Abstract
We developed a mouse model of embolic focal cerebral ischemia, in which a fibrin-rich clot was placed at the origin of the middle cerebral artery (MCA) in C57BL/6J mice (n = 31) and B6C3 mice (n = 10). An additional three non-embolized C57BL/6J mice were used as a control. Embolus induction, cerebral vascular perfusion deficit, and consequent ischemic cell damage were confirmed by histopathology, immunohistochemistry, laser confocal microscopy, and regional cerebral blood flow (rCBF) measurements. Reduction in rCBF and cerebral infarct were not detected in the control animals. An embolus was found in all C57BL/6J and B6C3 mice at 24 hours after injection of a clot. Regional CBF in the ipsilateral parietal cortex decreased to 23% (P < 0.05) and 17% (P < 0.05) of preembolization levels immediately and persisted for at least 1 hour in C57BL/6J mice (n = 6) and in B6C3 mice (n = 3), respectively. A significant decrease of rCBF was accompanied by a corresponding reduction of plasma perfusion in the ipsilateral MCA territory. Neurons exhibited marked reduction in microtubule-associated protein-2 immunostaining coincident with the area of perfusion deficit. The percent infarct volume was 30.3% ± 13.4% for C57BL/6J mice (n = 17), and 38.3% ± 15.3% for B6C3 mice (n = 7) at 24 hours after embolization. This model of embolic ischemia is relevant to thromboembolic stroke in humans and may be useful to investigate embolic cerebral ischemia in the genetically altered mouse and for evaluation of antiembolic therapies.
Focal cerebral ischemia in mice has been induced either by coagulating (Welsh et al., 1987) or by mechanically blocking (Yang et al., 1994) the middle cerebral artery (MCA). These models are not accessible to fibrinolysis and do not mimic human stroke, as up to 80% of human strokes are caused by thrombosis or embolism (Sloan, 1987; Albers, 1995). In addition, these models show great variability in volume of cerebral infarction between strains (Barone et al., 1993). With the development of genetically altered mice with targeted mutations leading to gene deficiency in the plasminogen–plasmin system, such as the tissue plasminogen activator knockout mice (Carmeliet et al., 1994; Carmeliet and Collen, 1995), and with the expected use of antithrombotic agents for treatment of stroke, a mouse model of embolic cerebral ischemia is required to elucidate function of the plasminogen–plasmin system in the maintenance of cerebral vascular patency and solubilization of fibrin clot, to assess the safety and efficacy of thrombolytic therapies, and to evaluate combination therapies.
We therefore developed a mouse model of embolic focal cerebral ischemia. We now report the methods for producing this mouse stroke model and present the pathologic features of embolic ischemia in this model.
MATERIALS AND METHODS
General preparation
Male C57BL/6J (n = 34) and B6C3 (n = 10) mice weighing 29 to 35 g were used in the experiments. C57B/6J mice were chosen because this strain of mice is commonly used to produce genetically altered mice. B6C3 mice were used to examine strain-related effects on our model. Using a face mask, animals were anesthetized with 3.5% halothane and anesthesia was maintained with 1.0% halothane in 70% N2O and 30% O2. Rectal temperature was maintained at 37° ± 1.0°C throughout the surgical procedure by means of a feedback-regulated water heating system. The right femoral artery and vein were cannulated with a PE-10 catheter for continuous monitoring of blood pressure and measurement of blood gases (pH, Pa
Preparation of the emboli
The method used to prepare a white embolus was adapted and modified from Overgaard et al. (1992). Briefly, femoral arterial blood from a donor mouse was withdrawn and retained in 10 cm of PE-50 tubing for 2 hours at room temperature and for 22 hours at 4°C. Five centimeters of the PE-50 tubing containing clot was cut and attached to a system built of two syringes interconnected by 40 cm PE-10 tube filled with saline. The clot was continuously shifted from one syringe to the other for 5 minutes. A single clot (10 mm × π 0.000625 mm2 = 0.02 μL) was transferred to a modified PE-50 catheter (0.15–0.18-mm outer diameter) filled with saline.
Animal model
Under the operating microscope (Carl Zeiss, Inc., Thorn-wood, NY, USA) by a midline incision, the right common carotid artery, the right external carotid artery (ECA) and the right internal carotid artery (ICA) were isolated and carefully separated from the adjacent vagus nerve. A 6-0 silk suture was loosely tied at the origin of the ECA and another suture was ligated at the distal end of the ECA. The right common carotid artery and ICA were temporarily clamped using a curved microvascular clip (Codman & Shurtleff, Inc., Randolf, MA, U.S.A.). The modified PE-50 catheter with a 0.15–0.18-mm outer diameter containing a clot was attached to a 100-μL Hamilton syringe and was introduced into the ECA lumen through a small puncture. The suture around the origin of the ECA was tightened around the intraluminal catheter to prevent bleeding, and the microvascular clip was removed. An 8-mm length of catheter was gently advanced from the ECA into the lumen of the ICA. Preliminary studies in 20 mice showed that at this position, the intraluminal catheter was 1 to 1.5 mm from the origin of the MCA. The embolus was then gently injected through the catheter (Fig. 1). After injection of the embolus, the catheter was immediately removed.
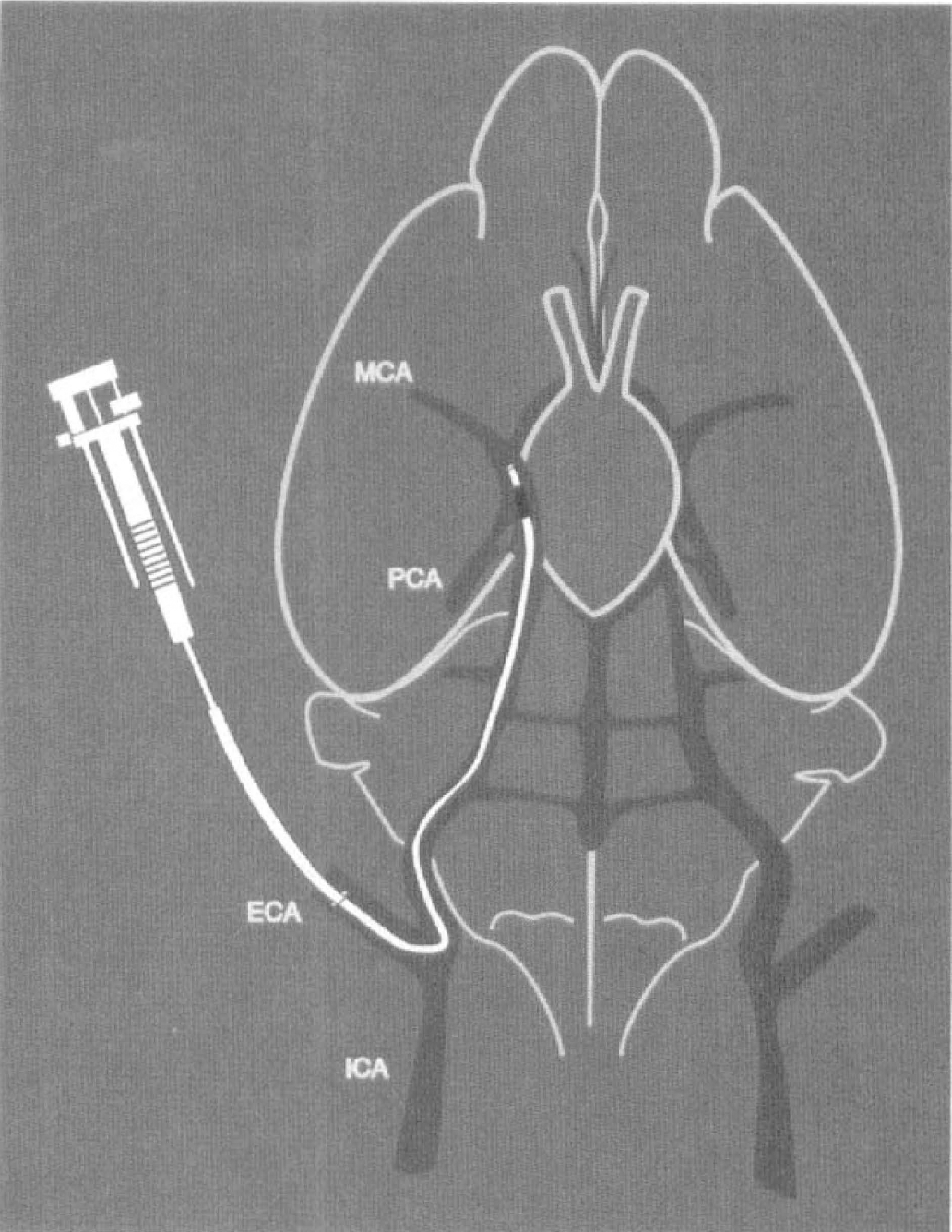
Schematic drawing of the catheter placed through the external carotid artery (ECA) and the internal carotid artery (ICA) of the mouse, with its tip at 1.5 mm proximal from the origin of the middle cerebral artery (MCA). A clot is injected through the catheter.
To evaluate the effects of placement of the catheter and injection of saline on regional cerebral blood flow (rCBF) as well as on ischemic cell damage, three C57B/6J mice were subjected to the same surgical procedures but without intravascular insertion of an embolus.
Monitoring of rCBF by laser Doppler flowmetry
Relative rCBF was measured using laser Doppler flowmetry, which was performed with a PeriFlux PF4 flowmeter (Perimed AB, Järfälla, Sweden) with relative flow values expressed as perfusion units. Using a micromanipulator, two probes were positioned 2 mm posterior to the bregma, 6 mm to each side of midline on the intact skull, being careful to avoid pial vessels after reflection of the skin overlying the calvarium. Because mouse skull and subarachnoid space are very thin, transcranial measurements of rCBF are consistent with craniectomy measurements (Hara et al., 1996). Regional CBF of both hemispheres were simultaneously measured before injection of a clot (to calculate the baseline flow) and immediately after and continuously for 1 hour after injection of a clot. The data were continuously stored in a computer and analyzed using Perimed data acquisition and analysis system (Perimed AB, Järfälla, Sweden). Regional CBF was expressed as a percentage of preischemic baseline values.
Preliminary studies in 20 mice showed that animals (12 of 20) achieving a successful occlusion of the MCA exhibited a blanching of the ipsilateral skull and a ≥70% reduction in rCBF immediately after injection of the clot. The remaining 8 mice, because of inadequate MCA occlusion, did not exhibit blanching of the ipsilateral skull and rCBF in these mice was reduced by 30% to 40% from preembolization levels. The major complications observed in the 8 mice with unsuccessful MCA occlusion were intracerebral and subarachnoid hemorrhage. Therefore, only animals exhibiting white color in the ipsilateral skull or a ≥70% reduction in rCBF observed immediately after injection of a clot were used in the current study.
Neurologic deficits
Neurologic examinations (Hara et al., 1996) were performed at 2 and 24 hours after injection of an embolus. The neurologic findings were scored on a 4-point scale: no neurologic deficit (0); failure to extend left forepaw fully (1); turning to left (2); and circling to left (3).
Blood analysis
Arterial blood gas (pH, Pa
Light microscopy
Twenty-four hours after injection of a clot, animals were anesthetized intramuscularly with ketamine (44 mg/kg) and xylazine (13 mg/kg). Mice were transcardially perfused with heparinized saline and 10% buffered formalin, and brains were removed. The lodgement of the clot was recorded. Using a mouse brain matrix, each brain was cut into 1-mm-thick coronal blocks, for a total of eight blocks per animal. The brain tissue was processed, embedded, and 6-μm-thick paraffin coronal sections from each block were cut and stained with hematoxylin and eosin (H&E) for histopathologic evaluation.
Phosphotungstic acid hematoxylin (PTAH) staining was performed on 6-μμm-thick coronal cerebral sections to examine fibrin deposition (Futrell et al., 1989).
Gross hemorrhage, defined as blood evident to the unaided eye on both faces of each block and on the H&E stained sections, was evaluated in each animal.
Formation of an embolus at the origin of the MCA was examined on the H&E stained coronal sections.
Confocal microscopy
Cerebral vascular perfusion patterns were examined using high-molecular weight fluorescein-dextran. A solution of fluorescein isothiocyanate (FITC)-dextran (0.1 mL of 50 mg/mL, 2 × 106 molecular weight, Sigma Chemical Co, St. Louis, MO, U.S.A.) was administered intravenously 1 or 2 hours after injection of an embolus and allowed to circulate for 1 minute. The animals (n = 6) were decapitated and the brains were rapidly removed and placed in 4% paraformaldehyde for 24 hours. Vibratome coronal sections (100 μm) were analyzed with a Bio-Rad MRC 1024 laser scanning confocal microscope mounted onto a Zeiss microscope (Bio-Rad, Cambridge, MA, U.S.A.). In addition, immunohistochemical staining for microtubule-associated protein-2 (MAP2) was performed for evaluation of early ischemic neuronal damage. Adjacent vibratome sections (100 μm) were incubated with a monoclonal antibody to MAP2 (1:50; clone AP20, Boehringer Mannheim Biochemicals, Indianapolis, IN, U.S.A.) for 3 days at 4°C. Texas Red-conjugated anti–mouse immunoglobulin antibody (Vector, Burlingame, CA, U.S.A.) was used as a secondary antibody.
A 10× or 40× objective was used for data acquisition. Areas of interest were scanned in 512 × 512 pixel format in the x/y direction using a 4× frame scan average. Twenty or sixty thin optical sections were scanned through a specimen along the z-axis with a 5- or 1-μm step size.
Measurement of infarct volume
Volume of cerebral tissue infarction was measured using a Global Lab Image analysis program (Data Translation, Marlboro, MA, U.S.A.). Each H&E stained coronal section was evaluated at 2.5× magnification. The area of infarction and the area of the ipsilateral hemisphere (mm2) were calculated on H&E stained sections by tracing the areas on the computer screen, and the volumes (mm3) were determined by integrating the appropriate area with the section interval thickness. The infarct volume was calculated using a formula derived from Swanson et al. (1990) in which infarct volume as a percentage of the contralateral hemisphere was calculated as 100 × (contralateral hemisphere volume – non–infarct ipsilateral hemisphere volume)/contralateral hemisphere volume.
Triphenyltetrazolium chloride (TTC) staining was also performed to demonstrate infarction 24 hours after injection of a clot. Brain sections from selected animals were incubated in isotonic phosphate-buffered saline (pH 7.4) containing 2% TTC (Sigma) at room temperature for 30 minutes and then stored in 10% neutral buffered formalin. Brain sections were photographed 3 to 5 hours after fixation.
Statistics
Data were analyzed using one-way analysis of variance followed by a Bonferroni t-test correction. Student's t-test was performed for analyzing rCBF. All values are presented as means ± SD. Statistical significance was set at P < 0.05.
RESULTS
Control animals
Reduction of rCBF and cerebral infarction were not detected in any control mice (n = 3). These mice did not exhibit any neurologic deficits.
Physiologic parameters
The arterial blood gas values and the mean arterial blood pressure were not significantly different between the two mouse strains before and 1 hour after injection of a clot (Table 1) and were within normal range.
Physiological parameters
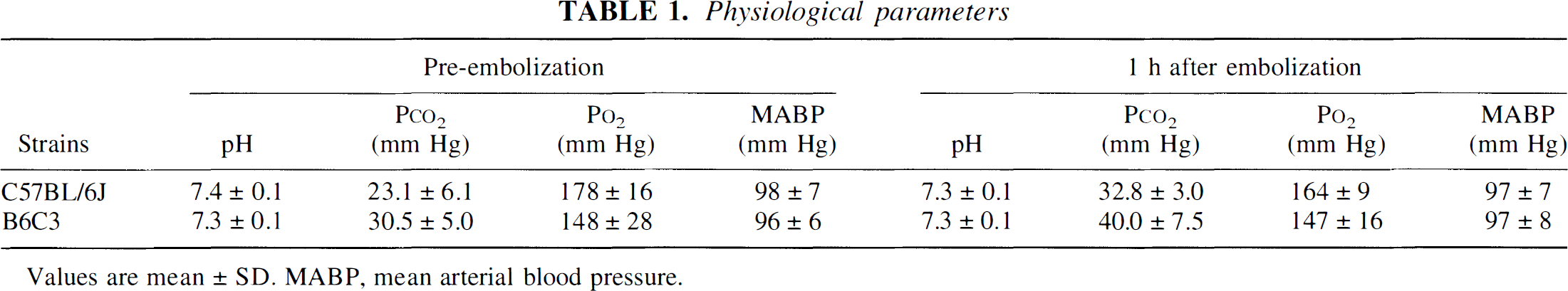
Values are mean ± SD. MABP, mean arterial blood pressure.
Neurologic deficits
Both C57BL/6J and B6C3 mice exhibited moderate to severe neurologic deficit 2 hours after MCA occlusion and showed mild to moderate neurologic deficit at 24 hours after MCA occlusion (Table 2).
Neurological deficits
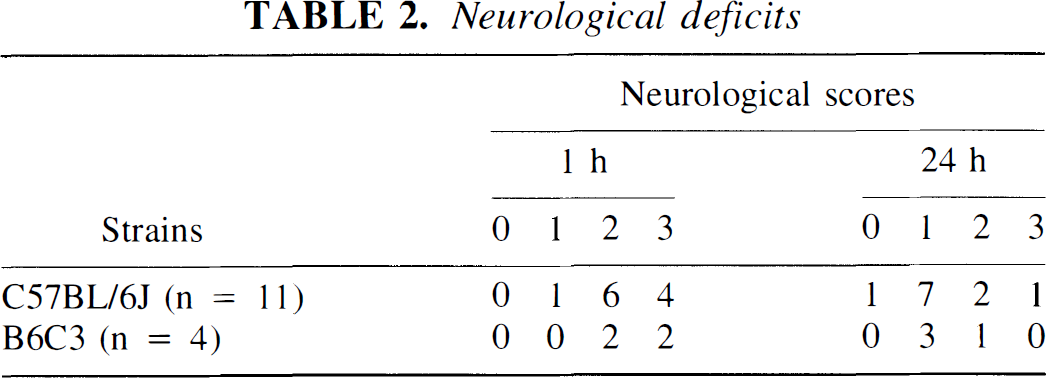
rCBF and density of perfused cerebral vessels
As measured by laser Doppler flowmetry, rCBF in the ipsilateral parietal cortex decreased to 15% ± 11.4% (P < 0.05) and 18% ± 7.0% (P < 0.05) of preinjection levels immediately after embolization and was sustained during 1 hour of measurement in C57BL/6J (n = 6) and B6C3 (n = 3) mice, respectively. The reduction in rCBF in the contralateral parietal cortex was not significant (Fig. 2).
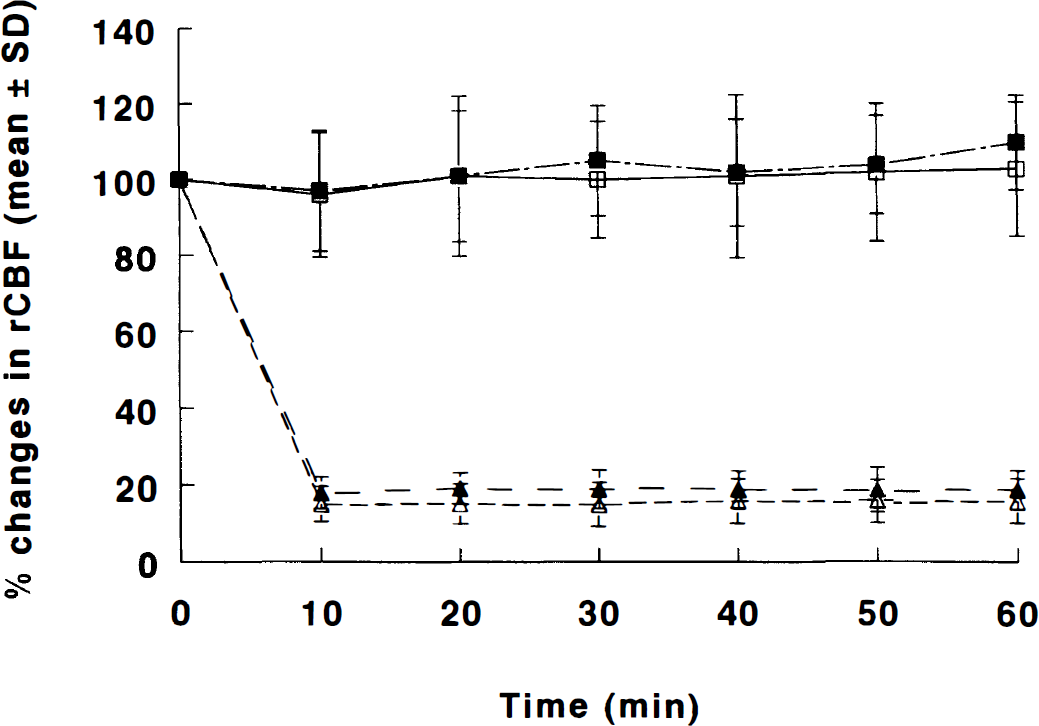
Regional cerebral blood flow measured by laser Doppler flowmetry on the intact skull before and 1 hour after embolization. C57/6J (n = 6) □ = ipsilateral and Δ = contralateral; B6C3 (n = 3) ▪ = ipsilateral and ▴ = contralateral.
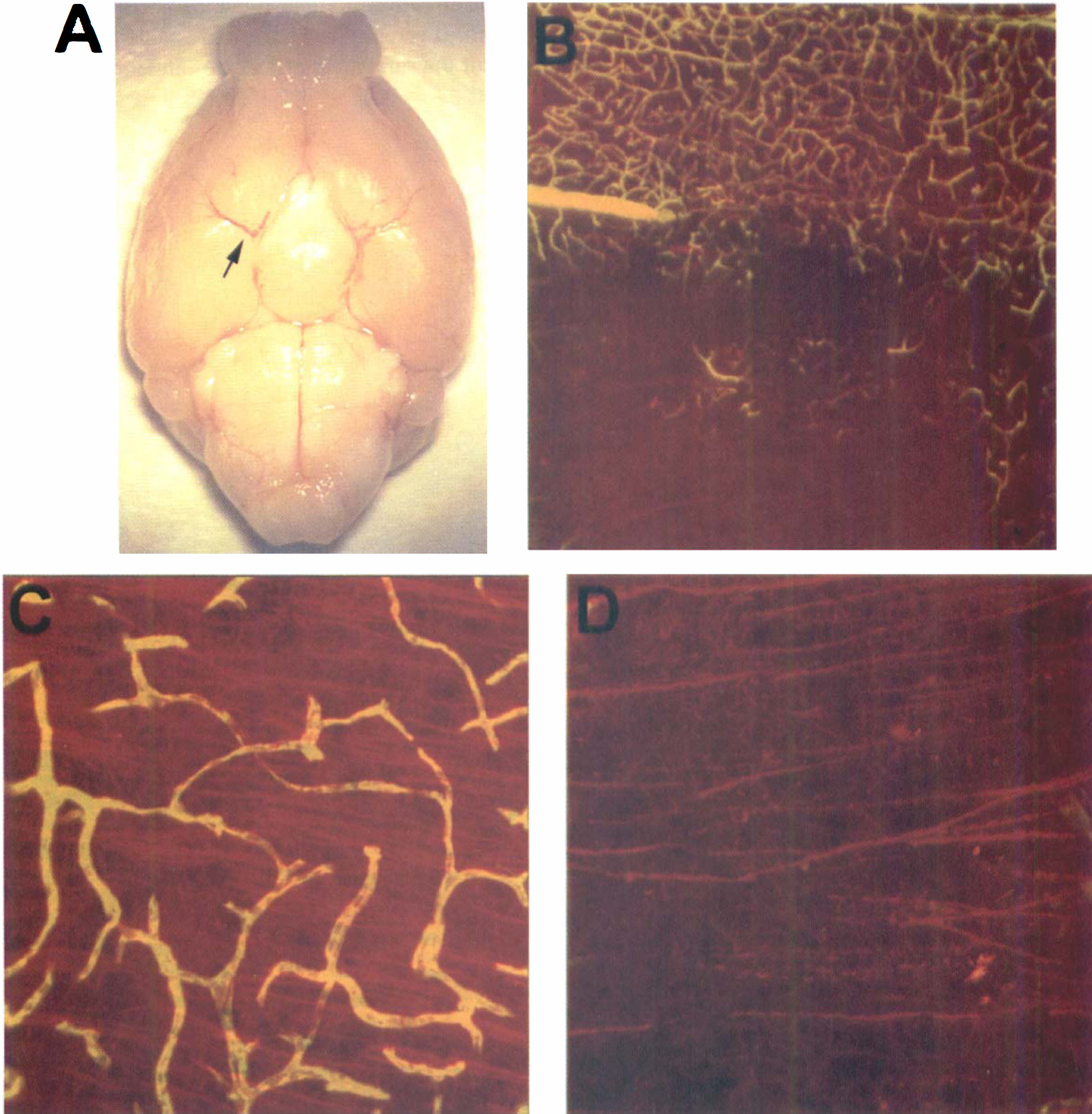
The significant reduction of rCBF after embolization paralleled a decrease in the density of perfused vessels. Nonperfused and underperfused regions were detected by injection of FITC-dextran in the ipsilateral area supplied by MCA at 1 hour after embolization (Fig. 3A and 3C). Nonperfused and underperfused regions were not detected in the contralateral hemisphere (Fig. 3B and 3D).
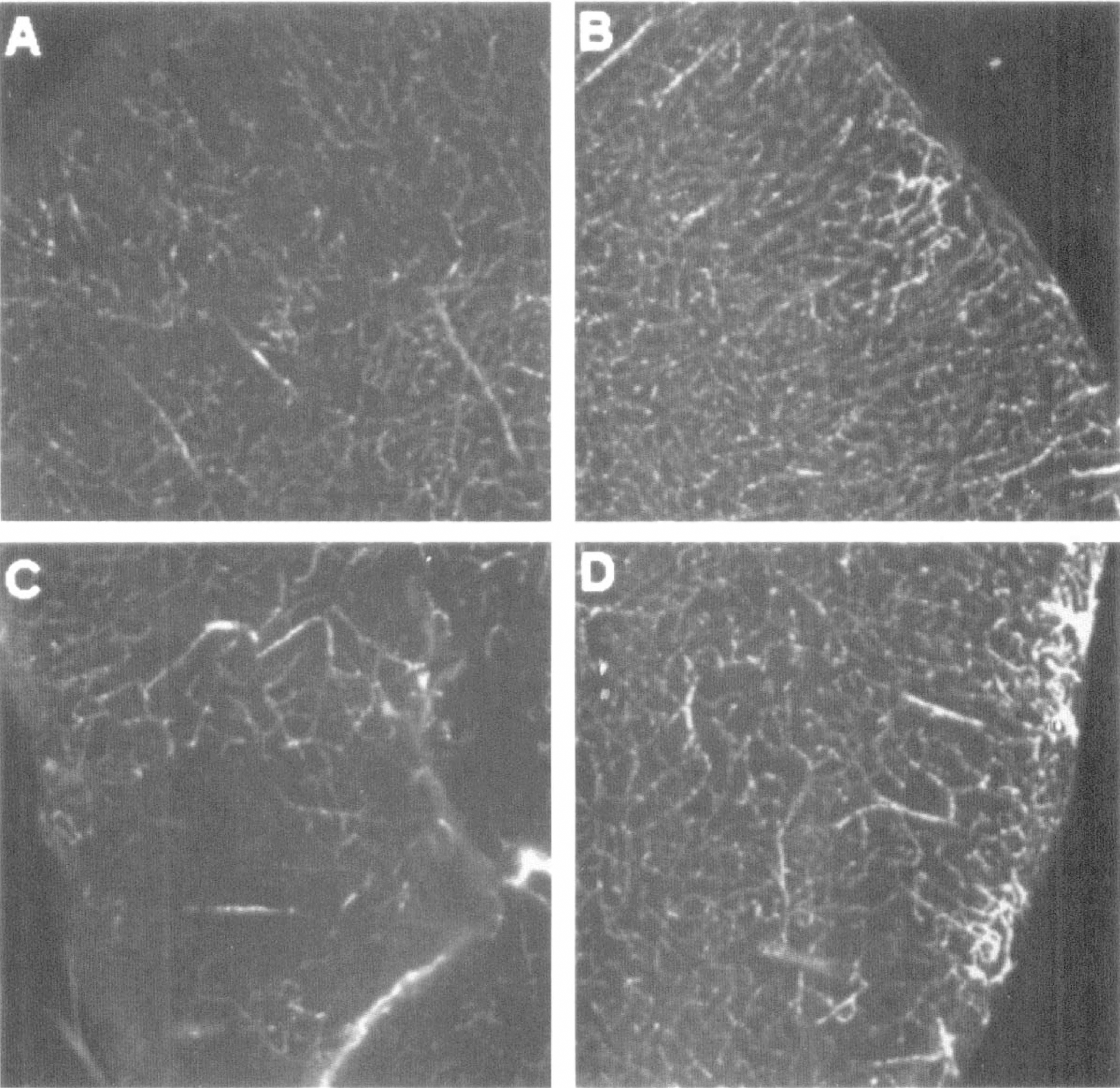
Confocal microscopy of a vibratome coronal section (100 μm) of a mouse 1 hour after injection of a clot. Perfused microvasculature is labeled by intravascular FITC-dextran.
Histopathology and infarct volume
Two mice of the C57BL/6J strain exhibited a subarachnoid hemorrhage and were excluded from the data analysis. An embolus was found in all C57BL/6J mice and B6C3 mice at 24 hours after injection of a clot. Most emboli were localized to the origin of the right MCA. Clot localization for all mice sacrificed at 24 hours is depicted in Fig. 4. Fibrin in the embolus was confirmed by PTAH staining (Fig. 5).
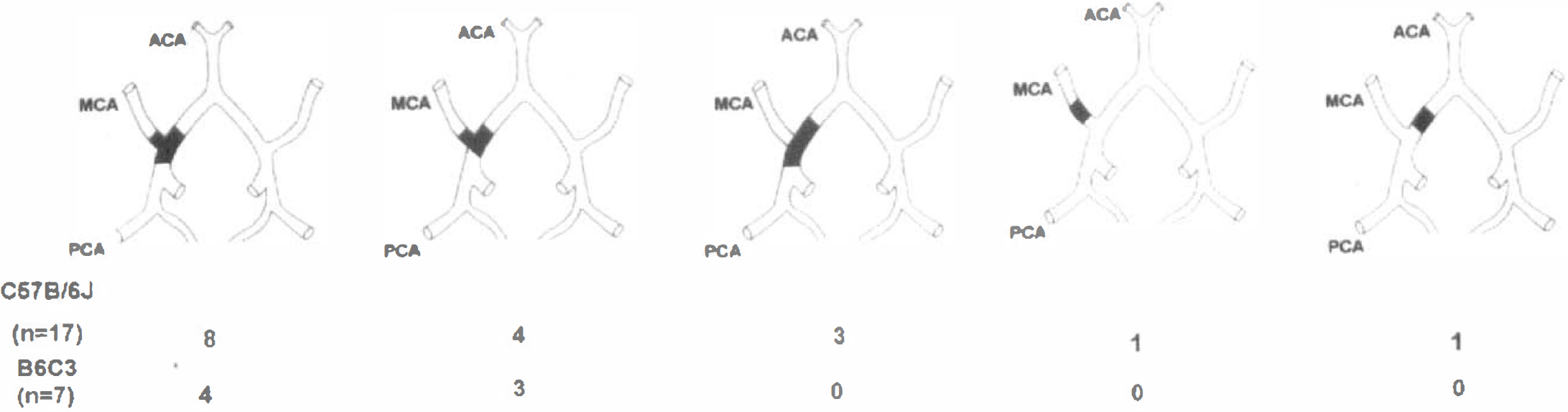
The distribution of lodgement of a clot.
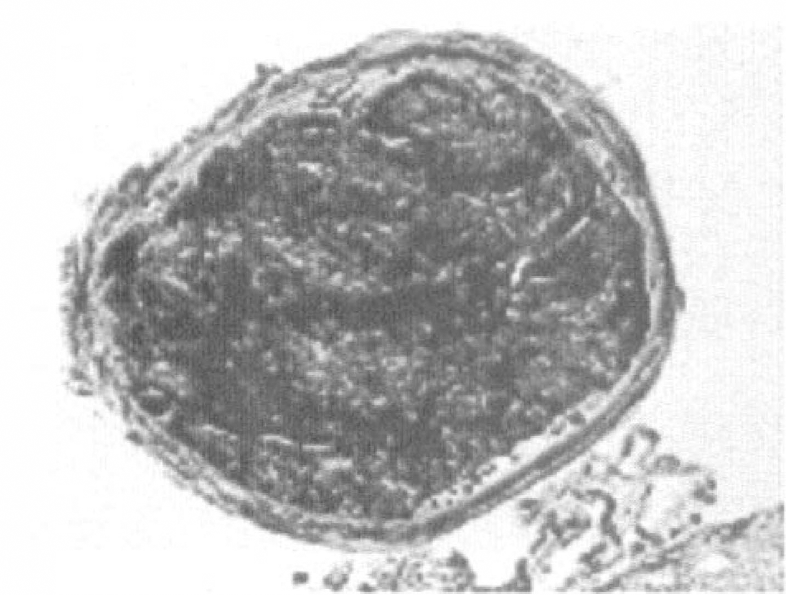
Phosphotungstic acid hematoxylin (PTAH) staining of coronal section of a mouse 24 hours after embolization. Photomicrograph shows blue fibrin strands in the clot in the intracranial segment of the ICA at the origin of the MCA.
Cerebral infarction induced by an embolus is illustrated by TTC staining (Fig. 6). The percent infarct volume as measured on H&E stained coronal brain sections was 30.29% ± 13.39% (n = 17) for C57BL/6J mice and 38.3% ± 15.3% (n = 7) for B6C3 mice. Table 3 summarizes the absolute values of the contralateral and the ipsilateral hemispheric volumes, indirect infarct volumes, and the percent infarct volumes to the contralateral hemisphere.
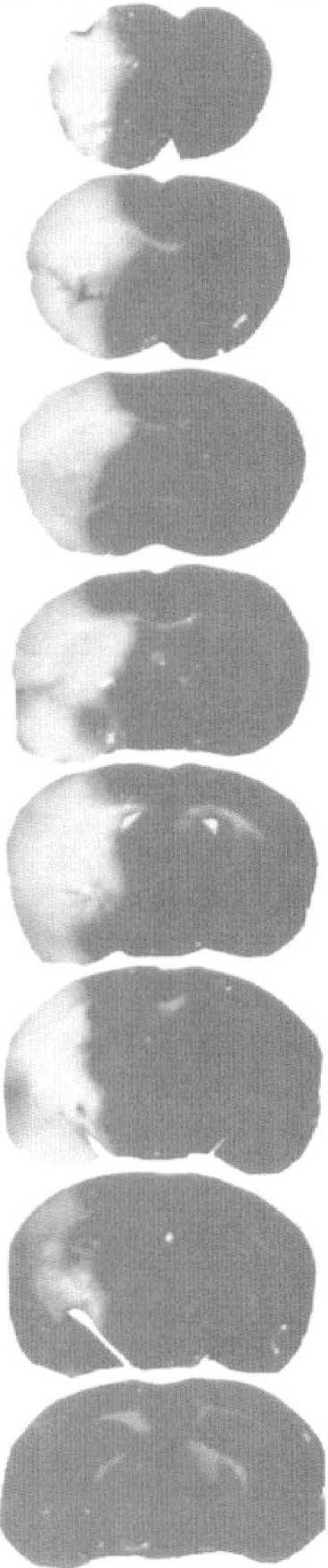
Triphenyltetrazolium chloride (TTC) staining of eight 1-mm coronal sections from a mouse 24 hours after embolization. Infarction was located in the territory of the right MCA.
The absolute hemisphere and infarct volumes in C57BL/6J and B6C3 mice
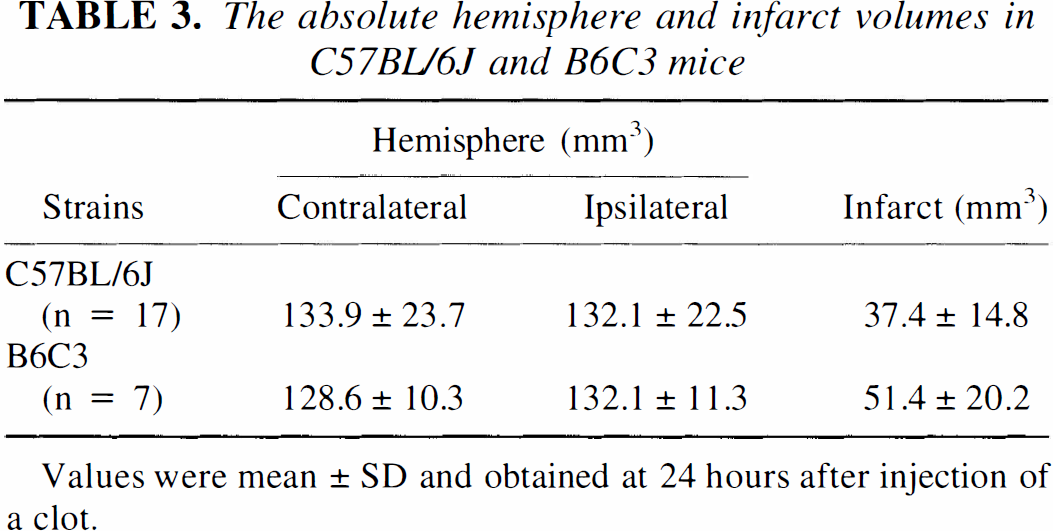
Values were mean ± SD and obtained at 24 hours after injection of a clot.
Confocal microscopic examination of sections stained for MAP2 revealed a reduction in MAP2 immunostaining in neuronal dendrites within the plasma perfusion-deficient area supplied by the MCA at 2 hours after embolization (Fig. 7). Eosinophilic neurons were detected in the ischemic region at 24 hours of embolization on H&E stained sections (Fig. 8).
Fluorescent labeling of microvasculature and neurons of a mouse 2 hours after injection of a clot.
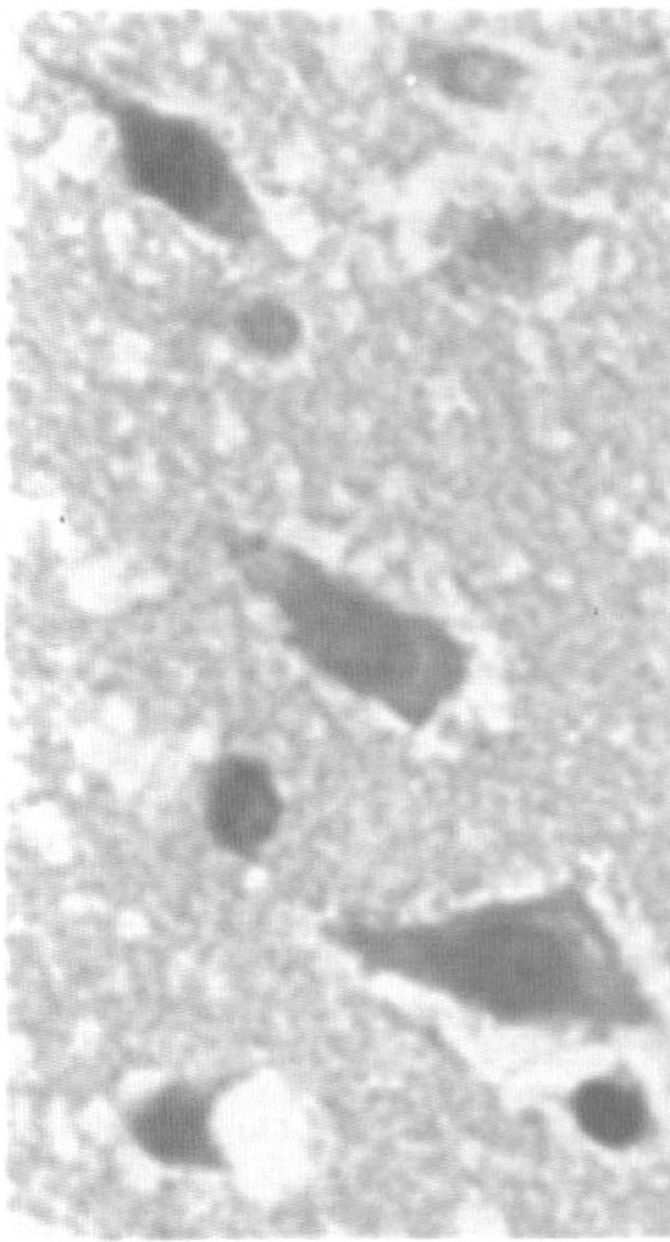
Photomicrograph of H&E staining coronal section shows eosinophilic neurons in the cortex from a mouse 24 hours after embolization (original magnification 120×).
Gross hemorrhage in the ipsilateral lesion was detected in 24% (4 of 17) in C57BL/6J mice and 14% (1 of 7) in B6C3 mice 24 hours after embolization.
DISCUSSION
In the present study, we have presented a new model of embolic focal cerebral ischemia in the mouse, in which a fibrin-rich clot is placed at the origin of the MCA. Embolus induction, cerebral vascular perfusion deficit, and consequent ischemic cell damage were confirmed by histopathologic and immunohistochemical analysis, neurologic examination, and CBF measurement, as well as by laser scanning confocal microscopy analysis of cerebral microvascular patency. This model may be employed to investigate pathophysiology of embolic focal cerebral ischemia and enable the quantitative study of the safety and efficacy of antiembolic therapies.
This new mouse model of embolic focal cerebral ischemia has several advantages: (1) A large vessel embolism similar to thromboembolic stroke in human is produced. (2) A fibrin-rich embolus is amenable to fibrinolysis. (3) The model provides a reproducible and predictable infarct volume within the territory supplied by the MCA.
Light microscopic examination confirmed that a single embolus was lodged at the segment of the intracranial ICA at the origin of the MCA. Up to 80% of ischemic stroke in humans is caused by thromboembolism (Albers, 1995). Occlusion of the MCA was documented in 86.7% of patients with stroke undergoing angiography within the first 8 hours of stroke onset and approximately 73% of MCA occlusions were induced by a single embolus (Del Zoppo et al., 1992). Our model thus mimics human stroke by precisely delivering a single small clot (0.2 μl) into the segment of the intracranial ICA 1 to 1.5 mm proximal to the origin of the MCA.
Regional CBF decreased to 15% to 18% of preembolization levels in the ipsilateral MCA territory and persisted at least 1 hour after embolization. The percentage reduction of rCBF is comparable to the values obtained in the suture model of MCA occlusion in mice (Huang et al., 1994; Hara et al., 1996). A significant decrease of rCBF is accompanied by a corresponding reduction of plasma perfusion in the ipsilateral MCA territory. In the present study, by using intravenous injection of a high-molecular weight FITC-dextran we have demonstrated that the perfusion deficit results in a reduction in the number of perfused capillaries after embolization. Complementary data are provided by MAP2 immunostaining. MAP2 immunostaining has been used as a sensitive marker of early ischemic neuronal damage in both global and focal cerebral ischemia (Matesic and Lin, 1994; Dawson and Hallenbeck, 1996). Neuronal dendrites exhibit marked reduction in MAP2 immunostaining coincident with the area of perfusion deficit, as shown in Fig. 6.
Infarct volumes in C57BL/6J mice are comparable to those obtained in the suture model of MCA occlusion in the same mouse strain (Huang et al., 1994; Hara et al., 1996). Differences in susceptibility to MCA occlusion have been reported for different mouse strains (Barone et al., 1993). However, with the new model significant differences of infarct volume in C57BL/6J mice and B6C3 mice were not detected.
There are two explanations of why our model provides a reproducible and predictable infarct volume within the territory supplied by the MCA. First, composition of the embolus influences the fibrinolysis (Zivin et al., 1988; Overgaard et al., 1993). The embolus used in the present study is fibrin rich as demonstrated by PTAH staining and has a high resistance to endogenous fibrinolysis (Overgaard et al., 1993). Our data demonstrate that an embolus was detected in all ischemic mice even 24 hours after embolization. Infarction was not detected in the animals subjected to injection of saline (control mice). Therefore in this model, cerebral ischemic infarction is induced by an embolus. Second, since 90% of animals have an embolus that blocks the ICA proximal to the origin of the MCA, or is at the origin of the MCA, a relatively uniform infarct volume is produced. This is in agreement with the suggestion that to reproduce human thrombosis, a segment of artery should be occluded (Lyden et al., 1985). Infarct volume is an important end point if efficacy of fibrinolysis is to be investigated. Although we did not measure the effect of fibrinolytic agents on this model, Overgaard et al. (1993) have shown that treatment with recombinant tissue plasminogen activator resulted in recanalization of the MCA in rats embolized with the fibrin-rich clot.
A disadvantage of our model is the incidence of subarachnoid and intracerebral hemorrhage (about 40% in C57BL/6J mice). A similar hemorrhage rate has been observed in the suture model of MCA occlusion in mice (Connolly et al., 1996). Although these animals can be identified by observing change in skull color or by monitoring rCBF, a relatively large number of animals of this particular strain are needed to accommodate these occurrences.
In summary, we have described a new mouse model of embolic focal cerebral ischemia that provides a reproducible and predictable infarct volume. This model of embolic stroke is relevant to thromboembolic stroke in humans and may be employed to investigate embolic cerebral ischemia in the genetically altered mouse and for evaluation of antiembolic therapies.
