Abstract
The underlying factors promoting increased mitochondrial proteins, mtDNA, and dilation to mitochondrial-specific agents in male rats following tMCAO are not fully elucidated. Our goal was to determine the morphological and functional effects of ischemia/reperfusion (I/R) on mitochondria using electron microscopy, Western blot, mitochondrial oxygen consumption rate (OCR), and Ca2+ sparks activity measurements in middle cerebral arteries (MCAs) from male Sprague Dawley rats (Naïve, tMCAO, Sham). We found a greatly increased OCR in ipsilateral MCAs (IPSI) compared with contralateral (CONTRA), Sham, and Naïve MCAs. Consistent with our earlier findings, the expression of Mitofusin-2 and OPA-1 was significantly decreased in IPSI arteries compared with Sham and Naïve. Mitochondrial morphology was disrupted in vascular smooth muscle, but morphology with normal and perhaps greater numbers of mitochondria were observed in IPSI compared with CONTRA MCAs. Consistently, there were significantly fewer baseline Ca2+ events in IPSI MCAs compared with CONTRA, Sham, and Naïve. Mitochondrial depolarization significantly increased Ca2+ sparks activity in the IPSI, Sham, Naïve, but not in the CONTRA group. Our data indicate that altered mitochondrial structure and function occur in MCAs exposed to I/R and that these changes impact not only OCR but Ca2+ sparks activity in both IPSI and CONTRA MCAs.
Keywords
Introduction
Mitochondria are recognized as essential energy producers and play a critical role in determining cellular viability but their importance in other aspects of vascular biology and pathology has been investigated only recently.1–16 While the brain is known to be one of the most mitochondrial rich organs due to its high energy demands, the cerebral vasculature also has extensive metabolic needs. Endothelium in the cerebral vasculature has the usual metabolic demands of this cell type similar to other circulations, but also has unique high energy requirements associated with the maintenance of tight junctions and the unique transport systems of the blood-brain barrier. Mitochondria in vascular smooth muscle (VSM) provide energy for adjusting vascular tone to meet brain metabolic demand and to protect the vulnerable microvasculature from upstream arterial pressure. Cerebral ischemia, with or without reperfusion, results in insufficient blood flow to the brain parenchyma and vasculature and leads to deranged energy metabolism, pH, and ion homeostasis in the ischemic core and penumbra region.17–21 While the effects of ischemic stress on mitochondria of different cells types such as neurons, astrocytes, and microglia in the brain have been studied in various experimental stroke models, 22 little information is known about the effects of ischemia/reperfusion (I/R) on mitochondria of the cerebral vasculature. We and others have shown that vascular mitochondria adapt to changing metabolic needs by adjusting energy production in the form of ATP and that mitochondria play an important role in the regulation of cerebrovascular function in both health and disease.2,3,7–10,13,14 In addition, we and other investigators have shown that activation of mitochondrial ATP sensitive potassium (mitoKATP) channels located on the inner mitochondrial membrane results in cerebral vasodilation under physiological conditions, both in vivo and in vitro.2,3,7,8,13,14,23 Vasodilation results from an integrated response to production of Ca2+ sparks in VSM together with production of endothelial nitric oxide (NO).2,3,7,8,13,14,23 We recently have shown that mitochondrial protein mass and DNA, the magnitude of mitochondrial depolarization to diazoxide (DZ), and the overall amount of dilation of ipsilateral (IPSI) MCAs to DZ were retained or enhanced in MCAs 48 h after transient middle cerebral artery occlusion (tMCAO) compared with MCAs from the non-occluded, contralateral (CONTRA) side or from sham-operated rats. 14 The relative contribution of endothelium to the dilator response to DZ was enhanced, whereas the VSM component was reduced. However, the precise intrinsic vascular mechanisms involved in these responses and the relationships between mitochondrial protein mass and mitochondrial structure in isolated MCAs with the mitochondrial bioenergetics and mitochondria-derived vascular function are unclear. Therefore, we examined cerebrovascular mitochondrial function in isolated MCAs under normal conditions and following tMCAO in male rats. Specifically, we determined mitochondrial morphology, expression of mitochondrial proteins, mitochondrial oxygen consumption rate (OCR), and mitochondria-derived Ca2+ sparks activity in isolated MCAs from healthy, Naïve rats and from rats exposed to tMCAO or Sham operation.
Material and methods
Animals
Age-matched, male, Sprague-Dawley rats (8–10-week old, n = 95) were obtained from Charles River Laboratories (Wilmington, MA, USA) and housed according to guidelines of the Institutional Animal Care and Use Committee of Tulane University, which were in compliance with National Institutes of Health Office of Laboratory Animal Welfare guidelines. The Institutional Animal Care and Use Committee of Tulane University approved the protocols, which were reported to be in compliance with ARRIVE (Animal Research: Reporting in Vivo Experiments) guidelines. Rats had free access to soft food and water, ad libitum. Animals were randomly assigned to Naïve, Sham, or tMCAO groups. Middle cerebral arteries from these three groups were isolated after euthanasia and labeled as Naïve, Sham, IPSI (ipsilateral to MCAO), and CONTRA (contralateral to MCAO), and used for OCR measurements, western blotting or electron microscopy. For the Ca2+ sparks measurements, the arteries were treated with both DMSO and DZ, resulting in the following subgroups: Naïve DMSO, Naïve DZ, Sham DMSO, Sham DZ, IPSI DMSO, IPSI DZ, CONTRA DMSO, and CONTRA DZ.
Ischemic stress
Rats were randomly assigned to either a Sham (without ligation or cutting of arteries or filament insertion) or tMCAO group, as described previously.
14
We also studied Naïve rats that were not subjected to any surgery. For tMCAO surgery, ischemia was induced under 100 mg/ml ketamine (KetaVed, St. Joseph, MO) and 20 mg/ml xylazine anesthesia, (i.p.) (Santa Cruz Biotechnology, Dallas, TX, respectively) for 90 min by modified Longa intraluminal filament method followed by 48 h reperfusion without anesthesia.
14
A midline incision was made in the neck and the right common and internal carotid arteries were isolated from the surrounding nerves and connective tissue. A rubber-coated silicone monofilament (Doccol, Sharon, MA, USA) was inserted into the internal carotid artery and advanced to the origin of the right MCA. The filament blocking blood flow (90 min) to the right MCA was removed and rats were allowed to recover for 48 h. Systemic analgesia, eye ointment, a rectal temperature probe, and a heating pad were used and rat vital signs were monitored during and after surgery. For example, body temperature was maintained relatively constant during the entire surgical period and the animals had normal coloration and did not need to be on a ventilator. After 48 h reperfusion and MCAs isolation, the infarcted brain area was determined using a 1% solution of 2,3,5-triphenyl-2H-tetrazolium chloride staining (TTC; Sigma-Aldrich, St. Louis, MO, USA) and ImageJ Software (NIH).
14
The tMCAO resulted in a consistent infarct area of 55.9 ± 2.5 % (mean ± SEM) expressed as the percentage of the area of the entire IPSI cortical hemisphere. No areas of infarcted tissues were present on the CONTRA cortical hemisphere. The low coefficient of variation establishes the uniformity of the tMCAO effects on the brains of the rats.
Mitochondrial morphology in MCAs following experimental stroke and in Sham and Naïve animals. (a, b) High magnification images of two IPSI MCAs 48 h after tMCAO. Mitochondria in VSM of IPSI arteries often appeared damaged and the density of mitochondria in VSM generally decreased (indicated by arrows on panels (a), (b), and the top of panel (e)). Damaged mitochondria showed various forms of mitoptosis, including localized areas where normal morphology of internal structures such as cristae were lost, generalized mitochondrial swelling or increased density of staining, which is consistent with condensation of internal structures. However, some post tMCAO VSM cells with extensively damaged mitochondria were found adjacent to neighboring VSM cells showing normal appearing mitochondria in close association with SR. Mitochondria in endothelial cells and adventitia (arrows) often appeared to be separating into two distinct mitochondria (fission), which would be consistent with increased mitochondrial levels in these cell types. The black vertical band in Panel A represents a folding artifact along the internal elastic lamina during processing and/or imaging of the section and does not distort the vascular morphology of adjacent cells. Mitochondria numbers in endothelium and adventitia appeared greater with normal morphology compared with MCAs from sham-operated rats. (c) High magnification image of MCA from a sham-operated rat. Endothelial cells, VSM, and adventitia had normal looking morphology with densities similar to our previously published images of Naïve rats.3,10 Mitochondria in VSM were largely arrayed in dense fields with interspersed SR in Sham arteries. In Sham and Naïve rats, occasional mitochondria observed in VSM lacked internal structures (arrow) indicating mitoptosis, a characteristic present in otherwise normal cells under physiological conditions. (d) High magnification image of a CONTRA MCA 48 h after tMCAO. Mitochondria in endothelium and VSM had normal morphology and density. Arrows show the location of mitochondria in endothelium. However, areas of low and high mitochondrial density were present in adjacent VSM cells in the same CONTRA artery, compared with a more consistent mitochondrial configuration among adjacent VSM cells in Naïve or Sham-operated rats. (e) Representative EM images of IPSI and CONTRA VSM cells showing significantly increased numbers of damaged mitochondria in VSM of IPSI MCAs compared with CONTRA. Arrows show structurally damaged mitochondria. (f) Quantitative measurements of the number of damaged mitochondria in IPSI (n = 7) and CONTRA (n = 6) arteries. Numbers of structurally damaged mitochondria in each VSM cell were greater in IPSI compared to CONTRA MCAs (P < 0.05). Mitochondria showing signs of structural damage are occasionally seen in normal VSM cells (Figure 1(c)) but are a consistent feature of IPSI MCAs. (g) Vascular expression of citrate-synthase in IPSI (I), CONTRA (C), Sham (S), and Naïve (N) arteries normalized to GAPDH. (Data are expressed as mean ± SEM; n = 8/each groups; P > 0.05). Differences in mitochondrial fission and fusion proteins and MnSOD in MCAs following experimental strokes and in Sham and Naïve animals. Vascular expression of the fusion proteins (a) Mfn-2, (b) OPA-1, as well as (c) acetylated MnSOD, and (d) total MnSOD shown in IPSI, CONTRA, Sham, and Naïve MCAs. (e) Histogram showing the expression of the fission protein DRP-1 in IPSI, CONTRA, and Sham arteries was adapted from Ibolya Rutkai et al.
14
Data are expressed as mean ± SEM. (n = 8/each group) *IPSI vs. Sham and Naïve, P < 0.05; #CONTRA vs. Sham and Naïve, P < 0.05; $CONTRA vs. SHAM, P < 0.05. 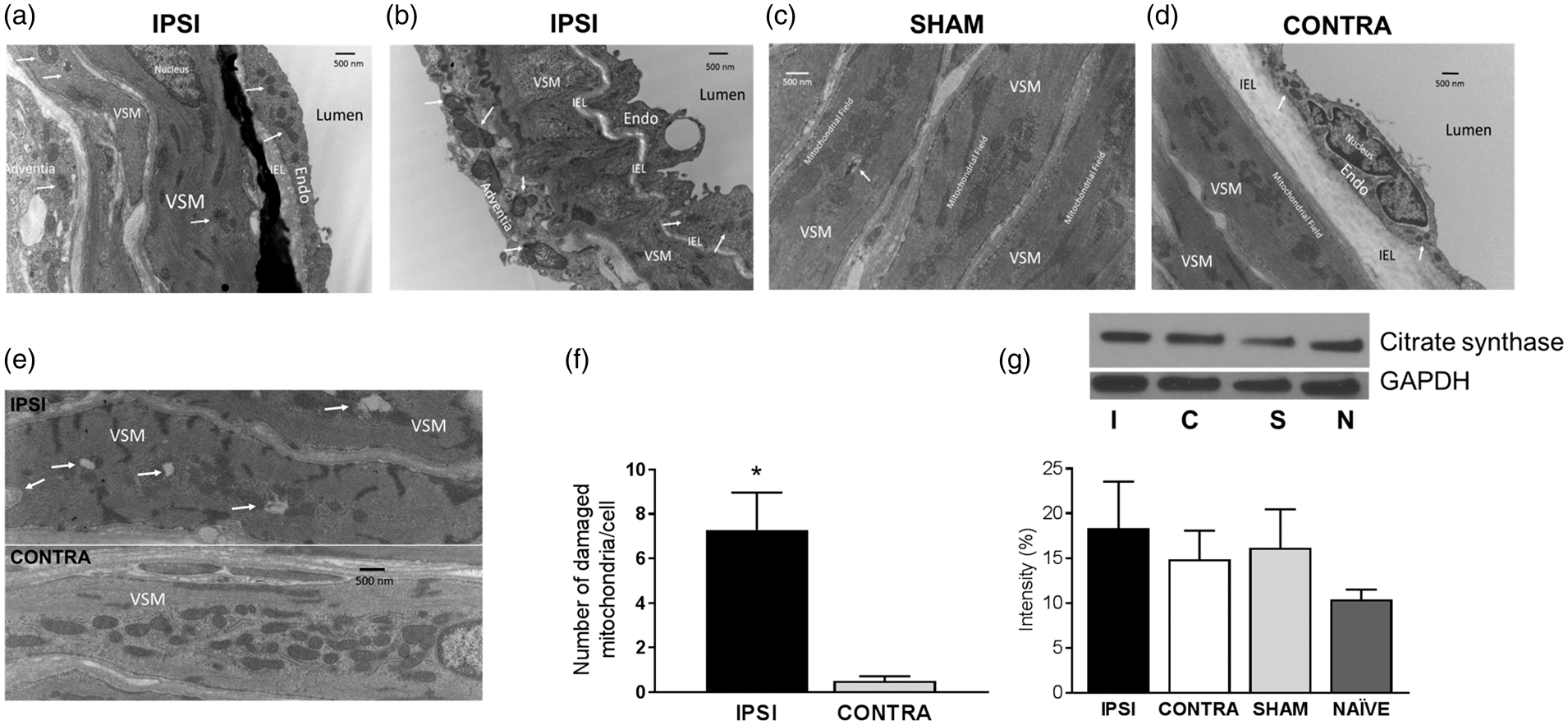
 IPSI vs. CONTRA and Sham.
IPSI vs. CONTRA and Sham.
Electron microscopy
Rats were euthanized with an overdose of ketamine and xylazine anesthesia (i.p.) and perfused with a PBS solution of 2% glutaraldehyde and 3% formaldehyde.9,10,13 The MCAs were removed, kept in perfusion solution for 1 h, post fixed in 1% osmium tetroxide, and embedded in Spurr's resin. Ultrathin (80–90 nm) artery sections were mounted on formvar-coated copper grids (200 mesh), air dried, and stained with uranyl acetate and lead citrate (7 min and 7 min, respectively). The sections were put on grids and viewed at different magnifications using a FEI Tecnai BioTwin 120 keV TEM with a digital imaging setup (Wake Forest University Health Sciences, Winston-Salem, NC, USA). We collected MCAs from 8 Naïve, 3 Sham, and 7 MCAO animals for electron microscopy.
Western blot
MCAs were isolated from rats 48 h after 90 min of tMCAO or Sham surgery, or from Naïve rats, then homogenized in ice-cold NP40 lysis buffer containing phosphatase and protease inhibitors (Invitrogen, Frederick, MD; #P8340 and #P2850, Sigma Aldrich, St. Louis, MO, USA) and centrifuged. The supernatant was used for Pierce BCA protein assay (Thermo Scientific) and the proteins were separated using a 4–20% SDS-PAGE gradient gel and transferred onto PVDF membrane. Afterward, casein blocking buffer (#92740200, Li-Cor, Lincoln, NE, USA) was used to block the non-specific binding sites for 60 min at room temperature and to dilute the primary antibodies. The membranes were washed with Tris-buffered saline containing 0.1% of Tween-20 (TBST, Sigma-Aldrich) in between the antibody application. The membranes were incubated overnight at 4℃ with the following primary antibodies: anti-Mitofusion-2 at 1:1000 dilution (86 KDa, #M6444, Sigma Aldrich), anti-OPA-1 at 1:1000 dilution (80–100 KDa, #612606, BD Transduction), anti-SOD2/MnSOD acetyl K68 at 1:500 dilution (24 KDa, #ab137037, Abcam), anti-MnSOD at 1:2000 dilution (25 KDa, #611581, BD Transduction), anti-Citrate-synthase at 1:1000 (predicted 52 KDa, #ab96600, Abcam), anti-beta actin at 1:5000 dilution (42 KDa, #A5441, Sigma Aldrich) or anti-GAPDH at 1:1000 (37 KDa, #2118s, Cell Signaling). Afterward, membranes were washed and incubated with the respective secondary antibody, either goat anti-rabbit IgG at 1:2500 dilution (#7074S, Cell Signaling Technology) or goat anti-mouse IgG at 1:5000 dilution (#7076P2, Cell Signaling Technology) at room temperature for 1 h. Chemiluminescence (LumiGLO, Gaithersburg, MD, USA) and autoradiography were used to visualize the final reaction. The relative optical densities of the immunobands were determined after scanning and normalizing to the corresponding β-actin or GAPDH loading control using the ImageJ Software.
Mitochondrial OCR measurement
The MCAs from IPSI, CONTRA, Sham operated, and Naïve rats were isolated from the hemispheres 48 ± 6 h after tMCAO surgery. Mitochondrial OCR was measured by the Seahorse Bioscience XFe24 Analyzer.
13
Arteries were placed in wells of a 24-well islet plate (#101122-100, Agilent Technologies, Santa Clara, CA), each containing 525 µl of Seahorse XF Assay medium (#102365-100, Seahorse Bioscience, 5.0 mmol/l glucose and 2.0 mmol/l pyruvate, pH 7.4 and 37℃). Our assay consisted of eight cycles for baseline measurements, a media injection after the third measurement, and five sequentially injected cycles of oligomycin (2 µmol/l), carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP, 1 µmol/l), and 1.5 µmol/l each of antimycin and rotenone. The protein concentration of each well was determined by Pierce BCA protein assay and used to normalize the raw OCR values by Wave 2.3.0 Software, then exported to GraphPad and Excel which calculated the components of mitochondrial respiration, as described previously. The OCR values were expressed as pmol/minute/µg protein. A schematic illustration is presented in Figure 3(a).
Mitochondrial respiration of MCAs. (a) Schematic of the Seahorse experimental design, showing sequence of agent injections to elicit different components of mitochondrial respiration. (b) Continuous tracings of protein normalized mitochondrial oxygen consumption rate expressed in pmol/min/µg protein unit in each group. Calculated values of mitochondrial OCR with statistical analyses shown for (c) non-mitochondrial respiration, (d) basal respiration, (e) proton leak, (f) ATP production, (g) maximal respiration, and H) spare respiratory capacity in IPSI, CONTRA, Sham, and Naïve MCAs. Data are expressed as mean ± SEM. *IPSI vs. NAÏVE, P < 0.05; #NAÏVE vs. SHAM, P < 0.05; $IPSI vs. SHAM and NAÏVE, 
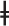 CONTRA vs. NAIVE, P < 0.05; &IPSI vs. CONTRA, SHAM, and NAÏVE, P < 0.05; @CONTRA vs. SHAM, P < 0.05.
CONTRA vs. NAIVE, P < 0.05; &IPSI vs. CONTRA, SHAM, and NAÏVE, P < 0.05; @CONTRA vs. SHAM, P < 0.05.
Calcium sparks imaging
Ca2+ sparks activity was measured in freshly isolated MCAs using a Zeiss laser scanning confocal system (LSM 510 and 7 Live, Jena, Germany) with a Zeiss C-Apochromat 63 × 1.2 NA water immersion objective. The MCAs were loaded with 5 µM of Fluo-4 AM and 20% Pluronic F-127 (Invitrogen, Frederick, MD, USA) dissolved in HEPES (mmol/l of 134 NaCl, 6 KCl, 1 MgCl2, 2 CaCl2, 10 HEPES, 0.026 EDTA, and 10 glucose) for 1 h, then washed using HEPES. 7 Afterward, MCAs were kept in room temperature HEPES for 30 min for de-esterification. For imaging, arteries were treated with 250 µM of DZ or an equivalent amount of DMSO (vehicle control). The collected images (512 × 512 pixel) were analyzed using the ImageJ Fiji Software (NIH) with the xySpark plugin (University of Leeds, UK). The number of sparks was calculated and normalized to 10 µm2 cell area.
Data analysis and statistics
Results were expressed as mean ± SEM and the number of independent measurements is indicated by “n”. Data comparison was performed using unpaired Student’s t-test and one-way ANOVA with Tukey’s post hoc analysis. P < 0.05 was considered statistically significant. There was no blinding done although the experiments were done in the presence of two of the co-authors and the data analysis was randomly assigned and performed by the co-authors.
Results
Electron microscopy
Distinctive and pronounced effects on mitochondrial morphology were apparent on IPSI (Figure 1(a) and (b), top of panel (e)) but not of SHAM (Figure 1(c)) or CONTRA MCAs (Figure 1(d)). In VSM cells of IPSI MCAs, mitochondria often appeared damaged with areas of disrupted, disintegrated, or condensed internal structures (Figure 1(e) and (f)). For these determinations, we carefully selected VSM cells from MCAs where we could observe virtually the entire VSM cell at the same magnification and in which the morphology was very clear. Additionally, the extensive mitochondrial fields typically seen in VSM in normal MCAs were reduced in area. However, in some VSM cells of IPSI arteries, morphology and density appeared to be within normal range for all the mitochondrial observed. In contrast, mitochondria in endothelial and adventitial cells in IPSI MCAs appeared to be more prevalent than in Sham MCAs and lacked evidence of the extensive mitoptosis seen in VSM cells. A characteristic feature of mitochondria in endothelial and adventitial cells of IPSI arteries was the close approximation of adjacent mitochondria with intact internal morphology (Figure 1(a) and (b)), which is suggestive of fission of one mitochondrion into two distinct mitochondria. However, due to the thinness of the endothelium and the variability in the direction of sections, it was difficult to quantify mitochondrial numbers without more extensive studies with three-dimensional reconstruction which is beyond the scope of the present study. In all MCA cell types in Sham (Figure 1(c)) operated animals, mitochondrial morphology, density, and relationship to other cellular structures were similar to Naïve arteries. 3 Thus, mitochondria in VSM cells form dense fields in close contact with the interspersed sarcoplasmic reticulum (SR). Mitochondria were less dense in endothelial and adventitial cells than in VSM cells and were present individually rather than forming mitochondrial fields. In MCAs from Sham and Naïve rats,3,13 occasional mitochondria in VSM appeared to have lost internal structures or organization, which supports the concept that limited mitoptosis is a routine characteristic of normal cells. Mitochondrial morphology, intracellular localization, and association with other cellular structures appeared to be within the normal range for CONTRA MCAs (Figure 1(d)). Typical mitochondrial fields were present in VSM cells of CONTRA MCAs and ECM, and adventitia did not exhibit any characteristics distinct from Sham or Naïve MCAs.
Western blot
Transient MCAO lead to changes in mitochondrial-related proteins in cerebral arteries. Expression of Mfn-2 and OPA-1 proteins were significantly (P < 0.05) decreased in IPSI (Mfn-2: 67.34 ± 4.08; OPA-1: 55.74 ± 6.41) and CONTRA MCAs (Mfn-2: 58.83 ± 4.096; OPA-1: 62.22 ± 6.95) compared with Sham (Mfn-2: 144.0 ± 13.00; OPA-1: 92.07 ± 6.72) and Naïve arteries (Mfn-2: 115.3 ± 10.76; OPA-1: 81.19 ± 6.05) (Figure 2(a) and (b)). There were no differences among the levels of the acetylated MnSOD among the groups (IPSI: 75.80 ± 15.15; CONTRA: 82.14 ± 16.44; Sham: 79.07 ± 17.18; and Naïve: 57.37 ± 13.06) (Figure 2(c)), whereas the expression of total MnSOD was significantly increased in IPSI MCAs (171.4 ± 24.24; P < 0.05) compared with Sham, Naïve (90.07 ± 17.92; 50.96 ± 9.93; respectively), and CONTRA arteries (113.4 ± 17.22) (Figure 2(d)). Our previous results, included here for completeness, showed increased Dynamin-related protein-1 (DRP-1) levels in IPSI compared with CONTRA and Sham MCAs (Figure 2(e)). 14 A trend toward an increase, which did not reach significance, in the expression of citrate-synthase, was observed in the IPSI arteries (18.33 ± 5.21) when compared with the CONTRA (14.88 ± 3.20), Sham (16.15 ± 4.31), and Naïve (10.4 ± 1.12) groups (Figure 1(g)).
Mitochondrial OCR measurement
Oxygen consumption rate in MCAs increased dramatically after tMCAO in IPSI compared with CONTRA, Sham, and Naïve groups (Figure 3). Specifically, I/R resulted in an increase in the OCR in IPSI MCAs (non-mitochondrial respiration: 148.3 ± 13.95; proton leak: 232 ± 53.7; ATP production: 209.5 ± 21.45; basal respiration: 293.2 ± 64.82; maximal respiration: 691.1 ±129.4; and spare respiratory capacity: 397.9 ± 80.12; n = 10) compared with CONTRA, Naïve, and Sham arteries (non-mitochondrial respiration: 96.64 ± 10.69; proton leak: 183.3 ± 36.69; ATP production: 69.34 ±24.92; basal respiration: 156 ± 26.57; maximal respiration: 416.8 ± 55.27; and spare respiratory capacity: 260.8 ± 45.72, n = 10) (Figure 3(b) to (h)). Mitochondrial respiration was greater in the Sham (n = 14–20) compared with Naïve MCAs (n = 20–29) for all mitochondrial respiratory parameters, including non-mitochondrial respiration (108 ± 24.67; 47.78 ±5.16, respectively), proton leak (111 ± 28.58; 69.72 ±12.55, respectively), ATP production (134.4 ± 21.22; 70.6 ± 6.66, respectively), basal respiration (133 ±32.58; 92.54 ± 15.47, respectively), maximal respiration (239.1 ± 40.42; 167.9 ± 17.89, respectively), and spare respiratory capacity (106.1 ± 13.07; 75.37 ± 5.89, respectively) (Figure 3(b) to (h)).
Calcium sparks measurement
Calcium sparks activity was affected by tMCAO. The most pronounced aspect of Ca2+ sparks activity (Ca2+ sparks number/10 µm2; n = 19) in the groups studied was the abnormally low level in IPSI MCAs during baseline conditions in the presence of DMSO (0.015 ± 0.004; n = 19, P < 0.05) compared with the CONTRA and Sham groups (0.057 ± 0.009; n = 26) (Figure 4), which correlated with the structural damage observed in VSM (Figure 1(e) and (f)). Diazoxide treatment increased Ca2+ sparks activity in all of the groups except the CONTRA MCAs. Administration of DZ significantly increased Ca2+ sparks activity in IPSI MCAs (0.120 ± 0.015; n = 39, P < 0.05), whereas it had little or no effect in the CONTRA group (0.066 ± 0.008, n = 35). The Ca2+ sparks activity of Naïve MCAs, measured in the presence of DMSO (0.104 ± 0.019, n = 27), was significantly increased after DZ treatment (0.182 ± 0.022, n = 32). Surprisingly, Ca2+ sparks activity in the Sham DMSO group was decreased (0.055 ± 0.011, n = 38) compared with the Naïve DMSO group, but treatment with DZ resulted in a significant increase in the Ca2+ events of Sham arteries (0.095 ± 0.019, n = 18), which was similar in magnitude to the Naïve DZ group.
Ca2+ sparks activity in isolated MCAs. Ca2+ sparks activity shown in the presence of DMSO and diazoxide (DZ) in isolated IPSI, CONTRA, Sham, and Naïve MCAs. Data are expressed as mean ± SEM. *DMSO vs. DZ within groups P < 0.05; #IPSI vs. CONTRA within groups P < 0.05.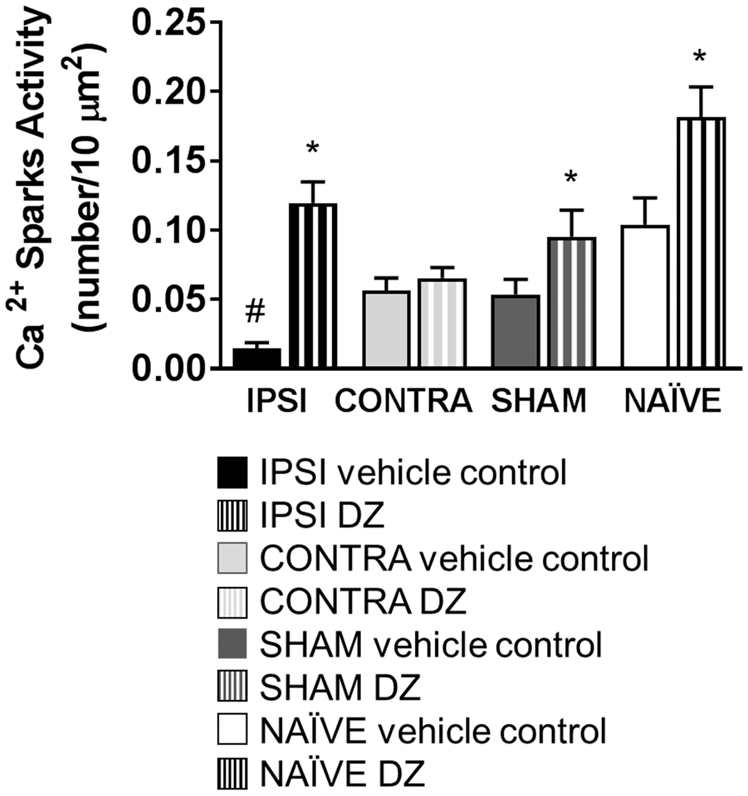
Discussion
There are three novel findings from our study with important implications for the understanding and treatment of strokes. First, mitochondrial and non-mitochondrial OCR were greatly enhanced in MCAs directly impacted by transient ischemia. All individual components of mitochondrial OCR were increased on the IPSI compared with the CONTRA, SHAM and Naïve MCAs. Increases in mitochondrial respiration were associated with augmented levels of numerous mitochondrial-related proteins and mitochondrial DNA, as shown in this and a previous publication. The previously reported increase in DRP-1 activity and reduced levels of fusion proteins shown in this paper are consistent with mitogenesis contributing to these changes. The increase in mitochondrial numbers appears to occur primarily in endothelium and adventitia, but because of technical limitations of convention electron microscopy methods, we cannot state this with certainty. Modest increases in mitochondrial proton leak, maximal respiration, and spare capacity occurred in CONTRA compared with Naïve MCAs, indicating the cerebral arteries also are affected at sites distant from direct ischemic stress. We did not examine the factors leading to increases in non-mitochondrial OCR but our previous studies indicate possible roles for cyclooxygenase, nitric oxide (NO) synthase, and NADPH-oxidase systems. 24 Second, calcium sparks activity was reduced during baseline conditions in the IPSI compared with other MCAs, and this physiological function was correlated with evidence of mitochondrial structural damage in VSM. Reduced Ca2+ sparks activity is consistent with our previous studies showing a predominant VSM defect to MCA dilation following mitochondrial activation after tMCAO.2,3,14 Diazoxide administration increased Ca2+ sparks activity in IPSI, SHAM, and Naïve but not CONTRA MCAs. The lack of responsiveness of CONTRA MCAs to DZ was surprising due to the normal appearance of mitochondria in all cell layers and the absence of significant morphological damage to mitochondria in VSM. Third, SHAM surgery has modest effects on mitochondrial OCR but substantial effects on Ca2+ sparks activity, indicating that even minimal surgery and/or anesthesia have prolonged effects on mitochondrial function in cerebral arteries lasting at least two days. In summary, tMCAO has diverse effects on MCAs directly exposed to ischemia and infarcted tissues as well as MCAs not subjected to ischemia and located distant from the stroke area.
A variety of techniques such as positron emission tomography, magnetic resonance imaging, electron paramagnetic resonance oximetry, perfusion imaging/diffusion-weighted imaging, and the T2*OC technique developed for the detection of the penumbra have been used to measure blood flow and tissue oxygenation in the brain.25–30 Occlusive stroke reduces blood flow and tissue oxygen not only in the area supplied by the occluded artery but in other areas of the cerebral circulation.17,19,28,31–34 Yaseen et al. found that intravascular pO2 in the superficial cortex of male, Sprague Dawley rats decreased with artery size. 35 Similar findings were reported by Devor et al. in female, Sprague Dawley rats and Sakadžić et al. in male CD-1 mice.36,37 Moderate reductions in blood flow to the brain such as carotid occlusion results in a 50% decrease in brain pO2 of male Mongolian gerbils, measured by electron paramagnetic resonance oximetry. 38 Liu et al. showed that prior to ischemia in male, Sprague Dawley rats, interstitial pO2 in the brain was significantly higher compared with the penumbra and ischemic core. 39 Transient MCAO resulted in a decrease in the interstitial pO2 in both the ischemic core and penumbra, which were significantly greater in the penumbra compared with the ischemic core. However, 1 h after the reperfusion, pO2 values in the ischemic core were increased and almost reached the pre-ischemic level, whereas the penumbra had a partial recovery. Similar findings were reported by Nakai et al. in MCAO models of male, Wistar rats where the cortical pO2 decreased to 5% of the pre-ischemic level during occlusion, and then was restored to 160% of the pre-ischemic level after reperfusion. 40
Rather than static entities, mitochondria function independently: they move within cells, form functional relationships with other mitochondria or organelles and structures, and undergo fission and fusion to meet the metabolic needs of cells.2,3,41–49 Mitochondrial fission must be balanced by mitochondrial fusion and vice versa under steady-state conditions to maintain ATP synthesizing capacity in proportion to cellular functions. Evidence from electron microscopy has shown that I/R resulted in detectable mitochondrial disruption and disintegration in VSM whereas mitochondria in adjacent endothelium and adventitia showed enhanced presence of mitochondria by visual observation. Although mitochondria in VSM are often clustered, mitochondria in endothelium normally occur singly. However, following I/R we observed that endothelial cells were often in close proximity to other mitochondria of similar size, which indicated fission events to promote increased mitochondrial numbers. Despite the loss of mitochondria in some VSM cells, adjacent VSM cells in the same artery often showed normal morphology and numbers. Thus, I/R effects are heterogeneous within the same cell type within the same artery. CONTRA arteries showed no generalized changes in mitochondrial features and mitochondria in all cell layers were similar in appearance to Sham and Naïve MCAs. Increased mitochondrial numbers per field was observed by Xie et al. in cortical tissues isolated from male rats with a peak occurring at 72 h after ischemic insult compared with the sham group. 50 Similar to our findings, they observed mitochondria with intact membranes as well as individual mitochondria with mild swelling in cortical tissue from male rats 24 h and 72 h after ischemia compared with sham animals. 50 Previously, we found an increased mtDNA/nDNA ratio in isolated MCAs 48 h after tMCAO as well as in rat primary cortical neurons exposed to 3 h oxygen glucose deprivation (OGD) [mimicking in vitro stroke conditions] and 12 h reoxygenation.14,51 In addition, the time course of the mtDNA changes in the cortical tissue of male rats after I/R and sham operation was determined by Xie et al. 50 ; mtDNA content increased simultaneously with reperfusion time, peaking at 72 h compared with the sham group. Interestingly, they found a reduction in mtDNA content 7 d after I/R, which was still greater than the control group. 50 These findings and observations confirm the presence of a dynamically changing mitochondrial milieu following I/R.
To determine the involvement of fission and fusion in the mitochondrial biogenesis, we investigated the expression of the dynamin-related protein-1, located on the cytoplasmic side of the outer mitochondrial membrane, which is one of the key mediators of mitochondrial fission. Activation of DRP-1 by phosphorylation in IPSI MCAs is suggestive of greater fission rate and mitogenesis. We and others have observed greater mitochondrial biogenesis and DNA content after ischemic or hypoxic insults in vascular and neuronal cells and tissue.2,14,44,46 Because I/R, or the imbalance between mitochondrial fission and fusion, may have contributed to the mitochondrial changes seen in IPSI MCAs, we extended our previous study to investigate the vascular expression of Mfn-2 and OPA-1, two of the most important fusion proteins. Mfn-2 plays an important role in the maintenance of the mitochondrial network, whereas OPA-1 is an intra-mitochondrial protein responsible for preserving the mitochondria, especially the cristae structure; the loss of these proteins might lead to alterations in both mitochondrial structure and function.41,42,44–47,49,52,53 We found significantly decreased vascular expression of Mfn-2 and OPA-1 after I/R compared with Sham and Naïve arteries. This decreased Mfn-2 expression might lead to inhibited or silenced fusion resulting in mitochondrial fragmentation and increased sensitivity of mitochondria in VSM of IPSI MCAs to apoptotic stimuli. Others have observed decreased Mfn-2 expression when using hypoxic HT22 cells as well as in a mouse tMCAO model. 47 Additionally, several studies have found increased NO synthase activity resulting in excess NO levels after I/R that might lead to reactive nitrogen species formation and thus may cause changes in the mitochondrial structure and initiation of mitochondrial fission in cultured cells and in vivo from mice exposed to tMCAO.2,3,14,54–56 Mitochondrial fission may be an asymmetric process, as shown in the electron microscopy images, resulting in a mitochondrion with maintained, normal structure adjacent to a fission partner with profound signs of damage. Fission and mitoptosis might represent novel cell defense mechanisms to reduce further mitochondrial damage by increasing the number of initial mitochondria and reducing the number of damaged mitochondria. However, the consequences of this increased biogenesis remain unclear. The increased mtDNA levels, measured by Xie et al. were accompanied by increased mRNA levels of mitochondrial biogenesis factors such as PGC-1α, NRF-1, TFAM, and increased levels of COX-I and COX-IV proteins in both whole cell lysate and mitochondrial fraction of cortical tissue homogenates 72 h after ischemic stress when compared with the control group. 50 Furthermore, they found that the citrate synthase activity was increased 24 h, 72 h, and 7 d after I/R, compared with the sham and non-reperfusion groups. 50 In our study, we observed a generalized increase in the citrate synthase levels of both the operated and sham animals compared with Naïve rats. Overall, IPSI arteries exhibited a slightly greater citrate synthase expression compared with CONTRA and Sham groups, although it was not statistical significant. Further studies are needed to determine the activity of this enzyme in isolated arteries. A positive correlation between the increased citrate synthase activity, mitochondrial number, and mtDNA has been reported by Xie et al. after I/R, suggesting an increased mitochondrial function. 50
We investigated whether stressed mitochondria are functional by measuring mitochondrial OCR in isolated MCAs. We used 24-well microplates in the OCR experiments and within a plate, 20 wells contained arterial samples and the remaining four wells were used as background. We were not able to perform simultaneous surgeries on rats to fill up the non-reusable, 24-well plates with freshly isolated arterial samples, which resulted in ±6 h deviation from the 48 h reperfusion time. We determined mitochondrial OCR in corresponding Naïve and Sham MCAs, and then measured the changes in the OCR of IPSI and CONTRA mitochondria, which were interpreted as changes relative to the values in the Naïve group. Interestingly, mitochondrial OCR was greater in Sham compared with Naïve MCAs, suggesting a possible side effect of the anesthesia, as both gaseous and injectable anesthetics can alter mitochondrial function in both experimental and clinical settings.38,57–62 Overall, we found a significantly greater OCR in male IPSI MCAs compared with CONTRA, Naïve, and Sham groups resulting in a greater non-mitochondrial respiration, proton leak, ATP production, basal respiration, maximal respiration, and spare capacity. Our results are consistent with other studies that showed increased citrate synthase activity after I/R in cortical tissues. Moreover, the spare respiratory capacity allowed us to estimate the ability of mitochondria to cope with a high energy demand when required to produce more ATP via oxidative phosphorylation.
The complexity of mitochondrial quality control, biogenesis, and function has been shown by a variety of studies. Marzatico et al. 63 investigated mitochondrial metabolism and enzymatic activity in isolated mitochondria from different brain areas of control and sham-operated rats and from rats exposed to subarachnoid hemorrhage. They found that mitochondrial respiration was affected by both subarachnoid hemorrhages and location in the brain. Recently, Andrabi et al. reported a reduced mitochondrial respiratory control ratio and mitochondrial oxygen consumption 24 h after I/R. 64 The difference in the mitochondrial function between our results and those of Andrabi et al. might be due to experimental factors. Several studies have demonstrated that mitochondrial function and numbers are reperfusion time-dependent, tissue specific, and show substrate-dependency. The reperfusion period in our study was twice that used in the Andrabi study indicating a time-dependent difference in the studies. Also, there was a substantial difference in the samples; isolated MCAs from rats were used in our experiments, whereas Andrabi et al. used isolated mitochondria from the rat, frontal cortex.
We also found increased non-mitochondrial respiration in IPSI MCAs especially compared with Naïve animals, suggesting increased oxidative stress and/or increased activity of oxygen consuming enzymes such as cyclooxygenases, NADPH oxidase, or nitric oxide synthase.24,65 We and others have found that levels of these enzymes, which produce ROS as well as other products, are increased in various models of experimental strokes. 24 While beyond the scope of the present study, we intend to investigate the processes responsible for enhanced non-mitochondrial OCR after tMCAO in future studies.
The role of proton leak in ROS formation and elimination is controversial: it has been reported that increased proton leak might contribute to cardiac protection by decreasing overall ROS production, whereas others have speculated that increased ROS production might lead to increased proton leak resulting in decreased ROS levels.11,55,66–77 Under physiological conditions, mitochondrial respiration efficiently produces high energy with modest ROS output. It has been shown that I/R may lead to increased mitochondrial membrane potential (ΔΨm), resulting in hyperpolarization and mitochondrial ΔΨm-dependent ROS formation.66,68,72,73 ΔΨm has been reported to be a regulator of ROS production via changes in ΔΨm such as hyperpolarization, which might contribute to an exponential increase in ROS production at Complexes I and III.66,68,72,73 Additionally, mitochondrial oxidative phosphorylation is diminished as the result of limited electron transport under ischemic conditions. Conversely, ischemia might induce depolarization and ATP depletion resulting in energy failure and a switch from oxidative phosphorylation to glycolysis. Andrabi et al. have reported reduced mitochondrial membrane potential and low TMRE fluorescence of cortical mitochondria after tMCAO, similar to our previous findings in isolated MCAs on the side of ischemia.14,64 Any of the described scenarios could lead to Ca2+ uptake and mitochondrial swelling, and alter the phosphorylation state of the Complex proteins. We have shown the consequences of ischemia and the effects of reperfusion. Although reperfusion restores blood flow, it contributes to further tissue deterioration and mitochondrial hyperpolarization.14,74 Both mitochondrial function and ΔΨm were restored within minutes after reperfusion, although this might be followed by hyperpolarization resulting in additional ROS production. However, the activities of Complexes I and IV have been reported to be maximally inhibited at 24-h reperfusion contributing to a collapsed ΔΨm and cell death. 72
We and others have found a decreased vasoreactivity in the presence of endothelium-dependent and endothelium independent vasodilator and vasoconstrictor agents such as acetylcholine, bradykinin, sodium nitroprusside, and serotonin of isolated, pressurized arteries from animals exposed to I/R or hypoxic injury compared with the control group.14,78,79 Interestingly, we have observed preserved mitochondrial mediated vasodilation of pressurized male MCAs in the presence of DZ, a mitochondrial KATP opener.13,14 We have elucidated how the I/R-mediated changes in mitochondrial structure and bioenergetics influence cerebrovascular function by determining the frequency of Ca2+ sparks, one of the subgroups of local Ca2+ events, in isolated MCAs in the presence and absence of DZ. Changes in the intracellular Ca2+ determined both tissue perfusion and vascular responses.11,23,76,79–81 We found a significantly lower Ca2+ sparks activity during baseline in IPSI MCAs compared with CONTRA, Sham, and Naïve arteries, and this effect is probably related to the observed structural damage to VSM mitochondria that we observed in the IPSI MCAs. However, treatment with DZ resulted in an eight fold increase in the Ca2+ sparks activity in IPSI MCAs, suggesting that a sufficiently large stimulus can still elicit the significant mitochondria-SR interactions even in stressed VSM. Nonetheless, the generation of substantial Ca2+ sparks activity after tMCAO in the presence of DZ still fails to elicit the normal degree of VSM-dependent dilation in normal MCAs. 14
There are several limitations of our study. For example, we studied animals at 48 h following tMCAO and thus could not document the time course of changes in mitochondrial function. We anticipate examining other time-points in future studies. In addition, technical limitations made it impossible to investigate responses of individual cells types rather than whole MCAs in our examinations of mitochondrial respiration and western blotting. We considered studies on MCAs following endothelium denudation, but were concerned that this approach would lead to damaged arteries and abnormal values of OCR. However, the Ca2+ sparks activity is specific to VSM and we were able to observe a clear relationship between the presence of structural damage in mitochondria and reduced frequency of calcium related events. Lastly, we could not count mitochondrial numbers and volume in the cells of the MCAs. A complete picture of the mitochondrial status of each of the MCAs studied would be ideal, but the time expenditure and extra equipment needed is currently beyond the scope needed for this study. Nonetheless, using strict criteria in VSM we could quantify obvious structural damage to mitochondria.
In conclusion, our data indicate that I/R initiated mitochondrial fission results in major changes in mitochondrial morphology and function in VSM and endothelial cells of IPSI MCAs. While the potential value of mitochondria as a therapeutic target in MCAs remains unclear, we believe our study contributes to a better understanding of the complex and cell specific mitochondrial processes present in the cerebral vasculature in response to transient ischemic stress.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health grants HL-077731 and HL093554 (DWB) and NS094834 (PVK), Louisiana Board of Regents Endowed Chairs for Eminent Scholars program (DWB), American Heart Association Post-Doctoral Fellowship Grant 15POST23040005 (IR), American Heart Association AWRP Summer 2016 Scientist Development Grant 17SDG33410366 (IR), and American Heart Association National Center NCRP Scientist Development Grant 14SDG20490359 (PVK).
Acknowledgements
The authors thank Nancy B. Busija, M.A., CCC-SLP for critically reading and editing the manuscript, Dan Liu M.S. and Catherine McNulty B.S. for technical help, and Ken Grant B.S. of the Cellular Imaging Shared Resource at Wake Forest Univ. Health Sciences for assistance with electron microscopy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
IR and DWB contributed to the experimental concept and design; IR, IM performed experiments; IR, IM, SW, GC, PVGK and DWB took part in the data analysis; IR and DWB was involved in the interpretation of result; IR and DWB prepared figures; IR, IM, SW, GC, PVGK, and DWB edited and approved the final version of the manuscript.
