Abstract
Mounting evidence has demonstrated that both innate and adaptive immune cells infiltrate into the brain after ischemic stroke. T cell invasion has been found in the ischemic region up to one month post experimental ischemic stroke and has been shown to persist for years in stroke patients. However, the function and phenotypic characteristics of the brain invading T cells after ischemic stroke have not been investigated. In the current study, we determined the function of brain invading T cells in the acute and chronic phase following experimental ischemic stroke induced by transient middle cerebral artery occlusion. We observed a significant increase of CD4+ and CD8+ T cells presented in the peri-infarct area at up to one month after experimental ischemic stroke. The brain invading T cells after ischemic stroke demonstrated close interaction with active astrocytes and a progressive proinflammatory phenotype as evidenced by the increased expression of T cell activation markers CD44 and CD25, proinflammatory cytokines INF-γ, IL-17, IL-10, TNF-α, and perforin, with corresponding transcriptional factors T-bet and RORc. Our results indicated a prolonged activation of brain invading CD4+ and CD8+ T cells after ischemic stroke which may play a role in the neural repair process after stroke.
Introduction
Inflammation is a critical part of the normal wound healing process, involving mostly innate immune cells such as neutrophils, macrophages, and mast cells.1,2 The central nervous system (CNS) has long been recognized as a site of “immune privilege” because of the existence of the blood brain barrier (BBB) and absence of lymphatic vasculature. As a consequence, the neural repairing process after ischemic brain damage is distinctive from the peripheral tissues by the involvement of CNS resident innate immune cells, such as microglia and astrocytes. Nonetheless, the BBB becomes permeable after ischemic stroke, allowing peripheral immune cells to infiltrate into brain parenchyma and participate in the repairing process. 3
Mounting evidence has demonstrated that peripheral innate immune cells such as neutrophils, 4 macrophages,5,6 and natural killer cells 7 are recruited to the infarct site after ischemic stroke in patients as well as experimental ischemic stroke models. In addition, adaptive immune cells have been found to infiltrate into brain parenchyma after ischemic stroke. Temporal and spatial patterns of T cell invasion after experimental ischemic stroke have been investigated using rodent middle cerebral artery occlusion (MCAO) models. Invading T cells were found to peak weeks after experimental ischemic stroke with predominant location in the peri-infarct zone8–10 (see Selvaraj and Stowe 11 ). Similarly, pathological studies have shown that T cell infiltration persists for years after stroke in human.12,13 Furthermore, the role of T cells in ischemic stroke outcomes have been explored in experimental ischemic stroke models. However, the action of T cells in the current research is highly heterogeneous in both methodology and results. On one hand, depletion of CD4+ or CD8+ T cells reduced infarct volume in experimental stroke.14,15 Blockage of lymphocyte invasion protected the brain against deleterious neuroinflammation after stroke.9,16 On the other hand, T cells that are specific to myelin antigens have been shown to reduce secondary neurodegeneration, enhance neurogenesis, and promote recovery after CNS injury.16–18
The recent discovery of functional meningeal lymphatic system suggests that the CNS is under immunosurveillance during steady state.19–22 Indeed, the CNS is not devoid of immune cells and cerebrospinal fluid (CSF) alone contains approximately half a million immune cells in a healthy adult. 23 Notably, the predominant cells in normal CSF are T cells (49% CD4+, 19% CD8+). 23 There is increasing evidence that immune cells, specifically T cells, play an important role in maintenance of CNS homeostasis and function.18,22,24–26 Our previous study discovered a regulatory T cell population in the normal rat brain with an activated/memory phenotype which may contribute to immunosurveillance and immunomodulation in the brain under physiological conditions. 27 However, the function and phenotypic characteristics of brain invading T cells after ischemic stroke have not been investigated. In the current study, we determined the function of brain invading T cells in the acute and chronic phases after experimental ischemic stroke. Our study indicated that experimental ischemic stroke induced long-term CD4+ and CD8+ T cell activation in the ischemic brain.
Materials and methods
Mouse experimental ischemic stroke model
Animal experimentation was approved by Institutional Animal Care and Use Committee (IACUC) of University of North Texas Health Science Center and was performed according to the IACUC, NIH and ARRIVE guidelines. Adult male C57B/6 mice (3 months old) were purchased from Charles River (Wilmington, MA, USA) and housed at the animal facilities with access to laboratory chow and water ad libitum under a fixed 12:12 light–dark cycle. Experimental ischemic stroke was induced by transient MCAO as described previously. 28 All surgeries were conducted under 2.0% isoflurane anesthesia. Body temperature was maintained at 37 ± 0.5℃ with a thermostat-controlled heating blanket during the procedure. A midline incision was made on the neck. Common carotid artery, external carotid artery and internal carotid artery (ICA) were dissected from the connective tissue. A silicon-coated 6-0 nylon monofilament was inserted into the left ICA and advanced till it occluded the origin of the middle cerebral artery (MCA). The MCA was occluded for 90 min after which reperfusion was reestablished by suture withdraw. The stroked animals were randomly assigned into two groups as described below. At specified times after stroke induction, mice were euthanized and spleens and brains were harvested for immune cell isolation. Animals which died prematurely were excluded from the study.
Isolation of immune cells from the brains and spleens
Mice were anesthetized by inhaling isoflurane and were intra-cardiacally perfused with 20 ml of ice-cold phosphate-buffered saline (PBS). Isolation of immune cells from the brains and spleens was performed at day 3 or one month post-MCAO as published earlier.27,29 Briefly, mouse cerebral hemispheres and spleens were cut into 1 mm3 pieces before digestion for 45 min at 37℃ with digestion buffer (RPMI 1640 medium supplemented with 10% fetal bovine serum, 1 mg/ml collagenase type IV, 50 µg/ml DNase I, 5 mM CaCl2). Digested tissues were then gently pressed through 40-µm cell strainers to prepare homogenized tissue suspension. The tissue suspension was then mixed with 4 volumes of 30% Percoll (GE Healthcare). The mixture was loaded onto 1 ml 70% Percoll, followed by centrifugation at 500 g for 20 min. Cells in the interface between 30% Percoll and 70% Percoll were collected for further use.
Flow cytometry analysis and cell sorting
For immune cell staining, isolated cells were stained with APC conjugated anti-mouse CD3e (145-2C11, eBioscience), PerCP anti-mouse CD4 (GK1.5, BioLegend), PE anti-mouse CD8a (53.6.7, BioLegend), PE-Cy7 anti-mouse CD44 (IM7, BioLegend), APC-Cy7 anti-mouse CD62L (MEL-14, BioLegend), APC-Cy7 anti-mouse CD25 (PC61, BioLegend). Cells were labelled with indicated antibodies on ice for 15 min before flow cytometry and cell sorting on a BD LSR-II flow cytometer and a BD influx Cell Sorter. For Ki67 (FITC-anti-Ki67, eBioscience) labelling, cells were fixed with Cytofix/perm buffer (BD Biosciences) for 15 min at room temperature, followed by permeabilization with 90% ice-cold methanol for 30 min. Cells were washed with PBS and labelled with Ki67 antibody for 1 h at room temperature and processed for flow cytometry.
Quantitative RT-PCR (Q-RT-PCR)
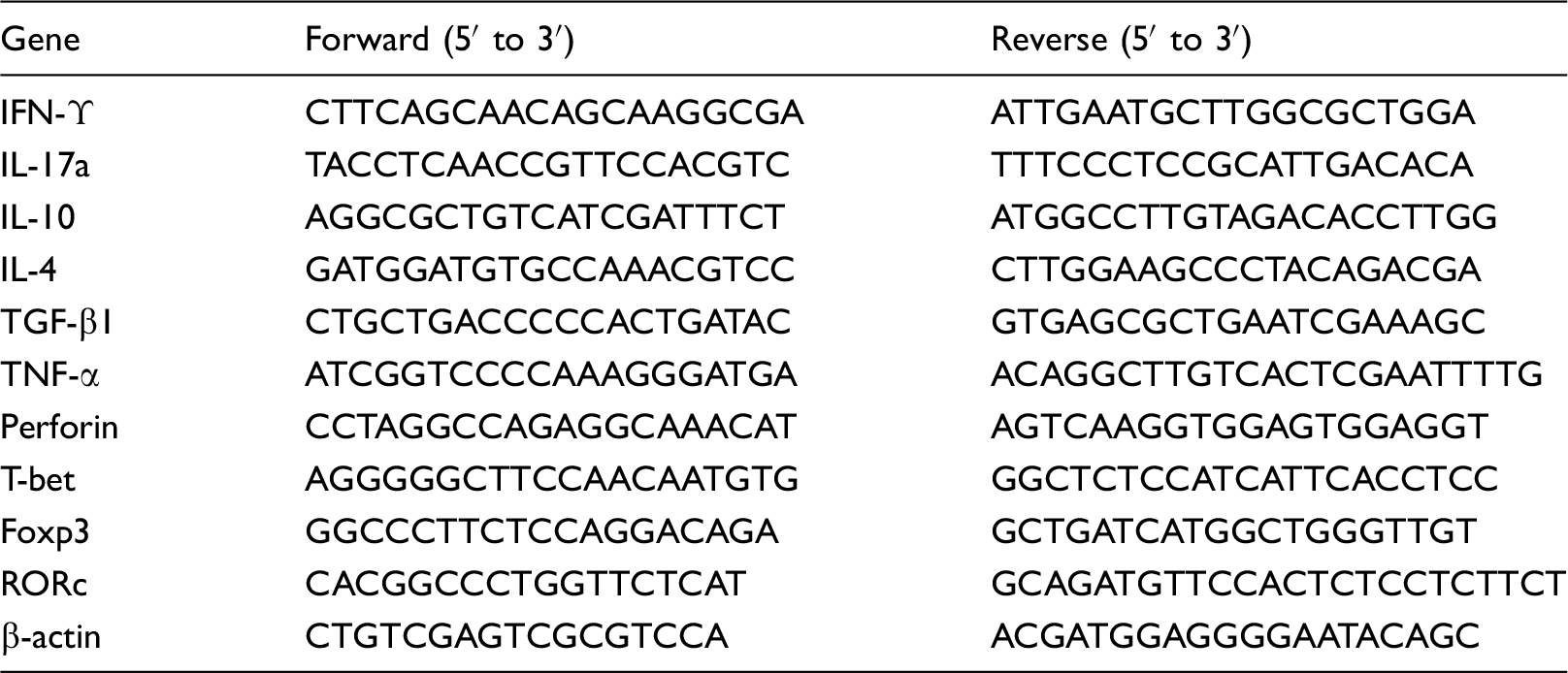
List of primer sequences used for Q-RT-PCR study.
Immunofluorescent staining
Mice were anesthetized by isoflurane inhalation and were intra-cardiacally perfused with 20 ml of 10% formalin. Five-micron paraffin-embedded brain sections were prepared and were incubated with antibodies against 2 µg/ml CD4 (eBioscience), CD8 (eBioscience), GFAP (Santa Cruz Biotechnology) and MAP-2 (Millipore) at 4℃ overnight. Sections were then incubated with 5 µg/ml of Alexa Fluor 488-conjugated goat anti-rat IgG and/or Alexa Fluor 594-conjgated goat anti-mouse IgG or Alexa Fluor 594-conjgated goat anti-rabbit IgG (Invitrogen). Sections were observed on an Axio Observer Z1 fluorescent microscope (Zeiss).
Statistical analysis
Graph Pad Prism 5 was used for statistical analysis. All the results are expressed as mean ± standard deviation (SD). Two-way analysis of variance and post-hoc Bonferroni analysis were conducted for multiple comparisons between groups. A p-value < 0.05 was considered to be statistically significant.
Results
Activated/memory T cells are present in the ischemic brain at one month after MCAO
To determine the long-term existence of T cells in the brain after ischemic stroke, mononuclear cells were isolated from the spleens and brains at three days or one month after ischemic stroke. As shown in Figure 1(a) to (d), more than 80% of splenic T cells were CD44-/CD62L+ naïve T cells at day 3 or one month after ischemic stroke. The proportions and activation status of splenic CD4+ and CD8+ T cells in the ischemic hemisphere were comparable at three days and one month after ischemic stroke. However, at three days after stroke, more than 50% of infiltrating brain T cells in the ischemic hemisphere (either CD4+ or CD8+) were CD44 positive, indicating their activated/memory phenotype. At one month after ischemic stroke, the proportion of CD44+ T cells in the ischemic hemisphere reached nearly 80%, suggesting that brain T cells were predominantly activated/memory T cells. Consistently, the proportion of CD44-/CD62+ naïve brain T cells (either CD4+ or CD8+) decreased to less than 5% at one month after stroke. Expression of another T cell activation marker, CD25, was also significantly increased in brain CD4+ T cells at one month after stroke, in comparison with splenic CD4+ T cells at day 3 after ischemic stroke (Figure 1(e)). Similarly, a much higher portion of brain invading CD4- T cells was CD25 positive as compared with CD4- T cells from the spleens (Supplementary data). The number of T cells isolated from the non-ischemic hemisphere was very low. Significant increase of T cell infiltration in the ischemic hemisphere was observed at 24 h after permanent and transient MCAO, while no significant change of T cells infiltration in the non-ischemic hemisphere was observed as compared with sham controls.
30
In our previous study, we had to pool several normal rat brains to obtain enough T cells for functional analysis.
27
In the current study, we were able to isolate low number of T cells from the contralateral hemisphere in two mice at each time point and significant increase of T cells were isolated from the ipsilateral hemisphere at three days and one month after ischemic stroke. Assessment of T cell abundance revealed a significant increase of both CD4+ and CD8+ T cells in the ipsilateral side of the brain at one month after ischemic stroke as compared with three days after stroke and contralateral hemisphere (Figure 1(f)). In the contralateral hemisphere, a low T cell amount was maintained at three days and one month after stroke.
Presence of T cells in ischemic mouse brains. (a & b) Flow cytometry dot plots showing T cell profile in spleens (a) and brains (b) at three days and one month after ischemic stroke induced by transient MCAO. (c & d) Quantitative analysis of activation status of CD4+ (c) and CD8+ (d) T cells in spleens and brains. (e) CD25 expression on splenic and brain CD4+ T cells at three days and one month after ischemic stroke. Upper panel: representative dot plots. Lower panel: statistic analysis. (f) Absolute abundance of T cells recovered from ischemic brains. *p < 0.05, **p < 0.01; ***p < 0.001. N=2 for the contralateral brain and N=4–5 for the ipsilateral brain per group. Absolute number of CD4+ T cells: 44, 70 in three days contralateral side; 67, 26 in one month contralateral side; 283, 463, 735, 600, 178 in three days ipsilateral side; 776, 1068, 794, 690 in one month ipsilateral side. Absolute number of CD8+ T cells: 61, 107 in three days contralateral side; 90, 50 in one month contralateral side; 154, 238, 305, 442, 164 in three days ipsilateral side; 514, 1002, 824, 563 in one month ipsilateral side.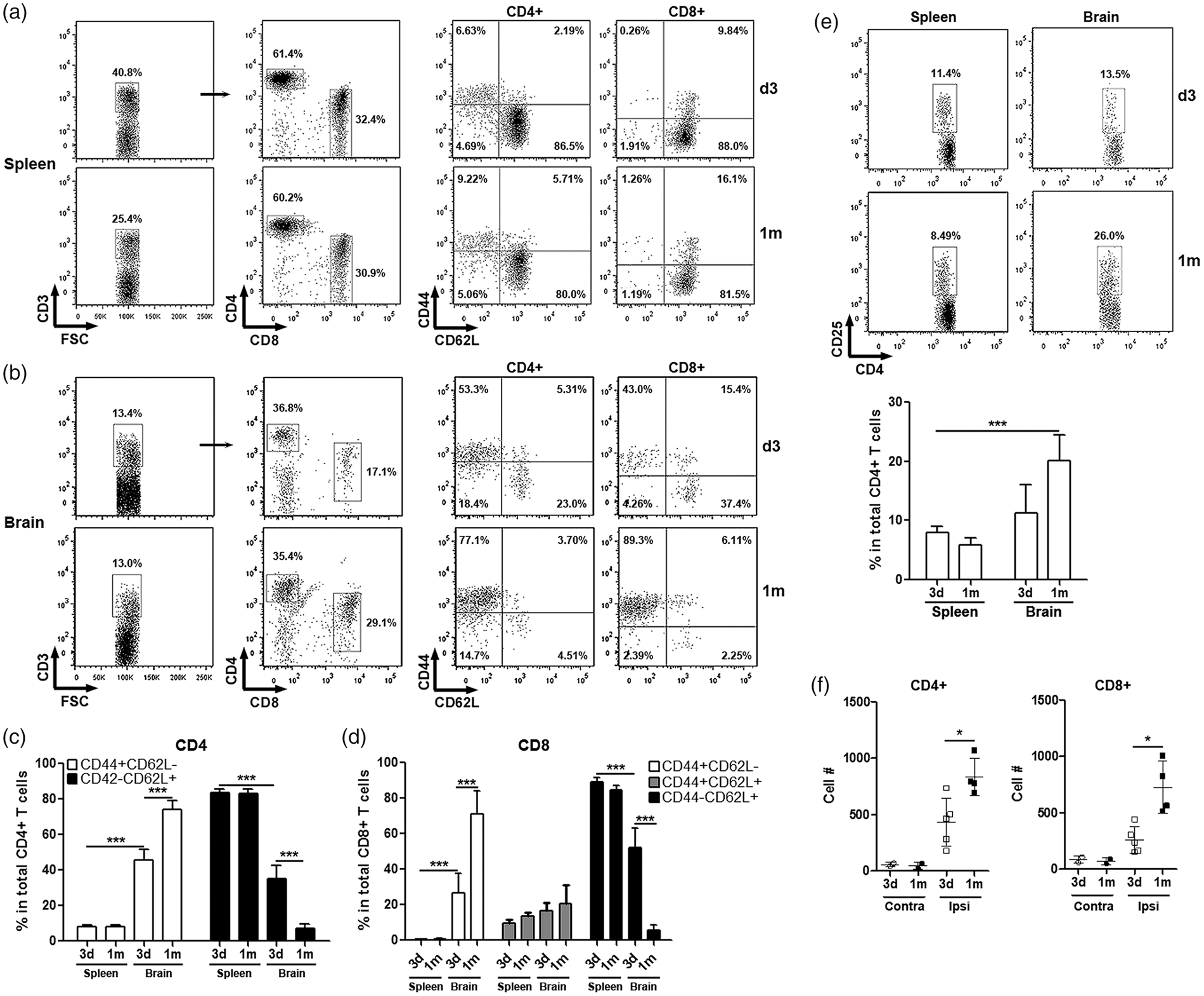
Brain T cells are highly functionally active at one month after experimental ischemic stroke
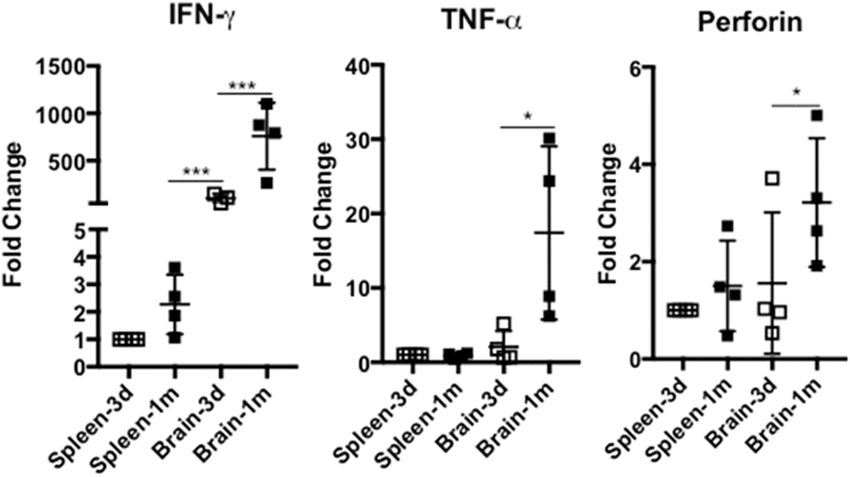
To evaluate the functional activity of T cells, gene expression of cytokines and master regulators in splenic and infiltrating brain CD4+ and CD8+ T cells were quantitated using q-RTPCR. Approximately 100–300 and 10,000 T cells of each T-cell subset from each hemisphere and spleen, respectively, at the indicated time after ischemic stroke were sorted for qRT-PCR. As shown in Figure 2(a) to (e), at three days after ischemic stroke, gene expression of IFN-γ, IL-17, and IL-10 was significantly upregulated in the infiltrating brain CD4+ T cells in comparison with splenic counterparts, while IL-4 gene expression was inhibited. At one month post ischemic stroke, brain CD4+ T cells had even higher IFN-γ and IL-17 gene expression while IL-4 expression was partially restored. Additionally, there was a non-significant trend of higher TGF-β gene expression in the infiltrating brain CD4+ T cells at one month after ischemic stroke. Furthermore, gene expression of T-bet and RORϒc was promoted in brain CD4+ T cells at three days and one month following ischemic stroke (Figure 2(f)). No significant alteration of Foxp3 gene expression was observed (Figure 2(f)). Similarly, gene expression of IFN-γ, TNF-α, and perforin in the invading brain CD8+ T cells was markedly upregulated at one month after ischemic stroke as compared with the infiltrating brain CD8+ T cells at three days after stroke and the splenic CD8+ T cells (Figure 3(a) to(c)).
QT-PCR of Expression of cytokines and master regulators in CD4+ T cells at three days and one month after ischemic stroke. (a to e) Cytokine IFN-γ, IL-17, IL-10, IL-4, and TGF-β expression in splenic and invading brain CD4+ T cells. (f) Expression of master regulators of T-bet, Foxp3, and RORc in splenic and brain invading CD4+ T cells. *p < 0.05, **p < 0.01; ***p < 0.001. N=4 per group. QT-PCR analysis of expression of INF-γ (a), TNF-α (b), and perforin (c) in splenic and brain invading CD8+ T cells at three days and one month after experimental stroke. *p < 0.05, **p < 0.01; ***p < 0.001. N=4 per group.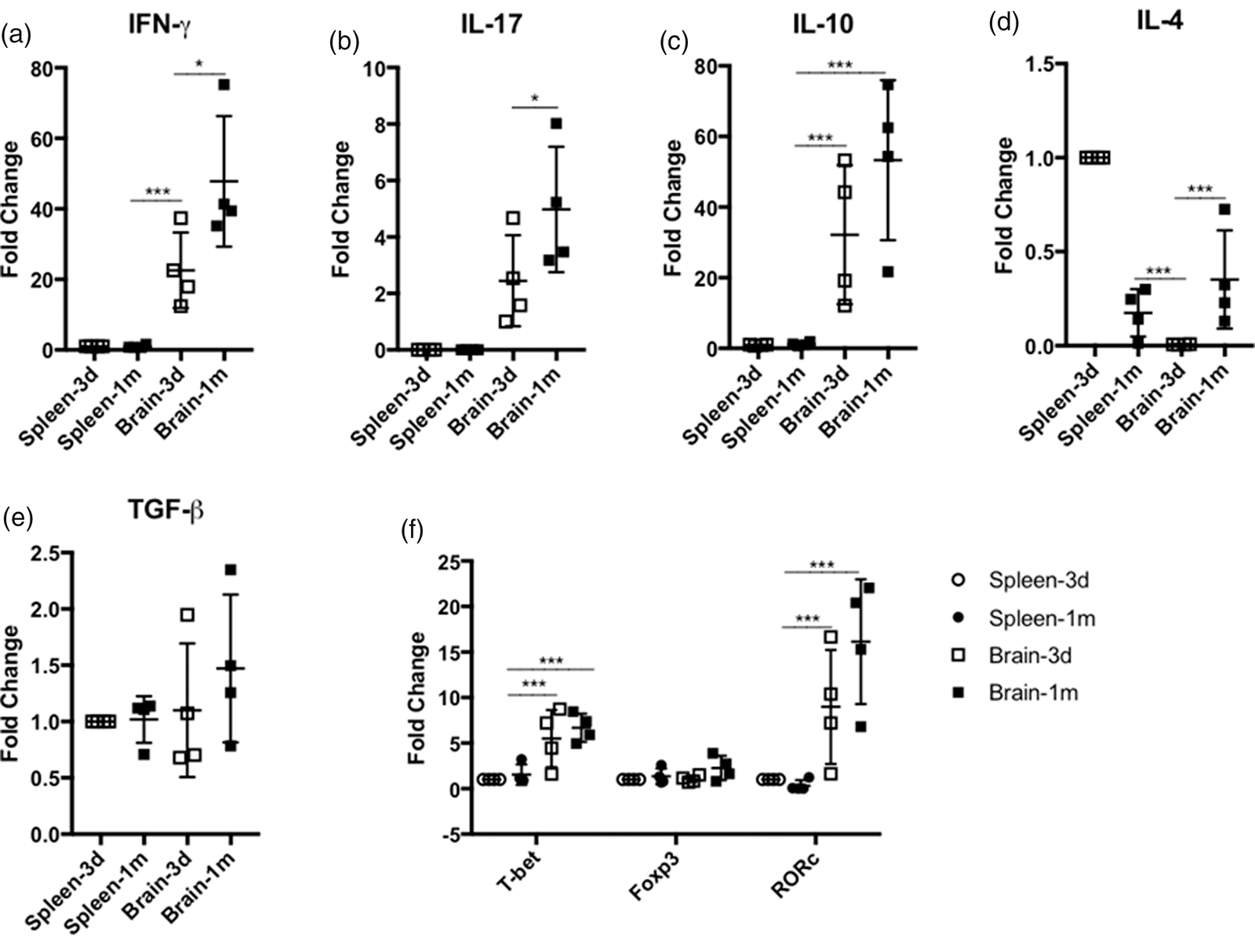
Brain T cells were proliferative and in close proximity to astrocytes
We expected that the increase of invading brain T cells abundance could be due to persistent extravasation of circulating T cells across BBB and (or) continuous T cell proliferation in the ischemic brains. To evaluate T cell proliferation, brain T cells were labelled with Ki67 and analyzed by flow cytometry. Compared with splenic T cells, both invading brain CD4+ and CD8+ T cells showed remarkably higher Ki67 labelling, suggesting that they were actively proliferating at one month post ischemic stroke (Figure 4(a)). Immunohistochemistry demonstrated that infiltrating brain CD4+ and CD8+ T cells were predominantly located in the peri-infarct region with extensive reactive astrocyte indicated by strong GFAP staining at one month after ischemic stroke. Fewer CD4+ and CD8+ T cells were found in the normal brain region distal to the infarct lesion where less reactive astrocyte and more MAP2 positive neuron was observed (Figure 4).
Proliferation and localization of invading brain CD4+ and CD8+ T cells at one month after ischemic stroke. (a) Ki67 staining of splenic and brain T cells. Left panel: representative histograms. Right panel: quantitative analysis of Ki67 expressed CD4+ and CD8+ cells in the spleen and brain after ischemic stroke. (b) Representative fluoresce microscopy (5× and 63×) of immunofluorescent staining of CD4+ (green), CD8+ (green) T cells, and reactive astrocytes (GFAP, red) in the ischemic brain at one month after ischemic stroke. Green: CD4+ and CD8+. Blue: DAPI. Red: GFAP. Arrows indicate CD4+ and CD8+ T cells.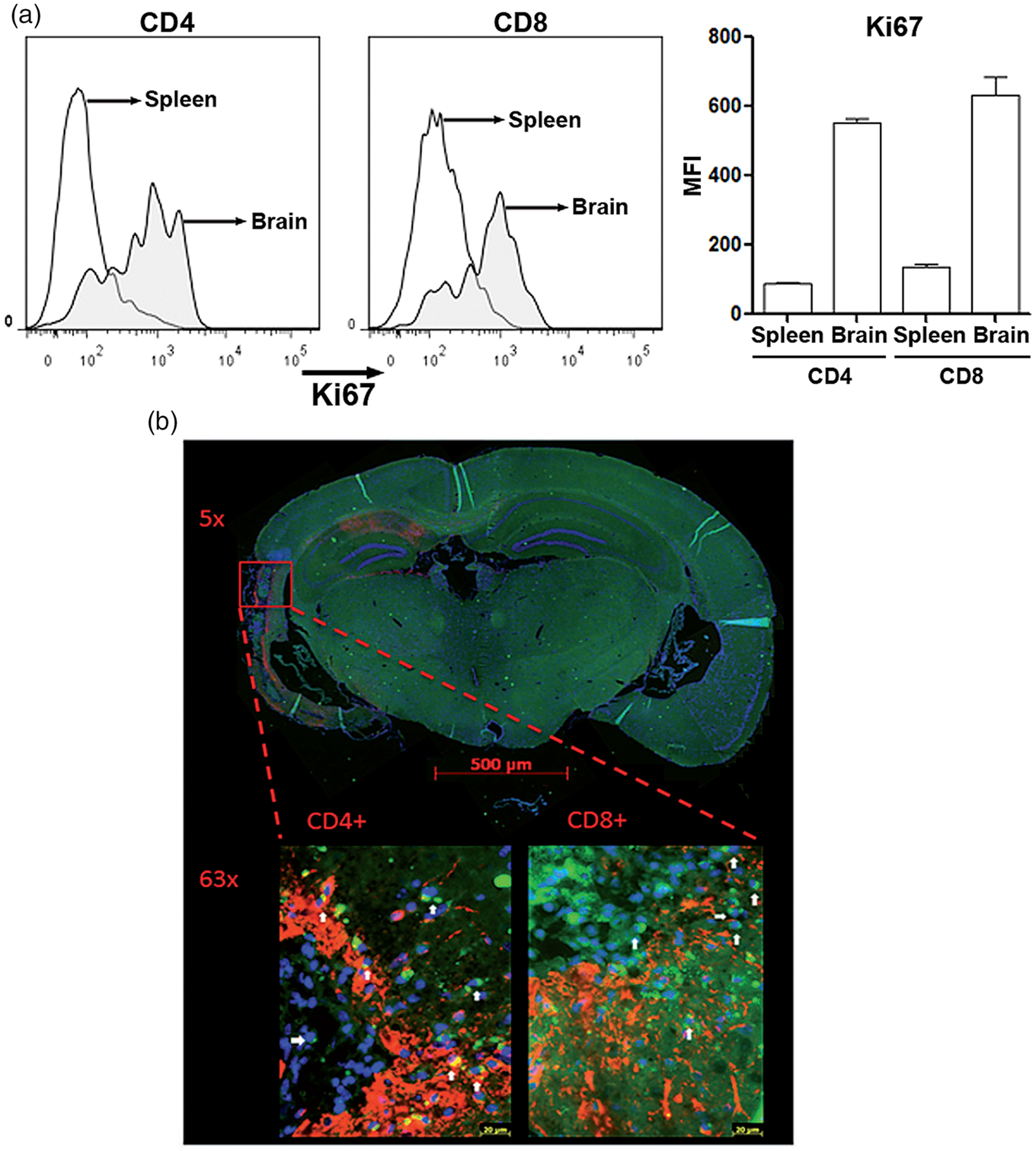
Discussion
Ischemic stroke triggers an adaptive immune response evidenced by infiltration of both innate and adaptive peripheral immune cells into the ischemic brain. The innate immune system is the first line of defense against injury and invading pathogens. The innate immune response is involved in the early phase of post ischemic stroke and includes the invasion and activation of peripheral and brain innate immune cells. Microglial infiltration into the ischemic region has been found to be robust and reach a peak at two to three days in rodent experimental ischemic stroke models.31,32 Similarly, massive increase and proliferation of neutrophils have been found in the infarcted brain tissue within three days following ischemic stroke. 31
In contrast to innate immunity, the adaptive immune system provides a delayed and specific response. Consistently, a delayed and long-term T cell infiltration in the ischemic brain has been found after ischemic stroke8–10 (see Selvaraj and Stowe 11 ). The spleen is one of the major immune cell reservoirs. Mature lymphocytes recirculate continuously between the spleen and blood. During acute infection, there is an exchange of T cells between the spleen and blood, and T cell activation in the blood has been found to reflect the major immunological reactivity that occurs in the spleen. 33 There is increasing evidence that the spleen may mediate the peripheral immune response related to secondary brain damage after ischemic stroke.34,35 In the current study, we compared the T cells phenotype profile between brain invading T cells and spleen T cells after ischemic stroke. Interestingly, a progressive increase of CD4+ and CD8+ T cells was observed in the ischemic hemisphere at up to one month after experimental ischemic stroke. Furthermore, both invading brain CD4+ and CD8+ T cells had remarkably higher Ki67 labelling, suggesting that they were proliferating at one month after ischemic stroke. We determined the function of invading brain T cells after ischemic stroke using T cell activation markers. CD44 is a widely expressed adhesion receptor and its upregulation has been found after activation of naïve T cells. We found an increase of CD44 expression in brain invading CD4+ and CD8+ T cells. CD25, also known as T cell activation antigen, is highly expressed on activated T cells. Consistently, our study demonstrated that the expression of CD25 was increased in brain invading CD4+ and CD4- T cells after ischemic stroke as compared with splenic T cells.
In the current study, we have chosen a transient MCAO mouse model with less brain invading immune cells than permanent MCAO model. 30 However, the permanent MCAO model has a much higher mortality and makes the long-term survival extremely difficult. We were not able to further sort the isolated CD4+ and CD8+ T cells for functional assays given the relative less brain invading T cells in the current model. Nonetheless, our flow cytometry of CD44/CD62L/CD25 indicated that the invading T cells were more active as compared with spleen T cells. In addition, we determined the gene expression profile of pro- and anti-inflammatory cytokines and their corresponding transcription factors in both splenic T cells and brain invading T cells after ischemic stroke. The newly developed RNA isolation technology enables us to recover high-quality total RNA consistently from fewer cells for gene expression analysis. Significant increases of pro-inflammatory cytokines (IFN-γ, IL-17, IL-10, TNFα, perforin) were found in the brain infiltrating CD4+ and CD8+ T cells at three days and one month after ischemic stroke. RORc, T-bet, and FOXP3 are key transcription regulators for the differentiation of Th17, Th1, and Treg cells, respectively.36–38 Significant increase of T-bet and RORc in the brain infiltrating CD4+ T cells was indicated at three days and one month after ischemic stroke, suggesting a Th1 and Th17 lineage differentiation.
The innate immune system plays a crucial role in the initiation and subsequent direction of the adaptive immune response. Reciprocally, adaptive immune cells regulate innate immunity. 39 Temporal spatial pattern of immune cell infiltration at the acute stage after ischemic stroke has been studied using distal permanent MCAO model in mice. Stroke induced BBB breakdown along the vascular tree.40,41 Interestingly, infiltration of different immune cell types seems have different temporal spatial pattern. For example, infiltrating CD3+ T cells have been predominately found in the peri-infarct region, while myeloperoxidase-positive neutrophils were predominately located in the necrotic area and the border of the ischemic zone at the acute phase after ischemic stroke. 8 The predominate localization of CD4+ and CD8+ T cells in the peri-infarct region where reactive astrogliosis developed after ischemic stroke indicating that the infiltrating CD4+ and CD8+ T cells after ischemic stroke may play roles in the repairing process after ischemic stroke.
In summary, the present study has provided novel evidence that ischemic stroke may induce long-term T cells invasion and activation in the brain in an experimental ischemic stroke model. We observed a significant increase of both CD4+ and CD8+ T cells presented in the peri-infarct area at up to one month post ischemic stroke. The invading CD4+ and CD8+ T cells after ischemic stroke demonstrated close interaction with reactive astrocytes and a progressive proinflammatory phenotype evidenced by the increased expression of T cell activation markers, proinflammatory cytokines, and corresponding transcriptional factors. Given the transient MCAO mouse model used in the present study, the number of T cells isolated from the ischemic hemispheres was relatively low which limited further functional analysis of the brain invading T cells after ischemic stroke. Nonetheless, our study indicated that the prolonged activation of brain invading T cells may affect the neural repairing process following cerebral ischemia. Understanding of the crosstalk between the T cells and the brain cells is still rudimentary. Our study warrants future research to determine the interaction between infiltrating brain T cells and the innate CNS immune system which may lead to significant insight for the understanding of neural repairing after ischemic stroke.
Supplemental Material
Supplemental material for Experimental ischemic stroke induces long-term T cell activation in the brain
Supplemental material for Experimental ischemic stroke induces long-term T cell activation in the brain by Luokun Xie, Wenjun Li, Jessica Hersh, Ran Liu and Shao-Hua Yang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by National Institutes of Health grants 1R21NS087209-01A1 (SY) and R01NS088596 (SY). Jessica Hersh was supported by NIH training T32 AG020494.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contribution
XL, WL, RL, and YS conceived and designed the experiments; XL, WL, JH, and RL performed the experiments. XL analyzed the data. XL, JH, RL, and YS wrote and edited the manuscript; all the authors reviewed the manuscript before publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
