Abstract
Elucidating metabolic effects of hypoxic-ischaemic encephalopathy (HIE) may reveal early biomarkers of injury and new treatment targets. This study uses untargeted metabolomics to examine early metabolic alterations in a carefully defined neonatal population. Infants with perinatal asphyxia who were resuscitated at birth and recovered (PA group), those who developed HIE (HIE group) and healthy controls were all recruited at birth. Metabolomic analysis of cord blood was performed using direct infusion FT-ICR mass spectrometry. For each reproducibly detected metabolic feature, mean fold differences were calculated HIE vs. controls (ΔHIE) and PA vs. controls (ΔPA). Putative metabolite annotations were assigned and pathway analysis was performed. Twenty-nine putatively annotated metabolic features were significantly different in ΔPA after false discovery correction (q < 0.05), with eight of these also significantly altered in ΔHIE. Altered putative metabolites included; melatonin, leucine, kynurenine and 3-hydroxydodecanoic acid which differentiated between infant groups (ΔPA and ΔHIE); and D-erythrose-phosphate, acetone, 3-oxotetradecanoic acid and methylglutarylcarnitine which differentiated across severity grades of HIE. Pathway analysis revealed ΔHIE was associated with a 50% and 75% perturbation of tryptophan and pyrimidine metabolism, respectively. We have identified perturbed metabolic pathways and potential biomarkers specific to PA and HIE, which measured at birth, may help direct treatment.
Introduction
Hypoxic-ischaemic encephalopathy (HIE) remains a persistent worldwide problem occurring in approximately 2 per 1000 term neonates.1,2 Surviving children are at risk of lifelong neurological impairment and represent a large global burden of disease. 3 The recent introduction of therapeutic hypothermia has improved survival rates and critically, reduced neurological disability and improved neurocognitive outcomes.4,5 However, to be effective, hypothermia must be initiated within 6 h of birth. 6 Unfortunately, the ability to recognise ‘at risk’ infants who would benefit from therapy this quickly is not always possible. Current methods of early diagnosis are either unreliable or require a high level of expertise. 7 A robust and non-user-dependent method to equivocally predict HIE at the cot-side and rapidly guide therapeutic hypothermia is needed.
Current understanding of the encephalopathy which stems from perinatal asphyxia, suggests the primary injury response is metabolic. 8 The hypoxic-ischaemic injury deprives the neonatal brain of oxygen and glucose, triggering a biochemical cascade of deleterious events. 8 Reduced oxygen inhibits normal function of the tricarboxylic acid (TCA) cycle and electron transport chain, causing a shift to anaerobic metabolism. Despite compensatory mechanisms, the infant’s ATP supply is quickly depleted and its dependant ion pumps fail. This disturbance of ionic homeostasis leads to an accumulation of free radicals, intracellular Ca2+ and extracellular glutamate as well as cell membrane depolarisation and neurotransmitter activation, sparking the activation of a cascade of pathological processes leading to neuronal cell death hours to days later. 9
The full extent of metabolic change that occurs in response to HIE remains unclear, as does its role in attenuating or exacerbating the injury sustained. To discover novel biomarkers of this early injury, better understanding of the metabolic response to perinatal asphyxia and the resultant HIE is necessary. An investigation of the complex interactions of individual metabolic pathways may yield new insight into the processes involved and potential options for therapeutic intervention.
Our group previously reported metabolic alterations in a population of infants with carefully defined perinatal asphyxia and HIE, using two semi-targeted metabolomic platforms.10,11 The current study aims to use untargeted metabolomics to more extensively examine the cord blood metabolome’s response to injury, allowing the investigation of two objectives: (i) identification of key biochemical pathways perturbed in HIE, which may help elucidate new treatment targets, and (ii) selection of disease specific biomarkers with the potential to characterise an infant’s phenotype. The ability to identify those infants at greatest risk of long-term disability would improve our ability to provide targeted neuroprotection, and hence improve long-term outcome.
Materials and methods
Neonatal study population
The BiHiVE study (
Infants were divided into those that had clinical or biochemical signs of perinatal asphyxia at birth, but recovered quickly with no clinical signs of encephalopathy (PA group) and those who developed an evolving clinical and electrographic encephalopathy consistent with HIE (HIE group). The overall grade of HIE (mild, moderate, or severe) was based on continuous multichannel EEG monitoring at 24 h of life.
Multichannel video-EEG monitoring provides an objective, standardised measure of injury severity 13 and was performed in all cases (HIE and perinatal asphyxia). EEG was analysed by an experienced neonatal neurophysiologist (GBB) and EEG grade was assigned at 24 h. EEG grading of HIE in this cohort has been previously described 14 using a validated modified grading system to assign a EEG grade of mild, moderate, or severe.15,16Clinical grade of HIE was assigned using the modified Sarnat score, as mild, moderate or severe also at 24 h.
During the same time period, a healthy control cohort was recruited as part of an ongoing birth cohort study, the Cork BASELINE Birth Cohort Study. 17 Infants were matched case-control one-to-one based on the following clinical variables: gender, gestational age, method of delivery, birth weight and centile, as well as sample storage time at −80℃.
Sample collection and storage
Umbilical cord blood (UCB) was drawn from cases and controls, processed and stored within 3 h of birth using the following strict standard operating procedure: UCB was collected within 20 min of placental delivery and placed in an additive free serum tube. After 30 min clotting time, it was centrifuged at 2400 × g for 10 min. The serum was removed, placed in a clean spin tube and centrifuged at 3000 × g for 10 min, all at 4℃. Aliquots of 250 microlitres of serum were placed in microtubes and stored at −80℃ until metabolomic analysis.
Metabolomic analysis of UCB serum with DI FT-ICR MS
Sample preparation
A modified Bligh-Dyer biphasic extraction method was used to extract polar metabolites from UCB serum.18,19 Extractions were conducted on ice to minimise metabolic activity. Assuming 90% water content of serum, 450 μl methanol was added to frozen samples and allowed thaw on ice. A further 1350 μl methanol, 1800 μl chloroform and 1440 μl water was added to achieve a ratio of 2:2:1.8. Samples were vortex mixed and allowed to stand for 10 min on ice. The extraction solution was centrifuged at 1800g for 15 min at 4℃ to allow separation of the upper polar phase consisting mainly of methanol/water with hydrophilic metabolites; 400 μl aliquots of the polar phase and a quality control (QC) sample, created by pooling 80μl aliquots from each sample, were dried down in a centrifugal evaporator (Thermo Savant, Holbrook, NY) and stored at −80℃ until re-suspension for MS analysis.
DI FT-ICR MS analysis and signal processing
The polar fractions from each sample were analysed in both positive and negative ion mode mass spectrometry. Each samples was re-suspended in either 80:20 methanol:water with 0.25% formic acid solution (positive ion analysis) or 80:20 methanol:water with 20 mM ammonium acetate solution (negative ion analysis). A blank sample consisting of only the re-suspension solution and analysed as per a real sample was run at the beginning and end of each analysis to detect mass spectral features not of biological origin.
All metabolomic analyses used a hybrid linear ion trap/Fourier transform ion cyclotron resonance 7-T FT-ICR mass spectrometer (LTQ FT Ultra, Thermo Fisher Scientific, Bremen, Germany) with a chip-based direct infusion nanoelectrospray ionisation (nESI) assembly (Triversa, Advion Biosciences, Ithaca, NY). nESI conditions consisted of a 200 nl/min flow rate, 0.3 psi backing pressure and +1.7 kV electrospray voltage controlled by ChipSoft software (version 8.1.0). Mass spectrometry conditions included an automatic gain control setting of 1 × 106 and a mass resolution of 100,000. Analysis time was 2.25 min (per technical replicate), controlled using Xcalibur software (version 2.0, Thermo Fisher Scientific). To increase metabolome coverage and maintain mass accuracy, spectra were collected using the ‘SIM stitching’ method, 20 in which data were acquired across seven selected ion monitoring (SIM) mass ranges or ‘windows’ to cover a total mass range of 70–590 Da for polar compounds. All data processing was conducted within Matlab software (version R2013, MathWorks, Natick, MA, USA) using custom written software except where stated. Duplicate replicate analyses of each sample were taken and filtered into a single peak list via a three step filtering process to enhance spectral reliability. 21
For each analytical batch, QC samples representative of the infant population being studied were included as a quality assurance measure. QC samples were injected multiple times at the beginning of each batch for equilibration and were then injected after every fifth sample and at the end of the batch to assess and correct the precision and reproducibility of individual mass spectral features.22,23 To reduce inter and intra batch variation, the Quality Control-Robust Spline Correction (QC-RSC) batch correction algorithm 24 was applied, in conjunction with three spectral cleaning algorithms, to remove unreliable, inconsistent or unreproducible features. For statistical analysis, the final intensity matrices were normalised using the probable quotient normalisation (PQN) method 25 and logarithm (log10) transformed to reduce technical variation.
Putative metabolite annotation and pathway analysis
Interpretation of mass spectral data requires first annotating with likely biochemical identifications based on accurate mass (putative annotations). These lists of data are better understood if they are grouped together into metabolic pathways based on known existing pathways from metabolic libraries. By comparing the percentage of significantly changing intermediates in these pathways to the original overall coverage (pathway analysis), pathways that are most affected are highlighted and it becomes easier to understand the biochemical shifts that are occurring with the disease process and may increase our confidence in altered putative annotations.
Metabolite features accrued in the raw data were putatively annotated by applying the Metabolite Identification Package (MI-Pack) workflow described previously. 26 Metabolite features identified in the processed raw data of mass spectral peaks were assigned one or multiple potential identifications based on accurate mass matching (with a 1.25 ppm error) against the Human Metabolome Database (HMDB) and Kyoto Encyclopaedia of Genes and Genomes (KEGG) libraries.27,28 The process of mass spectrometry analysis can lead to the formation of adducts of the original metabolite, e.g. [M+H]+, [M+Na]+, [M+K]+ are common adducts seen in positive ion mode, while [M-H]−, [M+Hac-H]−, [M+Cl]− are common negative ion mode adducts. 29 Common adduct forms were accounted for in our search, hence some metabolites are represented by more than one mass spectral feature. It is also important to remember that isomers (compounds which share the same mass) cannot be distinguished based on accurate mass alone. To avoid the overrepresentation of multiple isomers in the subsequent pathway analysis, empirical formulae rather than individual metabolite identity was used to determine pathway coverage for the pathway analysis. This approach equates to ‘level 2’ identification as defined by The Metabolomics Standards Initiative.30,31
Pathway analysis, also conducted using MI-PACK, focused on calculating the percentage perturbation of every individual human pathway as recorded in KEGG. The percentage perturbation of a biochemical pathway was determined by calculating the ratio of the number of empirical formulae significantly altered in that pathway, by the total number of empirical formulae detected in that pathway, i.e. pathways with high percentage perturbation had a large percentage of empirical formulae which were significantly different in the disease state compared to the number originally detected that mapped to that pathway. The same empirical formulae could occur in multiple pathways but each formula was counted only once for each pathway even when it may represent multiple metabolites in that pathway. This calculation was performed for the comparisons ΔPA and ΔHIE separately, then with the two groups combined compared to the control, to distinguish compounds related solely to the pathophysiology of either perinatal asphyxia or HIE.
The percentage perturbation of a biochemical pathway was determined by calculating the ratio of the number of empirical formulae significantly altered in that pathway, by the total number of empirical formulae detected from that pathway. This calculation was performed for the comparisons ΔPA and ΔHIE separately, then for empirical formulae altered in both groups combined, to distinguish compounds related solely to the pathophysiology of either perinatal asphyxia or HIE.
Statistical analysis
All statistical analyses were performed using MatLab (version R2013,MathWorks, Natick, MA, USA). Statistical comparisons of clinical patient data between cases and matched controls were performed using Student’s t-test or Mann–Whitney U test as required.
The relative standard deviation (RSD) of each metabolic feature detected was calculated for the QC samples (RSDQC). Mass features were included for further analysis only if the RSDQC < 20%, a measure of analysis precision. 32 Normality assumptions for each class were tested using the Lilliefor’s test. Comparisons between both HIE and matched controls (ΔHIE) and perinatal asphyxia (no encephalopathy) and matched controls (ΔPA) are reported. Alterations in each mass feature were calculated using a paired Student’s t-test for all matched case-control pairs with a Benjamini–Hochberg false discovery correction for multiple testing based on an assumed false discovery rate of 5%. 33 This yielded an adjusted p value, termed q value, for each variable. Any pair of samples with one or more missing values for that mass feature was excluded. For each mass feature, mean fold change differences were calculated for ΔHIE and ΔPA and reported with 95% confidence intervals. The differences in effect size between the arms of the study (ΔHIE vs. ΔPA) were tested using a two sample t-test and the associated p-values are reported. To determine if metabolites differed across the grade of injury, a one-way analysis of variance (ANOVA) with Tukey’s post hoc analysis was performed on the mean fold differences of the matched case-control pairs. Analysis was focussed on those peaks for which putative annotations could be determined.
Results
Study population
Clinical and demographic data of the study population.
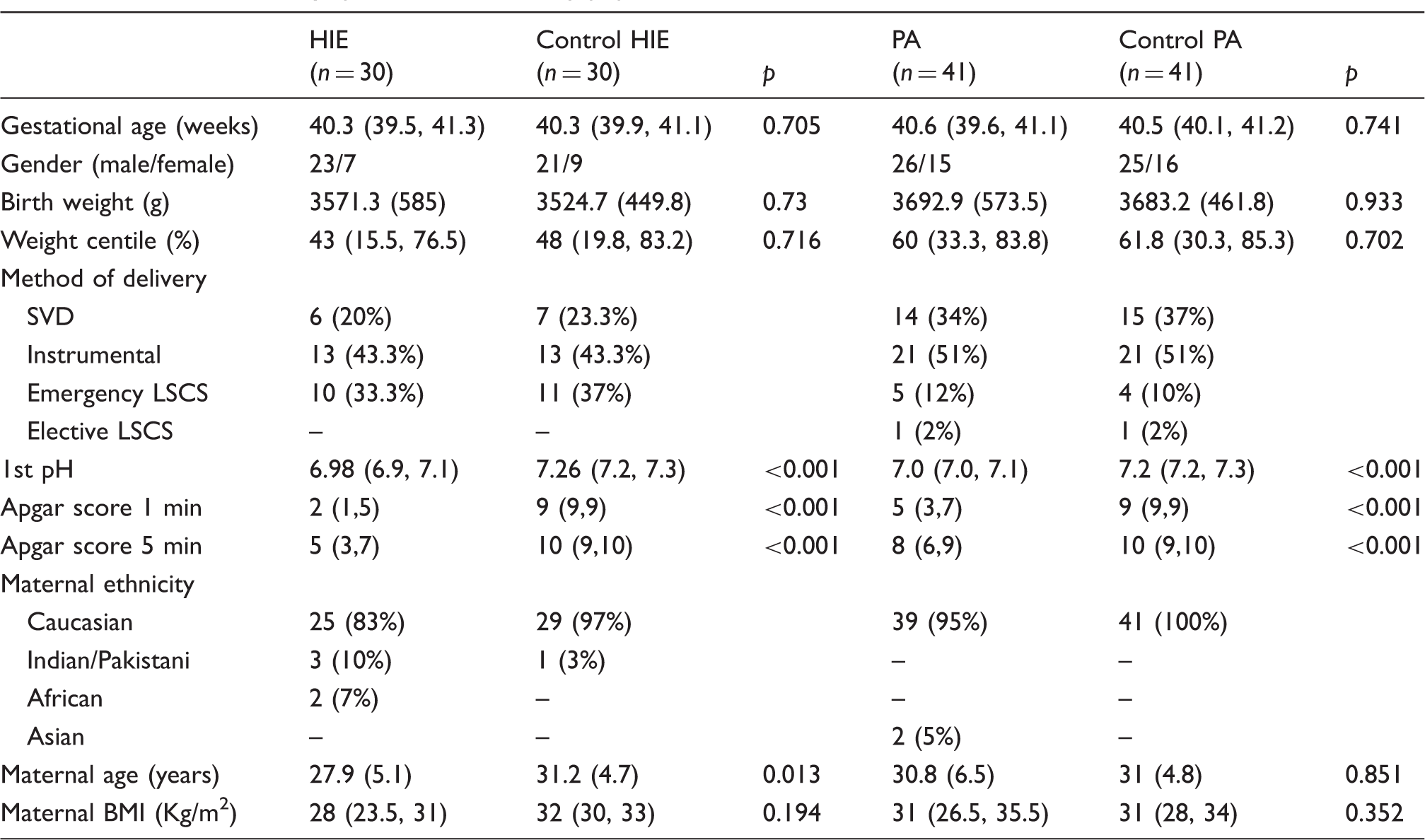
Note: Values are mean (SD), median (interquartile range), or n (%).
HIE: hypoxic-ischaemic encephalopathy.
Univariate analysis
The DI FT-ICR MS analysis of UCB serum yielded a total number of 1276 and 1737 polar mass spectral features in nESI positive (+) and negative (−) modes, respectively. Following spectral cleaning to remove unreliable, inconsistent or unreproducible features (>20% RSDQC), 1042 nESI(+) and 1191 nESI(−) metabolic features remained and were subject to analysis (Figure 1). A putative metabolite name was assigned to 17% (179) of masses detected in nESI(+) mode and 9% (109) in nESI(−) mode in total. It is important to note that metabolite annotations discussed should be viewed as preliminary annotations until fully validated using chemical standards and two orthogonal properties as defined by the Metabolomics Standards Initiative.
29
In many cases, multiple putative annotations have been assigned to single statistically significant metabolic features; however, we will discuss metabolites that have been previously detected as significant in this cohort,10,11 and those which have arisen from a single pathway.
Summary of the number of mass features detected and significantly altered in nESI(+) and nESI(−), respectively. Red Venn diagrams show the distribution of putatively annotated metabolic features, of those significantly altered for each comparison groups; perinatal asphyxia vs. matched controls (ΔPA) and HIE vs. matched controls (ΔHIE).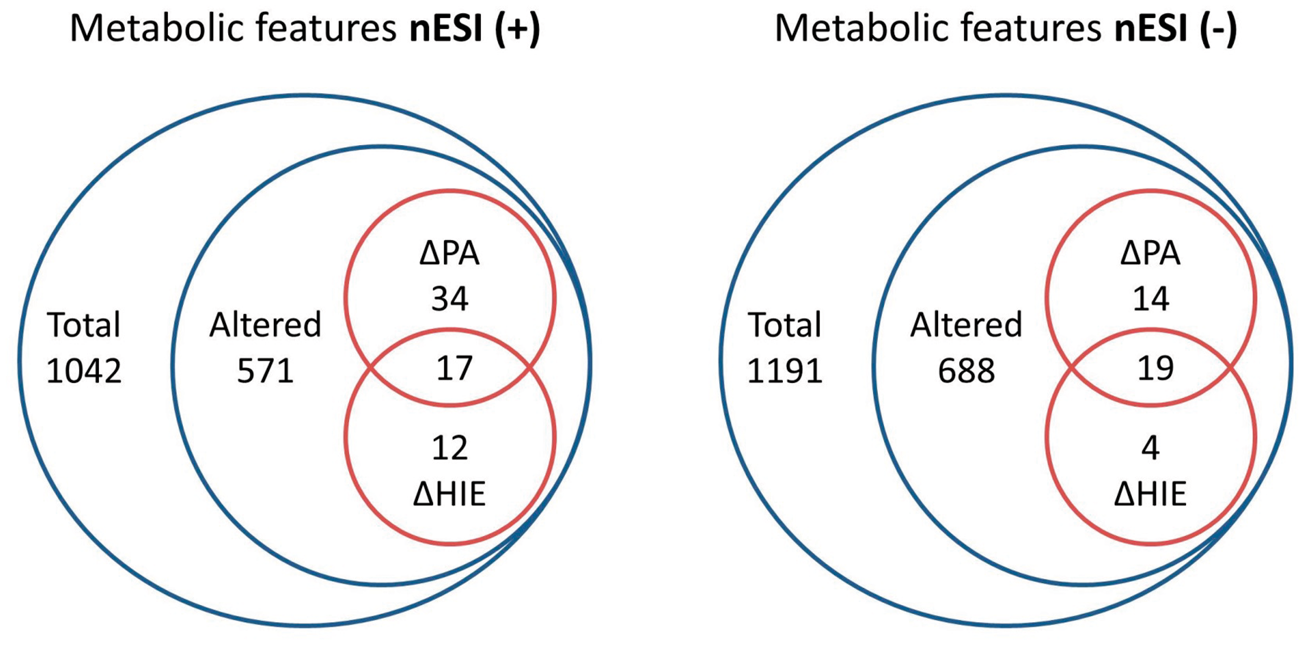
To select the most important spectral features associated with perinatal asphyxia and HIE, a fold change statistical comparison, of two separate groups, perinatal asphyxia vs. matched controls (ΔPA) and HIE vs. matched controls (ΔHIE) was performed. In total 571 (55%) nESI(+) and 688 (58%) nESI(−) metabolic features were significantly altered (p < 0.05), of which 63 and 37 were putatively annotated, respectively. A summary of these mass spectral features is provided in Supplementary Table 1. Assessment of the QC measurements for each putatively annotated metabolic feature confirmed excellent technical reproducibility for features with a median RSDQC of 11% nESI(+) and 9% nESI(−). 23
Putatively annotated metabolic features significantly altered post false discovery correction (q < 0.05) in the ΔHIE and/or ΔPA comparison groups.
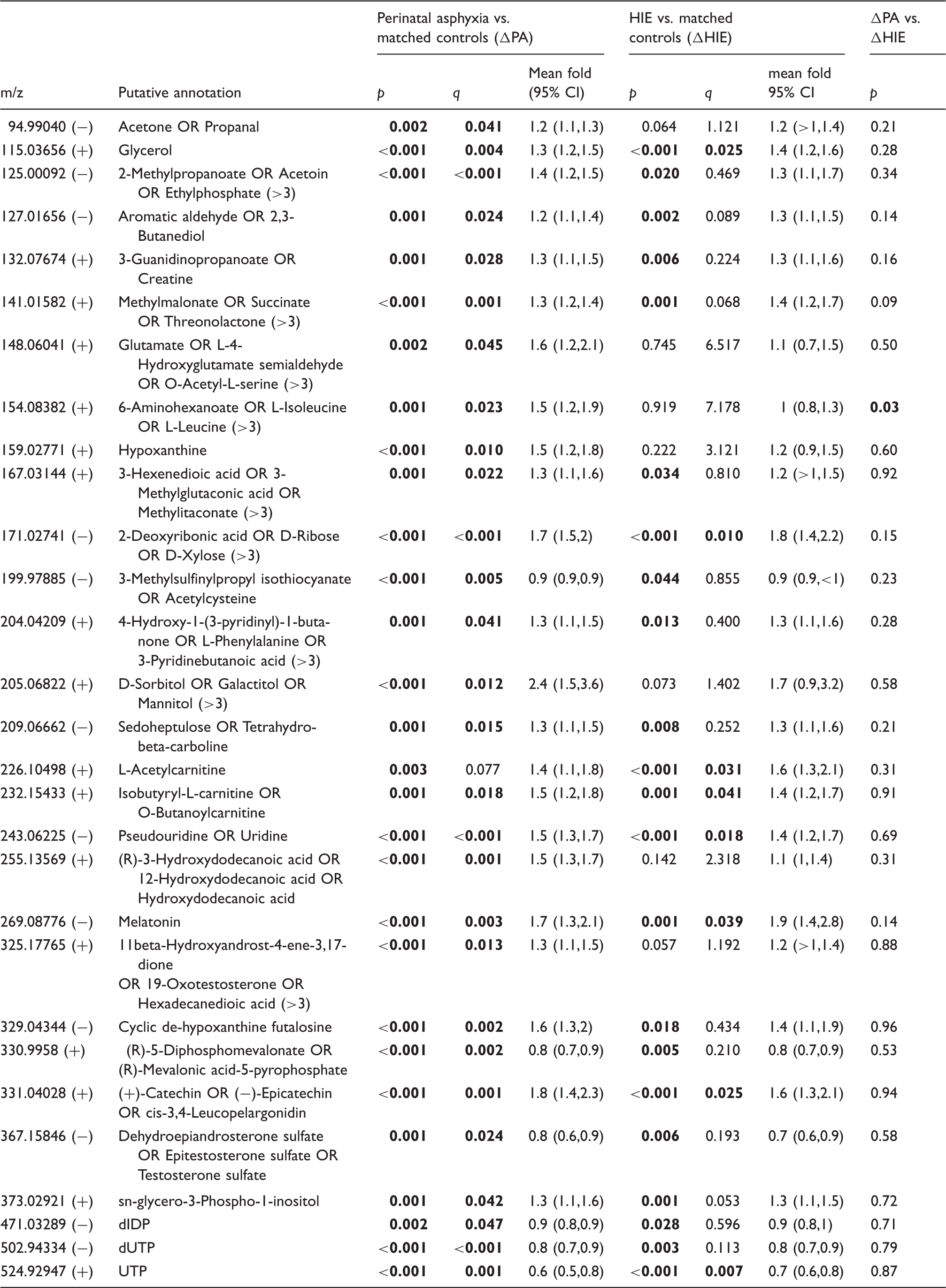
Note: (>3) indicates more than three possible putative annotations. Bold font indicates features that have passed the defined level of significance (q < 0.05).
>1 or <1 indicates a confidence interval of greater or less than 1, whose decimal points have been rounded.
HIE: hypoxic-ischaemic encephalopathy; UTP: uridine-triphosphate.
Significant elevations were detected in amino acids and acylcarnitines, compound classes previously found altered in these infants.10,11 Putatively annotated phenylalanine (m/z 204.04209, [M+K]+) was significantly altered in both comparison groups. Glutamate (m/z 148.06041, [M+H]+) and leucine (m/z 154.08382, [M+Na]+) were increased in ΔPA, but not in ΔHIE. Leucine was the only compound to be significantly altered (p = 0.03) between ΔPA vs. ΔHIE; however, this difference did not remain following correction for FDR. Acetylcarnitine (m/z 226.10498, [M+Na]+) and butanoyl-carnitine or isobutyryl-carnitine (m/z 232.15433, [M+H]+) increased significantly in both groups with fold changes (95% CI) of 1.6 (1.3, 2.1) and 1.4 (1.2,1.7) in ΔHIE, respectively.
Significant depletions in features linked to pyrimidine metabolism were also detected. Uridine (m/z 243.06225, [M-H]−) was highly significant, increasing 1.5 fold (1.3, 1.7 95%CI) in ΔPA and 1.4 fold (1.2, 1.7 95%CI) in ΔHIE. By contrast, uridine-triphosphate (UTP) (m/z 524.92947, [M+(41K)]+) and dUTP (m/z 502.94334, [M+Cl]−) were significantly decreased in both groups, UTP having a fold change of 0.7 (0.6, 0.8) in ΔHIE. Finally, acetone (m/z 94.9904, [M+K-2H]−) was significantly raised in ΔPA and glycerol (m/z 115.03656, [M+Na]+) was significantly raised in both injury groups, increasing 1.4 (1.2, 1.6 95%CI) fold in ΔHIE (p < 0.001).
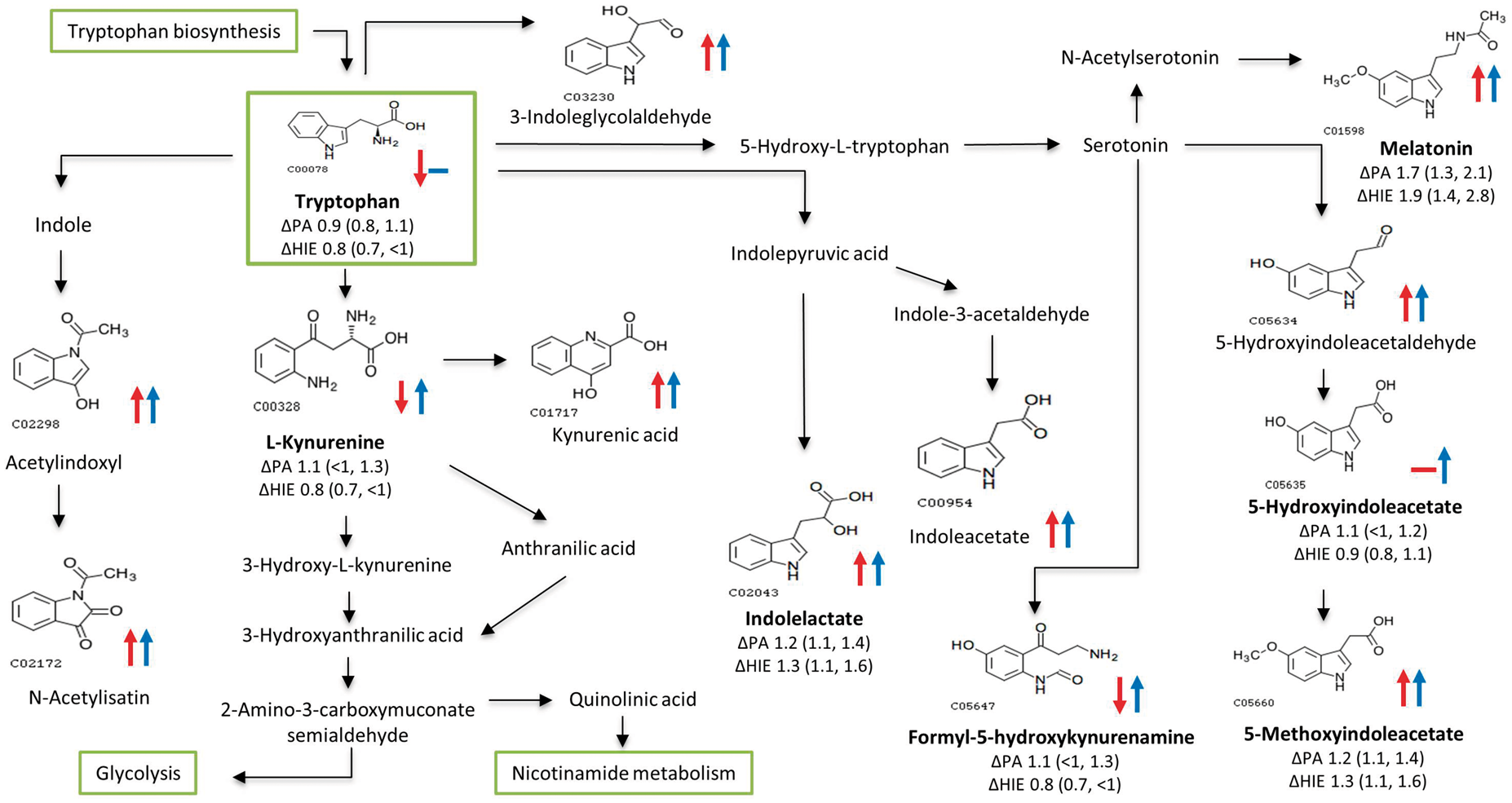
To assess whether any of the putatively annotated metabolites could be used to predict HIE severity, the mean fold change differences across the grades of HIE (compared to their matched controls; ΔMild, ΔModerate and ΔSevere) and ΔPA were analysed (Figure 2). This identified 14 putatively annotated metabolites which were differentially altered across the grades of HIE, see Table 3. No feature in this group passed false discovery correction. Features equating to succinate [M+H]+ and possibly a more unusual adduct of acetone [M-2H-K]− were significantly altered across grades injury severity. Alterations in these metabolites in severe HIE using NMR analysis in the same HIE cohort have been previously reported.
10
Metabolite log fold changes stratified by disease severity. Fold changes compare the difference in log transformed data in individual cases compared to their matched healthy controls in each disease group; perinatal asphyxia, mild HIE, moderate HIE and severe HIE. Putatively metabolic features significantly across the perinatal asphyxia and HIE severity groups (ΔPA vs. ΔMild vs. ΔModerate vs. ΔSevere). Note: (>3) indicates more than three possible putative annotations. Bold font indicates features passing the defined level of significance (p < 0.05). HIE: hypoxic-ischaemic encephalopathy.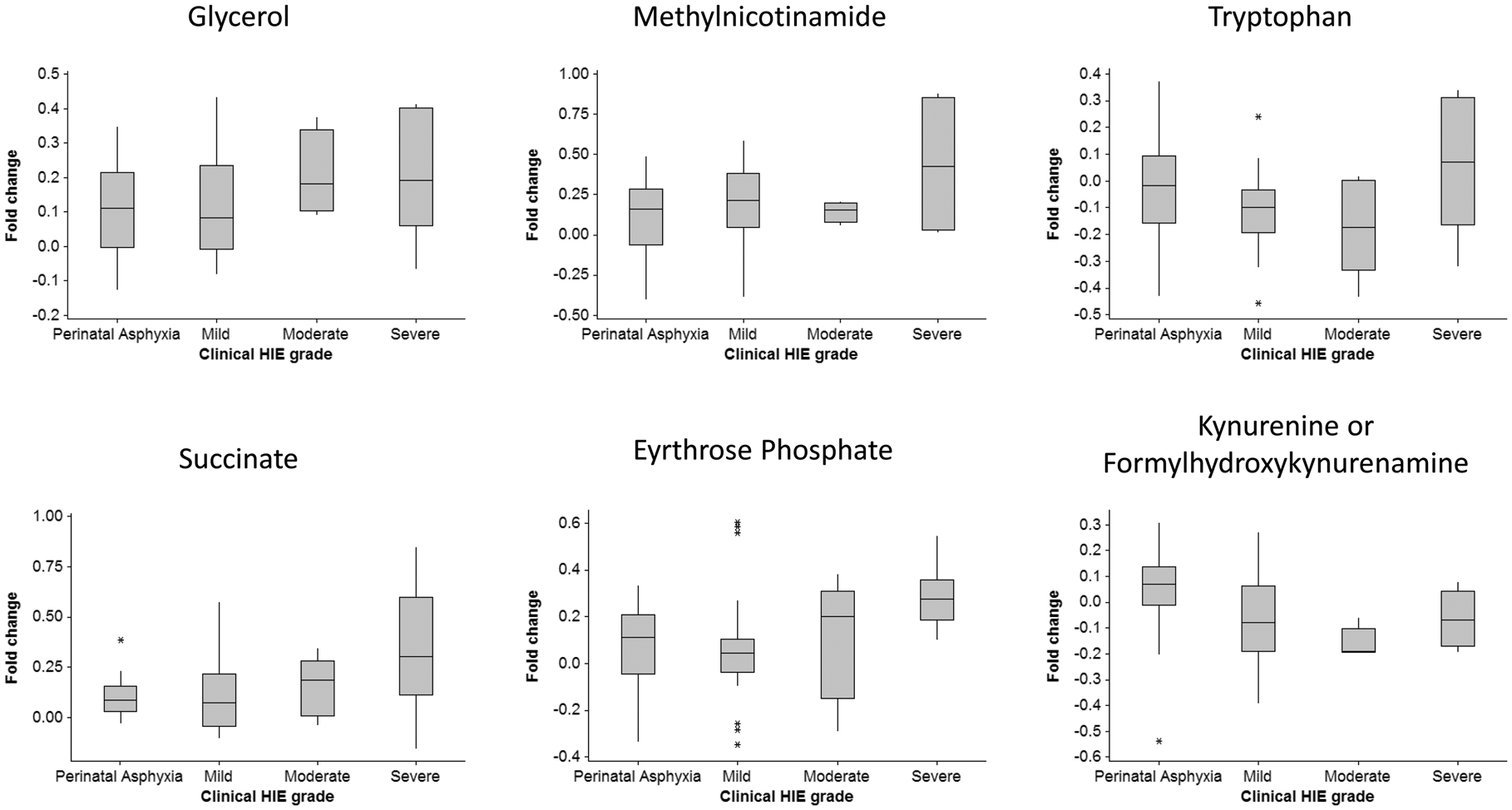
Pathways analysis
To probe potential biochemical pathway alterations in perinatal asphyxia and HIE, the significantly altered compounds found in the case vs. matched control comparisons from both positive and negatively charged ion datasets were combined. Certain metabolites (eight empirical formulae) were detected using both ionisation modes; therefore, an over-representation of the pathway may occur. However, this enabled the comparison of metabolites and ion adducts from nESI(+) and (−) to assess whether fold changes were in agreement, providing additional confidence in our findings.
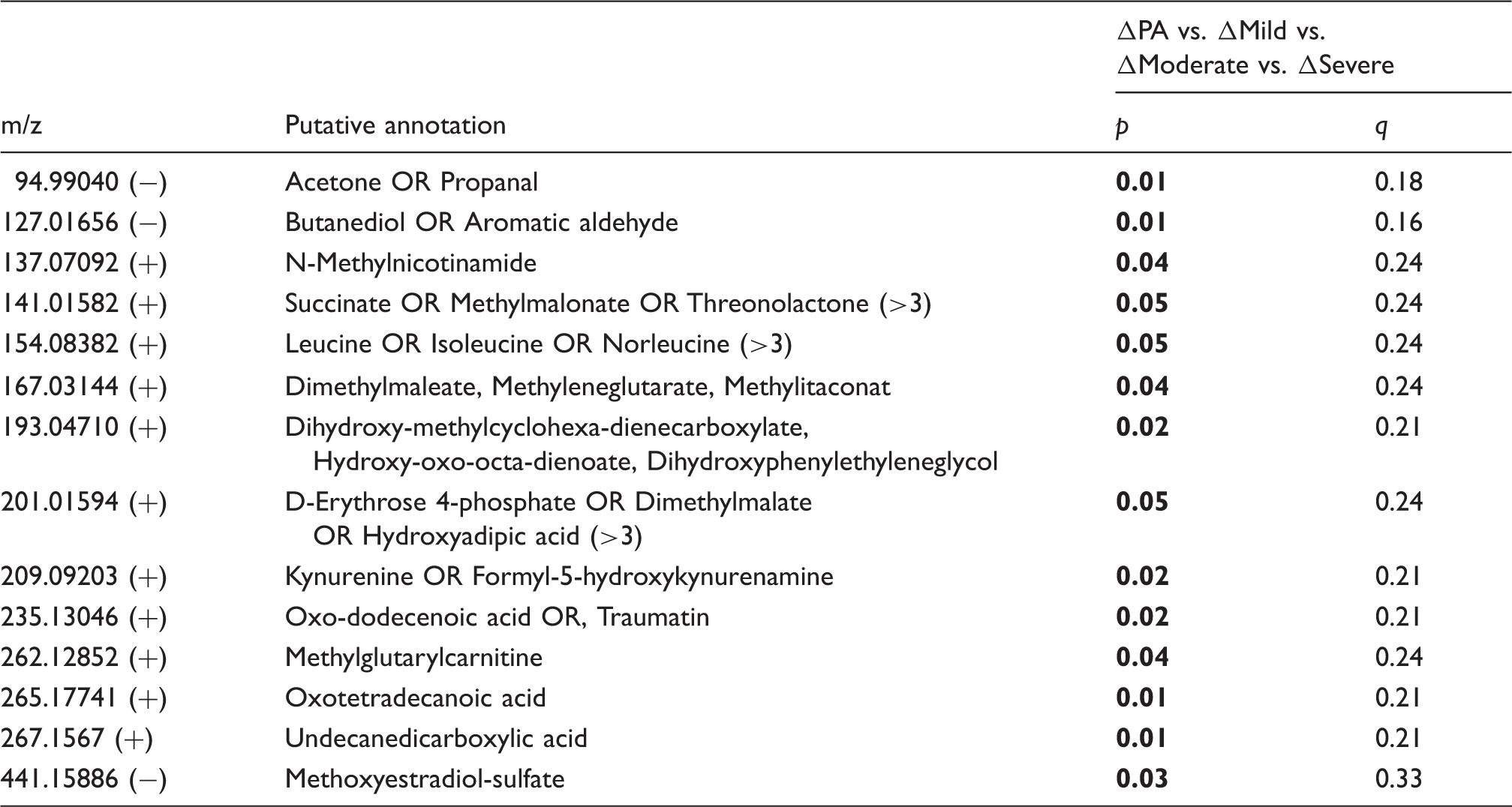
Figure 3 displays the foremost perturbed metabolic pathways, determined by the greatest percentage perturbation, in perinatal asphyxia and HIE from the cord blood metabolome. Only two pathways, pyrimidine metabolism and tryptophan metabolism, exhibited an increasing perturbation as the injury progressed from perinatal asphyxia to HIE. The pyrimidine pathway displayed perturbations of 75% in HIE, with three unique empirical formulae implicating glutamine (m/z 181.03847, [M+Cl]−) p = 0.044, hydroxypropanoate (m/z 113.02091, [M+Na]+) p = 0.015, and dihydrothymine (m/z 151.04777, [M+Na]+) p = 0.045. Uridine and its derivatives were altered in both groups, passing false discovery correction (Table 2), while urea was significantly decreased in HIE. Eight empirical formulae were detected from the tryptophan metabolism pathway which was 50% perturbed in HIE, see Figure 4. Putatively annotated kynurenine and tryptophan were unique to HIE, while melatonin (m/z 269.08776, [M+(37Cl)]−) p = 0.001 displayed the largest fold change (95% CI) = 1.9 (1.4,2.8).
Pathway analysis. Altered metabolomic pathways and networks of UCB serum as a result of HIE (ΔHIE in red), PA (ΔPA in green) and those altered in both comparisons (ΔHIE and ΔPA in blue). The y-axis displays the pathway name alongside the total number of empirical formulae from that pathway detected in the analysis. The x-axis displays the % perturbation for each pathway. For example, the TCA cycle was 100% perturbed meaning the three empirical formulae were detected were all altered as a result of PA and HIE. Alterations in tryptophan metabolism. The structure is shown for the compounds identified by FT-ICR-MS. Arrows indicate an increases (↑) or decreases (↓) in metabolite fold change; ΔHIE (red) and ΔPA (blue), while metabolite IDs in bold indicate the significantly altered compounds with fold change (95% CI) included.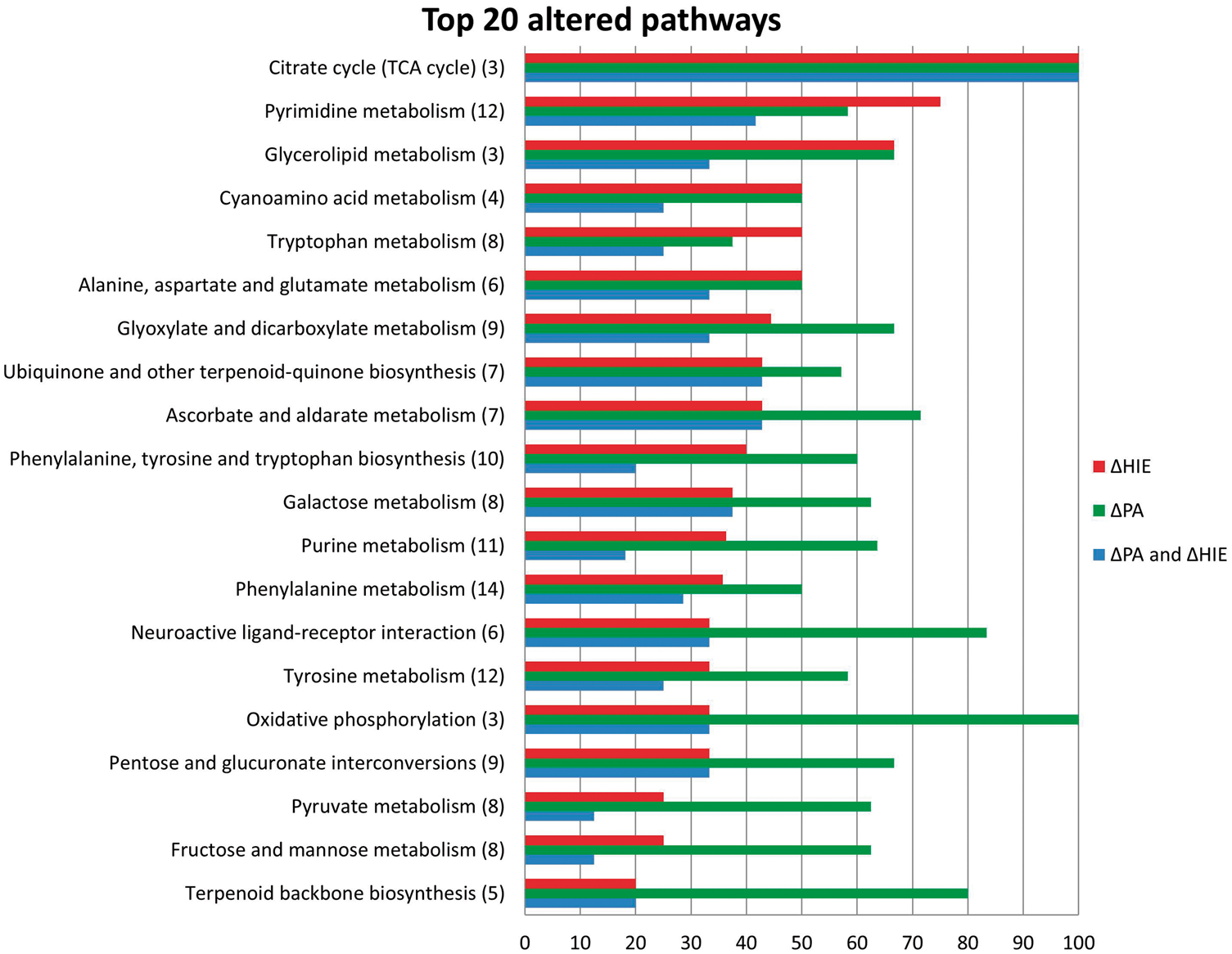
The overlapping biosynthesis of the phenylalanine, tyrosine and tryptophan pathway, as well as the individual metabolic pathways of these amino acids was disturbed, Figure 3. This disruption tended to be more wide spread in perinatal asphyxia, except for tryptophan metabolism. Our analysis detected 10 empirical formulae related to the biosynthesis of phenylalanine, tyrosine and tryptophan, implicating seven unique metabolites. Tryptophan was the only metabolite to be decreased in HIE, the others including phenylalanine and tyrosine increased in both HIE and perinatal asphyxia. Increases were also detected for erythrose-phosphate, deoxy-ketofructose phosphate and (hydroxyphenyl) pyruvate, whose alterations were attributable to perinatal asphyxia alone.
Discussion
This is the first study to characterise the human metabolome of perinatal asphyxia and neonatal HIE using an untargeted metabolomic approach. The aim was to identify novel metabolic pathway alterations to enhance understanding of the complex aetiology of perinatal asphyxia and HIE. This has enabled the identification of potentially important metabolic markers for the accurate prediction of HIE at birth, thus meriting further investigation.
A key strength extends to our ability to verify our findings through the implementation of a cross validation system. Upon biobanking this cohort, several micro-aliquots of serum were frozen from each infant allowing samples from each infant to be analysed on multiple metabolomic platforms. The current study represents the third time samples from this neonatal cohort have undergone metabolomic analysis, first a targeted and then a semi-targeted approach was taken.10,11 No single analytical platform has the sensitivity to detect all metabolites, therefore this three pronged approach ensures maximum coverage of the metabolome is achieved and increases confidence in our findings. For example, using 1H-NMR we have previously identified alterations in acetone, lactate, glycerol, creatine and succinate all of which were also found to be significantly altered using DI FT-ICR MS. 10 Using a combined targeted DI and liquid chromatography (LC) tandem mass spectrometry (MS/MS) approach, we also found changes in several amino acids that are confirmed herein. 11 The varying metabolomic detection methods used have ranged in quantitative ability, DI FT-ICR MS being relatively quantitative, therefore we report fluctuating degrees of metabolite changes but importantly the direction of change has remained consistent.
The heterogeneity of HIE suggests several pathway mechanisms will play a role in its pathophysiology. This study evaluated the metabolic components identified using DI FT-ICR MS that were significantly different in PA and HIE compared to their respective matched control groups. We reveal alterations in a wide range of metabolite classes highlighting several major metabolic pathways associated with these conditions.
Tryptophan metabolism
This study presents extensive coverage of the tryptophan metabolism pathway in perinatal asphyxia and HIE. In infants with HIE, we found total pathway disturbances in 50% of detected metabolites. Tryptophan is an essential amino acid necessary to maintain cellular health and homeostasis. It functions as a biochemical precursor for two important metabolic pathways; (i) serotonin synthesis and (ii) kynurenine pathway which produces nicotinamide adenine dinucleotide (NAD+). We have shown alterations in metabolites central to and stemming from, both of these pathways as displayed in Figure 4.
We report a significant decrease in tryptophan levels associated with neonatal HIE. Interestingly, when grades of HIE were examined, we found tryptophan levels were at their lowest in the infants with mild and moderate HIE. Three of the five severe infants for which tryptophan was detected, displayed a fold change increase. In the adult population, tryptophan depletion has been investigated in a wide variety of psychological, behavioural, and physical processes as well as, psychiatric disorders. 34 In particular, tryptophan decreases have been demonstrated to affect a number of cognitive processes, for example learning and memory skills in healthy individuals. 35 Several indole metabolites, metabolised from tryptophan to produce biologically active metabolites, were detected in the current study. Indolelactate was significantly increased up to 1.5 fold in HIE and interestingly, is generally found at significantly higher levels in umbilical foetal plasma. 36
The serotonergic network has been proposed as a candidate network, which is disrupted after neonatal hypoxia-ischaemia.37,38 This network is one of the first transmitters systems to peak in the developing brain. Serotonin functions as a neurotransmitter and is synthesised via 5-hydroxy-L-tryptophan by two enzymatic reactions including the hydroxylase and decarboxylase of tryptophan. Several metabolites downstream of serotonin were tentatively identified including significant alterations in hydroxyindoleacetate, methoxyindoleacetate and formyl-hydroxykynurenamine. Serotonin can also be converted to the neurohormone melatonin via n-acetylserotonin. We found melatonin to be significantly raised in this study, with levels elevating 1.7 fold in the perinatal asphyxia population compared to matched controls, and further still in HIE to 1.9 fold (p = 0.001). This is, perhaps, surprising due to the decrease in the upstream precursors of serotonin and melatonin, including tryptophan. We speculate that this may be due to decreased breakdown of Melatonin, or possibly increased production through alternate pathways. Again this requires validation in future studies. Melatonin is a naturally occurring neurohormone synthesised from tryptophan in the pineal gland. 39 Involved in physiological processes, it is considered a natural neuroprotectant and free radical scavenger, with the ability to promote the production of other antioxidant enzymes. 40 The neuroprotective effects of administered melatonin have been demonstrated in animal models of hypoxic brain injury, by decreasing microglial activation and astroglial reaction which prevents white matter myelination defects, as well as reducing the number of apoptotic cells.41,42Melatonin’s protective effects against induced oxidative mitochondrial damage, oxidative lipid and DNA damage have also been demonstrated.43,44 Robertson et al. 45 recently showed that melatonin augmented therapeutic hypothermia, as evident by a reduction in lactate/creatine and lactate/N-acetyl aspartate ratios in deep grey matter and an increase in the whole brain nucleotide/exchangeable phosphate pool. 45 Maternal melatonin treatment prior to the hypoxic-ischaemic insult reduced inflammation and apoptotic markers, 46 and delayed seizure onset. 47 In vitro, melatonin decreased the expression of CD11b in neutrophils, a marker of inflammation in neonatal encephalopathy. 48
Interestingly, oral dose melatonin is currently being trialled as a therapeutic agent; in term infants with moderate and severe HIE in St Louis USA, with neurodevelopmental outcome at 18 months being the primary efficacy measure (NCT01904786). 49 Reports on how endogenous melatonin responds to a hypoxic ischaemic insult are scarce; however, elevated levels have been documented in critically ill children with the authors hypothesising its rise may counteract the oxidative stress response. 50
This study detected two metabolites belonging to the kynurenine pathway. Kynurenine metabolites, enzymes and intermediates are neuroactive and responsible for the synthesis of coenzyme NAD+, which plays an important role in cellular respiration, energy production and DNA repair. 51 Tryptophan becomes deoxygenased to produce formyl-kynurenine which is rapidly metabolised to kynurenine. We report a significant reduction in kynurenine in HIE and raised levels of its neighbouring metabolite kynurenic acid in HIE and PA though not reaching statistical significance. Protective effects of kynurenic acid have been proposed, it is thought to reduce excitotoxin-induced neuronal death by antagonising excitatory glutamate receptors. 52 Conversely, a significant increase was previously reported in kynurenine/hydroxykynurenine in a piglet model of hypoxia. 53 Kynurenine can also progress down another branch of metabolism to produce hydroxykynurenine and hydroxyanthranilic acid which may cause either apoptotic or necrotic neuronal death in cultures. 52 Further downstream lies quinolinic acid, the focus of much study due to its interactions with N-methyl-D-aspartate receptors causing convulsion and excitotoxicity, therefore implicated in the pathogenesis of several human neurological diseases. 54
Pyrimidine metabolism
We report alterations in 75% of detected pyrimidine pathway metabolites. Pyrimidines are heterocyclic organic compounds whose derivatives cytosine, thymine and uracil are the constituents of nucleotides, the building blocks of RNA and DNA production. Uridine is the principle pyrimidine in humans and a precursor of brain membrane phospholipids in the form of UTP. 55 We have found a marked increase in uridine and pseudouridine in HIE. Beneficial effects of uridine have been proposed, hinged on its ability to enhance levels of cytidine-5-diphphosphocholine (CDP-choline), 56 a metabolite found to be increased in the retinal tissue metabolome after hypoxia-derived brain damage. 57 Uridine administered post hypoxic-ischaemic insults reduced infarct volume and expression of caspase-3 a marker of apoptosis. 58 Other protective functions include its potential to increase the synthesis of membrane lipid components which may ameliorate neuronal functions or its ability to maintain brain metabolism during ischaemia, as the catabolism of pyrimidines produces citric acid cycle intermediates. 59 Uridine’s neuroprotective action may also be related to the stimulation of P2Y receptors by the ligands UTP and UDP, previously shown to reduce apoptosis. 60
Pyrimidine synthesis is either recycled via a salvage pathway from normal catabolism or de novo through enzymes in the orotate pathway. De novo synthesis is the major source of UMP, UDP and UTP in the highly active tissue of the developing brain, though its use gradually declines throughout lifespan making the salvage pathway more important in adulthood. 61 Van Os et al. 62 measured spinal fluid purine and pyrimidine metabolites of hypoxic near term lambs. In agreement with our findings, they found the concentration of the pyrimidine metabolites uridine, pseudouridine and uracil were significantly higher in these lambs than controls, and corresponded with decreased electrocortical brain activity. Though potentially promoting protective mechanism, the accumulation of these pyrimidines during hypoxemia may also be attributed to the absence of high-energy phosphate compounds (ATP) needed to synthesise derivatives such as UTP and UDP. 63
Early studies measuring pyrimidine and purine metabolites (e.g. ATP, GTP, UTP, and CTP) in rodents discovered a marked decrease in metabolite levels compared with controls. Pyrimidines UTP and CTP displayed the greatest depletion, UTP having the slowest, most incomplete restoration to normal levels and it was the total uridine nucleotide pool that had the largest decline of 53%. 64 As mentioned the decline in these compounds may be due to depletion of the ATP needed for their formation. Their delayed restoration could indicate decreased synthesis or perhaps increased consumption. Similarly, in neonatal HIE we have found a significant decrease in UTP and dUTP. Finally, sugar conjugates of pyrimidines can modulate cellular and tissue pathophysiology, through interactions with G protein coupled receptors. We detected raised levels of UDP sugar conjugates UDP-D-xylose or UDP-L-arabinose (>2 putative annotations) in perinatal asphyxia and higher again in HIE and significant alterations in n-acetylneuraminate, a nucleotide sugar metabolism compound, in severe HIE compared to other grades.
Amino acids
Amino acids were another particular class of metabolites found disrupted in this cohort, and their alterations in both animal models of injury and infants have been previously reported.65–67 Pathway analysis revealed metabolites involved in the biosynthesis and metabolism of phenylalanine, tryptophan and tyrosine were disturbed particularly in both perinatal asphyxia and HIE. These interlinking amino acids are precursors to acetyl-CoA, the driving metabolite in the TCA cycle of energy production, whose derailment in perinatal asphyxia has been well documented. 8 Phenylalanine, an essential amino acid, synthesises tyrosine using the catalytic enzyme phenylalanine hydroxylase, and both are intrinsically linked to tryptophan. Tyrosine easily passes the blood–brain barrier and in dopaminergic cells of the brain, acts as the precursor for the neurotransmitters dopamine, norepinephrine and epinephrine. D-Erythrose-4-phosphate, an intermediate of phenylalanine, tyrosine and tryptophan biosynthesis and also an important intermediate in the pentose phosphate pathway, was found to be significantly altered in this cohort, particularly in mild and moderate HIE, therefore may have potential as a biomarker of injury in these infants. Consistent with this study, increased levels of cord blood phenylalanine and tyrosine have previously been detected in both arms of this cohort.10,11 It has also been documented that hypothermia can suppress tyrosine levels. 68
Several other amino acids were also putatively identified as altered in the current analysis including: taurine, isoleucine or leucine, arginine, glutamate and glutamine. We have confirmed alterations in taurine levels in perinatal asphyxia. 11 Taurine is an important intracellular metabolite and has been previously reported to be increased in CSF following neonatal HIE, and more recently to be altered in urine sampled for 48 h following perinatal asphyxia.67,69 The branched-chain amino acids (BCAA) leucine and isoleucine have previously displayed alterations in infants with perinatal asphyxia and HIE.10,11 BCAA act as alternate energy sources for the brain and muscles.70,71 Their increase has previously been documented in asphyxiated neonates, 67 while ratios of alanine and glycine to BCAA, in combination with the energy metabolites, correlate with the duration of hypoxia, 53 perhaps illustrating their mobilisation under conditions of reduced ATP production. Another important function of BCAA is their contribution of nitrogen to the glutamate/glutamine cycle. Astrocytes are thought to take up excess glutamate converting it to glutamine, to prevent excitotoxicity. Glutamine is shuttled back to the presynaptic neurons for recycling into glutamate, and the nitrogen taken is returned to astrocytes via branched chain amino acids. 72 We report elevations in cord blood glutamate and minor elevations in glutamine for both perinatal asphyxia and HIE.
Energy and oxidative phosphorylation
A hypoxic-ischaemic injury quickly diminishes the supply of high energy phosphate metabolites such as ATP, while inadequacies in oxygen availability inhibit the normal function of the TCA cycle and electron transport chain. The current study identified alterations in the TCA cycle intermediates: succinate, citrate and oxaloacetate in both perinatal asphyxia and HIE alike. Several metabolomic studies have reported on the accumulation and delayed recovery of these TCA metabolites post insult, as well as: fumarate, malate and α-keto-glutarate.10,57,68,73 Solberg et al. hypothesised that due to reduced ATP availability, these metabolites cannot enter the respiratory chain for energy production and may be cofactors in lactate accumulation. 57 Succinate may also have a role in the stabilisation and accumulation of hypoxia-inducible-factor1-α (HIF1 α), due to inhibition of prolyl-hydoxylase and a shift to anaerobic metabolism. 10
Reducing equivalents (NADH, FADH2) from the TCA cycle are converted to energy via oxidative phosphorylation (OXPHOS). OXPHOS operates via a series of redox reactions, creating a proton(H+) gradient which becomes phosphorylated by ADP and free inorganic phosphate (Pi) to release ATP into the mitochondrial matrix. Both ADP and Pi metabolites were significantly increased in perinatal asphyxia. A minor fold change was seen in the HIE population at birth. Altered brain energy stores whereby OXPHOS is inhibited, in the form of increased Pi and decreased nucleotide triphosphates (ATP and UTP) are a reliable sign of altered cerebral metabolism and hypoxic brain injury demonstrated by magnetic resonance spectroscopy (MRS).74,75
The present study had some limitations. Accurate metabolite annotation or identification is an important challenge facing the metabolomics community, particularly for studies such as this one undertaking untargeted techniques whereby a large number of features are detected. 76 Putative annotation is often necessary due to the extensive cost and technical challenges of identifying large numbers of unknown metabolites. To increase confidence in our identification, we applied a ‘transformation mapping’ approach to associate the empirical formula detected to known metabolic pathways. 26 This revealed several linked metabolites in a single pathway providing a higher level of confidence in the assigned metabolic annotation for each of the individual mass spectral features and enabled us to achieve the primary goal of the study. To translate these finding to the clinic, confirmation of identification and quantification using metabolite standards is needed. Future work will focus on confirming the identity of metabolic features reported with multiple putative identifications including a comparison to authentic chemical standards by LC-MS/MS.
A clear understanding of perinatal asphyxia and HIE at the biochemical level is an important challenge in the field of paediatrics and child health. Metabolomics now provides researchers with the tools to gain further insight into the disease mechanisms, pathophysiology and onset, through examination of metabolic pathway perturbations at the time of birth. This will enable the discovery of potential biochemical markers as well as new diagnostic and therapeutic strategies for neonatal disorders.
This is the first study to employ a highly sensitive and accurate mass, untargeted metabolomic technique to examine the cord blood metabolome of infants with both perinatal asphyxia and HIE. Our focus was to determine which pathways, if any were more significantly altered in HIE due to the more direct clinical application of this knowledge. We have identified novel biochemical pathway changes, for example that of tryptophan and pyrimidine metabolism, which have not previously been elucidated in HIE and PA. Due to the novelty of these findings, the mechanisms and interactions of some pathway metabolites in relation to HIE are unclear; however, some have been detected previously in animal studies. Furthermore, by using a third separate metabolomic approach on this rigorously biobanked and phenotyped cohort, we have confirmed many of our previous findings. With confidence we can now progress these metabolites to the next stage of biomarker validation in an independently recruited validation cohort. Importantly, this study has discovered perturbed metabolic pathways that are more significantly altered in HIE and many which overlap with PA, showing the difficulty of having a single HIE specific marker. However, measuring the interrelated metabolites of these HIE pathways may eventually provide the robust biomarker panel needed for the early prediction of HIE.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NMD is funded by Molecular Medicine Ireland [PRTLI cycle 5 of the Higher Education Authority of Ireland] as part of the Clinical and Translational Research Scholars Programme. The BiHIVE2 Study (Validation of Biomarkers in Hypoxic Ischaemic Encephalopathy) has been funded by the Health Research Board Clinical Scientist Award, CSA/2012/40 (www.medscinet.net/BIHIVE/, ![]() NCT02019147). This work was also supported by a Science Foundation Ireland Research Centre Award (INFANT – 12/RC/2272).
NCT02019147). This work was also supported by a Science Foundation Ireland Research Centre Award (INFANT – 12/RC/2272).
Acknowledgements
The authors wish to thank the midwives and clinical staff of the labour ward and neonatal intensive care unit for their help in identifying potential study candidates, as well as the parents and children of the study for their generous participation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conceived and designed experiments: NMD, JAK, BHW, WBD, DIB, GBB, DMM. Recruited cohort: BHW, GBB, DMM. Performed the experiments: NMD, JAK. Analysed the data: NMD, JAK. Interpreted the data: NMD, JAK, DMM. Contributed reagents/materials/analysis tools: WBD, DIB. Wrote the manuscript: NMD. Provided edits to the manuscript: NMD, JAK, BHW, WBD, DIB, GBB, DMM.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
