Abstract
Tumor necrosis factor (TNF)-
Keywords
Introduction
Tumor necrosis factor (TNF)-
The leading decoy receptor-type TNFI is etanercept, which is widely used to suppress TNF
Materials and methods
Fusion Proteins
The cTfRMAb-TNFR fusion protein was produced in stably transfected Chinese hamster ovary cells and purified by protein G affinity chromatography as described previously (Zhou et al, 2011a). Etanercept (Enbrel) was purchased from the Pharmacy of the UCLA Hospital. The TNF
Reversible Middle Cerebral Artery Occlusion Model
All experimental animal procedures were conducted according to Animal Welfare Act and the Public Health Service Policy on Humane Care and Use of Laboratory Animals. All of our study protocols were approved by the UCLA Animal Research Committee before the study began, and we were given permission to perform the study. The reversible MCAO mouse model was used, as described by Shimin et al (2009). Adult male C57Bl/6J mice (25 g) supplied by Jackson Labs (Bar Harbor, ME, USA) kept under standardized light/dark (12 h), temperature (25°C) and humidity (70%) conditions were used for the MCAO model. Mice were anesthetized with isoflurane (4% for induction and 2% for maintenance) in 30% O2. Body temperature was continuously monitored and maintained constant at 37°C throughout the surgical procedure using a Harvard thermal blanket with a rectal probe (Harvard Apparatus, Inc., Holliston, MA, USA). Mice were placed in a supine position; a midline incision was made in the neck and the common carotid artery and external carotid artery (ECA) were exposed. The branches of the ECA (superior thyroid and occipital arteries) were electrocoagulated. After occlusion of the common carotid artery with a microclip, the ECA was ligated distally to the cranial thyroid artery and an incision was made in the ECA. A silicon rubber coated 6-0 nylon monofilament (6021; Doccol Corp., Redlands, CA, USA) was inserted into the ECA and gently advanced 9 to 11 mm until resistance was felt for occlusion of the middle cerebral artery. The filament was secured in placed by ligation for duration of 60 minutes after which the nylon filament was carefully withdrawn to allow for reperfusion and the neck incision was sutured. Mice were euthanized either 23 hours or 7 days after 60-minute occlusion. Some mice died prematurely and were not included in the data analysis. No significant difference between the mortality rates among different treatment groups was observed. The average mortality across all the groups was 7% for mice subjected to 60-minute occlusion and 23 hours of reperfusion. The mice that died prematurely were not used to compute stroke volumes or neural deficits. The measurements of infarct volume and neural deficit were determined by an observer blinded to treatment group.
Drug Treatment Groups
Mice subjected to MCAO were divided into three treatment groups: (1) saline, (2) etanercept (1 mg/kg), and (3) cTfRMAb-TNFR (1 mg/kg). Drug treatment was administered intravenously via the tail vein with a 0.3-mL syringe and a 29-g needle at 45 or 90 minutes after arterial occlusion. The protein concentration in the intravenous injection solution was 0.25 mg/mL in ABS buffer and a total volume of 100
Neurologic Deficit Scoring
Before euthanasia at 1 day or 7 days after arterial occlusion, animals were monitored for neurologic deficit, and scored as follows: 0, no deficit; 1, failure to extend the contralateral forepaw fully; 2, intermittent circling; 3, sustained circling without moving forward; 4, walks only when stimulated with decreased level of consciousness.
Infarct Volume Calculation
After euthanasia, the brain was removed and cut into 2-mm coronal sections with a mouse brain matrix (Kent Scientific Co., Torrington, CT, USA). The brains were stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC; Sigma-Aldrich Inc., St Louis, MO, USA) and fixed overnight in 10% buffered formalin (Sigma-Aldrich Inc.). The sections were scanned with a UMAX PowerLook III flatbed scanner (UMAX Data Systems, Inc., Hsinchu, Taiwan) and the images processed in Adobe Photoshop. The area of either hemispheric or cortical infarct in each brain section was computed from the difference between the area of the contralateral hemisphere and the area of ipsilateral noninfarcted brain using NIH Image J software 1.62 (National Institutes of Health, Bethesda, MD, USA). Total infarct volume (mm3) was computed from the sum of infarct area for all sections from the same brain multiplied by the total section thickness (8 mm). Subcortical infarct volume was calculated as the difference between the hemispheric and cortical infarct volumes.
Statistical Analysis
Data are represented as mean±s.e.m., and statistical analysis was performed using GraphPad Prism 5.0 (GraphPad Software San Diego, CA, USA). One-way analysis of variance with Bonferroni's correction (for more than two groups) or Student's
Results
The TTC stains of coronal sections of mouse brain removed 24 hours after a 1-hour reversible MCAO are shown in Figure 1. The scans from a representative mouse in the saline treatment group, the etanercept treatment group, and the cTfRMAb-TNFR fusion protein treatment group are shown in Figures 1A, 1B, and 1C, respectively. The mean±s.e.m. of the stroke volume in the hemisphere, the cortex, and the subcortex for each of the treatment groups is given in Table 1, along with the mean±s.e.m. of the neural deficit scores. Treatment of mice with 1 mg/kg cTfRMAb-TNFR fusion protein at 45 minutes after the arterial occlusion caused a statistically significant 45% reduction in hemispheric stroke volume, a 48% reduction in cortical stroke volume, and a 42% reduction in subcortical stroke volume, which was associated with a 54% reduction in neural deficit score (Table 1). Conversely, treatment of mice with 1 mg/kg etanercept at 45 minutes after the arterial occlusion caused no significant changes in either stroke volumes or neural deficit scores (Table 1). If the time between occlusion and treatment with 1 mg/kg cTfRMAb-TNFR fusion protein was prolonged from 45 to 90 minutes after arterial occlusion, then a statistically significant 45% reduction in hemispheric stroke volume and a 62% reduction in cortical stroke volume was observed; however, the reduction in subcortical stroke volume or neural deficit score was not significant in this group (Table 1). In a separate series of studies, the duration between the 60-minute arterial occlusion and euthanasia was prolonged from 1 day to 7 days. Mice were treated with either saline or 1 mg/kg cTfRMAb-TNFR fusion protein at 45 minutes after arterial occlusion, and were euthanized at 7 days after the 1-hour MCAO. The TTC stain for a representative mouse in each group is shown in Figure 2. The treatment of mice with 1 mg/kg cTfRMAb-TNFR fusion protein at 45 minutes after arterial occlusion caused a statistically significant 32% reduction in hemispheric stroke volume, and a 38% reduction in subcortical stroke volume, which was correlated with 40% reduction in neural deficit score (Table 1).
Stroke volumes and neural deficit scores
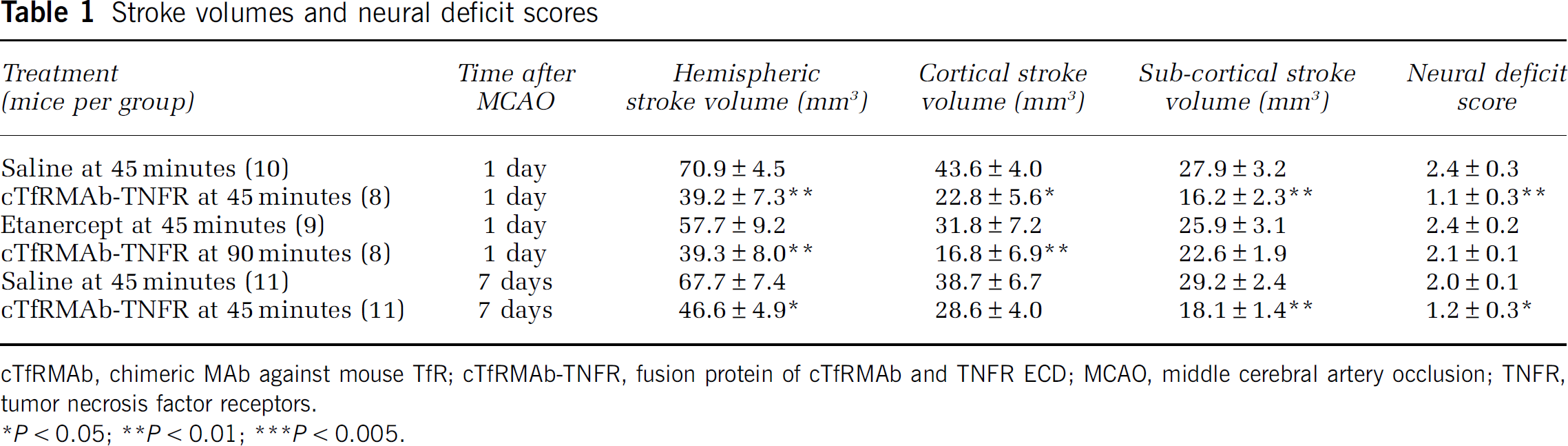
cTfRMAb, chimeric MAb against mouse TfR; cTfRMAb-TNFR, fusion protein of cTfRMAb and TNFR ECD; MCAO, middle cerebral artery occlusion; TNFR, tumor necrosis factor receptors.
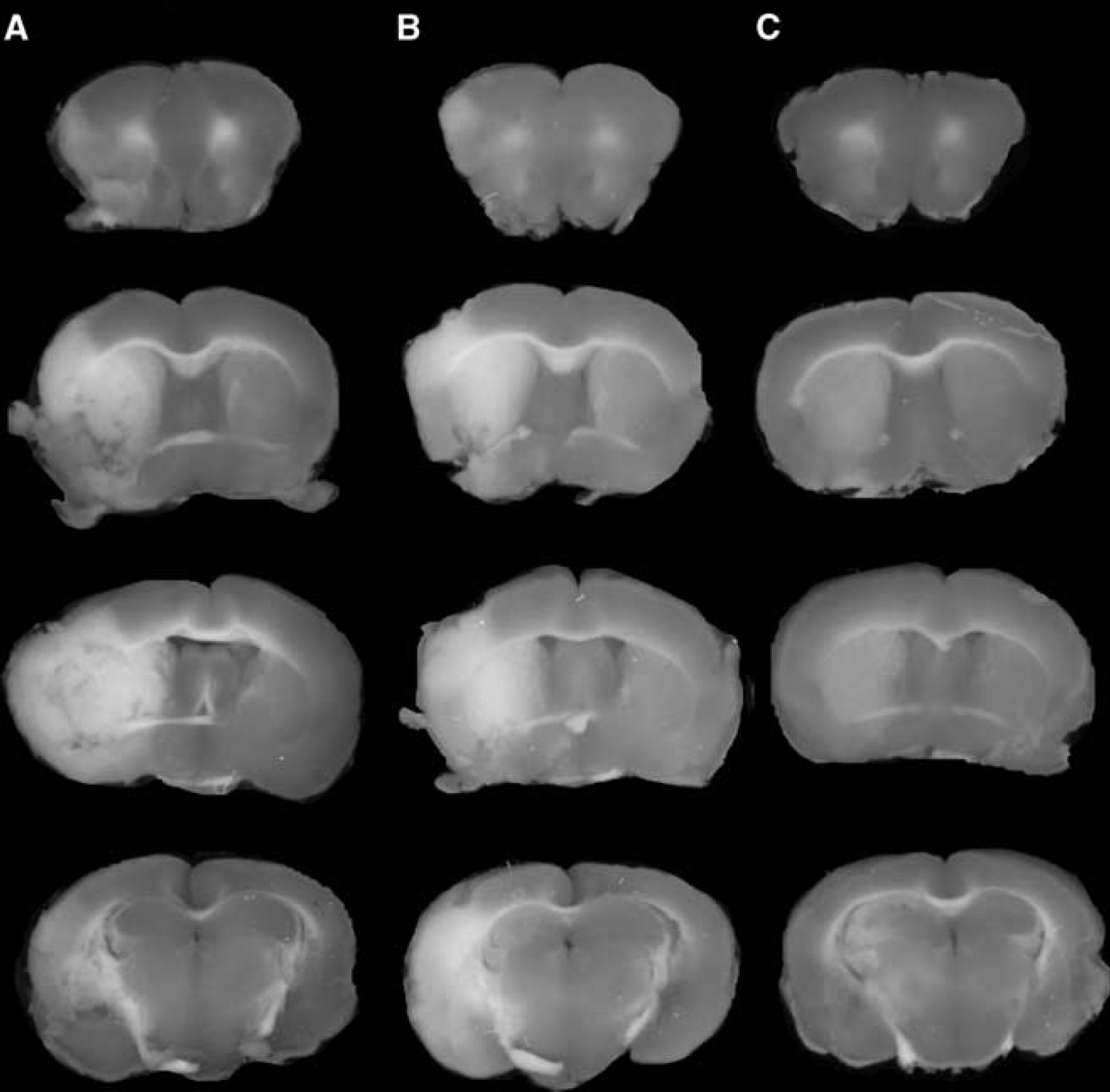
TTC stains of coronal sections from a representative mouse from each of three treatment groups: (
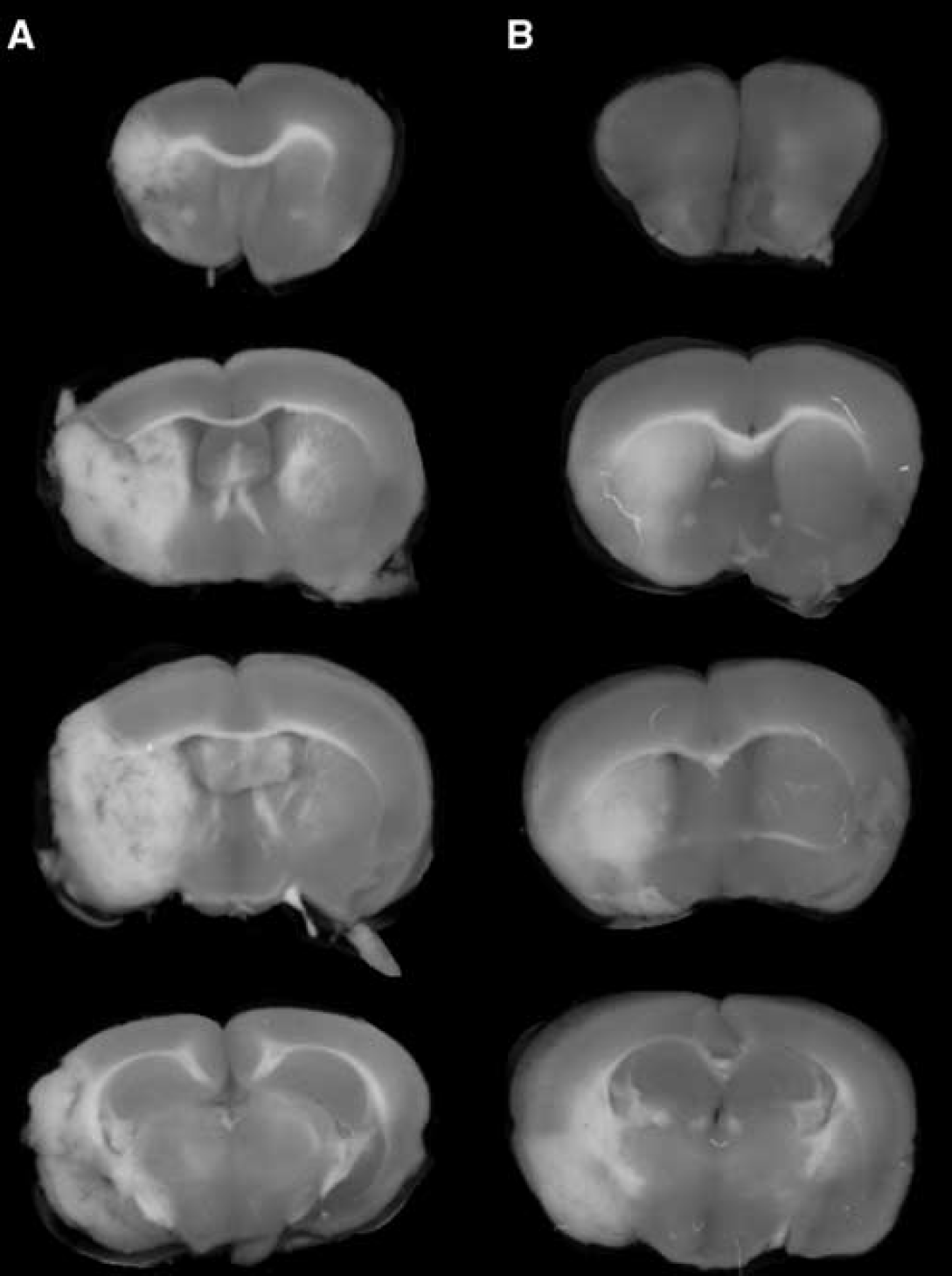
TTC stains of coronal sections from a representative mouse from each of two treatment groups: (
Discussion
The findings of this study show that the intravenous injection of a brain-penetrating TNFI, the cTfRMAb-TNFR fusion protein, is therapeutic in experimental stroke with a reduction in stroke volume that correlates with a reduction in the neural deficit (Table 1). The therapeutic effect is long lasting and persists at 7 days after the acute ischemia (Figure 2). In contrast, a nonbrain-penetrating TNFI, etanercept, has no effect on either infarct volume or neural deficit after intravenous administration (Table 1).
The lack of a therapeutic effect of intravenous etanercept in acute cerebral ischemia parallels similar observations in acute traumatic brain injury. The intravenous administration of 5 mg/kg etanercept is not therapeutic in traumatic brain injury, whereas the intracerebroventricular injection of etanercept is therapeutic in acute brain injury (Knoblach et al, 1999). The lack of a therapeutic effect of intravenous etanercept in acute stroke is consistent with the dual observations that etanercept does not cross the intact BBB (Boado et al, 2010), and that the BBB is intact in the early hours after stroke (Menzies et al, 1993; Belayev et al, 1996), when neuroprotection is still possible in stroke (Zivin, 1998). The maintenance of an intact BBB during the period after acute stroke when neuroprotection is possible means that biologic neuroprotective agents for stroke must be reengineered to penetrate the BBB. Etanercept is a fusion protein of the ECD of the type II human TNFR and the Fc domain of human IgG1 (Peppel et al, 1991). The TNFR ECD of etanercept can be reengineered for brain penetration by genetic fusion to a BBB molecular Trojan horse. The latter is a peptidomimetic MAb against an endogenous BBB receptor, such as the human insulin receptor (HIR). The HIRMAb (MAb against HIR) binds the endogenous BBB insulin receptor, and this binding triggers receptor-mediated transport across the BBB (Pardridge et al, 1995). A fusion protein of the genetically engineered HIRMAb and the human type II TNFR ECD has been engineered, and shown to bind both TNF
The present investigation assesses neuroprotection in stroke after a 60-minute reversible occlusion of the middle cerebral artery in the mouse. The 60-minute occlusion in the mouse may correlate with a more prolonged occlusion in humans, since the evolution of an infarction in the mouse MCAO model is faster than in other species (Carmichael, 2005). In the present study, the cTfRMAb-TNFR or etanercept fusion proteins were administered at a dose of 1 mg/kg intravenously. This therapeutic dose of the cTfRMAb-TNFR fusion protein produces a brain level of TNFR decoy receptor that saturates cerebral TNF
In summary, the present study shows that reengineering a biologic TNFI, the human type II TNFR decoy receptor, as an IgG-TNFR fusion protein that penetrates the BBB, enables therapeutic effects in acute experimental stroke in the mouse after intravenous administration. The role of TNF
Footnotes
Disclosure/conflict of interest
RJB is an employee of, and WMP is a consultant of ArmaGen Technologies, Inc.
