Abstract
Senescence-associated alterations in microglia may have profound impact on cerebral homeostasis and stroke outcomes. However, the lack of a transcriptome-wide comparison between young and aged microglia in the context of ischemia limits our understanding of aging-related mechanisms. Herein, we performed RNA sequencing analysis of microglia purified from cerebral hemispheres of young adult (10-week-old) and aged (18-month-old) mice five days after distal middle cerebral artery occlusion or after sham operation. Considerable transcriptional differences were observed between young and aged microglia in healthy brains, indicating heightened chronic inflammation in aged microglia. Following stroke, the overall transcriptional activation was more robust (>13-fold in the number of genes upregulated) in young microglia than in aged microglia. Gene clusters with functional implications in immune inflammatory responses, immune cell chemotaxis, tissue remodeling, and cell-cell interactions were markedly activated in microglia of young but not aged stroke mice. Consistent with the genomic profiling predictions, post-stroke cerebral infiltration of peripheral immune cells was markedly decreased in aged mice compared to young mice. Moreover, post-ischemic aged microglia demonstrated reduced interaction with neighboring neurons and diminished polarity toward the infarct lesion. These alterations in microglial gene response and behavior may contribute to aging-driven vulnerability and poorer recovery after ischemic stroke.
Introduction
Microglia are the principal resident immune cells of the central nervous system (CNS), accounting for 5–12% of total brain cells varying in different brain regions. 1 Microglia possess highly specialized plasticity with the capability of adapting to different local microenvironments.2–6 Aging markedly increases CNS inflammatory signatures, implicating that aged microglia have a sensitized phenotype.7–9 Notably, exposure to such aging-afforded neuroinflammatory environment has been shown to change the transcriptome profile of microglia in both human and rodents.10,11 Several studies examining aging-associated transcriptional changes in microglia mainly focus on neurodegenerative diseases and demyelination disorders.12,13 However, few studies have explored senescence-associated alterations in microglia in the context of ischemic or hemorrhagic stroke.
Aging is not only an independent risk factor for stroke incidence14,15 but also a major negative determinant for stroke outcomes, as advanced ages are associated with poorer neurological recovery in stroke patients and in animal stroke models.16–18 Although known factors such as increased co-morbidities likely contribute to aging-driven poorer outcomes after ischemic stroke, 19 senescence-associated alteration in microglial functions has emerged as a potential new mechanism.18,20,21 Studies examining inflammation-phenotypic (pro- vs. anti-inflammatory) markers in stroke models reveal a reduced anti-/pro-inflammatory ratio in microglia of aged mice compared to young adult mice, 18 suggesting that aged microglia shift towards a pro-inflammatory and detrimental phenotype after cerebral ischemia. However, such data must be interpreted with great cautions. The assessment of one or several specific markers might not accurately recognize the overall inflammatory phenotypes of post-ischemic microglia. Moreover, microglia possess other functional properties such as production and release of trophic and angiogenic factors that could have important impact on cerebral homeostasis and stroke outcomes. 22
To understand the aging effect on microglial genomic responses and its potential influence on stroke outcomes, we performed genome-wide transcriptional profiling of microglia purified from cerebral hemispheres of young adult and aged mice under both healthy and post-ischemic conditions. The current study aimed to determine: (1) senescence-associated alterations in microglial genomic profiles and their potential contribution to cerebral homeostasis in healthy aged mice; (2) age-specific genomic alterations in microglia after cerebral ischemia with functional implications of these cells in ischemic brain injury; (3) senescence-associated alterations in microglial genomic profiles and functional impairments that lead to worsened stroke recovery in aged mice.
Materials and methods
Methodological details beyond the descriptions below are provided in
Male young adult (10 weeks old) and aged (18 months old) C57BL/6 mice were subjected to focal cerebral ischemia induced by permanent occlusion of the left distal middle cerebral artery (MCA) and left common carotid artery (CCA). 18 To investigate age-related transcriptome differences in microglia in response to ischemic stroke, microglia were extracted from young and aged mouse brains by fluorescence-activated cell sorting (FACS) five days after brain ischemia or after sham procedures, and subjected to bulk RNA sequencing (RNA-seq). Infiltration of various peripheral immune cells into the post-stroke brain was assessed by flow cytometry. 23 Interaction between microglia and neighboring cells was examined in young and aged mouse brains after stroke by immunostaining and image analyses with the Imaris software.
All RNA-seq data are deposited at GEO (GSE145265). All statistics are summarized in Supplementary Table 1.
Results
Young and aged microglia acquire distinct morphological features in response to ischemic stroke
To induce ischemic stroke in both young adult (10 weeks old) and aged (18 months old) mice, we adopted a tandem occlusion model consisting of left distal middle cerebral artery occlusion (dMCAO) and left common carotid artery occlusion (referred to as “dMCAO” thereafter). dMCAO resulted in greater reduction of cortical cerebral blood flow (CBF) and a larger ischemic core in aged mice than in young mice (Figure 1(a) and (b)), consistent with previous reports. 18 Furthermore, aged mice developed larger brain infarct than young mice as measured three and 5fivedays after dMCAO (Figure 1(c) and (d)). These data indicated that aged mice are more susceptible to dMCAO-induced ischemic brain injury.
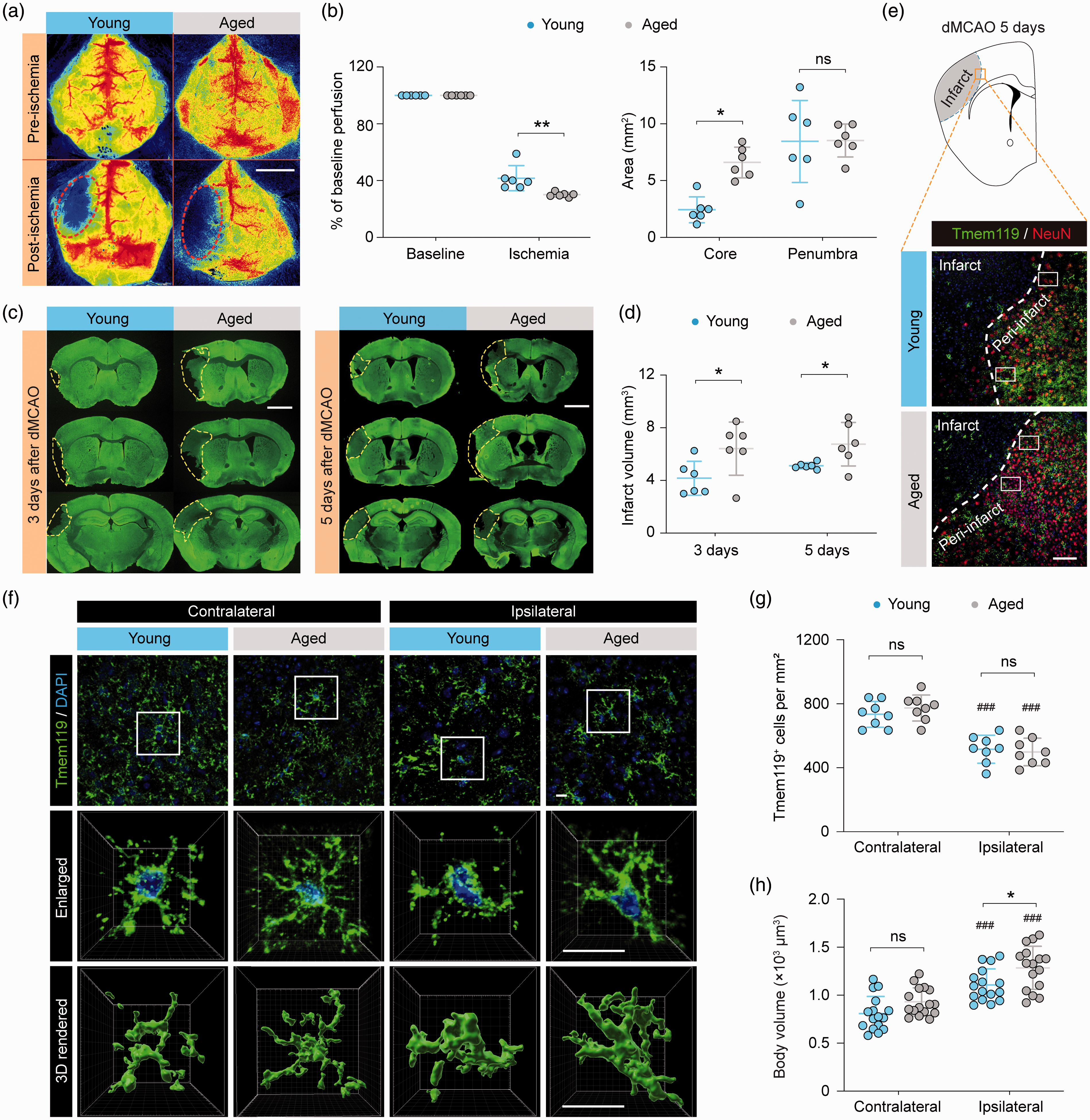
Morphological differences between young and aged microglia in response to ischemic stroke. Young adult (8–10 weeks old) and aged (18 months old) mice were subjected to dMCAO. (a,b) Cortical CBF was monitored by two-dimensional laser speckle imaging. (a) Representative laser speckle images show cortical CBF before (baseline) and 10 min after dMCAO. Dashed lines: the approximate boundaries of the ischemic area. (b) Summarized data on mean cortical CBF (left panel) and areas of the ischemic core (CBF reduction of >70% of baseline) and penumbra (CBF reduction of 50–70% of baseline; right panel).
It is well known that normal aging induces chronic low-level inflammation and increases microglial reactivity in the homeostatic brain. 24 However, whether young and aged microglia respond differently to ischemic brain injury remains poorly understood. Previous studies indicate that differences in microglial phenotypes between young and aged mice likely contribute to the deterioration of long-term stroke outcome in aged mice, under comparable chronic infarct sizes. 18 We examined the responses of microglia in young and aged mouse brains five days after dMCAO, using immunofluorescence staining of a specific microglial marker Tmem119. 25 In all brain sections double-labeled with Tmem119 and the neuronal marker NeuN, cortical infarct was easily identified based on the dramatically decreased density of NeuN immunofluorescence-positive cells (Figure 1(e)). Tmem119 immunofluorescence was readily detectable in all areas examined except within the infarct, where Tmem119 was drastically downregulated (Figure 1(e)). Therefore, Tmem119 immunofluorescence was analyzed mainly in the peri-infarct areas in all subsequent experiments. In the non-injured contralateral hemisphere, the numbers of Tmem119+ microglia were comparable between young and aged mice, and young and aged microglia had similar cell body volumes (Figure 1(f) to (h)). There were significantly less Tmem119+ cells in the ipsilesional peri-infarct regions than in the contralateral side in both young and aged mice (Figure 1(g)). A concomitant enlargement of cell body volume was also observed in the ipsilesional side (Figure 1(h)), indicating that these cells were activated in response to brain ischemia. Interestingly, the body volumes of aged microglia were significantly larger than young microglia five days after dMCAO (Figure 1(h)). This morphological difference indicated that aged microglia may have different functions from young microglia in the post-stroke brain, which could contribute to the previously reported worse long-term functional outcomes of aged mice after ischemic stroke.17,18
Aging primes microglia to a unique activation state in the homeostatic brain
To explore the intrinsic genomic differences between young and aged microglia, we used FACS to purify microglia (CD11b+CD45low cells) from the brains of young adult mice and aged mice five days after dMCAO or after sham operation, and performed bulk RNA-seq on isolated cells (Supplementary Figure 1(a) to (c)). FACS-purified cells expressed high levels of microglia signature genes, such as
Next, differential expression analysis was performed on RNA-seq data to determine the transcriptomic changes induced by aging in homeostatic microglia from non-injured sham brains. We identified 142 differentially expressed genes (DEGs; defined as genes with a fold change > 2 or <−2 and a false discovery rate (FDR)<0.05) in microglia from aged sham mice compared to young sham mice (Figure 2(a) and Supplementary Table 2), representing a moderate alteration of the genome (0.6% of total ∼23,000 genes). Aging downregulated 12 genes in microglia, including genes encoding the G protein-coupled receptors Gpr165 and Gpr34, and the heat shock protein family members Hspb1, Hspa1a, Hspa1b, and Hsph1 (Figure 2(b)). On the other hand, 130 genes were upregulated in aged microglia compared to young microglia, the functional implications of which were further explored by gene ontology (GO) enrichment analysis. A total of 263 GO terms were significantly overrepresented (
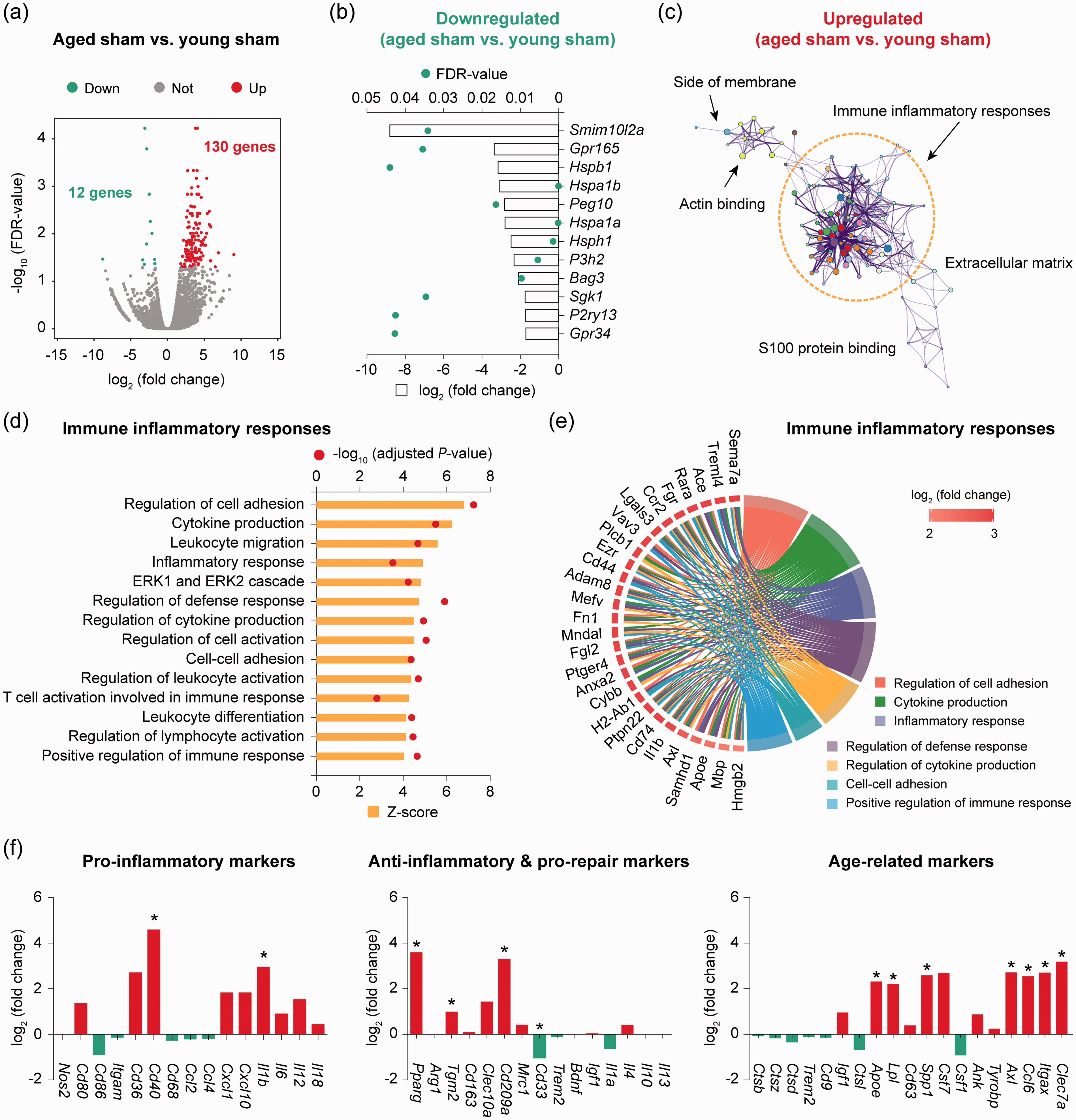
RNA-seq reveals transcriptomic differences between young and aged microglia in the homeostatic brain. Microglia (CD11b+CD45low cells) were sorted from the brain of young adult and aged mice after sham operation and subjected to bulk RNA-seq. (a) Differential expression analysis was performed on RNA-seq data. Volcano plot shows the differentially expressed genes (DEGs; fold change > 2 or < −2, false discovery rate (FDR) < 0.05) in aged microglia compared to young microglia. (b) Expression profiles of the 12 downregulated DEGs in aged microglia compared to young microglia. (c) Gene ontology (GO) enrichment analysis was performed using
We then annotated the relationship between specific genes and biological processes in the
We also analyzed the transcriptional signature genes defining pro-inflammatory, anti-inflammatory, or senescent microglial populations (Figure 2(f)). Two out of 15 pro-inflammatory genes (
Ischemic stroke induces distinct transcriptomic alterations in young and aged microglia
We sought to explore the differences in microglial gene expression profiles between young adult and aged mice in response to ischemic stroke by comparing their transcriptome five days after dMCAO relative to sham operation. We chose five days after dMCAO for microglial gene profiling as microglia are maximally activated at this time point after dMCAO.
18
In young microglia, dMCAO upregulated 250 genes and downregulated 21 genes (Figure 3(a) and Supplementary Table 4). Aged microglia showed only 18 upregulated genes and 40 downregulated genes after dMCAO (Figure 3(a) and Supplementary Table 5). Functionally, GO enrichment analysis of these DEGs predicted that 256 GO terms were activated (z-score> 2,
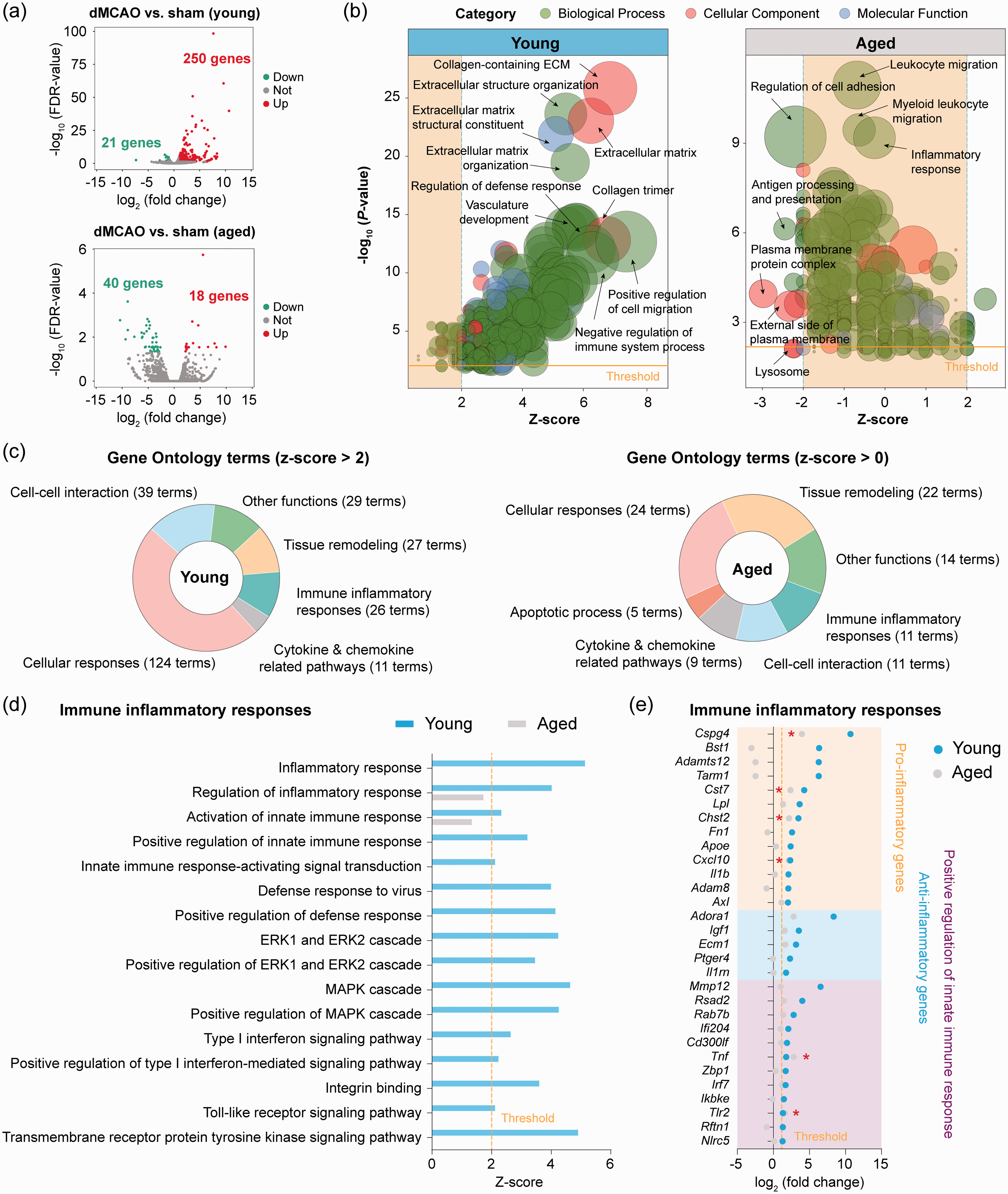
Transcriptional differences between young and aged microglia in response to ischemic stroke. Differential expression analysis was performed on RNA-seq data obtained from young and aged microglia five days after dMCAO or after sham operation. (a) Volcano plots show the DEGs (fold change > 2 or < −2, FDR < 0.05) in microglia from dMCAO brain versus sham brain in young adult (upper panel) and aged (lower panel) mice. (b) GO enrichment analysis was performed using
We further compared individual biological processes in the
We also extracted genes participating in the positive regulation of inflammatory responses (Figure 3(e)), which revealed 12 genes that were significantly upregulated (>2-fold, FDR < 0.05) in microglia of young adult mice. Several of these genes are well characterized for their regulatory roles in inflammation, including
Age-afforded decline in immune cell chemotaxis after ischemic stroke
We analyzed post-stroke gene expression profiles for pathways involved in cytokine and chemokine production in young and aged microglia. Eleven GO terms in these pathways were predicted to be activated (z-score > 2,
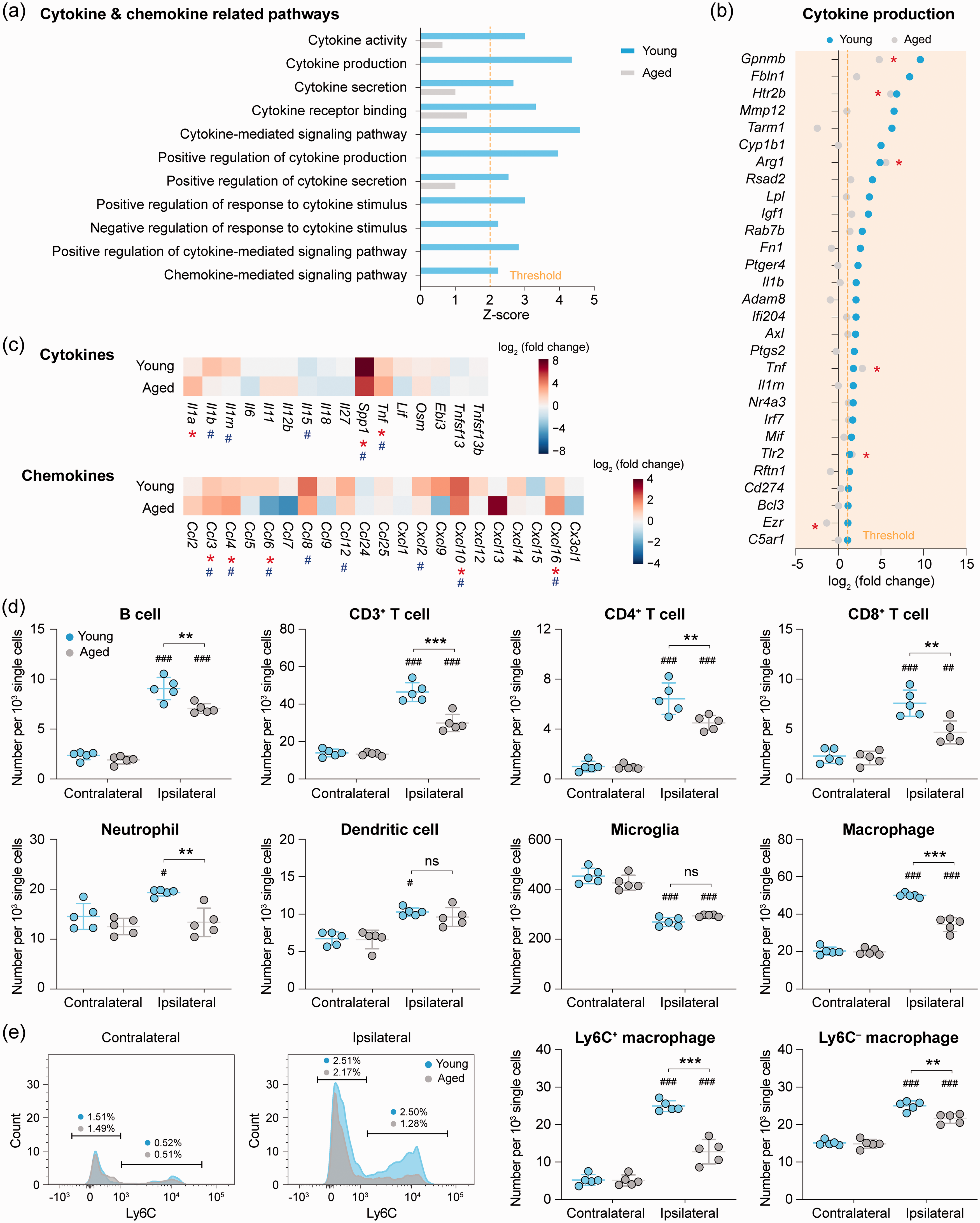
Reduction of cerebral immune cell chemotaxis in aged mice after ischemic stroke. (a) GO enrichment analysis was performed by
After ischemic stroke, a variety of chemokines are produced in the brain, attracting blood immune cells to enter the ischemic brain parenchyma.
50
Since our RNA-seq data showed that aging impairs stroke-induced upregulation of chemokine-coding genes in microglia, we hypothesized that such decline of chemokine gene expression may negatively impact immune cell infiltration after brain ischemia in aged mice. To test this hypothesis, we quantified the various immune cell populations in the brain of young and aged mice using flow cytometry five days after dMCAO (Supplementary Figure 4) as a means for functional verification of this impaired chemotaxis in aged microglia. Compared to the non-ischemic hemispheres contralateral to stroke, as expected there were robust increases in the amount of infiltrating immune cells in the ischemic hemisphere of young mice, including B cells, CD3+ T cells (both CD4+ and CD8+),
51
neutrophils, dendritic cells, and macrophages52,53 (both Ly6C+ and Ly6C–; Figure 4(d) and (e)). In contrast, indeed the infiltration of neutrophils and dendritic cells was completely abolished in post-stroke aged mice (
Activation of tissue remodeling pathways is diminished in aged microglia after ischemic stroke
We analyzed gene expression profiles for functional pathways involved in tissues remodeling. Twenty-seven GO terms in these pathways were predicted to be activated (z-score > 2,
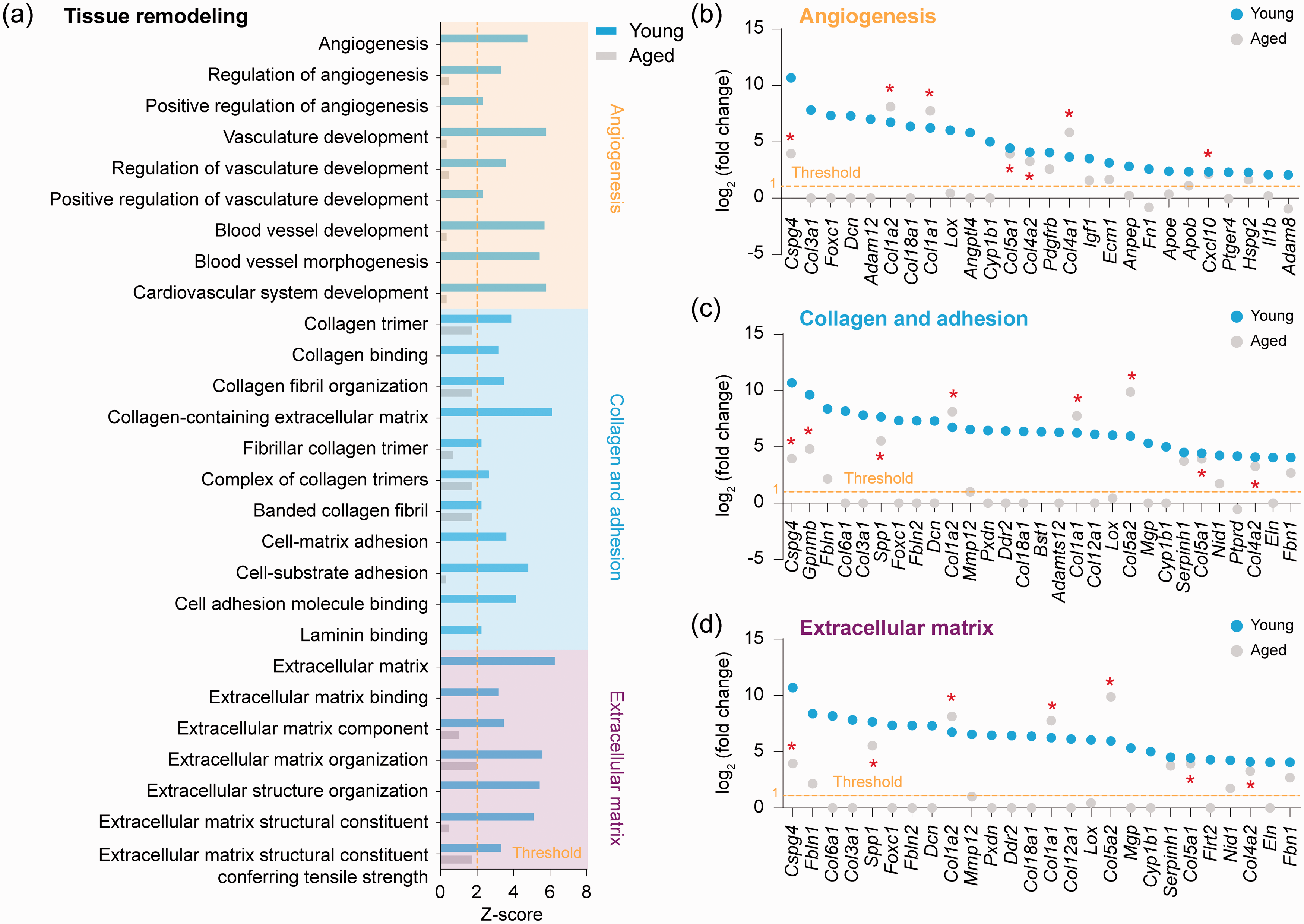
Transcriptional regulation of tissue remodeling-related genes in aged and young microglia after ischemic stroke. (a) GO enrichment analysis was performed by
Suppression of genes and pathways involved in cell–cell interactions in aged microglia after stroke
Following ischemic brain injury, activated microglia influence the activities of surrounding cells through cell–cell interactions,
54
and direct (cell contact) or indirect (
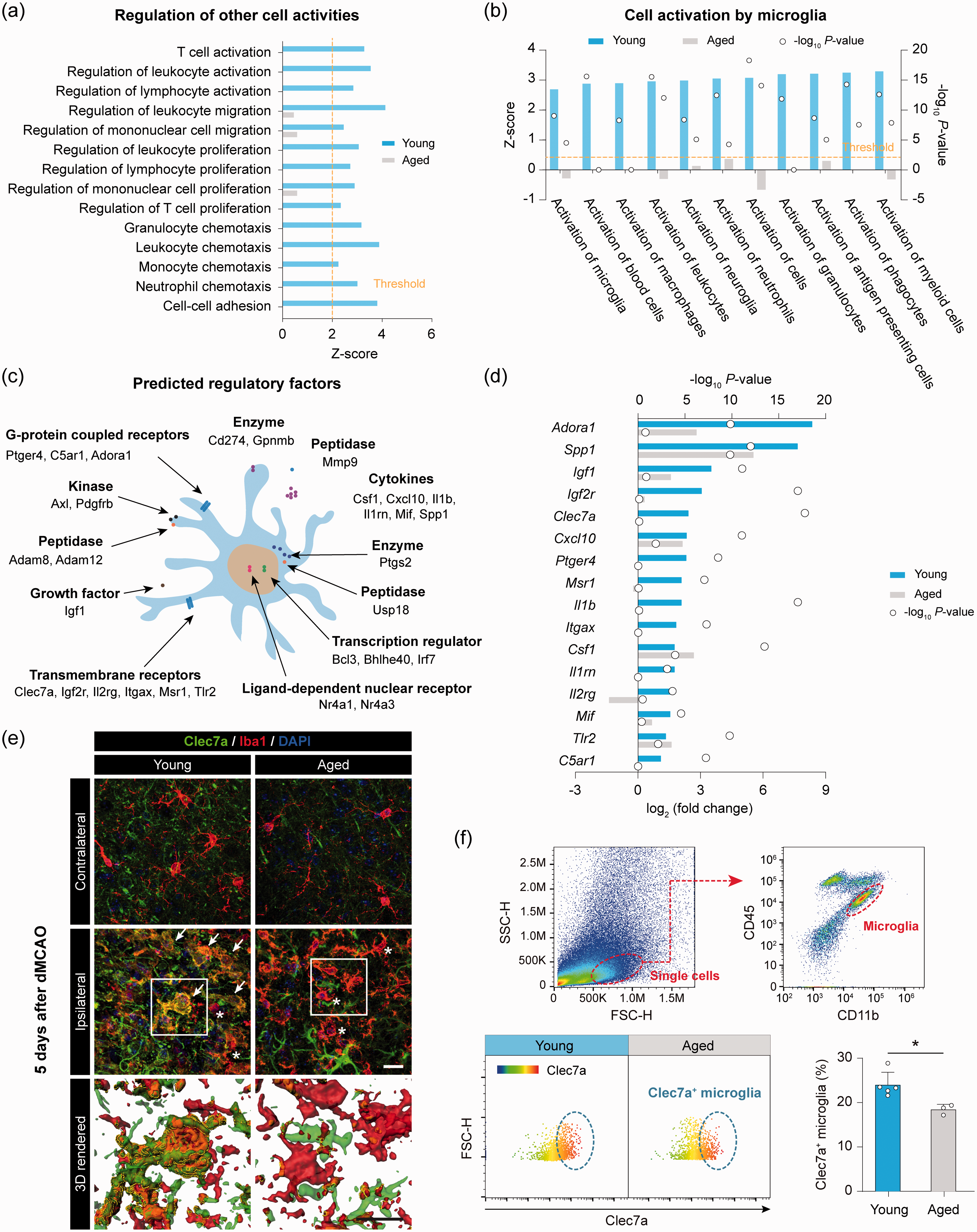
Impaired transcriptional regulation of cell–cell interaction molecules by aged microglia after ischemic stroke. (a) GO enrichment analysis was performed by
We next examined the genes in these pathways whose products are active in the plasma membrane or extracellular space and may therefore play a regulatory role on other cells, such as
We verified whether the age-dependent changes of gene expression at the RNA level led to changes at the protein level after stroke, focusing on
Aged microglia demonstrate less interaction with neighboring cells in the post-stroke brain
Given the functional importance of cell–cell interactions between microglia and surrounding cells, and since our RNA-seq data suggested that there are remarkable differences between young and aged microglia in their cell–cell communication behavior after stroke, we sought to explore such differences at the cellular level in the peri-infarct area (as depicted in Figure 1(e)) using double-label immunofluorescence staining followed by Imaris-assisted image analysis. To this end, we performed immunostaining using the microglial marker Tmem119 and the neuron marker NeuN, the astrocyte maker GFAP, or the oligodendrocyte marker APC to examine microglial interactions with neighboring cells in both young and aged mice five days after dMCAO (Figure 7 and Supplementary Figure 5).
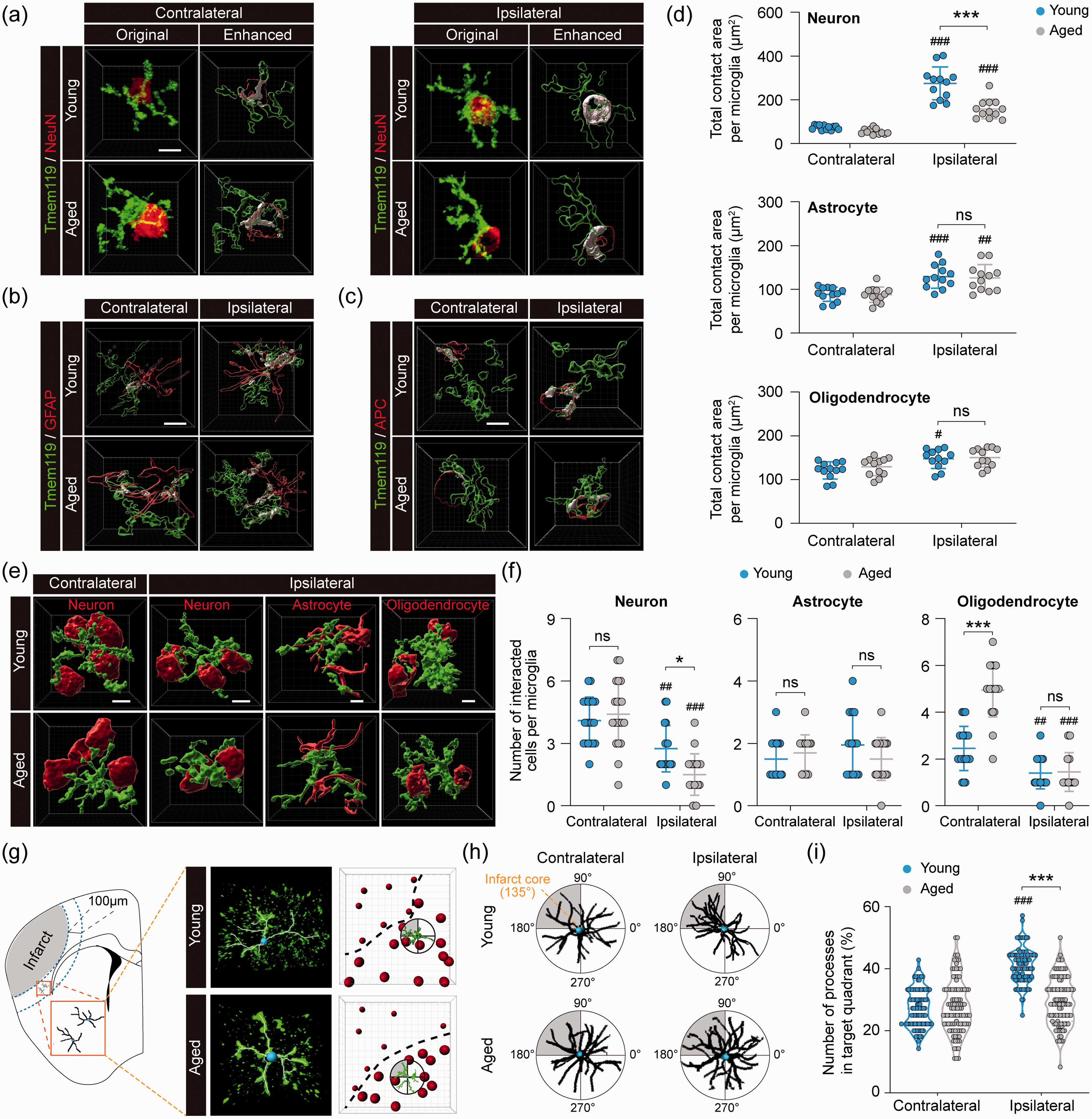
Aged microglia demonstrate reduced interactive responses to ischemic neurons and infarct in the post-stroke brain. Young adult and aged mice were subjected to dMCAO. The interaction between microglia and neighboring brain cells was examined by immunofluorescence staining five days after dMCAO in the ipsilesional peri-infarct regions and corresponding regions in the non-injured contralateral hemisphere. (a) Original images from confocal microscopy and Imaris-enhanced images showing the cell–cell contact areas between Tmem119+ microglia and NeuN+ neurons in white color. (b,c) Imaris-enhanced images showing the cell–cell contact areas between Tmem119+ microglia and GFAP+ astrocytes (b) or APC+ oligodendrocytes (c). See Supplementary Figure 5(a) to (c) for all original immunofluorescence images. (d) Summarized data of total contact area with neurons, astrocytes and oligodendrocytes per microglia.
Microglia can communicate with neighboring cells via direct cell–cell contact. Hence, cell–cell surface contact area is a rational parameter for cell–cell interactions. In the non-injured contralesional hemisphere, microglia formed contact with neurons, astrocytes, and oligodendrocytes, with no significant differences in the total cell–cell contact area per microglia between young and aged mice (Figure 7(a) and (b)). The total contact areas per microglia were significantly larger in the ipsilesional peri-infarct area than in the contralateral side for all three types of cells in both young and aged mice (Figure 7(a) and (b)), suggesting enhanced cell–cell engagement between microglia and neighboring cells after ischemia. Such changes were the most robust in microglia–neuron interactions after stroke, exhibiting approximately 3-fold and 1.8-fold increases in total contact area per microglia in young and aged mice, respectively (
Microglia can respond to brain injury by extending their processes towards the injury site, a reaction termed “polarity”. 58 To study microglial polarity after stroke, we used Imaris to analyze Tmem119+ cells in the peri-infarct area by quantifying their processes that were present in the quadrant proximate to the infarct (Figure 7(g) and (h)). In non-injured contralateral cortex of young and aged mice, microglial processes were evenly distributed in all four quadrants (Figure 7(h) and (i)). In the peri-infarct area of young mice, dMCAO significantly increased microglial polarity towards the infarct (Figure 7(h) and (i)). In contrast, such increased microglial polarity was not detectable in aged mice (Figure 7(i)). These results further supported our RNA-seq findings that aged microglia exhibit decreased communications with other cells, including injured or dying cells close to the infarct.
Discussion
The data presented in this report provide, for the first time, genome-wide transcriptional profiling of microglia purified from the brain of young adult and aged mice under both homeostatic and post-ischemic conditions. Major findings of our study include that (i) there are considerable transcriptional differences between young and aged microglia in the homeostatic brain, implicating a heightened chronic inflammation state in aged microglia; (ii) stroke-induced transcriptional activation was substantially suppressed in aged microglia compared to young microglia, especially those gene clusters relevant to immune inflammatory responses, immune cell chemotaxis, tissue remodeling and repair, and cell–cell interactions; (iii) consistent with the transcriptional profiling predictions, post-stroke cerebral infiltration of peripheral immune cells and microglial interactions with neighboring cells were markedly decreased in aged mice compared to young mice.
Transcriptomic comparison between young and aged microglia in the homeostatic brain identified 142 DEGs, representing a moderate alteration of the genome (0.6% of total ∼23,000 genes). The largest cluster of biological functions overrepresented by these DEGs is associated with immune inflammatory responses, including regulation of cell adhesion, cytokine production, leukocyte migration, and regulation of defense response. These results suggest that microglia in healthy aged mice are in an enhanced pro-inflammatory state compared to young microglia. This finding is strikingly consistent with previously reports, in which production of pro-inflammatory cytokines is found to be upregulated in microglia during aging.10,59,60 While the mechanism underlying such senescence-associated chronic inflammation remains elusive, emerging evidence supports the longstanding postulation of an infectious etiology of brain aging and age-prone neurodegenerative disorders, such as Alzheimer’s disease. 61 The “microbial hypothesis” suggests that a number of pathogens, including latent viruses, gut bacteria, periodontal bacteria, and pulmonary bacteria, may invade the CNS via the trigeminal nerve or the oral-olfactory route, or from systemic circulation by crossing the compromised blood–brain barrier during aging.62–65 More recent studies have emphasized the potentially important role of aging-associated increases in gut permeability and activation of the microbiota–gut–brain axis in chronic systemic inflammation, brain senescence and neurodegeneration.66,67
The major objective of this study is to explore aging-associated alterations in microglial transcriptome five days after ischemic stroke, as several key biological processes of neuroinflammation, including innate immunity responses and cerebral infiltration of circulating immune cells, peak at this sub-acute injury stage.68,69 Our data reveal that cerebral ischemia induces a considerable transcriptional activation in young microglia, upregulating approximately 1.1% of the transcriptome (250 of total ∼23,000 genes). Unsurprisingly, many of the functional gene-clusters which were predicted to be strongly activated in young microglia are associated with immune inflammatory responses (26 GO terms) and cytokine and chemokine pathways (11 GO terms). In contrast, ischemic stroke induced merely a modest transcriptional activation in aged microglia (18 genes upregulated), and none of the aforementioned neuroinflammation-relevant GO terms was activated in aged microglia. These results suggest that the immune inflammatory responses are severely suppressed in aged microglia after ischemic stroke compared to young microglia. As the transcriptional gene profile for chemokines predicts negative regulation on chemotaxis activity in aged microglia after ischemia, we quantified post-stroke cerebral infiltration of various types of immune cells in young and aged mice. Consistent with the gene profiling predictions, the infiltration of all types of examined immune cells was found to be diminished in aged mice compared to young mice after ischemic stroke. The suppressed immune inflammatory responses and brain infiltration of circulating immune cells seem to contradict to the poorer stroke outcomes in aged mice, as the general notion indicates that neuroinflammation exacerbates ischemic brain injury. However, this concept has been reversed by many recent studies, which suggest: (1) certain cytokines, such as IL-4, IL-10, IL-33 and TGFα are neuroprotective against ischemic brain injury;70–74 (2) certain infiltrating immune cells, such as bone-marrow monocytes-derived macrophages and regulatory T cells (Tregs), confer neuroprotection and/or are directly involved in neurovascular remodeling after ischemic stroke;69,75,76 (3) certain pro-inflammatory molecules, such as MMP-9, may be important for post-injury brain repair. 77
Another novel finding of this study is that several gene-clusters with functional implications in brain repair are robustly activated in young microglia, but not in aged microglia, after ischemic stroke. These gene-clusters include angiogenesis and vascular development (9 GO terms), collagen and adhesion (11 GO terms), and extracellular matrix organization (7 GO terms), all of which have strong relevance in post-stroke brain tissue remodeling and functional recovery. The suggestive involvement of microglia in promoting angiogenesis in post-ischemic brain is unexpected, yet it is a legitimate assumption. Our recent study has explored a similar functional role for infiltrating monocyte-derived macrophages using the dMCAO model. 69 In the latter study, PPARγ was identified as the key upstream regulatory molecule for the pro-angiogenesis gene-cluster in the myeloid-cell lineage, as conditional knockout of PPARγ selectively from these cells led to impaired angiogenesis in post-stroke brain. 69 Thus, our studies point to a novel source of molecular signals that contribute to neurovascular remodeling after ischemic stroke. This mechanism may be particularly significant for aging-associated impairment of stroke recovery. Among the post-stroke brain repair processes, diminished angiogenesis is strongly correlated with poorer stroke recovery in aged mice. 17
Microglia are highly dynamic cells that interact with other cells via direct (physical cell–cell touch) or indirect (release of soluble molecules) mechanisms.22,78 Our transcriptional profiling data suggest that the capabilities to interact and activate surrounding cells are remarkably dampened in aged microglia after cerebral ischemia. Gene clusters with functional implications for cellular activation are strongly activated in young microglia, but not in aged microglia, after ischemic stroke. Further analysis of the GO terms revealed that post-ischemic young microglia not only activate brain residential cells, such as neuroglia and microglia, but also regulate the migration, proliferation, and chemotaxis of infiltrating immune cells, such as neutrophils, lymphocytes and phagocytes. Our follow-up experiments partially confirmed the remarkable differences between young and aged microglia in their cell–cell communication behavior after stroke. First, we demonstrated that the protein expression levels for Clec7a, a pattern-recognition receptor on microglia that likely mediates cell–cell interactions, 57 are decreased in aged mice after stroke. Second, we found that the cell–cell physical contacts between microglia and neurons are suppressed in aged mice after stroke. Third, we showed that, in the peri-infarct brain regions, microglial polarity towards the infarct is nearly lost in aged mice after stroke, as compared to young mice. These results suggest, albeit do not prove, that microglia may influence stroke outcomes via functional cell–cell interactions. Future studies that elucidate the specific molecular signaling pathways underlying distinct microglial behaviors may help uncover senescence mechanisms. The permanent focal cerebral ischemia model that we used in this study involves occlusion of the distal MCA and ipsilateral CCA. This model has high clinical relevance, as the majority of stroke patients do not receive reperfusion therapy, especially for distal branch occlusions that are more common and remain beyond the reach of endovascular thrombectomy. In future studies, conclusions drawn from this study should be tested in other stroke models of ischemia reperfusion injury after proximal MCAO, which are applicable to the recent advances in clinical reperfusion therapies for a significant minority of stroke patients. 16
Several limitations of the present study should be noted. First, the RNA-seq profiling was performed using purified microglia from cerebral hemispheres. The CD11b+CD45low-based cell sorting approach, while ensuring high level of cell purity, would not include microglia that express high levels of CD45. Upon activation after cerebral ischemia, a subpopulation of microglia may shift to the CD11b+CD45high population, 70 and the latter cell populations were excluded for genomic profiling in this study. Second, although the bulk RNA-seq technology offers the advantage of deep gene profiling (∼23,000 genes), the identified DEGs were the result of averaging gene expression-level changes in the entire biological sample (approximately 1.0 × 107 cells per sample in this study). Thus, a caveat with bulk RNA-seq is that this approach would miss out the subtle changes in gene expression, especially when such changes occur only in small subpopulations of cells within the biological sample. The approach combining of bulk and single-cell RNA-seq would help overcome both roadblocks. Third, the DEGs resulting from RNA-seq do not always dictate molecular alterations at either the protein expression or functional levels. In the current study, we have confirmed several changes at protein and functional levels as predicted by RNA-seq, including Clec7a protein expression, immune cell infiltration, and cell–cell interactions in young and aged microglia after stroke. Nevertheless, future studies are warranted to further understand the functional significances of transcriptional alterations characterized in this report. Overall, our study used data-driven analytic approaches to provide unbiased genome-level characterization of age-associated differences in microglial transcriptome under homeostatic and ischemic conditions. The datasets generated by this study are useful resources for future studies to screen for potential regulators to modulate microglial functions in aged subjects. However, solid conclusions on whether certain regulators are causatively connected to stroke outcomes must be obtained with mechanism-oriented approaches, such as specific gain or loss-of-function manipulations.
In conclusion, this study performed transcriptome-wide comparisons between young and aged microglia under both homeostatic and post-stroke conditions. The alterations in microglial gene response and behavior may contribute, at least in part, to aging-afforded vulnerability and poorer recovery after ischemic stroke. Future elucidation of the microglia-dependent cellular and molecular pathways may help uncover novel therapeutic targets to boost functional recovery of older stroke patients.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-2-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-xlsx-2-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-3-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-xlsx-3-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-4-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-xlsx-4-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-5-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-xlsx-5-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-6-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-xlsx-6-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-7-jcb-10.1177_0271678X20925655 - Supplemental material for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia
Supplemental material, sj-xlsx-7-jcb-10.1177_0271678X20925655 for Genome-wide transcriptomic analysis of microglia reveals impaired responses in aged mice after cerebral ischemia by Ligen Shi, Marcelo Rocha, Wenting Zhang, Ming Jiang, Sicheng Li, Qing Ye, Sulaiman H Hassan, Liqiang Liu, Maya N Adair, Jing Xu, Jianhua Luo, Xiaoming Hu, Lawrence R Wechsler, Jun Chen and Yejie Shi in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Pittsburgh Medical Center (UPMC) Immune Transplant and Therapy Center grant (to JC, LRW, XH and YS) and the American Heart Association grant 17SDG33630130 (to YS). JC is the Richard King Mellon Professor of Neurology at the University of Pittsburgh and is also supported by the Senior Research Career Scientist Award and Merit Review grants (BX002495 and BX003377) from the U.S. Department of Veterans Affairs. LRW is the Henry B Higman Professor of Neurology at the University of Pittsburgh.
Acknowledgements
The RNA sequencing data analysis in this work used Ingenuity Pathway Analysis software licensed through the Molecular Biology Information Service of the Health Sciences Library System, University of Pittsburgh. We thank Patricia Strickler for administrative support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
JC and YS designed the research. LS, WZ, MJ, SL, QY, SHH, LL, MNA and JX performed the experiments. LS, YS, MR, LRW and JC analyzed and/or interpreted the data. JL mapped the RNA sequencing data. LS, JC and YS wrote the paper. MR, XH and LRW critically revised the paper.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
