Abstract
Even though opening endothelial tight junctions is an efficient way to up-regulate brain drug delivery, the extravasation of blood-borne components from the compromised tight junctions can result in adverse consequences such as edema and neuronal injuries. In this work, we developed a nanoagonist that temporarily opened tight junctions by signaling adenosine 2A receptor, a type of G protein-coupled receptor expressed on brain capillary endothelial cells. Magnetic resonance imaging demonstrated remarkable blood–brain barrier permeability enhancements and significantly increased brain uptakes of both small molecular and macromolecular paramagnetic agents after nanoagonist administration. Gamma ray imaging and transmission electron microscope observed tight junction opening followed by spontaneous recovery after nanoagonist treatment. Immunofluorescence staining showed the unspoiled basal membrane, pericytes and astrocyte endfeet that enwrapped the vascular endothelium. Importantly, edema, apoptosis and neuronal injuries observed after hypertonic agent mediated tight junction-opening were not observed after nanoagonist intervention. The uncompromised neurovascular units may prevent the leakage of blood-borne constituents into brain parenchyma and accelerate tight junction recovery. Considering blood–brain barrier impermeability is a major obstacle in the treatment of central nervous system diseases, nanoagonist-mediated tight junction opening provides a promising strategy to enhance brain drug delivery with minimized adverse effects.
Keywords
Introduction
Even though numerous drug candidates have been developed for the treatment of central nervous system (CNS) diseases, most of them are unable to translate into clinic due to their incapability to cross blood–brain barrier (BBB). As a dynamic physiological interface, BBB preciously regulates the movement of molecules, ions and cells between the blood and the brain. Due to its extremely low permeability, BBB provides a precisely regulated environment for neurological functionalities such as neuronal circuits, synaptic transmission and neurogenesis. However, it is also a formidable obstacle preventing delivery of therapeutics into brain. 1 Therefore, there is a tremendous need to increase brain drug uptake by tuning BBB permeability efficiently and safely.
BBB is made up of brain capillary endothelial cells (BCECs), pericytes, basal membrane (BM) and astrocytes. BCECs line along the vessel and completely encircle the vascular lumen. Like a thin layer, BM embraces the abluminal surface of BCECs. Pericytes are small cells attached on the abluminal side of the vessels. They directly contact with endothelial cells but are separated from brain parenchyma by the BM. Astrocytes communicate with the BCECs with their endfeet covering the BM and pericytes. BCECs play a predominate role in maintaining BBB tightness. Compared with the endothelium in the peripheral capillaries, BCECs exhibit fewer fenestrations, lower pinocytotic activity, 2 but higher expression level of efflux transporters such as P-glycoproteins (P-gp) that actively pump drugs out of the cells. 3 Meanwhile, tight junctions (TJs) seal the adjacent BCECs securely and possess a much higher electrical resistance (1000–2000 Ω cm2) than that of peripheral capillaries (2–20 Ω cm2), 4 which prevents the intracerebral uptake of substances with high polarity. Additionally, the average pore size of brain capillaries was determined as 0.4 nm, which is much lower than the value (6–7 nm) of the non-neural vessels.5,6 Therefore, brain drug deliveries via either paracellular or transcellular pathways are restricted. Although heterogeneous BBB disruption is observed in a variety of CNS disorders such as brain ischemia, 7 tumor 6 and neurodegenerative diseases, 8 delivering drugs by taking advantage of the pathological BBB leakage is not successful. First, uncompromised BBB is found in lesion periphery. 9 For example, even though the overall vascular permeability is increased in whole brain tumors, the BBB in tumor invasive margin usually keeps intact and prevents the drug delivery into this location where invasive cancer cells exist. 10 Second, brain capillaries maintain partial BBB characteristics even under pathological conditions. Although endothelial TJ breakdown is observed in numerous CNS diseases, BBB characteristics such as polarized distribution of the efflux transporters and down-regulation of the pinocytosis/fenestrae are persisted, which prevent the efficient brain uptake of drugs, especially the macromolecular drugs. Therefore, artificially increasing BBB permeability is necessary to improve the therapeutic response.
Current brain drug delivery strategies can be roughly classified into invasive and non-invasive approaches. For the invasive approach, drugs are injected directly into brain parenchyma/ventricles by a catheter. However, the requirements of accurate image-guidance, limited drug diffusive distance in brain parenchyma and high cost of hospitalization make it difficult for wide application. In non-invasive approaches, the therapeutics penetrate the BBB via transcellular traverse or paracellular diffusion. Receptor-mediated transcytosis (RMT) is a natural transcellular process through which receptor bound endogenous molecules traverse endothelial cells within endocytic vesicles. For example, low-density lipoprotein receptor-related protein-1 (LRP1) not only transports cholesterols from the blood into the brain but also is involved in a amyloid β-peptide (Aβ-peptide) clearance by delivering it from the brain into the blood.11,12 However, the transcytosis strategy requires the labeling of receptor targeting ligands on the drugs, 13 which inevitably attenuates their pharmacological activity. Additionally, the limited receptor density on vascular endothelium is another threshold preventing the efficient brain drug delivery. In contrast, intracerebral delivery by diffusing through the compromised endothelial TJs does not need any modifications on the therapeutics, which helps to maintain drugs’ pharmacological activities. Meanwhile, this strategy is also barely affected by physiological conditions such as receptor expression level. Therefore, temporarily opening TJs is a way of choice to increase brain drug delivery efficiency. Hypertonic agents such as mannitol were used to disrupt TJs by inducing osmotic pressure between the luminal and abluminal sides of the vessels. Nevertheless, the non-specific BBB breakdown leads to potential risks such as epileptic seizures 14 and vasogenic edema. 15 Microbubble enhanced focused ultrasound was intensively investigated recently to breakdown BBB in a specific region by creating shear forces that puncture the vessel wall. 16 Even though this strategy significantly increases drug uptake in brain tumors, uncontrollable BBB damage could lead to extravasation of blood-borne compartments such as erythrocyte, which not only disturbs the electrolytic homeostasis in the brain but also causes serious complications such as edema 17 and ischemia. 18 Therefore, tuning TJ tightness efficiently and safely is crucial for improving therapeutic response of the CNS diseases.
As the largest and most diverse class of cell surface receptors in eukaryotes, G protein-coupled receptors (GPCRs) are the targets of approximately 40% modern drugs prescribed worldwide.
19
After activation by an external stimuli, GPCRs not only modulate physiological behaviors with high velocity (ms–s) but also timely terminate above actions via agonist dissociation or receptor desensitization. Adenosine receptors (ARs) are a class of purinergic GPCRs with adenosine as endogenous agonist. In the four subtypes of ARs, A2AR shows important physiological functions such as regulating blood flow and anti-inflammation.
20
In previous work, we developed a series of A2AR nanoagonist (NA), in which multiple copies of agonistic agents were labeled on PAMAM dendrimer as a nano-scaffold.
21
These NAs specifically signal A2AR on BCECs, which induces cell contraction and paracellular TJ opening (Figure 1). Drugs are injected when the BBB permeability reaches its maximal value. The spontaneous TJ restoration after NA treatment minimizes adverse-effects such as edema induced by uncontrollable BBB leakage. Additionally, due to the prolonged circulation lifetime and amplified A2AR binding affinity, these NAs hold the potential to tune BBB permeability more efficiently and persistently than the monomeric agonists.
Schematic of the NA-mediated brain drug delivery. (a) NAs specifically activate A2AR on BCECs. (b) NA-mediated A2AR signaling triggers BCEC contraction and TJ opening. Drug is injected when the BBB permeability reaches its maximum and (c) Spontaneous BBB restoration after NA treatment minimizes the side-effects of BBB leakage.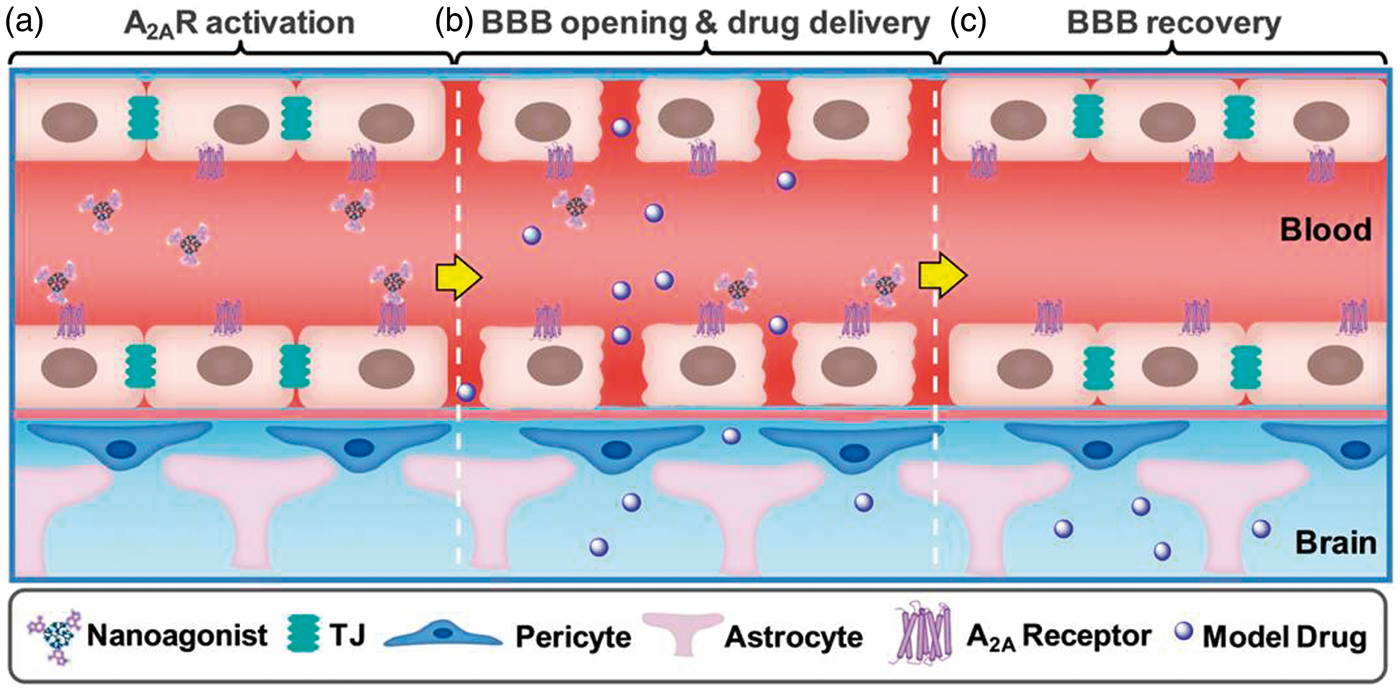
In this work, BBB permeability alternations in normal mice with intact BBB were evaluated by in vivo dynamic contrast enhanced magnetic resonance imaging (DCE-MRI) before and after intravenous (i.v.) injection of the NA. BBB restoration after NA treatment was monitored by in vivo gamma ray imaging in which radioactive tracer was administrated at selected time-points after NA injection. Edema, the most common pathological symptom after severe BBB leakage was monitored by T2-weighted (T2W) MRI. Structural variations of TJs were observed by transmission electronic microscopy (TEM). The spatial interaction between the neurovascular unit (NVU) components including endothelial cells, BM, pericytes and astrocyte endfeet was studied by double-stained immunofluorescence imaging. Pathological symptoms such as edema, apoptosis and neuronal injuries observed after mannitol treatment were not detected after NA administration. Our studies indicated that the NA-mediated TJ opening is a promising strategy for the treatment of CNS diseases by increasing brain drug delivery safely and efficiently.
Materials and methods
Materials
All chemical reagents were obtained from Aladdin Reagent (Shanghai, China) unless otherwise specified. PAMAM G5 dendrimer (MW: 28,826 Da) was purchased from Weihai CY Dendrimer Technology Co., Ltd (Weihai, China). Fetal bovine serum, trypsin and penicillin-streptomycin were purchased from Life Technologies Inc. (Carlsbad, USA). Rabbit anti-mouse AQP4, A2AR, Collagen IV, glial fibrillary acidic protein (GFAP) and active caspase-3 primary antibodies, Alexafluo488-labeled goat anti-rabbit secondary antibody were purchased from Abcam (Cambridge, USA). Goat anti-mouse CD34 primary antibodies, CruzFluor555-labeled donkey anti-goat secondary antibody were purchased from Santa Cruze (Dallas, USA). Maleimide-PEG2k-NHS and PEG2k-NHS were purchased from JenKem Technology Co., Ltd. (Beijing, China). Terminal dUTP nick end-labeling (TUNEL) apoptosis assay kit was purchased from KeyGEN Bio Teck (Nanjing, China).
Synthesis
Characterization
The molar ratio among dendrimer, polyethylglycol (PEG) and nucleotide on the nanoparticles was measured by 1HNMR (Mercury 400 spectrometer, Varian, Germany). Compounds dissolved in D2O were analyzed by integrating the characteristic proton of dendrimer (3.3–2.2 ppm), PEG (3.7 ppm) and regadenoson (9.0–7.5 ppm) in the 1HNMR spectrum. The particle size and zeta potential of the nanoparticles were measured using a dynamic light scattering detector (Zetasizer, Malvern, USA). Nanoparticles negatively stained by 2% phosphotungstic acid were imaged by a transmission electron microscope (TEM) (H-600, Hitachi, Japan). The radiochemical purity of 99mTc-albumin was determined by thin layer chromatography.
Animals
All animal experiments and procedures were approved by the Fudan University Institutional Laboratory Animal Care and Use Committee and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All sections of this report adhere to the Animal Research: Reporting in Vivo Experiments guidelines for reporting animal research. Male ICR mice, aging six to seven weeks were supplied by Shanghai Slac Lab Animal Ltd. (Shanghai, China) and housed under pathogen-free barrier conditions in animal facility at School of Pharmacy, Fudan University in a temperature- and humidity-controlled room (about 23℃ and 50%, respectively) under a controlled photoperiod (12 h light; 12 h dark). All mice were randomly assigned to groups.
In vivo MRI studies
In vivo MRI was conducted in a 7.0 T micro-MR scanner (Bruker BioSpec, Germany). Mouse body temperature was maintained by a thermostat-regulated heating pad, and the respiration rate was monitored by a Bruker Physiogard system with 30–40 breaths/min during the entire experimental procedure. According to the MR scanning protocol, the mice were first anesthetized with 1%–2% isoflurane in 20% oxygen, the tail vein was catheterized with a home-built catheter system using a small T-junction (Cole-Parmer) devised to minimize the dead volume in the tubing (<50 µL). NA (0.5 µmol/kg) was injected intravenously (i.v.) via tail vein through the catheter. Pre-contrast T1 weighted (T1W), T2 weighted (T2W) and T1-map images were acquired in turn: T2W image (repetition time (TR)/echo time (TE) = 3800/95 ms; acquisition matrix = 256 × 256; number of excitation (NEX) = 4; field of view = 20 × 20 mm; number of slices = 12; slice thickness = 1 mm, rare factor = 8, flip angle = 150°); Rapid acquisition with relaxation enhancement (RARE) T1W image (TR/TE = 600/11 ms; acquisition matrix = 256 × 256; NEX = 2; field of view = 20 × 20 mm; number of slices = 12; slice thickness = 1 mm, flip angle = 80°); RARE T1-map (TR: 3000, 1200, 800, 500, 300, 150 and 120 ms; TE: 11 ms, field of view = 20 × 20 mm; acquisition matrix = 256 × 256; number of slices = 2; slice thickness = 1 mm, flip angle: 180°, RARE factor = 2). The ΔT1 maps were obtained by subtracting the post-contrast T1 maps from the pre-contrast T1 maps; the resulting images were pseudo-colored with T1 values ranged from 0 to 5000 ms. DCE-MRI was obtained by using a fast low-angle shot (FLASH) axial T1 map (TR/TE = 16/2.8 ms; acquisition matrix = 128 × 128; NEX = 1; field of view = 20 × 20 mm; number of slices = 2; slice thickness = 1 mm) with multiple flip angles (5°, 10°, 15°, 20°, 25° and 30°) and T1W imaging (same parameters as FLASH T1 map with flip angle of 25°). At 20 s after the DCE-MRI acquisition, CA in 200 µL PBS with a dose of 0.05 mmol Gd3+/Kg was administered via the tail vein catheter. RARE T1W and RARE T1-map images post-injection (PI) of contrast agent (CA) were collected at the end of DCE-MRI.
DCE-MRI data processing method
DCE-MRI data were processed using in-house MATLAB (Mathworks, USA) programs. Arterial input function (AIF) was automatically extracted from the concentration-time curve pool by basically following the procedure described by Singh et al.
23
The only modification was computation of bolus arrival time and time-to-peak, with the method described by Kim et al.
24
To avoid partial volume effect, AIFs of PBS groups were scaled to set average cerebral blood volume of normal brain tissue as 4.0 mL/100 mL. The AIF curves of other groups were rescaled so that integrals of these AIFs were equal to AIFs of corresponding control groups (PBS groups). This correction is based on the assumption that the total amount of CAs should be the same. DCE-MRI models were selected following a bottom-up strategy. Patlak model25,26 was selected for most brain voxels, and it was linearized as follows
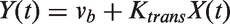
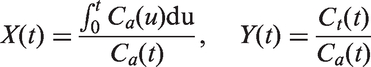
In vivo gamma ray imaging
Radioactive gamma ray imaging was performed on a BHP6602 gamma camera (HAMAMATSU, Japan). At 10 min, 30 min, 2 h or 4 h post-intravenous injection (i.v.) of PBS,
TEM studies
At selected times after injection of PBS, nanoparticle (0.5 µmol/kg) or mannitol (20 %, 10 mL/kg) via i.v., the mice were anesthetized, brains were removed and fixed in 4% paraformaldehyde (PFA) in PBS for overnight at 4℃. Brain specimens with a volume of 1.0 mm3 were excised, washed with de-ionized (D.I.) water, submitted to a second fixation with osmium tetroxide (OsO4) for 90 min and rinsed with D.I. water again. After dehydrated by a series of ethanol grades, 50%, 70% and 90%, the specimens were embedded in epoxy resin and cut into ultra-thin sections (50–60 nm). Then the sections were double stained with 3% uranyl acetate and lead citrate and viewed using a JEM 1230 TEM (JEOL, USA).
Development of brain edema model
The ICR mice were catheterized with a home-built catheter system, and 20% mannitol (10 mL/kg) was administrated intravenously. T2W-MRI was applied to measure the volume of brain edema at 24 h PI.
Brain water content measurement
Brain water contents (BWCs) before and after treatment were measured with the wet/dry mass method. Twenty male ICR mice were randomly divided into five groups (
Immunohistochemistry and confocal microscopic imaging
At 24 h PI of NA or mannitol, the mice were anesthetized and intracardially perfused with PBS followed 4% PFA. The brains were excised, fixed in 4% PFA and transferred to 30% sucrose aqueous solution for 12 h. Then the brains were embedded in optimum temperature compound, frozen and sectioned with a thickness of 10 µm. The slides were permeabilized with 0.3% Triton X-100 and blocked with a mixture of 1% BSA in PBS. After washing with PBS, the slices were incubated with rabbit anti-mouse AQP4 (1:100 dilution), active caspase-3 (1:120 dilution), Collagen IV (1:150 dilution) or GFAP (1:200 dilution) primary antibody, respectively, at 4℃ for overnight. After adequate washing, all the slices were incubated with goat anti-mouse CD34 (1:100 dilution) at 4℃ for overnight. After washing with PBS for three times, the sections were treated with CruzFluor555-labeled donkey anti-goat secondary antibody (1:100 dilution) at room temperature for 2 h. Alexa-fluo488-labeled goat anti-rabbit secondary antibody (1:500 dilution) for another 2 h followed the nucleus staining by 4′,6-diamidino-2-phenylindole (DAPI, 0.5 µg/mL). To study the AQP4 location variation after the TJ compromise, the brain sections were immuno-stained by a combination of rabbit anti-mouse AQP4 (1:100 dilution) and rat anti-mouse GFAP (1:100 dilution) primary antibodies at 4℃ for overnight. Alexa-fluo488-labeled goat anti-rabbit (1:500 dilution) and Alexa-fluo555-labeled goat anti-rat (1:300 dilution) secondary antibodies were used to label corresponding primary antibody. The immunofluorescence images were collected on a Zeiss LSM 710 META confocal laser scanning microscope (Carl Zeiss, Germany) by using a 60× oil lens. DAPI was excited with a 405-nm laser, and the emission was detected with a photomultiplier by a 420–480-nm band-pass filter. Alexa-fluo488 was excited with a 495-nm laser, and its emission was detected by a second photomultiplier using a 505–550-nm band-pass filter. Alexa-fluo555 and CruzFluor555 were excited with a 560-nm laser, and its emission was detected by a second photomultiplier using a 570–610-nm band-pass filter. The fluorescence images were processed by ZEN 2012 software. The fluorescence intensities of the images were quantified by ImageJ software (NIH). All data are mean ± standard deviations (SD) (
TUNEL staining
At 24 h after NA or mannitol treatment, mouse brain sections (10 µm) were pre-treated with 3% citric acid to minimize the non-specific staining. After fixation in 4% PFA for 30 min, the slides were permeabilized and incubated with DNase I for 30 min at 37℃. Then 70% (v/v) reaction buffer and 30% TdT enzyme were mixed, and the slides were incubated with above mixture at 37℃ for 1.0 h. Finally, the slides were treated with the newly prepared 10% Alexa-fluo488-labeled streptavidin and 90% labeling buffer at 37℃ for 30 min. After mounted and covered with a thin glass coverslip, the fluorescence images were collected on a confocal fluorescence microscope.
Histology study
At 24 h PI of NA or mannitol, the animals were anesthetized and intracardially perfused with PBS followed 4% PFA. The mouse brains were excised, fixed, dehydrated and then embedded in paraffin. Each section was cut with a thickness of 5.0 µm, stained with hematoxylin and eosin (H&E), and then visualized under optical microscope (Leica DMI 4000B, Germany).
Statistical analysis
The experimental data were statistically analyzed by using the Mann-Whitney U-test, and statistically significant difference was reported if
Results
Synthesis and characterization
NA Characterization of the nanoagonist. Chemical structure (a), TEM (b), 1H NMR spectrum (c), hydrodynamic size distribution (d) and zeta potential (e) of 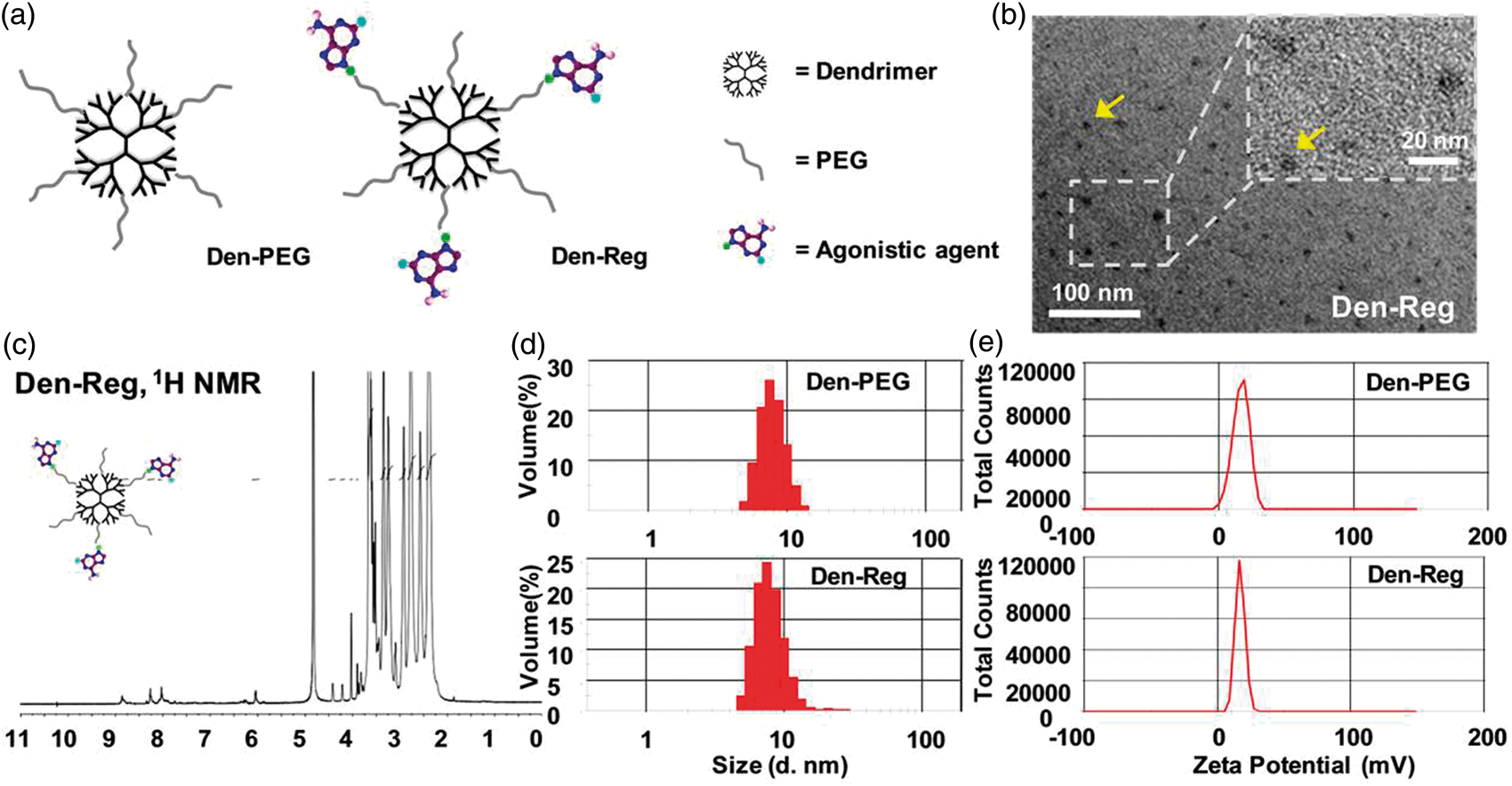
NA up-regulating BBB permeability in normal mice
The BBB permeability alternations before and after injection of NA via i.v. were non-invasively evaluated by DCE-MRI in normal ICR mice with intact BBB. Figure 3(a) demonstrated pixel maps of NA-mediated BBB permeability enhancement. (a) Representative 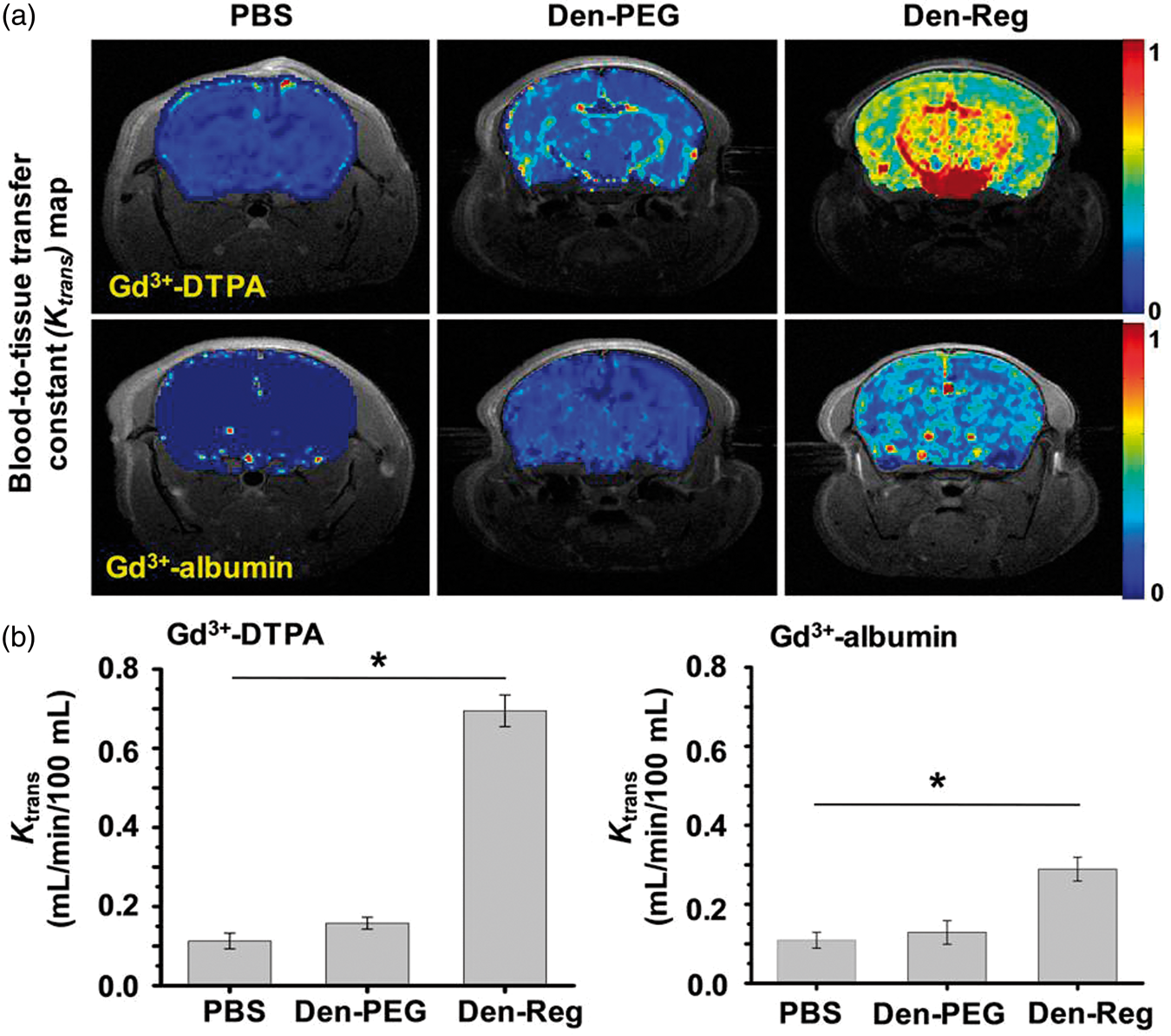
NA increasing brain uptake of CAs in normal mice
Brain uptake of CA was indicated as the hyper-intensive signal in the T1W-MRI (Figure 4(a)). ΔT1-map presenting the T1 values variation pixels by pixels before and after the combined treatment of NA/CA was also generated to quantify the CA uptake efficiencies (Figure 4(b)). Besides the parenchyma area such as striatum, ventral pallidum and thalamus, remarkable T1 value change was also observed in lateral ventricle and the neighboring choroid plexus as well as superior sagittal/cavernous sinus after the treatment of NA up-regulated brain uptake of both small and macromolecular CAs. (a) In vivo T1W-MRI images of normal mouse brain when Gd3+-DTPA (left panel) or Gd3+-albumin (right panel) was injected before and at 30 min post-administration of 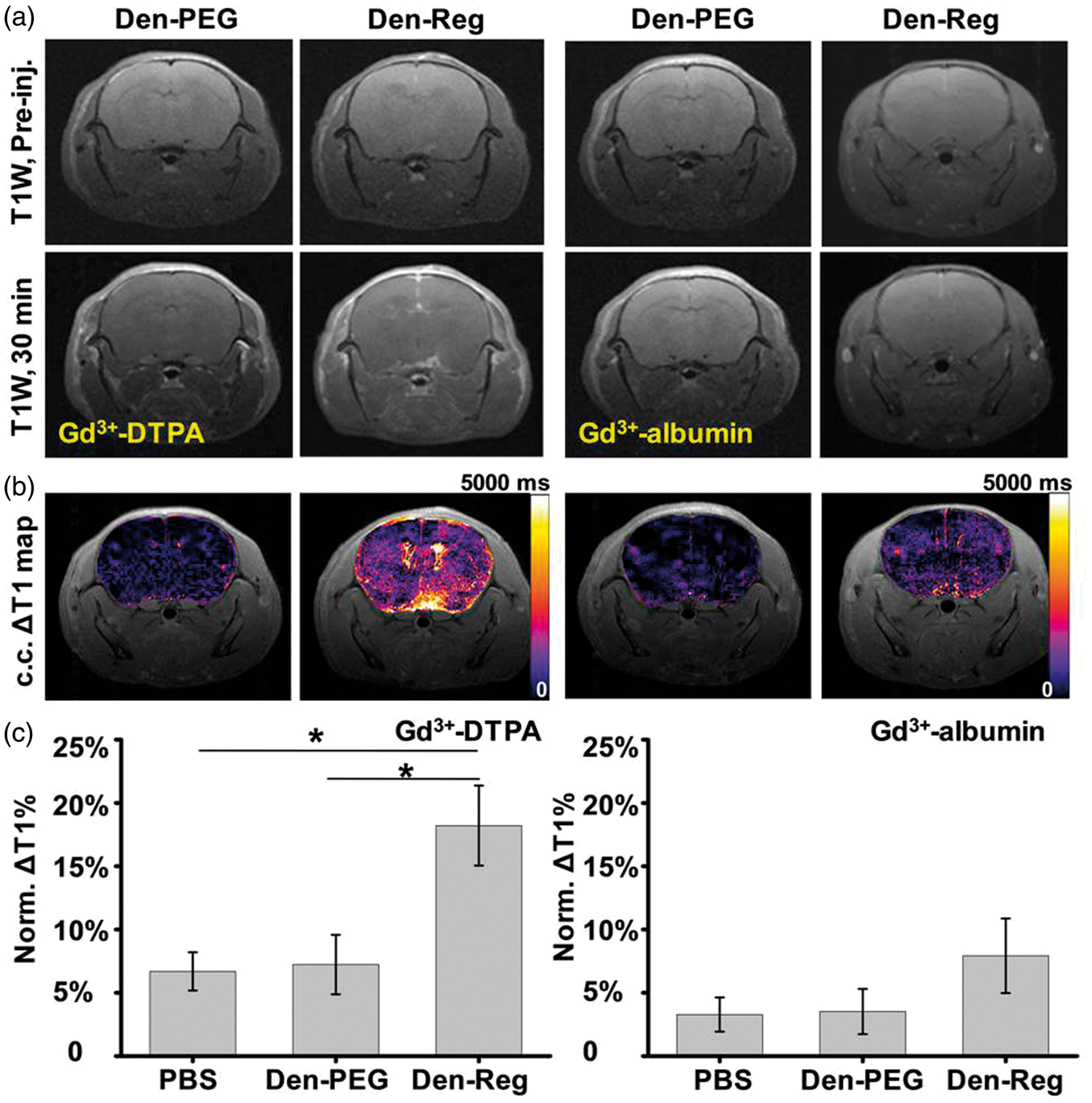
Spontaneous BBB restoration after NA treatment
Timely restoration of BBB functionality is crucial to minimize the side-effects caused by BBB leakage. In vivo gamma ray imaging showed that while the intracerebral radioactivity of 99mTc-albumin was evident when it was injected at 10 min, 30 min and 2 h PI of Spontaneous BBB restoration after NA treatment. (a) Representative in vivo gamma ray images of normal mice when radioactive model drug 99mTc-albumin (3.7 × 107 Bq/mouse) was injected at selected times PI of PBS, 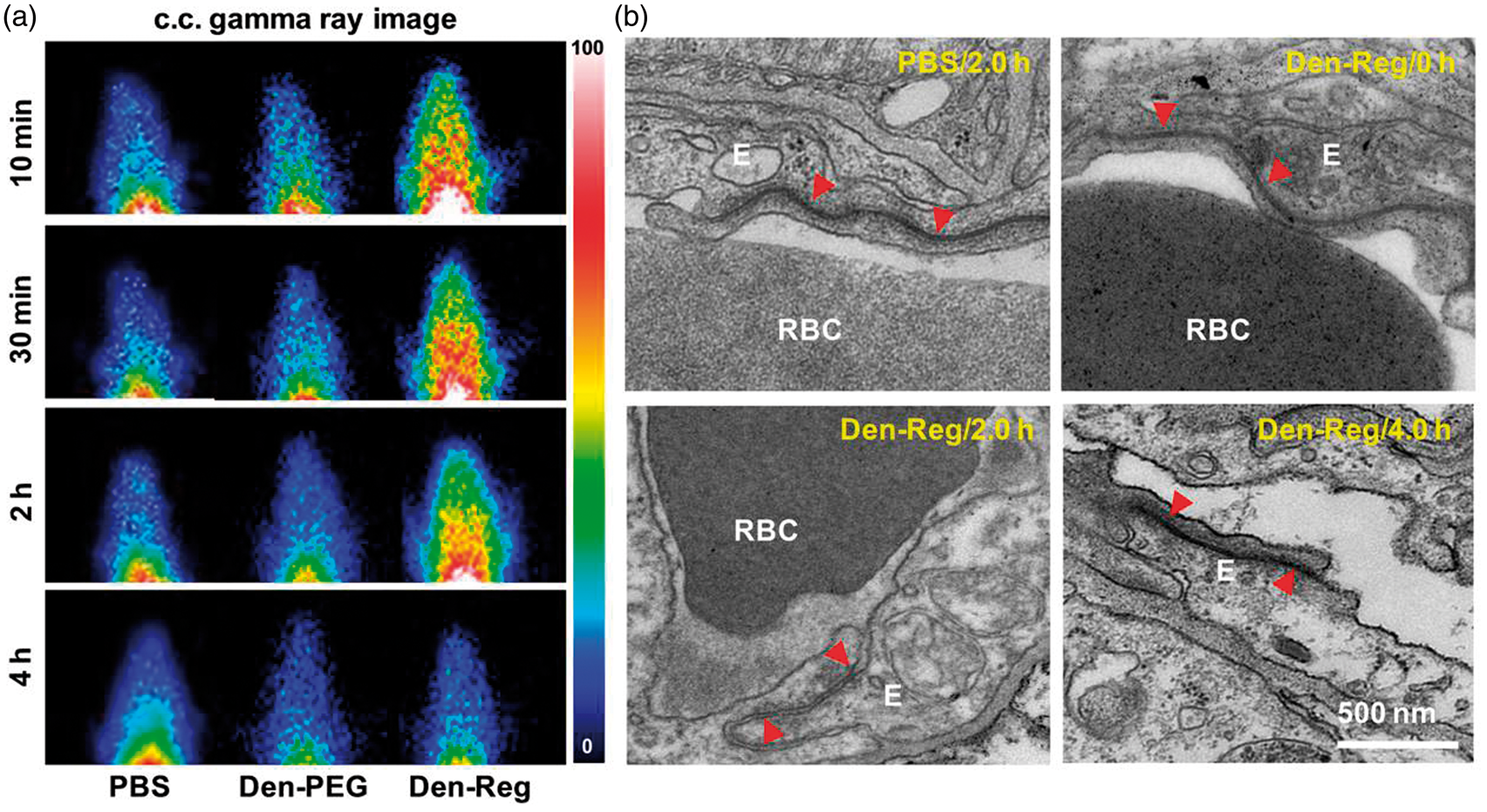
NA opening TJs without inducing edema and apoptosis
The excess influx of plasmic fluid from the leaky TJs will induce edema which is a common pathological symptom in brain disorders including traumatic brain injury, stroke, brain tumors and infections.
14
To evaluate the safety of the NA-mediated TJ opening, healthy mice were randomly divided into four groups and injected with PBS, Minimized brain edema after the NA-mediated TJ opening. (a) In vivo T2W-MRI images of normal mouse brain at 24 h PI of PBS, 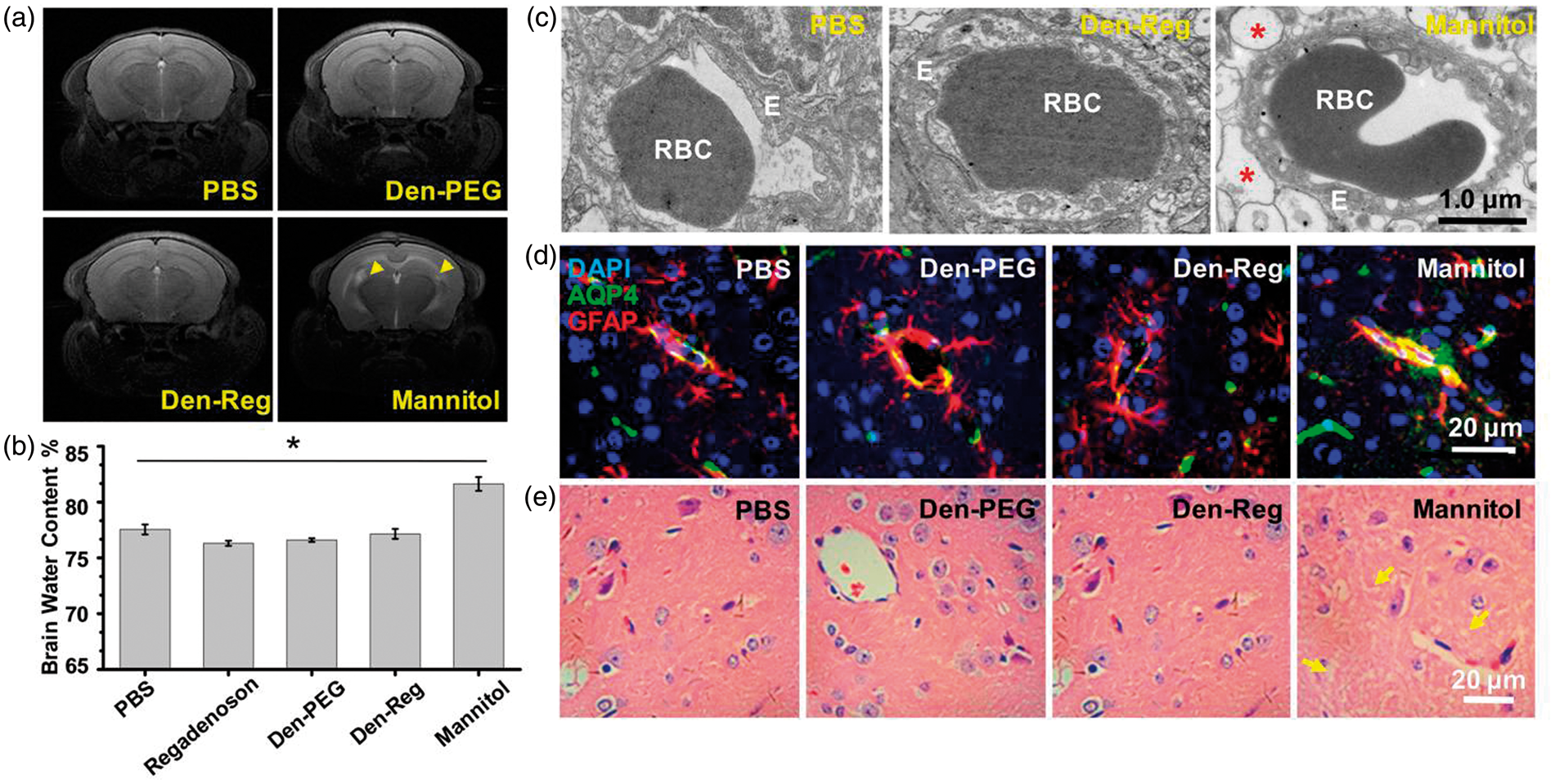
TEM images showed large pieces of vacuolated space surrounding the vessels in brain striatum at 24 h PI of mannitol (Figure 6(c)). However, the perivascular area was compact with cellular structures, and no swollen astrocyte endfeet of neuropil was shown after treatment of
The extravasation of erythrocytes after BBB breakdown was found in pathogenic states such as hyperthermia
33
and traumatic injuries.
34
The degradation of the erythrocytes in the brain results in the increase of ferrous iron concentration
35
and oxidative pressure,
36
which inevitably trigger apoptosis initiated neurologic deterioration. Caspase-3 as an apoptotic executioner is activated in both extrinsic and intrinsic apoptotic pathways.
37
Therefore, it is widely used as a biomarker to identify the apoptotic cells. As shown in Figure 7(a), while the active caspase-3 displayed as tiny vesicles located sporadically in brain striatum at 24 h PI of PBS, Minimized apoptosis and uncompromised NVU components after NA treatment. Active caspase-3 immunofluorescence images (a) and TUNEL staining images (b) of brain striatum at 24 h PI of PBS, 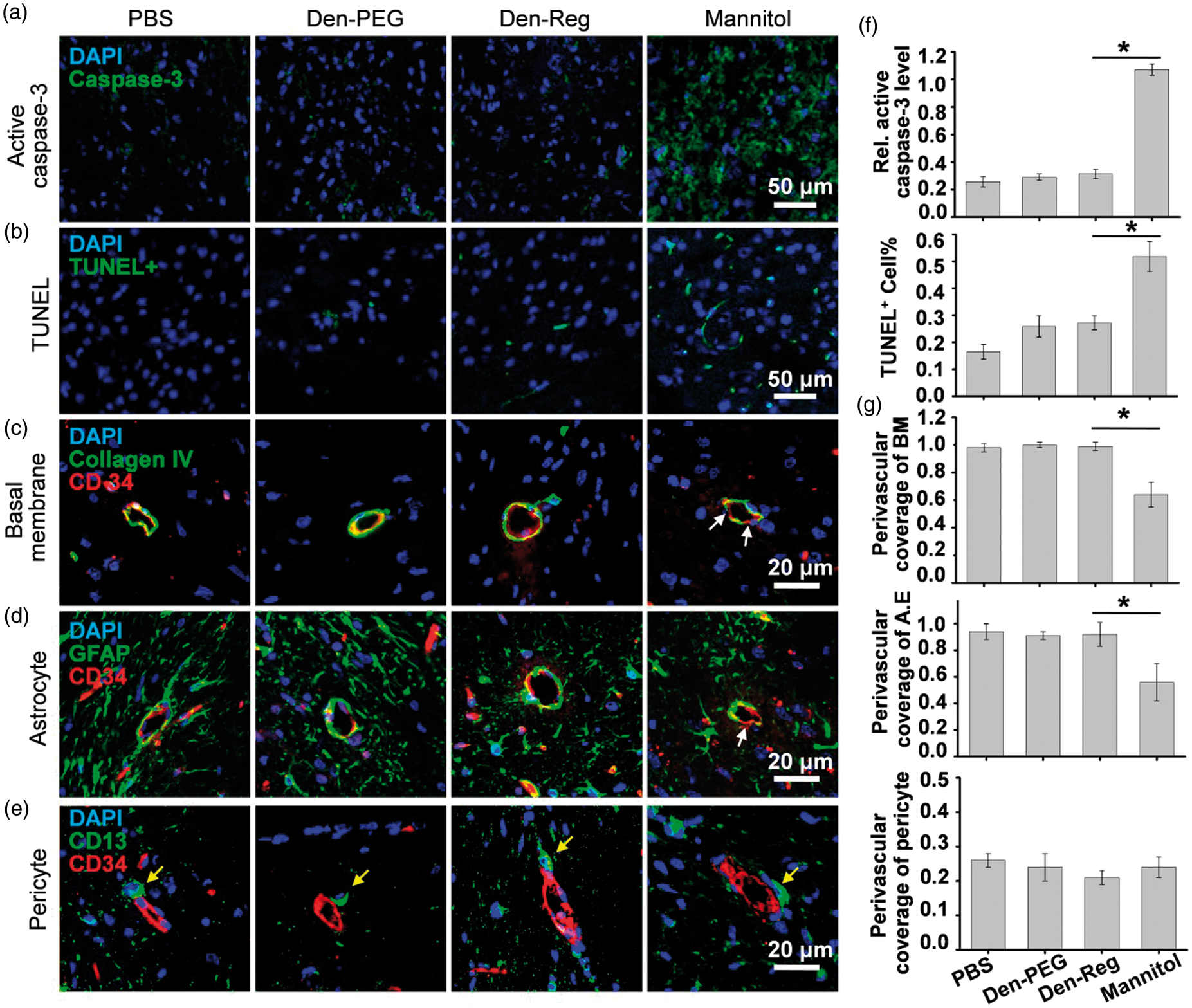
Discussion
Gaul and coworkers
38
first demonstrated that the signaling orphan GPCR, Moody, could temporarily increase
The extent of BBB disruption was traditionally studied by ex vivo Evan’s blue staining. However, this technology cannot dynamically evaluate BBB permeability in vivo. Vascular permeability induced by GPCR signaling was previously measured by T1W MRI.
39
However, this technology suffered low temporal resolution (≈3 min per scanning), low sensitivity and the requirement of multiple CA injections.
42
As a MR technology depicting anatomic structure as well as physiological dynamics, DCE-MRI determines vascular permeability alternations by tracking the pharmacokinetics of the administrated paramagnetic CA that diffuses between the blood and the extracellular extravascular space.
43
Compared to the T1W-MRI, DCE-MRI not only offers more vascular parameters such as blood-to-tissue transfer constant (
Temporarily opening paracellular TJs is a promising way to enhance brain delivery of drugs without compromising their pharmacological activities. However, manipulating brain delivery of drugs but not the blood-borne compartments is the key to reduce the side-effects caused by TJ disruption. NVU is a dynamic structure that comprises the co-coordinated BCECs, BM, astrocytes, pericytes and neurons. 48 A main function of NVU is to maintain CNS homeostasis by balancing the influx of nutrients and the efflux of wastes. As an important component of the NVU, BM is made up of type IV collagen, fibronectin, heparan sulfate and laminin, which enwrap the abluminal surface of endothelial cells as a thin layer. 49 Similar to a charge and molecular weight sieve, BM prevents the paracellular perfusion of excess protein-rich fluids and cellular blood elements into brain parenchyma. 50 BM injury leads to BBB leakage during pathogenic states such as brain ischemia 51 and hemorrhage. 52 Besides the BM, astrocytes also actively maintain the BBB characteristics. First, astrocyte endfeet encircle nearly 100% cerebral vessels and strictly control water transport between brain and blood by polarizing AQP4 distribution in the side facing the BCECs. 31 Second, astrocytes release nutrients and growth factors that not only keep BCEC active 53 but also help to restore TJ integrity timely after the its disruption. In contrast to the detachments of astrocyte endfeet and BM from the vessels after the mannitol treatment, BM and astrocytes kept intact at 24 h post-treatment of NA. As vital coordinators of the NVUs, pericytes54,55 actively regulate BBB functionalities such as mediating TJ associated protein expression levels in endothelial cells and aiding the polarization of astrocyte endfeet. 56 Considering that the degeneration of pericytes is occurred in multiple CNS diseases, 57 the barely changed location and perivascular percentage of the pericytes (Figure 7(e)) implied that the NA did not disturb the normal function of the pericytes. Previous work found that activation of A2AR on pericytes could trigger the opening of ATP-sensitive potassium (KATP) channels and induce hyperpolarizing current that acts as a vasoactive signal. 58 Even though the neurovascular effects of signaling A2AR on the pericytes were not clear yet, it may also change BBB permeability considering its key role in mediating the contractile properties of the vasculatures. Overall, the unspoiled structure and interaction between the NVU components may accelerate the TJ recovery and minimize adverse-effects after NA-mediated A2AR signaling.
Even though NA showed the promise to tune BBB permeability safely and efficiently, several issues should be addressed before its potential application. (1) Specifically tuning BBB permeability in the diseased tissue. Considering the heterogeneous expression of A2AR in the brain, 46 up-regulating BBB permeability exclusively in the diseased areas but not the normal brain tissues can reduce the side-effects induced by the non-specific vascular leakage. In our previous work, a two-order targeted nanoprobe was developed to visualize brain tumor with high signal to background ratio. 59 This nanoprobe first targeted to tumor neovasculatures, then traversed BBB via LRP-mediated RMT. Inspired by this study, it is possible to develop a NA modified both targeting ligand and agonistic agent to specifically open BBB in the lesion area. 60 By this way, adverse-effects induced by the non-specific BBB leakage could be minimized. (2) Improving the biocompatibility of the NA. Considering the globular architecture, uniform particle size and well-defined reactive groups, PAMAM dendrimer was chosen as a scaffold for NA development. However, due to the potential side-effects induced by its positive surface charge induced non-specific accumulation in kidney, nanocarriers with improved biocompatibility such as micelles will be developed to accelerate the translation of NAs. (3) Fine tuning the BBB opening time-window. The agonistic effect of the NA depends on its GPCR binding affinity as well as its circulation lifetime. Due to the flexibility to change the circulation lifetime as well as the number of the agonistic agent labeled on the NA, it is possible to maximize the brain drug uptake but minimize uncontrollable BBB leakage by adjusting the BBB opening time-window according to the pharmacokinetic behavior of the therapeutics.
Conclusion
In this work, we demonstrated that NA could up-regulate brain drug uptake by signaling A2AR, a type of GPCR expressed on the BCECs. This NA temporarily opens endothelial TJs but barely disrupts other NVU components that maintain the BBB characteristics. The unspoiled BM, pericyte and astrocyte endfeet embracing the vascular endothelium prevent the extravasation of the blood-borne components through the compromised TJs, and hence minimize the pathological symptoms that are often observed after mannitol-mediated TJ compromising. Considering the BBB impermeability is a universal challenge in the treatment of CNS diseases, the NA-mediated TJ opening provides a promising strategy to increase brain drug delivery with improved efficiency but reduced side-effects.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Basic Research Program of China (973 Program, 2013CB733801, 2013CB932500), the National Natural Science Foundation of China (Nos. 81371624, 81571741, 81301875), New Century Excellent Talents in University Award and the Shanghai Foundation for Development of Science and Technology (Nos. 13NM1400400, 15140901300) and the Opening Foundation of State Key Laboratory of Magnetic Resonance and Atomic and Molecular Physics (Nos. T151108).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
XG synthesized and characterized the NA, collected most of in vivo and ex vivo imaging data. YCW processed the MR images; YL developed a method to analyze DCE-MRI data; QY was in charge of immunofluorescence staining; MK and ZL took part of synthetic work and prepared figure pictures; SZ contributed to data analysis and statistics. CL was in charge of experimental design, data interpretation and manuscript preparation. All authors have made substantive contributions to this study, and all have reviewed the final paper before its submission. XG and YCW contributed equally to this work.
