Abstract
The relationship between plaque morphology, cerebral micro-embolic signals (MES) and platelet biomarkers in carotid stenosis patients warrants investigation.
We combined data from two prospective, observational studies to assess carotid plaque morphology and relationship with cerebral MES and platelet biomarkers in patients with recently symptomatic (≤4 weeks of transient ischaemic attack (TIA)/ischaemic stroke) versus asymptomatic carotid stenosis. Plaque morphology on ultrasound was graded with Grey-Scale Median (GSM) and Gray–Weale (GW) scoring. Bilateral transcranial Doppler ultrasound classified patients as ‘MES+ve’ or ‘MES-ve’. Full blood counts were analysed and flow cytometry quantified CD62P and CD63 expression, leucocyte-platelet complexes and reticulated platelets.
Data from 42 recently symptomatic carotid stenosis patients were compared with those from 36 asymptomatic patients. There were no differences in median GSM scores between symptomatic and asymptomatic patients (25 vs. 30; P = 0.31) or between MES+ve vs. MES-ve symptomatic patients (36 vs. 25; P = 0.09). Symptomatic patients with GSM-echodense plaques (GSM ≥25) had higher platelet counts (228 vs. 191 × 109/L), neutrophil–platelet (3.3 vs. 2.7%), monocyte–platelet (6.3 vs. 4.55%) and lymphocyte–platelet complexes (2.91 vs. 2.53%) than ‘asymptomatic patients with GSM-echodense plaques’ (P ≤ 0.03).
Recently, symptomatic carotid stenosis patients with ‘GSM-echodense plaques’ have enhanced platelet production/secretion/activation compared with their asymptomatic counterparts. Simultaneous assessment with neurovascular imaging and platelet biomarkers may aid risk-stratification in carotid stenosis.
Keywords
Introduction
Colour Doppler ultrasound (CDUS) is a non-invasive, widely accessible method of evaluating the degree of internal carotid artery (ICA) stenosis. Most decisions regarding intervention for symptomatic and asymptomatic carotid stenosis are heavily influenced by estimation of the severity of carotid stenosis with CDUS, 1 which should ideally be confirmed with another sensitive and specific non-invasive imaging modality (Computed Tomography Angiogram (CTA) or contrast-enhanced MRA). 2 However, the large disparity in the risk of TIA and stroke between patients with symptomatic severe (≥70%) 3 and asymptomatic moderate–severe (60–99%) carotid stenosis on ‘best medical therapy’ alone4–8 is not explained by the severity of carotid stenosis in question and may be influenced by several other factors related to the plaque,9–11 endothelial lining 12 or circulating blood. 12 Therefore, one needs additional information to improve our understanding of the pathogenesis of symptoms in subgroups of patients with recently symptomatic compared with asymptomatic carotid stenosis2,4,6,13 to optimise risk-stratification and prevention strategies in this patient population.
Duplex B-mode ultrasonography has been used to analyse the characteristics and morphology of carotid plaques in vivo to identify potentially ‘higher-risk plaques’ which may be associated with a higher risk of ipsilateral ocular or cerebral hemispheric ischaemic events.14–17 The commonly used Gray–Weale (GW) score categorises plaques into the following types based on visual inspection of the plaque by the operator: (1) predominantly echolucent; (2) substantially echolucent with small areas of echodensity; (3) predominantly echodense with small areas (<25%) of echolucency; (4) uniformly echodense and (5) not accessible because of heavy calcification. 18 The Grey Scale Median (GSM) score uses B-Mode ‘image normalisation and standardisation’ in an attempt to provide a more objective measure of carotid plaque echogenicity than the GW classification system.19,20 This process normalises the grey-scale values of all pixels in the original B-mode image according to specific input and output reference points (i.e. the vessel lumen and adventitia) (Figure 1). The distribution of the grey levels within the plaque is assessed and the median of the normalised grey values is calculated to establish the ‘grey scale median’ of the plaque, thus quantifying plaque echolucency and echodensity. Ultimately, the lower the GSM value, the more echolucent the plaque, with more echodense plaques having higher GSM values. Echolucent GSM scores have been found to correlate with intra-plaque haemorrhage 21 and percentage plaque necrotic lipid core area22,23 on histological examination. However, a limitation of GSM analysis is that plaques with >50% calcification produce acoustic shadowing artefacts which prevent adequate visualisation and assessment of the plaque. 20
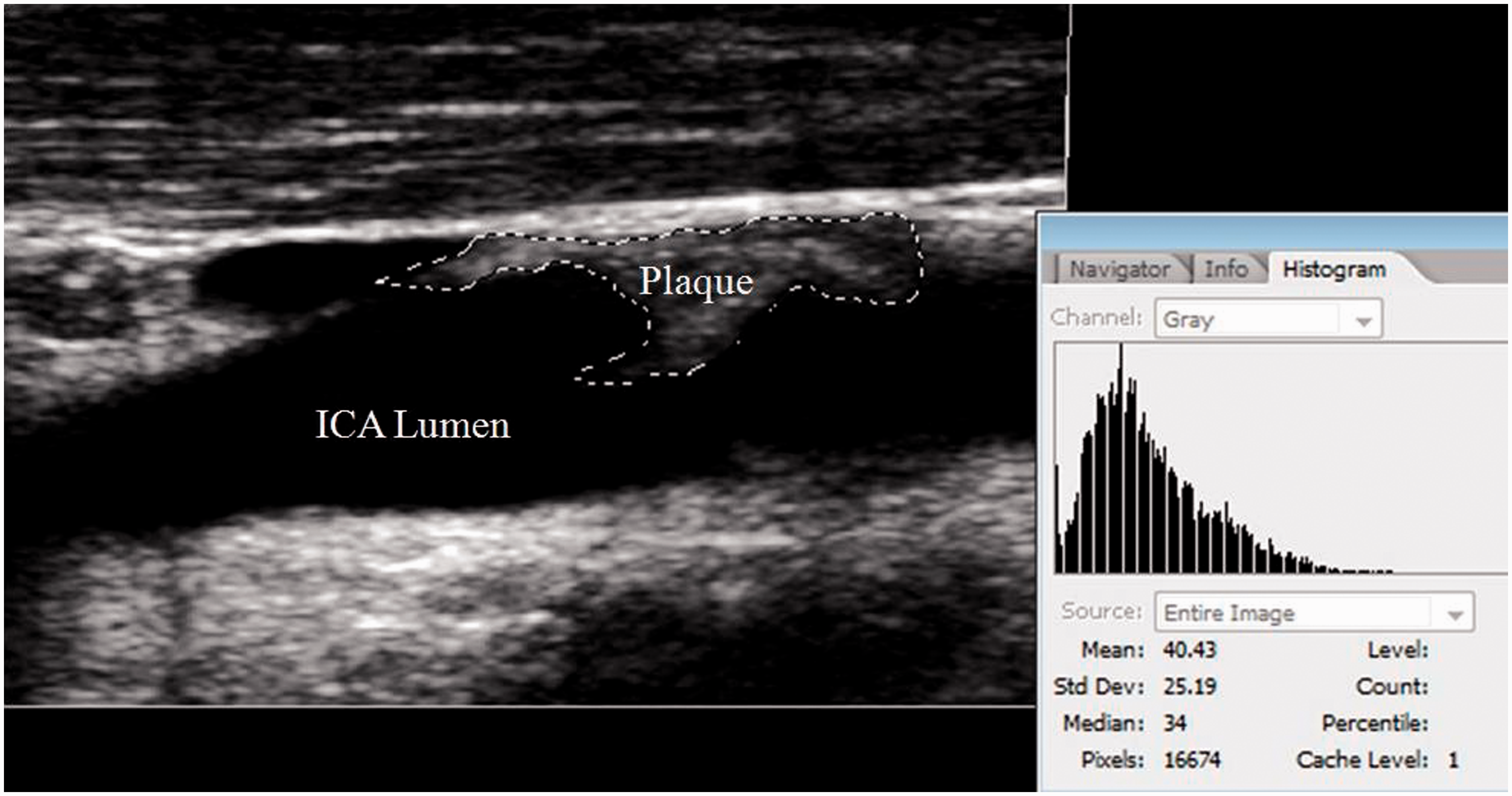
Example of gray-scale median (GSM) analysis of a stenosing, ‘predominantly echodense’ carotid artery plaque with a GSM of 34 using Adobe CS2 software.
Detection of micro-embolic signals (MES) on transcranial Doppler ultrasound (TCD) may identify asymptomatic and symptomatic carotid stenosis patients who may benefit most from optimal medical or surgical treatment to prevent TIA or stroke.6,24 However, there is some controversy in the literature regarding the relationship between plaque echodensity and the risk of MES. Tegos et al. found a weak, but significant inverse correlation between GSM values and the number of MES, with lower GSM values (more echolucent plaques) associated with an increased risk of MES in 80 moderate–severe stenosing plaques (67.5% of patients were asymptomatic; r = −0.22; P = 0.045). 25 Mayor et al. used their own classification system for plaque echolucency and reported that MES-positive patients had a higher proportion of echolucent plaques than MES-negative patients in a mixed asymptomatic (N = 48) and symptomatic (N = 94) patient cohort (P < 0.01). 26 Other studies have not found any correlation between GSM scores and MES positivity in symptomatic carotid stenosis patients (P >0.05).27,28
Prior platelet biomarker studies have revealed increased platelet counts in early29–31 and late30,31 symptomatic compared with asymptomatic moderate–severe carotid stenosis patients. In addition neutrophil–platelet 31 and lymphocyte–platelet complexes29,31 were increased in early symptomatic compared with asymptomatic patients. In the subgroups of patients with severe (>70%) stenosis, symptomatic patients had increased platelet counts in the early29–32 and late30,31,33 phases and increased neutrophil–platelet, 33 monocyte–platelet 33 and lymphocyte–platelet29,33 complexes in the early phase after TIA/stroke compared with asymptomatic patients. Interaction between platelets and exposed substrates from a recently ruptured plaque may enhance platelet activation and thrombus formation, as seen in patients with coronary artery disease.31,34 Furthermore, excessive platelet activation could promote carotid plaque instability itself via enhanced cell adhesion and smooth muscle cell proliferation, coagulation and proteolysis, as well as via synthesis of chemokines and pro-inflammatory cytokines, all of which enhance the inflammatory process to promote plaque development.31,34
Simultaneous evaluation of extracranial carotid plaque characteristics on Doppler ultrasound, TCD examination to assess MES status in vivo, in combination with assessment of platelet biomarkers ex vivo has the potential to enhance our understanding of the pathogenesis of symptoms in patients with carotid stenosis and assess response to treatment. 35 However, to our knowledge, no study has previously simultaneously assessed all three criteria in recently symptomatic vs. asymptomatic carotid stenosis.
Therefore, the aims of our study were to determine whether:
There were differences in atherosclerotic carotid plaque morphology on CDUS between highly clinically characterised patients with recently symptomatic and asymptomatic moderate–severe (50–99%) carotid stenosis; The incidence of MES was higher in recently symptomatic compared with asymptomatic patients with ‘analysable carotid plaque ultrasound data’; MES on TCD were more likely to be observed in association with more echolucent than more echodense plaques. Plaque echodensity on ultrasound influenced markers of platelet activation or turnover.
We hypothesised that:
Atherosclerotic plaques in patients with recently symptomatic carotid stenosis would be more likely to be echolucent on CDUS than in patients with asymptomatic moderate–severe carotid stenosis; MES would be more common in recently symptomatic than asymptomatic patients with analysable plaque ultrasound data; MES would be more common in recently symptomatic patients with more echolucent than those with more echodense plaques on CDUS; Platelet activation or turnover markers would be influenced by plaque echodensity.
Materials and methods
Consecutive eligible patients >18 years old with recently symptomatic or asymptomatic moderate or severe carotid artery stenosis identified on CDUS using standardised velocity criteria36,37 who were recruited to the Platelets and Carotid Stenosis (PACS) 29 or HaEmostasis In carotid STenosis (HEIST) studies 30 were screened for inclusion in this prospectively planned, multicentre observational study. Only patients who had both complete CDUS and TCD data available were included in this analysis (see below). Patients were recruited from the Rapid Access Stroke Prevention clinics, vascular surgery and general neurology clinics, stroke service, and neurology and vascular surgery wards at AMNCH/Tallaght University Hospital or St James’s Hospital.
The study was approved by the St James’s Hospital/AMNCH–Tallaght University Hospital Ethics Committee (Project/LREC Reference: 2011/31/02) under the St James’s Hospital/AMNCH–Tallaght University Hospital Ethics Committee guidelines on human experimentation. Written informed consent (or ‘proxy consent’ from a relative, where appropriate) was obtained in all cases.
Inclusion/exclusion criteria
Asymptomatic patients
Patients were included in the ‘asymptomatic carotid stenosis group’ if they were incidentally noted to have moderate (≥50–69%) or severe (≥70%) carotid stenosis on screening CDUS, and had never had TIA or stroke symptoms or had no history of TIA or stroke in the preceding 3 years.30,31
Symptomatic patients
Patients were included in the ‘recently symptomatic carotid stenosis group’ in this combined analysis if they had experienced a TIA or ischaemic stroke in the vascular territory supplied by a moderate (≥50–69%) or severe (≥70–99%) ipsilateral carotid artery stenosis within the preceding four weeks.30,31 The diagnosis of stroke and TIA was defined on clinical grounds, 38 as the design of these studies predated the ‘tissue-based’ definitions of TIA and stroke proposed by the American Heart Association/American Stroke Association. 39 A stroke was ‘clinically defined’ as a neurological deficit of sudden onset, with focal cerebral or ocular rather than global neurological dysfunction, with symptoms lasting for more than 24 h or resulting in death before 24 h, and in which, after adequate investigation, symptoms were presumed to be of non-traumatic vascular origin’. 38 A TIA was also ‘clinically defined’ rather than ‘tissue-defined’ as one of the above clinical syndromes, with symptoms lasting for <24 h, regardless of whether there was evidence of acute infarction on neuroimaging and neurovascular work-up. Before inclusion, all other potential ‘non-carotid sources’ of TIA and stroke were ruled out, so that all met strict criteria for ‘large artery atherosclerotic TIA/ischaemic stroke’ according to the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) classification. 40
Exclusion criteria included ipsilateral carotid occlusion, active infection, inflammation or neoplasia; platelet count <120 or >450 × 109/L; myocardial infarction, pulmonary embolism, deep vein thrombosis or major surgery within the preceding three months; prior primary intracerebral haemorrhage; known bleeding or clotting diathesis; ongoing unstable coronary or peripheral arterial disease; renal impairment (e.g. urea >10 mmol/L); or nonsteroidal anti-inflammatory drug (NSAID) intake other than aspirin within the preceding two weeks. Patients who were initially recruited were also subsequently excluded from the symptomatic group if there was evidence of a potential cardio-embolic source of embolism detected within three months of recruitment e.g. paroxysmal atrial fibrillation.30,31 Patients with suboptimal plaque imaging or heavy calcification on CDUS were excluded from this combined analysis.
Clinical assessment
As described previously, all patients underwent detailed neurovascular assessment by a neurology research registrar or supervising consultant vascular neurologist (DJHM) to confirm that asymptomatic patients met inclusion criteria, and to confirm a diagnosis of large artery atherosclerotic TIA or ischaemic stroke in the symptomatic cohort.30,31 Information regarding vascular risk factors, including hypertension, prior TIA or stroke, ischaemic heart disease, atrial fibrillation, valvular heart disease, diabetes mellitus, hyperlipidaemia, peripheral vascular disease, migraine, family history of stroke, medication intake (including anti-thrombotic therapy), smoking status, alcohol intake, illicit substance intake and the method of detection of carotid stenosis was collected prospectively. If antiplatelet therapy was altered by their treating physician in the early phase after presentation, patients were invited to undergo repeat blood testing approximately 14 days later if they had not undergone carotid intervention by that stage. Details regarding antiplatelet regimens, dose and duration of therapy were recorded. Results of routine haematological (full blood count (FBC)), coagulation (Prothrombin Time/Activated Partial Thromboplastin time), biochemical (renal and liver profiles) and blood glucose testing were collected prospectively. CT and/or MRI brain were performed in all symptomatic patients. A chest radiograph, electrocardiograph (ECG), 24-h ECG/Holter recording and transthoracic or trans-esophageal echocardiography were obtained in all symptomatic patients.30,31
CDUS examination
High-resolution colour Doppler and B-mode ultrasonography of the cervical arteries was performed by highly experienced vascular technologists on site within one week of recruitment to the studies in the majority of recently symptomatic patients, with the remainder assessed up to one month from symptom onset. Asymptomatic patients were assessed with CDUS of the cervical vessels within three months of recruitment in 81% of cases; identification of stenosis occurred up to six months before recruitment in 7/36 (19%) patients, but all were confirmed to be clinically asymptomatic by the time of recruitment.
The vascular technologists followed a standardised protocol on a Siemens Acuson Antares™, and used a 7.5 MHz linear-array transducer with a maximum dynamic range of 60 dB. Both CCAs and ICAs were imaged with patients lying supine and comfortable. The carotid bifurcation was imaged in longitudinal (anterolateral, lateral and posterolateral) and transverse projections. For this study, we selected patients with ‘at least moderately-stenosing plaques’, defined as localised protrusions of the vessel wall into the lumen that narrowed the lumen by ≥50% based on the velocity criteria outlined below. Patients were classified as having 50–69% ICA stenosis if the peak systolic velocity in the ICA (PSVICA) was ≥130 cm/s but <230 cm/s, the end diastolic velocity in the ICA (EDVICA) was <110 cm/s, and the ratio of the PSVICA to the PSV in the common carotid artery (PSVCCA) was <4.0. ICA stenosis severity was considered to be ≥70% if the PSVICA was ≥230 cm/s, the EDVICA was ≥110 cm/s, or the PSVICA:PSVCCA ratio was ≥4.0. 37 Of note, 38 of 42 (91%) recently symptomatic patients had another concurrent, non-invasive angiographic imaging modality performed in addition to CDUS (either extracranial MRA or CTA) to assess for concordance regarding the degree of carotid stenosis with the CDUS assessment. MRA or CTA was also performed in 19 of 36 (53%) asymptomatic patients.
Evaluation of plaque morphology on B-mode ultrasound
GSM and GW analysis
Gain settings during B-mode imaging were adjusted to allow for adequate visualisation of the carotid plaque and to minimise noise within the vessel lumen, so that structural details of the ‘far wall’ media–adventitia interface was clear. This ensured the availability of two reference structures for image normalisation, i.e. the ‘noiseless’ blood in the vessel lumen, and the echodense area of adventitia in the vicinity of the plaque. Plaque GSM analysis was performed in our lab using Adobe Photoshop CS2 software where >50% of the plaque area had analysable imaging information that was not obscured by acoustic shadowing from calcification. The number of pixels in the area of interest had to constitute over half the total number of pixels in the area of the plaque, but only the clearly visualised area was subjected to analysis. B-mode images were digitised, transferred to a personal computer for later analysis with the Adobe Photoshop CS2 software. Colour was eliminated by converting the images to grey scale using the ‘grey-scale’ software function. The ‘histogram facility’ and the ‘levels function’ on the software were employed to standardise the image for each plaque, and the GSM of two reference points was defined: (i) The GSM of the ‘noiseless’ blood in the centre of the carotid lumen was normalised to an ‘input’ value of 0–5, equivalent to ‘maximal echolucency’ and (ii) the GSM of the echogenic adventitia within the plane of imaging adjacent to the target plaque was normalised to an ‘output’ value of 185–195, equivalent to ‘maximal echodensity’.
The standardised images were analysed off-line by experienced, trained observers who calculated the GSM by consensus agreement after all data had been collected (JAK and CD for the PACS study; SJXM and JK for the HEIST study). Using a GSM ‘cut point’ of ≤25 for the definition of ‘plaque echolucency’ proposed by Varetto et al., patients were subsequently divided into ‘GSM echolucent’ (GSM ≤25) and ‘GSM-echodense’ (GSM >25) subgroups. 41 Analysis was performed blinded to symptomatic status, MES status and platelet biomarker status. The same trained observers classified plaques according to the GW score (types 1–5). 18 Results were simultaneously recorded and entered onto a database at the time each plaque was scored; formal statistical analysis was performed later (see below).
Transcranial Doppler ultrasound
Bilateral simultaneous 1-h TCD recordings of the middle cerebral arteries were performed with a Viassys Pioneer TC8080, as described previously around the time of study recruitment and blood sampling.29–31 As reported previously, inter-observer agreement regarding the presence of MES versus artefact between two of the authors who analysed a representative section of data from the HEIST study (SJXM and JK), blinded to clinical details, symptomatic status and recorded MES status, was ‘excellent’ (97.4% concordance; Cohen’s unweighted kappa statistic 0.95). 30 All remaining TCD data analysis was performed locally and independently by one of these two authors. Symptomatic patients were classified as ‘MES+ve’ if they had ≥1 MES detected ipsilateral to the stenosed carotid artery of interest. Asymptomatic patients were considered ‘MES+ve’ if ≥1 MES was detected ipsilateral to an asymptomatic ≥50% carotid stenosis. Other patients were deemed ‘MES-ve’.30,31
Platelet biomarkers
All subjects were rested for at least 20 min, and venepuncture performed from a free-flowing vein using a 21G butterfly needle and a Vacutainer® system with a luer adaptor, as previously described.30,42,43 A FBC was measured in Ethylenediaminetetraacetic acid (EDTA) Vacutainer® tubes between 2 and 4 h after venepuncture on a Sysmex XE-2100™ haematology analyser (Sysmex UK Ltd.). 30 Platelet activation was assessed with whole blood flow cytometry (Beckman-Coulter EPICS XL-MCL™) to quantify platelet surface activation marker expression (CD62P and CD63) within 90 min of venepuncture, 42 and the percentages of circulating neutrophil–platelet, monocyte–platelet, and lymphocyte–platelet complexes within 3 h of venepuncture using previously described 44 and validated methodology.30,32,42,43 Reticulated platelets were similarly quantified via labelling with Thiazole Orange (Retic-COUNT™, Becton Dickinson, San Jose, USA), as previously described. 30
Statistical methods
The distribution of all variables was tested for normality with the Anderson–Darling test of normality. Paired or unpaired t-tests were used for comparison of paired and unpaired parametric variables, respectively. The Wilcoxon signed rank test and Wilcoxon rank sum test were used for comparison of paired and unpaired non-parametric variables, and the Kruskal–Wallis rank sum test for comparison of multiple non-parametric variables, where appropriate. All statistical calculations were performed using Minitab 16® (Minitab 2015). P < 0.05 was considered statistically significant.
Results
Forty-two recently symptomatic and 36 asymptomatic patients with moderate–severe ICA stenosis had complete CDUS and TCD data available for analysis from both the PACS and HEIST studies. The demographic and vascular risk factor profiles of included patients are presented in Tables 1 to 3. There was a higher proportion of active smokers in the recently symptomatic compared with the asymptomatic group; otherwise, there were no significant differences between groups (P >0.05). Four asymptomatic patients had previously had an ipsilateral carotid endarterectomy, with a mean interval between surgery and recruitment of 5.25 years (range: 3–8 years). Of note, there were no significant differences in prescribed anti-platelet regimens between groups overall (Table 2). Furthermore, there were no significant differences in prescribed antiplatelet regimens between MES+ve and MES-ve symptomatic patients (Table 3). Within the symptomatic MES-ve subgroup, antiplatelet monotherapy with aspirin or clopidogrel was more commonly prescribed than aspirin–clopidogrel or aspirin–dipyridamole combination therapy (P = 0.03; Table 3).
Demographic and vascular risk factor profiles of patients.
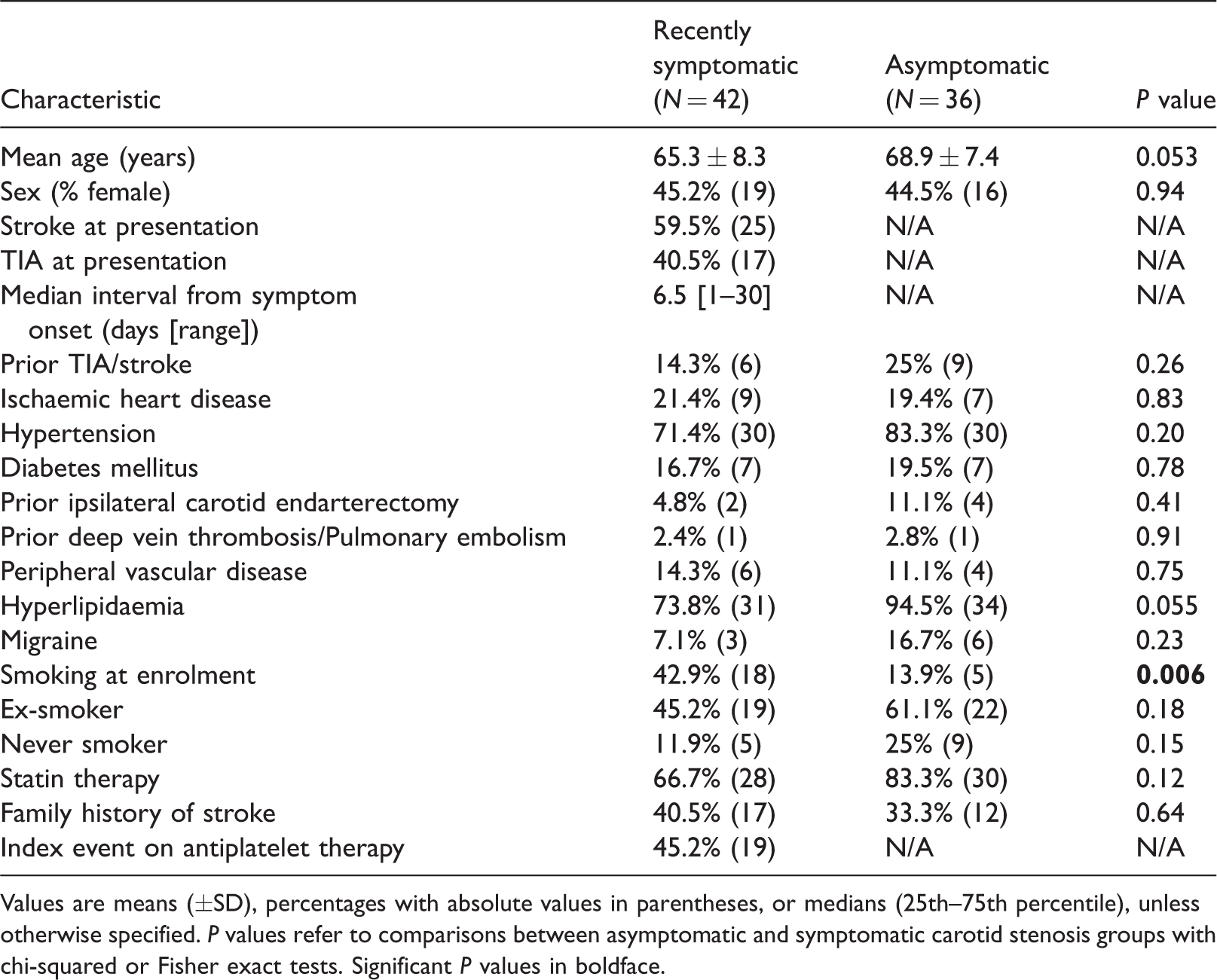
Values are means (±SD), percentages with absolute values in parentheses, or medians (25th–75th percentile), unless otherwise specified. P values refer to comparisons between asymptomatic and symptomatic carotid stenosis groups with chi-squared or Fisher exact tests. Significant P values in boldface.
Degree of stenosis, prescribed antiplatelet regimens and percentage of patients with micro-embolic signals (MES) on transcranial Doppler ultrasound (TCD).
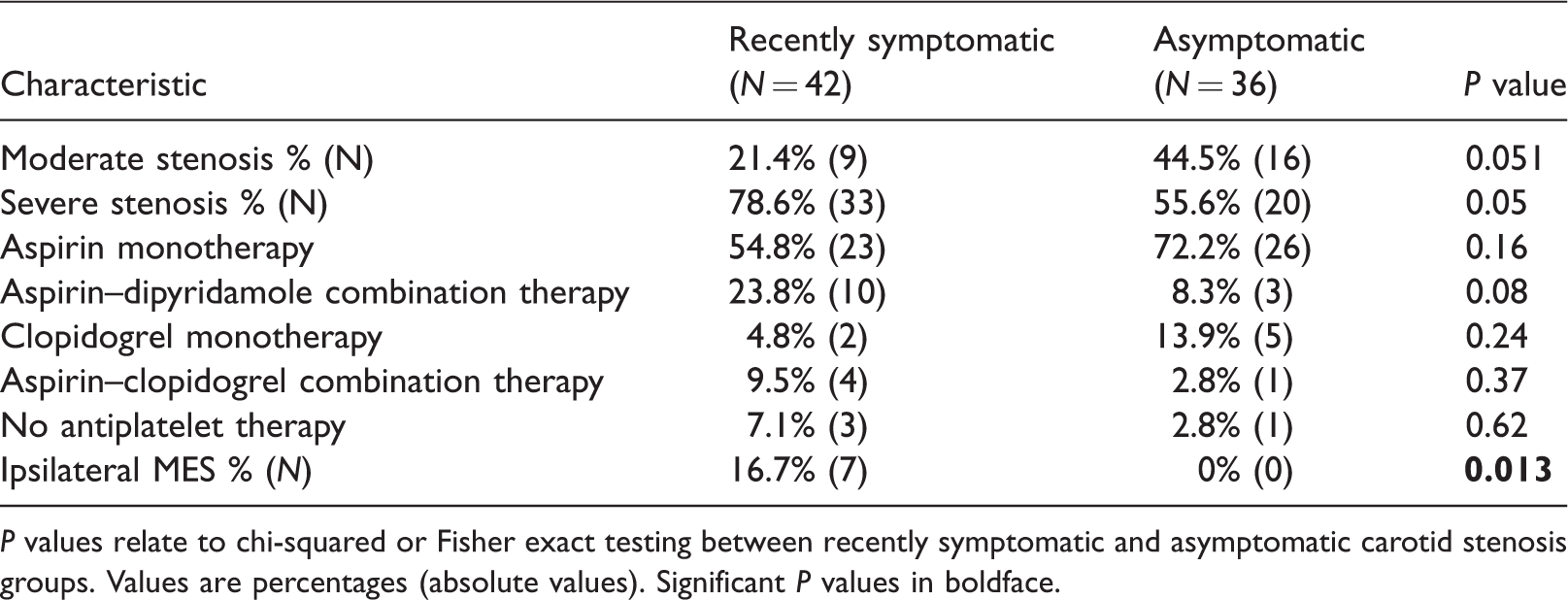
P values relate to chi-squared or Fisher exact testing between recently symptomatic and asymptomatic carotid stenosis groups. Values are percentages (absolute values). Significant P values in boldface.
Prescribed antiplatelet regimens in symptomatic patients who were MES+ve and MES-ve.

P values refer to the comparisons of the proportion of MES+ve vs. MES-ve patients who were on no antiplatelet agents, aspirin or clopidogrel monotherapy, or aspirin–clopidogrel or aspirin–dipyridamole combination therapy. *P value refers to the comparison of the proportions of patients who were prescribed antiplatelet monotherapy vs. combination therapy within the individual MES+ve and MES-ve subgroups. Significant P values in boldface.
Plaque morphology on CDUS
There were no significant differences in median GSM scores between recently symptomatic and asymptomatic carotid stenosis patients (25 vs. 30; P = 0.31), or in patients sub-categorised into moderate (50–69%) or severe (70–99%) stenosis (Table 4). Symptomatic patients who presented with a qualifying TIA did not have significantly different GSM scores to those who presented with an ischaemic stroke (20 vs. 29; P = 0.283). Using a GSM ‘cut-point’ of ≤25 to differentiate GSM-echolucent patients (GSM ≤ 25) from GSM-echodense (GSM >25) patients, 41 there were no significant differences in the proportions of recently symptomatic vs. asymptomatic patients who had echolucent or echodense plaques. Furthermore, there were no differences between symptomatic and asymptomatic groups overall in the proportions of patients in each GW scoring category (Table 5).
Ultrasound characteristics of patients with Gray-Scale Median (GSM) scores, including in those who were MES+ve and MES-ve.
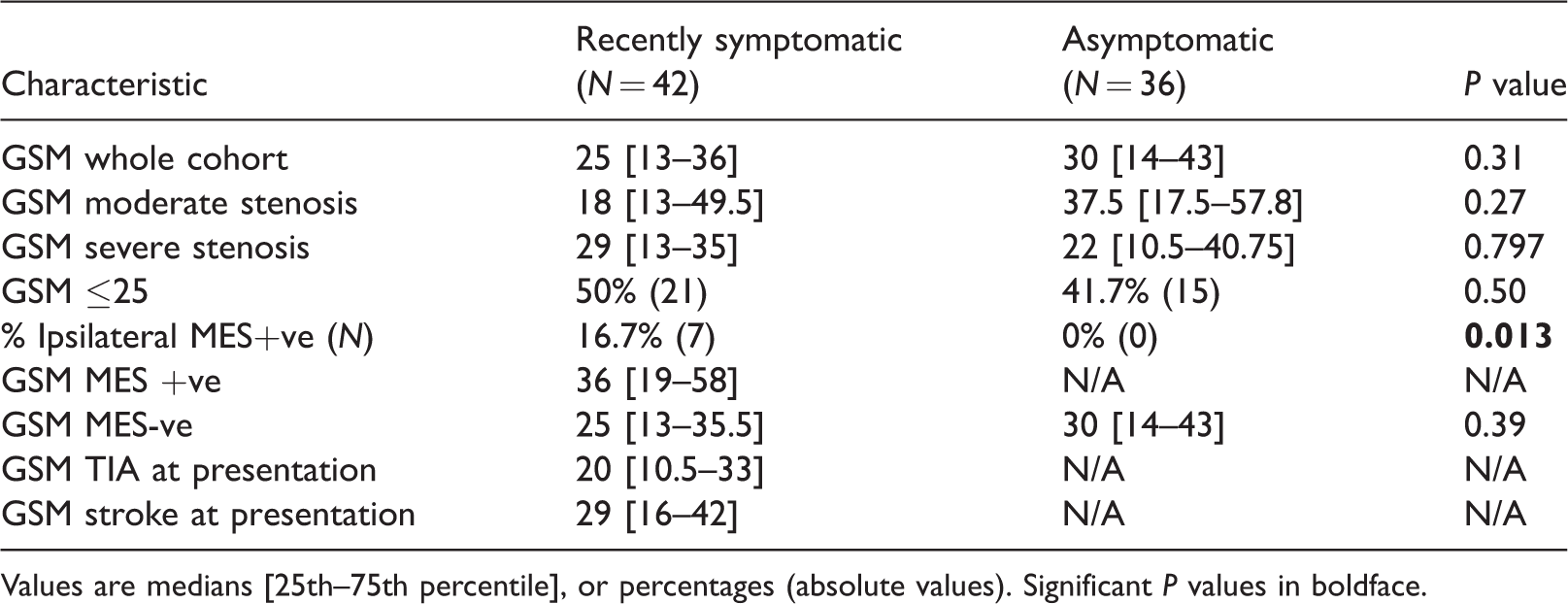
Values are medians [25th–75th percentile], or percentages (absolute values). Significant P values in boldface.
Proportions of patients with different Gray–Weale (GW) scores, categorised according to symptomatic status, stenosis severity, with subgroup data from MES-ve patients.
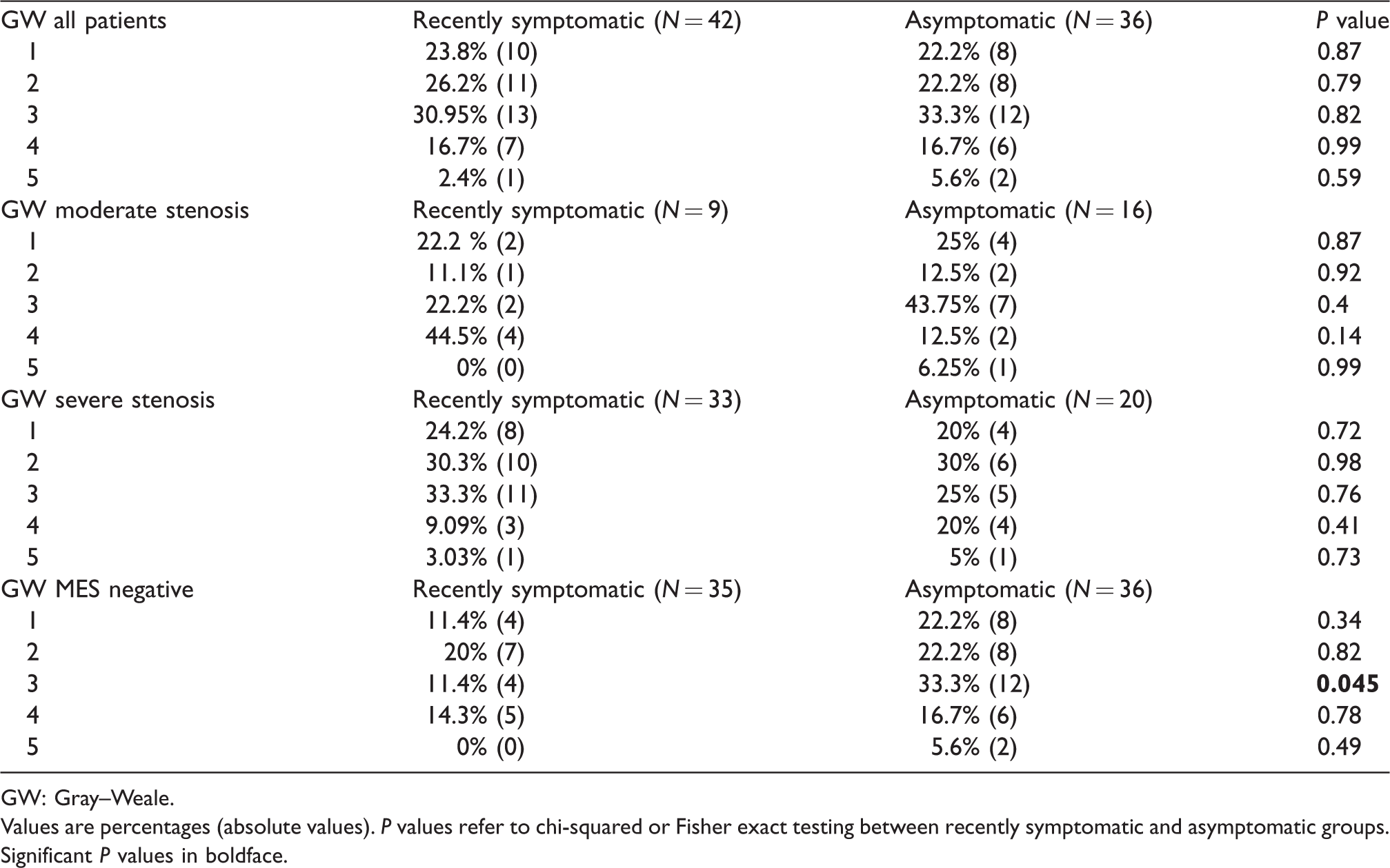
GW: Gray–Weale.
Values are percentages (absolute values). P values refer to chi-squared or Fisher exact testing between recently symptomatic and asymptomatic groups. Significant P values in boldface.
TCD data and relationship with plaque morphology
MES were more frequently observed in recently symptomatic than asymptomatic patients who had complete CDUS and TCD data available for analysis (16.7% (7/42) vs. 0% (0/36); P = 0.01, Table 2).
There were no significant differences in median GSM scores between symptomatic MES+ve vs. MES-ve patients (36 vs. 25; P = 0.09, Table 4). Within the MES-ve subgroup, recently symptomatic patients were less likely to have a type 3 GW plaque than their asymptomatic counterparts i.e. predominantly echodense with <25% echolucency; (P = 0.045), but there were no other differences in GW plaque morphology between these symptomatic and asymptomatic subgroups (Table 5).
Relationship between GSM–plaque morphology and platelet biomarkers
Pre-planned subgroup analyses were performed to compare GSM-echolucent patients (GSM ≤ 25) with GSM-echodense (GSM >25) patients 41 within the symptomatic and asymptomatic groups initially, and then between respective symptomatic and asymptomatic subgroups (Table 6). There were no differences in the proportions of patients with severe (≥70–99%) stenosis between echodense and echolucent symptomatic (76% vs. 81%; P = 0.76) or between echodense and echolucent asymptomatic patients (73% vs. 43%; P = 0.096). There were no differences in the proportions of echodense symptomatic vs. asymptomatic patients (P = 0.85). Severe stenosis was more frequent in echolucent symptomatic than echolucent asymptomatic patients (81% vs. 43%; P = 0.006).
Platelet biomarkers in recently symptomatic and asymptomatic patients.
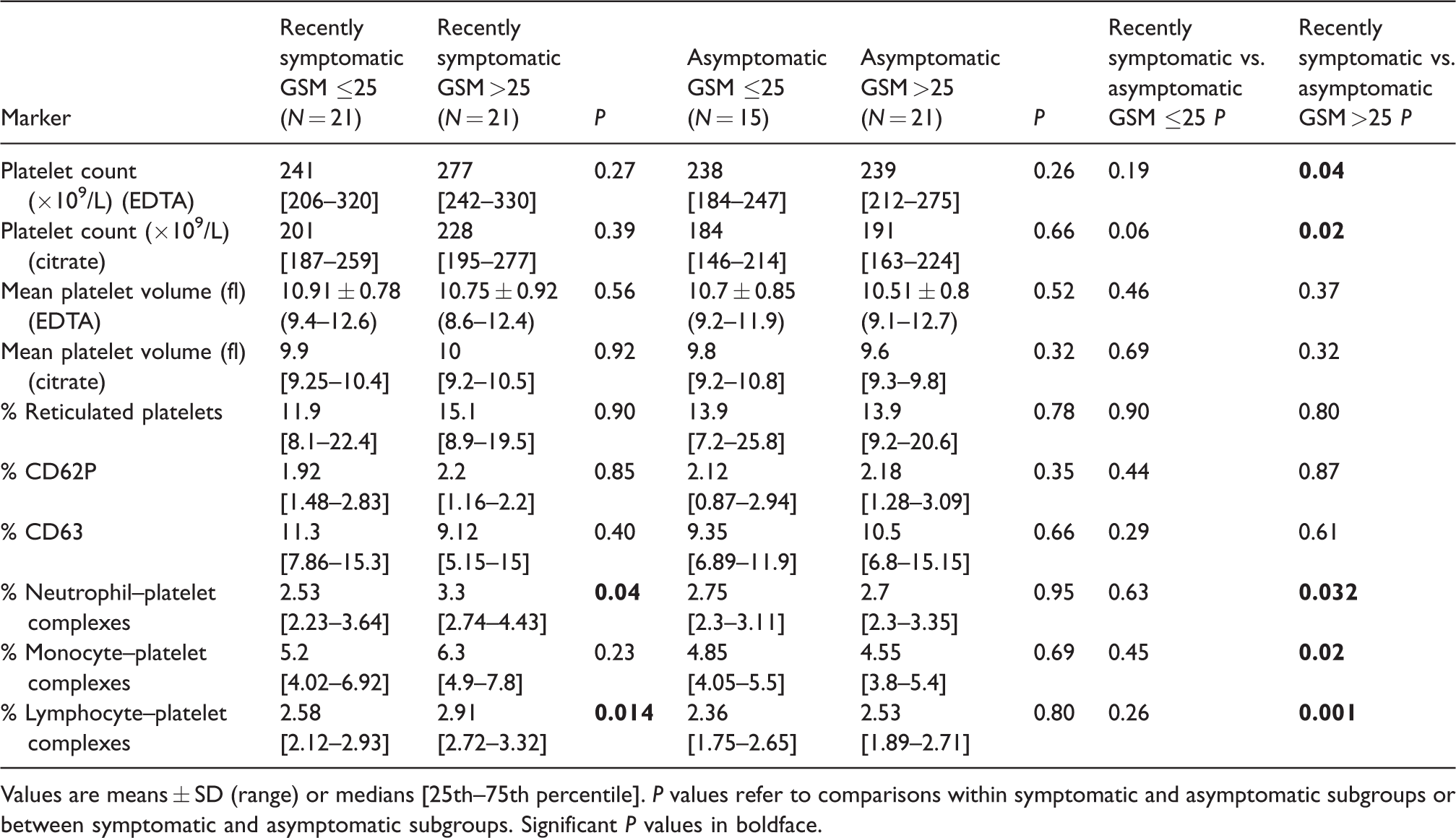
Values are means ± SD (range) or medians [25th–75th percentile]. P values refer to comparisons within symptomatic and asymptomatic subgroups or between symptomatic and asymptomatic subgroups. Significant P values in boldface.
Within the symptomatic group, echodense patients had increased circulating neutrophil–platelet complexes (3.3 vs. 2.53%; P = 0.04) and lymphocyte–platelet complexes (2.91 vs. 2.58%; P = 0.01) compared with echolucent symptomatic patients (Table 6). Platelet counts were higher in EDTA (277 vs. 239 × 109/L; P = 0.04) and citrate (228 vs. 191 × 109/L; P = 0.02), and neutrophil–platelet complexes (3.3 vs. 2.7%; P = 0.03), monocyte–platelet complexes (6.3 vs. 4.55%; P = 0.02) and lymphocyte–platelet complexes (2.91 vs. 2.53%; P = 0.001) were increased in recently symptomatic compared with asymptomatic echodense patients (Table 6). There were no significant differences in the percentage of reticulated platelets between echodense or echolucent symptomatic or asymptomatic patients (P >0.05) (Table 6).
Discussion
This innovative pilot study which simultaneously assessed patients with both the GSM and GW scoring systems in a highly phenotyped carotid stenosis population did not show any significant differences in ‘plaque echodensity’ between early symptomatic and asymptomatic moderate–severe carotid stenosis patients overall. These data are in keeping with prior studies on this topic28,45,46 but are not in agreement with others.21,41,47,48
MES were more common in recently symptomatic than asymptomatic patients with complete ultrasound and TCD data, consistent with prior data from our group and others.12,29,49–51 This study confirms that the presence of MES on TCD is a reliable ‘neuroradiological biomarker’ which can partly explain the higher risk of TIA or stroke in symptomatic compared with asymptomatic patients in vivo. Contrary to one of our a priori hypotheses, we did not confirm that MES+ve patients had more echolucent plaques than MES-ve patients, with no suggestion of even a trend to support this hypothesis. The finding that early symptomatic MES-ve patients were less likely to have a type 3 GW plaque (predominantly echodense with small areas of echolucency) than their asymptomatic MES-ve counterparts just reached statistical significance. Because there were no other significant differences in GW categories between any of the MES-ve subgroups, this result may reflect a type I error.
Although Tegos et al. reported a significant correlation between GSM scores and MES count (r = −0.22; P = 0.045), they identified a very high proportion of patients with MES positivity, with 61.5% of symptomatic and 37% of asymptomatic patients noted to be MES+ve after only a 30 min recording. Oikawa et al. found that average GSM scores did not differ between groups with and without MES in 70 patients with ≥70% carotid stenosis (21% asymptomatic) (P = 0.15); 52 in total, 27% had MES detected on intra-operative TCD. Kolkert et al. performed intra-operative monitoring prior to carotid clamping in 96 patients (12 asymptomatic) with 50–99% stenosis and found no difference in GSM between symptomatic and asymptomatic patients (37.8 vs. 37.6; P = 0.97). 28 The median GSM of plaques in patients with ipsilateral ‘ischaemic lesions’ on neuroimaging was similar to plaques in patients without ipsilateral lesions (36.0 ± 14.6 vs. 37.8 ± 16.9; P = 0.64). Bivariate correlation analysis revealed no significant correlation between GSM values and numbers of intra-operative MES 30 min before clamping (Spearman’s correlation ρ = 0.039; P = 0.75); however, the precise proportion of patients with MES positivity was not reported. These intra-operative data cannot be directly compared with those from our recently symptomatic or asymptomatic patients who were assessed outside of the immediate intra-operative setting. Therefore, there is insufficient evidence to definitively conclude whether MES positivity is related to GSM scores or not because published data are somewhat conflicting. Larger studies in well clinically phenotyped cohorts of carotid stenosis patients, with contemporaneous assessment of both GSM scores and TCD monitoring for MES are required to further evaluate this relationship.
To our knowledge, this is the first study to assess both carotid plaque morphology on ultrasound, MES status on TCD and circulating biomarkers of platelet production/secretion/activation and turnover. Previous studies by our group have shown that there is evidence of both enhanced platelet production/secretion/activation29–31,33 and turnover 30 in recently symptomatic vs. asymptomatic carotid stenosis patients overall. In these studies, total platelet counts and leucocyte–platelet complexes were the most sensitive indicators of platelet production/secretion/activation in symptomatic compared with asymptomatic patients. Jurk et al. found elevated monocyte–platelet complexes in 48 asymptomatic moderate-severe carotid stenosis patients with ‘echolucent’ plaques compared with healthy controls; however, plaque echogenicity was not graded objectively. 53
We found no significant differences in the percentage reticulated platelets on whole blood flow cytometry between recently symptomatic echodense vs. echolucent subgroups, or between recently symptomatic and asymptomatic GSM subgroups in this combined analysis (Table 6). However, newer, automated assays to quantify the reticulated platelet fraction appear to be more sensitive at detecting differences between subgroups of carotid stenosis patients. 30 This automated assay of the reticulated platelet fraction was not available at our institution during the conduct of the PACS study, and we did not have sufficient data from the HEIST study alone to assess the potential relationship between automated reticulated platelet fraction measurements and plaque morphology.
Histological examination of plaques removed at endarterectomy has shown that plaques with large areas of calcification exhibited the highest pre-operative GSM values, and those with large haemorrhagic areas or with a predominant necrotic lipid core exhibited the lowest GSM values, with fibrous plaques having intermediate GSM values.22,48 There were no differences in platelet activation markers between recently symptomatic vs. asymptomatic patients with echolucent plaques on GSM analysis. However, subgroup analysis revealed that GSM-echodense plaques were associated with increased platelet activation status compared with GSM-echolucent plaques in recently symptomatic patients (higher neutrophil–platelet and lymphocyte–platelet complexes). Furthermore, platelet counts (as a marker of platelet production/secretion) and all subsets of leucocyte–platelet complexes were increased in recently symptomatic compared with asymptomatic GSM-echodense patients. Although some of these findings could have arisen due an acute phase response following TIA or stroke in symptomatic patients, it is possible that excessive platelet activation could promote primary echodense plaque formation and plaque instability, thus leading to symptoms in the first instance. However, our study was not designed to address that issue and one would need to prospectively follow-up asymptomatic patients with different plaque types, in combination with assessment of platelet activation status to determine whether one or a combination of both of these biomarkers predict outcomes over time. Our results appear to be in keeping with those of a prior study which reported a significant inverse correlation between lipid core size on carotid plaque MRI and circulating CD62P levels on whole blood flow cytometry (P < 0.05); however the degree of stenosis and the proportions of patients who were symptomatic were not described in that study. 11 Patients with a detectable lipid core also had significantly lower circulating platelet GPIIb levels (P = 0.02) and monocyte P-selectin glycoprotein ligand-1 levels (P = 0.01) than those without a detectable lipid core. 11
This pilot study had some limitations. The number of subjects included was limited and some analyses may have been subject to a type II error, thus limiting our ability to identify differences between groups and subgroups. However, these pilot data will inform power calculations for future larger studies on this topic. MES status was determined on the basis of a single, validated 1-h, bilateral TCD recording. A more prolonged TCD recording period, e.g. with newer ‘ambulatory TCD’ devices 54 would most likely have increased the observed incidence of MES in asymptomatic and symptomatic carotid stenosis patients, and enhanced our ability to assess the relationships between our chosen laboratory and neuroimaging biomarkers. However, more prolonged TCD recordings were beyond the scope of the initial phases of the PACS and HEIST studies, but warrant inclusion in future studies. GW classification and GSM scoring were performed by two sets of investigators independently in the PACS and HEIST studies, respectively. The data were then combined for analysis, but all investigators strictly adhered to a standardised imaging and interpretation protocol to minimise inter-observer variability. Newer, potentially more specific computerised methodology to assess plaque echogenicity or inhomogeneity on ultrasound, such as assessment of the juxta-luminal black (hypoechoic) area55,56 or total plaque area 55 were not performed, but deserve inclusion in future studies. It is difficult to control for inter-frame variation in GSM scoring which is estimated to be approximately 5%, 57 and this remains a limitation of the GSM scoring methodology. Our study might have been subject to some selection bias as we only included patients who had complete CDUS and TCD data available from PACS and HEIST; however, we do not think this explains the observed differences between groups because the same inclusion criteria were applied to all subjects from each study.
Overall, the available literature on ultrasound-based assessment of carotid plaque morphology and the ultrasound data from this pilot study have provided conflicting results regarding the potential utility of assessing plaque echodensity with ultrasound alone to identify ‘higher risk’ asymptomatic and symptomatic carotid stenosis patients. Interpretation of these data in different national and international centres is hampered by differences in methodology and ‘cut points’ used for the definition of echolucent, mixed, or echodense plaques, as outlined above. However, data from this study do improve our understanding of the underlying biological mechanisms which may contribute to the disparity in the risk of TIA or stroke in subgroups of patients with recently symptomatic compared with asymptomatic carotid stenosis, and in subgroups of symptomatic patients with different plaque types. Larger studies which pool data from internationally standardised and reproducible methods of assessing plaque echodensity, in conjunction with robust TCD and platelet biomarker data 58 have the potential to improve risk stratification of carotid stenosis patients to enhance primary and secondary TIA/stroke prevention in routine clinical practice.1,59
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Murphy’s research was funded by the Trinity College Dublin Innovation Bursary, the Meath Foundation, Ireland, Joint IICN/Merck Serono Fellowship in Neuroscience, The Vascular Neurology Research Foundation, Ireland, and by unrestricted educational grants from Bayer HealthCare Ireland and Verum Diagnostica, GmbH. Dr. Lim’s research was funded by the Meath Foundation, Ireland, The Irish Institute of Clinical Neuroscience (IICN)/Novartis Ireland Fellowship Grant, The Irish Heart Foundation Stroke Prevention Bursary, and by unrestricted educational grant funding from Biogen Idec Ireland. None of the above charities or funding bodies had any influence on design or conduct of this study, or had any influence on the decision to submit the final manuscript for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contribution statements
All named authors and collaborators qualify for authorship and contributed to the manuscript as follows: SJX Murphy contributed to study design, data acquisition, analysis and interpretation, and manuscript writing. ST Lim contributed to data acquisition and critically appraised the manuscript for important intellectual content. JA Kinsella contributed to study design, data acquisition and analysis and critically appraised the manuscript for important intellectual content. C Dooley and J Kelly contributed to data acquisition, analysis and interpretation. S Tierney, B Egan, TM Feeley, SM Murphy, RA Walsh, DR Collins, T Coughlan, D O’Neill, JA Harbison, P Madhavan, SM O’Neill, MP Colgan, JF Meaney and G Hamilton contributed to data acquisition and critically appraised the manuscript for important intellectual content. DJH McCabe was the study PI, designed the study, contributed to data acquisition, analysis, interpretation, and manuscript writing. All source data from this study have been stored on a secure, password-protected database by the first and supervising authors.
Authors’ note
The manuscript has not been submitted elsewhere and has not been published elsewhere in whole or in part, except as an abstract.
