Abstract
Chronic kidney disease (CKD) independently increases the risk of stroke and small vessel disease (SVD). This study compared SVD and a transcranial Doppler (TCD)-based marker of intracranial vascular resistance (pulsatility index, PI) in post-stroke patients with and without CKD. Between January 2015 and December 2017, 118 individuals with stable eGFR (50 with CKD) had cerebral MRI and TCD within three months of a stroke. The means of bilateral PI in anterior (anterior cerebral [ACA] and middle cerebral arteries [MCA]) and posterior vessels (posterior cerebral [PCA] and vertebral arteries [VA]) were computed. CKD strongly correlated with higher distal resistance (median CKD ACA PI 1.2, IQR 1.0 to 1.35 vs. controls 0.91 IQR 0.79 to 1.1 [p < 0.0001]; median MCA PI 1.14 IQR 1.03 to 1.39 vs. controls 0.93 IQR 0.79 to 1.1 [p < 0.0001]) and MRI SVD burden (median CKD SVD 4.98 × 104 IQR 2.66 to 7.76 × 104 voxels vs. controls median SVD 6.7 × 103 IQR 2.4 to 24.0 × 103 [p < 0.0001]). In conclusion, in patients with recent stroke, CKD is an independent determinant of increased intracranial vascular resistance in both anterior and posterior cerebral circulations. MRI SVD volume is significantly associated with anterior and posterior circulation PI.
Background
Although chronic kidney disease (CKD) independently increases the risk of stroke and burden of ischemic small vessel disease (SVD), stroke prevention measures in patients with kidney disease are generally similar to those in populations with normal renal function.1–4 Kidney function uniquely impacts the systemic vasculature and extracranial arterial stiffness;5–7 however, little is known regarding the effects of CKD on intracranial hemodynamics.
Transcranial Doppler (TCD) ultrasound is an accurate, non-invasive bedside measurement that assesses cerebral hemodynamic values including distal vascular resistance to flow (pulsatility index, PI). 8 To determine the effect of CKD on intracranial arterial stiffness and therefore PI, this study compared TCD PI indices in the anterior and posterior cerebral circulations in patients with and without CKD after stroke. In addition, impacts of CKD on SVD volumes determined from cerebral MRI were assessed.
Methods
Design
A retrospective review of adult (age >18 years) acute post-stroke patients at the Wake Forest Baptist Medical Center (WFBMC) was performed between January 2015 and December 2017. The study was approved by the WFBMC institutional review board, and was conducted in accordance with the ethical standards stated by the Declaration of Helsinki, 1983. Consent was waived due to the retrospective nature of this study. From a consecutive sample of 636 stroke patients, those who had a TCD ultrasound, brain MRI and stable serum creatinine concentration (≥3 measures over three months using the Jaffe method, including a measurement at three months) within three months of their stroke were included. TCDs recorded <72 h from ischemic stroke were not included due to possible acute phase hemodynamic changes post-stroke. Patients with unstable serum creatinine concentrations (defined as a change of 0.3 mg/dl between stroke admission and three-month follow-up) or with less than three months of available serum creatinine concentrations were excluded (Figure 1). Patients with carotid stenosis (>50% in carotid arteries) or intracranial vascular occlusion were also excluded from this study.
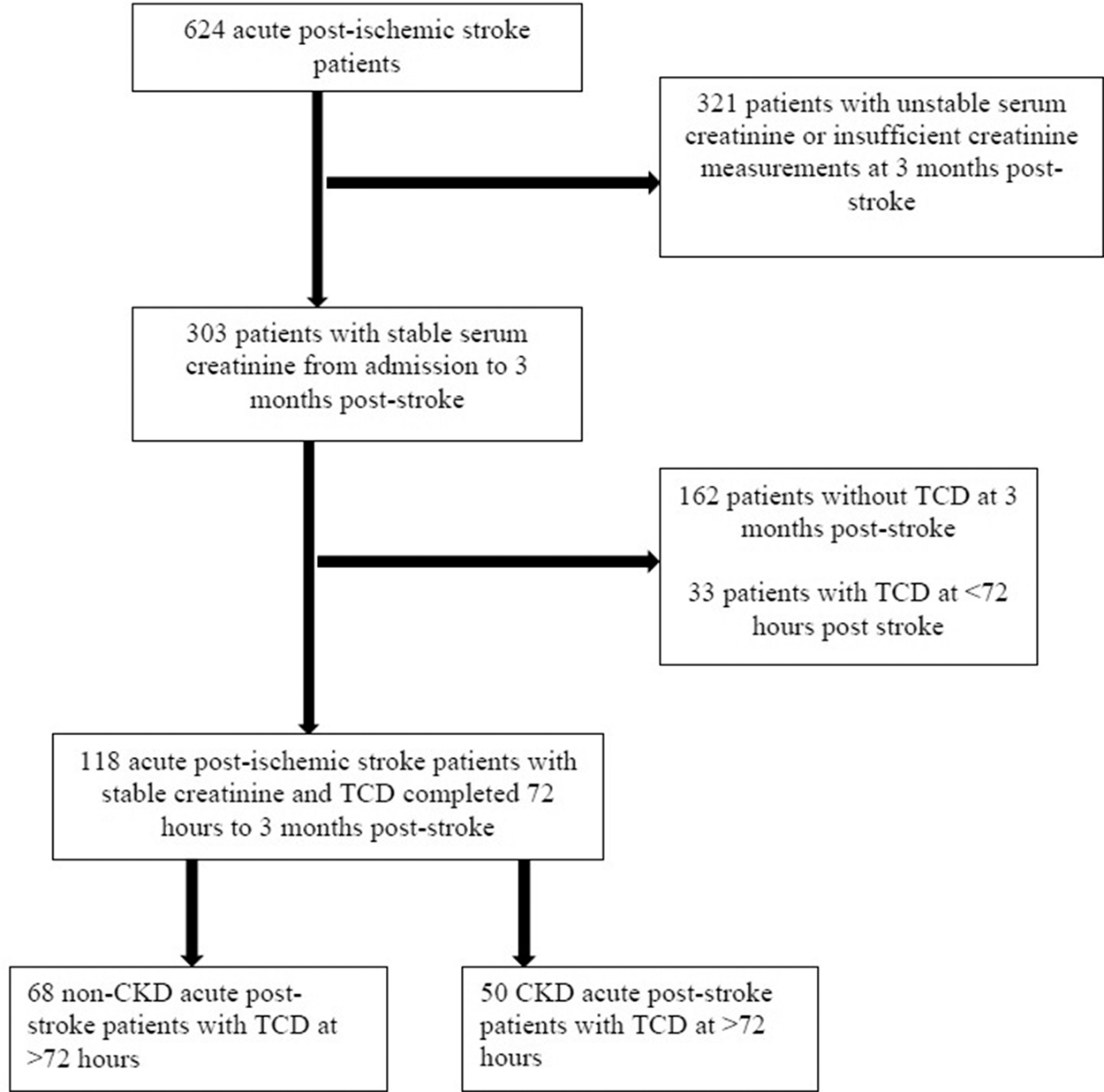
Patient selection of acute post-stroke patients for study cohort.
Measurements
Demographic and clinical data were collected by retrospective chart review. The stage of CKD was determined by the CKD-Epidemiology Collaboration (CKD-EPI) equation to determine the estimated glomerular filtration rate (eGFR). 9 Clinical TCD examination was performed using the transcranial color duplex technique. There was sampling at multiple depths of the MCA, ACA, and PCA by trans-temporal window, and the VA via sub-occipital approach. A zero angle of insonation was used, and the highest PI for each segment was used. TCD PI values were reported as the mean of the bilateral values for the anterior circulation vessels, anterior cerebral arteries (ACA) and middle cerebral arteries (MCA), as well as the posterior circulation, posterior cerebral arteries (PCA) and vertebral arteries (VA).
Statistical analysis
Patients were grouped by the presence or absence of CKD (presence was defined as CKD Stage 3 or greater, eGFR <60 ml/min/1.73 m2). 8 Age was dichotomized to >65 or ≤65 years, as prior TCD population studies have established increased values for PI beyond age 65. SVD volume was calculated using software ITK-SNAP (Version ITK-SNAP 3.8). 10 Statistical analysis was performed using SAS software JMP Pro version 13.0, SAS Institute Inc. Means and medians as appropriate with corresponding standard deviations and interquartile ranges, respectively, were used as measurements of central tendencies. Comparison between groups was done using Fisher’s exact test for proportions. Continuous variables were analyzed using Wilcoxon rank sum test. With TCD PI in each vascular distribution as the dependent variable, multivariate models were constructed to test whether CKD independently increases the PIs. A p value <0.05 was considered statistically significant.
Results
From the total sample of 636 acute stroke patients at WFBMC, 118 had a stable eGFR for >3 months, and had an MRI and TCD study done within three months of their acute stroke. Of these, 50 patients had CKD stage 3 with an eGFR <60 ml/min/1.73 m2. Table 1 displays the demographic, clinical and vascular comorbidities. Patients with CKD were significantly older and had higher prevalence of diabetes and coronary artery disease.
Patient characteristics based on the presence of CKD (eGFR <60 ml/min/1.73 m2).
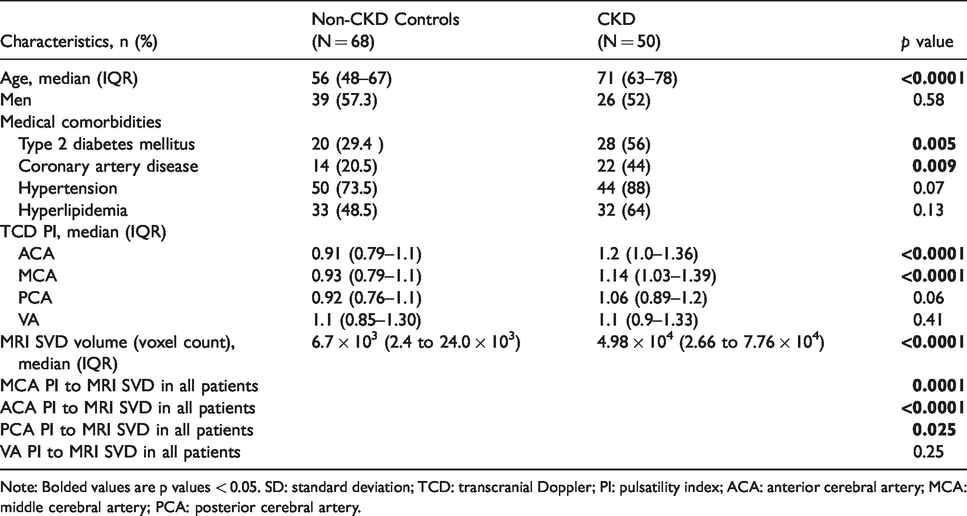
Note: Bolded values are p values < 0.05. SD: standard deviation; TCD: transcranial Doppler; PI: pulsatility index; ACA: anterior cerebral artery; MCA: middle cerebral artery; PCA: posterior cerebral artery.
In the anterior cerebral circulation, the presence of CKD was significantly associated with higher distal vascular resistance (median CKD ACA PI 1.2, IQR 1.0 to 1.36 vs. controls 0.91 IQR 0.79 to 1.1 [p < 0.0001]; median MCA PI 1.14 IQR 1.03 to 1.39 vs. controls 0.93 IQR 0.79 to 1.1 [p < 0.0001]). In the posterior cerebral circulation, the presence of CKD revealed a trend toward higher distal resistance (median CKD PCA PI 1.06 IQR 0.89 to 1.2 vs. controls 0.92 IQR 0.76 to 1.1 [p = 0.06]); however, results were not statistically significant (Table 1, Figure 2).
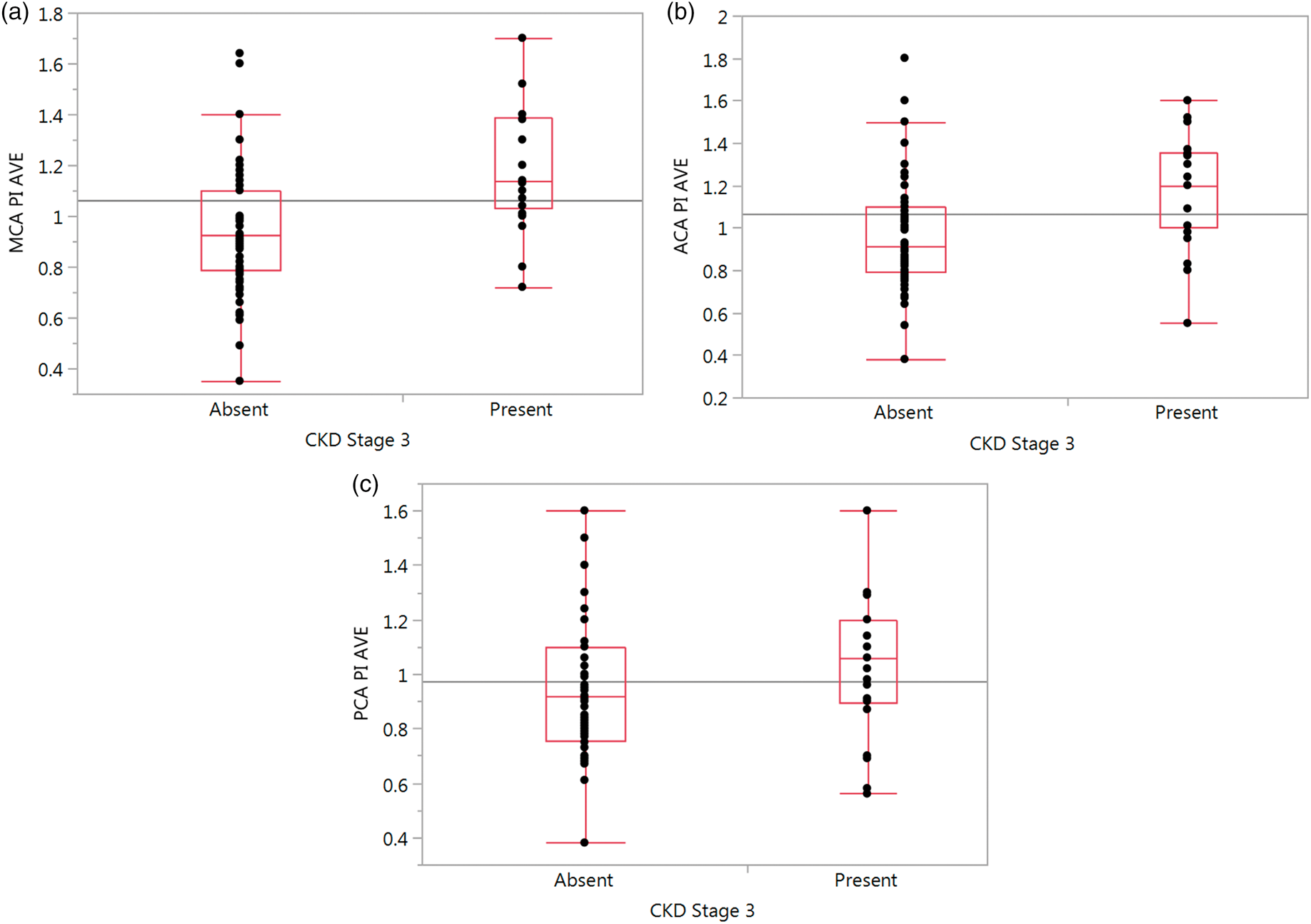
One-way analysis of average cerebral artery PI by presence of CKD ≥3 (EGFR <60) for (a) MCA, (b) ACA, and (c) PCA vessels.
Next we constructed four multivariate models using standard least squares approach with average ACA, MCA, PCA and VA PIs as outcome variables. Risk factors known to affect vascular stiffness and therefore PI, namely age (dichotomized as >65 and ≤65), hypertension, hyperlipidemia, type 2 diabetes, coronary artery disease, and gender were included in the model. In the anterior circulation, CKD was independently associated with higher MCA PIs (p = 0.02) and in the posterior circulation, with higher VA PIs (p = 0.03) CKD did not reach statistical significance as an independent contributor to PI in multivariate models for ACA and PCA vessels (Table 2).
Multivariate model of intracranial vascular resistance, log worth.
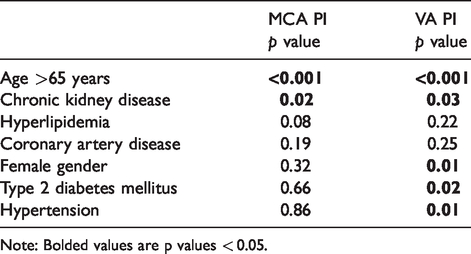
Note: Bolded values are p values < 0.05.
Patients with CKD had a significantly higher SVD lesion burden (median CKD SVD 4.98 × 104 IQR 2.66 to 7.76 × 104 voxels vs. controls median SVD 6.7 × 103 IQR 2.4 to 24.0 × 103 [p < 0.0001]) (Figure 3). This association retained significance in a multivariate model (p < 0.0001) when the following vascular risk factors were included; age (dichotomized as >65 and ≤65), hypertension, hyperlipidemia, type 2 diabetes, coronary artery disease, and gender. In addition, the SVD volume on MRI was significantly associated with anterior circulation distal resistance (MCA PI p = 0.0001, ACA PI p < 0.0001) and with the posterior circulation PCA (PCA PI p = 0.025) using linear regression (Table 1).
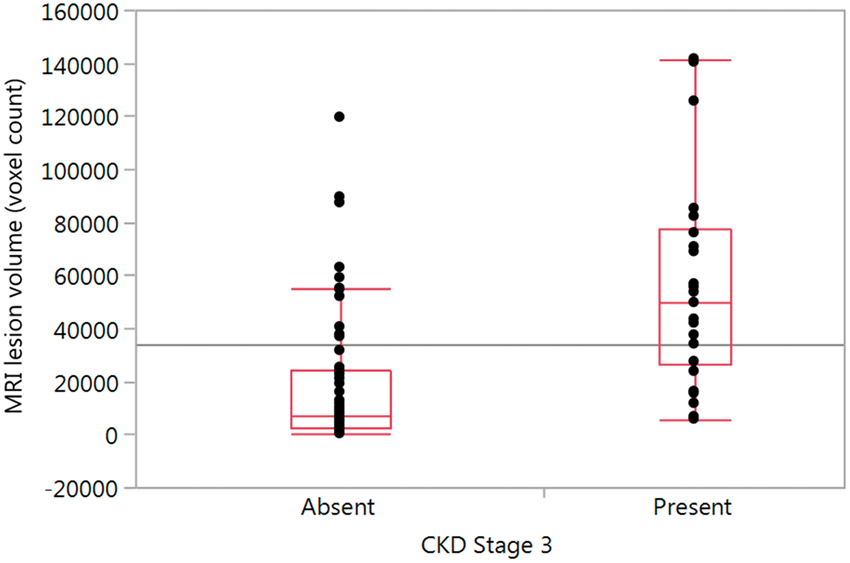
One-way analysis of MRI lesion volume (voxel count) by presence of CKD stage ≥3 (EGFR < 60).
Discussion
In this retrospective study involving patients post-stroke, those with CKD stage 3 or higher were found to have significantly higher burden of SVD lesions and higher distal intracranial resistance in the anterior cerebral circulation, independent of other vascular risk factors. SVD volume on MRI was significantly correlated with TCD-measured distal intracranial resistance.
This is the first TCD study of intracranial cerebral hemodynamics in patients with CKD. It is also the first to suggest a TCD correlate to the known increased SVD lesion burden in patients with CKD. CKD is known to promote endothelial dysfunction, where inflammation, dysfunction of nitric oxide metabolism, and vascular calcification lead to remodeling and stiffening of large arteries.6,7,11,13 Based on results from longitudinal studies, kidney function is known to be an independent determinant of carotid artery and aorta remodeling and stiffening. 6,12 In the Rotterdam study, eGFR was independently associated with lower cerebral blood flow (CBF); each 1 standard deviation reduction in eGFR was associated with a 0.42 ml/min/100 mL lower CBF (95% CI 0.01 to 0.83). 14 The present findings suggest that CKD affects cerebral hemodynamics by increasing resistance to blood flow in the anterior circulation and posterior circulation.
Kidney disease predicts the radiographic presence of cerebral small vessel disease and white matter lesions. 3 Cerebral white matter lesions are likely mediated by endothelial dysfunction, ischemic arteriosclerosis and reduced perfusion. 15 The Northern Manhattan Stroke Study (NOMAS) followed a large number of stroke-free subjects for vascular outcomes. 4 NOMAS showed an eGFR of 15–60 ml/min/1.73 m2 was associated with a higher volume of white matter hyper-intensities, including after adjustment for conventional cardiovascular risk factors. 4 More recently, a subgroup analysis from the Systolic Blood Pressure Intervention Trial (SPRINT) identified increased white matter lesion burden in patients with reduced eGFR (<60 mL/min/1.73 m2) and a high urine albumin-to-creatinine ratio. 16 The results of the present retrospective study support the previously reported impacts of CKD on volume of SVD, and note a correlation between the bedside TCD PI values and SVD lesion burden in patients with and without CKD.
A strength of this study is the application of TCD technology in carefully phenotyped patients after an acute stroke. Limitations include that patients with CKD were older (median age 71 years) and frequently had coronary artery disease and type 2 diabetes mellitus; however, our multivariate models were adjusted for these confounders (Table 2). We also lacked data on smoking status. TCD data obtained <72 h post-stroke were not included in this study due to possible acute-phase hemodynamic changes post-stroke, though the time-course for cerebral hemodynamics to reach a stable baseline post-stroke is as yet unknown. The sample was relatively small, because the study was limited to post-stroke patients. As such, it was not powered to assess the effects of ancestry or co-existing medical comorbidities on TCD values. We also did not stratify based on the stage of CKD, beyond presence versus absence of CKD stage 3 or higher based on eGFR <60 ml/min/1.73 m2. The binary nature of our CKD variable may not reflect patients with borderline eGFR values. Data were cross-sectional and could not address longitudinal relationships between the progression of CKD and changes in cerebral hemodynamics. Future studies are needed to expand the scope of this report by assessing factors such as ancestry and stage of CKD in a larger and younger population affected with CKD.
Non-invasive TCD markers of cerebral hemodynamics in patients with CKD hold great promise as clinically valuable, disease-specific, bedside-screening measures to identify patients at high risk for stroke, as well as to potentially permit modification of risk factors for stroke. Correlations between TCD PI and MRI SVD volume are of importance for the non-invasive assessment of longitudinal trends in SVD burden among patients at risk for stroke.
Summary
Chronic kidney disease independently increases the risk of stroke and the burden of ischemic small vessel disease. In this study, the presence of stage 3 or higher CKD was associated with increased distal resistance in the anterior cerebral circulation of post-stroke patients. Anterior circulation and posterior circulation distal resistive indices also correlated with the degree of SVD on MRI. Prospective TCD studies assessing intracranial PI and MFV changes and white matter disease burdens in larger samples may be helpful for non-invasive screening and informing risk factor modification in patients with CKD at high risk for neurovascular disease.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SG, AS, CT and BIF designed research; SG and JRG performed data collection; SG, JRG, and SVD analyzed data; SG wrote the manuscript; CT and BIF were responsible for editing the manuscript; all authors revised the article critically for important intellectual content.
