Abstract
Hypoxic stress is a common occurrence during human pregnancy, yet little is known about its effects on the fetal brain. This study examined the fetal hemodynamic responses to chronic hypoxia in an experimental mouse model of chronic maternal hypoxia (11% O2 from E14.5 to E17.5). Using high-frequency Doppler ultrasound, we found fetal cerebral and ductus venosus blood flow were both elevated by 69% and pulmonary blood flow was decreased by 62% in the fetuses exposed to chronic hypoxia compared to controls. This demonstrates that brain sparing persists during chronic fetal hypoxia and is mediated by “streaming,” where highly oxygenated blood preferentially flows through the ductus venosus towards the cerebral circulation, bypassing the liver and the lungs. Consistent with these changes in blood flow, the fetal brain volume measured by MRI is preserved, while the liver and lung volumes decreased compared to controls. However, hypoxia exposed fetuses were rendered vulnerable to an acute hypoxic challenge (8% O2 for 3 min), demonstrating global blood flow decreases consistent with imminent fetal demise rather than elevated cerebral blood flow. Despite this vulnerability, there were no differences in adult brain morphology in the mice exposed to chronic maternal hypoxia compared to controls.
Introduction
Pregnancy complication may cause acute or chronic hypoxic stress to the fetus. Short-term episodes of fetal hypoxia occur during compression of the umbilical cord 1 and during labour and delivery, 2 while chronic hypoxia is associated with a number of conditions including high-altitude pregnancy, 3 maternal smoking, 4 maternal respiratory disease, 5 maternal anemia 6 and pre-eclampsia. 7 Chronic fetal hypoxia is known to cause fetal growth restriction and is associated with impaired neurodevelopment8,9 and increased risk of disease later in life. 10
The response of the fetus to acute hypoxia has been well characterized 11 and is associated with brain sparing, a mechanism by which more oxygenated blood is directed to the brain and heart at the expense of organs such as the liver and the lungs.12–14 Recently, this physiologic response to acute hypoxia has been demonstrated by our group in fetal mice using blood oxygen level-dependent (BOLD) magnetic resonance imaging (MRI) and Doppler ultrasound. 15 Varying the maternal inspired gas mixture between 100% and 8% oxygen, we observed the mean cerebral blood velocity to rise by 15 ± 8% under acute hypoxic conditions.
In contrast, the fetal response to chronic hypoxia is not well understood. A recent study using a hypoxia chamber and instrumented pregnant sheep demonstrated that the fetal brain sparing response persists and is sustained during chronic fetal hypoxia 16 and motivates an investigation of the hemodynamic changes induced by hypoxia. While sheep have been used extensively in pregnancy research, 17 the mouse offers several advantages for modelling and studying complications of human pregnancy. Mice reproduce many of the features of human pregnancy,18,19 including a hemochorial placenta with similar vascular and cellular structure. Moreover, a variety of advanced imaging techniques have been developed for monitoring pregnancy in the mouse. We have recently reported a methodology for evaluating blood flow distribution in the mouse fetus with high-frequency ultrasound. 20 Together with MRI, these technologies enable a detailed analysis of the fetal hemodynamic changes induced by chronic hypoxia.
To date, studies using mouse models of chronic maternal hypoxia (for review: Jang et al. 21 ) have reported fetal growth restriction,22–26 effects on pulmonary development,23,27 altered heart growth and abnormal heart morphology22,28 reduced placental perfusion 24 and altered placental morphology.26,29 Using micro-computed tomography and scanning electron microscopy, we previously found that the murine placenta adapts to chronic maternal hypoxia by capillary expansion, thinning of the interhaemal membrane and increased utero-placental blood flow. 30 In the present study, Doppler ultrasound is used to characterize the fetal hemodynamic responses to chronic maternal hypoxia during late gestation in pregnant mice. We studied the effects of chronic hypoxia on fetal cerebral blood flow, right ventricular outflow, and the pulmonary circulation. An acute hypoxic condition (8% O2 for 3 min, termed acute-after-chronic) was used to challenge the fetus and assess its capacity to compensate for additional hypoxia stress. Fetal organ growth and brain morphology in a survived cohort of pups were assessed using ex vivo MRI.
Methods
Animals
Fifty-one healthy adult CD-1 virgin female mice (six-to-nine weeks of age) from Charles River Laboratories (St Constant, QC, Canada) were used and mated in-house. Facility health reports indicated that the mice were free of viral and parasitic pathogens. The morning that a vaginal copulation plug was detected was designated as Embryonic Day 0.5 (E0.5). Full term for CD-1 dams is 18.5 days. A sample size calculation was performed based on the variation in control E17.5 CD-1 blood flow data from Zhou et al. 20 With a sample size of 12 mice per group, we are powered at 0.8 to detect changes in blood flow on the order of 20% using ultrasound. One cohort of dams (13 chronic hypoxia and 13 control) was imaged using ultrasound biomicroscopy at E17.5. Pups from a second cohort of dams (thirteen chronic hypoxia and twelve control) were survived, weighed weekly and then sacrificed for ex vivo MRI at two developmental time points: either at weaning (post-natal day 23 (P23)) or young adulthood (P59). Mice were co-housed in a standard cage, with ad libitum access to food and water, in a pathogen-free environment on a 12-h light:dark cycle. All of the experimental procedures were performed during the light phase. All animal experiments were approved by the Animal Care Committee at The Centre for Phenogenomics, conducted in accordance with guidelines established by the Canadian Council on Animal Care and complied with the ARRIVE guidelines.
Chronic hypoxia protocol
At E14.5, mice were randomly assigned to either the chronic hypoxia or control group. The chronic hypoxia group remained in a standard cage and were placed in a sealed chamber (medium A-chamber, 30 inch W × 20 inch D × 20 inch H, Biospherix, NY, USA) with computer control to maintain a 11% O2 (balance N2) environment. Soda lime was placed in the bottom of the chamber to scavenge excess CO2.
Ultrasound biomicroscopy
A high-frequency ultrasound system (Vevo 2100 and 40 MHz transducer; VisualSonics, Toronto, Ontario, Canada) was used to make fetal blood flow measurements in dams at E17.5, as described in detail previously.15,20 The dams were removed from the hypoxia chamber and anesthetized with isoflurane (4% for induction and 1.5% for maintenance in 100% O2), endotracheally intubated (22 gauge catheter) and mechanically ventilated (135 cycles per minute) using a pressure-controlled ventilator (TOPO Small Animal Ventilator; Kent Scientific, Torrington, CT, USA). To verify normocapnic conditions throughout the experiment, the ventilation parameters have been previously validated using a transcutaneous blood gas analyzer (TCM4, Radiometer Canada, London, ON, Canada) to measure maternal partial pressure of carbon dioxide. 15 The maternal temperature was maintained at 35–37℃ and maternal heart rate and temperature were monitored throughout the experiment. This experimental design allows the study of differences in fetal hemodynamics resulting from angiogenesis and vascular remodeling that occur over days and are insensitive to the mice being removed from the hypoxia chamber for imaging while breathing 100% O2. In each dam, one fetus with the most favourable spatial orientation for visualization in the lower abdomen was chosen for imaging.
For velocity measurements, the pulsed wave Doppler sample volume was adjusted to cover the entire vessel lumen and the angle of insonation was kept as small as possible (<60°). The diameter measurements were determined using either B-mode or M-mode recordings with the ultrasound beam perpendicular to the vessel of interest, at the same location as the Doppler spectrum. The intensity-weighted mean velocity of the Doppler spectrum was traced as a function of time and used to measure the velocity-time integral (VTI). The flow was calculated by multiplying the VTI by the vessel cross-sectional area and the fetal heart rate. All parameters were averaged over three cardiac cycles. The pulsatility index (PI) was calculated as the difference between peak systolic and end diastolic velocities, divided by the mean velocity over the cardiac cycle.
The fetal vessels of interest included the middle cerebral artery (MCA), main pulmonary artery (MPA), ductus arteriosus (DA), ductus venosus (DV), and the umbilical artery (UA). The diameter of the fetal MCA is too small for accurate measurement and therefore we used mean velocity as a measure of blood flow. The MPA and DA were imaged in an oblique slice showing flow between the right ventricular outflow tract and the descending thoracic aorta. The MPA diameter and flow velocity were measured just beyond the pulmonary valve and the DA measurements were taken at the distal segment of the DA. The pulmonary blood flow (PBF) was calculated from the difference between the MPA and DA flows.
To investigate the effects of acute hypoxia, after the blood flow measurements were acquired for each vessel at 100% O2, the gas mixture inhaled by the dam was changed to 8% O2 (balance N2) for a total of 3 min (acute-after-chronic hypoxia model). The gas conditions were chosen to be consistent with our previous study, 15 where this large change in oxygenation of the fetal organs clearly results in brain sparing in healthy fetal mice. Three minutes was chosen based on our previous work, 15 where we reported the onset of bradycardia at ∼2.5–3 min with an acute hypoxic challenge. Doppler recordings were taken continuously for the first 2 min and then diameter measurements were collected. To compare the changes in blood flow during the acute hypoxic challenge between mice, the time averaged mean velocity during hypoxic (νavg(hypoxia)) and hyperoxic (νavg(hyperoxia)) conditions were determined by the νavg at 1.5 min from the start of each of the gas cycles (before the onset of bradycardia) under 8% O2 and 100% O2, respectively. The change in νavg with hypoxia was calculated as: (νavg(hypoxia) – νavg(hyperoxia))/νavg(hypoxia). The duration of the procedure was ∼1 h and after the imaging session, the dam was sacrificed and the fetuses and placentas were dissected and weighed.
The intubation of one dam from the chronic hypoxia group was unsuccessful and thus we only have measurements at 100% O2 for this dam. For two dams in the chronic hypoxia group during imaging of the MPA/DA and for one dam during imaging of the DV, we were unable to locate the diameter during the acute hypoxic challenge. The UA measurements were not collected for one of the dams in the chronic hypoxia group and the UA diameter measurement was not collected for four additional dams in the chronic hypoxia group and one control.
Ex vivo MRI
To measure fetal organ growth, pregnant dams at E17.5 from the chronic hypoxia (8 fetuses from 3 litters) and control group (9 fetuses from 3 litters) were sacrificed by cervical dislocation. The fetuses were harvested and fixed in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS) and 2 mM ProHance at 4℃ for three days. To detect changes in neuroanatomy associated with chronic maternal hypoxia, mice at P23 (chronic hypoxia: 23 mice (12 females and 11 males) from 8 litters, control: 21 mice (11 females and 10 males) from 7 litters) or P59 (chronic hypoxia: 21 mice (11 females and 10 males) from 5 litters, control: 27 mice (14 females and 13 males) from 5 litters) underwent a transcardiac perfusion. 31 Mice were anesthetized with ketamine/xylazine and perfused through the left ventricle with 30 mL PBS, 1 µL/mL heparin and 2mM ProHance, followed by 30 mL of 4% PFA and 2 mM ProHance in PBS. The brains within the skull were incubated in 4% PFA containing 2 mM ProHance overnight at 4℃. Both the fetal and brain samples were stored in a solution containing PBS, 2 mM ProHance and sodium azide.
A multi-channel 7.0 T magnet (Varian Inc. Palo Alto, CA, USA) and a custom-built solenoid array were used to image 16 samples in parallel. 32 An anatomical scan was performed using a T2-weighted, 3D fast spin-echo sequence using a cylindrical k-space acquisition 33 with TR = 350 ms, TE = 12 ms, echo train length = 6, two averages, field-of-view 20 mm × 20 mm × 25 mm, matrix size = 504 × 504 × 630, isotropic image resolution = 40 µm.
Volume analysis
The brain, lungs and liver were manually segmented in 3D on each of the fetal MR images. For the adult brain MR images, an automated image registration approach implemented in the Pydpiper toolkit 34 and using the advanced normalization tools (ANTs) deformation algorithm 35 was used. The images were iteratively linearly and non-linearly registered together to create an average image of all the scans. The Jacobian determinants provided an estimate of the local volume changes at every voxel in the brain for comparison between the chronic hypoxia and control groups. 36
Statistical analysis
All statistical tests were conducted using the R statistical software (www.r-project.org). The fetal and placental weight data were analyzed using a mixed effects model with group (control, chronic hypoxia) as a fixed effect and litter as a random effect. Mean velocity, blood flow, PI and fetal organ volume measurements are presented as box-and-whisker plots. The cerebroplacental ratio (CPR) was calculated as the MCA PI/UA PI. All data were tested for homoscedasticity (Levene’s test) and are reported as means with 95% parametric confidence intervals (CIs). A value of p < 0.05 was taken to be significant. To determine if chronic hypoxia had an effect on fetal blood flow, a linear mixed effect model was used to analyze the variations in the flow with group (control, chronic hypoxia) and vessel (MCA, MPA, DA, DV, PBF, UA) as fixed effects and allowing for interactions between the two. Mouse ID was treated as a random factor to account for variation between mice. PI and fetal organ volumes were analyzed using t-tests to compare chronic hypoxia-exposed and control fetuses. The body weight growth curves for each mouse of the survived cohort were fit with a natural spline with five degrees of freedom. A linear mixed effect model was used to determine whether there was an effect of group (control, chronic hypoxia) or sex (male, female) on the spline parameters. For the MR volumetric data, a linear model was computed at every voxel relating the relative Jacobian determinant to the fixed effects of group, sex and group-by-sex interaction. Multiple comparisons were controlled for using the false discovery rate (FDR) 37 and statistical significance was defined at an FDR threshold of 10%. No corresponding multiple comparisons correction was made for the blood flow, PI and fetal organ volume measurements (the p values reported are uncorrected).
Results
Following maternal exposure to hypoxia during late gestation (E14.5–E17.5), the dams (N = 13) weighed significantly less when compared to controls (N = 13) (47 (CI: 43–51) vs. 53 (CI: 50–56) g, p = 0.004). In addition, the fetuses exposed to chronic maternal hypoxia (n = 152) weighed 15% less than controls (n = 140) (0.96 (CI: 0.93–0.99) vs. 1.13 (CI: 1.11–1.15) g, p < 0.0001), indicating that the chronic hypoxia fetuses failed to reach their growth potential. In contrast, placental weights remained similar between the chronic hypoxia and control groups (0.12 (CI: 0.11-0.13) vs. 0.121 (CI: 0.117-0.125) g) and there was no difference in litter size (12 (CI: 11–13) fetuses/litter) or number of fetal resorptions (0.08 (CI: 0.04–0.12) resorptions/total implantation sites per mouse). The fetal–placental weight ratio 38 was significantly decreased in the chronic hypoxia group compared to controls (8.4 (CI: 8.1–8.7) vs. 9.7 (CI: 9.3–10.1), p < 0.0001). This decrease is consistent with fetal growth restriction, where the placenta may fail to adapt its capacity to provide oxygen and nutrients to compensate for the smaller fetal size. 39 The chronic hypoxia fetuses had elevated heart rates compared to controls (298 (CI: 275–321) vs. 238 (CI: 208–268) beats per minute, p = 0.002).
To further investigate the pathophysiology of growth restriction following chronic maternal hypoxia in late gestation, the fetal hemodynamic responses were evaluated using high-frequency ultrasound. Mimicking the study of placental dysfunction in human growth restricted fetuses, 40 we looked for evidence of the following: (i) brain sparing, (ii) DV mediated flow redistribution, (iii) decreased CPR and (iv) absent or reversed end-diastolic blood velocity (AREDV) UA waveforms.
Maternal exposure to chronic hypoxia resulted in brain sparing and preferential streaming of umbilical venous blood via the DV
To investigate our primary hypothesis that exposure to chronic maternal hypoxia would result in fetal blood flow redistribution, we measured fetal cerebral blood flow (determined using MCA blood velocity waveforms), DV blood flow, PBF and UA blood flow using high-frequency ultrasound in chronic hypoxia (n = 13) and control (n = 13) fetuses. The DV provides a shunt for highly oxygenated blood to bypass the liver and preferentially cross the foramen ovale into the left atrium and ventricle, towards the cerebral circulation.41–44 As can be seen in Figure 1, chronic hypoxia resulted in significant changes in basal blood flow (p = 0.0003). There was a significant group-by-vessel interaction and therefore we performed post hoc t-tests to investigate the effect of hypoxia on blood flow for each vessel. Fetal cerebral blood flow was found to be elevated by 69% in the fetuses exposed to chronic maternal hypoxia compared to controls (18.6 (CI: 14.7–22.5) vs. 14.0 (CI: 12.0–16.0) mm/s, p = 0.03). The blood flow through the DV was 69% higher in the fetuses exposed to chronic maternal hypoxia compared to controls (0.16 (CI: 0.11–0.21) vs. 0.10 (CI: 0.07–0.13) mL/min, p = 0.01). The diameter of the DV was 15% larger in the chronic hypoxia fetuses compared to controls (0.25 (CI: 0.24–0.26) vs. 0.22 (CI: 0.20–0.24) mm, p = 0.008). In addition, there was a positive correlation between mean MCA velocity and DV flow (p = 0.0004).
(a) Mean velocity and (b) blood flow in control (grey) and chronic hypoxia (blue) fetuses measured using high-frequency ultrasound. The fetuses exposed to chronic maternal hypoxia showed flow redistribution: increased MCA velocity, increased DA and DV blood flow and decreased pulmonary blood flow (PBF). The box-and-whisker plots show medians and quartiles. The pulmonary blood flow was calculated from the measurements of the MPA and DA flows. n = 13 controls and n = 13 chronic hypoxia fetuses except for the UA where n = 12 controls and n = 8 chronic hypoxia. *p < 0.05, **p < 0.005. DA: ductus arteriosus; DV: ductus venosus; MCA: middle cerebral artery; MPA: main pulmonary artery; PBF: pulmonary blood flow; UA: umbilical artery.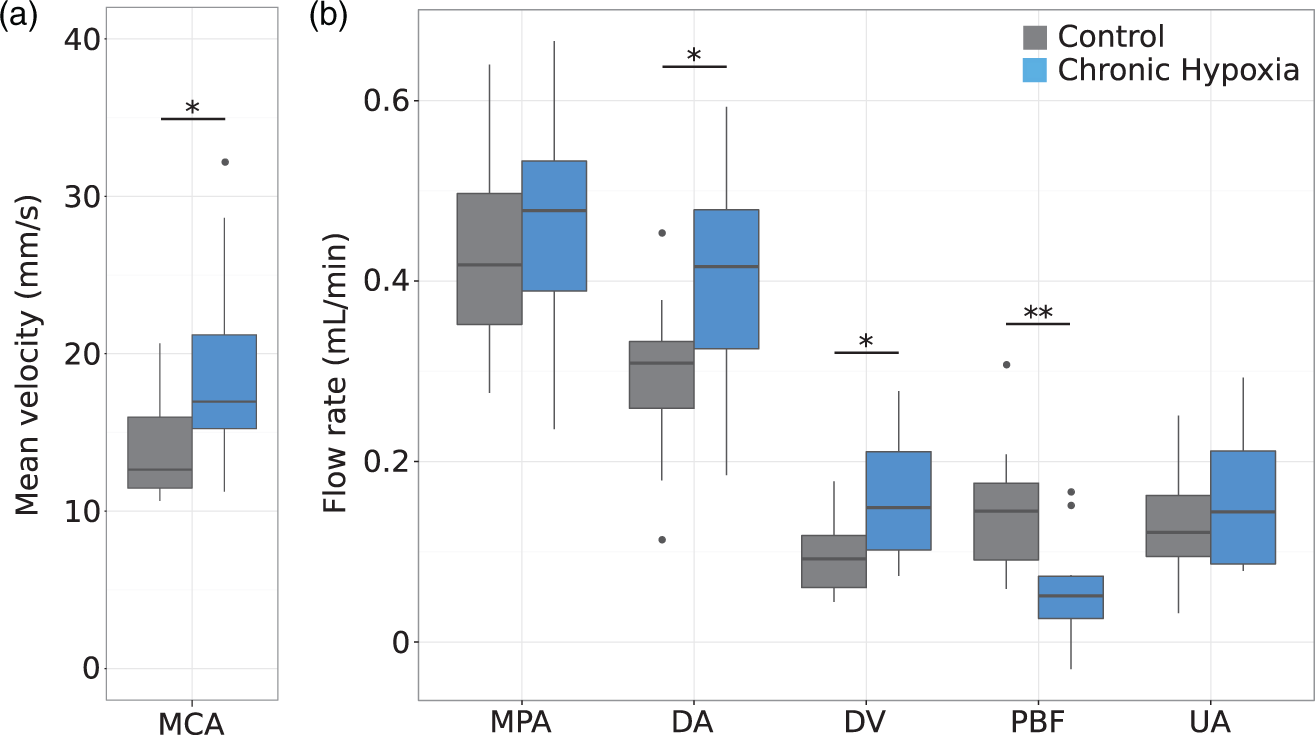
Right ventricular outflow and flow to the fetal pulmonary circulation were determined by measuring flow through the MPA and the DA. MPA blood flow remained similar between the two groups, while the DA flow was elevated in the fetuses exposed to chronic maternal hypoxia compared to controls (0.41 (CI: 0.33–0.49) vs. 0.29 (CI: 0.24–0.34) mL/min, p = 0.01). There was no difference in the diameter of the MPA and a trend towards an increased DA diameter in the chronic hypoxia fetuses compared to controls (0.50 (CI: 0.47–0.53) vs. 0.47 (CI: 0.44–0.50) mm, p = 0.1). This resulted in a significant decrease of 62% in PBF in the chronic hypoxia fetuses compared to controls (0.05 (CI: 0.02–0.08) vs. 0.14 (CI: 0.10–0.18) mL/min, p = 0.002).
To determine overall fetal well-being, UA blood flow was measured. There was no difference in UA blood flow between the chronic hypoxia fetuses (n = 8) and controls (n = 12).
Signs of accelerating placental dysfunction in fetuses exposed to chronic maternal hypoxia
Representative Doppler waveforms are shown in Figure 2. Qualitatively, the fetuses exposed to chronic maternal hypoxia showed evidence of reverse DV flow (Figure 2(b)) and AREDV waveforms in the UA (Figure 2(d)). Reverse DV flow was evident in 10 of 13 fetuses exposed to chronic maternal hypoxia and in 4 of 13 of the controls, while AREDV was present in 5 of 12 chronic hypoxia fetuses compared to 2 of 13 controls.
Representative DV and UA Doppler flow waveforms for (a,c) control and (b,d) chronic hypoxia fetuses. The blood flow through the DV is significantly higher in the fetuses exposed to chronic maternal hypoxia compared to controls. Note the presence of reverse DV flow and AREDV in the UA of the chronic hypoxia fetuses.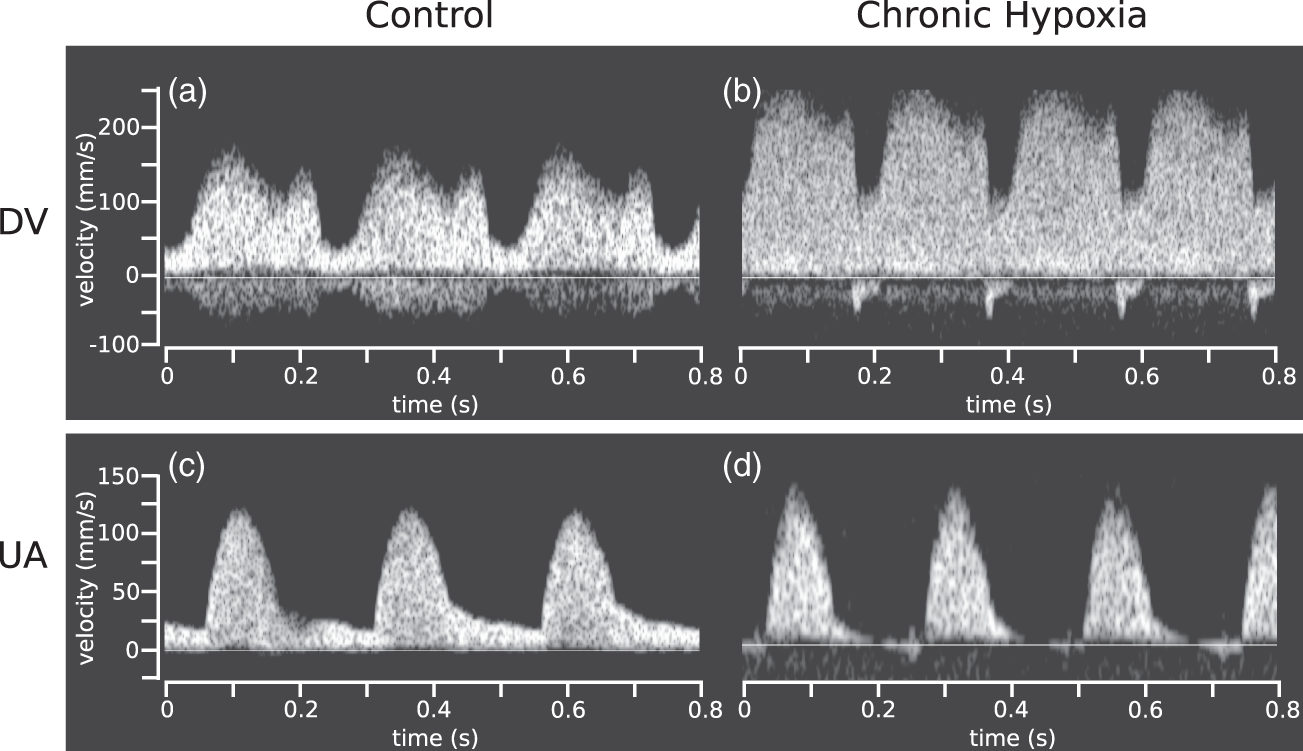
The fetuses exposed to chronic maternal hypoxia had an elevated PI in the DV (0.8 (CI: 0.6–1.0) vs. 0.4 (CI: 0.3–0.5), p = 0.003) and a decreased pulsatility in the MCA (1.1 (CI: 1.0–1.2) vs. 1.3 (CI: 1.2–1.4), p = 0.02) compared to controls; however, there was no difference in PI between groups for the UA (Figure 3). There was a trend towards a decreased CPR in the chronic hypoxia fetuses compared to controls (0.58 (CI: 0.52–0.64) vs. 0.67 (CI: 0.60–0.74), p = 0.06).
Doppler indices in control (grey) and chronic hypoxia (blue) fetuses. In the fetuses exposed to chronic maternal hypoxia, (a) the DV pulsatility index was elevated by 98% (p = 0.003), (b) MCA pulsatility index was decreased by 13% (p = 0.02), (c) the UA pulsatility index was similar and (d) the cerebroplacental ratio was decreased by 13% (p = 0.06). The box-and-whisker plots show medians and quartiles. n = 13 controls and n = 13 chronic hypoxia fetuses for DV and MCA; n = 13 controls and n = 12 chronic hypoxia for UA and CPR. *p < 0.05, **p < 0.005.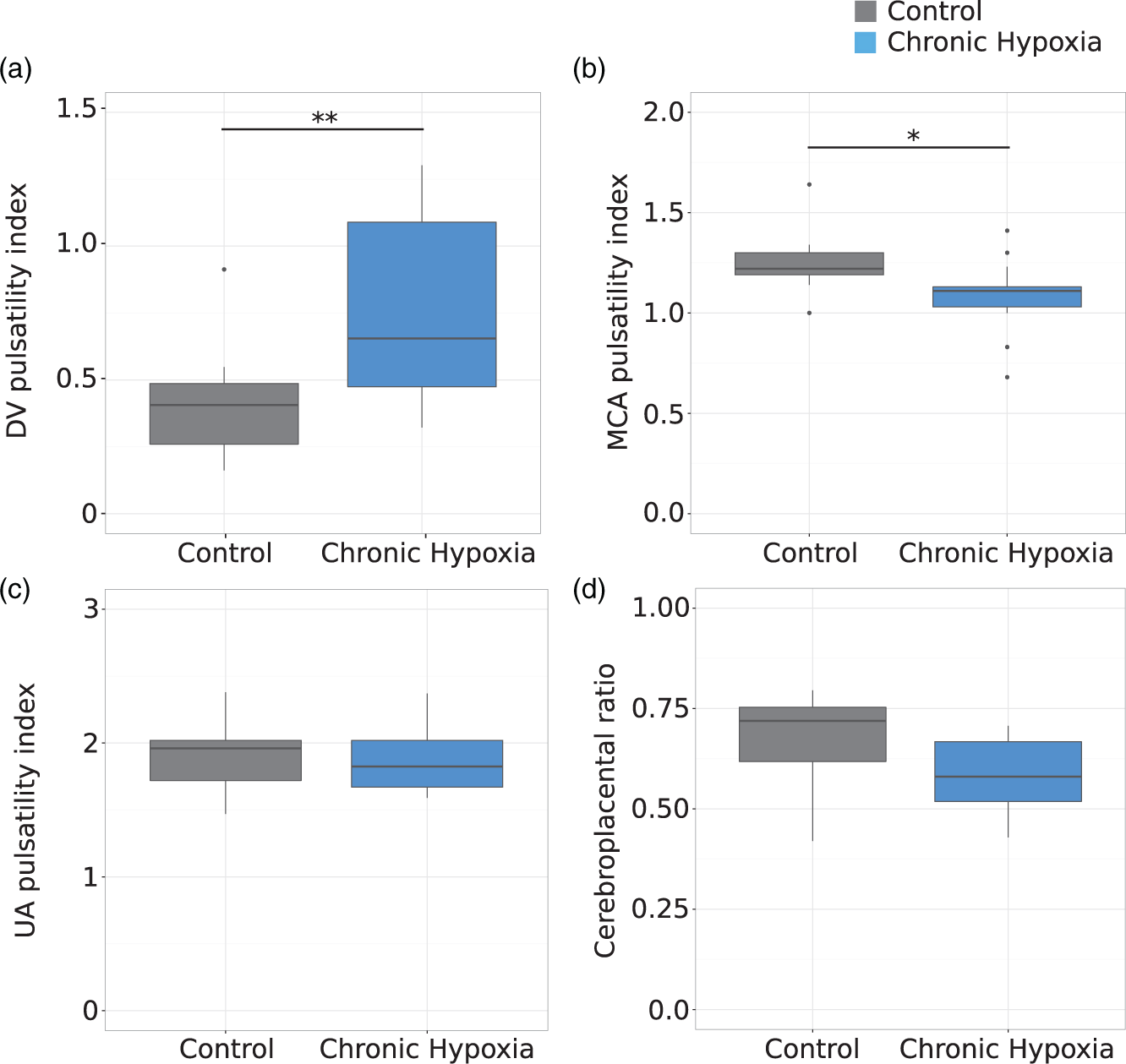
Fetal brain volume is preserved while lung and liver volumes decrease with chronic hypoxia
The physical manifestations of brain sparing following chronic maternal hypoxia were studied using ex vivo MRI to assess fetal organ volumes (Figure 4). The fetuses exposed to chronic maternal hypoxia showed evidence of asymmetric growth restriction, measured as an increased ratio of brain volume/body weight (75.3 (CI: 68.5–82.1) μl/g vs. 64.6 (CI: 60.3–68.9) μl/g, p = 0.006). Interestingly, the absolute brain volume did not differ between the chronic hypoxia fetuses and controls, indicating protection of brain growth. However, the liver and lung volumes were 22 and 27% smaller in the fetuses exposed to chronic maternal hypoxia compared to controls (59.4 (CI: 49.7–69.1) mm3 vs. 76.6 (CI: 66.1–87.1) mm3, p = 0.01 and 25.4 (CI: 21.8–29.0) mm3 vs. 34.8 (CI: 28.6–41.0) mm3, p = 0.01, respectively).
(a) Mid-sagittal section of an E17.5 fetus acquired by ex vivo MRI (resolution = 40 µm3) and (b) superposition of the manual segmentation of the brain (red), liver (blue) and lung (green). (c) Organ volume normalized to fetal weight (μl/g) and (d) absolute organ volume (mm3) for the control (grey) and chronic hypoxia (blue) fetuses. The chronic hypoxia fetuses showed asymmetric growth restriction: increased brain volume/body weight and decreased liver and lung volumes. The box-and-whisker plots show median and quartiles. n = 9 controls and n = 8 hypoxia. *p < 0.05.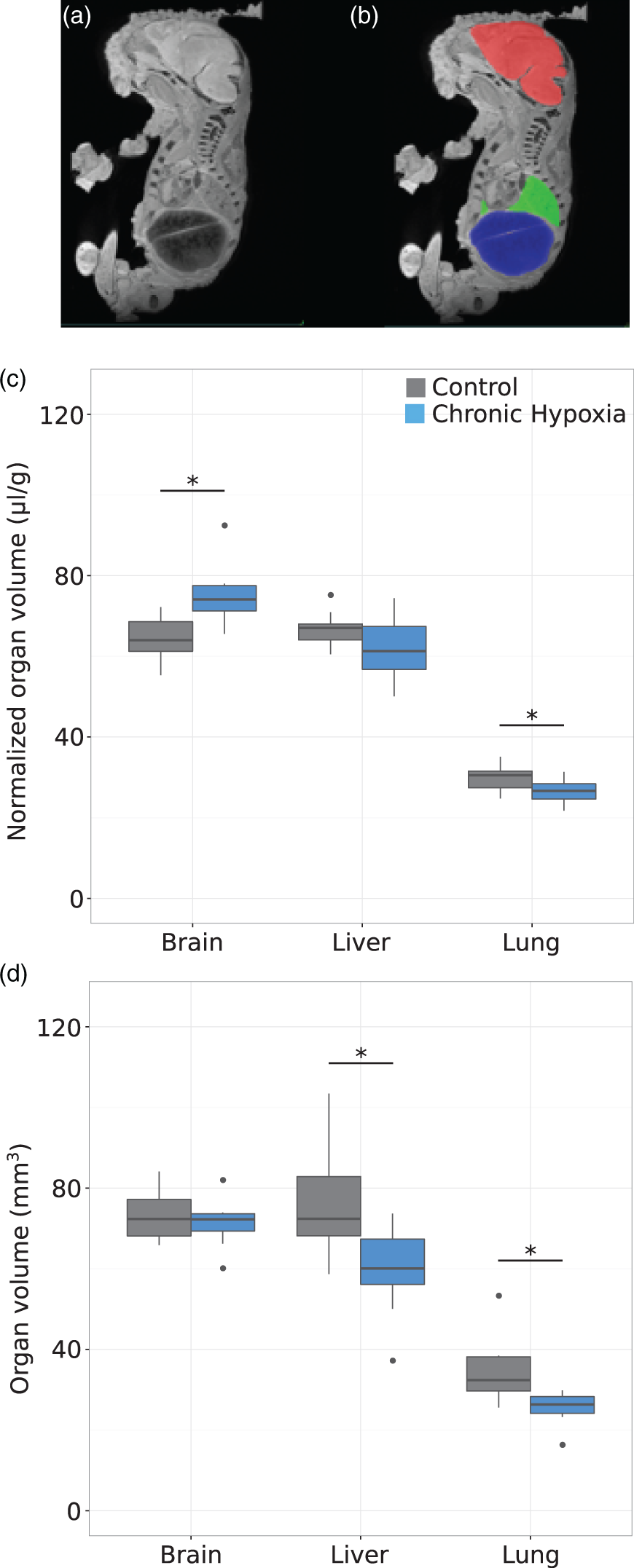
The chronic hypoxia-exposed fetal capacity for compensation is exceeded under an acute hypoxic challenge
To assess the fetal capacity to compensate for additional hypoxia stress, the effect of an acute hypoxia challenge (dam breathing 8% O2 for 3 min after removal from chronic hypoxic environment) on the fetal blood flow redistribution was studied (Figure 5). With an acute hypoxic challenge, the cerebral and DV blood flow increased by 15 (CI: 10–20)% and 31 (CI: 20–42)%, respectively, in control fetuses. The increased flow in the DV was achieved by vasodilation (23 (CI: 16–30)%). In addition, the MPA and DA blood flow for the control fetuses increased by 4 (CI: −8 – 16)% and 28 (CI: 12–44)% respectively, resulting in a decrease of PBF by 38 (CI: 10–66)% as more blood is directed to the DA. The UA blood flow increased by 16 (CI: −11 – 43)%. In contrast, the blood flow in all of the vessels of interest in the fetuses exposed to chronic maternal hypoxia decreased when challenged. The decrease was accompanied by either a decrease or no change in vessel diameter and heart rate. After 2.5–3 min of the hypoxia challenge, the control fetuses experienced significant bradycardia, while the chronic hypoxia fetuses did not show a significant change in heart rate.
Mean velocity/blood flow in control (grey) and chronic hypoxia (blue) fetuses for (a) MCA, (b) MPA, (c) DA, (d) DV, (e) PBF, (f) UA at (i) baseline (after chronic normoxia/hypoxia exposure) and after an acute hypoxia challenge (acute-after-chronic); (ii) the percent change with the acute hypoxia challenge. (g) The average systemic fetal heart rate (beats per minute, bpm) at (i) baseline and after an acute hypoxia challenge; (ii) the percent change with the acute hypoxia challenge. The control fetuses showed brain sparing physiology under the acute hypoxia challenge: increased MCA velocity, increased DA and DV blood flow and decreased PBF. For the fetuses exposed to chronic maternal hypoxia, the blood flow in all of the vessels of interest decreased when challenged. The box-and-whisker plots show medians and quartiles. n = 13 controls and n = 12 chronic hypoxia for the MCA; n = 13 controls and n = 10 chronic hypoxia for MPA, DA and PBF; n = 13 controls and n = 11 chronic hypoxia for the DV; n = 10 controls and n = 8 chronic hypoxia for the UA. *p < 0.05, **p < 0.005.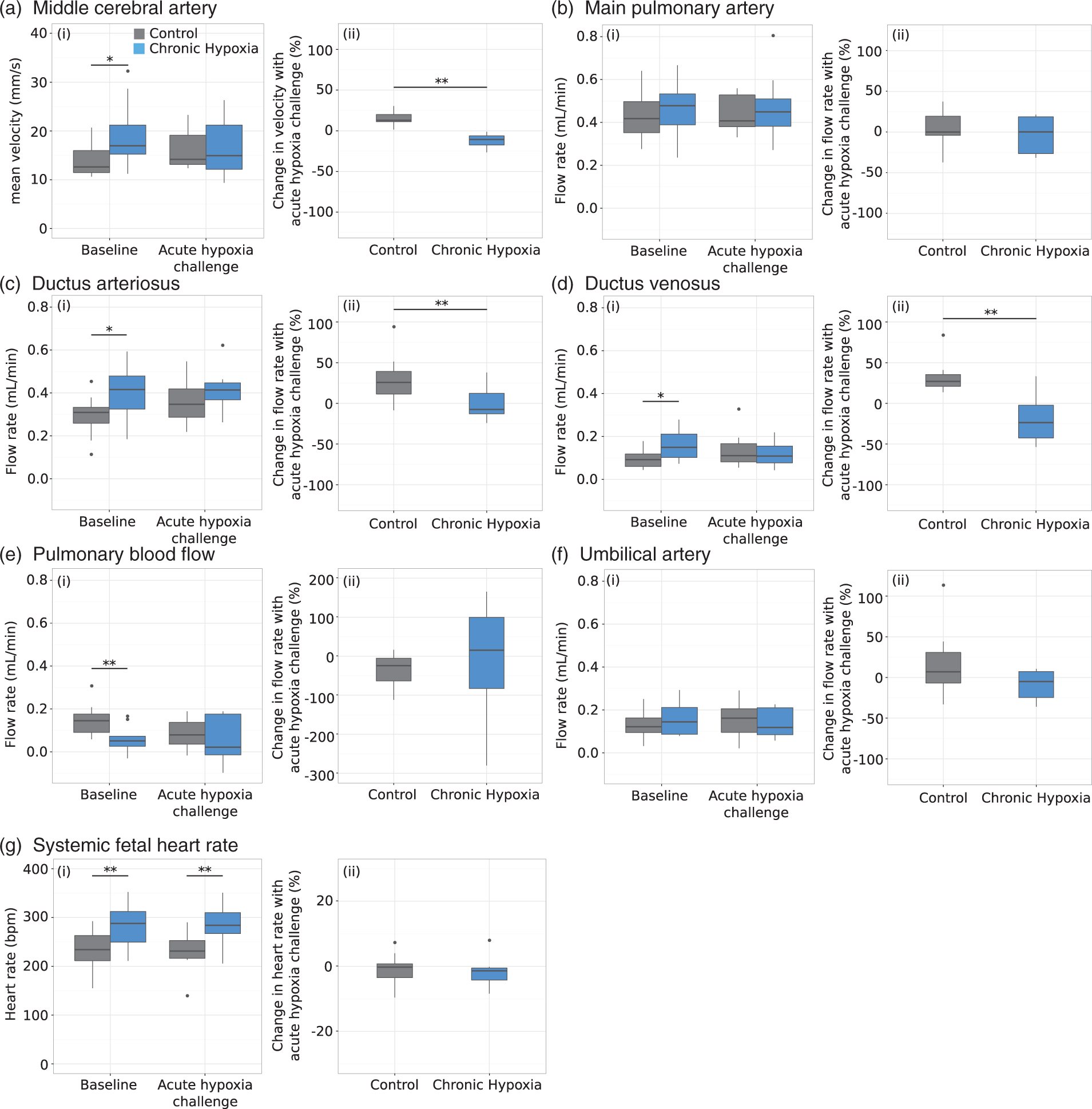
Under the acute hypoxia challenge, the DV PI increased by 81 (CI: 52–110)% and 23 (CI: −12–58)% in the control and chronic hypoxia fetuses, respectively, and there was no longer any statistically significant difference between the groups (p = 0.2). There was no change in the MCA PI in the control group between gas conditions; however, in the chronic hypoxia fetuses, the MCA PI increased by 2 (CI: −4 – 8)% under acute hypoxia. The UA PI increased by 3 (CI:−3 – 9)% in the control group under the acute hypoxia challenge and there was no difference in the UA PI for the chronic hypoxia fetuses between gas conditions. The consequence of the increased MCA PI in the chronic hypoxia fetuses is that there is no longer a statistically significant difference between the chronic hypoxia and control group under acute hypoxia conditions (p = 0.07) and there is no difference in CPR (p = 0.2).
Exposure to chronic maternal hypoxia does not induce changes in adult brain morphology
A second cohort of mice was survived to measure differences in post-natal development and brain morphology associated with chronic maternal hypoxia. The analysis of variance of the post-natal growth curves showed a group effect and a sex effect. Visually, the data suggest a trend towards a larger difference between the chronic hypoxia and control group for the males than females (Figure 6(a) and (b)) that is reflected in the group-by-sex interaction term (p = 0.1) of the analysis of variance. There was also a trend towards a smaller total brain volume in the chronic hypoxia mice compared to controls at P23 (414 (CI: 405–423) vs. 425 (CI: 416–434), p = 0.06), whereas the brain volume was similar between the groups at P59. A voxelwise comparison between chronic hypoxia and control brains at P23 revealed focal increases in relative volume in the cerebral peduncle (ventral tegmental area), pontine central grey matter and areas of the cerebellar white matter (Figure 6(c)). Once the mice reached adulthood (P59), there were no significant voxelwise brain volume differences between the chronic hypoxia and control groups.
Change in body weight with development for the (a) female and (b) male control (black circles) and chronic hypoxia (open squares) mice. (c) Anatomical differences in the brains of chronic hypoxia versus controls at P23. Top: sagittal view of the average ex vivo MR image. The green lines and numbers correspond to the coronal sections below. The images are overlaid with color maps indicating regions of relative volume that are significantly larger in mice exposed to chronic maternal hypoxia compared to controls (10% FDR). Focal significant differences are seen bilaterally in the cerebral peduncle (ventral tegmental area) (1), pontine central grey matter (2) and cerebellar white matter (3). n = 10–12 per sex from 5 to 8 litters per group.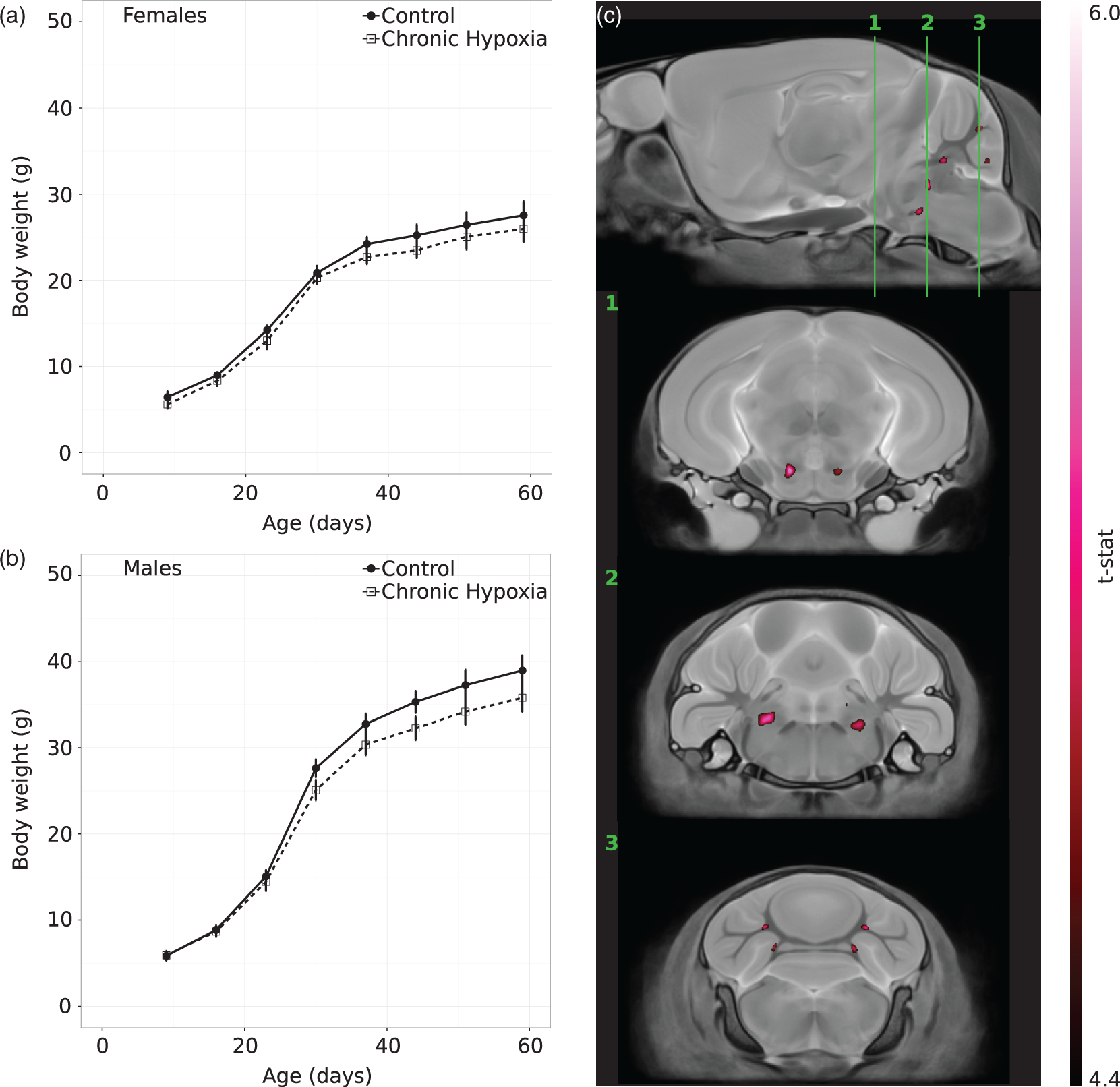
Discussion
In the present study, high-frequency ultrasound was used to study the fetal hemodynamic responses to chronic maternal hypoxia in late gestation. The increase in blood flow to the brain, facilitated by decreased cerebral blood flow impedance, demonstrates that the brain sparing response persists during chronic fetal hypoxia in mice. This response is mediated by “streaming,” where highly oxygenated umbilical venous blood preferentially flows across the DV and through the foramen ovale towards the cerebral circulation, bypassing the liver and the lungs. While this phenomenon has been described in humans and fetal sheep, 45 to our knowledge, this is the first demonstration of streaming in fetal mice. The mechanism of preferential streaming through the DV is supported here by the significant increase in DV blood flow and the strong correlation between mean MCA velocity and DV blood flow. Further evidence supporting brain sparing in the chronic hypoxia fetuses is the increased DA blood flow and reduction in PBF. Under normal physiology, blood flow from the right ventricle passes through the DA to the descending aorta, with a portion entering the pulmonary circulation. If the vascular impedance is lower in the cerebral circulation than in the subdiaphragmatic circulation, then there can be reversal of blood flow from the DA, through the aortic isthmus to the brain. 46 The fetal MR volumetric data indicate that brain sparing under chronic maternal hypoxia results in protection of brain growth, at the expense of the liver and lung volumes. This is supported by the MR volumetric data from the survived cohort where we detected only small structural changes within the brain of the chronic hypoxia mice at weaning and no differences in brain morphology by adulthood. This is consistent with humans born small-for-gestational age, who often show rapid catch-up growth during the first year of life. 47 Moreover, the trend towards a difference in growth trajectories for male and female mice following exposure to chronic hypoxia is consistent with evidence that males are more susceptible than females to hypoxic injury in utero.48,49
Our finding of brain sparing following chronic maternal hypoxia in mice is consistent with a recent study in pregnant sheep16 and with late-onset intrauterine growth restriction (IUGR) in humans where active regulation of cerebral oxygenation has been demonstrated using ultrasound50,51 and MRI. 52 The asymmetric growth restriction observed in the present study is also consistent with chronically hypoxic fetal sheep53 and human IUGR fetuses.54,55 As described in humans, the presence of brain sparing and venous redistribution following chronic maternal hypoxia is evidence of placental dysfunction. Decreased CPR, reverse DV flow and elevated DV pulsatility indices are hallmarks of accelerating placental dysfunction. 40 Another characteristic response to placental dysfunction is AREDV in the UA; however, this was only present in a small number of hypoxia-exposed fetuses (5/12) and there was no difference in UA PI between groups. It has been shown in sheep that progression to AREDV only occurs when 60–70% of the villous vascular tree has been damaged. 56 It is not clear whether the inconsistent observation of AREDV in the present study reflects a difference in the degree of placental dysfunction compared to the sheep study or a difference in the fetal response to maternal hypoxia as compared to villous occlusion.
Based on the lower average fetal litter weight, we anticipated that the UA blood flow would be lower in the chronic hypoxia fetuses compared to controls. 57 Surprisingly, the blood flow was similar between the groups. Reduced fetal blood oxygen saturation in IUGR fetuses has been reported in invasive animal studies, 58 human cordocentesis 59 and in our recent MRI study using T2 oximetry in fetal blood. 52 Therefore, it is likely that UA blood flow was maintained in the chronic hypoxia fetuses to compensate for the lower UA blood oxygen saturation. The placental adaptations could also explain the maintained UA blood flow but this was not investigated in the present study.
The hemodynamic response to an acute hypoxia challenge of the control fetuses closely matched the flow patterns observed in fetuses following exposure to chronic maternal hypoxia. Both showed evidence of brain sparing with increased cerebral and DV blood flow, increased DA blood flow and decreased PBF. Under the acute hypoxia challenge, the chronic hypoxia and control fetuses had similar DV PI, MCA PI and CPR. The increase in the MCA velocity in the control fetuses of 15% and no change in MCA PI is consistent with our previous work. 15 Brain sparing is an adaptive vascular response, as evidenced by the vasodilation of the DV in the control fetuses during the acute hypoxia challenge. However, it is not clear in the fetuses exposed to chronic maternal hypoxia if the compensatory response remains active or if structural changes have occurred to make permanent this adaptive response.
In the chronic hypoxia fetuses, the acute-after-chronic hypoxia condition resulted in decreased blood flow in all of the vessels of interest, suggesting that the fetus can no longer compensate for the decreased oxygen transport across the placenta. This highlights that fetuses exposed to chronic maternal hypoxia are at increased risk of fetal demise caused by an acute episode of hypoxia; for example, those that commonly occur during labour and delivery.
During acute hypoxia, bradycardia was observed consistently in the control group. Bradycardia is known to occur in human fetuses during hypoxic conditions 60 and is triggered by a carotid chemoreflex. 61 Interestingly, this fetal heart rate response was diminished in the chronic hypoxia fetuses, suggesting blunting of this fetal chemoreflex defense response. This is consistent with studies of chronic fetal hypoxia conducted in chronically instrumented sheep.62–64
It must be noted that the acute hypoxia challenge used here was performed after removal of the dam from the hypoxia condition and reoxygenation (acute-after-chronic) and should be distinguished from studies with an acute-on-chronic experimental paradigm. One advantage of the acute-after-chronic model is that both the chronic hypoxia and control groups have the same partial pressures of oxygen during baseline measurements, enabling comparison of the magnitude of the response to the acute challenge. 11 To our knowledge, there is only one other study that has reported the effects on the fetal cardiovascular system using an acute-after-chronic model. 63 Consistent with our findings, they report an attenuated cardiovascular response to an acute hypoxia challenge and note that this renders the sheep fetus more susceptible to hypoxia. A few experimental sheep studies have investigated the effects of acute-on-chronic hypoxia62,65,66 but results have varied. The study by Kamitomo et al. reported no difference in cerebral blood flow between the high altitude and low-altitude groups and a similar increase in cerebral blood flow between groups after an acute hypoxia challenge, whereas the work of Tissot van Patot et al. found the group raised at high altitude had elevated cerebral blood flow prior to the acute hypoxia challenge and a blunted response to the challenge. Compared to our result of fetal decompensation with an acute hypoxia challenge, the difference may be explained by the milder conditions of the chronic hypoxia protocol in sheep or by the difference in experimental model (acute-after-chronic vs. acute-on-chronic). Two of the studies reported blunted bradycardia with acute-on-chronic hypoxia62,66 which is consistent with the results presented here.
A limitation of the present study is that the sex of the fetus imaged was not determined. Recent evidence suggests that the physiological outcomes of maternal hypoxia are sex dependent26,67 and it is possible that some of the variability in our measurements could be the result of sex differences. For example, several of the fetuses had cerebral and DV blood flow within the range of the control group. One possible explanation is that these fetuses are female and less susceptible to hypoxic insult. Another limitation is that the food intake of the dams exposed to chronic hypoxia was not measured; however, a recent study using a similar chronic hypoxia protocol reported no effect of hypoxia exposure on maternal food intake. 26 Finally, unlike many of the studies in chronically hypoxic sheep, the dams were anesthetized during the ultrasound imaging. While the isoflurane levels were kept constant for each animal ensuring reliable comparisons between groups, the absolute blood flow values will not replicate measurements from an awake animal.
In summary, we have presented an easy-to-implement experimental mouse model of chronic maternal hypoxia that mimics many of the features of human chronic fetal hypoxia including asymmetric fetal growth restriction and brain sparing physiology. The use of experimental mice allowed us to investigate the fetal hemodynamic response to chronic hypoxia and the mechanisms that lead to fetal distress. This mouse model and the presented methodology for studying blood flow distribution in the mouse fetus could aid in the development of new diagnostic procedures for fetal monitoring and could be used to evaluate novel interventions to promote fetal well being.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work was provided by the Canadian Institutes of Health Research MOP130403.
Acknowledgements
We thank Yu-Qing Zhou for his help with the ultrasound data collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
LSC wrote the manuscript and performed the data analysis. JH performed the MRI experiments and completed the manual segmentation of the MR images. JPL, MS, CKM and JGS conceived of the study and are responsible for the overall design of the research. All of the authors approved the submitted version of the manuscript.
