Abstract
Intracerebral hemorrhage (ICH) is an important stroke subtype, but preclinical research is limited by a lack of translational animal models. Large animal models are useful to comparatively investigate key pathophysiological parameters in human ICH. To (i) establish an acute model of moderate ICH in adult sheep and (ii) an advanced neuroimage processing pipeline for automatic brain tissue and hemorrhagic lesion determination; 14 adult sheep were assigned for stereotactically induced ICH into cerebral white matter under physiological monitoring. Six hours after ICH neuroimaging using 1.5T MRI including structural as well as perfusion and diffusion, weighted imaging was performed before scarification and subsequent neuropathological investigation including immunohistological staining. Controlled, stereotactic application of autologous blood caused a space-occupying intracerebral hematoma of moderate severity, predominantly affecting white matter at 5 h post-injection. Neuroimage post-processing including lesion probability maps enabled automatic quantification of structural alterations including perilesional diffusion and perfusion restrictions. Neuropathological and immunohistological investigation confirmed perilesional vacuolation, axonal damage, and perivascular blood as seen after human ICH. The model and imaging platform reflects key aspects of human ICH and enables future translational research on hematoma expansion/evacuation, white matter changes, hematoma evacuation, and other aspects.
Introduction
Intracerebral hemorrhage (ICH) accounts for about 14% of all incident stroke cases 1 and is characterized by high mortality, severe tissue damage, and often permanent functional deficits. 2 ICH is therefore considered a major global socioeconomic problem, exhibiting an increasing incidence in many countries. 3
ICH is often caused by degenerative changes in the cerebral vasculature, but is also a feared complication of focal cerebral ischemia. Similar to ischemic stroke, hypoperfusion has been shown in the perilesional area and contributes to continuous tissue damage.4,5 Because of its prolonged therapeutic window, the perihematoma may serve as a potential therapeutic target. However, the underlying mechanisms of tissue degeneration remain only poorly understood and very limited progress has been made in the development of therapeutic strategies for ICH.
One reason for the lack of knowledge and therapeutic strategies may be that current experimental ICH models are limited. Most researchers use rodent models6,7 that are invaluable for basic research and offer a number of significant advantages. Nevertheless, translation into patients may be limited by major species differences such as gyrencephaly and significant difference in the relative amount of white matter.8,9
More recently, a piglet model has been developed and first studies demonstrate the possibility to investigate hematoma resolution and white matter changes after ICH in a larger animal model. 10 However, this model addresses the pediatric facet of ICH and certain aspects of the disease related to aging may not be covered, thus warranting the development of an ICH model in adult large animals.
A second major limitation of preclinical ICH research is the availability of advanced neuroimaging techniques. This would allow (i) direct comparison of pathophysiological parameters to the human situation, such as edema and white matter damage, and (ii) investigation of imaging markers needed for therapeutic target engagement and surveillance. Therefore, image appraisal and automatic post-processing analyses are needed for objective and reproducible data analysis in preclinical studies. However, lesion delineation is often performed manually or semiautomatically, partly due to the high spatial variability and complex three-dimensional shape of the hemorrhage, reducing reproducibility of subsequent analysis. 11
We here present the development and characterization of an ICH model in adult sheep including advanced neuroimaging, addressing the properties of acute moderate ICH in gyrencephalic subcortical brain tissue. The model features (i) an appropriate survival rate to study perilesional effects, (ii) reproducible lesion localization and size, (iii) simulation of the clinical environment in particular regarding neuroimaging, and (iv) a quantitative, reproducible, and objective image analysis pipeline for tailored and automatic image post-processing.
Material and methods
Experimental animals, inclusion criteria and anesthetic protocols
All experimental procedures were conducted according to the German Animal Welfare Act and German regulation for animal welfare and approved by the Experimental Animal Committee of the Regional Council of Leipzig (register number 26/09). All experiments, documentation and reporting were compliant with the ARRIVE guidelines (Animal Research: Reporting in Vivo Experiments). Fourteen (5/9 male/female) one-year-old hornless outbreed Merino sheep weighing 49–70 kg were obtained from the experimental farm of the Faculty of Veterinary Medicine of the University of Leipzig. Basic neurological functions were assessed one day before surgery 12 to exclude any preexisting cerebral pathologies. Food, but not water, was restricted 12 h before surgery. Detailed hemograms of all animals were taken five days before surgery.
All imaging and invasive procedures were conducted in prone position under deep general anesthesia and ventilation (see Supplementary Table 1).
Presurgical image acquisition and surgical planning
Imaging
Baseline MR imaging (1.5T Phillips MRI, Gyroscan Intera; Philips, Koninklijke, The Netherlands) was performed prior to experimentation in each animal to prepare stereotaxic neurosurgery using a frameless device. 13 An array of three MR-sensitive fiducial markers defining individual positions of the scanned head were installed just prior to imaging. The arrays were adopted on an individualized tooth bar (Rogue Research Inc., Canada) and mounted to the individual maxillary dent profiles using hydroplastic (TAK Systems, US). The device was later reused during surgery with the respective subject. Structural MR sequences (voxel size in mm, field direction, TE/TR, SENSE, scan duration in min) T1w (0.39 × 0.39 ×1.0 mm, transversal, 4.78/25.000, yes, 19 min), T2w TSE (0.234 × 0.234 × 2.0 mm, transversal, 10/2317.988, yes, 15 min) and T2* (0.469 × 0.469 × 4.0 mm, transversal, 18/1305.312, yes, 15 min) were acquired.
Each animal was allowed to recover from imaging for four days before inducing ICH. During pre- and postsurgical imaging, procedure general anesthesia was maintained by 1% isoflurane with 40% oxygen (Siemens Ventilog 900D, Siemens AG, Germany) and by perfusion of 6 mg × kg − 1 × h−1 propofol (Braun-Melsung, Germany).
Surgical planning
Surgical trajectory for ICH induction was planned individually using Brainsight™ neuronavigation software package (Brainsight™, Rogue Research Inc., Canada). Injection was precisely targeted to the radiate corona considering individual anatomy rather than using fixed stereotactic coordinates to avoid the trespass of blood into subarachnoidal space or into the ventricular system. The trajectory was planned for the shortest distance to the ICH localization by avoiding passing any sulci or the frontal sinus, or major vessels.
Surgery and monitoring
Surgical procedure
Fluid homeostasis was maintained with a continuous intravenous infusion of 2 mL/kg/h Ringer-Lactate-Solution (Braun Melsungen AG, Germany) throughout surgery (see Supplements for details). The fiducial marker array was carefully placed at the respective maxillary dents followed by shaving and disinfection of the head. Next, the c-clamp was attached to the head using four skull-screws, followed by mounting the working arm and the position reflector for the 3D infrared-camera (Polaris, Northern Digital Inc., Canada) connected to the computer component of the neurosurgical device. The position of the head was adjusted to the appropriate MRI dataset by identifying each fiducial marker with a reflecting pointer. After adjusting the trajectory and ICH position with the working arm according to presurgical planning, a 4 mm burr hole was drilled using a rose burr at 10,000 r/min (microspeed@uni, Aesculap, Germany). An autologous arterial blood sample (10 mL) was obtained from the saphenous artery via an 18G leader catheter and transferred into a heparinized (250 IU) syringe. A 7¼″ Hamilton needle was carefully inserted along the trajectory until the planned ICH localization was reached. Initially, in four animals, blood volume ranging from 1 to 4 mL was continuously infused to identify detrimental intracranial pressure (ICP, see below) conditions. ICH was induced in all further animals by continuous infusion of 2 mL blood within 5 min. The needle remained in place for another 10 min before being slowly removed over another 10 min. The drill hole was closed with surgical bone wax and the wound was closed before the neurosurgical device was dismounted. Subjects were allowed to recover for 2 h in prone position under persistent general anesthesia.
Monitoring
Electrocardiogram (ECG), continuous arterial blood pressure (ABP) at metatarsal artery, external oxygen saturation (SPO2) at the tongue, heart rate (HR), and body temperature (rectal, T°) were continuously recorded during the surgery. ICP was monitored by a fiberoptic transducer-tipped combined pressure–temperature catheter (Integra® Camino® Parenchymal Intracranial Pressure/Temperature Kit) to detect detrimental cortical pressure conditions by ICP >20 mmHg. Time periods of (a) 3 min before surgery (baseline), (b) 10 min during surgery (ICH) including blood application, and (c) 10 min after needle withdrawal were analyzed (Table 2). With respect to individual baseline values, the parameter shift was determined by subtracting baseline values from values recorded during and after ICH.
Postsurgical imaging
Multimodal MRI data were acquired 6 h post ICH induction under general anesthesia to investigate peracute effects after ICH: T1w, T2wTSE, T2* (sequence details see above), diffusion-weighted image (DWI, voxel size: 0.742 × 0.742 ×6 mm, b-value: 0 and 1000, gradient directions: 3, field direction: transversal, TE/TR: 80/4201.254, SENSE: yes, scan duration: 5 min) and perfusion-weighted image (PWI, voxel size: 1.25 × 1.25 × 4 mm, timing: 0.64 s, field direction: coronal, TE/TR: 27.9/18.946, SENSE: yes, contrast enhancement: i.v. 20 mL gadolinium (Gadovist, Bayer Vital GmbH), scan duration: 5 min).
Image post processing
A chart illustrating all steps of the workflow is given in Supplementary Figure 1. The procedure includes the following steps.
Preparation
All DICOM data sets were stored in NIFTI-1 format using software dcm2nii. 14 Assessment of each dataset regarding sufficient image quality and presence of occasional non-ICH brain pathologies was performed by a blinded experienced investigator (FF) before further processing. Post-processing was performed using Matlab2010a with SPM8 (Statistical Parametric Mapping 8, Wellcome Trust Centre for Neuroimaging, UK) 15 and “Fiji is just ImageJ” 16 (see Supplementary Figure 1). Additionally, the sheep reference dataset (https://www.mcgill.ca/bic/resources/brain-atlases/ovine-brain-atlas) was integrated in the SPM8 software routine. 17
Initial processing (Step 1)
All images were reoriented according to ovine stereotactic space and resliced to 0.5 mm isotropic voxel size to enable further comparative analyses. The lesion was defined as a spatial divergence of intensity in structural MR sequences (T1w and T2w) within the brain tissue. Thus, the combined information from the structural MR sequences was required to quantitatively interpret lesion size (see Figure 1(a)). T1w, T2w TSE and T2w* images (S) were averaged according to equation (1)
Exemplary structural MRI of intracerebral hemorrhage (ICH) model in sheep and key features for the structural image processing. (a) Exemplary coronal and transversal sections T1w, T2w TSE, and T2w* MRI sequences from ICH in a sheep brain registered to the ovine standard space.
17
Crosshairs represent the coordinate [x;y;z] of the ovine space at [−19;0;8]. The 3D-rendered surface provides the spatial localization of the lesion and depicts gray (GM) and white matter (WM), as well as the ventricle. (b) A T1w image attenuated for T2-signals (LogS0S1) served as input for the unified segmentation procedure of SPM8 to adequately reflect the heterogeneous lesion. The tissue probability based on the ovine tissue probability maps for GM and WM were modified by the lesion probability, which served as a separate tissue class.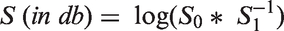
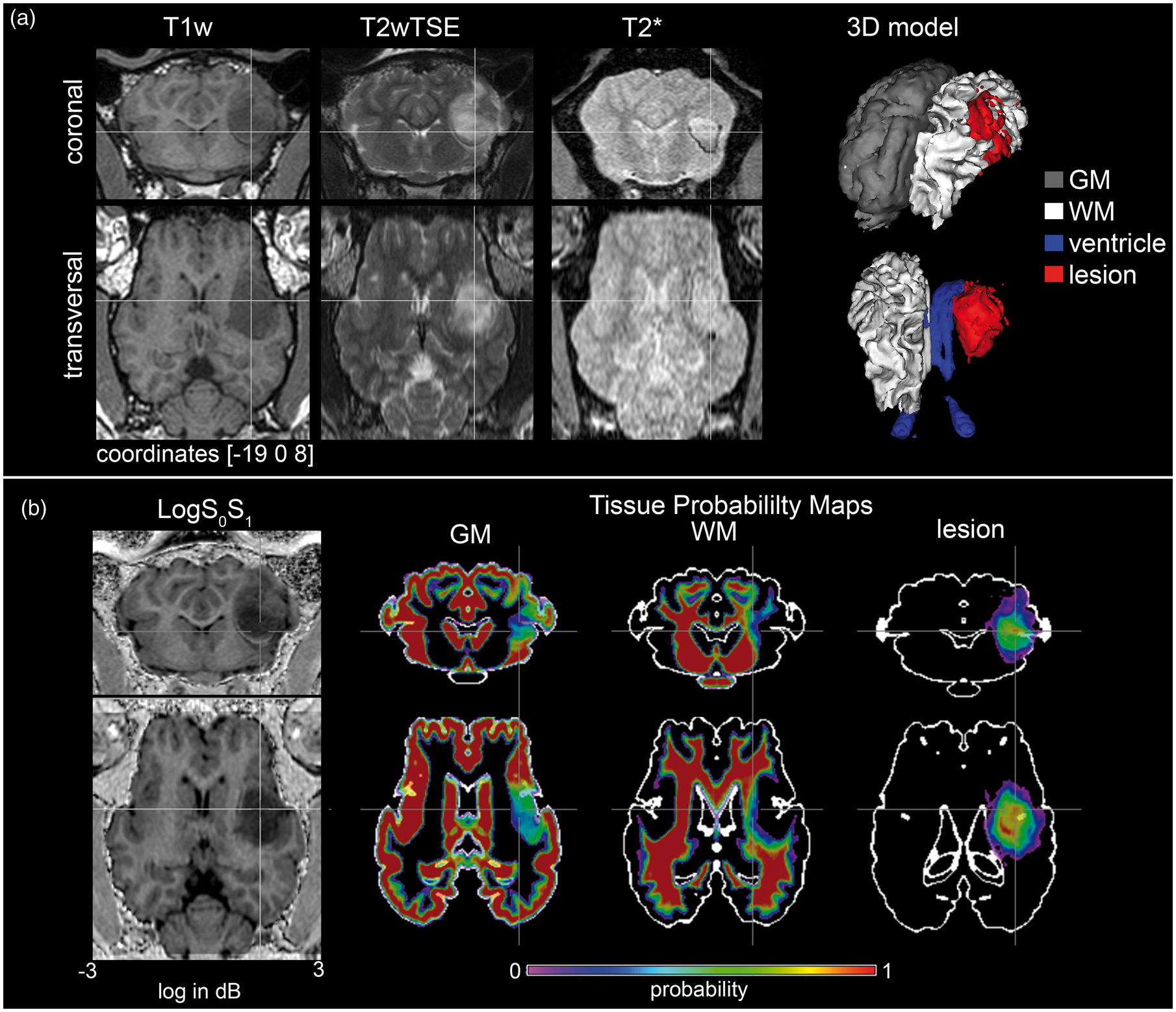
Established ovine tissue probability maps (TPMs) 17 were modified to enable the classification (=segmentation) of the additional tissue class ‘lesion’ (lesion probability map, LPM) as follows:
Preparation of TPM/LPM (Step 2)
The lesion was manually segmented using FIJI. The resulting binary lesion mask was averaged to create the LPM. LPM values were subtracted subsequently from the present TPM GM and WM. TPM for CSF was divided into external CSF space and ventricles. The obtained dataset including TPM/LPM for GM, WM, lateral ventricle, lesion, and external CSF were finally implemented in SPM8 for subsequent segmentation procedure (see Figure 1(b)).
‘New Segmentation’ in SPM8 (Step 3)
Each logS0S1 was automatically classified into binary GM, WM, lateral ventricle, and lesion (LlogS0S1) masks by using standard parameters for the unified segmentation procedure of SPM8 software for further analyses. 18 Additionally, brain tissue masks were generated by summing GM, WM, and lesion, respectively, and the ipsi- and contralateral part from each tissue mask were parcellated.
PWI and DWI data processing (Step 4)
The free diffusion of water within the blood cavity was identified as hyperintensities in DWI sequences (LDIFF). LDIFF was calculated voxel-wise by subtracting the corresponding contralateral brain region voxels by flipping over the image of the contralateral hemisphere after skull stripping and smoothing. All voxels above the standard deviation (SD) of the contralateral hemisphere were assigned to contribute to LDIFF.
Additionally, restricted diffusion of the adjacent tissue identified by apparent diffusion coefficient (ADC) imaging was calculated as follows: All voxels in the brain with ADC <800 × 10−6 mm/s2 were defined to show apparent diffusion (LADC). 19 The sum of LADC and LDIFF represent the total diffusion deficit volume LDWI.
PWI deficit volume was calculated by generating the mean-transit-time (MTT) maps. Regions of restricted perfusion were calculated voxel-wise by subtracting corresponding ipsilateral voxels from the contralateral ones by flipping over the image of the contralateral hemisphere after skull stripping and smoothing. All voxels above SD were assigned to contribute to the perfusion deficit (LPWI).
All animals undergoing ICP measurements (
Quality of segmentation procedure
The automatic segmentation procedure was compared to manual processing by calculating DICE’s similarity coefficient according to equation (2)
20
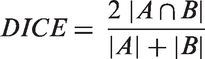
Volumetry
Total brain and hemispherical volume for GM, WM, and ventricle as well as lesion (LlogS0S1) and deficit volumes (LPWI, LADC, LDIFF, LDWI) were calculated in mL. The ratios between DWI and PWI deficit volumes (LDWI:LPWI, LlogS0S1:LPWI, LADC:LDIFF) were created. The volume of LlogS0S1 was correlated to the injected blood volume.
Morphometry
The degree of spatial co-localization of structural lesion and PWI:DWI volumes was revealed by calculating DICE’s similarity coefficient between LlogS0S1 and LDWI, LlogS0S1 and LPWI, LDWI and LPWI, LADC and LPWI as well as between LDIFF and LPWI according to equation (2).
The lateralization index (Ilat) was calculated as follows

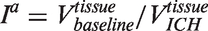
Voxel-based-morphometry
Voxel-based-morphometry (VBM) was performed to test for a spatial shift of the lateral ventricles due to ICH 21 . The VBM8 Toolbox (Gaser, University of Jena, Department of Psychiatry; http://dbm.neuro.uni-jena.de/vbm/) was used. All segmented and normalized ventricle masks were smoothed by applying an FWHM of 2 mm with respect to target volume size.
Macroscopic and histological examinations
Anatomical and histological preparation
Animals were sacrificed by pentobarbital overdose (Euthadorm, 100 mg/kg, i.v., CP-Pharma GmbH, Germany) immediately after imaging. Death was confirmed by two veterinarians. Subjects were decapitated and heads were perfused with 3 L of phosphate-buffered saline solution (pH 7.4) throughout the maxillary arteries followed by 15 L of 4% phosphate-buffered paraformaldehyde (pH = 7.4; PFA; Sigma Aldrich Ltd, Taufkirchen, Germany). The calvarium was removed after being stripped from soft tissue using an oscillating saw (HEBU medical, Germany) and the dura mater was opened to enable
Staining procedure
After de-waxing, sections were stained using routine staining methods, including hematoxylin-eosin, cresyl violet and luxol fast blue.
In addition, consecutive sections were labelled immunohistochemically for axonal neurofilaments, myelin basic protein (MBP), astroglia (glial fibrillary acid protein, GFAP), microglia (Iba-1), and a plasma marker (fibrinogen) (see Table 2). After de-waxing, endogenous peroxidase activity was blocked by exposure to H2O2 (3% v/v, aqueous solution) for 8 mi. After high-pressure heat-induced antigen retrieval (30 s, 125oC, in pH 7.8 Tris-citrate buffer), non-specific binding was blocked with phosphate-buffered saline containing 0.1% v/v Triton-X100 and 3% (w/v) bovine serum albumin (PBT-BSA) for 60 min at room temperature. Sections were then exposed to primary antibodies at 4℃ overnight. Primary antibodies were diluted on the day of use in PBT-BSA.
Primary antibody labelling was visualised using a peroxidase-conjugated secondary reagent (Envision® kit, K4065, Dako, Carpinteria, CA) and diaminobenzidine (DAB) chromagen. Sections were then counterstained with Mayer’s haematoxylin. As a negative control, neighbouring sections were treated identically but with no primary antibody (not shown).
All histochemical labelling and image analysis were performed by an investigator blinded to clinical and MRI data. Neuropathological examination was performed by a senior registered neuropathologist (LRB) on brain tissue from animals without ICP measurement.
Statistics
All values were given in mean ± SD. All data were tested for normal distribution using Kolmogorov–Smirnov and Shapiro–Wilk tests. No relevant deviations from normality were found. Group-wise comparisons of the retrieved parameters absolute and relative tissue, lesion and deficit volumes were performed using repeated measurement ANOVA in Matlab2010. A
Spatial statistics via VBM were performed using a paired
Results
Pre-surgical procedures
No pathological hemogram or neurofunctional deficits were observed. Results of blood sample analyses are given in Supplementary Table 3. Baseline MRI did not reveal any structural brain abnormalities.
Surgical procedure
Monitoring (mean ± SD/quartile I-III).
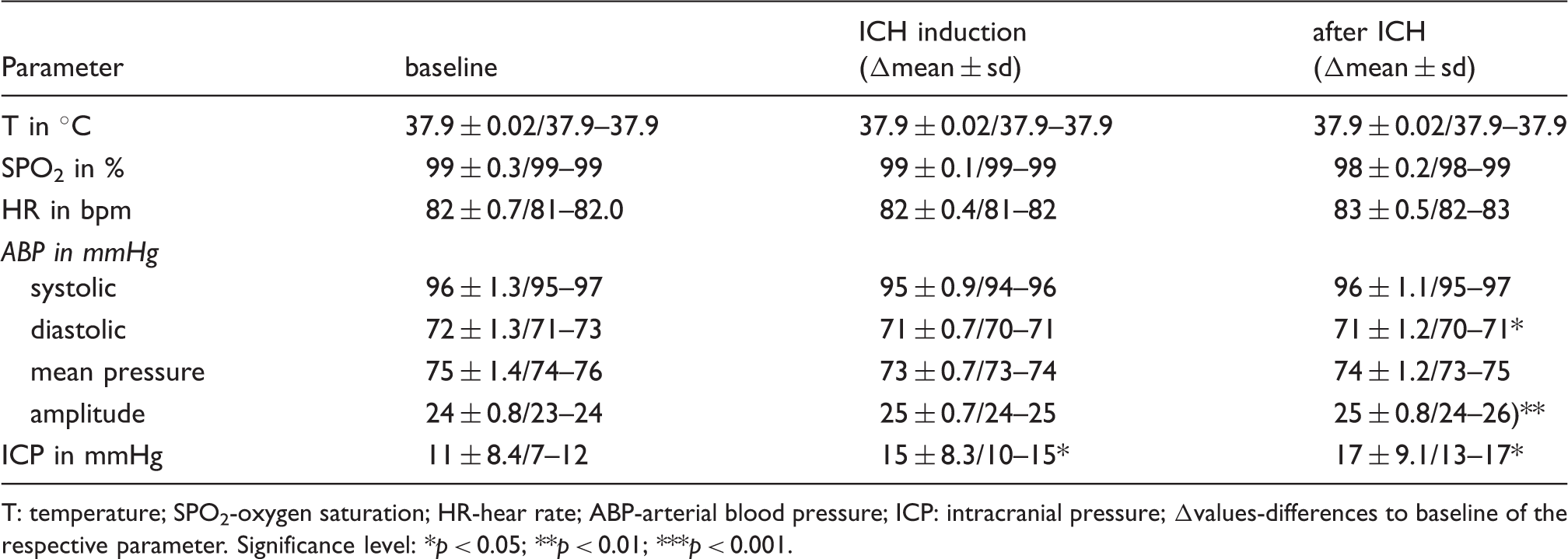
T: temperature; SPO2-oxygen saturation; HR-hear rate; ABP-arterial blood pressure; ICP: intracranial pressure; Δvalues-differences to baseline of the respective parameter.
Significance level: *
Descriptive statistics (mean ± SD/quartile I-III) of tissue volumes, lateralization index (Ilat), tissue ablation coefficient (Ia) as well as ventricle brain index (Iv:b).
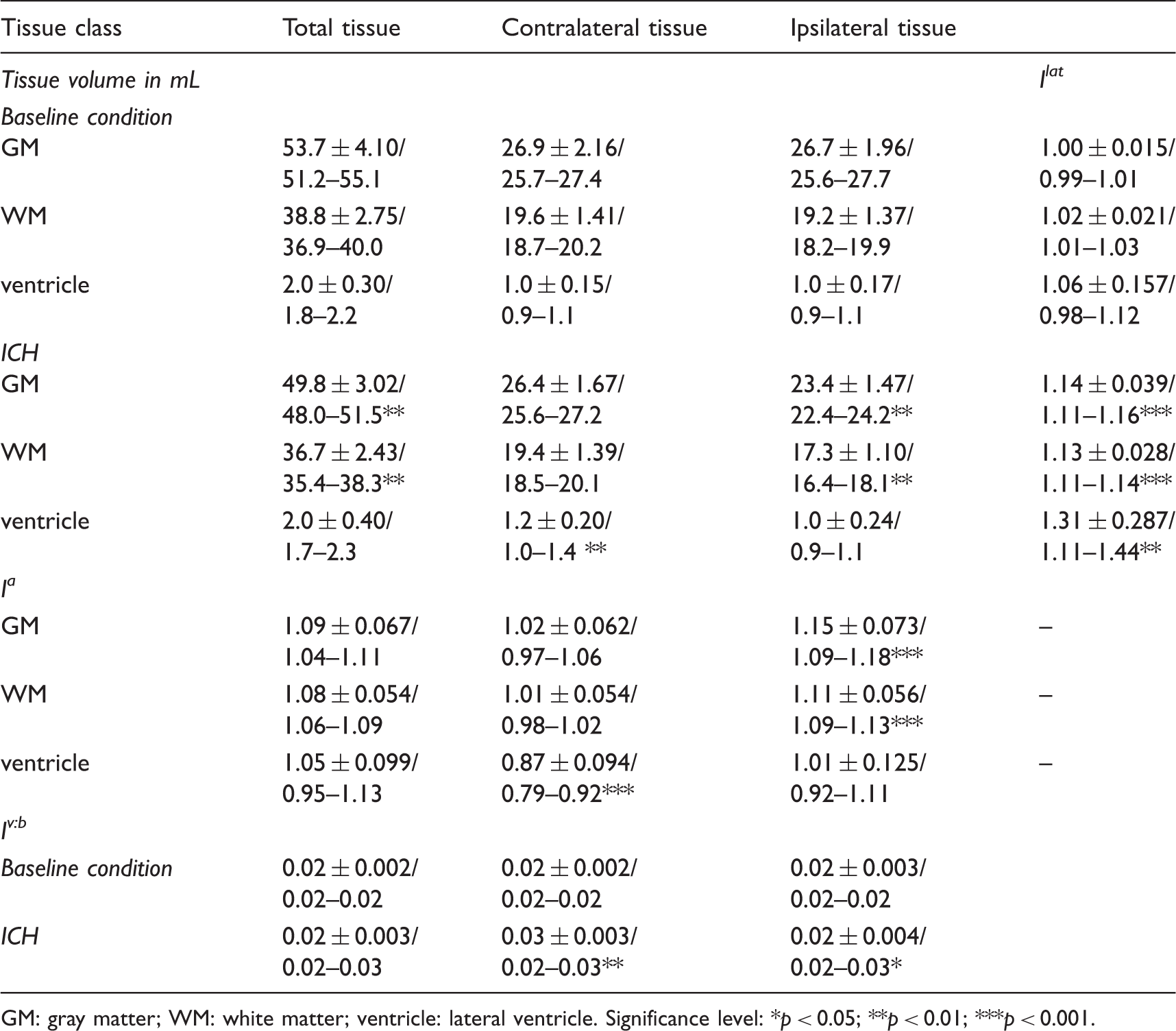
GM: gray matter; WM: white matter; ventricle: lateral ventricle.
Significance level: *
Quality of image processing
Visual inspection of automatically segmented GM, WM, ventricle, and lesions masks revealed no misclassified voxels, but the manual segmentation procedure of the lesion led to a distinct aliasing of the lesion mask. The DICE similarity coefficient between the automatically and manually segmented lesions was 0.71 ± 0.08.
Morphology and volumetry
GM and WM
Tissue volumes at baseline were 53.7 ± 4.1 mL (GM) and 38.8 ± 2.8 mL (WM), respectively. Ipsi- and contralateral GM and WM (see Table 2) volumes were symmetrically distributed and a lateralization of GM and WM was not detected at baseline (
However, GM ( Volumetry and morphology of gray (GM) and white (WM) matter as well as ventricles with and without intracerebral hemorrhage (ICH). (a) GM and WM volume of both hemispheres at baseline and ICH: The gray, dotted line represents the median of the unaltered tissue volumes. (b) shows the tissue ablation (Ia) and lateralization index (Ilat) for GM, WM, and ventricle of both hemispheres at baseline and after ICH. (c) Ventricle-to-brain index (Iv:b) of contra- and ipsilateral hemisphere are given at baseline and after ICH. (d) The voxel-based-morphometry revealed a compression of the ipsilateral lateral ventricle (green, left), while the olfactory ventricles were dilated bilaterally after ICH (green, right). ICH location is indicated as red ellipsis.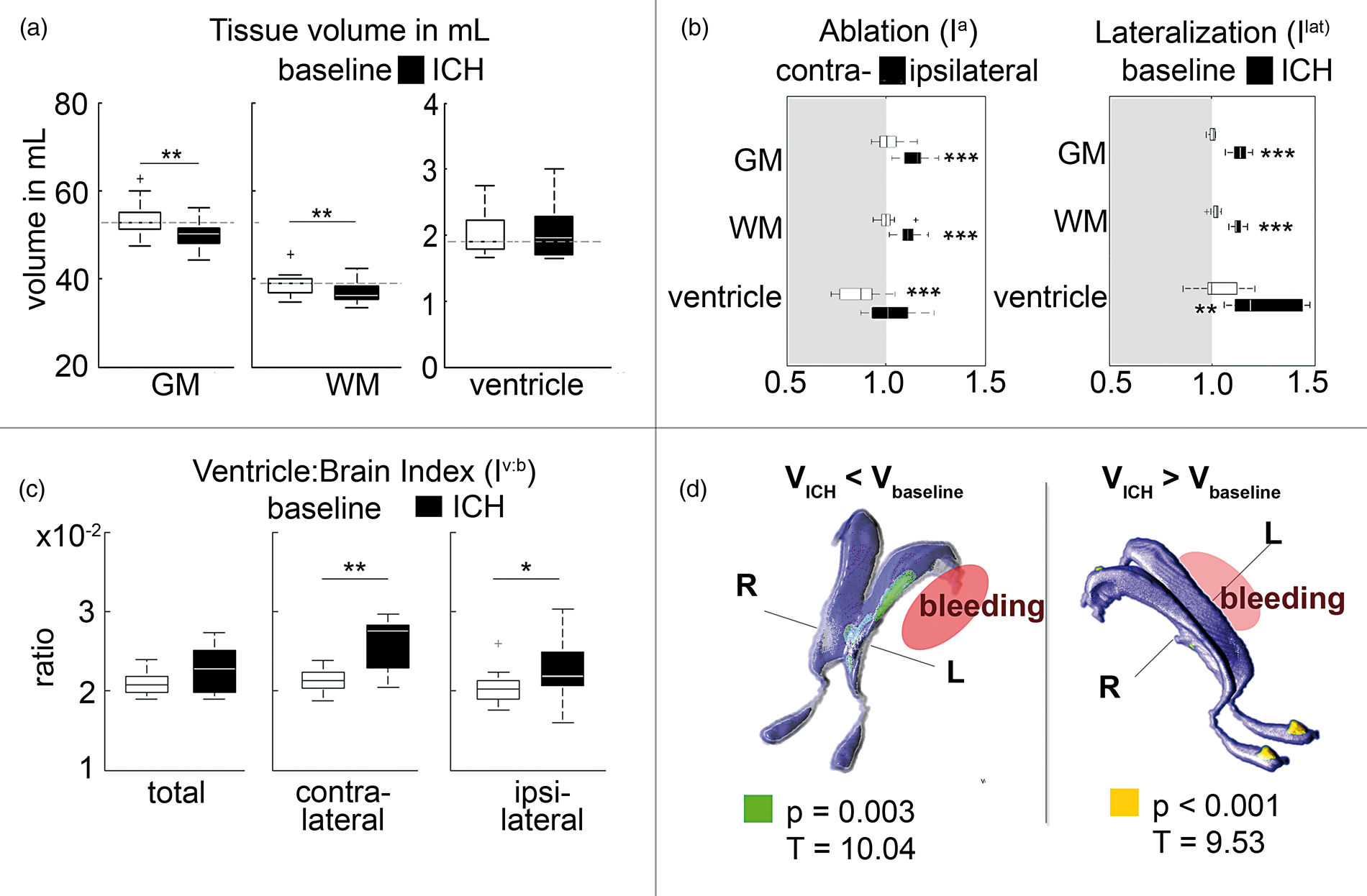
Ventricles
No significant lateralization of the ventricles by Ilat was detected at baseline (
The overall volume of both ventricles was unaltered after ICH induction (
A significantly decreased Ia was calculated for the contralateral hemisphere (
The central part of the ipsilateral lateral ventricle was significantly smaller after spatial voxel-wise testing using VBM (
Lesion and deficit
The structural lesion (T1w 3d, T2w TSE and T2*) appeared as a circumscript hyper- (T2w) or hypotense (T1w) cavity in structural imaging sequences (see Figures 1(a) and 3(a)). logS0S1 images also revealed a clear circumscriptive lesion, but corrected for hypertense T2 effects (see Figure 1(b)). The segmentation procedure revealed an overall lesion volume LlogS0S1 with 2.1 ± 0.76 mL which equates to 106% of the injected blood.
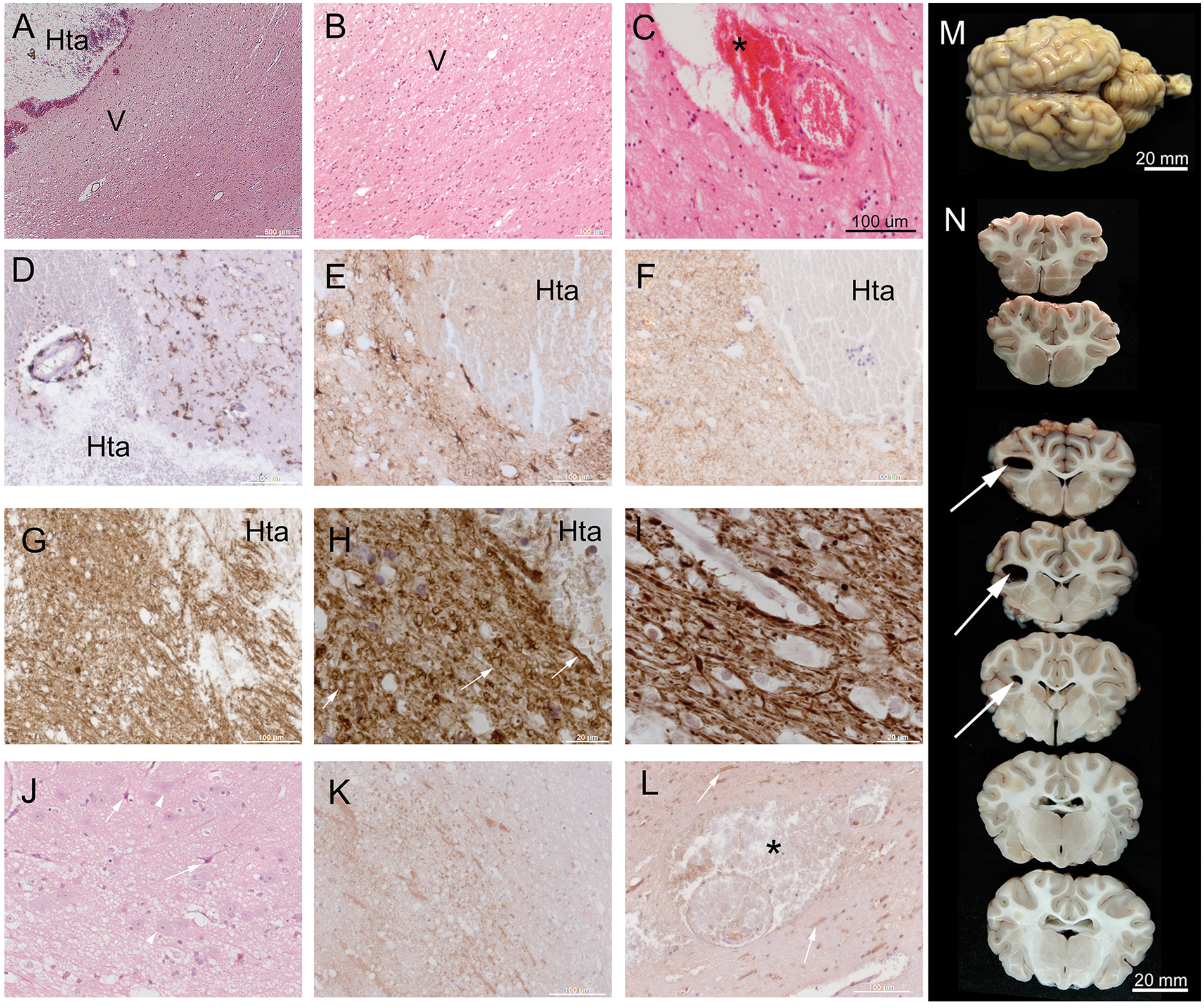
Parametric perfusion (mean-transit-time, MTT) and diffusion (apparent diffusion coefficient, ADC) maps and the lesion/deficit volumes. (a) T2w TSE image (left), related map of apparent diffusion-coefficient (ADC, middle), and perfusion-weighted-imaging via map of mean-transit-time (PWI, right, including T2 TSE overlay) were aligned to ovine stereotaxic space. Crosshairs located at [−17, 0, 8]. (b) Lesion and deficit mismatches: Mismatch analysis of the ratio of LDWI:LPWI and LlogS0S1:LPWI deficit volumes revealed a significant perfusion excess ( Histopathology of brain tissue around experimental ICH in sheep brain. (A) Low magnification view of hematoma (Hta) within subcortical white matter: Parenchymal tissue close to the hematoma exhibited vacuolation (V) suggesting ischemic damage. (B) Higher magnification view of region shown in (A) shows the margin of vacuolated white matter (V). (C) Blood (asterisk, *) occurred in the perivascular space around a small arterial vessel, close to the hematoma. (D–G) Sections showing tissue adjacent to the hematoma immunolabelled for microglial cells (Iba-1, panel D) or astrocytes (GFAP, E) myelin (MBP, panel F) or axonal neurofilaments (panel G). (H, I) Higher magnification of neurofilament labelled tissue shows swelling and bulbing of axons (arrows). (I) Neurofilament labelled healthy white matter from the same animal, for comparison. (J) Shrunken, eosinophilic pyramidal neurons (arrows) were seen in overlying cortical grey matter (arrows). Many non-shrunken neurons were also evident (arrowheads). (K, L) sections labelled for the plasma marker fibrinogen. Generalised and cellular fibrinogen was detected close to the hematoma (K). Fibrinogen positive cells were also seen relatively distant from the hematoma (arrows in panel L). Fibrinogen and blood cells (asterisk) were evident in the perivascular space around a small blood vessel. (M, N) ICH altered sheep brain specimen. Coronal gross sections (upper = anterior, bottom = posterior) reveal sharply demarcated intracerebral hematoma (white arrow) in radiate corona. Panels A, B, C, J: standard haematoxylin-eosin stain. D–I, K: immuno-histochemical labelling with DAB chromogen (brown) and hematoxylin nuclear counterstain (blue). Scale bars (A–l) 500 µm (a), 20 µm (H, I), 100 µm (b–G, K–L) and 20 mm (M,N).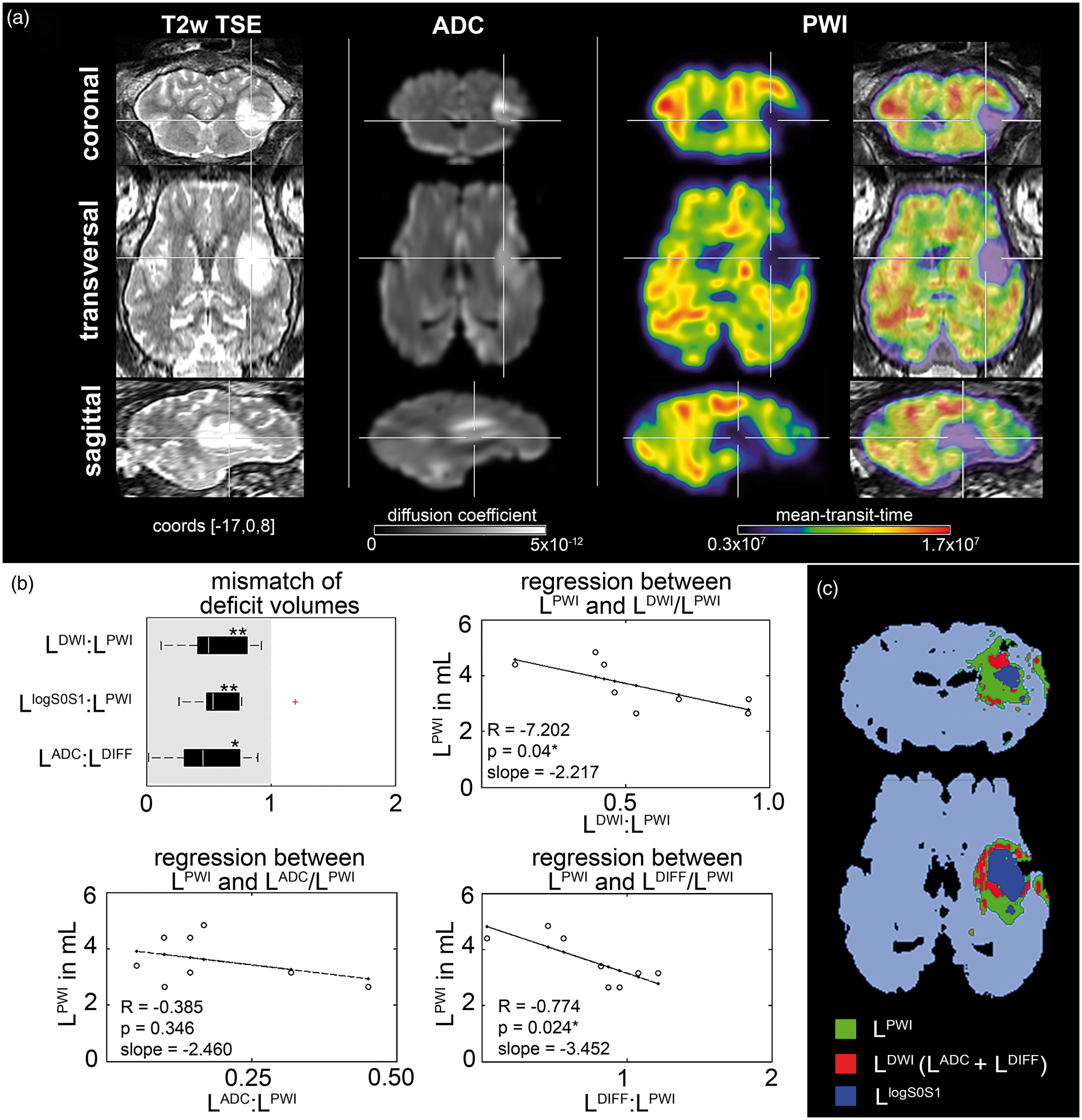
The volume of free water diffusion LDIFF (1.2 ± 0.55 mL) could be clearly segmented. A restricted diffusion volume LADC of 0.6 ± 0.36 mL with lowered ADC values as compared to unaltered brain tissue surrounded the cavity. This resulted in an overall diffusion disturbed volume LDWI of 1.9 ± 0.7 mL. A perfusion deficit measured by MTT delay was detected with 3.6 ± 0.85 mL which surrounded both LlogS0S1 and LDWI (Figure 3(c), see Supplementary Table 5 and Supplementary Figure 2 for more details).
Structural lesion, perfusion and diffusion deficit
DICE’s coefficient and corresponding volumetric mismatch correlated significantly (R2 = 0.745,
Volumetric mismatch and spatial similarity by DICE’s coefficient between lesion and deficit volumes (mean ± SD/quartile I-III).
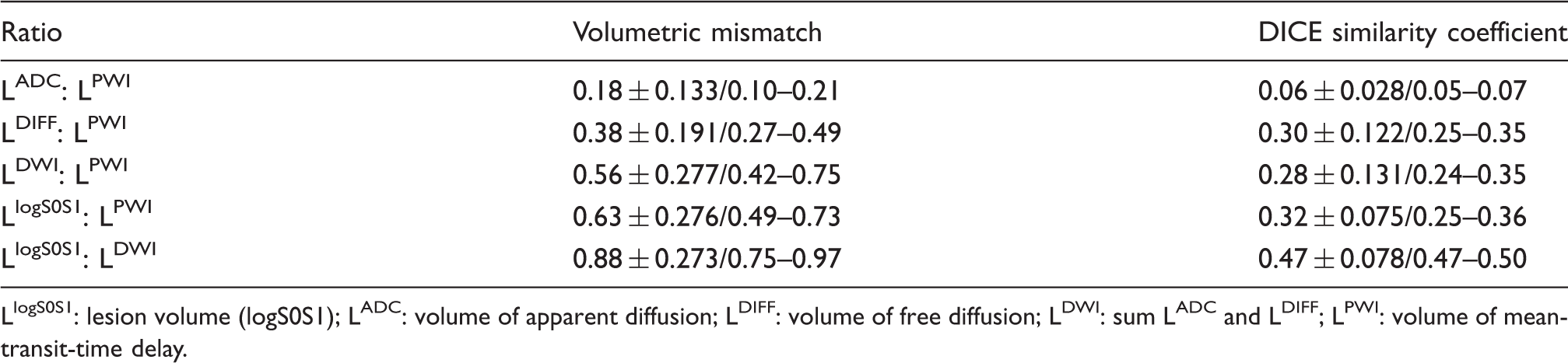
LlogS0S1: lesion volume (logS0S1); LADC: volume of apparent diffusion; LDIFF: volume of free diffusion; LDWI: sum LADC and LDIFF; LPWI: volume of mean-transit-time delay.
Remarkably, significant relations between PWI deficit volume and DWI/PWI mismatch (
Anatomical and histopathological examinations
Macroscopic investigation of fixed brain tissue revealed a circumscribed, blood-filled cavity containing hematoma (Figure 4(n)). The shape of the cavity varied between animals; therefore, the degree of grey matter and white matter involvement was unequal. There was no evidence of ventricular disruption or leakage of blood to the subdural CSF space.
Histological examination revealed ischemic tissue changes adjacent to the cavity in all animals examined. These changes included white matter vacuolation (Figure 4(a) and (b)) and blood in the perivascular space around small vessels (Figure 4(c)). Such tissue alterations extended to brain tissue that was relatively distant (>10 mm) from the blood cavity (
Intact microglia and astrocytes were evident adjacent to the hematoma (Iba-1, GFAP labeling, examples in Figure 4(d) and (e)). MBP-labeled sections confirmed white matter vacuolation (Figure 4(f)). Swelling and bulbing of axons within white matter were seen adjacent to the blood cavity in neurofilament-labeled sections (Figure 4(g) to (i)). In the overlying grey matter, some large neuronal cells (not all) exhibited shrinkage of the cell body and nucleus, eosinophilia, and fading (Figure 4(j)), consistent with early stage “neuronal hypoxic-ischemic” changes.
Plasma protein fibrinogen was detected in and around the hematoma (Figure 4(k)). This was expected since we injected whole blood including plasma. Fibrinogen labeling was also observed within neuronal cells distant (>10 mm) from the lesion in some subjects (
Discussion
We developed an ICH model in adult sheep by stereotactic infusion of autologous heparinized blood as well as an advanced neuroimaging routine and image analysis pipeline. The model reflects a space-occupying intracerebral bleeding of moderate severity predominantly affecting white matter. It allows for application of multimodal clinical imaging protocols to quantitatively characterize the ICH and perilesional features in an objective and reproducible manner. Here, we showed that the perilesional area after acute ICH consists of diffusion disturbances and restricted perfusion along with histological alterations of neurons, their axons, and the perivascular space.
Ovine ICH model
As a novel model for adult ICH in sheep, we simulated acute moderate ICH by injecting a blood volume of ∼2% of the total brain volume. This did not induce mortality during experimentation, but human non-fatal ICH affects up to 15% of the brain. 22 The ovine ICH model is feasible for long-term neurofunctional assessment, e.g. as described in Boltze et al. 12 This will further augment experimental options in future investigations by using the sheep model, but was beyond the scope of this studies. Long-term mortality rate remains to be determined but is expected to be very low due to the well controllable lesion induction. Larger lesion volumes and prolonged study designs need to be applied to further investigate dynamic properties of clinical ICH. This will allow to explore therapeutic interventions, hematoma clearance, and edema expansion. 23 In contrast to other experimental ICH modalities such as collagenase 24 or incision of cerebral vessel, 25 autologous blood injection allows to study well-controlled effects without spontaneous pre-terminal mortality in vivo, which is an advantage for longer term studies. One limitation of our model is the use of autologous blood injection. More realistic models for ICH such as collagenase injection are available. 26 These models are more volatile in lesion configuration and outcome, nicely mimicking the situation in human ICH patients. 27 However, we decided to employ the blood injection model because of its more reproducible lesion configuration to establish an imaging-based analysis method. Its similarity to human ICH on the histological scale was proven by histopathological assessments. Future studies may now rely on the more realistic, collagenase-based ICH model for the evaluation of therapeutic and diagnostic concepts. Animal survival as well as follow-up behavioral assessments is essential in this context.
Although stereotactic application enables precise blood deposition, the resulting cavity slightly varies in spatial characteristics. This may be due to local brain tissue texture or, to a minor extend, the individual trajectory angle. The stereotaxic system allows for navigation according to inter-subject coordinates that are available in the ovine brain coordinate system. 17 This, however, comes at the cost of somewhat limited consideration of individual anatomy.
Neuroimaging and image analysis pipeline
There is an urgent need for a quantitative comparison of key pathophysiological parameters such as hematoma expansion, blood clearance, edema, white matter changes, and iron in human and animal ICH. Large, gyrencephalic animal models enable the generation of a wide spectrum of detailed image information by using clinical imaging equipment28,29 allowing for direct comparison to the patient data.
Automatic processing pipelines are the method of choice when considering reproducibility, practicability, and exact reporting. 11 Although image classification into tissue classes is well-established, automatic lesion segmentation, in particular of large territorial lesions, is still complicated and often necessitates manual interventions. A potential reason may be that commonly available software solutions are optimized for human diagnosis.
Here, we show that an automatic classification of the induced lesion and brain tissues is possible when considering ICH localization in the ovine brain MRI as an additional tissue class. Moreover, the approach enables creating a model-specific image processing pipeline, which is based on former evaluations in sheep. 17
Quality of image processing
Overall, the visual inspection of automatic segmented brain masks did not reveal obviously misclassified voxels. Further, there was relatively high spatial similarity between manual and automatic processing shown by DICE’s coefficient and as compared to similar approaches in other animal models including large lesions. 30 Thus, the automatic lesion segmentation procedure is a reproducible and practicable method.
The automatic segmentation procedure described in this paper needs to be adapted to accommodate lesions in different anatomical brain areas. Although a certain spatial variability of the hemorrhage is considered and, therefore, the automatic segmentation procedure should cover different ICH volumes and shapes, further implementation of the lesion probability map may improve usability and, prospectively, support more variable model approaches. 31
Tissue changes after ICH in sheep
Structural ICH alterations were analyzed by the routine MR sequences T1w, T2w TSE, and T2*. However, there is a debate which sequence is optimal to depict lesion volume most accurately, especially when assuming hyperintensities due to perilesional edema.32,31 We defined the structural lesion as T1w modality with T2 signal attenuation to consider such alterations which were detected by T1 and T2 MR signals. Other MR sequences also depict aspects of ICH, such as fluid-attenuated inversion recovery (FLAIR) for selective edema visualization or susceptibility-weighted imaging (SWI) for visualization of iron content, but at the cost of lower tissue contrast (FLAIR, SWI) and prolonged scan duration (SWI) 13 compared to the herein applied sequences. However, additional image modalities such as positron-emission-tomography may contribute prospectively to visualize perilesional perfusion and/or metabolism. 33
Using the aforementioned definition, alterations of global brain tissue volumes in GM and WM were detected as expected. The ventricular changes corresponded to an increased Iv:b. Thus, it can be concluded that the ipsilateral ventricle was compressed at the lateral aspect while both olfactory ventricles were dilated, which was confirmed by VBM analyses.
This may be due to the space-consuming ICH and/or a dysfunction of CSF efflux. However, it is not clear whether the findings result from passive, space-occupying effects alone or if these are supported by active processes such as vasoconstriction and subsequent edema by perivascular hemoglobin as indicated by the histopathological findings.34,35 It needs to be taken into account that the olfactory ventricles are larger in sheep than in human or rodent, 36 which may increase the absolute dimension of the observed effect and a species-specific reaction cannot be entirely excluded. Further studies have to clarify the underlying mechanism of this phenomenon.
We further demonstrate perilesional restriction of diffusion and perfusion at 6 h after ICH which corresponds to findings in patients.37,38 The herein defined structural lesion LlogS0S1 represents not only T1w abnormalities but includes also T2w hyperintensities such as cytotoxic cell edema and free-floating water. 39 Therefore, the almost comparable volumes of the defined LlogS0S1 and the diffusion deficit LDWI were not surprising. However, the LDWI deficit includes not only free floating water (LDIFF) but also a region of restricted water diffusivity (LADC). The mismatch between both is relatively wide and, therefore, a limited diffusion solely due to compressed tissue may be unlikely and both findings indicate additional and/or separate reasons leading to perihematomal alterations.
Restricted diffusion may contribute significantly to progression of perilesional edema. In fact, it is not clear if the tissue of restricted diffusion (LADC) is assigned to disappear or may be part of a potential therapeutic target. 40 Recent studies showed that perihematomal diffusion restriction depends on ICH volume, but does not independently predict outcome although the diffusion restriction correlated with in-hospital mortality and disability rate or death. 41 However, other studies indicate that the perihematomal edema expansion is associated with poor clinical outcomes.22,42 Reports from patients after ICH and from other animal models also showed perihematomal tissue changes including perilesional edema. 43 In our model, histopathological findings adjacent to the hematoma revealed WM vacuolation, axonal disruptions, edema, and ischemic neuronal cell changes (Figure 5). Therefore, we propose to consider a hypothesis of accompanying compressive and vasoconstrictive effects in the early acute phase of ICH.
The volume of PWI deficit was larger compared to the volume of structural lesion or diffusion disturbances. This further supports genesis of tissue alterations next to ICH which are also evident in ICH patients. 44 Indeed, histological examination showed perivascular fibrinogen in areas distant to the hematoma (which therefore unlikely emerged from the injected blood), possibly indicating a blood–brain barrier alteration. This is consistent with findings in rodent models of ICH. 45 Interestingly, the PWI deficit negatively correlated with DWI/PWI mismatch and LDIFF:LPWI mismatch, but not with the volume of ADC. As recently reported in a subarachnoid hemorrhage model, 46 perivascular hemoglobin may contribute significantly to acute vasoconstriction. However, further studies are required to clarify the role of vasoconstriction in the pathogenesis of ICH.
Together with histological findings adjacent to the hematoma, the perfusion-restricted tissue may further contribute to brain edema formation 47 but may also serve as a potential therapeutic target. 48 In fact, the pathogenesis of cytotoxic edema is controversially discussed since other researchers did not find a link between perilesional CBF restriction, blood pressure, and perilesional edema in patients. 49 The ovine ICH model may allow to resolve and clarify these issues in further studies.
Conclusion
This novel gyrencephalic ovine ICH model offers a platform to further investigate (i) hematoma expansion, a key feature of human ICH pathology, (ii) hematoma clearance, when larger blood volumes (ICH diameters) are applied, (iii) therapeutic hematoma evacuation, that is performed in current clinical ICH trials (e.g. MISTIE and MISTIE III, NCT01827046) 23 and offers the potential of drug/biomaterial administration into the cavity, and (iv) white matter damage. The translational platform will allow refinement of clinical trial parameters, including dosing, therapeutic window assessment, and target engagement.
Supplemental Material
Supplemental material for Lesional and perilesional tissue characterization by automated image processing in a novel gyrencephalic animal model of peracute intracerebral hemorrhage
Supplemental material for Lesional and perilesional tissue characterization by automated image processing in a novel gyrencephalic animal model of peracute intracerebral hemorrhage by Johannes Boltze, Fabienne Ferrara, Atticus H Hainsworth, Leslie R Bridges, Marietta Zille, Donald Lobsien, Henryk Barthel, Damian D McLeod, Felix Gräßer, Sören Pietsch, Ann-Kathrin Schatzl, Antje Y Dreyer and Björn Nitzsche in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was co-funded by European Union (EFRE-No: 100109012). AHH and LRB are funded by Alzheimer's Society (UK) (PG146/151) and Alzheimer's Research UK (PPG2014A-8).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
JB, BN, FF and MZ designed the study and wrote the manuscript. Animal experiments were carried out by FF, DL, SP, AKS, AD and BN. MR Image analyses were performed by SP, FG and BN. AHH and LRB performed histochemistry and histological imaging. All Authors edited the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
