Abstract
Regulatory changes in cytokine permeation across the blood—brain barrier (BBB) may have crucial roles in central nervous system (CNS) autoimmune disease. Accordingly, we examined the interactions of interleukin (IL)-15 with the cerebral vasculature after induction of experimental autoimmune encephalomyelitis (EAE). In contrast to the influx of 125I-IL15 from blood to the CNS in normal mice and the persistence of IL15 influx in the spinal cord of EAE mice, influx was reduced in the EAE brain. Analyses of disappearance kinetics, FITC (fluorescein isothiocyanate)-albumin space, and delivery of IL15 by
Introduction
Interleukin (IL)-15 is a 14-kD pleiotropic cytokine in the 4-α-helix bundle cytokine family, which also includes IL2, IL3, IL4, IL6, IL21, granulocyte-macrophage colony-stimulating factor, granulocyte colony-stimulating factor, erythropoietin, growth hormone, and prolactin (Budagian et al, 2006). Interleukin-15 has both proinflammatory and antiinflammatory effects and is a potent antiapoptotic cytokine and T-cell chemoattractant (McInnes et al, 1996; Wilkinson and Liew, 1995). The roles of IL15 and its receptors in vascular function and neuroinflammation are supported by their presence in the endothelia (Nilsen et al, 1998), microglia, and brain in general (Hanisch et al, 1997; Kurowska et al, 2002). The constitutive expression of IL15 in neurons and the induced expression in glial cells (Maslinska, 2001) suggest its crucial role in normal brain function and in central nervous system (CNS) development. Results from studies with IL2 knockout mice suggest that IL15 might be involved in hippocampal neurogenesis (Beck et al, 2005). Interleukin-15 induces dose-dependent increases of non-REM (rapid eye movement) sleep in rabbits after intracerebroventricular injection (Kubota et al, 2001), but the mechanism is not clear.
Interleukin-15 is a crucial target in the treatment of inflammatory and autoimmune diseases (Baslund et al, 2005; McInnes and Gracie, 2004; Waldmann, 2004). In autoimmune diseases, IL15 seems to be detrimental as it inhibits self-tolerance mediated by activation-induced cell death and induces other proinflammatory cytokines, including tumor necrosis factor-α (TNF-α) and IL1β (McInnes et al, 1997; McInnes and Schett, 2007). The concentration of IL15 is increased in both the serum and cerebrospinal fluid (CSF) of patients with multiple sclerosis, and the high serum/CSF ratio in these patients is believed to suggest a predominant role of the peripheral production of IL15 in immunopathology (Rentzos et al, 2006). Both IL15 and IL2 seem to worsen rheumatoid arthritis (Baslund et al, 2005).
Daclizumab (Zenapax, Hoffmann-La Roche Inc., Nutley, NJ, USA), an antibody against IL2, has shown success in a recently completed phase II clinical trial of relapsing and remitting multiple sclerosis after monthly intravenous injections (Rose et al, 2007). This provides a strong basis for determining the interactions of IL15 with the blood—CNS barriers in the established animal model for multiple sclerosis—experimental autoimmune encephalomyelitis (EAE).
The blood—brain barrier (BBB) and blood—spinal cord barrier constitute a regulatory interface that allows the selective entry of certain peptides and cytokines, yet bars many others. Experimental autoimmune encephalomyelitis is associated with a dynamic pattern of BBB disruption that shows specificity in different brain and spinal cord regions during the course of the disease. The peak of neurologic deficits is associated with the maximal increase of BBB permeability to vascular markers, and there is an enhancement of the transport system for TNF (Pan et al, 1996). Tumor necrosis factor is a strong activator of the IL15 system in cerebral endothelia. It increases IL15 by reducing its protein turnover initially and upregulating mRNA expression afterwards, and it also increases the production of IL15Rα at both mRNA and protein levels (Yu C
The transport of peptides and cytokines across the BBB can occur in either direction, involving an influx from blood to the CNS and an efflux from the CNS to blood. For the latter, there are known efflux transporters present in cerebral endothelia and perivascular astrocytes. The well-studied efflux transporter, P-glycoprotein (Pgp), is a 12-transmembrane protein located at the apical/luminal surface of endothelial cells and belongs to the ABC (adenosine triphosphate-binding cassette) family of energy-dependent transporters, which has a broad spectrum of substrates. Regulation of Pgp expression and activity occurs in inflammation (Yu et al, 2007, 2008). T-helper-1 and monocytes/macrophage cytokines induce robust Pgp upregulation in mixed glial culture (Lisak et al, 2009). Neither IL15 itself (Pan et al, 2008c) nor the related cytokine IL2 (Waguespack et al, 1994) enters the CNS by a saturable transport mechanism in the basal state, although neuroinflammation induced by lipopolysaccharide increases the permeation of IL15 from blood to the CNS (Pan et al, 2008c). Interleukin-2 leaves the brain through a brain-to-blood transport system (Banks et al, 2004), yet the fate of IL15 in CSF has not been studied. Therefore, we determined the regulatory changes in IL15 permeation from blood to the CNS (a summation or balance of influx and possible efflux if IL15 is a substrate for ABC transporters), and also measured the brain-to-blood transport in EAE mice.
Materials and methods
Experimental Autoimmune Encephalomyelitis Induction
The animal protocol was approved by the Institutional Animal Care and Use Committee. Eight-week-old female SJL/J mice (Jackson Laboratories, Bar Harbor, ME, USA) were injected subcutaneously with an emulsion of proteolipid protein fragment, PLP139–151 (AnaSpec, San Jose, CA, USA), along with adjuvant. The dose of PLP 139–151 was 80 μg per mouse, delivered subcutaneously to the lower flank area, evenly divided into three spots. The initial experiments used RIBI adjuvant (RIBI Immunochem Research, Hamilton, MT, USA); as it is no longer available commercially, the later experiment used the Sigma Adjuvant System (Sigma, St Louis, MO, USA), which is an incomplete Freund's adjuvant similar to RIBI, and eventually complete Freund's adjuvant (CFA) (DIFCO Laboratories, Detroit, MI, USA) with two pertussis toxin (List Biological Laboratories, Campbell, CA, USA) boosters (200 ng in 200 μL PBS (phosphate-buffered saline) per mouse, one on the day of immunization and another 48 h later). PLP + RIBI produced a uniform and monophasic EAE with an onset of weakness on days 9 to 10, a peak of disease on day 14 with EAE scores of 2 to 3, and complete behavioral recovery by day 23 (Pan et al, 1996). PLP + CFA with two pertussis toxin boosters induced a biphasic distribution of the EAE score, with the first peak occurring on day 14 and the second peak about day 20. On the basis of preliminary data of the behavioral development of EAE, the mice were studied 2 days before the emergence of the classical symptoms of weakness of tail and hind limbs. Different batches of mice were used for the different studies described below.
Blood-to-Central Nervous System Influx of Interleukin-15 After Intravenous Injection
Carrier-free recombinant mouse IL15 (R&D Systems, Minneapolis, MN, USA) was labeled with 125I (Perkin Elmer, Waltham, MA, USA) using the chloramine-T (Sigma) method. Bovine serum albumin (BSA; Sigma) was labeled with 131I, also using the chloramine-T method. The iodinated proteins were separated from free iodine by elution on Sephadex G-10 columns (Sigma). The specific activity of 125I-IL15 was ~120Ci/g, and that for 131I-albumin was ~60 Ci/g.
Multiple-time regression analysis (Kastin et al, 2001) was carried out to quantify the influx of IL15 from blood to the CNS. Two groups of mice were studied: EAE mice and adjuvant-only controls (RIBI adjuvant), both at 7 days after induction (
Brain Distribution of Fluorescein Isothiocyanate-Albumin After Intravenous Delivery
Fluorescein isothiocyanate (FITC)-albumin (Sigma) solution (5 mg/mL) was freshly prepared in oxygenated modified Zlokovic's perfusion buffer as follows (g/L): NaCl (7.19), KCl (0.3), CaCl2 (0.28), NaHCO3 (2.1), KH2PO4 (0.16), MgCl2 6H2O (0.37),
Degradation Assays
Our previous study using size-exclusion chromatography showed that 125I-IL15 remains stable in the blood and brain 10 mins after intravenous injection (Pan et al, 2008c). Thus, the stability of 125I-IL15 at a longer duration was determined here. Thirty minutes after intravenous injection of 125I-IL15 and 131I-albumin as described above, arterial blood was collected into a tube containing protease inhibitor cocktail (Thermo Fisher Scientific, Rockford, IL, USA) on ice, and the mice were immediately decapitated. Whole brain was homogenized on ice in PBS in the presence of protease inhibitor cocktail, and supernatant was collected. The processing control involved the collection of blood and brain to which radioisotopes were added
In situ Brain Perfusion
To determine whether the ILl5-specific receptor, IL15Rα, and the common γ-chain receptor, IL2Rγ, have a role in the permeation of IL15 from blood to the CNS in EAE mice induced by PLP with the Sigma Adjuvant System,
Efflux Transport of Interleukin-15 After Intracerebroventricular Injection
In the first study to determine whether the efflux of IL15 occurred by a regulatory process in normal mice, four groups of urethane-anesthetized 2-month-old male B6 mice (Jackson Laboratories) were studied (
In the second study to determine whether EAE mice show an altered brain-to-blood transport of IL15, three groups of mice were studied (
Real-time Reverse Transcriptase-PCR
Two groups of mice were compared: control and EAE mice on day 7 after induction by PLP+RIBI (
Results
Abolished Influx of Interleukin-15 and Albumin from Blood to Brain in EAE Mice
In control SJL/J mice, 125I-IL15 had a significant influx from blood to brain, with a Ki of 0.214 ± 0.089 μL/g min and a Vi of 14.79 ± 1.17 μL/g. In EAE mice (induced by PLP + RIBI and studied before the emergence of symptoms), the Ki was −.140 ± 0.134 μL/g min and Vi was 17.74 ± 1.53 μL/g. The significant (
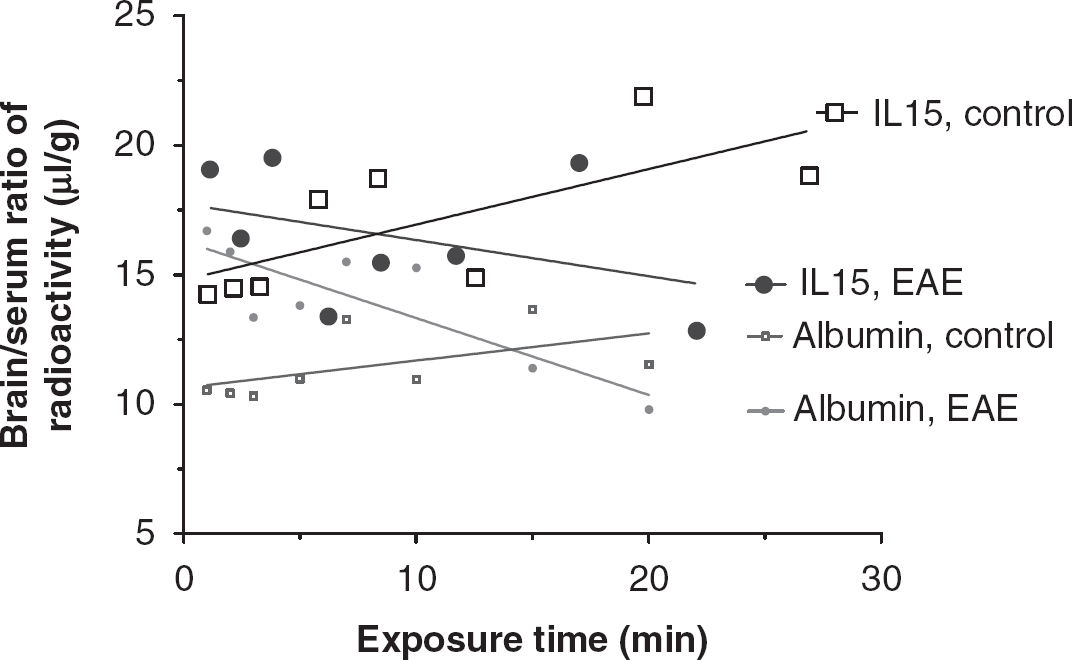
The influx of 125I-IL15 from blood to the brain was determined in groups of control and EAE mice receiving PLP139–151 and RIBI adjuvant (
Decreased Influx of Albumin But Not IL15 From Blood to Spinal Cord in EAE Mice
In the control spinal cord of the SJL/J mice, the influx rate Ki of 125I-IL15 was 0.426 ± 0.045 μL/g min, and the initial volume of distribution Vi was 9.26 ± 0.59 μL/g. In EAE mice, Ki was 0.267 ± 0.090 μL/g min, whereas Vi was 11.15 ± 1.10 μL/g. Although both groups had significant regression lines, the difference between them was not significant. The spinal cord influx of IL15 differed from the pattern in the brain, but the influx of 131I-albumin was similar to that observed in the brain. The control group had a Ki of 0.274 ± 0.073 μL/g min and a Vi of 6.27 ± 0.74 μL/g. The EAE group had a Ki of −.053 ± 0.085 μL/g min and a Vi of 10.40 ± 0.86 μL/g (Figure 2). The difference in albumin influx in the spinal cord between the control and EAE mice was significant (
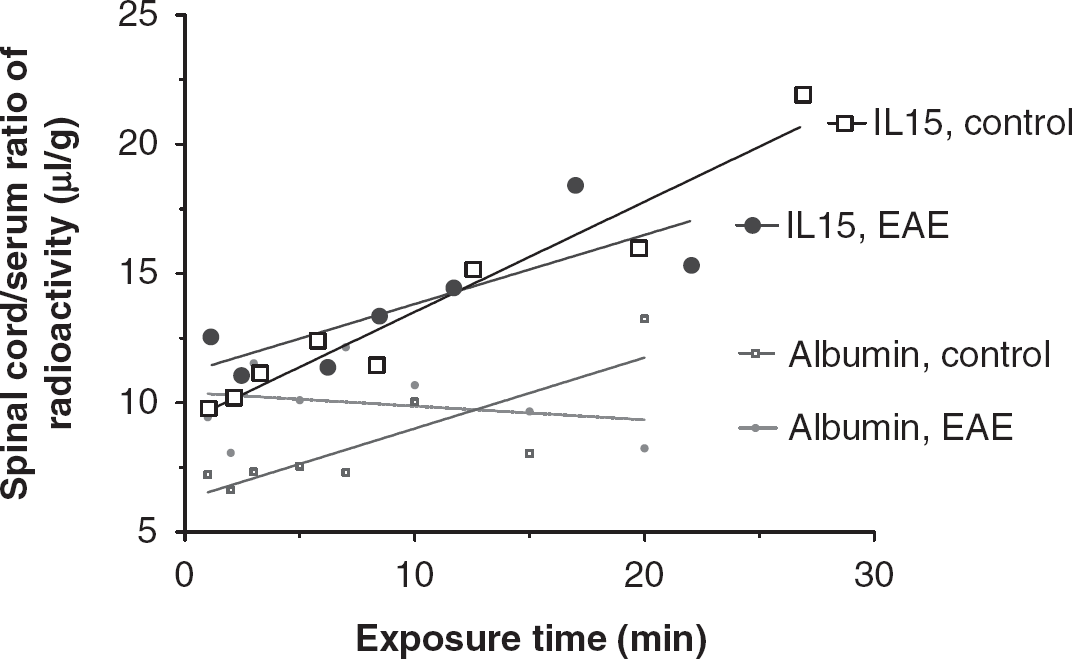
The increased influx of 125I-IL15 from blood to the spinal cord in EAE mice (PLP + RIBI) did not differ significantly from that in the control group. The influx rate of 131I-albumin, however, was significantly reduced in EAE mice.
Regional Differences in IL15 Permeation and the Level of IL15 Expression In Situ
Major regional differences in the BBB permeation of 125I-IL15 were observed in control mice. The highest Ki was observed in the lumbar spinal cord (0.703 ± 0.125 μL/g min). There were several areas with a moderate Ki of ~0.3 μL/g min, including the hippocampus, hypothalamus, cerebellum, and cervical and thoracic spinal cord. A low permeation of IL15 in descending order was observed in the ponsmedulla, midbrain, cerebral cortex, and striatum. Interestingly, the striatum, which had the lowest influx rate (0.092 ± 0.052 μL/g min) in controls, maintained a positive but minimal influx in EAE mice, whereas all other brain areas showed negative Kis attributed to rapid efflux. In the thoracic and lumbar spinal cord, where BBB disruption was maximal in EAE mice, there was a significant reduction in the influx of 125I-IL15 that still maintained positive values (Figure 3A).
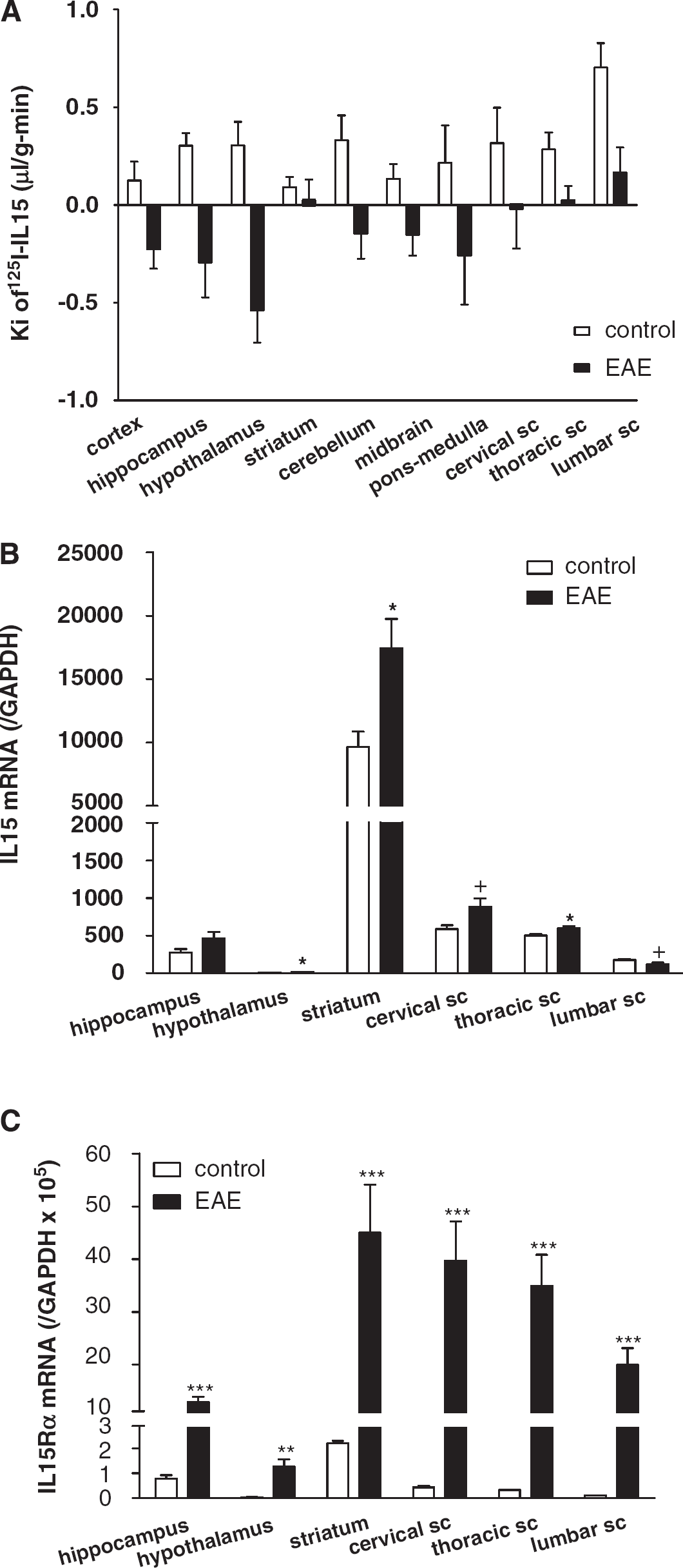
Regional variation in BBB permeability to 125I-IL15 and levels of
In normal mice, the relationship between BBB permeability and
Vascular Space in the Experimental Autoimmune Encephalomyelitis Mice
In EAE mice induced by PLP + CFA, there was an increase in the density of distribution of FITC-albumin in cerebral microvessels. More microvessels were delineated by FITC-albumin after intravenous delivery. In the striatum of control mice that received only adjuvant either 11 or 29 days earlier, there was a marked regional variation in the filling of microvessels. The EAE mouse 11 days after induction (asymptomatic) showed a mild increase in FITC-albumin fluorescence intensity (arrow). The EAE mouse 29 days after induction (with a score of 0, having just recovered from EAE) showed a major increase of delineated microvessels and a greater fluorescence intensity of FITC-albumin (Figure 4A). This suggests hyperemia and an increase in vascular space. In the hypothalamus, there was a moderate increase in the capillary density of postcapillary venules at the external rim of the median eminence on both days 11 and 29 after EAE induction, without apparent changes in the microvessels in the arcuate nucleus (Figure 4B). In none of these mice was there an extravasation of FITC-albumin. This indicates that the BBB remained relatively intact in most regions of the brain.
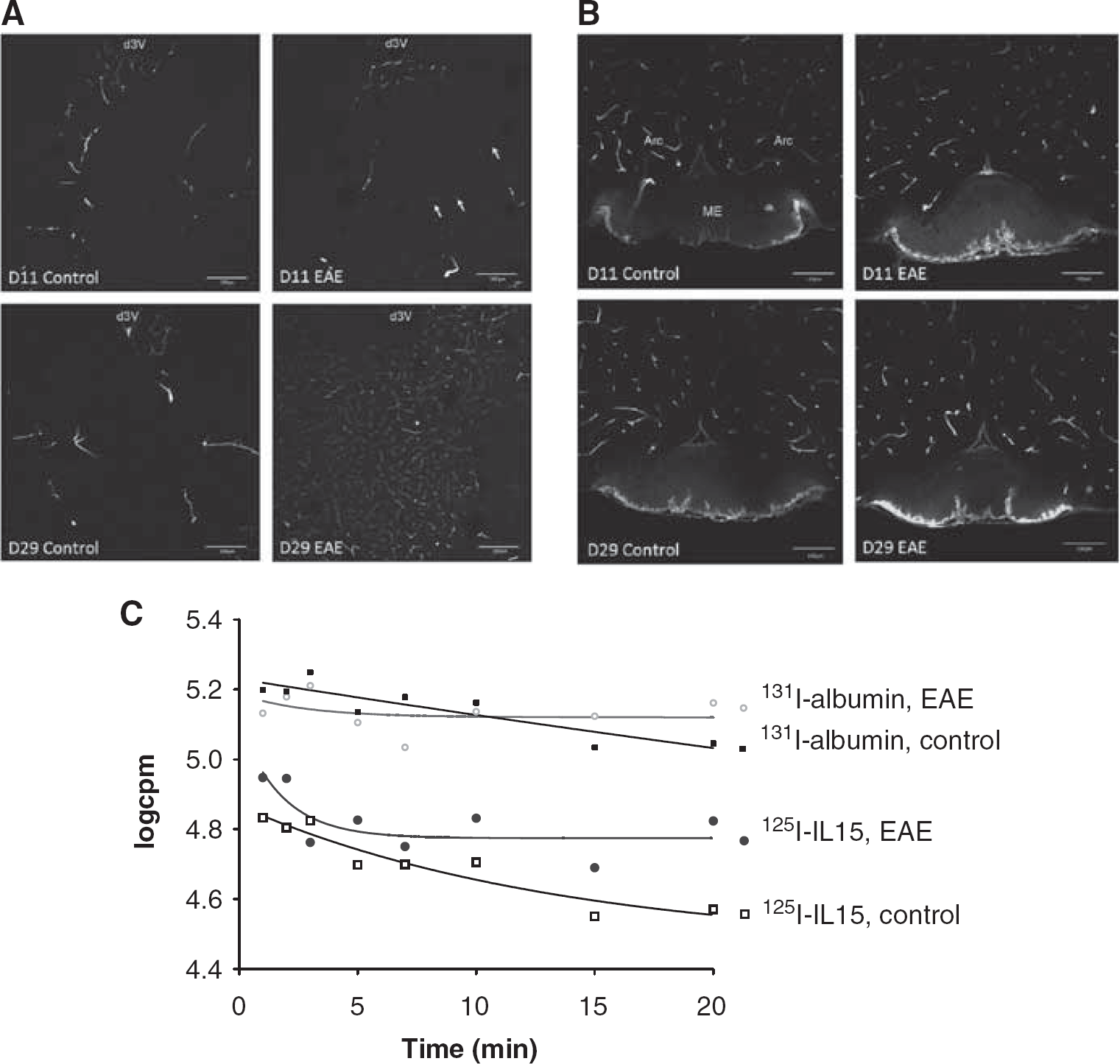
The vascular space of control and EAE mice after induction with PLP and CFA. (
The disappearance half-life of 131I-albumin after intravenous injection differed significantly in EAE and control mice. The F-test showed the best fit of the concentration of 131I-albumin (log c.p.m.) with the following equation for one-phase exponential decay:
Interleukin-15 and Albumin Remained Intact 30 mins After Intravenous Delivery
125I-IL15 coeluted with 131I-albumin in brain homogenates (Figure 5A) and serum (Figure 5B) 30 mins after intravenous injection. As shown in Table 1, at least 80% of the total radioactivity in the brain eluant 30 mins after intravenous injection represents intact 125I-IL15. Some of the degradation occurred during tissue processing, as shown by the result of 90% intact 125I-IL15 in the brain after
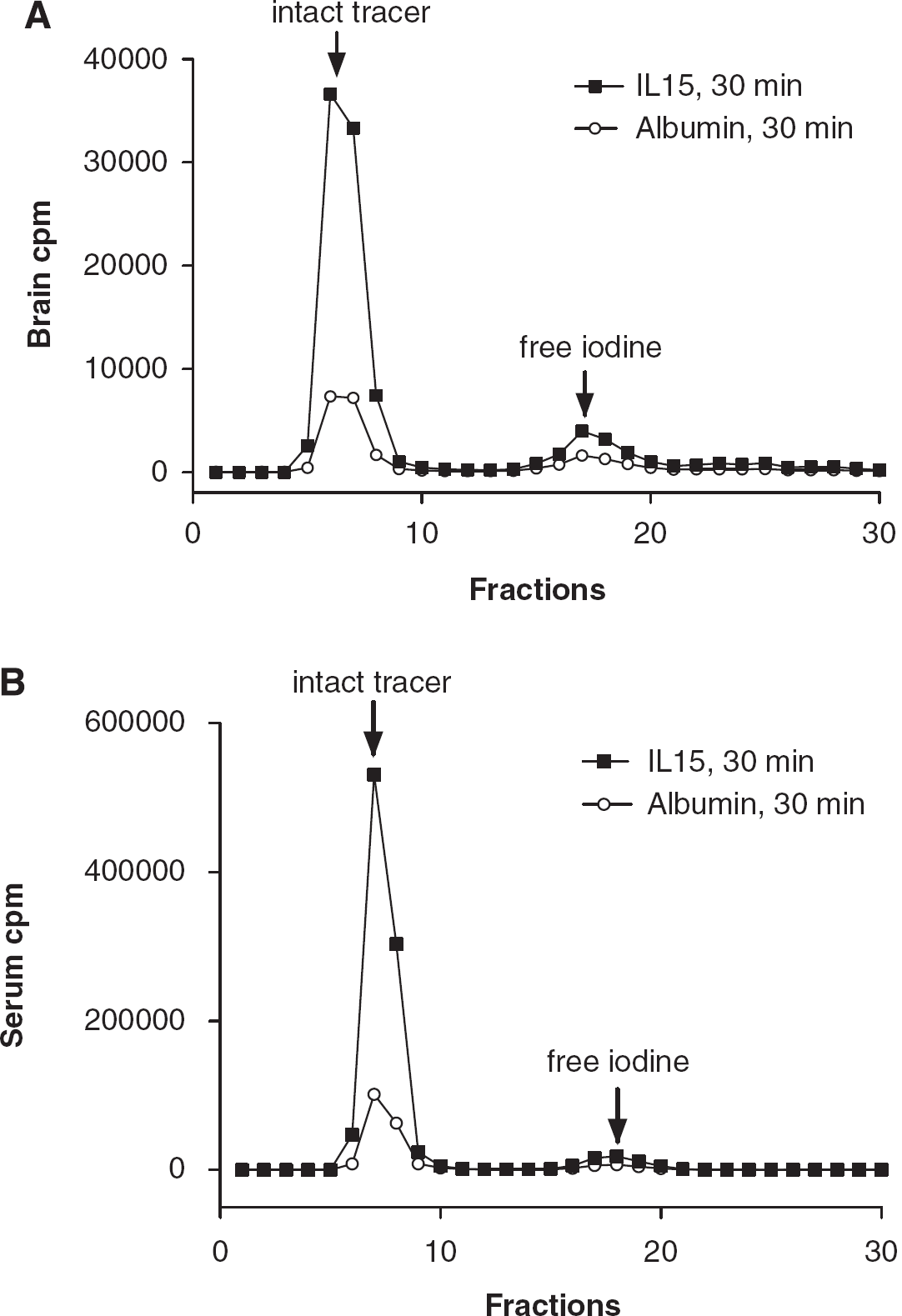
The degradation assay of 125I-IL15 and 131l-albumin. In both brain (
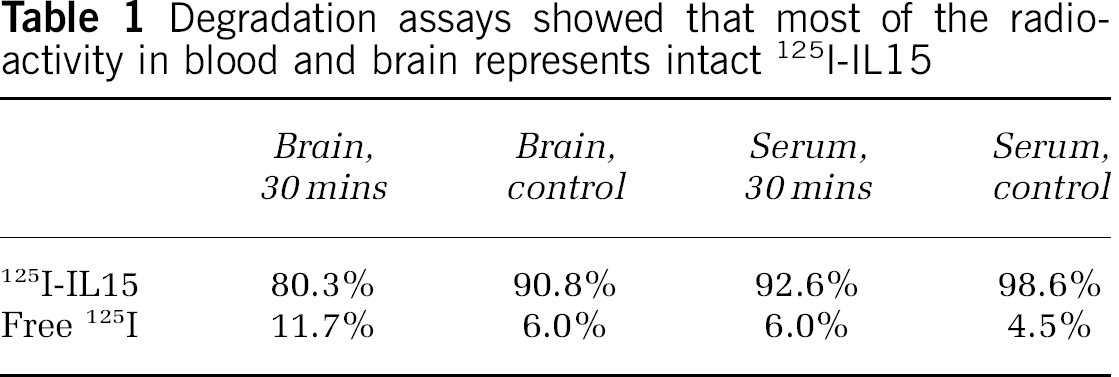
Degradation assays showed that most of the radioactivity in blood and brain represents intact 125I-IL15
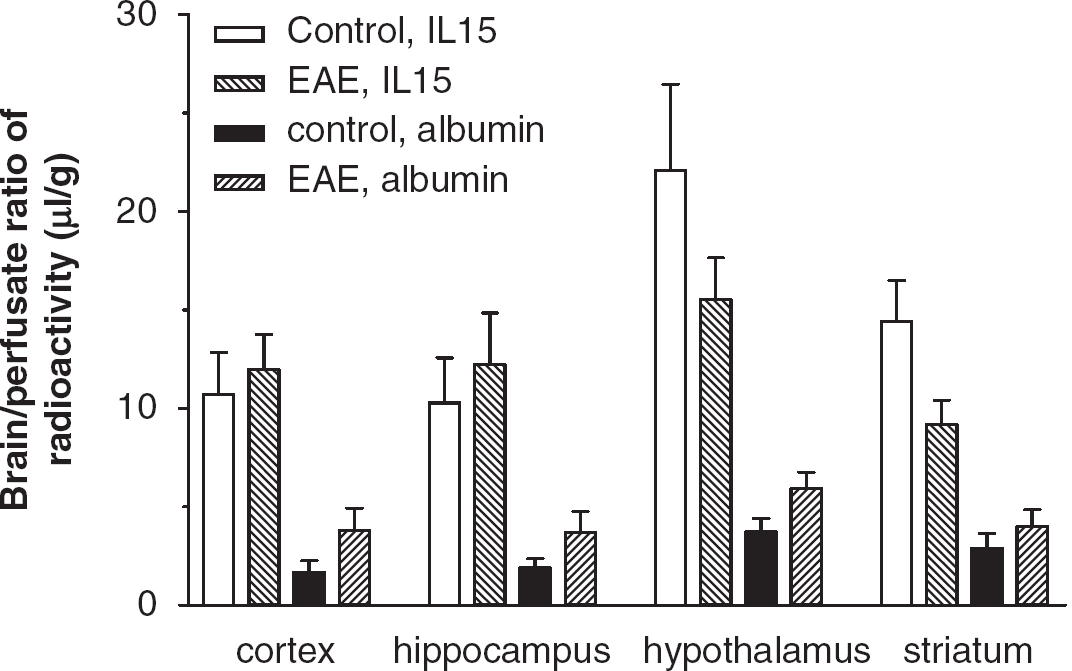
In Situ Brain Perfusion Overrides the Decrease of IL15 Permeation Across the Blood-Brain Barrier
The brain/perfusate ratio of 131I-albumin was higher in EAE mice induced by PLP with the Sigma Adjuvant System as they began to show signs of weakness. This was observed in all brain regions and in the whole brain, but the increase in the adjuvant-only controls was not significant. The brain/perfusate ratio of 125I-IL15 did not show significant changes, despite a tendency toward a reduced uptake in the hypothalamus and striatum (Figure 6). In neither the control nor EAE mice did the coadministration of sIL15Rα or sIL2Rγ (after 1 h of incubation with the radioactively labeled tracers) have a significant effect on the brain uptake of 125I-IL15 or 131I-albumin. The lack of decrease of IL15 uptake in the brain of EAE mice argues against the presence of an efflux transport system. Rather, it supports the possibility that the negative Ki in the multiple-time regression analysis after intravenous injection was caused by the rapid disappearance of IL15 and albumin in blood to reach the cerebral vasculature during the first few minutes, followed by redistribution with backflux.
The Brain-to-Blood Transport of Interleukin-15 in Normal and EAE Mice
In normal mice, the rate of 125I-IL15 exiting the brain could be predicted by the following equation: log10(
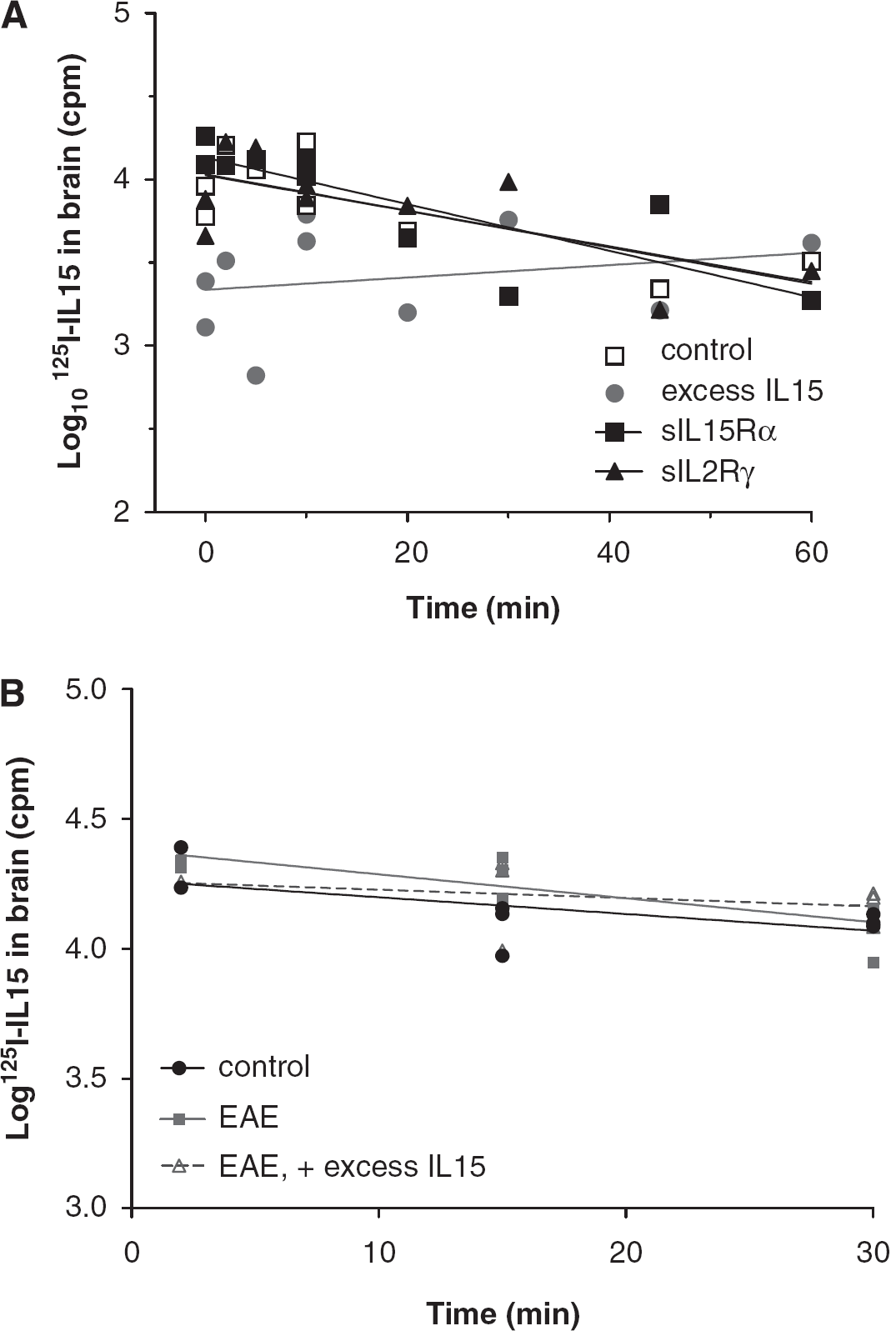
Brain-to-blood transport of IL15, shown by the logarithm of residual radioactivity in the brain was plotted against time after intracerebroventricular injection. (
In EAE mice induced by PLP + CFA and showing early signs of weakness, there was no significant change in the disappearance of 125I-IL15 from the brain in comparison with that in control mice (
Discussion
In this study, we show that the development of EAE was associated with a decreased brain/serum ratio of IL15 and vascular marker albumin over time. The influx rate of IL15 into the spinal cord remained high, despite a decrease in the spinal cord/serum ratio of albumin over time. Multiple-time regression analysis is typically applied to situations in which the blood concentration of a tracer is relatively stable and permeation across the BBB is low, as observed for most nonvasoactive peptides and proteins (Kastin et al, 2001). In situations with a rapid extraction of a blood-borne substance by the CNS, an oscillation of efflux and influx, or extremely unstable blood concentrations, the method may no longer be valid. In the case of EAE mice, the negative Ki seemed to be caused by a rapid filling of the cerebral vascular space within the first 5 mins and by a later redistribution of serum proteins (including albumin and IL15) over time. Although not perfect for this situation, the multiple-time regression analysis reflects dynamic interactions of IL15 and albumin with cerebral vasculature. Therefore, the increase in Vi in the brain was equally as important as the persistent Ki in the spinal cord. This is further supported by results from
Our previous findings of IL15 permeation across the BBB after lipopolysaccharide challenge by intraperitoneal injection showed relatively smaller changes than would be expected from the increase of vascular permeability shown by albumin (Pan et al, 2008c). The results of this study with EAE mice showed an entirely different pattern of altered BBB permeation. As a negative influx rate of 131I-albumin may be caused by a reduction in CNS vascular space resulting from either vasoconstriction or hypoperfusion, we determined the regional distribution of FITC-albumin after intravenous injection. There was no vasoconstriction; rather, EAE mice showed an increase in vascular space, apparent in the striatum. Neither was there extravasation of FITC-albumin indicative of focal BBB disruption. An increased vascular space was probably mainly responsible for this rapid turnover of exogenous albumin. This is also supported by findings that albumin nanoparticles show increased distribution in meningeal and perivascular areas in the lumbar spinal cord (Merodio et al, 2000). In addition, perivascular extravasation of serum albumin, immunoglobulins, and exogenous horseradish peroxidase, are all prominent in the brainstem and cervical spinal cord in EAE (Rabchevsky et al, 1999). Concurrently, laser flow Doppler measurement at the cortical surface did not show a significant change in cerebral perfusion (Pan W, unpublished observations). The Ki for IL15 in the spinal cord remained high, suggesting an increase in the spinal cord uptake of IL15. Therefore, EAE is associated with an increased cerebral vascular space and an accelerated turnover of serum proteins. The altered kinetics of CNS extraction of IL15 from blood have never been observed in other disease models in mice, including neuroinflammation by lipopolysaccharide (Pan et al, 2008d), spinal cord injury (Pan et al, 2003a), brain trauma (Pan et al, 2003b), or stroke (Pan et al, 2006).
As EAE is a heterogenous disease with regional variation in CNS pathology, we determined whether the regional difference of IL15 permeation was related to the level of expression of IL15 and its receptors within the CNS. Owing to technical limitations in protein analyses by western blotting or immunohistochemistry (issues of sensitivity and specificity), mRNA was measured. The changes in IL15 permeation and
An efflux system could be present at the cerebral endothelia, such as the Pgp system at the luminal surface of microvessels (apical surface of the endothelial cells), or on the brain side of the barrier structures. Cytokines, such as TNF, have been shown to increase Pgp activity and facilitate the efflux of its substrates across the BBB (Bauer et al, 2007; Yu et al, 2007). Thus, we further determined the influx of IL15 by intracardial (
Once the blood-borne IL15 reaches the CSF through either the BBB or blood—cerebrospinal fluid barrier, there is an increase in the CSF/blood ratio. However, in the human disorder of multiple sclerosis, which EAE models, there is an increased serum/CSF ratio of IL15 (Rentzos et al, 2006). This decrease in the CSF/serum ratio might suggest brain-to-blood transport. Thus, we determined the fate of IL15 after permeation across the BBB or intrathecal production by intracerebroventricular injection. In normal mice, IL15 exits the brain by CSF reabsorption, as shown by the similar disappearance half-life of IL15 and albumin. Albumin enters the CNS through a variety of routes collectively termed ‘extracellular pathways,’ and the CSF levels of albumin are ~0.5% of serum levels (Banks, 2004). Albumin exits CSF by reabsorption at the granular process in the subarachnoid space, making it a reliable marker of CSF turnover. However, excess IL15 seemed to inhibit the process, with a concurrent reduction of both IL15 and albumin disappearance from the CSF. Results from the use of soluble receptors indicated that brain-to-blood transport was unlikely to be receptor-mediated. Overall, the prolonged half-life of 125I-IL15 and 131I-albumin in the CSF in the presence of excess IL15 suggests altered CSF dynamics. This was changed in EAE mice, in which excess IL15 was no longer effective.
Thus, EAE is associated with drastic changes in the vascular space, with lack of saturation of the blood-to-CNS influx and brain-to-blood transport, and with the transcriptional upregulation of IL15 and its specific receptor IL15Rα in the brain. The results indicate a complex role of the BBB in mediating communication between blood-borne IL15 and the CNS during the development of EAE.
Footnotes
The authors declare no conflict of interest.
