Abstract
Age-related memory loss shares similar risk factors as cardiometabolic diseases including elevated serum triglycerides (TGs) and low-density lipoprotein cholesterol (LDL-C) and reduced high-density lipoprotein cholesterol (HDL-C). The mechanisms linking these aberrant blood lipids to memory loss are not completely understood but may be partially mediated by reduced integrity of the hippocampus (HC), the primary brain structure for encoding and recalling memories. In this study, we tested the hypothesis that blood lipid markers are independently associated with memory performance and HC viscoelasticity—a noninvasive measure of brain tissue microstructural integrity assessed by high-resolution magnetic resonance elastography (MRE). Twenty-six individuals across the adult lifespan were recruited (14 M/12 F; mean age: 42 ± 15 y; age range: 22–78 y) and serum lipid profiles were related to episodic memory and HC viscoelasticity. All subjects were generally healthy without clinically abnormal blood lipids or memory loss. Episodic memory was negatively associated with the TG/HDL-C ratio. HC viscoelasticity was negatively associated with serum TGs and the TG/HDL-C ratio, independent of age and in the absence of associations with HC volume. These data, although cross-sectional, suggest that subtle differences in blood lipid profiles in healthy adults may contribute to a reduction in memory function and HC tissue integrity.
Introduction
Advancing age is the primary risk factor for Alzheimer’s disease (AD), which is the most common form of dementia and is characterized by neurodegeneration and irreversible memory loss. As the number of older adults in developed societies continues to increase, the number of AD cases is projected to triple by 2050 if preventive measures are not undertaken. 1 Age-related cognitive decline shares many of the same modifiable risk factors as cardiometabolic diseases, especially when they emerge earlier in life, prior to the development of clinically relevant memory loss. 2 In this regard, elevated blood triglycerides (TGs) and low-density lipoproteins (LDL-C) and reduced high-density lipoprotein cholesterol (HDL-C) are established clinical risk factors and independent predictors of ischemic heart disease3–9 and have been associated with cognitive impairment and late-life risk for AD.10–14 Despite their association with cognitive aging, the underlying mechanisms linking aberrant lipid profiles to neurodegeneration and memory loss in humans are not fully understood.
Insight into the mechanisms of cognitive aging can be gained by studying the viscoelastic mechanical properties of the hippocampus (HC), a brain structure essential for memory encoding and performance. 15 In this regard, magnetic resonance elastography (MRE) is a novel and noninvasive imaging modality for measuring brain viscoelastic mechanical properties in humans 16 , 17 and reflects the distribution and organization of neurons, axons, and glial cells.18–20 Indeed, alterations in neuronal microstructure via measures of viscoelastic properties have been demonstrated in neurodegenerative diseases including AD,21–23 Parkinson’s disease, 24 , 25 multiple sclerosis, 26 , 27 and amyotrophic lateral sclerosis, 28 as well under conditions of increased inflammation. 18 , 29 Changes to brain tissue viscoelastic properties assessed using MRE also occur during normal healthy aging, and properties of the HC specifically have been strongly associated with memory performance30–32 such that they are potentially more predictive of episodic memory than traditional clinical measures of HC volumes. 33 As such, MRE-based assessments of HC tissue integrity in healthy adults could provide important insight into pre-clinical associations between blood lipids and age-related memory loss that precede HC atrophy. Therefore, in this study we determined if blood lipids are independently associated with HC viscoelastic properties and memory function in healthy individuals across the adult lifespan. We hypothesized that elevated serum TGs, elevated LDL-C, and reduced HDL-C would be independently associated with lower memory performance. We also hypothesized that aberrant serum lipids would be associated with reduced HC integrity—defined as either a higher HC damping ratio (ξ) or lower HC shear stiffness (µ)—and that these associations would occur independent of age, sex, and other cardiometabolic risk factors. We further predicted that these associations would occur in the absence of an association with HC volume.
Materials and methods
This study was reviewed and approved by University of Delaware’s (UD) Institutional Review Board and all experimental procedures and protocols conformed to the standards outlined in the Declaration of Helsinki. The purpose of the study, along with the possible benefits and risks, was explained to all subjects and their written informed consent was obtained prior to participation. All measurements were made in the Neurovascular Aging Laboratory and the Center for Biomedical and Brain Imaging at UD between August 2018 and December 2019.
Study participants and recruitment
Twenty-six healthy adult men and women (14 M/12 F) between the ages of 22 and 78 years were recruited for this study from Newark, DE, and surrounding areas. All participants were non-smokers and were apparently free from chronic clinical diseases and major psychological and neurological disorders as assessed by medical history and baseline blood chemistries. Participants were excluded if they exhibited clinically abnormal blood chemistries such as a GFR <60 ml/min/1.73 m2 and values that were ±2.5 × the upper or lower limit of the normal range. Participants taking any statin medications and medications acting on the central nervous system (CNS) were excluded as well. Participants were also excluded if they were unable or unwilling to participate in an MRI scan (e.g. metal implants, pacemakers, claustrophobia). Women of childbearing age were tested for possible pregnancy (hCG Pregnancy Test Kit, McKesson Consult) before entering the MRI. Subjects refrained from all over-the-counter medications for 48 h prior to the experimental visits and refrained from alcohol and vigorous exercise 24 h prior to experimental visits. Height and weight were measured during the initial screening. BMI was calculated as body mass in kilograms over square of height in meters. Physical activity was obtained from the Modifiable Activity Questionnaire and was measured as metabolic equivalents (METs) in hours per week during leisure time activity. 34
Blood lipid analysis
A standard blood lipid panel (Quest Diagnostics, USA) was performed at the time of initial screening following a 12 h overnight fast. Measured lipid markers included serum TGs, total blood cholesterol (TC), and HDL-C. LDL-C concentrations were calculated from the Martin–Hopkins equation. Generally, the Friedewald calculation (LDL-C = TC − HDL-C − TG/5) is commonly used to obtain blood LDL-C concentrations in which TG/5 is used to represent very low-density lipoprotein cholesterol (VLDL-C). 35 However, Martin et al. derived a more accurate factor that calculates VLDL-C from TGs, ranging from 3.1 to 11.9 depending on individual TG and non-HDL-C concentrations. 36 We also calculated an index of atherogenic risk that includes the TG/HDL-C ratio. 37 , 38
Memory function
Episodic memory was assessed using the California Verbal Learning Test (CVLT-III). 39 The CVLT-III consists of the examiner reading a pre-recorded list of 16 words (List A) with four items belonging to each of four categories (e.g. vegetables, animals, modes of transportation, and furniture). Participants are asked to recall as many items as possible, in any order, over five trials. Another list of 16 words (List B) is administered after List A for one trial as an interference. After List B, participants are asked to freely recall words in any order from List A and then categorize List A into their four semantic categories (cued recall), without the examiner re-reading the list. Following a 20-min delay, free and cued recall of words from List A is assessed followed by a Yes/No recognition from List A. For this study, raw scores from the delayed recall portion of the test were used, as robust associations between HC structural changes and delayed recall memory exist both in healthy aging as well as in AD patients.40–42
Magnetic resonance elastography
All participants completed a 50-min scanning session at the UD Center for Biomedical and Brain Imaging on a 3 T Siemens Magnetom Prisma whole-body MRI scanner with a 64-channel head coil (Siemens, Erlangen, Germany). MRE images were acquired using a 3 D multiband, multishot spiral sequence at 1.25 mm3 isotropic resolution (240 × 240 mm2 field of view (FOV), 96 slices, repetition time (TR)/echo time (TE) = 3360/70 ms) in order to capture high-resolution displacement data with an acquisition time of 10 min and 45 s. 43 The sequence was synchronized with an external 50 Hz vibration frequency delivered to the head by a pneumatic actuator system and soft pillow driver (Resoundant Inc., Rochester, MN, USA) that has already been reported to be acceptable over a wide age range 30 as depicted in Figure 1. Motion from the actuator was sampled with flow-compensated motion encoding gradients to generate full vector displacement fields, following iterative image reconstruction, as described previously by our collaborators. 44
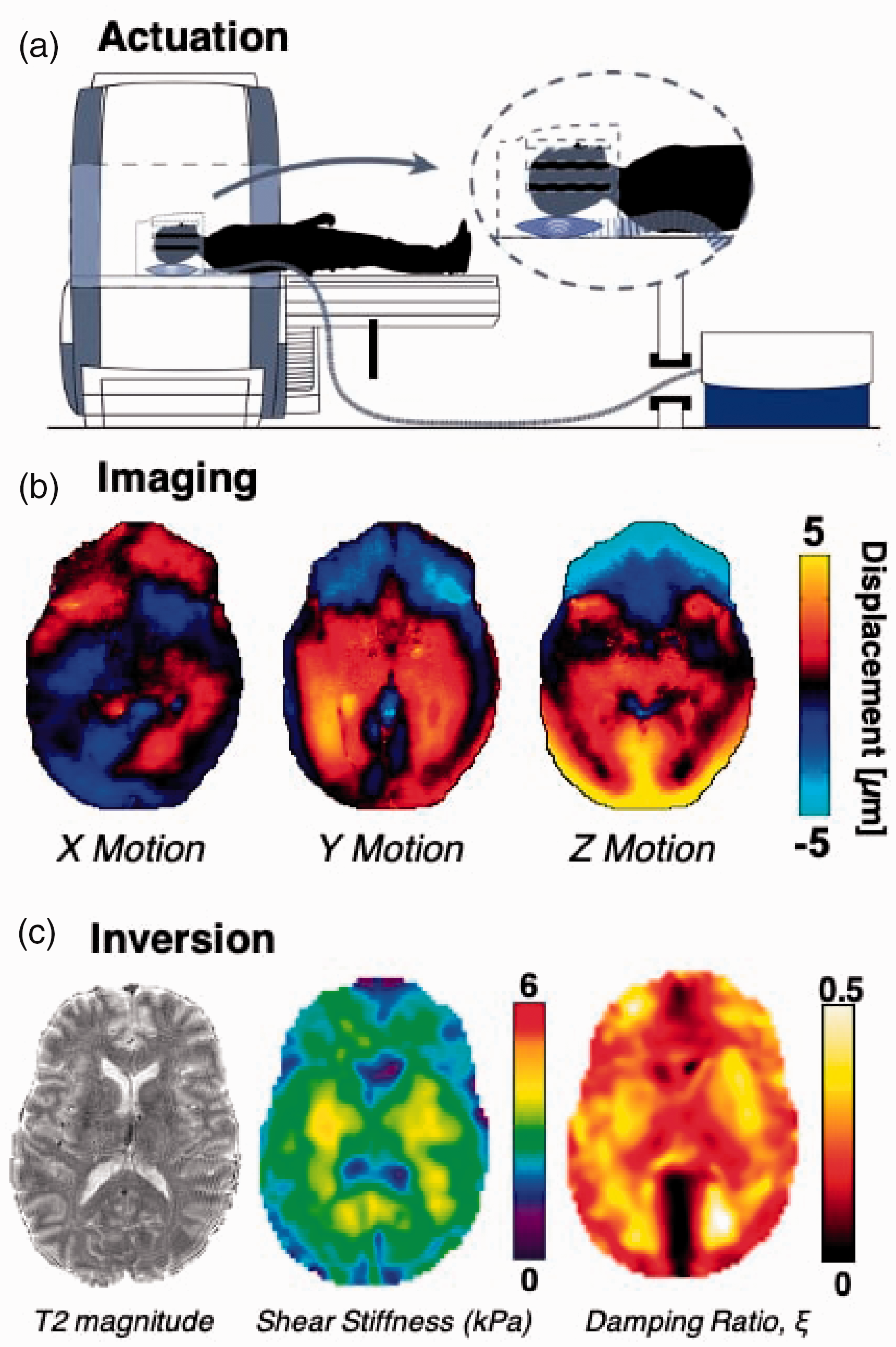
MRE protocol. (a) Shear waves generated via external vibration at 50 Hz from an active driver to a head pillow-driver; (b) shear waves imaged as tissue displacement in three directions; (c) nonlinear inversion algorithm to determine shear stiffness (µ) and damping ratio (ξ).
Bilateral HC masks were obtained from a T1-weighted magnetization-prepared rapid gradient echo structural scan at 0.9 mm3 resolution (240 × 240 mm2 FOV, 192 slices, TR/TE = 2300/2.32 ms) and segmented using FreeSurfer 6.0. They were then registered from the anatomical T1 image space to MRE space using the FLIRT tool in FSL (FMRIB Software Library v. 6.0.0). 45 Bilateral HC volumetric measures were determined from Freesurfer outputs and were normalized using estimated total intracranial volume to correct for participant head size. 46
MRE data quality was assessed using octahedral shear-strain based signal-to-noise ratio (OSS-SNR) and a cut-off of OSS-SNR >3 was considered as acceptable data for stable inversion.
47
A nonlinear inversion algorithm, combined with soft prior regularization (SPR), was used to estimate the viscoelastic complex shear modulus (G = G′ + G*) of the HC.
48
,
49
SPR employs prior anatomical information to reduce regional variability and promote mechanical homogeneity in regions of interest;
50
SPR weighting of 10−12 was applied in this study. Maps of the complex shear modulus were used to calculate viscoelastic parameters, shear stiffness (µ = 2
Statistical analyses
Normality of residuals of each variable was assessed using the Shapiro–Wilk test. 56 Bivariate correlation analyses (Pearson correlations) were used to identify whether blood lipid markers and potential covariates were associated with any of the dependent variables. The Spearman rank correlation test was performed on variables that were not normally distributed (BMI, TG, and CVLT Delayed Recall scores). Because age is a strong predictor of both HC integrity and memory, 57 it was included in the bivariate correlation analysis. Sex, habitual physical activity, and BMI are also associated with brain viscoelasticity 31 , 53 , 58 and blood lipid profiles; 59 , 60 therefore, these variables were included as potential covariates. Multiple linear regressions were performed to identify the association between each covariate and independent variable of interest from the bivariate correlation analyses and the primary dependent variables. Any variable that was not normally distributed was log transformed prior to being entered into the regression models. Inferences from simple linear regressions were age-adjusted data based on residuals of the independent and dependent variables; however, for simplicity, figures are presented with raw data. Statistical significance was set at p < 0.05. All analyses were performed with RStudio v.1.2.1355 (RStudio Inc., Boston, MA, USA) and GraphPad Prism 8.0 (GraphPad Software Inc., San Diego, CA, USA).
Results
Participant characteristics
Twenty-six adult men and women between the ages of 22 and 78 years were included in this study. Basic characteristics and clinical laboratory values are presented in Table 1. Participants were generally healthy and free of overt cardiovascular diseases or other chronic conditions and the group mean laboratory values were within normal clinical ranges.
Participant characteristics.
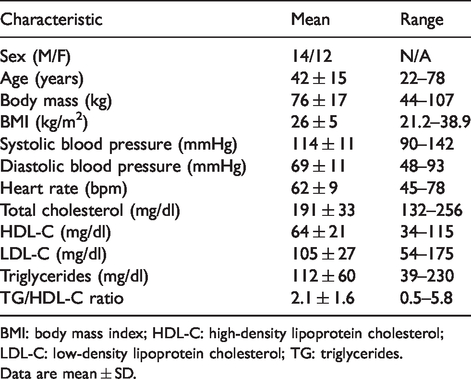
BMI: body mass index; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglycerides.
Data are mean ± SD.
Bivariate correlation between serum lipids and hippocampal structure and function
Bivariate correlations of serum lipid markers and their relation to memory and HC viscoelastic properties are presented in Table 2. HDL-C was significantly associated with delayed recall memory (rs = 0.48, p = 0.01) and HC stiffness (r = 0.43, p = 0.03), while serum TGs were strongly associated with HC damping ratio (rs = 0.63, p = 0.0005). TC was also significantly associated with HC stiffness (r = 0.42, p = 0.03); however, we observed no significant relation between LDL-C and HC stiffness (r = 0.25, p = 0.22), suggesting that this association was solely due to the influence of HDL-C. Hence, TC was not included in subsequent multiple linear regressions to avoid overfitting the models. Although not statistically significant, we also observed negative associations between delayed recall memory with both TGs (rs = −0.37, p = 0.06) and LDL-C (rs = −0.36, p = 0.08) as well as a negative association between HDL-C and HC damping ratio (r = −0.34, p = 0.09). Collectively, these results support an association between serum lipid profiles and HC function (i.e., memory) and integrity.
Bivariate correlation between dependent variables and potential covariates.
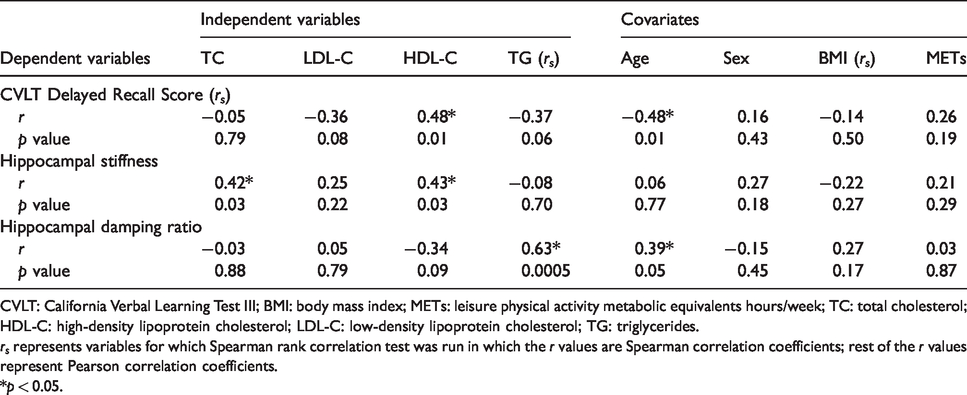
CVLT: California Verbal Learning Test III; BMI: body mass index; METs: leisure physical activity metabolic equivalents hours/week; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglycerides.
rs represents variables for which Spearman rank correlation test was run in which the r values are Spearman correlation coefficients; rest of the r values represent Pearson correlation coefficients.
*p < 0.05.
Because serum lipids, HC structure, and memory are potentially influenced by age and other cardiometabolic risk factors or lifestyle behaviors, we next determined whether any of our independent or dependent variables were influenced by other covariates including age, sex, BMI, and physical activity (Table 2). Age was significantly associated with delayed recall memory (rs = −0.48, p = 0.01) and nearly associated with HC damping ratio (r = 0.39, p = 0.05), therefore we included age as a covariate in all subsequent models. There were no associations between any of the other covariates with our independent or dependent variables.
Independent association between serum lipids and hippocampal structure and function
Next, we performed multiple linear regressions to determine the independent associations between serum lipid markers of interest and HC viscoelastic properties or memory performance while correcting for age and other lipids of interest (Table 3). Because we observed no significant associations between LDL-C and HC viscoelastic properties, we only included TGs and HDL-C in our models. Consistent with our bivariate correlations, we observed a significant association between TGs and HC damping ratio (β = 0.54, p = 0.012) while correcting for age and HDL-C (Table 3). We no longer observed a significant association between HDL-C and HC stiffness after correcting for age and TGs; however, the effect size remained strong and was close to significant (β = 0.43, p = 0.066). Finally, after correcting for blood lipid markers of interest, only age was independently associated with delayed recall memory (β = −0.45, p = 0.02); however, the explained variance (R2 = 0.28) was larger than that explained by age alone (R2 = 0.23), suggesting at least some contribution of blood lipids to the total variance in cognitive function.
Multiple linear regression models.
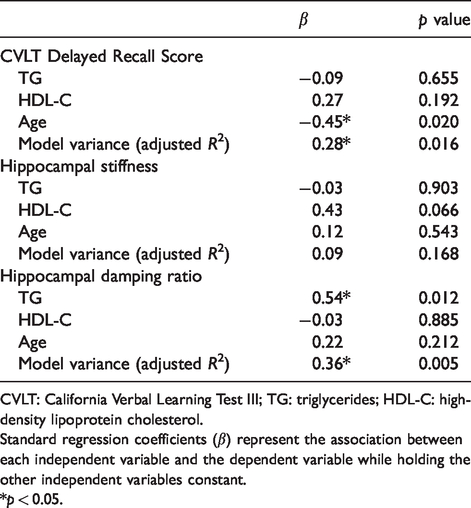
CVLT: California Verbal Learning Test III; TG: triglycerides; HDL-C: high-density lipoprotein cholesterol.
Standard regression coefficients (β) represent the association between each independent variable and the dependent variable while holding the other independent variables constant.
*p < 0.05.
Association between TG/HDL-C ratio and hippocampal structure and function
Because serum TGs and HDL-C emerged as the blood lipid markers most associated with HC viscoelastic properties, we next assessed the association between the TG/HDL-C ratio (an established clinical marker of atherogenic risk) and HC viscoelastic properties and memory while correcting for age. Consistent with our findings in the previous analyses, we observed a significant association between the TG/HDL-C ratio and HC damping ratio (Figure 2(a); r = 0.54, p = 0.005) and a close association with HC stiffness, although not significant (Figure 2(b); r = 0.36, p = 0.07). Interestingly, we also observed a significant association between the TG/HDL-C ratio and delayed recall memory, even when corrected for age (Figure 2(c); r = 0.47, p = 0.02). Representative images depicting a lower HC damping ratio in a participant with lower TG/HDL-C ratio characterized by a low serum TG concentration and high HDL-C concentration (Figure 2(d)) compared to a higher HC damping ratio in a subject with higher TG/HDL-C ratio characterized by a high serum TG concentration and low HDL-C concentration (Figure 2(e)) are also presented.
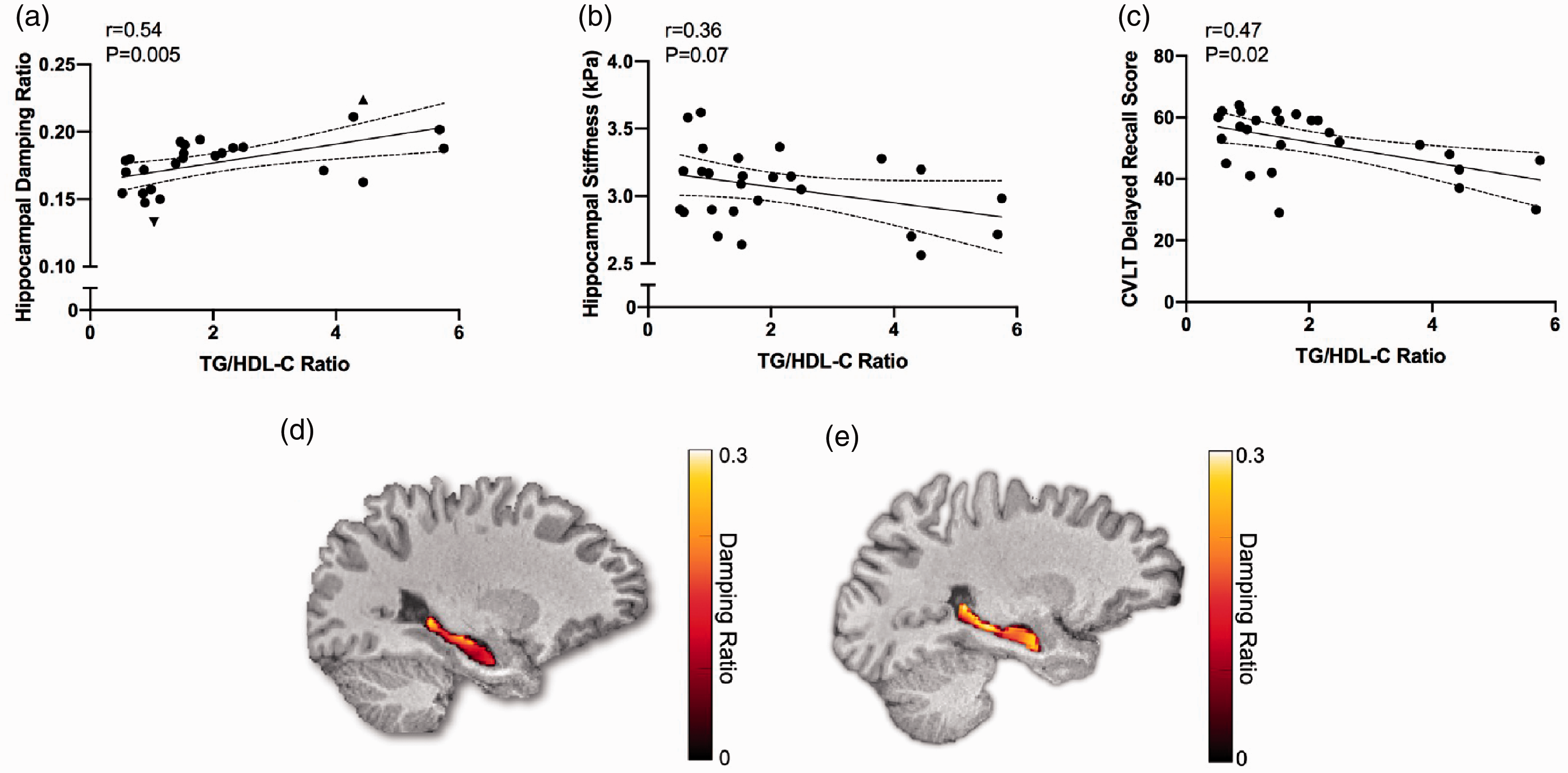
Serum lipids, memory function and brain mechanical properties, and representative images of hippocampal damping ratio. Linear regression analysis between TG/HDL-C ratio and (a) hippocampal damping ratio; closed downward facing triangle symbol represents individual in (d) and closed upward facing triangle symbol represents individual in (e); (b) hippocampal stiffness and (c) CVLT delayed recall score; r and p values are adjusted for age; dashed lines represent 95% confidence interval; statistical significance set at p < 0.05; N = 26 in all panels. (d) image of participant with a low TG/HDL-C ratio (TG = 79 mg/dl; HDL-C = 76 mg/dl; TG/HDL-C = 1.04) and relatively low hippocampal damping ratio (0.13) compared with (e) image of participant with high TG/HDL-C ratio (TG = 222 mg/dl; HDL-C = 50 mg/dl; TG/HDL-C = 4.44) and relatively high hippocampal damping ratio (0.22).
Serum lipids and HC volumes
Finally, to investigate whether serum lipids were associated with standard volumetric measures of the HC, we examined their association with HC volumes. These associations between HC volume and serum TGs (r = 0.01, p = 0.96), HDL-C (r = 0.10, p = 0.61), and TG/HDL-C ratio (r = 0.02, p = 0.92) were not significant after correcting for age (data not shown).
Discussion
The primary finding of this study is that serum concentrations of TGs and the TG/HDL-C ratio are associated with memory performance and HC viscoelastic properties in healthy, cognitively normal adults, independent of age. Importantly, these associations exist in the absence of any relation with HC volumes suggesting a potential mechanistic role of serum lipids in the loss of HC viscoelastic properties in the absence of HC atrophy or clinically relevant memory loss. Collectively, our results may provide important insight into the early events linking cardiometabolic risk factors to late life memory impairment.
A novel aspect of our study is the use of high-resolution MRE to characterize HC structure and integrity. MRE has recently emerged as an important imaging modality for assessing brain tissue viscoelasticity 16 , 17 , 55 , 61 and reflects the microstructural composition and organization of neural structures.18–20 Two important properties can be derived from MRE that provide important insight regarding the structural integrity of the brain. Damping ratio, or the relative viscous-to-elastic behavior of tissue, has been correlated with the density of solid phase connections of soft tissues in animal models 18 , 19 and has been proposed to reflect neuronal microstructural tissue organization in vivo. 20 Accordingly, a lower HC ξ indicates a more elastic solid likely with densely packed neuronal connections, whereas a higher ξ reflects a more viscous tissue, likely with fewer neuronal connections. 20 In contrast, brain tissue stiffness assessed by MRE has been correlated with neuronal density, myelin content, and neural network strength in mouse and bovine animal models, respectively; 20 , 62 , 63 thus, a higher HC µ has been suggested to reflect an increased number of neurons within a stronger cellular matrix. 20 In this regard, normal aging is characterized by an increase in HC ξ and a decrease in HC µ, with a change in either property suggested to represent a loss of brain tissue integrity, 20 which in turn is associated with memory impairment 51 , 54 and potentially the transition from mild cognitive impairment to AD. 64
Our results provide important mechanistic insight into early events linking changes in blood lipids to the loss of HC integrity and function and may help explain why cardiometabolic risk factors during midlife predict memory loss later in life. The most striking observations were positive, independent associations between serum TGs and the TG/HDL-C ratio with HC damping ratio suggesting that even mildly aberrant blood lipids may disrupt existing neuronal connections and organization prior to any clinically detectable HC atrophy. Though not significant after correcting for age and other lipids, we also observed an association between HDL-C and HC stiffness that may suggest an important independent role of HDL-C in maintaining neuronal composition and integrity and is worthy of future investigation.
Although previous studies have demonstrated a similar link between serum lipids and memory function, 11 , 13 , 14 our study is the first to relate these important cardiometabolic risk factors to markers of HC tissue integrity in otherwise healthy humans. Reductions in brain tissue viscosity (loss modulus G”) and elasticity (storage modulus G’) have been observed in mice following acutely induced neuroinflammation 18 and may mimic the effects of chronic neuroinflammation that occurs in the HC of older adults. 65 In this regard, TG transport occurs within VLDL particles which can bind to toll-like receptors on vascular endothelial cells leading to the release of pro-inflammatory cytokines and the development of vascular endothelial dysfunction.66–69 Although VLDL and LDL particles themselves are not known to be present in the CNS, there is some evidence that lower-density lipoproteins may penetrate the blood–brain barrier (BBB) during conditions of increased inflammation. 70 Thus, elevated serum lipids may contribute to a loss of HC integrity either directly, or through a mechanism involving reduced cerebral perfusion, which is an established early clinical feature of AD.71–73
TG-rich lipoproteins (VLDL and LDL) exchange their TGs with cholesterol esters from HDL-C via cholesteryl ester-transfer protein; 68 , 74 thus, higher levels of HDL-C facilitate removal of harmful lipids which may explain why we also observed an association between the TG/HDL ratio and HC damping ratio. A major component of plasma HDL is apolipoprotein-A-I (ApoA-I), which has been shown to be protective against cognitive decline and AD. 14 , 75 , 76 HDL/ApoA-I inhibits expression of pro-inflammatory cytokines by suppressing expression of adhesion molecules such as VCAM-1 and ICAM-1. 77 Accordingly, the mechanistic role of HDL-C and its associated lipoproteins on HC integrity and function needs to be explored further.
Lifestyle factors, such as consumption of an unhealthy diet, contribute to the increase of several cardiometabolic risk factors and may reduce HC integrity through the modulation of blood lipids. In this regard, dietary intake of added sugars (i.e., all caloric sweeteners added to food during processing) have been shown to increase the production of blood TGs and lower concentrations of HDL-C, in large part via the metabolism of fructose by the liver. 78 , 79 Several animal studies have linked a high-sugar (along with high-fat) diet to elevated blood TGs, 80 , 81 increased inflammation,82–84 and reduced concentrations of the neuroprotective protein brain-derived neurotrophic factor. 85 , 86 Thus, it is possible that chronic consumption of sugar-sweetened food and beverages, particularly those containing fructose, may contribute to age-related memory loss by altering HC integrity; however, this hypothesis has not yet been tested in humans. Future randomized controlled trials examining the effects of a high added sugar diet (e.g., containing foods sweetened with high fructose corn syrup), on HC integrity and memory performance in humans are needed before changes to public health policy or nutritional guidelines can be made.
It is important to note that our study was conducted in generally healthy individuals, most of whom had clinically normal serum concentrations of TGs and HDL-C suggesting that even modest, pre-clinical changes in lipid metabolism may be sufficient to evoke changes to HC structure and function. Importantly, we did not observe a significant association between blood lipids and HC volumes, even after correcting for age, suggesting that elevated TGs or reduced HDL-C can induce a loss of HC microstructural integrity at any age in the absence of clinically significant brain tissue atrophy. These observations from our study could have important therapeutic implications for slowing or reversing the progression of age-related memory loss prior to the development of irreversible neuronal tissue loss; however, it is important to note that the present study was cross-sectional and did not investigate a causal link between serum lipids and markers of HC structure or function. Longitudinal studies are needed to determine whether changes in blood lipids mediate the loss of HC integrity or memory performance with aging.
Although the focus of our study was on healthy individuals, our findings have important implications for cognitive aging and dementia risk. Future studies should examine whether these associations are impacted by apolipoprotein-E (ApoE) genotype, which not only plays a significant role in lipid metabolism, but is involved in brain amyloid-beta (Aβ) transport and BBB integrity.87–89 Individuals with one or more copy of the E4 variant of ApoE are at greater risk for AD, while the E2 variant has been shown to be neuroprotective and is associated with increased HC volume and improved cognitive performance. 90 , 91 Thus, it will be important to characterize the influence of ApoE genotype on the association between serum lipids and HC integrity in future studies to further elucidate the mechanisms underlying age-related memory loss.
In summary, this is the first study to explore the relation between serum lipid markers and HC viscoelastic properties in healthy adults. We found that elevated serum TGs are associated with a reduction in HC integrity while HDL-C may help maintain HC integrity. Collectively, our findings suggest that the TG/HDL-C ratio is inversely associated with memory performance, even after correcting for age, and precedes any loss of HC volume which underscores the importance of cardiometabolic risk factors on memory function before any clinical changes of cognitive impairment can occur. Future research should explore how diet and other lifestyle factors that are known to modulate serum lipids affect HC integrity to delay or reverse cognitive aging.
Footnotes
Authors’ contributions
FS, CLJ, and CRM conceived and designed the study; FS, PLD, TMD, and JCH performed the study procedures; FS, PLD, LVH, and MLC analyzed the data; FS, PLD, and LVH prepared the figures; FS, PLD, LVH, TMD, JCH, MLC, CLJ, and CRM interpreted the results; FS and CRM drafted the manuscript; all authors critically revised and approved the final manuscript.
Acknowledgments
We thank our research nurse, Wendy Nichols, for assisting with blood draws and our undergraduate research volunteer, Elizabeth Habash, for help with data entry.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following grants from National Institutes of Health K01AG054731, R01AG058853, P20GM113125, and P20GM103653.
