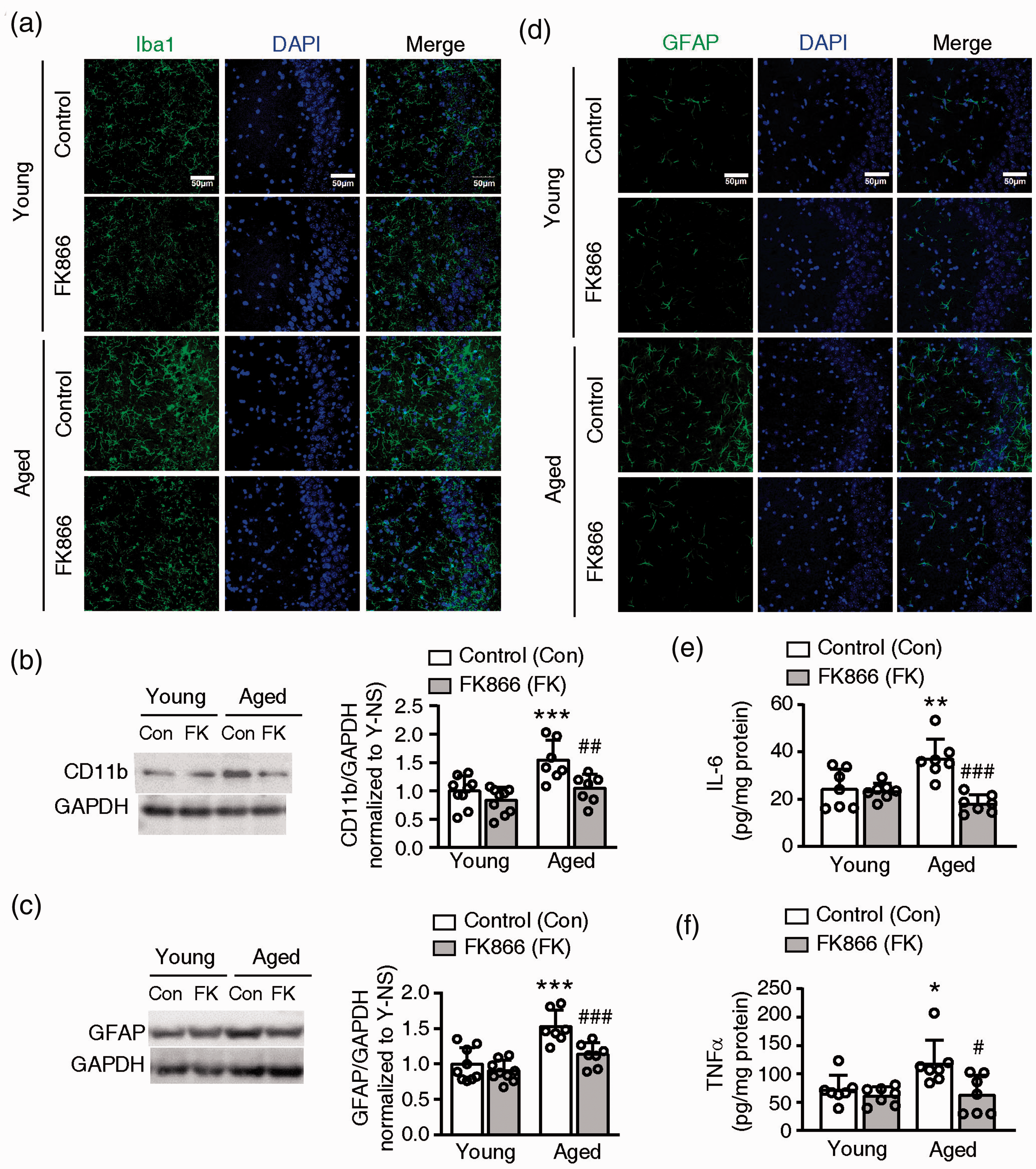
Effects of FK866 (0.5 mg/kg ip qod for six weeks) on chronic neuroinflammation in the mouse brain. (a) Representative images of Iba1, a microglia marker, fluorescent staining in the hippocampus CA3 of mouse brains. Three mice for each group were stained. (b) Western blotting analysis of CD11b, a microglia marker, in mouse brains. Left panel: the representative western blotting bands of CD11b and GAPDH. Right panel: the statistical analysis of CD11b expression level. N = 9 for young groups and N = 7 for aged groups. (c) Representative images of GFAP (green), an astrocyte marker, fluorescent staining in the hippocampus CA3 of mouse brains. Three mice for each group were stained. (d) Western blotting analysis of GFAP in the mouse brain. Left panel: the representative western blotting bands of GFAP and GAPDH. Right panel: the statistical analysis of GFAP expression level. N = 9 for young groups and N = 7 for aged groups. (E and F) The level of IL-6 (e) and TNFα (F) in the mouse brain. N = 7. *P < 0.05, **P < 0.01, ***P < 0.001, compared with control (Con) young mice, #P < 0.05, ##P < 0.01, ###P < 0.001, compared with aged control mice, one-way ANOVA with Newman-Keuls multiple comparisons test.
The western blotting analysis showed CD11b and GFAP expression significantly increased in the aged control mouse than the young control mouse (Figure 4(b) and (d)). The administration of FK866 did not change CD11b and GFAP expression in the young mouse but significantly decreased their expression in the aged mouse (Figure 4(b) and (d)).
The level of TNFα and IL-6 significantly increased in the aged control mouse comparison to the young control mouse. FK866 significantly decreased the level of TNFα and IL-6 in the aged mouse but had no effects on the young mouse (Figure 4(e) and (f)).
FK866 at 0.5 mg/kg (qod, ip) enhance autophagy and decreased the protein aggregation in aged mouse brain
Using western blotting, we found that the level of LC3-II, an indicator of autophagy, significantly decreased in the aged control mouse compared to the young control mouse. FK866 significantly increased the LC3-II level in the aged mouse, but it did not affect the LC3-II level in the young mouse (Figure 5(a) and (b)).
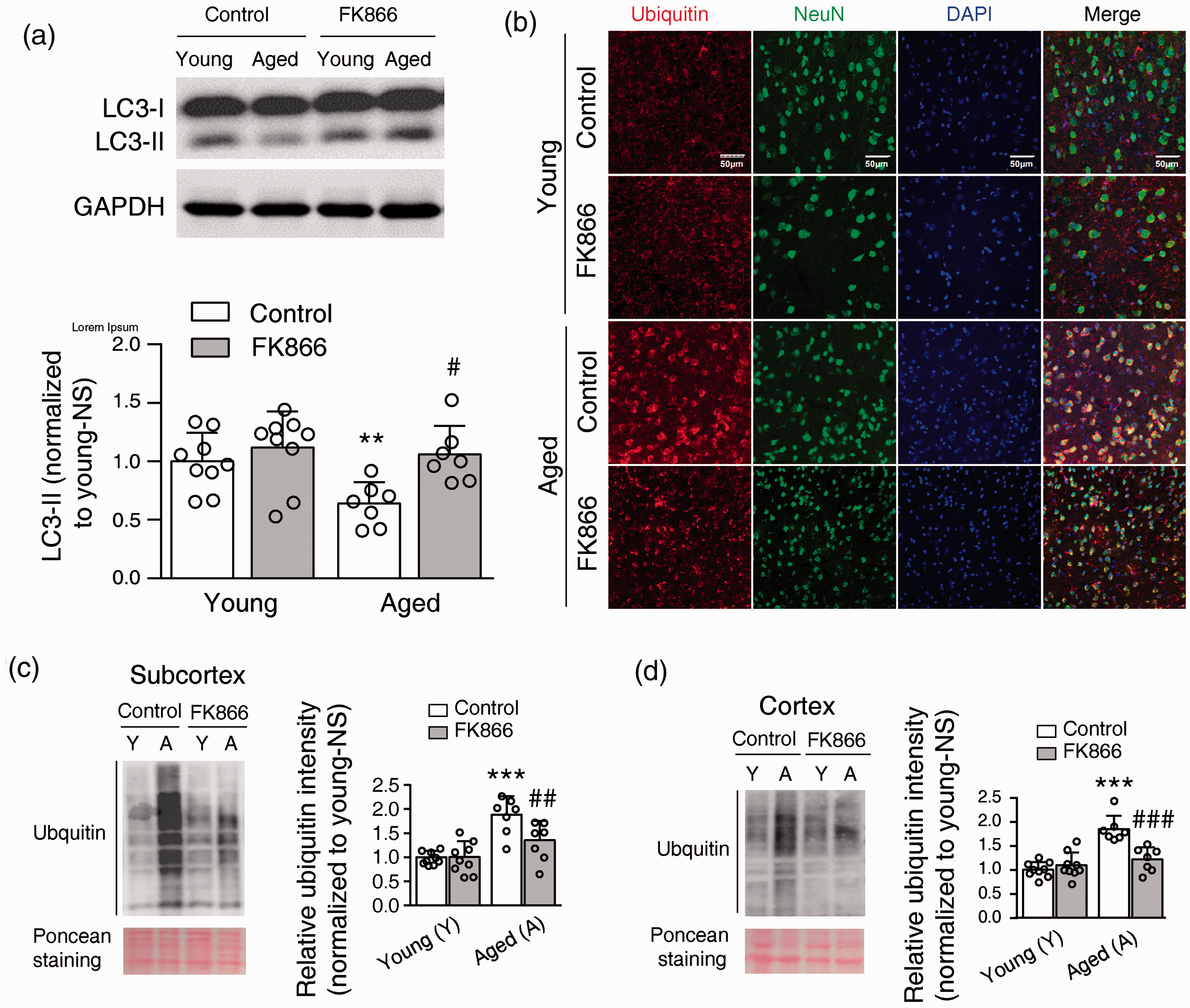
Effects of FK866 (0.5 mg/kg ip qod for six weeks) on the level of aggregates in the mouse brain. (a) FK866 enhanced autophagy in aged mouse brain. Upper panel, a representative image of LC3 and GAPDH Western blot. Lower panel, statistical analysis of LC3-II expression. N = 9 for the young groups and N = 7 for the aged groups. **P < 0.01, compared with young control mice, #P < 0.05, compared with aged control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. (b) Representative images of ubiquitin (red) and NeuN, a marker of the neuron, double fluorescent staining in the cortex of mouse brains. Three mice for each group were stained. (c and d) Western blotting analysis of ubiquitin in the insoluble part of subcortex (c) and cortex (d) of the mouse brain. Left panel: the representative western blotting bands of ubiquitin and the ponceau staining as a loading control. Right panel: the statistical analysis of ubiquitin level in the insoluble part of the mouse brain. ***P < 0.001, compared with young control mice, ##P < 0.01, ###P < 0.001, compared with aged control mice, one-way ANOVA with Newman-Keuls multiple comparisons test. N = 9 for young groups and N = 7 for aged groups.
Ubiquitin is a marker for protein aggregation. 3 Using immunofluorescence staining, we found that ubiquitin is widely distributed in the neurons. The fluorescence intensity of ubiquitin appears to higher in the aged control mouse than in the young control mouse. FK866 had no obvious effect on the distribution and expression of ubiquitin in the young mouse but appeared to decrease the intensity of ubiquitin staining in the aged mouse (Figure 5(b)). Using western blotting analysis, we assessed the ubiquitin level in the insoluble fraction to determine the protein aggregation. The ubiquitin level was much higher in the aged control mice compared to the young control mice, both in the subcortex and cortex of the mouse brain. The treatment of FK866 significantly reduced ubiquitin level in the aged mouse but no effect on the young mouse (Figure 5(c) and (d)). As such, FK866 can enhance autophagy and decrease protein aggregation in the aged mouse brain.
Effect of FK866 at 0.5 mg/kg (qod, ip) on the level of total NAD, NAM and NMN in mouse brain
To determine how does FK866 affect the NAD savaging synthesis pathway, we measured the NAM, NMN, and NAD level in the mouse brain at 24 hr after the last injection. The total NAD level in the aged control mouse brain was significantly lower in comparison to the young control mouse brain (Figure 6(a)). The NMN level was slightly higher in the aged control mouse brain in comparison to the young control mouse brain (Figure 6(b)). FK866 treatment did not change the total NAD level and NMN level in the mouse brain, either young or aged mouse (Figure 6(a) and (b)).
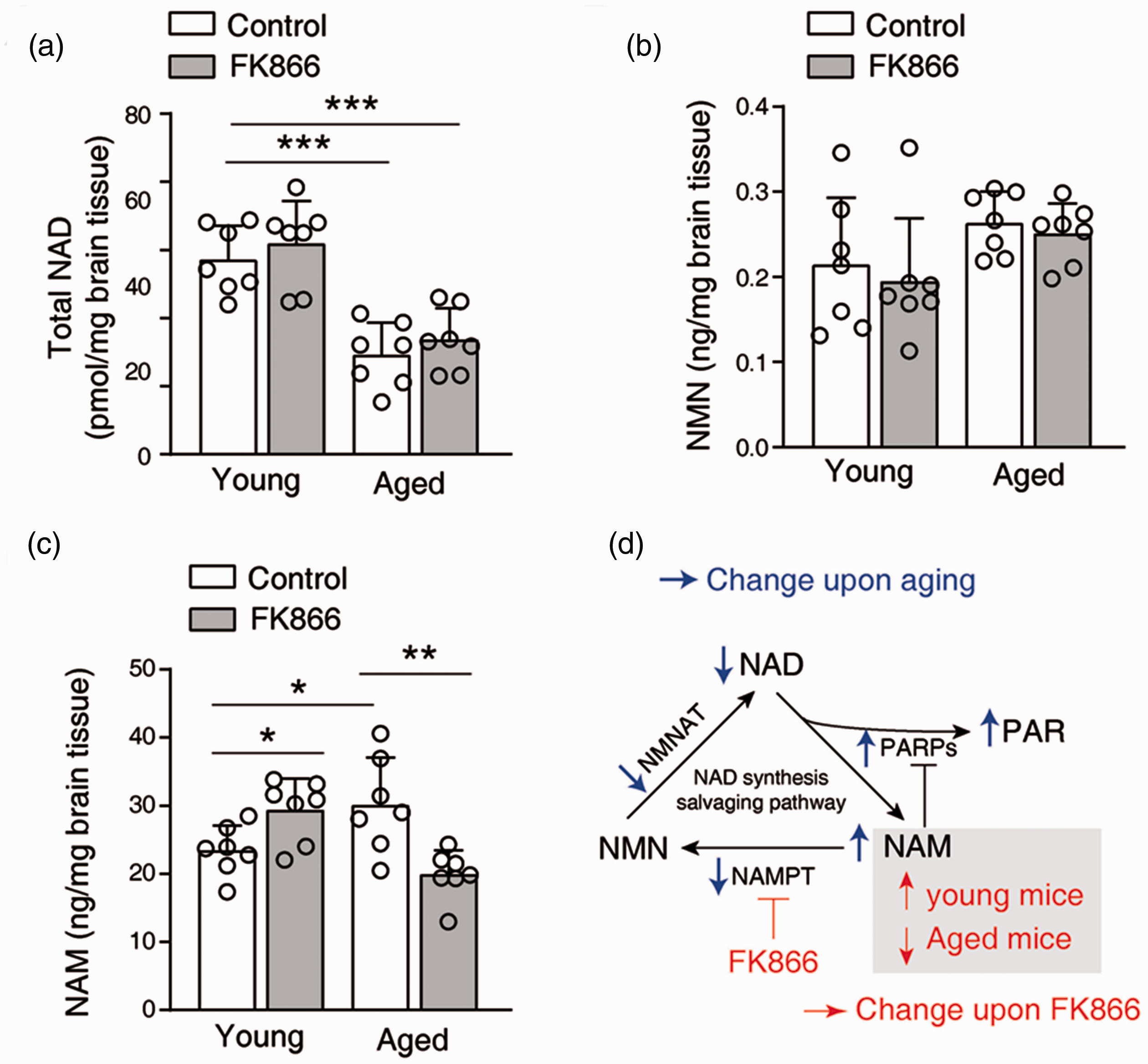
Effects of FK866 on the salvaging NAD synthesis pathway. (a–c) The level of total NAD (a), NMN (b), and NAM (c) in the sub-cortex of mice brain at 24 hr after the last injection of FK866. *P < 0.05, **P < 0.01, ***P < 0.001, one-way ANOVA with Newman-Keuls multiple comparisons test. (d) A diagram illustrates the NAD synthesis salvaging pathway, and the changes upon aging (blue arrows) and upon the administration of FK866 (red arrows).
The NAM level was significantly higher in the aged control mouse brain in comparison to the young control mouse brain. Surprisingly, FK866 increased the NAM level in the young mouse brain but decreased the NAM level in the aged mouse brain (Figure 6(c)).
Effect of FK866 on neuronal death
The Nissel staining showed that the administration of FK866 did not cause a visible neuronal morphological change in the young and aged mouse (Figure S6). In the primary cultured rat neurons, FK866 at >10−8 M for 24 hr significantly decreased the cell viability (Figure S7(a)), and at >10−7 M increased lactate dehydrogenase (LDH) release indicating the neuronal death (Figure S7(b)). Thus, only a high concentration of FK866 would induce neuronal death.
Discussion
Here we show that a long-term treatment of FK866, at low and non-toxic concentration, significantly ameliorates the cognitive impairment and increases the locomotor activity of aged mice. Accordingly, FK866 enhances autophagy, decreases cellular protein aggregation, and inhibits glial-responses in the brain of aged mice.
Firstly, FK866 improves the cognition of the aged mice thanks to decreasing protein aggregation via enhancing autophagy. As illustrated in Figure 1(a), toxic and misfolded proteins accumulate due to the decline of energy metabolism, the disruption of endoplasmic reticulum homeostasis, and the increase of oxygen free radicals in the aged brain.7,35 The proteasomal activity responsible for the degradation of toxic and misfolded proteins also declines upon aging.7,36 The toxic and misfolded proteins can thus aggregate and form aggresome, which can be cleared in the autophagy-lysosome pathway. 35 Since autophagy activity also decreases upon aging, aggresome may accumulate in the aged brain.37,38 The formation of protein aggregate would injury neurons and lead to sustained neuroinflammation in the aged brain.39,40 The autophagy enhancers can increase the clearance of aggresome and alleviate cognitive impairment in aged brain and neurodegenerative diseases. 6 In the present study, we show that FK866 enhanced autophagy and decreased protein aggregation in the aged mouse brain. Thus, FK866 can directly protect neurons, and FK866 can indirectly inhibit the glial-response.
Secondly, FK866 improves the aged mice’ cognition thanks to the direct and indirect inhibition of glial-mediated neuroinflammation. Chronic neuroinflammation upon aging can be a result of or a cause for the activation of microglia and astrocyte.1,2 Indeed, it has been reported that direct inhibition of neuroinflammation by FK866 can provide a neuroprotective effect.29,30 We have previously shown that NAMPT functions as a cytokine secreted by microglia. FK866 can inhibit oxygen-glucose-deprivation induced microglia activation and inhibit the release of NAMPT. 31 Recently, Yang et al. showed that FK866 inhibits LPS-induced inflammatory responses in monocytes. 26 Thus, besides indirectly inhibiting neuroinflammation by reducing protein aggregation, FK866 may also directly inhibit aging-induced microglia activation.
The results here appear counter-intuitive since previous studies have shown a strong correlation between aging and impaired NAD metabolism. For example, conditional knockout of Nampt gene can cause neuronal injuries and cognitive impairment.18,41 These can be explained by the compartmentalized distribution of NAMPT and NAD. NAMPT is distributed in the cytoplasm, nucleus, and mitochondria. The cytoplasm- and nucleus-located NAMPT is likely more sensitive to FK866 than the mitochondria-located NAMPT. 42 The in vitro IC50 of FK866 on NAMPT inhibition is several nanomolar. But we showed that >10−8 M FK866 can reduce the activity of neuronal mitochondria as characterized with decreased cell viability; >10−7 M FK866 can cause neuronal death. The Nampt gene knockout eliminates NAMPT from the entire cell, regardless of its subcellular location. Together, the inhibitory effect of FK866 is highly dose-dependent and the cells may only be irreparably damaged when the mitochondrial NAMPT is inhibited, which is phenotypically similar to gene knockout.
Another interesting observation is that FK866 has almost no effect on the young mice. The reasons can be due to the difference between young and aged brains. First, NAMPT is expressed differently in the microglia of young and aged mice. NAMPT is absent in the microglia in the young mouse but is highly expressed in the microglia of aged mice. 11 NAMPT in microglia is responsible for microglia activation and can be released as a cytokine. 31 Thus, FK866 may only inhibit microglial response in the aged brain. Second, as NAD-consuming enzymes, PARPs are over-activated in the aged mice. As illustrated in Figure 6(d), the over-activation of PARPs would decrease NAD and increase NAM. 43 Consistently, we observed that NAM increased and NAD decreased in the aged mice. Under the low dose, we found that FK866 did not affect the NMN and NAD level. FK866 increased NAM in the young mouse, which is expected. However, FK866 decreased NAM in the aged mouse. NAM is not only a NAD precursor but also a PARPs inhibitor. 44 In the aged mouse, we reason that NAM level can increase at the beginning of FK866 administration during the one-month treatment. With the increase of NAM, the over-activation of PARPs in aged brain can be inhibited by NAM, which leads to a negative feedback. With the inhibition of PARPs, NAM production gradually decreased. The inhibition of PARPs can also be beneficial for aged brains. 12
Clinical studies have shown that FK866 is safe as an anti-cancer drug. 45 In the present study, the dose of FK866 (0.5 mg/kg for every other day) is two orders of magnitude lower than what is used in anti-tumor studies. For example, to kill cancer cells in vivo, FK866 at 20-40 mg/kg/day has been used.46–48 To kill the cancer cells in vitro, FK866 higher than 10−8 M is required.20,49 In the aged mice, FK866 caused the elevation of serum glucose under the dose (1 mg/kg, biw) in the pilot experiment. This side effect could be due to the inhibition of NAMPT in islet β cells. 50
In summary, we have shown that FK866, an anti-cancer drug, can be repurposed for the treatment of age-related diseases. FK866 treatment at low dose can enhance autophagy, increase the clearance of aggresome, and inhibit glia-mediated chronic neuroinflammation, which all help the alleviation of cognitive impairment of the aged mice. As the accumulation of protein aggregation and chronic neuroinflammation are associated with aging and neurodegenerative diseases, we foresee that the low-dose FK866 can be a novel treatment for such age-related diseases.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211006291 - Supplemental material for Nicotinamide phosphoribosyltransferase inhibitor ameliorates mouse aging-induced cognitive impairment
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211006291 for Nicotinamide phosphoribosyltransferase inhibitor ameliorates mouse aging-induced cognitive impairment by Min Zeng, Tao-Feng Wei, Cong Chen, Chen Shen, Tong-Yao Gao, Xian Xie, Ming Wu, Yun-Bi Lu and Wei-Ping Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X211006291 - Supplemental material for Nicotinamide phosphoribosyltransferase inhibitor ameliorates mouse aging-induced cognitive impairment
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X211006291 for Nicotinamide phosphoribosyltransferase inhibitor ameliorates mouse aging-induced cognitive impairment by Min Zeng, Tao-Feng Wei, Cong Chen, Chen Shen, Tong-Yao Gao, Xian Xie, Ming Wu, Yun-Bi Lu and Wei-Ping Zhang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Key R&D Program of China (2018YFA0507700), the National Natural Science Foundation of China (81573400 and 81971304) and the Zhejiang Provincial Natural Science Foundation of China (LZ20H010001, LY18H170001).
Acknowledgement
We thank Prof. C. Tang for critical reading of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
MZ performed the behavioral experiments and fluorescent imaging in the formal experiments. TFW performed the Western blotting experiments in the formal experiments. CC assisted the Western blotting experiments and NAD, NAM, NMN detection in the formal experiments, and fluorescent imaging in the pilot experiments. CS assisted the behavioral experiments. XX performed the behavioral experiments in the pilot experiments. TYG performed that TNFα and IL-determination, MW, YBL and WPZ contributed the design and data analyze. YBL and WPZ wrote the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
