Abstract
Cerebral blood flow (CBF) is the most common parameter for the quantification of brain's function. Literature data indicate a widespread dispersion of values that might be related to some differences in the measurement conditions that are not properly taken into account in CBF evaluation. Using recent high-resolution imaging of the complete cortical microvasculature of primate brain, we perform extensive numerical evaluation of the cerebral perfusion. We show that blood perfusion associated with intravascular tracers should be normalized by the surface of the voxel rather than by its volume and we consistently test this result on the available literature data.
INTRODUCTION
Cerebral blood flow (CBF) is a key parameter for quantifying normal, altered, or pathologic brain functioning, both in medical current practice and in cognitive neuroimaging studies. Relative comparison of this parameter is generally used for the purpose of identifying a given effect after a treatment or a condition change at the individual level.
1
Nevertheless, there is also a great interest in providing consistent perfusion estimation which, with different methods, differs by a fair amount.2–4 As stated by Kudo
In this contribution, we challenged intrinsic CBF pixel size-independent normalization with a new standpoint. We analyzed, at the fundamental level, the blood perfusion inside the cerebral cortex starting from an exhaustive description of its vessel network from pia surface down to capillary scale inside gray matter. We specifically focus on the contribution of intravascular blood flow to cerebral perfusion.
MATERIALS AND METHODS
Synchrotron microtomography
7
has been used to image Barium particles in solution injected in primate brain microvascular networks with a 1.43 μm3/voxel resolution. The animal (5-year-old male marmoset
The blood perfusion inside this highly complex network is then computed with a network method
10
that permits the evaluation of the blood pressure and flux inside each individual vessel segment. Finally, the statistical analysis of the data discussed in the following sections uses classic Student's
RESULTS
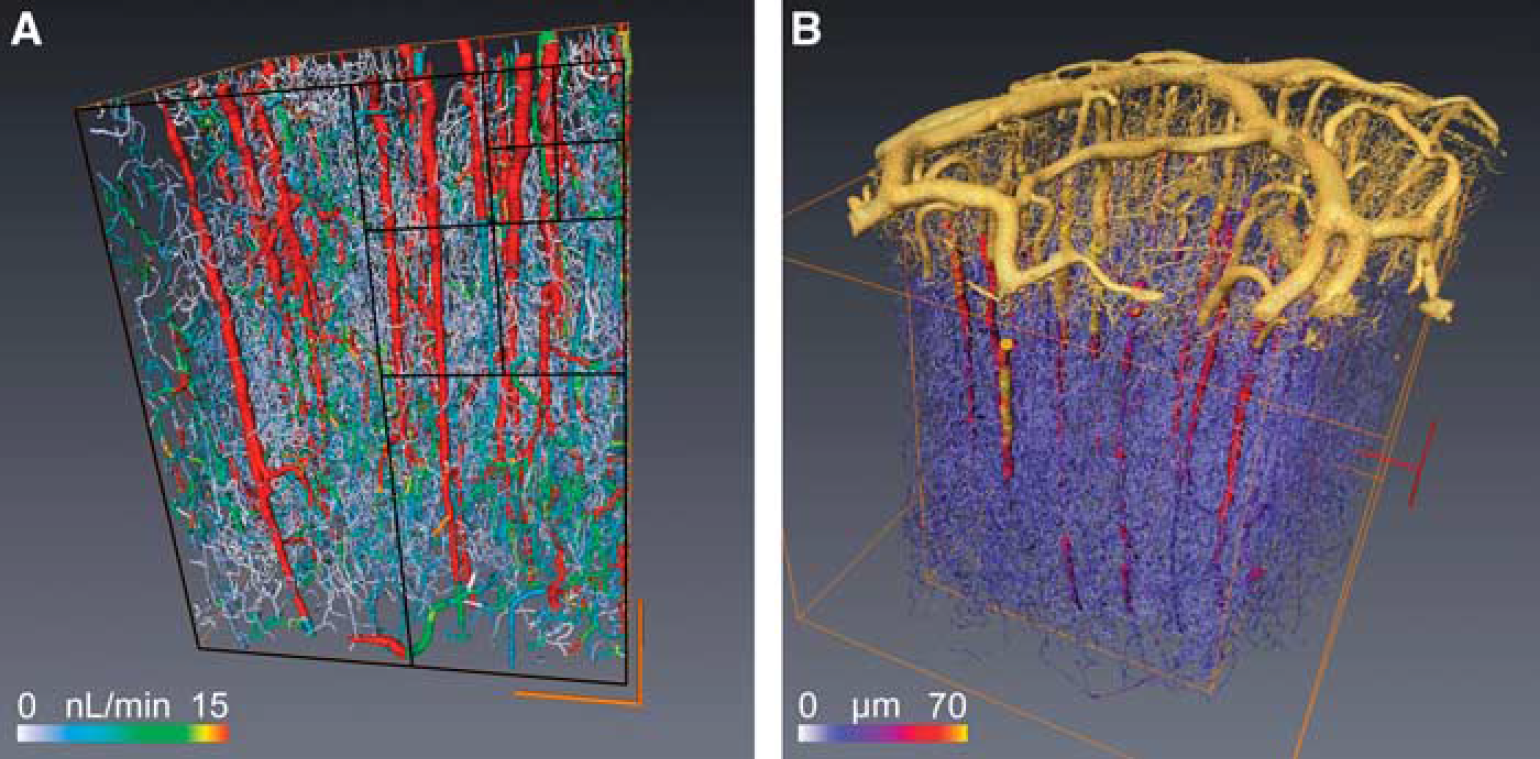
Figure 1A illustrates the largest cube of 8 mm3 that could be extracted from the complete reconstruction of a cylindrical sample of primate cortex excluding the pia matter (Figure 1B), which is essential for applying relevant boundary conditions.
10
The resulting prediction for the blood pressure and flow distribution depends on the considered blood rheological model; we already analyzed this issue in a recent study.
10
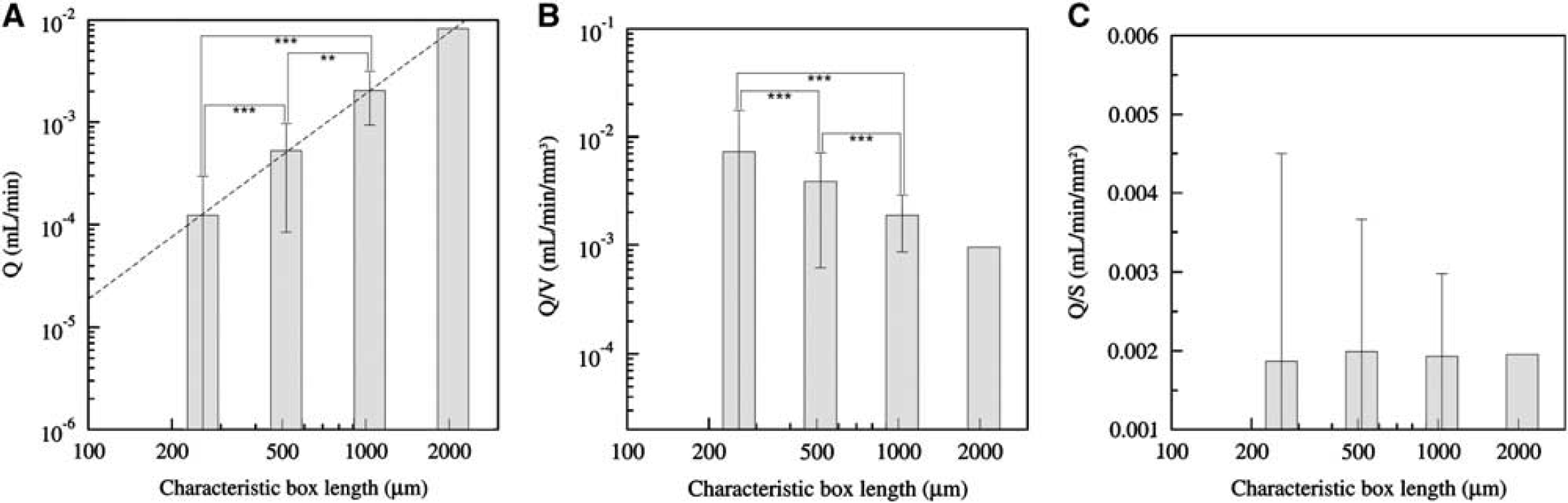
Here, we show that the CBF measurement depends on the size of the processed virtual voxel, i.e., the size of the cubic box. From evaluating CBF using different voxel sizes, we have been able to analyze its size dependence. The total blood perfusion flux Q in a given voxel is directly evaluated from summing the input contributions of all the vascular segments that are also equal to the output ones since blood is incompressible. It obviously increases when sampling more and more vessels so that it is significantly different when varying the voxel-box size (
(
(
DISCUSSION
The results obtained in the previous section can be understood from a more theoretical viewpoint by considering that each contribution to the voxel perfusion Q results from the contribution of each vascular segment input. It is thus proportional to the sum of the input flow coming from each input segment. When the vascular segment number is large, the total perfusion is then well approximated by the average flux per segment multiplied by the total number of segments. Since the voxel size is large (superior to 2503 μm3) compared with the vessel's spatial correlation length (estimated between 50 and 80 microns 12 ), it can be considered that the surface vessel density is almost homogeneous along each voxel surface (because correlation length provides the scale above which the vessels random distribution can be considered as homogeneous for providing a sufficient statistical sampling). In this context, it is possible to approximate the number of input vessels by half the voxel surface times the vessel density (the other half being the total number of output vessels for which the blood leaves out flows outside the considered box). Since the total perfusion Q is proportional to the number of vessels, it is thus proportional to the voxel surface S, as confirmed by numerical simulations and Figure 2C. One might wonder whether this reasoning could lead to different consequences if, instead of using input arterial units, we chose the venular output ones, since their proportion is different (penetrating arteries represent 2/3 of all penetrating vessels 10 ). Since inputs and outputs contribute to the same amount of total flux (a fact that we have explicitly checked in the numerical results), the weaker proportion of veins will be compensated by a larger average flux, and the proportionality of the total output perfusion flux with voxels surface is unchanged. Similarly, one could also question the fact that our results might be influenced by the undesirable contribution of the large penetrating vessels to the perfusion. Filtering the penetrating vessels contribution to the input flux leaves a similar surface rescaling to the CBF (not shown).
Furthermore, since we have only considered the influence of blood convection in our CBF evaluation, the role of supplementary diffusive processes on the proposed surface rescaling is questionable. Basic transport theory states that material transport goes as the flux times the area, implying that flux measurements should be normalized by the tissue area. Our findings thus highly suggest to consider a new reference parameter for brain perfusion that could be denoted as CBFs (s for surfacic CBF) which should scale as CBFs = CBF.V/S where V and S are the volume and the surface of the considered voxels. It is difficult to provide a convincing confirmation of this proposition from considering the available CBF data of the literature. First, because even though a large number of CBF measurements can be collected, voxel volume and surface values are rarely provided (even deduced from the acquisition set-up). Second, CBF values are found in various species (rabbit, 1 rat, 2 dog, 13 human,3,14–21 and pig 22 ) for various ages, sex, and brain regions, using various methods (CT, single-photon emission computed tomography, positron emission tomography, and magnetic resonance imaging). For low-resolution measurements, the possible joined contribution of gray and white matter could influence local CBF. Furthermore, the voxels are rarely cubic, and, most of the time, they are elongated parallelepiped (for which pial vessels might contribute). All these considerations could weaken the relevance of the proposed scaling for explaining the observed CBF variability when tested on the available data of the literature. Hence, our proposition should be challenged in the future using new dedicated measurements.
Finally, it is interesting to mention that the filtration velocity (or Darcy flux 23 ), which is considered in porous media, is also a flow per surface unit (and not per volume unit) consistent with our finding for the brain perfusion. We hope that this work could improve CBF overall quality assessment and its validity domain, thus contributing to its better use in preclinical and clinical studies.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors thank Pr A Steyer for interesting discussions.
