Abstract
Neuronal death due to ischemic stroke results in permanent deficits in sensory, language, and motor functions. The growth-restrictive environment of the adult central nervous system (CNS) is an obstacle to functional recovery after stroke and other CNS injuries. In this regard, Nogo-A is a potent neurite growth-inhibitory protein known to restrict neuronal plasticity in adults. Previously, we have found that treatment with monoclonal antibody (mAb) IN-1 to neutralize Nogo-A immediately after stroke enhanced motor cortico-efferent plasticity and recovery of skilled forelimb function in rats. However, immediate treatment for stroke is often not clinically feasible. Thus, the present study was undertaken to determine whether cortico-efferent plasticity and functional recovery would occur if treatment with mAb IN-1 was delayed 1 week after stroke. Adult rats were trained on a forelimb-reaching task, and the middle cerebral artery was occluded to induce focal cerebral ischemia to the forelimb sensorimotor cortex. After 1 week, animals received mAb IN-1 treatment, control antibody, or no treatment, and were tested for 9 more weeks. To assess cortico-efferent plasticity, the sensorimotor cortex opposite the stroke lesion was injected with an anterograde neuroanatomical tracer. Behavioral analysis demonstrated a recovery of skilled forelimb function, and anatomical studies revealed neuroplasticity at the level of the red nucleus in animals treated with mAb IN-1, thus demonstrating the efficacy of this treatment even if administered 1 week after stroke.
Introduction
Stroke is a prominent cause of serious, long-term disability (Bonita, 1992; Bonita et al, 1994) and the third leading cause of death in the United States (Minino et al, 2002). Total health costs for disability due to stroke in the United States are estimated at $53.6 billion annually (Morbidity and Mortality Chart Book on Cardiovascular, Lung, and Blood Diseases, 2004). Ischemic strokes comprise over 88% of all strokes, making them the most common type of cerebrovascular injury (Heart Disease and Stroke Statistics-2004 Update, 2004). Ischemic conditions in the brain cause neuronal death, leading to permanent sensorimotor and cognitive deficits. While thrombolytic therapy with a strong neuroprotective effect is sometimes administered up to 3 h after stroke, currently, there is no effective treatment for stroke after the acute stage (del Zoppo, 2000). Given that immediate treatment for stroke is often impossible in the clinical setting, the need for new treatment strategies for stroke is urgent.
A major obstacle to functional recovery after stroke is the growth-restrictive environment of the adult central nervous system (CNS), mainly due to neurite growth-inhibitory proteins, especially in myelin (see Schwab (2004) for review). Nogo-A is one of the known neurite inhibitory proteins present in myelin (Chen et al, 2000; Filbin, 2003; GrandPre et al, 2000; Prinjha et al, 2000; Schwab, 2004). Nogo-A, a protein product of the nogo gene, is a potent neurite growth inhibitor that is predominantly expressed in the CNS. In CNS myelin, confocal and immunoelectron microscopy have shown that Nogo-A is expressed in oligodendrocyte cell bodies and processes and is localized in the innermost adaxonal and outermost myelin layers (Huber et al, 2002). Nogo-A may exert its activity through stimulation of GTPase RhoA, which activates Rho kinase, or ROCK, and through elevation of intracellular calcium (Bandtlow, 2003; He and Koprivica, 2004; Schweigreiter et al, 2004). These proteins are known to regulate cytoskeletal changes through modulation of actin polymerization (Olson et al, 1995). Neutralization of Nogo-A promotes growth of axons in vitro (Oertle et al, 2003) and in vivo, leading to enhanced neural plasticity (Kartje et al, 1999; Thallmair et al, 1998), regeneration, and functional recovery (Brosamle et al, 2000; GrandPre et al, 2002). These findings illustrate its importance as a target for treatment of CNS injury.
Several studies have shown that recovery of function after injury is much better in the neonatal brain, presumably due to the formation of new neuronal connections, that is, structural plasticity. For example, functional reorganization of the unablated hemisphere occurs after unilateral motor cortical lesions in neonatal rats, due to the less restrictive environment of the neonatal CNS (Kartje-Tillotson et al, 1985, 1987). In neonatal rats receiving sensorimotor cortical lesion, the uninjured contralateral cortex forms bilateral projections to de-afferented subcortical targets from the opposite, unablated cortex (Kartje-Tillotson et al, 1986; Kolb et al, 1992; Leong and Lund, 1973; Naus et al, 1985; Rouiller et al, 1991; Yu et al, 1995). Importantly, remodeling of uninjured neuroanatomical pathways is accompanied by functional recovery after CNS injuries in young rats. However, re-routing of neural pathways in response to CNS lesions is less pronounced in adult rats after lesions, but can be facilitated by neutralizing antibodies against the neurite inhibitory protein Nogo-A (Wenk et al, 1999; Kartje et al, 1999).
Our laboratory has investigated antibody-mediated Nogo-A neutralization at the time of stroke. The monoclonal antibody (mAb) IN-1 is specific to the amino component of Nogo-A (Fiedler et al, 2002), that is, amino-Nogo/NiG, a potent inhibitory domain of Nogo-A (Niederost et al, 2002; Schweigreiter et al, 2004). Previously, we found that immediate treatment with the mAb IN-1 resulted in improvement of the impaired forelimb associated with the ischemic stroke (Papadopoulos et al, 2002). Furthermore, this improvement was associated with neuroanatomical plasticity from the uninjured cortex at the level of the red nucleus. The cortico-rubral pathway, part of the cortico-rubro-spinal system, has been implicated in precise limb movements, which are directly affected by lesions to the sensorimotor cortex (Jarratt and Hyland, 1999; Whishaw and Gorny, 1996). There is some evidence that delayed nogo-A neutralization after stroke results in functional recovery and neuroanatomical plasticity. For example, the administration of anti-Nogo-A antibodies specific to NiG 24 h after stroke has been shown to promote recovery of skilled forelimb movements as well as motor corticospinal tract (CST) remodelling (Wiessner et al, 2003). However, to date, it is unknown how long the time-to-treatment window with anti-Nogo-A antibodies can be extended. This information is of crucial importance from the clinical viewpoint when treating stroke patients in subacute or even chronic settings.
The purpose of the present study was to determine whether animals treated with anti-Nogo-A antibody at a later time point after stroke, that is, 1 week, would exhibit functional recovery and neuroplasticity. We show here that neutralization of Nogo-A 1 week after stroke in adult rats resulted in marked improvement of skilled forelimb function as well as corticorubral plasticity.
Materials and methods
Animals
Adult male Long Evans black-hooded rats (250 to 350 g, ages 10 to 12 weeks) (Harlan, Indianapolis, IN, USA) were divided into three experimental groups: (1) stroke plus mAb IN-1 (n = 9); (2) stroke plus control antibody (n = 4); (3) stroke only (n = 7). Animals were number-coded to ensure investigators were masked to treatment groups. Experiments were approved by the Joint Institutional Animal Care and Use Committee of Loyola University and Hines Veterans Affairs Hospital.
Skilled Forelimb-Reaching Task
Using a restricted diet of 12 g of rat chow per day, animals were reduced to 95% of their ideal body weight before testing; this diet was maintained throughout the testing period. The skilled forelimb-reaching task was used according to previous work to assess skilled motor functional recovery after stroke (Papadopoulos et al, 2002; Wiessner et al, 2003) and other injuries to motor pathways governing forelimb function (Z'Graggen et al, 1998). The testing apparatus consisted of a transparent Plexiglass chamber with an elliptical opening in one wall and a shelf attached outside the opening. Small sucrose pellets (45 mg; Research Diets, Inc., New Brunswick, NJ, USA) were placed one at a time onto the pellet shelf at a distance of 1.5 cm from the opening of the chamber. During a 2- to 4-week training period, limb preference was determined. To meet the criteria for inclusion in the study, rats had to reach, grasp, and bring to their mouth a minimum of 16 pellets on the first attempt (maximum of 20) in a 5-min period using the preferred limb. The results of the last three testing sessions were averaged together to establish a baseline value for each animal. After baseline was established, each animal received a middle cerebral artery occlusion (MCAO) and then was tested daily for 1 week to determine whether a deficit was present. Only animals that had a deficit signified by successfully reaching six or less pellets (≤37.5% of baseline) during a 5-min testing session were included in the study. Animals that remained in the study were tested daily and taped once weekly throughout the remaining 8 weeks until sacrifice.
Middle Cerebral Artery Occlusion
Strokes were induced via occlusion of the middle cerebral artery (MCA) as described previously (Chen et al, 1986), with some modifications. After training and baseline testing to determine limb preference, rats underwent an MCAO in the hemisphere contralateral to, and thus corresponding to, their preferred limb. Rats were anesthetized with sodium pentobarbital (50 mg/kg, intraperitoneally) and placed in a stereotaxic instrument. A craniotomy was made in either the right or left parietal bone with the use of an operating microscope. The resultant burr hole was extended to follow the distal branches of the MCA to where it transversed the rhinal sulcus. The MCA was permanently ligated with a 10-0 nylon suture and transected with microscissors. Then, the common carotid artery (CCA) ipsilateral to the occluded MCA was permanently ligated with a 4-0 silk suture, and the contralateral CCA was temporarily occluded with an aneurysm clip for 60 mins.
Antibody Application
Antibodies were applied in the same method as performed in previous work (Emerick et al, 2003; Papadopoulos et al, 2002; Z'Graggen et al, 1998) (see Figure 1). The mAb IN-1 was raised against the Nogo-A band from SDS-polyacrylamide gel electrophoresis separations of rat spinal cord myelin (Caroni and Schwab, 1988b). At 6 days after MCAO, animals with a stable deficit were reanesthetized with sodium pentobarbital (50 mg/kg, intraperitoneally), and a cell suspension (6 μL) containing a total of 1 × 105 mouse hybridoma cells secreting either mAb IN-1 or the control antibody, anti-horseradish peroxidase (anti-HRP) was stereotaxically injected with a Hamilton microsyringe into the hippocampus ipsilateral to the MCAO (coordinates: 4 mm caudal, 5 mm lateral and 5 mm ventral to bregma). This site was chosen to avoid damaging areas of motor cortex and to allow the antibody to reach the ventricular system and circulate in the CSF (Papadopoulos et al, 2002).
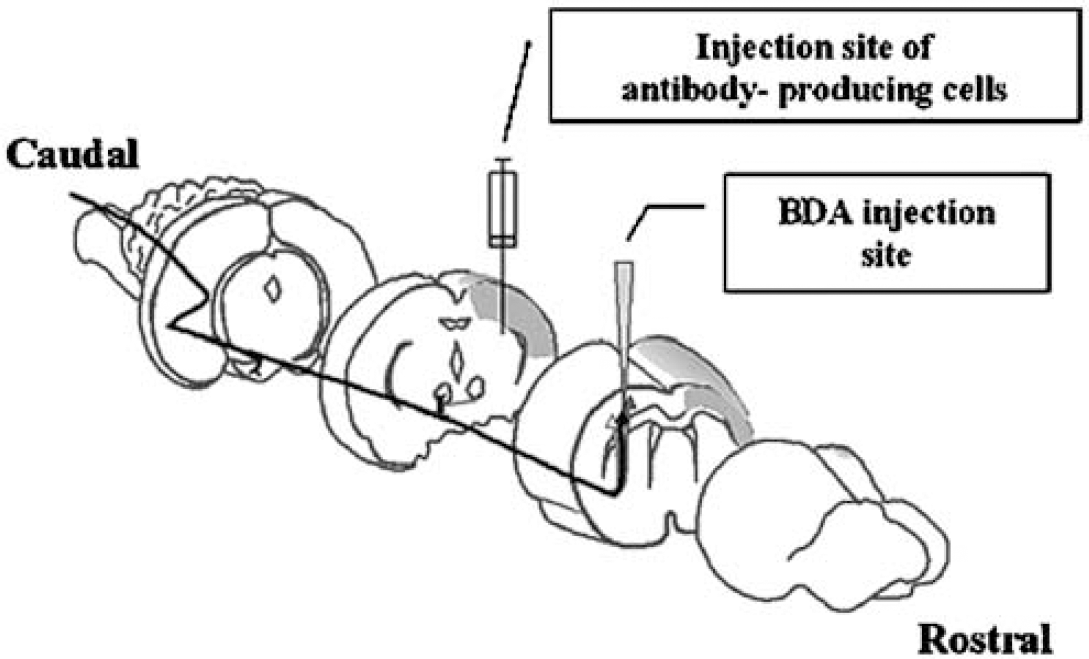
A schematic diagram illustrating the experimental procedures. Corticofugal fibers from the right forelimb motor cortex to the termination areas in the red nucleus are represented. In the left hemisphere is shown the stroke lesion (shaded) and the antibody-secreting hybridoma cell implantation site. The BDA tracer injection site is contralateral to the stroke lesion (adapted from Wenk et al, 1999).
These mouse hybridoma cells produce an IgM antibody against the rat neurite growth-inhibitory protein Nogo-A. As a control, an mAb raised against HRP (anti-HRP, an IgG antibody without inhibitory effect on HRP enzymatic activity) was generated from the same parent myeloma line. As a hybridoma line producing an IgM against HRP is not available, the IgG-producing hybridoma was used. Hybridoma cells were grown in Iscove's modified Dulbecco's medium using standard mammalian tissue culture techniques. Before implantation into the brain, hybridoma cells were tested for inhibitory neutralizing activity in a bioassay using neurons on a myelin-protein substrate, and were tested for antibody production by FITC-coupled anti-mouse antibodies. The stroke-only group of rats received vehicle only, that is, media, at the same injection site. All animals received cyclosporin A (10 mg/kg, intraperitoneally) daily starting 1 day before, and continuing for 7 days after cell implantation, to prevent rejection of the hybridoma xenograft; stroke-only animals also received cyclosporin A according to the same schedule. Based on this paradigm, hybridoma cells survive for approximately 2 weeks and continue to secrete antibody during such time. According to our past experience and published reports, antibody-secreting hybridoma cells given as treatment will continue to secrete antibody for at least 2 weeks after implantation in the immune-suppressed animal, and this is equal between the mAb IN-1 and anti-HRP antibody-secreting hybridomas (Emerick and Kartje, 2004; Schnell and Schwab, 1990; Thallmair et al, 1998; von Meyenburg et al, 1998; Wenk et al, 1999; Z'Graggen et al, 1998).
Biotinylated Dextran Amine Tracing
After the period of behavioral observation and data collection (9 weeks), all rats were re-anesthetized with sodium pentobarbital (50 mg/kg, intraperitoneally) and placed in a stereotaxic instrument. A craniotomy was made to expose the sensorimotor cortex of the hemisphere contralateral to the stroke lesion (Figure 1). Using a Hamilton microsyringe, 1 μL of a 10% solution of the anterograde tracer biotinylated dextran amine (BDA, Molecular Probes, Eugene, OR, USA) was injected at two sites (1 to 3 mm lateral to bregma, at a depth of 1.5 mm from the cortical surface) in the uninjured caudal forelimb motor cortex as described by Neafsey et al (1986). At 2 weeks after BDA injection, rats were overdosed with sodium pentobarbital and transcardially perfused with saline followed by 4% paraformaldehyde.
Neuroanatomical Analysis
Anatomical structures were identified using the atlas of Paxinos and Watson (1998). Brains were removed, postfixed overnight in 4% paraformaldehyde, cryoprotected in 30% sucrose, and kept at -80°C before processing. Tissue was cut on a Leica cryostat in serial 50 μm coronal sections and reacted for BDA-positive fibers by the semi-free-floating method (Herzog and Brosamle, 1997); alternate sections were processed for Nissl stain and analyzed for lesion size and location. All slides were coded to ensure investigators were blind to treatment groups. Coronal sections were analyzed by computer-aided image analysis using NIH Image version 1.51 (National Institutes of Health, Bethesda, MD, USA). A Macintosh computer (Centris 650) interfaced with a digitizing board (Data Translation, Marlboro, MA, USA) to a digital camera (model MOS VK-C150; Hitachi; Brisbane, CA, USA) was used with a Leitz DMR microscope.
Quantification of Cortico-Rubral Fiber Number
Biotinylated dextran amine-positive fibers at the level of the parvocellular red nucleus were analyzed by calculating ratios of BDA-labeled fibers between red nuclei in each hemisphere using an adaptation of a previously described method (Emerick and Kartje, 2004). Using five consecutive sections for each animal, a 2500 μm2 grid was placed in four standardized locations within the red nucleus in each hemisphere and fibers present within the grid were counted and totalled, yielding an estimate of fiber density within the red nucleus in each hemisphere. To provide an internal control for fiber tracing, the total fiber count in the red nucleus within the lesioned hemisphere was divided by the total fiber count in the red nucleus within the unlesioned hemisphere to yield the cortico-rubral fiber ratio. Histological data from three stroke/control Ab and two stroke-only animals were combined after a Student's t-test showed no significant difference between these controls.
Stroke Size Analysis
The volume of the stroke lesion for each animal was quantitatively analyzed on Nissl-stained sections, according to the atlas of Paxinos and Watson (1998) (+4.7 to -5.2 mm from bregma). Using the method described by Kawamata et al (1997), the area of the damaged hemisphere was subtracted from the area of the intact hemisphere; this value was divided by the area of the intact hemisphere, then multiplied by 100 to give stroke lesion volume as a percent of the intact hemisphere. Stroke size data were omitted for three animals: two in the stroke/mAb IN-1 group and one in the stroke-only group.
Statistics
Analysis of all data was performed with SPSS version 12.0. (SPSS, Inc.). For behavioral data, a repeated-measures analysis of variance (ANOVA) with a Tukey HSD post hoc test was used to test for overall significance, differences in initial deficits, and to determine the time points at which the predicted recovery differed among the groups. For anatomical data, a Student's t-test was used to compare the mean fiber ratios at the level of the red nucleus between stroke/mAb IN-1-treated rats and control rats. For all analyses, P<0.05 was considered significant. All data are presented as mean values±standard error of the mean (s.e.m.).
Results
Functional Recovery in Animals Treated with Monoclonal Antibody IN-1 One Week after Stroke
To determine whether blockade of the neurite inhibitory protein Nogo-A 1 week after stroke resulted in functional recovery, rats were tested on a skilled forelimb-reaching task (Figure 2). At baseline testing before stroke, all animals achieved the same success score in grasping pellets with no significant difference between groups (P = 0.21). At 1 week after stroke, all the three groups showed a severe deficit in pellet grasping (mean of two pellets, or 11% of baseline), with no significant differences between groups (P = 0.63). By 5 weeks after stroke, rats treated with mAb IN-1 had significantly greater mean pellet-reaching success scores compared with the stroke/control antibody group (P<0.05), but not compared with the stroke-only group. At week 7 and continuing through week 9 after stroke, animals treated with mAb IN-1 had significantly greater mean pellet-reaching success than both control groups (weeks 7 and 8 after stroke, P<0.05; week 9 after stroke, P<0.001). There were no significant differences between the stroke/control Ab and the stroke-only groups at any time point. In addition, at nine weeks after stroke, the last time point tested, animals in the stroke/mAb IN-1 group were able to successfully grasp a mean of 12.7 pellets, or 75% of their baseline success level. However, rats receiving control antibody or stroke only were only able to grasp a mean of 5 (26% of baseline) and 6 (29% of baseline) pellets by 9 weeks after-stroke, respectively.
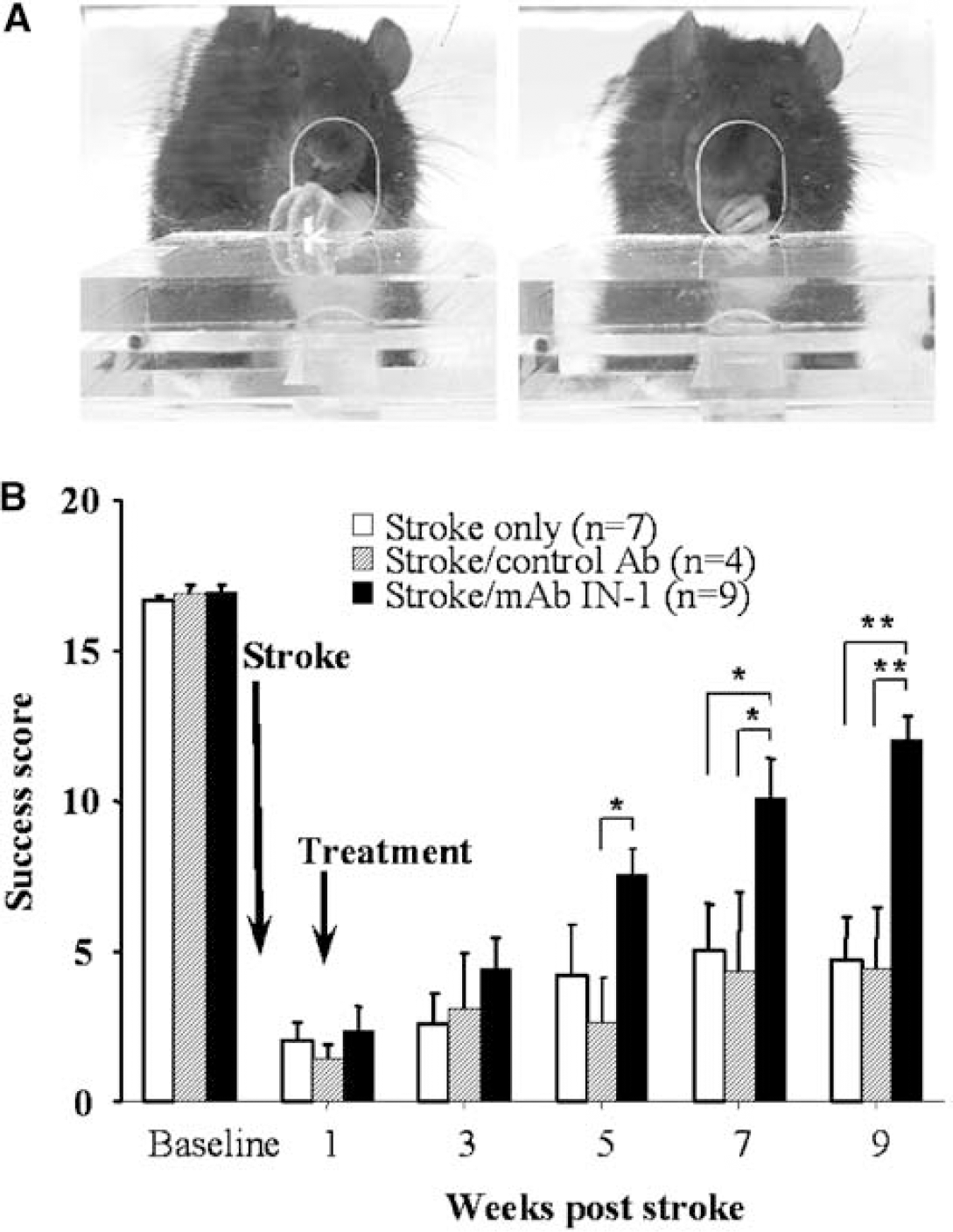
The skilled forelimb-reaching task. (
Quantification of Stroke Lesions in the Sensorimotor Cortex
All stroke lesions were localized in the sensorimotor cortex ipsilateral to the MCAO (Figure 3). Analysis of stroke size showed no stroke volume difference between groups (stroke/mAb IN-1 = 10±3%; all controls = 14±4%; P = 0.25).
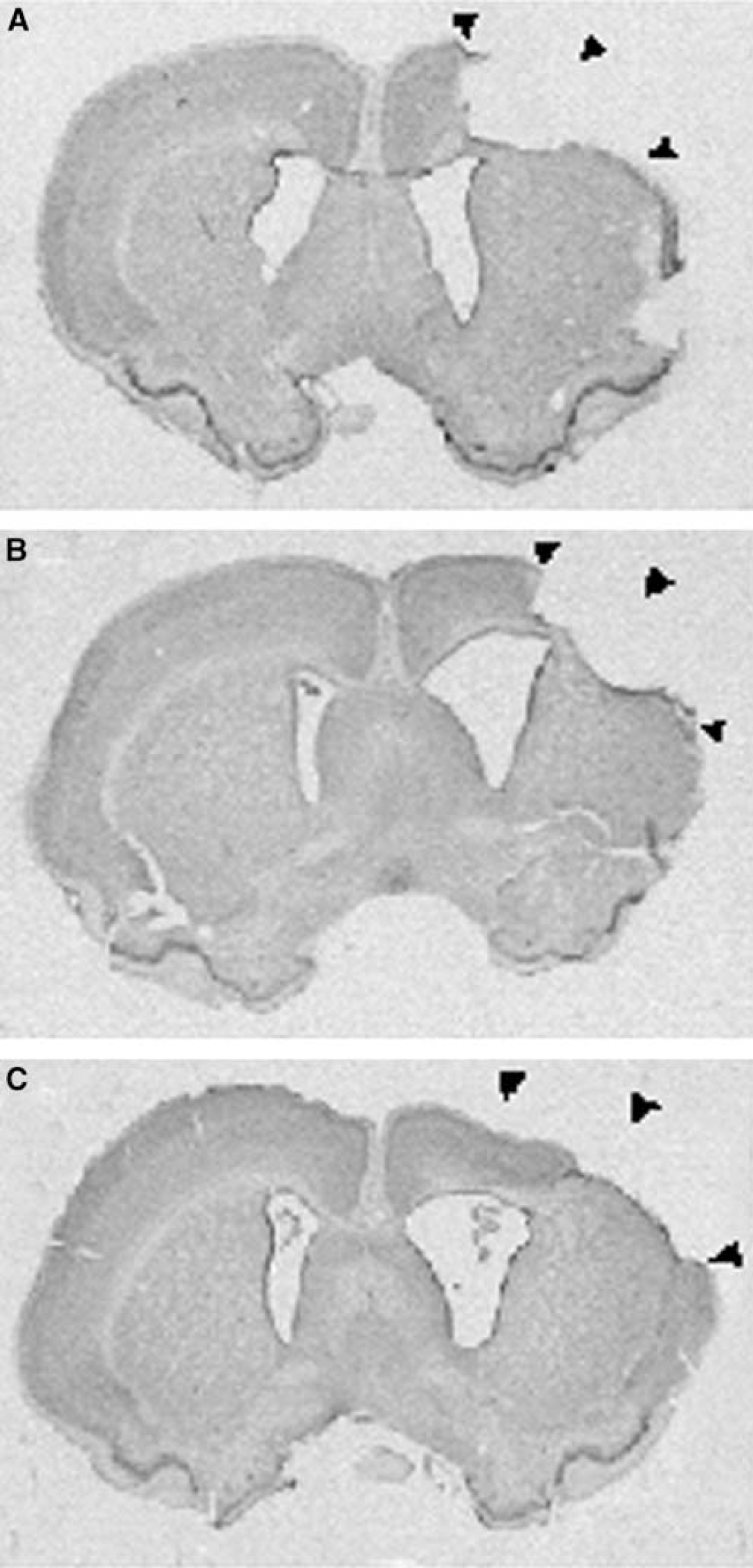
Representative photomicrographs of ischemic stroke lesions in the sensorimotor cortex. (
Cortico-Rubral Plasticity in Animals Treated with Monoclonal Antibody IN-1 1 Week after Stroke
To relate behavioral changes to the development of new cortico-efferent pathways, the cortico-rubral projection from the spared hemisphere was examined with the anterograde neuroanatomical tracer, BDA. In the intact CNS, the corticorubral pathway is primarily an ipsilateral projection with a minor contralateral component. In all groups, the parvocellular region of the red nucleus on the BDA-injection side showed more dense BDA-positive fibers than on the contralateral side (Figures 4A and Figure 4B), as expected. However, mAb IN-1-treated rats appeared to have a higher density of BDA-positive fibers in the contralateral de-afferented red nucleus when compared with control groups. Quantitative analysis confirmed these results and showed significantly higher corticorubral fiber ratios in stroke/mAb IN-1 animals as compared with controls (P<0.05) (Figure 5B). Nissl-counterstained sections also displayed BDA-positive fibers with varicosities in close apposition to cell bodies in the de-afferented red nucleus (Figure 4C).
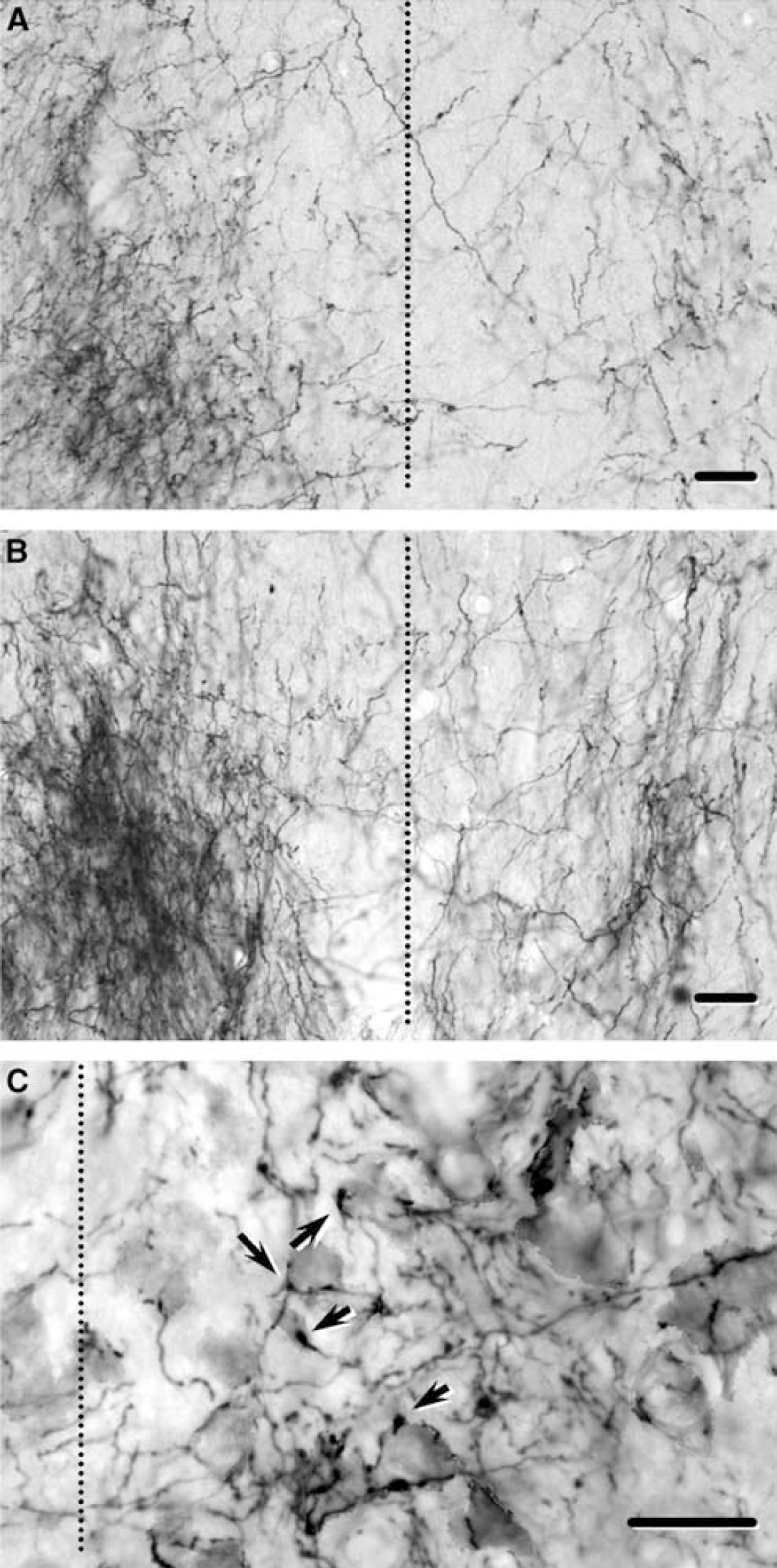
Biotinylated dextran amine-positive fibers in the red nucleus in control rats (
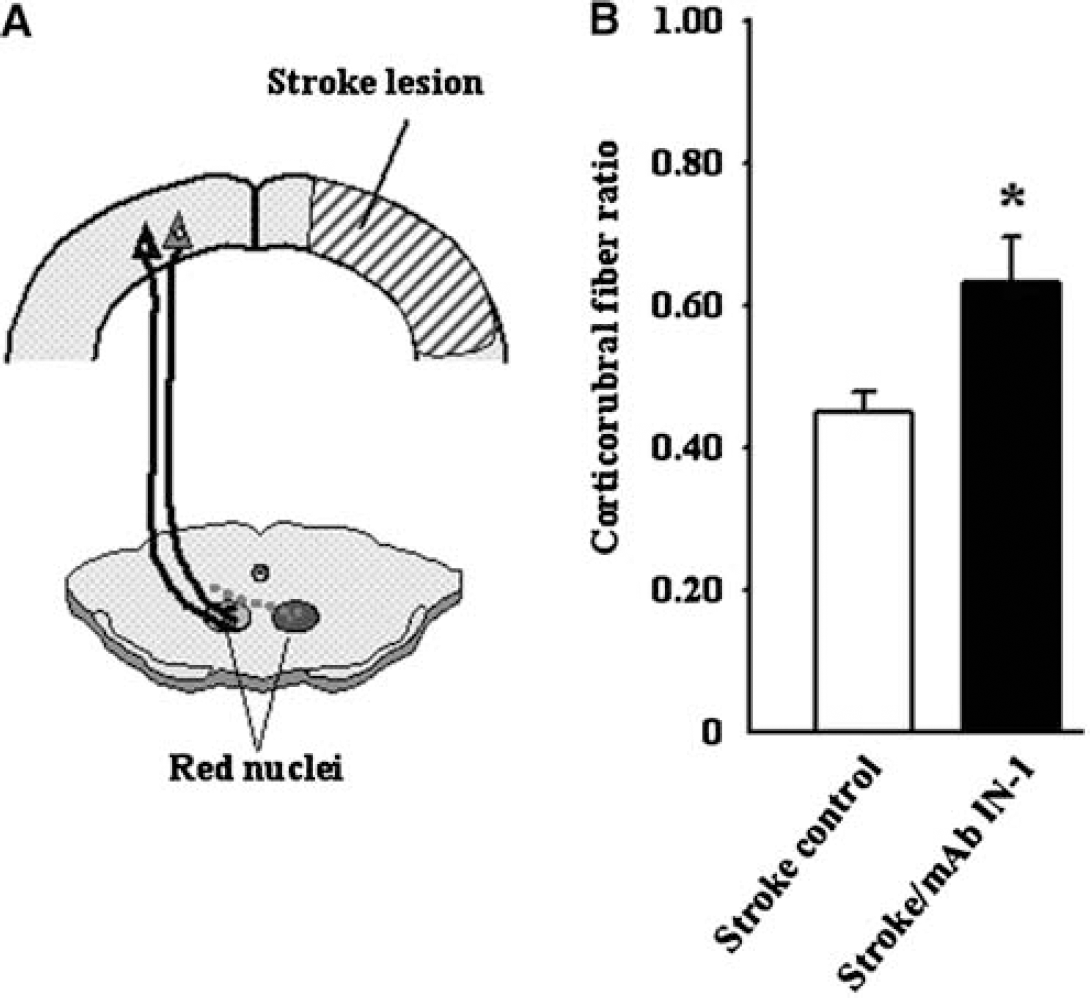
Rats treated with mAb IN-1 1 week after stroke show increased cortico-rubral plasticity, compared with controls. (
Discussion
Our results show that intracerebral treatment with the mAb IN-1 to neutralize Nogo-A 1 week after stroke in adult rats promotes recovery of skilled forelimb function as measured in a forelimb task, requiring fine digital dexterity. Furthermore, these recovered rats demonstrated cortico-efferent plasticity, as shown by increased corticorubral fibers in the de-afferented red nucleus. This indicates that, with the proper treatment, it is possible to unlock the capacity for growth and repair of the adult injured CNS. Our results show that this capacity is retained at least 1 week after ischemic stroke.
Knowledge of the growth-inhibitory character of the adult CNS environment is derived from studies showing that neutralization of the myelin-associated neurite growth inhibitor Nogo-A decreases the inhibitory activity of myelin (Brosamle et al, 2000; Caroni and Schwab, 1988a; Chen et al, 2000; Fiedler et al, 2002). We have previously shown that, by neutralizing Nogo-A with the mAb IN-1 at the time of stroke, neurons from opposite, spared forelimb motor cortex projected to de-afferented areas in the lesioned hemisphere, a process associated with functional recovery (Papadopoulos et al, 2002). In the present study, at 9 weeks after stroke, rats administered mAb IN-1 1 week after stroke demonstrated ~75% recovery of forelimb function, as measured by a skilled forelimb-reaching task. Similarly, Papadopoulos et al (2002) found that at 8 weeks after stroke, rats that had been administered mAb IN-1 immediately after stroke demonstrated 80% recovery of forelimb function on the exact same task. Interestingly, in the present study, animals administered mAb IN-1 1 week after stroke showed significant recovery at 5 weeks after treatment, whereas animals administered mAb IN-1 at the time of stroke (Papadopoulos et al, 2002), showed significant recovery a week later, that is, 6 weeks after treatment. Therefore, delaying treatment with Nogo-A blockade 1 week after stroke may actually shorten the time to recovery, while the level of recovery appears to be unchanged with such a delay. Studies comparing various treatment timepoints are underway to determine the optimal time-to-treatment window. In a related study using a specific anti-Nogo-A antibody 24 h after stroke in adult rats, functional recovery in skilled forelimb use on the Montoya Staircase task was seen by 6 weeks after stroke (Wiessner et al, 2003). In that study, increased numbers of CST fibers originating in the uninjured motor cortex were found to sprout across the midline in the cervical spinal cord in animals treated with anti-Nogo-A antibody. Further, similar to the work by Papadopoulos et al (2002), increased axonal sprouting was correlated to improved successes in the reaching task. A similar study in rats targeted the Nogo-66 receptor (NgR), a common receptor for the Nogo-66 inhibitory domain of Nogo-A, oligodendrocyte myelin glycoprotein/Omgp, and myelin-associated glycoprotein/MAG (Filbin, 2003; Lee et al, 2004). This study reported functional recovery and cortico-efferent plasticity when an antagonist to NgR was administered continuously for over 2 months beginning 7 days after stroke. Interestingly, NiG, a potent inhibitory domain of Nogo-A to which the mAb IN-1 binds, has recently been shown to mediate growth inhibition independent of NgR, and the binding site for NiG is physically distinct from that of Nogo-66 on neural tissue (Schweigreiter et al, 2004). Therefore, neutralization of Nogo-A with specific antibodies targeting NiG remains a powerful potential treatment option to promote cortico-efferent plasticity and functional recovery after ischemic stroke.
In addition to behavioral and neuroanatomical evidence of the capacity of the adult CNS to recover lost function after cortical injury and anti-Nogo-A treatment, our laboratory has shown that rats administered mAb IN-1 at the time of motor cortex injury displayed functional reorganization of the opposite unablated cortex (Emerick et al, 2003). In this work, intracortical microstimulation of the non-injured motor cortex resulted in a significant increase in movements of the lesion-impaired forelimb in animals treated with mAb IN-1. These studies indicate that anti-Nogo-A treatment after unilateral cortical injury provides a permissive environment in which new growth from the uninjured cortex results in functional connections that restore motor movements.
Other processes may play important roles in functional reorganization after focal ischemic stroke. For instance, changes in neuronal excitability due to reductions in binding of gamma amino butyric acid (GABA)-A receptors (Qu et al, 1998), as well as increases in dendritic spine density and growth, and synaptogenesis (Jones et al, 1996) have been found after unilateral ischemic lesions. Stroke lesions have been shown to induce the expression of growth-promoting factors, for example, brain-derived neurotrophic factor (BDNF), basic fibroblast growth factor (bFGF), and growth-associated molecules, for example, growth-associated protein-43 (GAP-43) (Nudo, 1999). Blockade of one of these factors, bFGF, after cortical lesions in the rat resulted in decreased spontaneous recovery in the affected forelimb as measured by the skilled forelimb-reaching task (Rowntree and Kolb, 1997). The adult CNS thus retains a repertoire of growth- and repair-related responses needed for new axonal growth and synapse formation. These studies indicate that these growth-related factors might be peaking in the early weeks after injury. Therefore, treatment with mAb IN-1 1 week after stroke might be an opportune time to take advantage of the growth-promoting microenvironment.
Related work has shown that at 1 week after a high unilateral lesion of the CST in the rat without treatment, an increase in gene expression of growth factors, extracellular matrix proteins, and axonal guidance molecules, in addition to compensatory sprouting from the uninjured CST, was found in the spinal cord (Bareyre et al, 2002). Rats administered mAb IN-1 at the time of CST lesion had increased target-specific, stable compensatory sprouting within the spinal cord. Anti-Nogo-A treatment was associated with increased gene expression of growth-associated proteins, including GAP-43, BDNF, and insulin-like growth factor (IGF), but also with increased expression of cytoskeletal proteins. Taken together, these results suggest that at least two mechanisms must be present for a meaningful compensatory reorganization of the injured CNS. The first is the release from inhibition resulting from effects of Nogo-A neutralization, and the second is the denervation-induced modulation of guidance molecules and growth-related factors that allow fibers to reinnervate denervated targets in an organized manner.
Functional recovery after CNS lesions in the perinatal period is associated with a high degree of neuroanatomical plasticity. For example, unilateral cortical lesions in the neonatal rat results in the development of bilateral connections with the striatum (Kolb et al, 1992), thalamus (Yu et al, 1995), red nucleus (Naus et al, 1985), tectum, basilar pontine gray (Kartje-Tillotson et al, 1986), and spinal cord (Rouiller et al, 1991). These new, lesion-induced pathways are thought to be the underlying substrate for functional recovery after perinatal injury. Interestingly, we have found similar structural plasticity after unilateral cortical lesion in adult rats followed by immediate anti-Nogo-A therapy (Emerick and Kartje, 2004; Kartje et al, 1999; Papadopoulos et al, 2002; Wenk et al, 1999). In the present study, using delayed anti-Nogo-A blockade, we again found structural plasticity in the red nucleus, a well-established subcortical target of the forelimb motor cortex, and an important nucleus for motor control.
In this regard, special attention has been paid to the cortico-rubral tract and the cortico-spinal tract, pathways which undergo neuroanatomical remodeling after motor cortex lesions (Iwaniuk and Whishaw, 2000; Papadopoulos et al, 2002) or CST lesions (Raineteau et al, 2001, 2002) after anti-Nogo-A treatment. The cortico-rubral pathway in the adult rat is mainly an ipsilateral projection with a very minor contralateral contribution (Naus et al, 1985; Z'Graggen et al, 1998). Although the CST directly controls fine finger and hand movements in humans, monkeys, and rats, the cortico-rubro-spinal system is also implicated in precise limb movements, a function that parallels that of the CST (Naus et al, 1985). While selective lesions of the rubro-spinal tract induce only slight deficits in reaching, when the motor cortex is removed in addition to portions of the rubrospinal tract, greater deficits are seen (Whishaw et al, 1990). Electrophysiology studies have shown that, with regard to the distribution of onsets of neuronal spiking, neuronal activity in the rubro-spinal tract during a skilled forelimb-reaching task closely resembles that of the CST (Hyland, 1998; Jarratt and Hyland, 1999). Neuroanatomical data from the present study show that, compared with controls, animals administered mAb IN-1 1 week after stroke had an increase in BDA-positive fiber density in the de-afferented red nucleus. The extent of neuroanatomical remodeling observed in the present study does not appear to correspond with the remarkable behavioral recovery on the skilled forelimb-reaching task after stroke and delayed mAb IN-1 treatment. It is not known what quantity of fiber growth within or towards de-afferented regions is necessary to facilitate functional recovery. Our studies would suggest that a complete pathway is not needed to show some return of function after a CNS lesion (Papadopoulos et al, 2002). As our animals did not completely recover, perhaps more fiber sprouting would have increased recovery.
Anti-Nogo-A treatment to promote axonal regeneration and functional recovery was first demonstrated in vivo after spinal cord injury in a set of seminal studies (Bregman et al, 1995; Schnell and Schwab, 1990). When treatment with the mAb IN-1 and neurotrophin-3 (NT-3) was delayed for 2 weeks after spinal cord injury, rats exhibited improvement in functional recovery, and regenerating CST fibers were found (von Meyenburg et al, 1998). This was one of the first studies to show that transected adult corticospinal axons retain the ability to regenerate at least a few weeks after injury. In another study, delayed blockade of NgR 1 week after spinal cord injury was shown to promote functional recovery and axonal outgrowth in the spinal cord (Li and Strittmatter, 2003). Thus, delayed anti-Nogo-A therapy is a promising treatment to enhance recovery after CNS injury. In conclusion, if the degree of debilitating functional impairment caused by stroke can be minimized through anti-Nogo-A therapy administered in a more practical time window after ischemia, then a new era of stroke treatment options may evolve.
Footnotes
Acknowledgements
Special thanks goes to Josh Rosales for his help in preparing figures.
