Abstract
Ischemic stroke is a leading cause of death worldwide and clinical data suggest that children may recover from stroke better than adults; however, supporting experimental data are lacking. We used our novel mouse model of experimental juvenile ischemic stroke (MCAO) to characterize age-specific cognitive dysfunction following ischemia. Juvenile and adult mice subjected to 45-min MCAO, and extracellular field recordings of CA1 neurons were performed to assess hippocampal synaptic plasticity changes after MCAO, and contextual fear conditioning was performed to evaluate memory and biochemistry used to analyze Nogo-A expression. Juvenile mice showed impaired synaptic plasticity seven days after MCAO, followed by full recovery by 30 days. Memory behavior was consistent with synaptic impairments and recovery after juvenile MCAO. Nogo-A expression increased in ipsilateral hippocampus seven days after MCAO compared to contralateral and sham hippocampus. Further, inhibition of Nogo-A receptors reversed MCAO-induced synaptic impairment in slices obtained seven days after juvenile MCAO. Adult MCAO-induced impairment of LTP was not associated with increased Nogo-A. This study demonstrates that stroke causes functional impairment in the hippocampus and recovery of behavioral and synaptic function is more robust in the young brain. Nogo-A receptor activity may account for the impairments seen following juvenile ischemic injury.
Introduction
Ischemic stroke is a pathology that has eluded successful pharmacologic therapy (aside from endovascular revascularization in select patients), despite tremendous efforts. This, in conjunction with increasing appreciation that stroke affects people of all ages, demands that novel strategies to improve the outcomes of survivors be realized. Recent pre-clinical trials have shifted focus from acute neuroprotective strategies to exploring treatments that can improve functional status when administered at delayed time points. Much of this research has focused on cortical injury and restoration of motor deficits,1,2 with less emphasis on cognitive domains. Learning and memory problems are a common feature after focal ischemia, with 30–50% of survivors showing impairments in the first year after stroke.3–6 Therefore, the focus of this study centers on hippocampal dysfunction after stroke to examine a new strategy to restore memory at chronic time points after focal ischemia. Recently, we provided evidence that reversing learning and memory impairments in the adult brain is possible at chronic time points after stroke, 7 bolstering the idea that delayed pharmacologic therapies may be a viable therapeutic strategy to improve cognitive outcomes after stroke. Here, we investigate a novel target, Nogo-A receptor, in furthering a delayed therapeutic approach after stroke in the young brain.
Children who survive arterial ischemic stroke encounter long-term neurologic deficits with varying degrees of disability, 8 that can affect learning during critical school age years. Clinical studies suggest that children recover from stroke to a greater degree than those suffering stroke around the time of birth or in adulthood.8–10 Despite the apparent potential for the juvenile brain to recover after stroke, the impact of age on the brain's response to focal ischemia remains understudied. In order to understand the physiology after stroke that may be unique to the juvenile developmental time period, we used a recently developed mouse model of juvenile arterial ischemic stroke 11 to describe the electrophysiologic and behavior phenotypes that characterize the changes in synaptic function that occur after focal ischemia in the young brain.
Nogo-A is a neurite growth inhibitory factor that has been shown to be important in regulating structural changes throughout the central nervous system.12,13 Nogo-A has been well studied in axonal regeneration after CNS injury in adults13,14 and as a negative regulator of structural plasticity during development.12,15 More recently, Nogo-A has been found to restrict memory formation 16 and hippocampal long-term potentiation (LTP)17,18 via the interaction of the Nogo-66 domain of the Nogo Receptor 1 (NgR1). Remarkably, restriction of LTP can be rapidly attenuated via acute neutralization of Nogo-A signaling. 17 Consistent with this rapid synaptic signaling, Nogo-A and NgR1 are expressed in hippocampal pyramidal cells and located in the synapses of the mature CNS. 19 Therefore, we used the new juvenile arterial ischemic stroke model to test the hypothesis that increased Nogo-A signaling at hippocampal synapses actively impairs plasticity following ischemic stroke and reduced Nogo-A expression at delayed time points is associated with resilience and recovery unique to the developing brain.
Methods
Experimental animals
All experimental protocols were conducted in accordance with the American Association for the Accreditation of Laboratory Animal Care (AAALAC) policies and approved by the University of Colorado School of Medicine Institutional Animal Care and Use Committee (IACUC) and conformed to the National Institutes of Health guidelines for care and use of animals. C57Bl/6 mice (Charles River Laboratory) were 21–25 days old (juvenile) or 60–90 days old (adult) at the time of sham or MCAO. The mice were housed in a standard 12-h light and 12-h dark cycle and had free access to food and water. All experiments in the study adhered to the ARRIVE guidelines for animal experiments. Mice were randomly assigned to experimental groups and the investigator was blinded for all analyses.
Middle cerebral artery occlusion model
MCAO was induced using the reversible intraluminal filament method as previously described.7,11,20 Briefly, focal cerebral ischemia was induced for 45 min under isoflurane anesthesia (induction 3.0% and maintenance 1.5–2.0%). Head and body temperature were maintained at 36.5 ± 1.0 ℃ throughout the MCAO surgery. A laser Doppler probe (Moor Instruments, Oxford, England, UK) was placed over the ipsilateral (right) parietal cortex to assure adequate occlusion. Only animals with a blood flow reduction to less than 25% of baseline were included. The suture was removed after 45 min to restore blood flow to the affected MCA territory.
Electrophysiology
Hippocampal slices were prepared at 1, 7 or 30 days after recovery from MCAO or sham surgeries. Mice were anesthetized with 3% isoflurane in an O2-enriched chamber. Mice were transcardially perfused with ice-cold (2–5℃) oxygenated (95% O2/5% CO2) artificial cerebral spinal fluid (ACSF) for 2 min prior to decapitation. The composition of ACSF was the following (in mmol/L): 126 NaCl, 2.5 KCl, 25 NaHCO3, 1.3 NaH2PO4, 2.5 CaCl2, 1.2 MgCl2 and 12 glucose. Horizontal hippocampal slices (300 µm thick) were cut with a Vibratome 1200S (Leica) and transferred to a holding chamber containing ACSF for at least 1 h before recording.
Synaptically evoked field potentials were recorded from hippocampal CA1 slices that were placed on a temperature controlled (31 ± 0.5oC) interface chamber perfused with ACSF at a rate of 1.5 ml/min. Excitatory post-synaptic potentials (fEPSP) were produced by stimulating the Schaffer collaterals and recording in the stratum radiatum of the CA1 region. The fEPSPs were adjusted to 50% of the maximum slope and test pulses were evoked every 20 s. Paired pulse responses were recorded using a 50-ms interpulse interval (20 Hz) and expressed as a ratio of the slopes of the second pulse over the first pulse. A 20-min stable baseline was established before delivering a theta burst stimulation (TBS) train of four pulses delivered at 100 Hz in 30 ms bursts repeated 10 times with 200 ms interburst intervals. 21 Following TBS, the fEPSP was recorded for 60 min. The averaged 10-min slope from 50 to 60 min after TBS was divided by the average of the 10-min baseline (set to 100%) prior to TBS to determine the amount of potentiation. Analog fEPSPs were amplified (1000×) and filtered through a pre-amplifier (Model LP511 AC, Grass Instruments) at 1.0 kHz, digitized at 10 kHz and stored on computer for later off-line analysis (Clampfit 10.4, Axon Instruments). The derivative (dV/dT) of the initial fEPSP slope was measured. For time course graphs, normalized fEPSP slope values were averaged and plotted as the percent change from baseline. In experiments using NEP(1–40) (Tocris), the drug was made in 1 mM aliquots in saline and diluted in ACSF to final concentrations as indicated in the results. In experiments using Nogo-P4 (Alpha Diagnostics Intl, Inc.), the drug was made in 4 mM aliquots in saline and diluted in ACSF to final concentrations as indicated in the results. Electrophysiology studies using NEP(1-40) and Nogo-P4 were performed in paired fashion. Specifically, acute slices were obtained from mice seven days after MCAO or sham surgeries and were superfused with ACSF (control) or ACSF + drug. Experiments were only included in final analysis if data were obtained under control and the investigational drug-exposed slices from the same animal. Two electrophysiologists (RD and JO) independently verified all LTP results in this report.
Behavioral test
The contextual fear conditioning (CFC) paradigm was used as a hippocampal-dependent memory task, as described previously.7,22,23 The apparatus consisted of two fear conditioning chambers with shock grid floors, consisting of 16 stainless steel rods connected to a shock generator (Coulbourn Instruments, Model H13-15, Whitehall, PA, USA). During training, mice were allowed to habituate to the conditioning chamber for two separate 2 min pre-exposure sessions followed by a foot shock (2 s/1.0 mA electric shock) immediately after the second exposure. Memory was tested 24 h later, determined by percentage of freezing behavior, measured in 10-s intervals across a 5-minute test by a blinded observer and was defined as the absence of movement except for heart beat/respiration.
Western blot analysis
Single whole hippocampi were homogenized in N-PER (Thermo Scientific, Rockford, IL, USA) and nuclei and debris were removed by centrifugation (1000g × 10 min). Total protein content was measured by using a microplate bicinchoninic acid assay (Thermo Scientific, Rockford, IL, USA), and protein was resolved via electrophoresis on 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis gels for 1 h at 150 V. Protein was transferred to a polyvinylidene difluoride membrane for 1 h at 100 V, and incubated at room temperature (22 ℃) with gentle rocking in 5% milk in Tris-buffered saline with 0.1% Tween-20. Blots were incubated with anti-Nogo-A (1:2000; Santa Cruz Biotechnology, Inc, USA) or anti- β-actin (1:5000; Sigma-Aldrich, St Louis, MO, USA) overnight at 4 ℃, and washed five times, followed by a 1-h incubation with a horseradish peroxidase-conjugated goat anti-mouse (1:4000; Thermo Scientific). Blots were then washed in Tris-buffered saline with Tween five times for 5 min each, and bands were detected using SuperSignal chemiluminescent substrate kits (Thermo Scientific) and a ChemiDocTM MP Imaging System (Bio- Rad, Hercules, CA, USA). Quantification of integrated volume of bands was performed using IMAGE LAB software version 4.0 (Bio-Rad).
Antibodies
Primary antibodies used for Immunohistochemistry were rabbit anti-Nogo-A (1:300, Abcam), mouse anti-neuronal nuclei (NeuN, 1:500, Millipore), mouse anti-GFAP (1:500, Santa Cruz Biotechnology, Inc, USA), goat anti-Iba-1 (Iba-1, 1:300, Abcam), with Alexa Fluor 488 or 594-conjugated IgG (1:600; Jackson Immuno) secondary antibody.
Immunohistochemistry
For Nogo-A expression, staining of 50 µm sections consisted of phosphate-buffered saline washes (1 × PBS, 3 × 5 min), 2 h incubation in blocking serum (10% normal donkey serum in 0.3% Triton X-100), 48 h incubation at 4 ℃ in primary antibody, PBS washes (3 × 5 min), 1 h incubation in secondary antibody, PBS washes (3 × 5 min), mounting and coverslip with anti-fade mounting medium (Vectashield, H-1000). Confocal microscopy was used to confirm co-localization of Nogo-A and Hoechst using an Olympus FV1000 laser scanning confocal microscope and Olympus Fluoview imaging software (Center Valley, PA, USA). The cell counter plug-in on Fiji software 24 was used for cell count analyses of medial hippocampus, averaged across two sections per mouse.
Statistical analysis
All data are presented as mean±SD. Sample size and power analyses were performed using previous data generated in our laboratory. To determine group size for LTP recordings, to observe a 40% change in LTP between two groups with a standard deviation of 20 and an alpha error of 5% and a beta error of 80%, a group of 6 slices per group are required. For behavior studies, to observe a 25% change in object exploration between two groups with a standard deviation of 15%, with an alpha error of 5% and a beta error of 80%, eight animals per group are required for behavior experiments. Statistical analysis was performed using Student's t-test for two-group comparisons and one-way ANOVA with Tukey post hoc test for comparison of multiple groups (95% confidence intervals in Supplemental Figure 2). The effect of NEP(1-40) and Nogo-P4 on LTP after MCAO was analyzed by paired t-test. Differences were considered statistically significant at two-tailed p < 0.05.
Results
Synaptic function is impaired in the CA1 area of the hippocampus after MCAO
To determine the effect of experimental stoke on hippocampal function of developing brains, juvenile mice (p21–25) were exposed to transient MCAO and extracellular field recordings of CA1 neurons were analyzed. We recently described persistent impairment of synaptic function after experimental stroke in adult mice.
7
Field excitatory potentials from hippocampal slices obtained from juvenile sham mice or mice exposed to 45-min MCAO were measured by placing a stimulating electrode in stratum radiatum and a recording electrode in the CA1 dendritic area. Seven days after sham surgery in juvenile mice, a brief TBS (40 pulse TBS) resulted in potentiation (LTP), with fEPSP slope increasing to 154 ± 15% (n = 8, Figure 1). In contrast, juvenile mice show impaired LTP in the ipsilateral hippocampus 7 days after MCAO (93 ± 18%, n = 7), but remarkably fully recover 30 days (166 ± 43%, n = 6) after MCAO. LTP was impaired in the contralateral hippocampus at 24 h after MCAO (130 ± 23%, n = 7 in 24 h MCAO, p < 0.05), but not impaired either 7 days (165 ± 20%, n=7) or 30 days (175 ± 20%, n = 5) following MCAO. Figure 1(c) shows a summary of these results. Confidence intervals for all multiple comparisons are shown in Supplemental Figure 2(a). There was no effect of stroke on overall excitability (input-output curve) or presynaptic probability of release (paired-pulse ratio) (data not shown).
Focal cerebral ischemia transiently impairs synaptic plasticity in juvenile mice. (a) Time plots of fEPSP slope from sham mice (n = 7, black), mice at 7 days (n = 7, blue), or 30 days (n = 5, green) after MCAO in ipsilateral slices. Arrow indicates timing of theta-burst stimulation (40 pulses, 100 Hz). (b) Time plots of fEPSP slope from sham mice (n = 7, black), mice at 24 h (n = 7, white), mice at 7 days (n = 7, red), or 30 days (n = 5, cyan) after MCAO in contralateral slices. (c) Quantification of change in fEPSP slope after 60 min following TBS normalized to baseline in ipsilateral slices and contralateral slices, set at 100%. Each point represents a hippocampal slice that was recorded with no more than two slices per animal used. (d) Quantification of freezing behavior 24 h after contextual fear conditioning in a novel environment 7 or 30 days after MCAO. Each point plotted represents each animal tested. Data presented as mea ±SD, *p < 0.05 by ANOVA.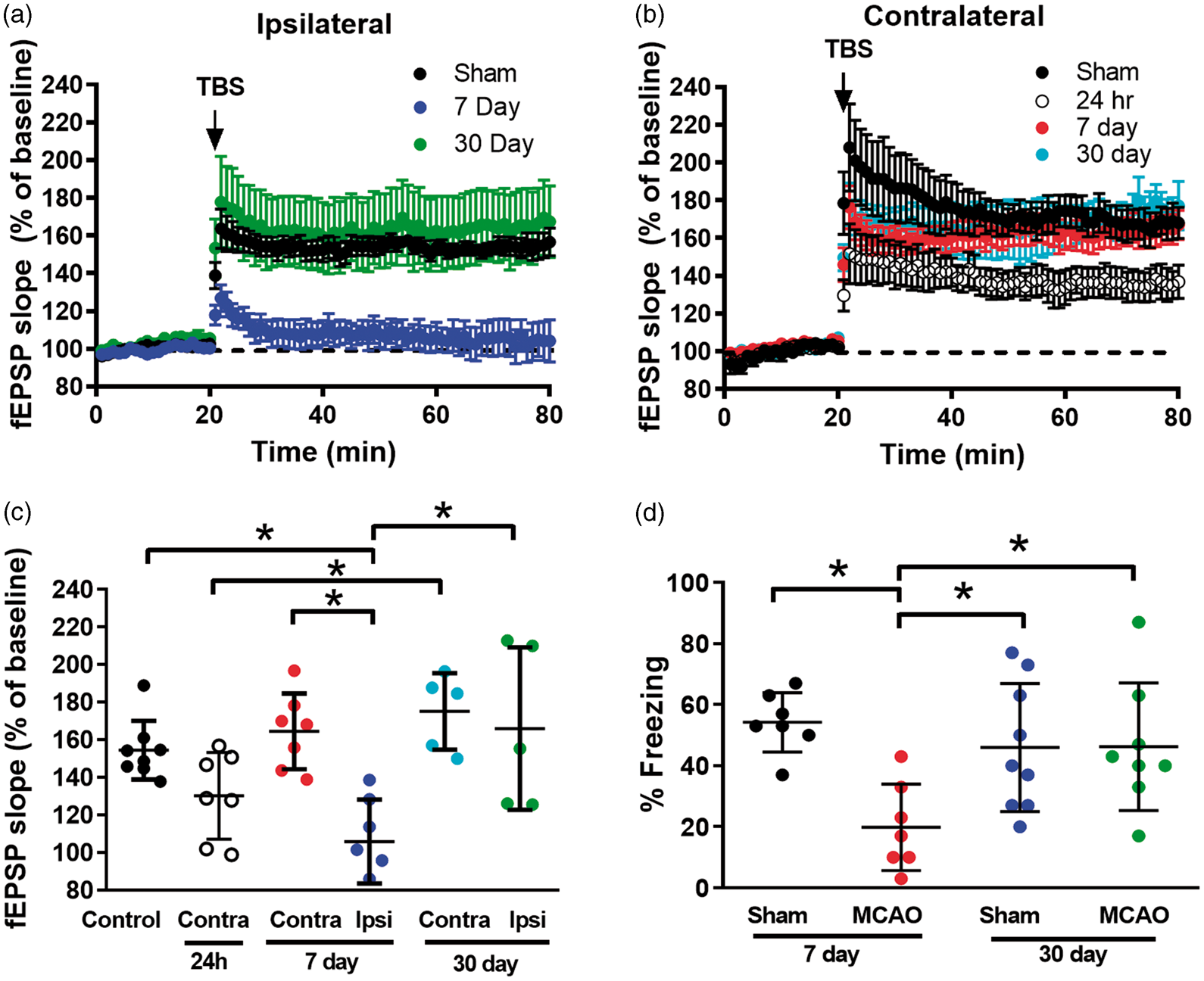
LTP is well correlated with learning and memory and we have shown memory impairments observed after focal cerebral ischemia in adults. 7 Therefore, we tested the effect of stroke on learning and memory of juvenile mice by performing CFC, a test of hippocampal-dependent cognition and memory.7,23,25,26 We found that seven days after sham surgery, mice exhibited freezing behavior in 54 ± 10% of time epochs (n = 7, Figure 1(d)) 24 h after training, consistent with intact memory. However, mice seven days after MCAO demonstrated a decrease in freezing behavior (20 ± 14%, n = 7, p < 0.05), indicating impaired memory. A separate set of mice tested 30 days after MCAO, froze 46 ± 20% of time epochs (n = 9), indicative of recovered memory function compared to the seven-day MCAO juvenile mice (Figure 1(d), p < 0.05). Confidence intervals for all multiple comparisons are shown in Supplemental Figure 2(b). These results are similar to 30-day sham mice (46 ± 21%, n=8). Taken together, these data indicate that juvenile mice have impairments in synaptic plasticity and memory that recover as the mouse matures into adulthood.
Nogo-A expression transiently increases after juvenile MCAO
It has been suggested that Nogo-A has a role in rapid synaptic signaling, restricting the induction of LTP.
17
To test the hypothesis that Nogo-A is responsible for the impairment of synaptic plasticity after juvenile stroke, we examined the temporal expression of Nogo-A within the CA1 region of the hippocampus after MCAO. Figure 2 reveals a 90% increase in Nogo A expression in the ipsilateral CA1 compared to the contralateral CA1 seven days after MCAO (normalized Nogo-A/β-actin ratio 1.62 ± 0.3 in ipsilateral [n = 6] versus 0.86±.01 in contralateral [n = 6], p < 0.05 by paired t-test, Supplemental Figure 1). We went on to confirm an increase in Nogo-A in ipsilateral hippocampal CA1 neurons seven days after MCAO using immunohistochemistry. Consistent with our Western blot analysis, juvenile MCAO caused an increase in the number of Nogo-A positive cells in the ipsilateral hippocampus (Figure 2(a)) compared to the contralateral hippocampus (Figure 2(b)) seven days after recovery from MCAO (n = 8 per group). The number of Nogo-A positive cells in the ipsilateral hippocampus at 30 days (n = 8) after MCAO was not different from 7-day or 30-day contralateral hippocampus (n = 8; Figure 2(i)). Together, these data suggest that transient impairment of memory and synaptic function correlate with increased hippocampal Nogo-A in juvenile mice after MCAO.
Ischemia increases Nogo-A expression specifically in juvenile hippocampus. Nogo-A immunoreactivity increases the ipsilateral CA1 (a) but not in the contralateral CA1 (b) seven days after MCAO in juvenile mice. In adult mice, there is very little Nogo-1 staining in ipsilateral (c) or contralateral CA1 (d) seven days after MCAO. (c′) Expanded image of inset box in Figure (a) showing that Nogo-A (E-red) and GFAP (F-green) colocalize, whereas Iba-1 (G-blue) does not. (i) Quantification of data showing that Nogo-A-positive cells are increased in ipsilateral CA1 seven days after juvenile MCAO compared to the contralateral CA1 seven days after juvenile MCAO, sham CA1 and adult CA1, n = 8 per group. Data presented as mean ± SD, *p < 0.05.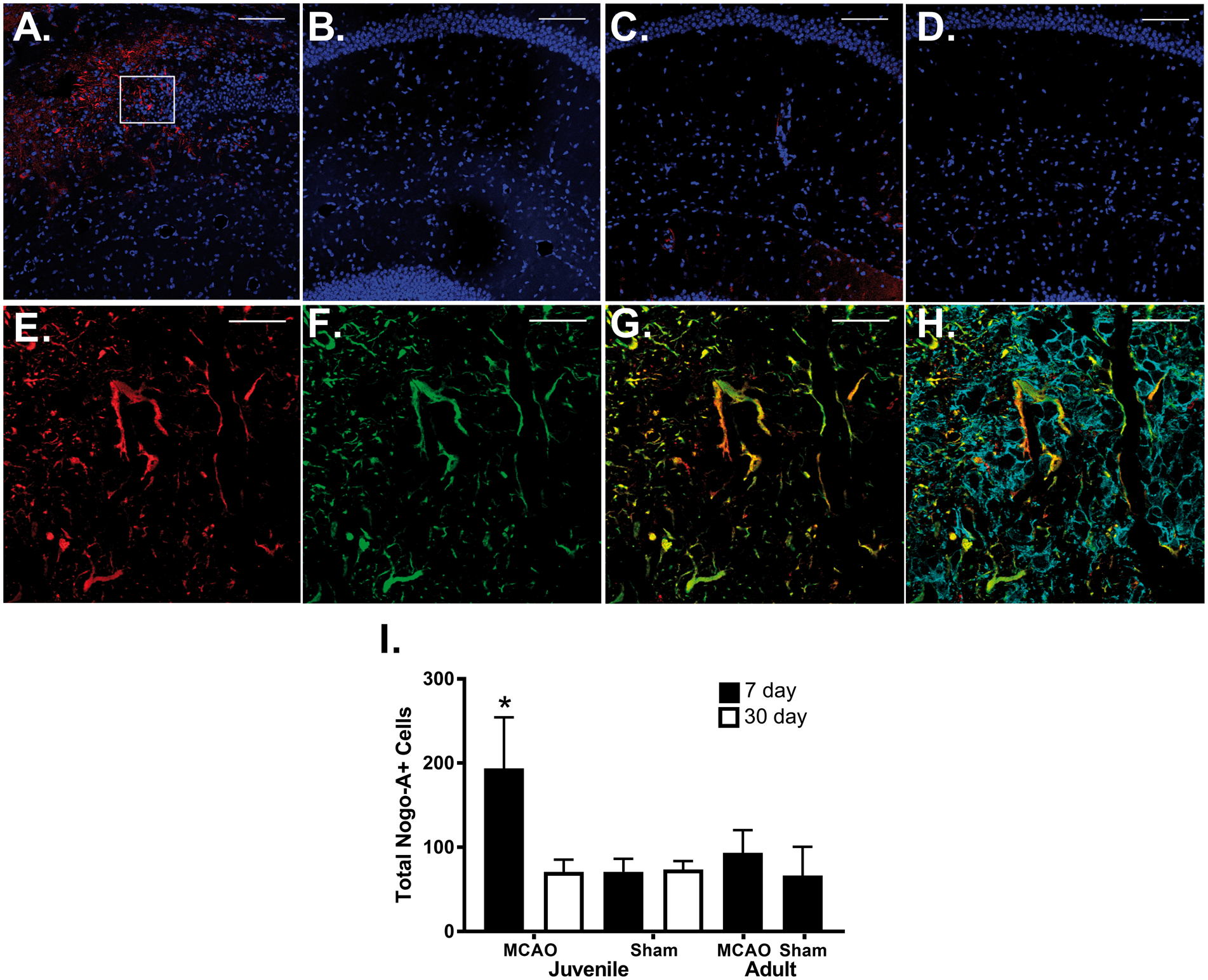
Recently, we reported persistent impairment of LTP after stroke in adult mice. 7 Therefore, we tested whether increased Nogo-A expression may play a role in this impairment. In slices from adult mice seven days after MCAO showed no difference in the number of Nogo-A positive cells when compared to sham (Figure 2(c), (d) and (i)).
We further interrogated the source of increased Nogo-A expression seen in the ipsilateral hippocampus. Hippocampal sections were co-labeled with Nogo-A (Figure 2(e)), GFAP (astrocyte stain; Figure 2(f)) and Iba-1 (microglia stain; Figure 2(g)). Figure 2(h) shows merged images showing significant overlap of Nogo-A and GFAP, whereas there is no co-staining of Nogo-A an Iba-1. Together, these data suggest that astrocytes may be the source of increased expression of Nogo-A at sub-acute time points after juvenile stroke.
Delayed inhibition of Nogo receptors rescues synaptic plasticity after juvenile MCAO
To test whether the increase of Nogo-A following juvenile MCAO is a causative contributor to impaired LTP, we applied the Nogo receptor antagonist NEP(1-40) to ipsilateral slices from mice seven days after juvenile MCAO. Figure 3 shows paired experiments (slices from the same animal used for control and NEP(1–40) recordings) from mice seven days after sham or MCAO surgery. All slices were exposed to NEP(1-40) (1 µM) or vehicle 30 min prior to TBS and throughout the duration of LTP experiment. Sham slices showed an increase in LTP from 146 ± 17% in vehicle (n = 7) to 172 ± 21% with NEP(1–40) (n = 7, Figure 3(a), p < 0.05, paired t-test), consistent with prior observations on the effect of Nogo-A receptor antagonists on LTP.
17
In paired experiments using ipsilateral hippocampal slices from juvenile mice seven days after MCAO, exposure to NEP(1–40) resulted in reversal of synaptic dysfunction from 103 ± 25% in vehicle to 143 ± 21% in NEP(1–40) (n = 7 each, Figure 3(b), p < 0.05, paired t-test). There was no difference on paired-pulse stimulation after administration of NEP(1–40) (data not shown).
Nogo-1 receptor antagonist rescues synaptic impairment when applied seven days after juvenile MCAO. (a) Time plots of fEPSP slope from mice seven days after sham experiments (black, n = 7) and paired slices bath applied with NEP (1–40) (1 µM, n = 7, red). Arrow indicates timing of theta-burst stimulation (TBS, 40 pulses, 100 Hz). (b) Time plots of fEPSP slope from mice seven days after MCAO experiments (black, n = 7) and paired slices bath applied with NEP (1–40) (1 µM, n = 7, red). C. Quantification of change in fEPSP slope after 60 min following TBS normalized to baseline. Paired experiments are indicated by the line between the pairs where slices from the same juvenile mouse prepared seven days after sham or MCAO were tested with vehicle or NEP (1–40). *p < 0.05 by paired t-test.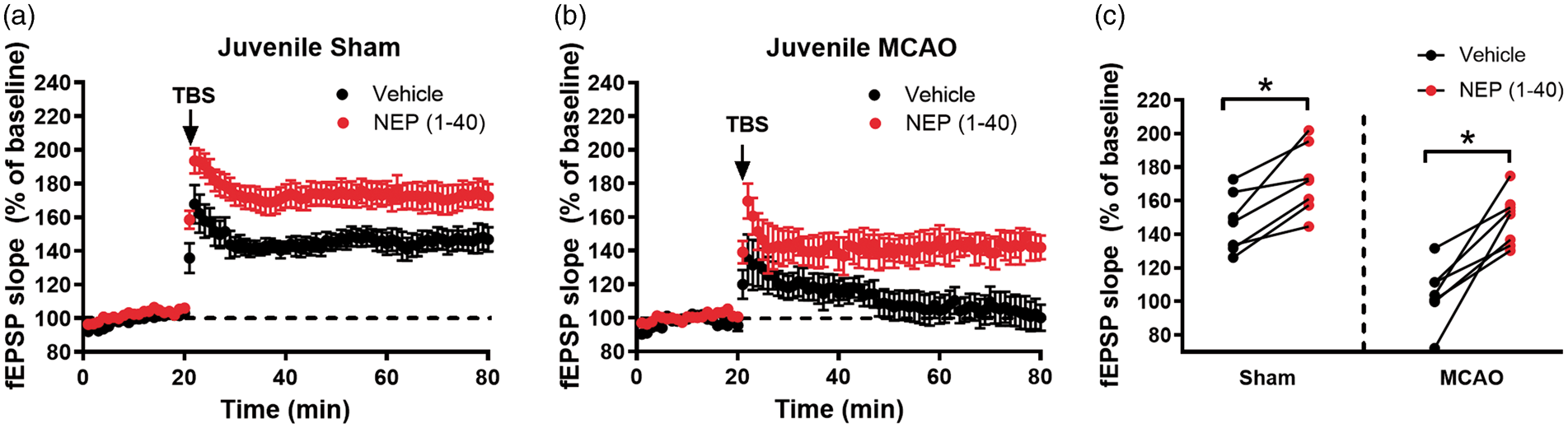
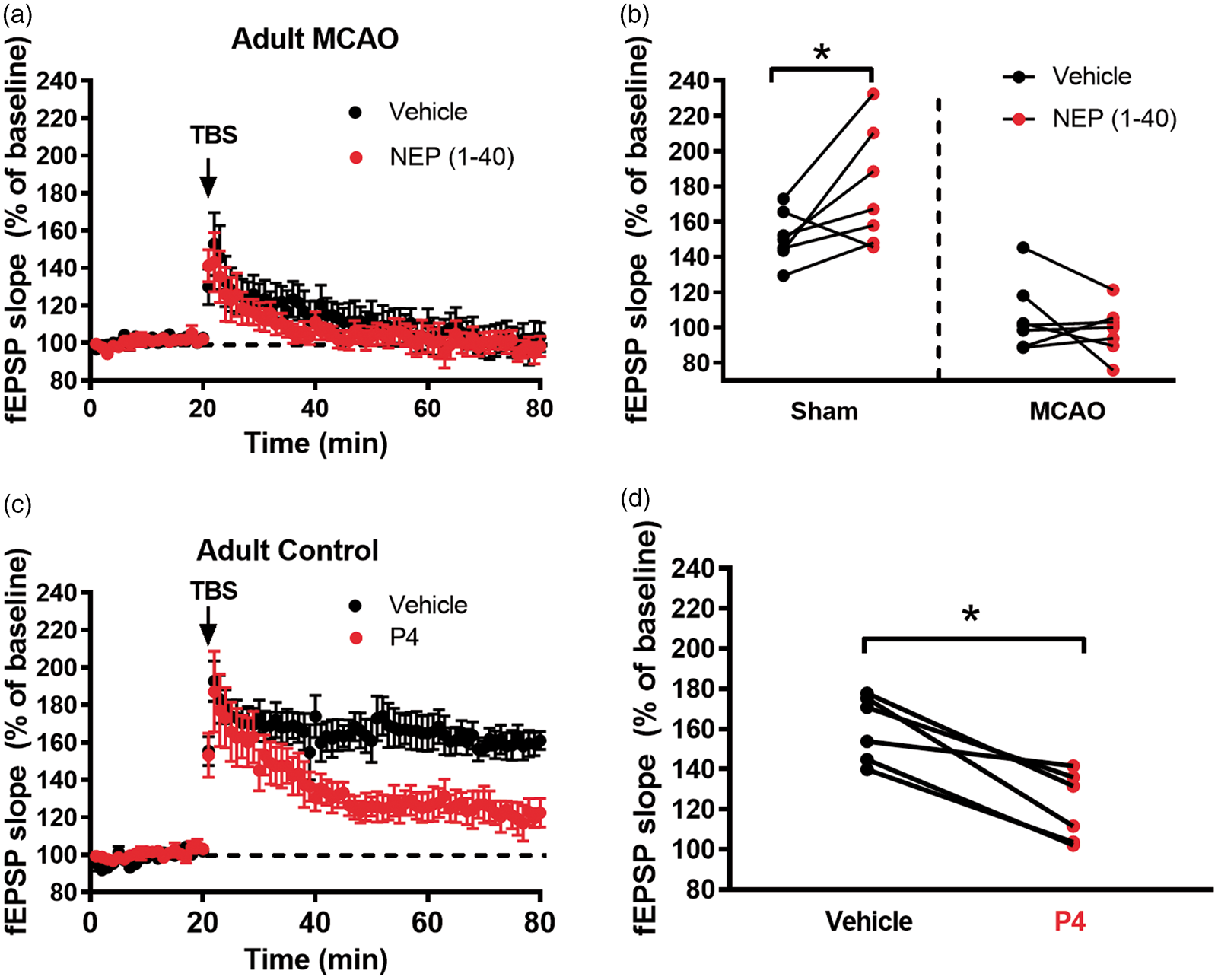
We next investigated whether inhibiting Nogo-A receptors in adult mice after MCAO would reverse MCAO-induced deficits in synaptic plasticity. Given the IHC results showing no change in Nogo-A expression after stroke in adult mice, we hypothesized that inhibition of Nogo receptors would not have an effect. In sham mice, using the same experimental design as in the juvenile mice above, exposure to NEP(1–40) resulted in an increase in LTP (150 ± 16% in vehicle vs. 178 ± 31% after NEP(1–40), n = 8 each, p < 0.05, paired t-test, Figure 5(b)). However, there was no change in the impairment of synaptic plasticity in slices seven days after adult MCAO with or without NEP(1–40) (101 ± 23% in vehicle vs. 99 ± 13%, respectively, n = 8, Figure 5(a) and (b)). Consistent with our recent report showing no difference in striatal injury following MCAO in juvenile and adult animals,
27
our analysis of infarct volume showed equivalent injury at both ages (Figure 4). Finally, to verify that Nogo-A receptor signaling is intact in the adult hippocampus, the Nogo-A inhibitory peptide Nogo-P4, a 25-amino acid peptide corresponding to part of the Nogo-66 domain of Nogo-A,
28
was used on acute slices from control animals. In paired experiments from the same mouse, vehicle-treated mice exhibited an average potentiation of 160 ± 16% (n = 6, Figure 5 (c) and (d)) that was reduced to 121 ± 17% (n = 6, p < 0.01 by paired t-test) in Nogo-P4-treated slices. Nogo-P4 had no effect on paired pulse stimulation after 30 min of bath application (data not shown).
No age difference in infarct volume following MCAO. Representative TTC-stained brain slices in adult (a) and juvenile (b) mice. (c) Quantification of infarct volume in adult (n = 8) and juvenile (n = 7) mice. Data were presented as % infarct relative to contralateral structure. Nogo-1 receptor antagonist does not affect LTP impairment in adult mice. (a) Time plots of fEPSP slope from mice seven days after MCAO experiments (black, n = 7) and paired slices bath applied with NEP (1–40) (1 µM, n = 7, red). Arrow indicates timing of theta-burst stimulation (TBS, 40 pulses, 100 Hz). (b) Quantification of change in fEPSP slope after 60 min following TBS normalized to baseline paired experiments are indicated by the line between the pairs where slices from the same adult mouse prepared seven days after sham or MCAO were tested with vehicle or NEP (1–40). (c) Time plots of fEPSP slope from adult control mice (black, n = 7) and paired slices bath applied with Nogo-P4 (4 µM, n = 7, red). (d) Quantification of change in fEPSP slope after 60 min following TBS normalized to baseline. Paired experiments are indicated by the line between the pairs where slices from the same mouse tested with vehicle or Nogo-P4. *p < 0.05 by paired t-test.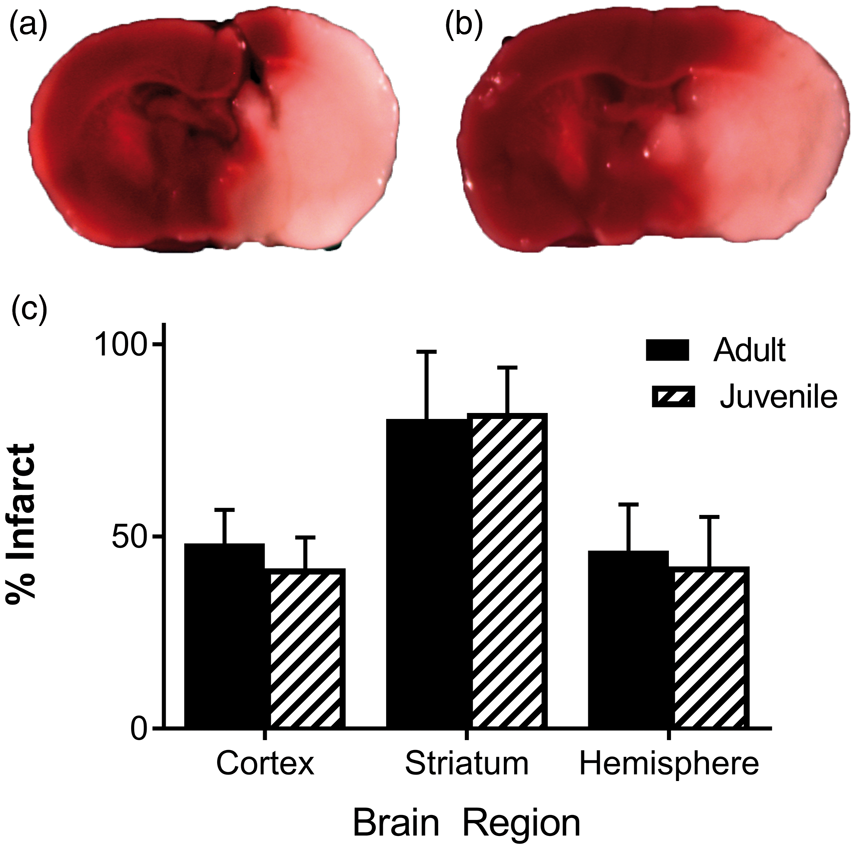
Discussion
This study presents evidence that transient focal ischemia impairs hippocampal memory and synaptic function in the sub-acute time frame in the developing brain. While this is consistent with recent findings in the adult brain after transient focal ischemia,7,29,30 we report here that juvenile mice showed remarkable recovery of hippocampal function compared to adults, exhibiting near complete recovery by 30 days after MCAO. We have been interested in delayed therapies to improve cognitive outcome after ischemia7,23 and given recent findings that Nogo-A is involved in restricting LTP, we hypothesized that Nogo-A signaling is involved in synaptic impairment after juvenile stroke. We show that MCAO causes increased astrocytic Nogo-A expression in the hippocampus, consistent with synaptic impairment by Nogo-A.17,28 Surprisingly, we observe MCAO-induced expression of Nogo-A specifically in juveniles, with no change observed in adult animals. Indeed, treatment with the Nogo receptor antagonist NEP(1–40) reversed synaptic plasticity impairment ex vivo in slices taken from juveniles seven days after MCAO, but not in adult slices. Therefore, increased Nogo receptor activity may be a mediator of impaired plasticity and memory specifically in the juvenile brain following MCAO. We are unaware of a commercially available Nogo-A receptor antagonist that reliably crosses the blood–brain barrier to test in vivo memory rescue after stroke. Nevertheless, our study suggests that inhibition of Nogo-A receptors is a potential therapeutic strategy to improve long-term functional recovery after childhood stroke.
The observation that synaptic function in the hippocampus is impaired after MCAO is important given that the hippocampus is vascularized by the posterior cerebral artery (PCA) and not the MCA. 31 We and others have previously shown that the hippocampus has no TUNEL-positive staining 24 h after MCAO surgery and adult animals,7,30 consistent with PCA vascularization. Consistent with these studies, we did not observe an infarct in the hippocampus using TTC staining. Further, differences observed in hippocampal deficits and recovery following MCAO are not related to the magnitude of cortical/subcortical injury. The data presented here are consistent with our recent study describing equivalent neuronal injury in the striatum of juvenile and adult mice in our 45-min MCAO model, 27 assuring that our model produces the expected injury in both age groups. Therefore, impaired LTP and memory deficits observed in the current study indicate that regions of the brain that remain uninjured following stroke can exhibit alterations in synaptic function. Hippocampal changes after MCAO have been well documented in human literature, among both adults and children. Ipsilateral hippocampal volume loss has been demonstrated in adult patients after unilateral MCAO, and has been shown to correlate to cognitive deficits after stroke.32,33 Interestingly, MR spectroscopy also shows neuronal changes in the contralateral hippocampus without gross volumetric loss,32,33 which is consistent with data from our group and others that functional changes in contralateral, as well as ipsilateral hippocampus after MCAO in adult rodents.7,34 Recently, hippocampal volume loss has also been demonstrated after pediatric patients with MCA territory stroke and has been shown to be age dependent. 35 The current study shows that ipsilateral LTP is also impaired in juveniles seven days post MCAO. The mechanisms of hippocampal structural and functional changes after MCA-territory stroke are unclear and are the subject of active ongoing research, which include changes in neurotransmitter receptors such as NMDA-R and GABA dysfunction.7,29 Indeed, our finding that MCAO impairs hippocampal LTP is consistent with the rich literature demonstrating hippocampal dysfunction following experimental ischemic stroke. The novel observation reported in the current study is that the young brain responds differently than the adult brain in terms of LTP and memory, and that the mechanisms of hippocampal dysfunction after stroke are likely different at different ages. Father studies elucidating age-dependent differences in hippocampal dysfunction after focal arterial ischemic stroke are needed.
The endogenous recovery we show in the young brain is a novel finding. Endogenous recovery of hippocampal function is an important observation in light of data suggesting survivors of childhood stroke suffer from considerable neurologic impairment, including impaired memory and cognitive function36–39 that can persist through school age. 40 Importantly, the prognosis following stroke in children is generally better than adult stroke, 41 which correlates with the data presented here. The spontaneous recovery observed following juvenile MCAO in the current study is in stark contrast to our recent report showing persistent impairment of LTP and memory deficits in the adult brain. 7 Interestingly, we recently reported a similar spontaneous recovery in the juvenile brain following global cerebral ischemia induced by cardiac arrest and CPR. 23 While we show near full recovery over a month's time, it is well accepted that several days in the young mouse brain correlate to several months to years of brain development in a child. 42 Therefore, we believe that our data indicate that strategies which accelerate recovery during a child's school age years have the potential to represent an important advancement in stroke rehabilitation.
Many compounds have been found to reduce brain injury when given in the acute phase following stroke. Unfortunately, the translation to clinical use has largely been unsuccessful. Recent studies have identified the need to move beyond neuroprotection as a therapeutic strategy and begin to target the impaired neurophysiology of surviving neurons after ischemia.1,7,23 Specifically, we have established that electrophysiology is an important tool to investigate cellular dysfunction in surviving neurons after stroke. This has allowed us to identify novel pathways to improve neuronal function.7,23 Here we identify a novel approach to improve functional recovery by targeting altered Nogo-A signaling in the hippocampus. This strategy restores hippocampal function in an age-dependent manner. Nogo-A is perhaps best known as playing an integral role in axonal guidance during the CNS development and maturation. Nogo-A signaling is known to be abundant during early development,12,15 with Nogo receptor (NgR1) expression peaking in the hippocampus during the first postnatal week. 43 Nogo-A is also known to be an inhibitor of neurite outgrowth in the adult CNS after injury. 15 Experimental data have demonstrated that anti-Nogo-A antibody therapy may be a potential treatment for improved motor outcome after stroke, owing to its effects on axonal growth.44,45 However, this mechanism does not explain the current results, as we observe effects within minutes in the acute slice preparation. Indeed, a more relevant role of Nogo-A to the present study is its role in attenuating LTP on a rapid time scale via NgR1 and intracellular signaling involving Lingo1.17,18,28 NgR1 has also been proposed to regulate memory formation. 16 More recently, it has been shown that acute Nogo-A signaling influences actin cytoskeleton dynamics to restrict dendritic spine plasticity and limit AMPA receptor insertion upon LTP induction.46,47 However, the pathological relevance for increased Nogo-A on synaptic function in the hippocampus is less clear, though the fast time scale in which Nogo-A acts on synaptic plasticity hints at different mechanisms than the slow time scale of action on neurite formation. We find that LTP and memory behavior are impaired after MCAO in young animals when Nogo-A expression is increased. Nogo-A is shown to be the causative agent by inhibiting NgR1 and reversing impairment of LTP. Further, we find that while adult mice have impaired LTP after MCAO, Nogo-A expression is not increased by MCAO and, therefore, it is not surprising that inhibiting NgR1 has no effect on reversing impaired synaptic plasticity. As an important control, we show that stimulating NgR1 in adult CA1 neurons impaired LTP, demonstrating intact Nogo-A-NgR1 signaling in the adult. Therefore, the lack of effect of NgR1 inhibition in the adult following MCAO is most readily explained by a lack of stroke-induced increase in Nogo-A levels. Together, our data indicate that Nogo-A plays an important role in impairing synaptic plasticity after stroke in the young brain, but not in the young adult, mature brain. The reasons for this are unclear. Interestingly, antagonists of Nogo-A signaling have also been observed to improve hippocampal-dependent memory function after stroke in aged animals when given chronically (eight-week treatment duration) beginning one week after ischemia. 48 This study observed that inhibition of Nogo-A improved memory function without stimulating structural dendritic or synaptic changes in the hippocampus, though axonal growth was not examined. Therefore, further studies assessing the role of Nogo-A in aged animals using electrophysiological analyses of synaptic function would be of great interest.
The fact that Nogo-A expression increases after brain injury is not necessarily surprising given its role in axonal refinement during the recovery phase. 15 Nogo-A has long been recognized to have a possible role in cortical axonal plasticity and motor impairments after stroke, 13 even when given at delayed time points, 14 delivered chronically over several weeks in adult and aged rats. 49 Nogo-A has also been shown to restrict dendritic arborization in organotypic hippocampal cultures 19 and may play an important role in stabilization of neuronal networks and compensatory circuits after injury. Previously, Nogo-A has been found on oligodendrocytes and neurons.12,15,50–53 Our data suggest an astrocytic source of Nogo-A in stratum radiatum of the ipsilateral hippocampus after juvenile MCAO, revealing a previously unidentified source of Nogo-A after ischemic injury in the hippocampus. Further, the hippocampus has a relative dearth of oligodendrocytes, but has been shown here and by others to have high Nogo-A and NgR1 expression, particularly at synapses, though without altering synaptic density.12,15 It has been suggested recently that Nogo-A is important in regulating the balance between synaptic plasticity and stability of neural circuits under physiologic conditions. 28 The data presented here suggest that Nogo-A activation may inhibit plasticity signaling after focal ischemia in the developing brain.
Conclusion
We provide the first direct evidence that the developing brain has the ability to recover hippocampal function after focal ischemia. We further describe the novel role of Nogo-A expression in the impairment of synaptic function in the juvenile brain following ischemia. Importantly, both the endogenous recovery of hippocampal function and the role of Nogo-A expression appear to be unique to the juvenile brain, as we provide evidence that these observations do not occur in the adult brain following focal ischemia, highlighting the need for critical appraisal of age as a variable after ischemia. Our strategy of delayed NgR1 inhibition several days after juvenile stroke affords the opportunity to reverse learning and memory impairments at time points well beyond the time of insult. In this way, we observe a therapeutic window that provides the promise to alter the learning potential of the developing brain long after what is provided by the current standard of care. Thus, this work establishes a new therapeutic strategy to reverse functional consequences of focal ischemia in the developing brain.
Supplemental Material
Supplemental material for Experimental pediatric stroke shows age-specific recovery of cognition and role of hippocampal Nogo-A receptor signaling
Supplemental Material for Experimental pediatric stroke shows age-specific recovery of cognition and role of hippocampal Nogo-A receptor signaling by James E Orfila, Robert M Dietz, Krista M Rodgers, Andra Dingman, Olivia P Patsos, Ivelisse Cruz-Torres, Himmat Grewal, Frank Strnad, Christian Schroeder and Paco S Herson in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgments
We thank Dr. Traystman for his assistance in early study design and critical revisions of early versions of the manuscript.
Author's contributions
James E. Orfila: Study conception and design, acquisition of data, analysis and interpretation of data, drafting of manuscript.
Robert M. Dietz: Study conception and design, acquisition of data, analysis and interpretation of data, drafting of manuscript, critical revision.
Krista M. Rodgers: Acquisition of data, analysis and interpretation of data, drafting of manuscript.
Andra Dingman: Acquisition of data, analysis and interpretation of data, drafting of manuscript.
Olivia P. Patsos: Acquisition of data, analysis and interpretation of data, drafting of manuscript.
Ivelisse Cruz-Torres: Acquisition of data, analysis and interpretation of data, drafting of manuscript.
Himmat Grewal: Acquisition of data, analysis and interpretation of data, drafting of manuscript.
Frank Strnad: Acquisition of data, analysis and interpretation of data, drafting of manuscript.
Christian Schroeder: Acquisition of data, analysis and interpretation of data, drafting of manuscript
Paco S. Herson: Study conception and design, analysis and interpretation of data, critical revision.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants RO1NS046072, RO1NS092645, 1K08NS097586, AHA/Bugher 14BFSC1769001, AHA 14GRNT18190012.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
