Abstract
The cerebral microvasculature consists of pial vascular networks, parenchymal descending arterioles, ascending venules and parenchymal capillaries. This vascular compartmentalization is vital to precisely deliver blood to balance continuously varying neural demands in multiple brain regions. Optical imaging techniques have facilitated the investigation of dynamic spatial and temporal properties of microvascular functions in real time. Their combination with transgenic animal models encoding specific genetic targets have further strengthened the importance of optical methods for neurovascular research by allowing for the modulation and monitoring of neuro vascular function. Image analysis methods with three-dimensional reconstruction are also helping to understand the complexity of microscopic observations. Here, we review the compartmentalized cerebral microvascular responses to global perturbations as well as regional changes in response to neural activity to highlight the differences in vascular action sites. In addition, microvascular responses elicited by optical modulation of different cell-type targets are summarized with emphasis on variable spatiotemporal dynamics of microvascular responses. Finally, long-term changes in microvascular compartmentalization are discussed to help understand potential relationships between CBF disturbances and the development of neurodegenerative diseases and cognitive decline.
Keywords
Introduction
Brain function is maintained by a continuous supply of cerebral blood flow (CBF). Reductions or cessation in CBF lead to irreversible and sometimes lethal brain damage. Not surprisingly, milder long-term cerebral hypoperfusion can lead to future cognitive decline. 1 Studies in animals indicate that cognitive impairment is associated with cerebrovascular dysfunction, in particular neurovascular uncoupling. 2 Neurons, the primary computation element in the brain, consume large amounts of energy essentially for propagating action potentials and maintaining postsynaptic ion fluxes. 3 These energetic demands are primarily met by the delivery of blood oxygen and glucose. As neurons work simultaneously across the brain to execute numerous functions, CBF is spatiotemporally modulated accordingly to balance local needs over a broad range of activity levels. 4 This supply and demand coupling is artfully coordinated by cell-to-cell communications among neurons, glia, and vascular cells, collectively called the neurovascular unit (for recent reviews, see literature5–9)
The combination of different physiological systems that compose the neurovascular unit makes it intrinsically complex. Imaging systems like positron emission tomography (PET) and functional magnetic resonance imaging (fMRI) have been crucial to our understanding of brain physiology. For example, increases in neuronal activity lead to regional increases in blood flow that exceed tissue oxygen consumption but not glucose metabolism. 10 Although the necessity of these disproportionate changes remains somewhat unclear, the magnitude of the changes in blood flow (and metabolism) is tightly correlated to the changes in neuronal activity. 11 Optical methods are proving to be essential tools for piecing together the dynamics and signaling pathways underlying neurovascular regulation.
Many techniques can be used to capture the dynamics of cerebral vasculature. In this article, we focus on in vivo optical studies that have helped shape our understanding of cerebral microvascular responses at the systemic and local levels, such as intrinsic optical imaging, fluorescence microscopy, two-photon microscopy and optogenetics. To this end, we briefly review cerebrovascular anatomy focusing on the cortical microvasculature. Then, we review cerebral microvascular responses to global perturbations (e.g. blood gas and blood pressure changes), followed by cerebral microvascular responses to local neural activation. Optical studies employing transgenic mice encoding optogenetic actuators in specific cell targets are providing invaluable insight into the cellular underpinnings of microvascular responses. Recent studies based on these animal models have found separately that excitatory and inhibitory neurons are able to evoke cerebrovascular responses with somewhat different temporal properties.12–15 Moreover, optogenetic modulation of astrocytes induced spatially propagated vascular responses that are comparable to photo-activation to excitatory neurons.16–18
Cerebrovascular responses to global systemic perturbation
In the cerebral cortex, the vascular system can be divided into two-independent compartments (i.e. pial and parenchymal vascular systems) according to location, morphology, and neural innervation. Pial arteries and veins are situated in the arachnoid spaces of the cortical surface. Parenchymal arterioles and venules branch from the pial arteries and veins, respectively, and dive into the parenchyma, referred to as descending arterioles and ascending venules. Those arterioles and venules further branch off at different cortical layers, approximately every 60 µm and 25 µm depths from the cortical surface measured in the mouse cortex, respectively, 19 and eventually all connect with parenchymal capillaries.20,21 Parenchymal capillaries are all microvessels whose diameter are smaller than the outer diameter of red blood cells (RBCs). It is noted that pial capillaries are not detected in the normal adult rodent brain,22,23 because all arteries eventually dive into parenchyma (i.e. arterio-venous shunts are not commonly observed on the cortical surface). Parenchymal vessels are also typically surrounded by astrocytic end-feet 24 and by pericytes with varying morphologies, 25 and observed to respond local neural demand (see following section of the parenchymal vessel response to local neural demand). In this article, we call microvessels those with caliber less than approximately 0.3 mm where the apparent hematocrit decreases with decreasing vessel diameter due to the faster traffic of RBCs relative to blood plasma, known as the Fåhræus–Lindqvist effect. 26 In these microvessels, flow dynamics are dominantly influenced by the viscosity of the fluid relative to its inertial force (i.e. small Reynolds number; <0.2). 27 It should be noted that parenchymal vessels are constructed and operate in this regime (i.e. <240 µm in human cortex 28 and <80 µm in rat cortex 29 ). Independent of vascular diameter, parenchymal blood flow can vary depending on arterial hematocrit (the volume ratio of RBCs),30,31 viscosity of blood plasma,32–34 deformability of the RBC,35,36 and endothelial surface layers (i.e. mechanical and electromechanical properties of glycocalyx on the endothelial surface).37,38
Pial vascular responses
Pial arteries and veins are located on the cortical surface and play an important role in balancing the blood supply among the various cortical regions.39,40 Pial arteries can form network structures, called arterio-arterial anastomosis that bridge together arteries of different major arteries. 41 This unique characteristic can allow bidirectionality of blood flow from different parent arteries, 42 allowing for a regional balancing of blood supply. 43 Although it has been shown that anastomosis, also known as collateral flow routes, can prevent and rescue ischemic damage once a major supply vessel is disrupted, 44 its role as an active or passive participant in blood flow regulation is not known.
It was hypothesized that a higher density of pial arterial routes acquired with phylogenic evolution allows for a more precise regulation of blood supply to smaller regions of the cortex and that in developed animals, these facilitate the organization of smaller neural units. 40 Considering that the brain continually processes information across multiple cortical regions with ever-changing levels of activity, these regions experience varying degrees of energetic demands. Hence, it can be inferred that pial arteries and their anastomosis help maintain parenchymal microvascular pressure constant across different brain regions. This organization could also prevent blood stealing during focal increases in blood flow to a particular cortical region. 45 Mathematically, blood flow is generated by a pressure gradient along the vessels. If bidirectional flow exists somewhere in the pial arterial networks, this implies that the pressure difference among the pial arterial segments is small. In line with this view, it was shown that pial arterial pressures fall within a 30 mm Hg range with diameters between 455 and 25 µm.46–48 These in vivo measurements suggest that a ‘uniform pressure reservoir’ may exist in pial arterial networks.
Pial arteries and veins respond to systemic changes in blood gas (e.g. concentrations of dissolved oxygen and carbon-dioxide gasses)49–52 and arterial blood pressure.53,54 Direct examination of vessel distensibility in isolated human pial arteries (200 to 1200 µm in diameter) showed that the myogenic tone of the pial arteries increases seemingly in uniform fashion with decreases in the inner vessel diameter. 55 Nevertheless, as previous researchers have hypothesized, a dual CBF control theory appears to be present where the reaction of the pial arteries to hypercapnia and autoregulation differs depending on vessel size. 54 In response to carbon dioxide inhalation, animal experiments in vivo consistently show that smaller sized pial arterioles dilate to a greater extent than larger ones,50,51,56–58 which suggest that smaller arterioles have more tone and greater vascular reserve. In contrast, no detectable changes in diameter of pial vein were observed for hypercapnia, 50 but other studies have reported significant dilation in smaller pial veins.51,58 This could result from increases in venous pressure and venous blood velocity to match the increases in arterial blood flow. 54 For changes in systemic arterial blood pressure, segment-dependent dilation and constriction of pial arteries (30–395 µm in diameter) have been observed in response to hypertension (<200 mmHg). 47 Because pial arterial responses to intravascular pressure were independent of arterial blood gas and tissue metabolite concentrations, a neurogenic control of pial arterial responses (e.g. via extrinsic neural innervation) was also postulated. 59
Regulation of the vascular tone at the level of pial vessels is not always associated with parenchymal vessels, such as penetrating arteries which have significant influence on regional CBF. 60 The sphincters at the off-shoots of small arteries from pial arteries that dive into the cortex have extensive innervation with cholinergic and adrenergic nerves, but less so for vessels forming arterio-arterial anastomoses. 61 In pre-cortical and pial arteries, nerve fibers run along the penetrating arteries and their innervation is known as extrinsic innervation (for reviews, see Hamel 62 ). This anatomical feature highlights a non-local means of pial arterial tone modulation that can be independent of regional CBF. In the cat brain, sympathetic stimulation induced a pial arterial constriction without affecting CBF. 63 A similar dissociation between the pial arterial diameter responses and parenchymal perfusion has been reported with the topical application of vasoactive agents, 64 cortical spreading depression, 65 and neurovascular coupling after adaptation to chronic hypoxia. 66 These reports suggest that the dissociation of the responses between the pial and penetrating arteries is present under certain conditions.66,67 For example, studies with isolated preparations of pial and penetrating arteries show dilation or constriction responses to noradrenaline application are present for penetrating arteries, but not for pial arteries. 60 Because penetrating arteries serve as a bottle neck to feed parenchymal tissue, 68 impaired vasodilation at this level can cause insufficient tissue perfusion. Collectively, it can be inferred that pial arteries balance a rather wide CBF level over broad cortical regions, whereas penetrating arteries ensure focal supply of CBF to meet regional neural demand arising in parenchymal tissue.
Parenchymal vascular responses
Descending arterioles and ascending venules, also called radial vessels, are oriented perpendicular to the cortical surface. Descending arterioles branch from pial arteries and shortly dive into the parenchymal tissue. This unique branching structures (i.e. T-junctions; a vertical branch from pial to the cortex) may interrupt natural flow, but appear to be very effective controlling blood supply to their district territories.42,68 Visualization of the dynamic of these parenchymal microvessels in vivo was not possible until the development of laser scanning microscopy (i.e. two-photon microscopy). 69
It was shown that 2–3% CO2 gas inhalation caused a mean dilation of 11% and 7% for parenchymal arteries and veins in the rat cortex, respectively. 70 Another study also showed that descending arterioles whose diameters at rest were not uniform (15–20 µm) over different cortical depths (0–500 µm) become more uniform (10–25% dilation) in response to hypercapnia (3% CO2 gas inhalation) in the mouse cortex. 67 This observation indicates that the tone of the descending arterioles varies over cortical layers, which may be associated with local demands.
In the rodent cortex, 80–90% of the parenchymal microvessels are categorized as capillaries whose diameter is less than 8–10 µm, while arterioles and venules constitute about 5–10%. 71 Other studies with in vivo two-photon microscopy showed volume fractions of 12%, 40%, and 48% for parenchymal arteries, capillaries (≤8 µm in diameter), and veins in the mouse cortex, 72 and 91% for capillaries (<10 µm in diameter) and the remaining 9% for other microvessels (10–60 µm in diameter) in the rat cortex. 23 Variations in the reported volume fraction among published reports could be partly due to a different threshold for binarization.
Historically, it has been assumed that the volume fraction of parencnhymal arteries and veins is 15% and 85% (including 0.4% of pure capillaries), which stems from observations of vascular anatomy in the bat wing. 73 If one considers capillaries to be less than 7 µm in diameter (including pre-capillary arterioles and post-capillary venules in this category from the original article), it turns out that volume fractions of 14%, 6% and 80% are estimated for arteries, capillaries and veins, respectively in the bat wing. Unlike the bat wing, however, the cerebral cortex has dense microvessels and a mean capillary diameter of 5–7 µm, which is similar between rodents and humans, according to three-dimensional scanning electron microscopy (SEM) microvascular corrosion casts.74–79
Just like distinctions are emerging from different types of pericytes and mural cells, capillaries should be separated into several sub-types depending on their location, distance from descending arteries or ascending veins, and endothelial and/or pericyte phenotypes.25,80 However, it is usually difficult to categorize the capillary phenotype based on visual inspection of morphology apart from branch positions. Perfusion fixation experiments showed that mean capillary diameter was 4.9 µm and 5.9 µm in in the rat cortex under hypocapnia (PaCO2 = 22 mmHg) and hypercapnia (PaCO2 = 96 mmHg) conditions, respectively, 81 and 6.2 µm, 7.5 µm, and 10.0 µm in the cat cortex under PaCO2 = 17, 40, and 76 mmHg, respectively. 82 In consistent with these reports measured under steady-state conditions, in vivo experiments show that a mean capillary diameter dynamically increased from 5.33 µm to 5.66 µm with increase of PaCO2 from 33.25 to 50.26 mmHg in the rat cortex. 83 Although mean capillary diameter increased from 3.7 µm to 4.1 µm in the rat cortex during inhalation of 2–3% CO2 gas (PaCO2 from 33.0 mmHg to 45.4 mmHg), they found that smaller capillaries (less than 4.0 µm in diameter and the majority of the capillary population) tend to increase significantly, while larger capillaries showed either no changes in diameter or even constriction (4% of the capillary population). 70 However, it remains unclear whether these responses are controlled actively or not. It is possible that these capillary responses are the result of the local fluidic/rheological environment (such as passive adjustment due to solely hemodyanamic factors). Because cerebrovascular adaptation to chronic hypoxia (8–9% oxygen in room) experiments showed that restructuring of capillaries takes a long time, namely that a mean capillary diameter gradually increased from 4.9 µm to 6.8, 7.5 and 8.5 µm over days 0, 7, 14 and 21, respectively, from hypoxia exposure in the mouse cortex, 20 the physiological limits of capillary dilation may be achieved by hypercapnic perturbation.
Cerebrovascular response to local neural activity
The activity of neurons in cortex (and other brain areas) is incredibly complex. Different types of neurons work to process and promote or prune the flow of information.84–86 Locally in the cortex, neurons process functionally distinct forms of information based on anatomical location, but also participate and modulate activity over broad brain regions. 87 Local and regional activity patterns have been traditionally measured using methods like electrophysiology. More importantly, numerous studies have captured these patterns of neuronal activity using blood flow-sensitive and blood oxygenation-sensitive imaging methods, which highlight a tight relationship between neuronal activity (spiking as well as field potential activity) and changes in CBF and tissue metabolism (for reviews, see literature88–90). Recent multi-modal optical studies have measured and verified this tight relationship across the cortex, where on-going neuronal activity measured using GCaMP (a genetically encoded calcium indicator) can explain on average about 40% of the cortical changes in blood volume (and up to 80% in some imaging periods). 91
Pial vascular responses
It is thought that pial arteries play a major role in controlling parenchymal blood flow. 41 This is partly based on the anatomical observation that pial arteries have relatively free space around the vessel wall compared to parenchymal vessels. Since pial vessels are located in the arachnoid space, these vessels are surrounded by cerebrospinal fluid; on the other hand, parenchymal vessels have limited perivascular space, which was thought to be limited for vasomotion. 41
Precise delivery of blood to active brain regions is also important for the detection of activated brain regions by hemodynamic-based neuroimaging. It was reported that the mean distance of cortical descending arterioles is dependent on the phylogenic evolution of vertebrates with distances of 500, 470, 300, 250, and 150 µm reported for chicken, rabbit, cat, dog, and monkey, respectively. 40 These values maybe underestimated because of the presence of penetrating branches underneath arteries, invisible from the cortical surface. 42 Indeed, in vivo two-photon microscopy studies allow for detection of these penetrating arteries and show shorter distances between cortical penetrating arteries in rats and mice: 130 µm and 220 µm, respectively.68,92 Although it may be considered that higher order animals consume more energy per unit brain tissue, the energy consumption rate is likely reflected in the capillary density of parenchyma tissue.93,94 Likewise, the density of penetrating arteries was also hypothesized to reflect the size of the spatial unit that is fed by blood independently of the neighboring areas. 40 If one penetrating artery is solely responsive to supply blood to a region, the density of the penetrating arteries would determine the spatial resolution of the hemodynamic-based neuroimaging. However, high-resolution imaging of hemodynamic signals has revealed localized signal changes to smaller regions, 95 indicating the presence of additional regulating mechanisms downstream of descending arterioles (e.g. pre-capillary sphincter and pericytes, see following section of the parenchymal vessels).
Studies by Duvernoy report an artery density in the human cortex that is four to five times higher than that of veins 28 which differs from observations in rodents (around 0.8 times in the rat cortex 96 ). These observations indicate that a number density of the cortical arteries and veins depend on species and cortical regions measured. Also, size-dependent variations in the vessel density should be considered for their quantitative comparisons. 97 It is also noted that a number of parenchymal arteries selectively decreases with aging, compared to the parenchymal veins. 98 The age-dependent selective elimination of the parenchymal arteries may lead to apparent decreases in vascular compliance in the aging subjects relative to young ones. 99
In response to somatosensory stimulation, the temporal dynamics of the pial arterial diameter is well-correlated with the changes in parenchymal blood flow measured with laser Doppler flowmetry.66,100 This observation indicates a concurrent vasodilation of pial and penetrating arteries under normal conditions. 66 This vasodilation response is shown to be largest at the pre-cortical terminal arteries, and spreads from small arteries to branches of pial arteries and then pial arteries, 101 which is also known as the proximal integration model. 102 Further, the propagation of pial arterial dilation has been reported to be independent of the direction of blood flow and to subside with distance from the active region. 103 The speed of propagation through pial arteries was around 2.4 mm/s in the anesthetized rat somatosensory cortex, 103 which is consistent with the conduction speed estimated for vasomotor responses in isolated cerebral arteries of rats, 104 but faster than the propagation of intracellular calcium signaling in vascular endothelial cells.105,106 As known in the peripheral vasculature,107–111 this vascular conduction responses through the pial arteries could involve hyperpolarization mechanisms via endothelial cells.112–114
In vivo animal experiments have shown that the spread of vasodilation through the pial arterial network during functional activation involves endothelial signaling. 115 Also, selective impairment of vascular endothelial cells (de-endothelialization) leads to the elimination of prolonged hemodynamic responses in the rat cortex. 116 Although the functional role of conduction and propagation of arterial dilation remains relatively uncharacterized, these animal studies indicate that endothelium-dependent vasodilation mechanisms may contribute to late focal increases in CBF, whereas hyperpolarization mechanism may participate in early onset of widespread arterial dilation.
Predominant increases in the diameter of pial arteries rather than veins have been consistently observed for short stimulation paradigms of the functional hyperemia. 117 But, the dilation of pial veins has become more evident when the stimulus duration is prolonged. 117 Consistent with the direct observations of the vasodilation, fMRI studies in the cat visual cortex show rapid and spatially localized increases in arterial blood volume responses to a 40-s visual stimulus, followed by a slow and less-specific venous volume change. 118 Similar changes in arterial blood volume to 15-s somatosensory stimulation in the rat cortex have been reported. 119 Optical studies biased to the surface of the brain vasculature have also shown that engagement of local neuronal activity through visual or sensory stimuli produces the fastest vascular changes, which are dominated by small pial arteries, while the pial venous volume follow the parenchymal blood volume responses.120,121 Overall, these studies consistently show that the dilation of venous vasculature is relatively small and slow, indicating that activity-induced diameter changes in venous vessels are likely a passive response to the changes in bulk blood flow.
Parenchymal vascular responses
Although many studies have measured diameter changes in penetrating arteries and veins in animals using in vivo two-photon microscopy.20,29,68,122–138 fewer studies have directly investigated their dynamic responses to neural activation.14,66,67,139–146 Dynamic imaging of intra-cortical arterial and venous responses is not trivial due to the axial scanning nature of laser scanning microscopes. Axial scanning is typically parallel to radial vessels, requiring three-dimensional (3D) acquisitions (or multiple slices) to attempt to cover the dynamics of these vessels. Nonetheless, repeated slice scanning (cross-section imaging of the radial vessels) at different cortical depths converges on the notion that vascular responses originate in active layers67,139 and that parenchymal vascular responses propagate upwards from descending arterioles to pial arteries.14,66,137,139,146 In contrast, stimulation of the nucleus basalis of Meynert produce dilation of the penetrating arteries of the cortex without influencing pial arteries. 67 On the other hand, depth-resolved fMRI studies show that the fastest onset of the BOLD response to sensory stimulation originates at cortical layer IV in the somatosensory cortex.139,147 Furthermore, the earliest and largest hemodynamic responses to sensory stimulation were also observed in layer IV using optical coherence tomography (OCT) angiography. 148 However, caution should be exercised in interpreting these hemodynamic signals in terms of the underlying neural activity, because layer-specific blood volume and BOLD responses are not always consistent with the layer-specific neural activity. 149 Additionally, CBV responses to neural stimulation stem mostly from the dilation of penetrating arteries, whereas BOLD signals are known to stem from the venular end of the vasculature. 150 These vascular compartment-dependent sources of fMRI signals (CBV vs. BOLD) should be considered carefully, specifically in employing high-resolution fMRI.
An important and controversial question is whether cerebral capillaries actively dilate or not in response to neural activation. Some studies report no detectable changes in parenchymal capillary diameters with photolysis of caged calcium ions in astrocytes, 122 or with the local elevation of extracellular potassium concentration, 137 bicuculline-induced seizures 124 or optogenic and sensory stimulation. 141 Other studies showed capillary dilation (1% to 40% from pre-stimulus baseline) in response to sensory stimulation,140,143–146,151 which is in close association with intracellular calcium drops in the neighboring pericytes.145,146 Several factors might contribute to these apparent discrepancies, such as variations in animal physiology (systemic arterial blood gas and blood pressure), anesthesia, and the type and strength of the stimulus applied. More importantly, we must consider the heterogeneity of capillaries and their definition. Generally, a pure capillary is a vessel that has a diameter of less than 6–8 µm in vivo which stems from vessels having a smaller intraluminal space than the outer diameter of RBCs in stasis (6–8 µm in mice and rats). In literature, some papers consider capillaries (mean diameter of 6–7 μm) as vascular segments one to three branching-orders away from penetrating arteries. 144 On the other hand, other studies categorized these microvessels as ‘pre-capillary arterioles’, because the vessel is covered with α-SMA (alpha-smooth muscle actin) expressing mural cells. 25 It is hard to distinguish capillaries from pre-capillary arterioles or post-capillary venules based on their diameters with microscopic in vivo observation without staining the mural cells. To harmonize findings between groups, it might be helpful to perform a histological characterization of anatomical features by certain cell-types. 25 Alternatively, the specific location of sampled vessels should be reported, such as their axial distance and branching order from their connecting arterioles or venules, in addition to the lumen diameter.
Recent studies have shown that pericytes play a role in controlling parenchymal blood flow at the capillary level (for reviews, see literature152–154), and that pericyte-containing capillaries, especially on the end proximal to arterioles, are able to regulate vessel tone.124,140,155 Some of these pericytes contain contractile elements indicating a potentially significant role in local vascular regulation.25,156 Another action site for microvascular flow regulation is a ring-shaped compression observed at the branch from arterioles as well as capillaries and venules in the microvascular casts. 157 This unique structure is referred to as a pre-capillary sphincter. 158 The contributions of pre-capillary sphincters and pericytes in healthy conditions of microvascular flow regulation are still debated and require further investigation.
From a technical point of view, measurements of the capillary diameter are not trivial because of the optical detection limit. For example, under normal conditions, the vessel diameter response to neural activity is typically around 10% of the mean diameter for microvessels (a mean of 4.8 µm in diameter in the rat cortex). 159 This indicates that capillaries dilate on average by 0.5 µm in diameter during neural activation. This change in caliber is almost equivalent to the optical detection limit (i.e. a size corresponding to a single pixel size in the image), so increased SNR (signal to noise ratio) is required to resolve them reliably. Another concern is that capillary diameter responses show complex heterogeneous patterns. Three-dimensionally reconstructed volume data of cortical capillary networks showed that activity-induced capillary dilation (1.2%–2.0% relative to the relative to the baseline) and also constriction (1.1%–1.3% relative to the baseline), account for a total volume increase of 2.4% in the rat cortex. 23 Our experiments with three-dimensional reconstruction (0.26 mm × 0.26 mm with 256 by 256 pixels through 128 slices with 1.6-μm step over cortical layer II/III of cerebral microvessels for 30-s somatosensory stimulation in awake mice confirmed this observation and showed that 11.5% of the capillaries dilated, while 82% of the capillary locations showed no detectable changes in diameter, while 6.5% of capillaries showed some reduction in diameter. 71 These observations suggest that the majority of capillary diameter responses are subtle and near the detection limit of the typical optical methods. To overcome issues stemming from the response heterogeneity and detection limits, extensive averaging of capillary images is effective to measure subtle changes in their diameter response to neural activation. 151 Another alternative is to measure the transit-time of vascular labels, where heterogeneity is represented by the temporal spread of its vascular passage.160–162 To further understand potential physiological mechanisms underlying heterogeneous capillary flow, it is also important to determine whether the resistance of the endothelium and/or blood cells changes actively (such as with swelling, deformability and endothelial surface layers)163–168 or whether the surrounding cells (i.e., mural cells) help change the luminal space.140,141,143,145,146,155,169,170 The propagation of vasoactive signals from parenchymal capillaries to the up-stream arteries through endothelium-dependent or -independent mechanisms is also under debate.115,137,144,171
Finally, the impact of parenchymal capillary diameter in CBF regulation should be carefully considered. In the cerebral cortex, the pressure drop from small pial arteries to pial veins was reported to be between 40% and 52% over broad ranges of the mean arterial blood pressure (measured in the anesthetized cats). 172 Numerical simulations with realistic 3D models of microvascular networks of the mouse cortex predicted a layer-dependent pressure drop through the vascular compartments, i.e. different contribution of penetrating arteries versus cortical capillaries. 173 In the cortex, several types of the penetrating arteries with different length and branching patterns) were identified.28,79 For example, small penetrating arteries on the pial surface preferentially feed superficial layers, whereas large ones dive deep into tissue with fewer branches and preferentially feed deep layers. 28 These variations in penetrating arteries may also further contribute to the layer-dependence of flow regulation in the cortex.
Photo-activation of brain cells to investigate cell type-specific microvascular regulation
The rapid growth of optogenetic tools has enabled studies of neurovascular regulation that have taken advantage of optogenetics’ strengths, namely accurate time control and genetic specificity. Channelrhodopsin has been the most popular optogenetic channel to modulate membrane potential and neuronal activity.174,175 Initial studies demonstrated the feasibility of this tool through patch–clamp studies that showed no significant alterations in cellular physiology.
176
Genetic targets selective for cortical pyramidal neurons (excitatory), inhibitory neurons and other cell types have provided additional insights into neurovascular mechanisms and contributions from supporting cell types such as astrocytes and pericytes (Figure 1).177,178
Compartmentalization of cerebral microvasculature and excitatory and inhibitory neurons. The cerebral microvasculature consists of pial arteries, veins, parenchymal descending arterioles, venules, and parenchymal capillaries with transition of pre-capillary arterioles and post-capillary venules. The 3D reconstructed image (1 × 1 × 1 mm3) of cortical microvessels (left top, labeled with sulforhodamine 101 in blood plasma) was obtained with in vivo two-photon microscopy in the mice. Arterial vascular tone is regulated by vascular smooth muscle cells (top), endothelial cells (middle), and pericytes (bottom), with varying contributions from each in different vascular compartments. Vascular smooth muscle cells in arteries are oriented radially, while they are few in number and oriented axially and planar in veins. Endothelial cells are positioned axially and cover all of the luminal interface. Pericytes are rather localized. Those morphological differences should have reflect different roles within these cell-to-cell networks, such as in signal propagation. Neurons can be specifically activated with optogenetic proteins. Excitatory and inhibitory neurons have different distribution and morphology across the cortical layers (right). Nevertheless, similar contributions to microvascular responses with optogenetic stimulation were observed.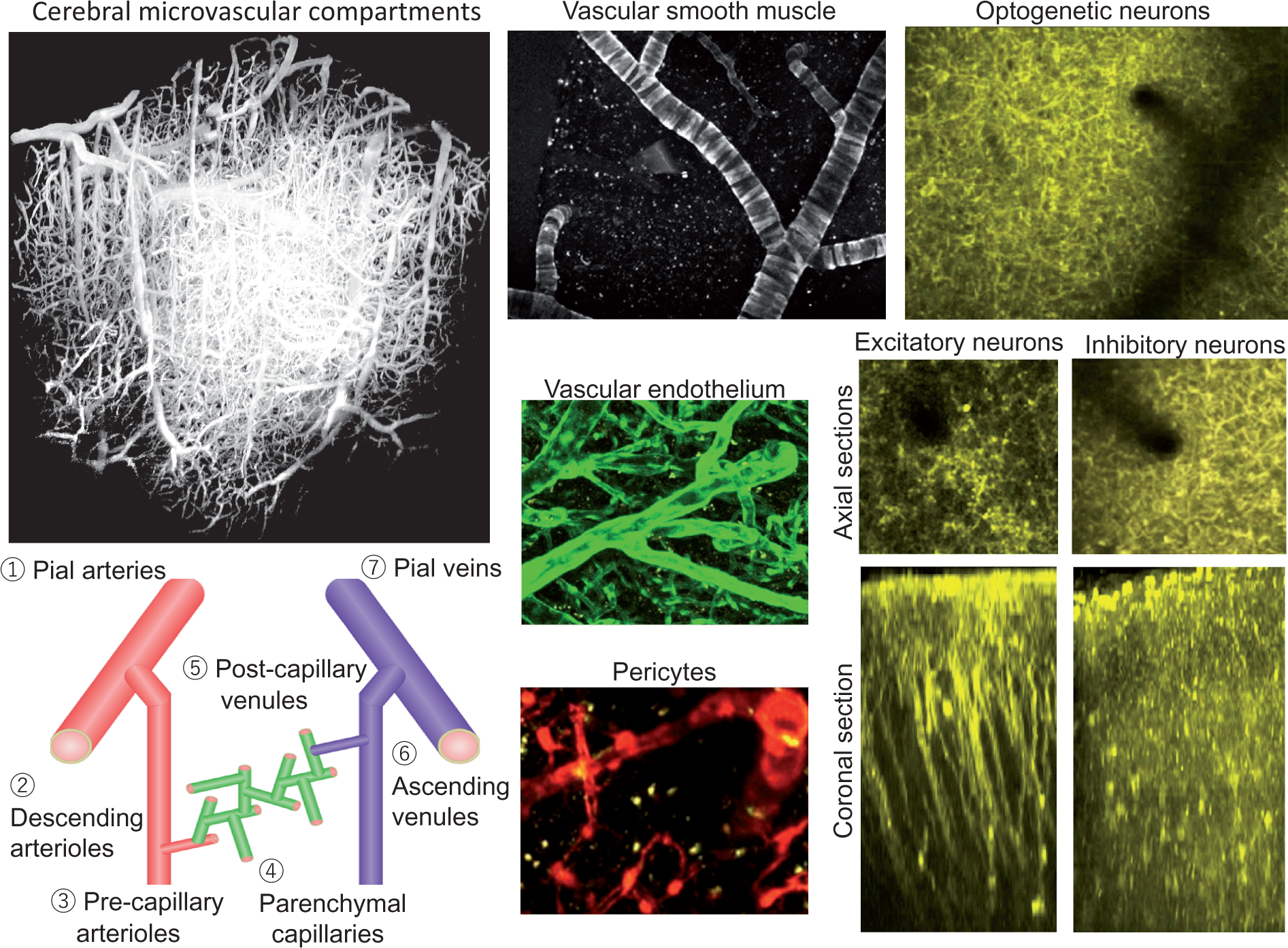
Neurons
In general, two main pathways have been identified in local neurovascular regulation, prostaglandin E2 (PGE2) and nitric oxide (NO). 179 The PGE2 pathway involves cyclooxygenase-2 (COX-2) in neurons or cyclooxygenase-1 (COX-1) in astroglia, while the NO pathway involves NO synthase (NOS)-expressing inhibitory neurons.122,180–182
Rodents expressing channelrhodopsin in excitatory neurons have been used to verify that modulation of cortical excitatory activity modulates cortical hemodynamic responses accordingly.183–185 These findings are similar to those obtained by sensory stimulation except that these optogenetic studies preferentially target Layer 5 (and Layer 2/3 in some studies) pyramidal excitatory neurons, while sensory stimulation is initiated in Layer 4.186 Nonetheless, modulation of the optogenetic stimulus, modulates both the evoked neuronal activity and size of the vascular response. 185 More importantly, the application of glutamate receptor blockers maintained neurovascular coupling in similar proportion to pre-blocker application. 187 Because the application of these blockers eliminate most sensory activity arriving in cortex, these optogenetic models are very well positioned to dissect the main signaling pathways engaged by local excitatory neurons.
Similar studies performed in transgenic mice expressing channelrhodopsin in inhibitory neurons have revealed that optogenetic stimulation of inhibitory neurons drives strong hemodynamic responses measured by intrinsic optical imaging and arterial diameter using two-photon microscopy.13–15,188 These studies show that inhibitory neuron activity appears to have a significant impact on neurovascular regulation, with perhaps higher efficiency than that of excitatory neurons (i.e. higher gain).13,188 Interestingly, the magnitude of the diameter change relative to pre-stimulation baseline increased with depth, similar to sensory stimulation, although the photo-stimulation evoked neuronal activity is strongest in the superficial layers. 14 This finding might be due to a bias in the vascular regulation capacity of inhibitory neurons, such as from nitric oxide expressing neurons, and to a heterogeneous distribution of these neurons in cortex. Another interesting observation is the mitigation of the post-stimulus vasoconstriction observed in control optogenetic responses in these mice with the application of BIBP, a potent and selective antagonist for the Neuropeptide Y receptor Y1 (NPY-Y1), suggesting that NPY-expressing neurons might modulate this vascular response feature when present. 14 These studies have verified that the evoked vascular responses are not the result of aberrant activity, such as from disinhibition. 189 Collectively, these reports are identifying cell-type specific signaling mechanisms for local neurovascular coupling, but further studies are needed to clarify the contributions from sub-populations of neuronal cell types.
Comparing the evoked hemodynamic responses from excitatory (Thy1-ChR2) and inhibitory (VGAT-ChR2) optogenetic mouse models shows similar responses with some differences. Under light anesthesia, the vascular responses obtained in the excitatory optogenetic model peak about 1.5 to 2 s sooner than those from the inhibitory optogenetic model. The anesthetic condition used also did not show a detectable post-stimulation vasoconstriction which might be due to the impact of the anesthesia on the activated network (Supplementary Figure 1, normalized differences in vascular responses are shown in the bottom panel). Nonetheless, the temporal features of these responses generally imply that the signal transduction pathways from these neuronal populations appear to have similar kinetics.
Glia
Astrocytes and astrocytic end-feet cover most of the parenchymal vasculature. 24 Their anatomical location positions them as gatekeepers of essential metabolites, such as glucose.190–193 The propagation of signaling pathways that originate from neuronal activity converge on energetic demands from recycling neurotransmitters and/or relay for signaling pathways such as PGE2.3,194–197 Photo-activation of channelrhodopsin-2 (ChR2) expressed in astrocytes causes rapid dilation of descending arterioles and pial arteries, leading to widespread increases in CBF. 16 However, whether stimulated astrocytes directly communicate with vascular cells or whether astrocytic networks evoke arterial dilation remains unclear. 192 Pharmacological results show that the astrocytic CBF response was inhibited by topical application of either carbonexolone or barium chloride (BaCl2), which may indicate that ChR2-astrocytic vasodilation involves signaling through astrocytic gap junctions and reticular inward potassium signaling in vascular smooth muscle cells.192,198
Future perspectives: Cerebral microvascular plasticity and dysfunction
Many studies have aimed to understand the dynamic responses of cerebral microvessels to evoked neural activity, but fewer have studied the long-term relationship between neural demand and vascular supply, such as during normal development or aging.199–206 It has been shown that metabolic demands stimulate neovascularization, which involve astrocytic guidance (in particular vascular endothelial growth factors), 207 to build new networks of capillaries to fulfill regionally varying demands. 208 However, in mature brains where the neurovascular unit is already stabilized, neovascularization needs other players (e.g. microglia, macrophage and pericytes),209–213 to modify the existing structures, such as during hypoxia-induced cerebral angiogenesis.214,215 Additional research is also needed to determine mechanisms underlying neurovascular adaptation, such as in association with plastic changes in neural functions. 216 It is also unknown whether neurovascular adaptation appears to be different in species, genders, and brain regions. Identifying common and different mechanisms would help maximize treatment, especially if reconstructing the neurovascular unit with cell therapy is necessary, as currently investigated for stroke and neurodegenerative diseases.217,218
Hemodynamic responses are likely impacted by aging. There is literature evidence from animals and humans that task-induced vascular responses dampen in normal aging.205,219,220 Results showing both modified neurovascular coupling relationships with age 219 or age-dependent decreases in neuronal activity 220 might explain the aging effects on neurovascular coupling. For resting CBF, human PET studies show an age-dependent decline in most of the cerebral cortex.221,222 On the other hand, animal experiments show different aging effects on microvascular compartmentalization. 223 Sonntag et al. 98 showed a lower number of descending arterioles than ascending venules in aged animal brains compared to young. It is well-known that a ratio of descending arterioles and ascending venules are different between rodent and primate brains. 224 Because rodent data are biased to young animals (not many studies use aged animals), variations due to aging effects should be carefully examined. Also, whether region-dependent variations are present in vascular anatomy225–227 need to be clarified to determine how the neurovascular unit is impacted by aging. In particular, a causal relationship with aging for microvascular compartmentalization and/or impairment of neurovascular coupling, as seen in white matter diseases, 228 is largely unknown.
In summary, optical methods are powerful tools to directly visualize and modulate cortical activity. With submicron-scale spatial resolution, the spatio-temporal dynamic properties of microvascular responses are being routinely investigated in healthy and diseased animal models in vivo. However, it is still difficult to integrate measured 2D slice image data to estimate volume changes that would approach human imaging coverage. Three-dimensional reconstruction and quantification of imaging data with 2D planes are needed for volume estimation by means of image analysis software. Optogenetic manipulation to specific cells are also sensitive to many factors, such as density of expression for optogenetic proteins, optogenetic protein type and mutation, sensitivity and intensity of photo-activation, and anesthesia used. Careful examination on these factors is strongly recommended.
Supplemental Material
Supplemental material for Optical imaging and modulation of neurovascular responses
Supplemental material for Optical imaging and modulation of neurovascular responses by Kazuto Masamoto and Alberto Vazquez in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by: JSPS KAKENHI (16K01944) to KM, and NIH R01-NS094404 to AV.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
