Abstract
Serial magnetic resonance imaging (MRI) was performed to investigate the temporal and spatial relationship between the biphasic nature of blood–brain barrier (BBB) opening and, in parallel, edema formation after ischemia–reperfusion (I/R) injury in rats. T2-weighted imaging combined with T2-relaxometry, mainly for edema assessment, was performed at 1 h after ischemia, after reperfusion, and at 4, 24 and 48 h after reperfusion. T1-weighted imaging was performed before and after gadolinium contrast at the last three time points to assess BBB integrity. The biphasic course of BBB opening with a significant reduction in BBB permeability at 24 h after reperfusion, associated with a progressive expansion of leaky BBB volume, was accompanied by a peak ipsilateral edema formation. In addition, at 4 h after reperfusion, edema formation could also be detected at the contralateral striatum as determined by the elevated T2-values that persisted to varying degrees, indicative of widespread effects of I/R injury. The observations of this study may indicate a dynamic temporal shift in the mechanisms responsible for biphasic BBB permeability changes, with complex relations to edema formation. Stroke therapy aimed at vasogenic edema and drug delivery for neuroprotection may also be guided according to the functional status of the BBB, and these findings have to be confirmed in human stroke.
Introduction
In ischemic stroke therapy, thrombolysis of the occluded cerebral artery is the most beneficial approach to effective reperfusion, which aims at salvaging the ischemic penumbra (Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group, 1995). Rapid reperfusion, although desired, contributes to secondary injury through a cascade of events, including hemodynamic disturbances, inflammatory processes, free radical formation and breakdown of the blood–brain barrier (BBB) (del Zoppo, 1994; Hacke et al, 1996). When severely impaired, there is an increased risk of deleterious vasogenic edema, brain herniation and death (Hacke et al, 1996). Experimental studies have consistently shown a biphasic pattern in BBB permeability changes after ischemia–reperfusion (I/R) injury, which has not been categorically confirmed in stroke patients (Belayev et al, 1996; Huang et al, 1999; Kuroiwa et al, 1985; Latour et al, 2004). Severe restraints are in place to subject the severely ill patient to time-consuming and exhaustive repeated magnetic resonance imaging (MRI) in the acute phase of cerebral edema. In addition, the role of biphasic BBB opening characteristics in brain edema evolution and resolution is still rather ill defined.
Magnetic resonance imaging has the potential to be the standard for assessing BBB permeability characteristics using small molecule paramagnetic contrast agents (Stoll et al, 2009). Furthermore, quantitative T2-mapping can be performed in parallel to detect dynamic changes in tissue water content, which invariably reflects the state of the barrier function at the BBB (Verheul et al, 1992). A number of studies have analyzed the temporal evolution of biphasic BBB characteristics and, separately, the temporal course of vasogenic edema formation after I/R injury. To the best of our knowledge, no study has been published that simultaneously characterized the complex relationship between BBB opening and vasogenic edema formation in a temporal and spatial manner in identical animals.
In this study, we investigated the dynamics of edema formation in both the dimensions of time and space and in relation to biphasic BBB opening after 90 mins of transient middle cerebral artery occlusion (tMCAO) in rats.
Materials and methods
Experimental Design
All experiments were performed in accordance with the council directive (86/609/EEC) of European communities and the institutional guidelines for animal care after ethics committee approval. Male Wistar rats (Charles River Laboratories, Sulzfeld, Germany) weighing between 250 and 300
Surgical Procedure
Transient middle cerebral artery occlusion was performed as previously described (Longa et al, 1989; Spratt et al, 2006). Briefly, anesthesia was induced with 5% isoflurane in a mixture of 70% nitrous oxide and 30% oxygen. After endotracheal intubation, anesthesia was maintained using 1.5% isoflurane. Body temperature was maintained at 37°C to 37.5°C throughout surgery. A cannula was inserted into the left femoral vein for contrast agent administration. Left common carotid artery bifurcation was exposed through a midline neck incision and the occipital artery branches of the external carotid artery (ECA) were isolated, ligated and dissected. After a careful isolation of the internal carotid artery, a 3 cm long silicone-coated, polyamide 4-0 monofilament (Ethicon) was advanced through the internal carotid artery to the MCA until a mild resistance was felt. After the intra-luminal suture was placed, the neck incision was closed with a silk suture. Sham operation was performed in the same manner, except for MCA occlusion. The animals were then allowed to recover. One hour after occlusion, the animals were re-anesthetized with 1.5% isoflurane and MRI was performed for the time point BR. Thereafter, animals were subjected to reperfusion by gently pulling back the filament.
Magnetic Resonance Imaging
All experiments were conducted on a standard 3T clinical dedicated head MR scanner (Magnetom Allegra, Siemens Healthcare, Erlangen, Germany), in which gradients achieve a 40 mT/m amplitude with a slew rate of 400 T/m/s per axis. The MR scanner was complemented only by a dedicated four-channel phased array rat head coil assembly (RAPID Biomedical GmbH, Rimpar, Germany). All MRI acquisitions throughout this longitudinal study have been conducted at a constant receiver gain, which was initially set during the optimization phase of the standard Siemens product sequences for performing small animal imaging.
To detect ischemic changes especially at early time points (BR and AR), DW-EPI images were acquired with repetition time (TR)=3300 ms, echo time (TE)=105 ms, field of view=6.7 cm, image matrix=104 × 104 and at five different ‘
To assess BBB permeability changes, T1-spin echo (SE) images were acquired before (pre-contrast) and after contrast agent (post-contrast) administration. The images were acquired with TR=900 ms and TE=10 ms. After acquiring pre-contrast images, Gd-DTPA (MW: 590 Da. Magnevist, Shering, Germany) was administered (0.2 mmol/kg) and post-contrast T1-SE images were acquired after 25 mins. This dose was chosen from preliminary experiments showing that, after subtraction and during the calculation of enhanced volumes, there was significant variability in the volumes estimated by two authors of this paper (DRP and DB) when using lower doses of MRI contrast agent (0.1 mmol/kg).
For assessment of brain water content/edema formation, T2-relaxometry was performed at all time points (Control, BR, AR, 04PR, 24PR and 48PR) using a SE sequence with a TR of 3330 ms and using seven TE values (29, 58, 88, 117, 146, 175 and 204 ms).
For TSE and SE acquisitions, a field of view of 2.5 cm, with an image matrix of 128 × 128, was used. The spin-echo and EPI images had an in-plane resolution of 200 and 600 μm, respectively, with 1 mm slice thickness. Sequences were applied in the following order: DW-EPI, T2-TSE, T2-relaxometry, T1-SE and post-contrast T1-SE.
Data Analysis
All required parameters were acquired from a single coronal slice (1 mm thick) located at 7 mm posterior to the anterior tip of the frontal cortex. A preliminary analysis showed that this slice revealed the largest volume of infarct at all three relevant time points, with a good assessment of the midline shift, and was best suited for standardized T2-relaxometry in the striatum and cortex. The variation in infarct volume was also found to be lower for this slice region at 24 and 48 h, compared with that at adjacent regions with similar infarct volumes (data not shown).
Quantitative T2-relaxometry was performed at three regions of interest: the ipsi- and contralateral striatum and on the ipsilateral cortex. In the striatal region, circular regions of interest have been defined over the entire striatal region, excluding the ventricles (Figure 1).
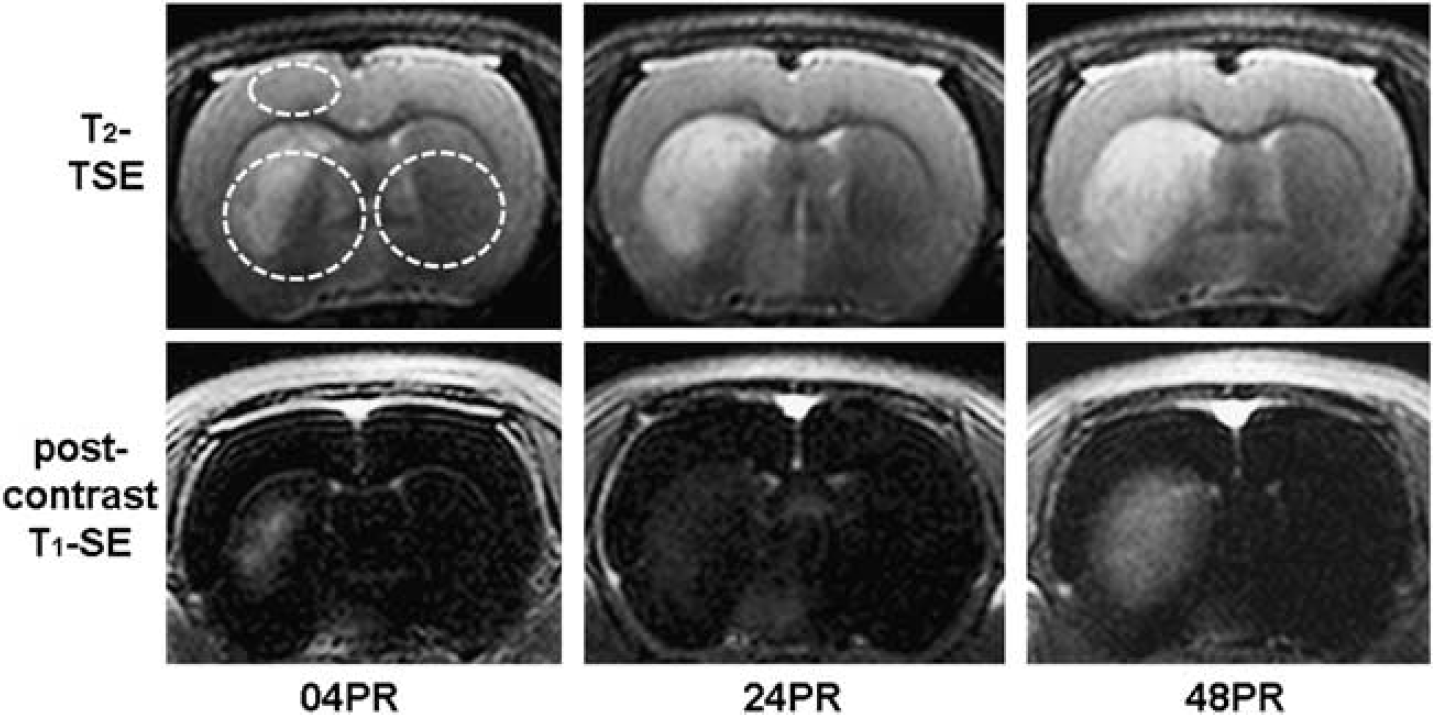
Representative images of T2-TSE for edema analysis (upper) and post-contrast T1-SE (lower) for blood–brain barrier permeability at 04, 24 and 48 h after reperfusion. Highlighted areas (as upper left image) have been considered for T2-relaxometric estimations.
Monoexponential nonlinear curve fitting was performed using Graphpad Prism Version 5.00 for Windows (Graphpad Software, San Diego, CA, USA) to determine T2-relaxation parameters. Volumetric estimations of cerebral hemispheres were again performed on T2-weighted images using the available built-in tools of Siemens
Analysis of BBB permeability changes was conducted on subtracted maps from the pre- and post-contrast T1-SE images to highlight regions of Gd-DTPA extravasation. Gd-DTPA permeable BBB volume (PBV) in cubic centimeters (cm3) representing brain tissue with leaky BBB and the average pixel intensity (T1SIdiff) of the hyper-intense enhanced regions derived from subtraction maps were calculated using the built-in tools. The obtained values of the mean pixel intensity for the subtracted images (T1SIdiff) are software generated (Siemens syngo 2004A) and standard across the current generation of Siemens scanners. A product of T1SIdiff and PBV (T1SIdiff × PBV) has been considered to account for the observed temporal and spatial changes in the average pixel intensity of enhanced regions (T1SIdiff) and the brain volume with leaky BBB (PBV). The T1SIdiff × PBV product serves as an indicator to quantify the overall entry of contrast agent into the brain over time. For sham analysis, the average PBV from the experimental group was projected onto subtracted images of sham animals, and T1SIdiff was determined within this region.
Statistical Analysis
Throughout the study, values are treated as mean±s.e. (s.e.m.). Repeated-measures analysis of variance, followed by Tukey–Kramer
Results
Three out of 11 animals did not survive the duration of this study, making for an effective
Biphasic Blood–Brain Barrier Opening and Progression of Brain Volume with Leaky Blood–Brain Barrier After Ischemia–Reperfusion Injury as Investigated by Post-Contrast T1-Sequences
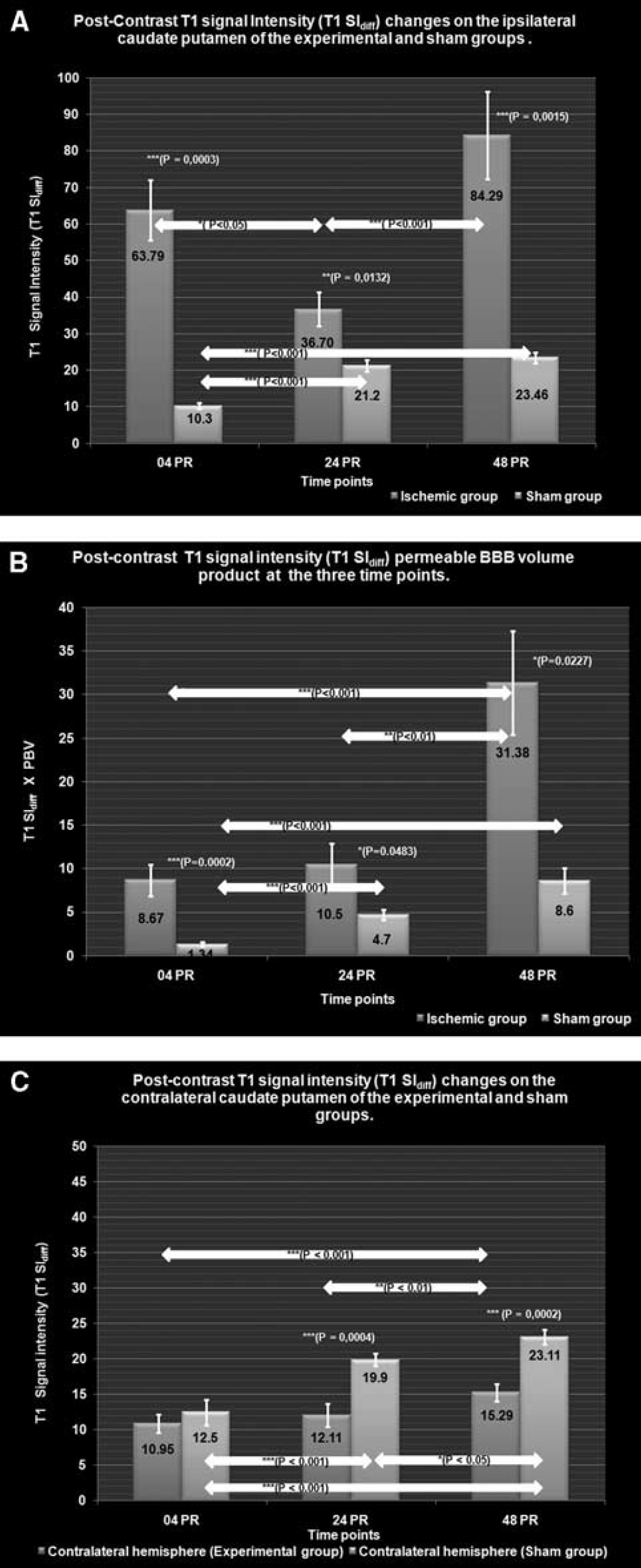
Temporal profiles of BBB leakage (post-contrast T1SIdiff) and overall extravasated contrast agent (T1SIdiff × PBV product) for the ipsilateral hemisphere are compared with those of sham (Figure 2A and 2B). Post-contrast T1SIdiff profiles of the contralateral striatum are again compared with those of sham (Figure 2C).
(
At 04PR, the post-contrast T1SIdiff value at the ipsilateral striatum of the experimental group was found to be significantly elevated (63.79±8.2) compared with that of the sham group (10.3±0.79,
By 24PR, post-contrast T1SIdiff (36.70±4.61) declined significantly to that at 04PR (
The 48PR post-contrast T1SIdiff value (84.29±11.99) was significantly higher than 24PR (
Ipsilateral and Remote Edema Formation as Investigated by the Temporal Profile of Transverse Relaxation Times
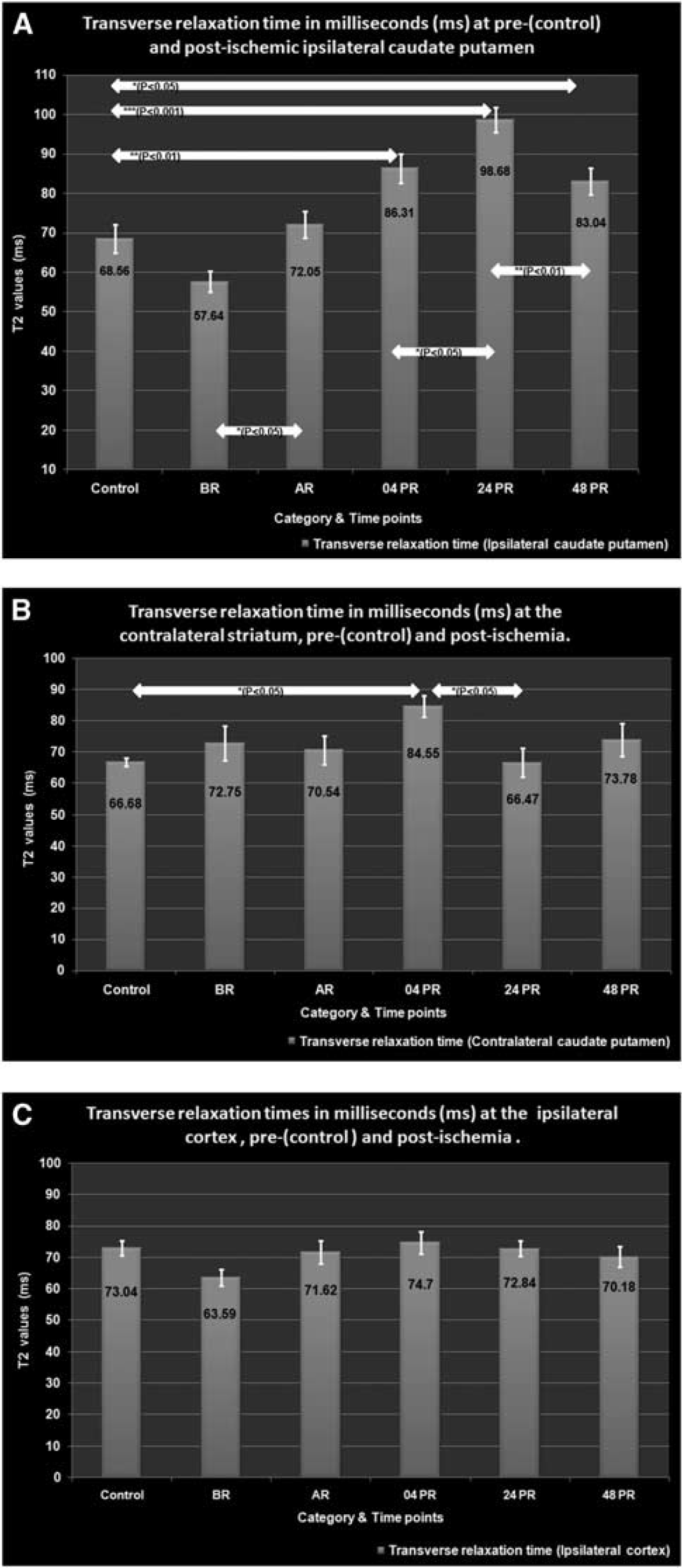
Transverse relaxation times from healthy, ischemic and reperfused tissues are shown for ipsi- and contralateral striatum along with the cortical region on the ipsilateral side (Figure 3A–3C).
(
The control T2-value indicative of an increased water content obtained at the cortex was 73.04±2.313 ms, with little inter-hemispherical difference, whereas striatal values were 68.56±3.58 and 66.68±1.38 ms at the ipsi- and contralateral hemispheres, respectively.
During tMCAO (BR), T2-values dropped at the ipsilateral cortex (63.59±2.52 ms) and striatum (57.64±2.625 ms), whereas the contralateral striatal T2-value was slightly elevated (72.75±5.617 ms). However, none of these changes were statistically significant. After reperfusion (AR), the T2-value at the ipsilateral striatum showed a significant increase to 72.05±3.4 ms (
By 04PR, T2-values at the ipsilateral (86.31±3.71 ms;
At 24PR, the ipsilateral striatal T2-value (98.68±3.12 ms) increased significantly compared with that of both 04PR (
By 48PR, the T2-value at the ipsilateral striatum (83.04±3.4 ms) showed a significant (
Discussion
Using serial MRI at 3T, we detected the phasic nature of BBB opening after I/R injury. The injured BBB showed dynamic temporal changes in permeability characteristics and might mediate local and remote vasogenic edema. Furthermore, the first phase of differential permeability characteristics at the BBB lasting up to 24 h led to progressive edema accumulation, whereas the second phase did not contribute to edema formation and may mediate edema resorption.
On reperfusion, three distinct phases of increased BBB permeability changes attributed to endothelial paracellular/tight junction (TJ) opening have been documented (Belayev et al, 1996; Huang et al, 1999; Kuroiwa et al, 1985). Increased BBB permeability (also termed BBB opening, BBB disruption, BBB leakage) is commonly characterized by the extravasation of blood-pool agents that under normal physiological conditions do not cross the BBB to enter brain parenchyma. Such blood-pool agents include the Evans Blue dye/albumin complex (MW 65961 Da) for histological studies, 3H-sucrose (MW 342 Da) for autoradiography and Gd-DTPA (MW 590 Dalton) for
Magnetic Resonance Imaging of Blood–Brain Barrier Permeability Characteristics
After 4 h of reperfusion and consistent with studies using Evans Blue, we observed the first significant increase of BBB permeability as indicated by an increase in T1SIdiff (Figure 2A). This observation denotes the first phase of biphasic permeability changes and may be attributed to increased inflammatory and oxidative stress on the BBB, in conjunction with an enzymatic degradation of extracellular matrix (Heo et al, 2005; Wang and Shuaib, 2007). Inflammatory mediators, such as thrombin, histamine and bradykinin, cause BBB permeability changes by actin polymerization-dependent endothelial cell rounding and formation of inter-endothelial gaps (Ahmmed and Malik, 2005). Proteolytic enzymes such as matrix metalloproteinases (MMPs) mediate the degradation of TJ proteins (TJPs) at such an early time point (Yang et al, 2007). The deleterious role of oxidative stress has also been shown by the reversible disruptive effect of hydrogen peroxide on the physiological, biochemical and immunocytochemical parameters of TJP integrity (Meyer et al, 2001). RNA levels of vascular endothelial growth factor (VEGF) and Angiopoetin-2 have been described to increase as early as 1 to 3 h after ischemia, whereas proteins peak later during severe vasogenic edema. As angiogenic and endothelial cell destabilizing effects have been shown to lag behind in their peak levels, their contributory roles in this initial opening seem to be minimal (Croll and Wiegand, 2001). A correlation has been shown between neutrophil infiltration into brain parenchyma and the overlying time-frame of biphasic permeability responses (Sandoval and Witt, 2008). Peak neutrophil infiltration may, however, not be the cause but may be facilitated by the disassembly of TJPs as a result of MMP activation and/or because of oxidative stress after I/R injury. As the observed enhancement is a characteristic feature of tMCAO and is not shown by permanent MCAO models, it is reasonable to speculate that the increased oxidative stress resulting from reperfusion may be the principal factor contributing to reversible TJP disruption, thereby bringing about the observed Gd-DTPA extravasation.
The first phase of biphasic BBB permeability was followed by a significant drop in BBB permeability measured by T1SIdiff at 24 h; however, a complete recovery of BBB integrity did not occur as T1SIdiff was still higher than that of sham (Figure 2A). In fact, brain tissue with leaky BBB (as measured by PBV) doubled in volume compared with that at 4 h after reperfusion and the net amount of extravasated Gd-DTPA measured as T1SIdiff × PBV product remained almost similar to that of the 4 h post-reperfusion.
Even though the precise mechanisms of this partial recovery in barrier function found at this time are not well understood, two independent mechanisms might contribute. Reactive oxygen species-mediated disassembly of TJPs has been shown to reverse within a period of 6 h, whereas ongoing effects of inflammatory mediators maintain endothelial cell rounding, resulting in an endothelial gap so large that even fully functional TJPs cannot form functional tight junctions limiting paracellular diffusion (Meyer et al, 2001). Such a situation at the BBB may still allow the extravasation of water, whereas the free flow of Gd-DTPA molecules is partially hindered by re-assembled TJPs causing the observed drop in post-contrast T1SIdiff.
After these observations at 24PR, we observed an increase in BBB permeability and brain tissue with leaky BBB as indicated by both T1SIdiff and PBV at 48PR. The angiogenic and endothelial cell destabilizing effect of VEGF and Angiopoetin-2 levels is known to peak at 48PR and may contribute to the observed BBB permeability changes (Croll and Wiegand, 2001). Monocyte chemoattractant protein-1 secretion by both astrocytes and brain endothelial cells also coincides well with the time course of events described and directly alters the BBB permeability characteristics associated with redistribution of occludin, ZO-1, ZO-2 and claudin-5. This also facilitates the entry of neutrophils to the site of the lesion (Stamatovic et al, 2005). Infiltrating neutrophils, being a rich source of MMP-9, have also been shown to mediate vasogenic edema and hemorrhagic transformation in a transient ischemic model of stroke (Gidday et al, 2005).
We observed a consistent increase in brain tissue with leaky BBB (PBV) up to 48 PR, with the most dramatic increase observed at the 24PR. Interestingly, the entire PBV presented rather homogenously at the time points of 24 and 48 PR in and outside the initial ischemic core (Figure 1).
Throughout the experiment, BBB permeability as measured by T1SIdiff at the contralateral side of the experimental group has been consistently lower than that of sham controls, probably due to a compression of cerebral vessels because of raised intracranial pressure by cerebral edema. Moreover, we observed an increase in post-contrast T1SIdiff values in the sham group as well as in the contralateral hemisphere of the experimental group, most likely attributed to isoflurane anesthesia. Although not thoroughly investigated, isoflurane has been shown to induce cerebrovascular dilation mediated through endothelial nitric oxide synthase and the formed nitric oxide in humans and animals in a dose-dependent manner (Werner et al, 2005). The confounding effects of isoflurane on the observed ipsilateral T1SIdiff may not be as direct, as this region also possesses the infarct core along with profound edema. Low basal cerebrovascular tone results in a decreased capacity to vasodilate, whereas high cerebrovascular tone expands the auto-regulatory range (Werner et al, 2005). Persistent edema observed at the ipsilateral hemisphere with increased ICP results in a low cerebrovascular tone to maintain adequate CBF. Any additional effect of isoflurane to cause further vasodilatation is rather doubtful.
Edema Development in Relation to Blood–Brain Barrier Permeability
Quantification of edema formation in both hemispheres using MRI was performed by T2-relaxometry over time and correlated to BBB permeability in parallel. At the early time point of 4PR, we could detect increased brain water content/edema as indicated by an elevated T2-value not only at the ipsi- but also at the contralateral striatum. Such remote effects at the nonischemic contralateral hemisphere have thus far been shown in permanent MCAO and cold brain injury models only (Izumi et al, 2002; Shinohara et al, 1990). In permanent MCAO, peak edema at the contralateral regions was only apparent at 24 to 48 h after occlusion. However, in the cold brain injury model, edema mediated by leaked protein was observed at the lesion site and periphery after 1 h, and by 24 to 48 h, the leaked protein was strongly stained at both hemispheres (Shinohara et al, 1990). The authors attribute this to ‘routed protein migration’ and consider such ‘remote’ effects within the realm of ‘Diaschisis’. A similar assumption may also be valid in the case of tMCAO for two reasons. First, the initial BBB permeability change would have resulted in the extravasation of serum proteins, which, along with the cellular debris after necrosis, could proliferate through the white matter tracts. Second, with an intact BBB at the contralateral striatum, the net transport of water needs to be driven by osmotic forces because of solute movements, as there are no primary active, ATP-driven, water pumps (Kimelberg, 2004).
By 24 h, the observation of a constant amount of extravasated contrast agent into the brain as observed by a constant T1SIdiff × PBV product to that at 4PR, accompanied by a peak edema observed by T2-values and volumetric swelling of the ipsilateral hemisphere, may indicate an ongoing unperturbed water shift from the vascular to the brain compartment. Contrary to the earlier time point, the observed edema is unlikely to be caused by significant BBB leakage for proteins, as indicated by the partial recovery of BBB function at this time point. Potential candidates mediating edema accumulation may include members of the nonselective cationic transient receptor potential channels, which serve as redox sensors in vascular endothelium (Groschner et al, 2004). Transient receptor potentials may form the molecular basis of oxidant-activated cation channels and are increasingly recognized for their role in regulating barrier function (Ahmmed and Malik, 2005; Simard et al, 2007). Some members of the transient receptor potential family, especially the canonical transient receptor potentials, are well recognized for their lack of cation specificity and the ability to generate substantial monovalent conductances that govern membrane potential and cationic gradients. According to the biophysical properties reported so far, inward currents through most canonical transient receptor potential channels are carried by both Na+ and Ca2+ ions in physiological conditions (Eder et al, 2005). However, the entry of Na+ has been implicated in physiological Ca2+ signaling phenomena, such as Ca2+ oscillations, and also in pathophysiological Ca2+ overload (Craner et al, 2004; Paltauf-Doburzynska et al, 2000). Such a mechanism would explain the observation that BBB permeability to sodium occurs at 12 to 48 h after MCA occlusion, which, together with an antecedent intracellular shift to sodium, causes a massive water and sodium influx (Gotoh et al, 1985). Again, continued oxidative stress resulting from reperfusion, entry of neutrophils and generation of superoxide by microglia activated by extravasated albumin and other serum components may continuously drive the upregulation and proliferation of such ion channels and would therefore be responsible for the massive edema observed at these times (Abraham et al, 2002).
At 24 h after reperfusion, we also observe a significant drop in the T2-value at the contralateral striatum compared with the T2-value at 04PR when remote edema has been consistently shown to be attributed to extravasated protein migration through white matter tracts. In the immediate absence of any scavenging mechanisms to remove these proteins and/or their degradation products, we expected a further increase in vascular edema at this time point, especially in the presence of the upregulation of ion channels as discussed before. The unexpected significant fall in T2-values at 24 h, however, may possibly be because of the reduction in the oxy-hemoglobin content resulting from vessel compression by the existing bilateral edema and the increased cerebral oxygen extraction and/or by a decreased volume of oxygenated blood volume. In this scenario, the resultant T2-value being a function of both water content and blood oxygenation effects happens to be tilted downwards.
Consistent with previous studies, we found a decrease in brain water content/edema, as indicated by the fall in T2-values and a reduction in the ipsilateral hemispherical volume at 48 h, despite an increase in BBB permeability and the BBB disrupted area, as indicated by both T1SIdiff and PBV (Belayev et al, 1996; Kuroiwa et al, 1985; Wang and Shuaib, 2007). This indicates a resorption of vasogenic edema, possibly brought about by a variety of mechanisms, including the uptake of extravasated proteins by glial cells and the resulting reduction in oncotic force, migration of edema into the cerebrospinal fluid spaces and also because of effects from proliferating endothelium. (Hatashita and Hoff, 1988, 1990; Klatzo, 1985). Of late, selective water channels at the astrocytic end feet, the aquaporins, especially aquaporin-4, have also been proposed to have an important role in the pathophysiology of brain edema both in rodents and humans, with a peak at 48PR (Zador et al, 2009). Furthermore, the T2-values at the contralateral striatum showed a nonsignificant but re-bound increase in values. As this value is higher than that of the control, it may indicate the persistent presence of remote edema at these times, which was duly masked at the 24 h time point due to increased ICP.
T2-relaxometry is also influenced by flow effects, alteration in the amounts of oxy- and deoxy-hemoglobin, tissue oxygenation and the exchange of nuclear spin magnetization between the ‘free’ and ‘bound’ proton pools (Calamante et al, 1999). We observed this ‘blood oxygenation level dependant’ effect in the very early phase of ischemia by a consistent fall in T2-values, which affected the whole ischemic hemisphere even in regions that did not show pannecrosis (Longa et al, 1989; Young et al, 2007). As expected, after reperfusion, the increased inflow of oxygenated blood resulting from reactive hyperemia, which invariably develops after the release of arterial occlusion, elevated the transverse relaxation time at the ipsilateral striatum.
Biphasic Blood–Brain Barrier Permeability—a Surrogate Marker for the ‘New Penumbra’?
In a recent editorial, Eng Lo coined the term ‘new penumbra’ to define the transition of most targets of ischemic stroke therapy (VEGF, MMPs, nitric oxide, glutamate release, JNK pathway, among others) from mediators of injury to effectors of repair after acute stroke (Lo, 2008). Indeed, the lack of understanding of this ‘Janus-faced’ nature of many targets of neuroprotection in stroke research might represent the increasing frustration with a number of proposed neuroprotectants (Endres et al, 2008). The observed phasic permeability characteristics of BBB and the resultant shifts in edema may possibly be orchestrated by a variety of mediators, from growth factors (i.e., VEGF, epidermal growth factor, brain-derived growth factor), MMPs, nitric oxide to inflammatory cytokines (i.e., IL-6, IL-1ß, C-reactive protein, TNF-
However, confirming that a similar characterization of BBB changes in humans is a prerequisite for such timely pharmacotherapy demands great efforts from acute stroke patients and neuroradiologists in the early hours after thrombolysis. In addition, because of inherent variability in human stroke, biphasic BBB behavior may not occur as uniform as in experimental settings, but in overlapping time intervals, which may complicate the analysis of MRI data. Using a human 3T MR scanner with identical sequences to the application in human stroke allows us to use an identical analysis as used in this study.
Summary
In this MRI study conducted on a relatively low-field clinical scanner, we were able to define the timely correlation between the biphasic opening of BBB and the dynamics of brain water content during the first 48 h after focal cerebral ischemia. The phasic phenomena of altered BBB permeability might not only contribute to local but might also possibly mediate widespread edematous changes including resorption at late time points. Magnetic resonance imaging equivalents of BBB permeability characteristics and edema formation are exceedingly complex and are continuously influenced by raised ICP, altered CBF, blood oxygenation level dependant effects and effects of inhalational anesthetics. The biphasic BBB opening and edema formation as shown in this study may reflect injury and repair after ischemia and reperfusion at the neurovascular unit and need to be confirmed in acute stroke patients using similar MRI sequences. Stroke therapy aimed at vasogenic edema and neuroprotection strategies may be guided according to the functional status of BBB.
Footnotes
Acknowledgements
DR Pillai was supported by a grant from ‘Bayerische Forschungsgemeinschaft’ and by the Bavarian State Ministry of Sciences, Research and the Arts, ‘ForNeuroCell’ grant. We thank the ‘Achtzehn Medical Service’ (Erlangen, Germany) for donation of anesthetic equipment.
The authors declare no conflict of interest.
