Abstract
Blood–brain barrier (BBB) dysfunction has been implicated in ischemic risk following aneurysmal subarachnoid hemorrhage (aSAH), but never directly imaged. We prospectively examined whether post-bleed day 4 dynamic contrast-enhanced magnetic resonance (DCE-MR) BBB permeability imaging could predict development of delayed cerebral ischemia (DCI). Global MR-derived BBB permeability (
Keywords
Introduction
Delayed cerebral ischemia (DCI) is a multifactorial process often concomitant with radiographic cerebral vasospasm that occurs in approximately 30% of patients after aneurysmal subarachnoid hemorrhage (aSAH).1,2 Neurological deficits from DCI account for the majority of in-hospital morbidity and mortality in aSAH patients, and dramatically impact functional outcomes. Prospective identification of those patients at highest risk for DCI following aSAH is nonetheless challenging, as current assessments of ischemic risk are largely based on crude measures of subarachnoid blood,3–5 and radiographic vasospasm can occur with or without clinical or ischemic sequelea. 6
While the exact mechanisms underlying cerebral vasospasm and DCI following aSAH remain poorly understood, the importance of early microvascular dysfunction and the inflammatory response in these processes has been established in laboratory, translational, and clinical models.7–15 Under normal homeostatic conditions, the brain is a largely immune-privileged organ due to the presence of vascular endothelial cell tight-junctions comprising the blood–brain barrier (BBB). Following aSAH, pathologic extravasation of leukocytes through the BBB microvasculature leads to an upregulation of extravascular inflammatory mediators, the scale of which is proportional to vasospasm and DCI risk.10,13 BBB dysfunction has also been directly linked to DCI risk through serum elevations of basal membrane catalytic enzymes such as matrix metalloproteinases (MMPs).16–18 Together, these data suggest that high-resolution imaging of BBB permeability could act as a novel predictor of DCI following aSAH.
In this study, we assess the ability of advanced dynamic contrast-enhanced magnetic resonance (DCE-MR) permeability imaging,19,20 to identify early and subtle BBB changes following aSAH and prospectively predict risk of DCI.
Materials and methods
This study was approved by and conducted in accordance with the ethical standards of the Institutional Review Board (IRB) of the University of Southern California, and performed in compliance with Health Insurance Portability and Accountability Act regulations. Patient consent for procedures, data collection and review was obtained based on institutional guidelines. Data on consecutive patients presenting with aSAH were collected. In addition to standard aSAH care (detailed in the Supplemental Materials and Methods), a DCE-MR scan on post-bleed day (PBD) 4 (prior to the day 7 peak vasospasm period) 21 was obtained. Patients were screened daily for vasospasm using transcranial Doppler (TCD) ultrasonography, and those with elevated TCDs or neurologic declines underwent computed tomographic angiography (CTA) to assess for radiographic vasospasm.
Primary study outcome was development of DCI (defined as a new onset focal neurologic deficit with radiographic vessel narrowing without other pathologic explanations) within 14 days of aSAH. Patients were classified into three groups based on clinical course: no vasospasm or ischemia, radiographic vasospasm only, and DCI, with MR-derived BBB permeability compared across groups.
DCE-MR imaging
DCE-MR images were obtained and processed as detailed in the Supplemental Materials and Methods. Briefly, user defined regions-of-interest (ROIs) were placed in standardized representative vascular territories (Figure 1). Patlak linear regression analysis with individual vascular input functions was used for voxel-by-voxel calculation of the BBB transfer constant between blood plasma and the extravascular extracellular space ( DCE-MR blood–brain barrier imaging (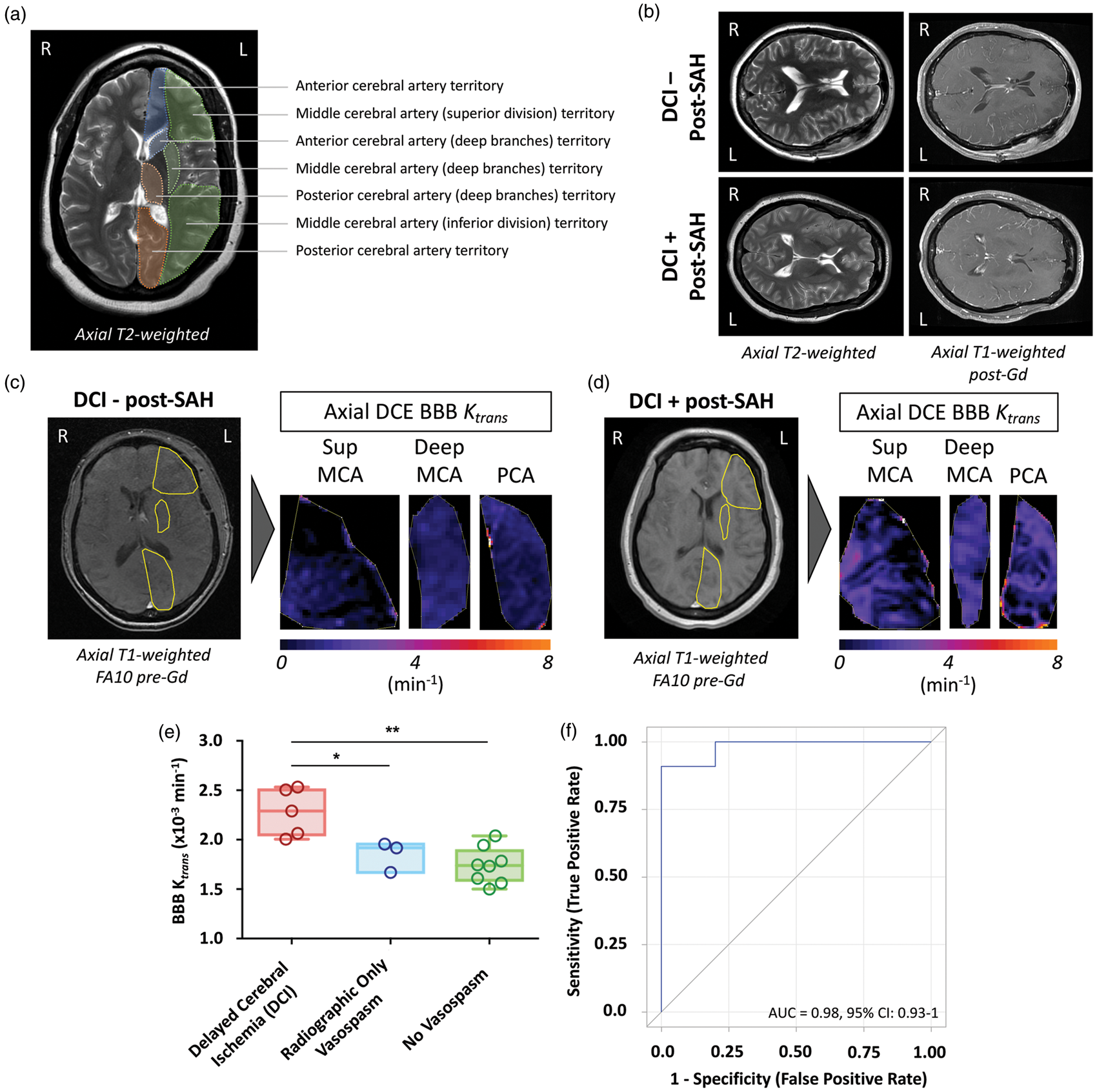
Data analysis
See Supplemental Materials and Methods for full data analysis. Briefly, data normality was assessed using a scatter plot and Anderson–Darling test. The random effect model was used to assess inter-group and inter-ROI variations and interactions, prior to post hoc contrast or ANOVA testing. Hochberg’s step-up procedure was used for correction of multiple testing errors. Receiver operating characteristic (ROC) curves were used to assess the discrimination power of DCE-MR to predict DCI. SAS9.4 software (SAS Institute, Cary, NC) was used for all data analysis. An alpha level of 0.05 was used as the experimental wise error.
Results
Baseline patient data and vasospasm outcomes
Baseline patient data and clinical outcomes.
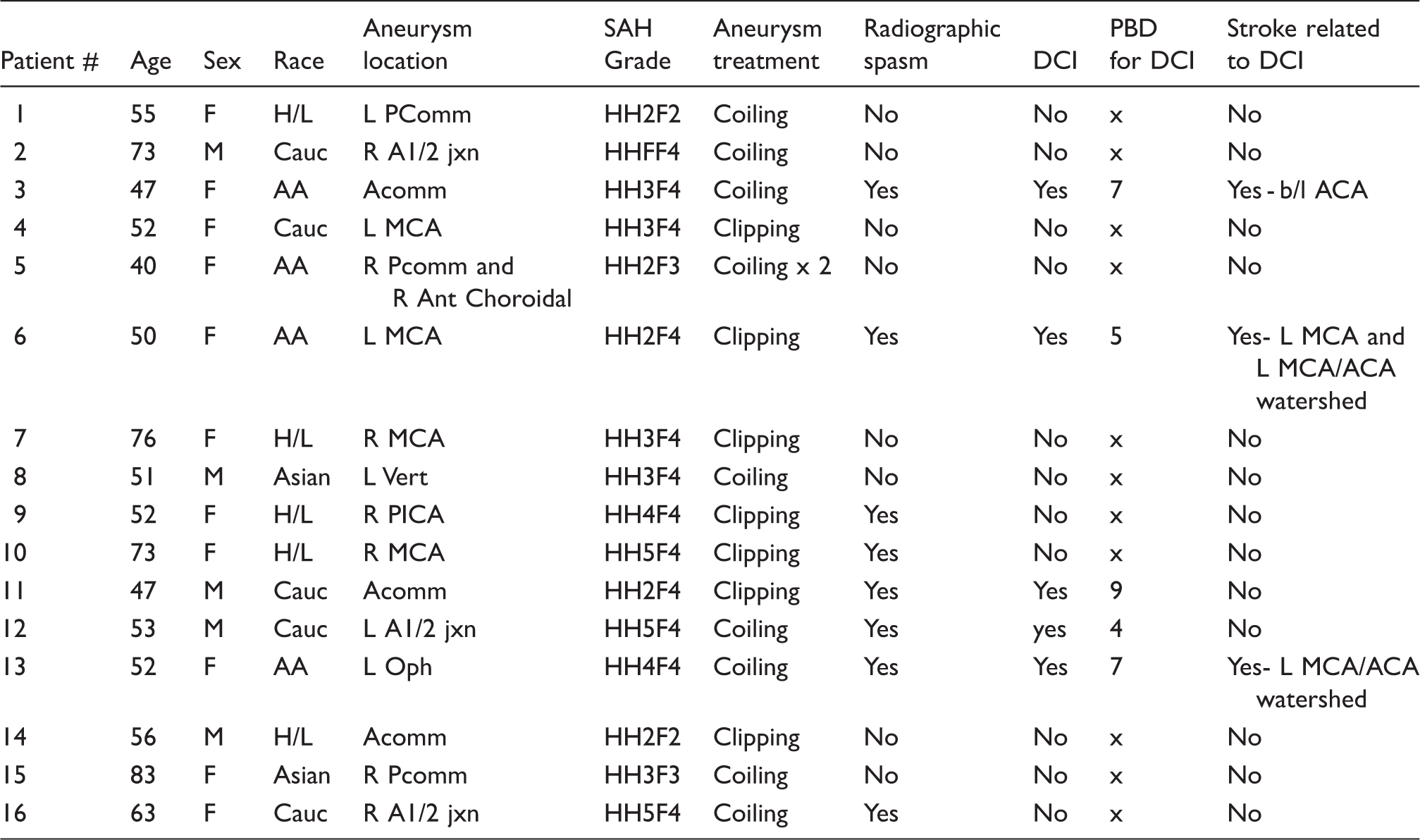
AA: African American; Acomm: anterior communicating artery; ACA: anterior cerebral artery; Cauc: Caucasian; MCA: middle cerebral artery; F: female; H/L: hispanic/latino; HH/F: hunt Hess/Fisher scales; M: male; Oph: ophthalmic artery; Pcomm: posterior communicating artery; PICA: posterior inferior cerebellar artery; Vert: vertebral artery.
DCE-MR predicts clinical vasospasm
Three DCE-MR parameters were assessed. A statistically significant intersubject variability in the volume of the extravascular extracellular space per unit volume of tissue (
Average DCE-MR
Discussion
The link between BBB permeability and DCI following aSAH has been explored in previous works, but not directly imaged. The first step in this process, microvascular dysfunction, begins immediately after aSAH and includes pathologic vasoconstriction, luminal platelet aggregation, and activation of early inflammatory cascades.14,15 Increased BBB permeability also occurs within 24 h post-hemorrhage as a result of endothelial dysfunction from oxidative stress and structural breakdown, as well as basal lamina degradation.14,15 These BBB alterations allow migration of macrophages and neutrophils into the subarachnoid space to ‘clean up’ extraluminal red blood cells as they begin to undergo hemolysis.22–31 There is no recognized pathway for these leukocytes to return to the circulation, however, and after approximately two to four days, they begin to undergo apoptosis. 32 These combined events promote the release of pro-inflammatory and vascular endothelial cell-derived spasmogenic molecules, including intercellular adhesion molecule-1 (ICAM-1), endothelin 1, and E-selectin, and are thought to contribute to the development of large-vessel vasospasm and DCI.8,33–36
While it is therefore logical that existing DCI risk scales are largely based on aSAH blood volumes,3–5,37 which should be proportional to the resulting inflammatory response, these scales have relatively poor predictive value. Several molecules have nonetheless linked BBB disruption following aSAH directly to DCI risk, suggesting the BBB as a potentially more clinically predictive target than SAH volume. MMPs released by microglia, astrocytes, and endothelial cells in the brain during extracellular matrix remodeling are one such molecule and have been previously identified as contributing to inflammatory conditions in the nervous system. 38 Multiple clinical studies measuring circulating soluble MMPs following sSAH have also demonstrated significantly higher MMP levels (specifically MMP-9) prior to the development of DCI.17,18 Additionally, in animal models of SAH, downregulation of MMP-9 expression has been shown to decrease the incidence of cerebral ischemia, 39 consistent with a contributory role of BBB dysfunction in DCI. Several other factors have also been implicated in BBB dysfunction after aSAH, including vascular endothelial growth factor, mitogen-activated protein kinase, hypoxia-inducible factor-1α, von Willebrand factor, and the upregulation of aquaporins,17,40–43 some of which have been linked to DCI risk.
Building on these works, we hypothesized that global changes in the BBB, if measurable, should be more pronounced in patients that experience DCI versus those who do not. Our data support this hypothesis, as DCE-MR permeability did not vary significantly among ROIs in the same patient, but still predicted the development of DCI as compared to radiographic vasospasm only or no vasospasm/ischemia. These findings are also consistent with the increasing recognition of the complex global vascular pathophysiologic processes that separate DCI from asymptomatic, ‘mechanical’ large-vessel narrowing. 44 Although our data differ subtly from a recently published study linking focal rather than global computed tomography perfusion (CTP) changes (an indirect marker of BBB dysfunction) with DCI, 45 this retrospective analysis used only regional in-patient controls and did not assess CTP in patients with radiographic vasospasm only or no vasospasm/ischemia, limiting the interpretation and clinical utility of this work.
Mechanistically, while it is unclear whether the early BBB dysfunction characterized by DCE-MR plays a causal role in subsequent DCI development, or is itself a concomitant marker of more influential upstream microcirculatory pathologic processes, the ability of BBB modulating agents to affect DCI risk suggests at least some causality between these processes. 39 As the previously identified serum markers of vasospasm risk only indirectly assess BBB permeability,17,18 the ability to directly assess global BBB permeability changes that predict DCI risk via non-invasive DCE-MR represents a major advancement for aSAH research. Moreover, because DCE-MR imaging was completed on PBD 4, correlating with peak levels of circulating biomarkers associated with BBB dysfunction but preceding the highest DCI risk period,17,18,21 these data provide a clinically relevant window for potential alterations in the management of high-risk aSAH patients in future studies.
Despite the relatively small sample size of our study, its prospective design allowed for robust statistical analyses to determine not only a significant correlation between DCE-MR and DCI, but also the discrimination power of this imaging modality using ROC curves. With a statistical power comparable to several recent reports assessing other imaging modalities after aSAH,45–48 our data highlight the clear clinical potential of MR permeability testing after aSAH. Future, larger studies will nonetheless be needed to confirm the findings of this pilot work.
Conclusion
BBB MR-permeability imaging can accurately identify patients at highest risk for developing DCI following aSAH.
Supplemental Material
Supplemental material for Permeability imaging as a predictor of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage
Supplemental material for Permeability imaging as a predictor of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage by Jonathan J Russin, Axel Montagne, Francesco D’Amore, Shuhan He, Mark S Shiroishi, Robert C Rennert, Jena Depetris, Berislav V Zlokovic and William J Mack in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
Brain Aneurysm Foundation Carol Harvey Memorial Grant; NIH ES024936 WJM; NIH #AG 052350 and Leducq Foundation #16 CVD 05 BVZ; KLFTR000131 MSS.
Acknowledgements
The authors thank Dr. Steven Cen for his assistance with statistical analysis for this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
JJR and WJM contributed to the study concept and design. JJR, AM, FD, SH, MSS, RCR, JD, BVZ and WJM contributed to data acquisition, analysis, and interpretation of data, and/or manuscript drafting/critical revising for intellectual content. All authors approved the submitted version of the manuscript to be published.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
