Abstract
Acute hypertensive response is a common systemic response to occurrence of intracerebral hemorrhage which has gained unique prominence due to high prevalence and association with hematoma expansion and increased mortality. Presumably, the higher systemic blood pressure predisposes to continued intraparenchymal hemorrhage by transmission of higher pressure to the damaged small arteries and may interact with hemostatic and inflammatory pathways. Therefore, intensive reduction of systolic blood pressure has been evaluated in several clinical trials as a strategy to reduce hematoma expansion and subsequent death and disability. These trials have demonstrated either a small magnitude benefit (second intensive blood pressure reduction in acute cerebral hemorrhage trial and efficacy of nitric oxide in stroke trial) or no benefit (antihypertensive treatment of acute cerebral hemorrhage 2 trial) with intensive systolic blood pressure reduction compared with modest or standard blood pressure reduction. The differences may be explained by the variation in intensity of systolic blood pressure reduction between trials. A treatment threshold of systolic blood pressure of ≥180 mm with the target goal of systolic blood pressure reduction to values between 130 and 150 mm Hg within 6 h of symptom onset may be best supported by current evidence.
Keywords
Intracerebral hemorrhage (ICH) consists of a hematoma within the brain parenchyma usually originating from the branching point of the small penetrating arteries located in the basal ganglia, thalamus, cerebral lobes, pons, and cerebellum. 1 The hematoma progressively enlarges within the first few hours either from continued bleeding from the original source or recruitment of additional points of ruptures via mechanical trauma to adjacent micro vessels. 2 Accumulation of intravenous contrast in brain parenchyma (leakage of contrast from damaged small arteries) in acute period of intracerebral hemorrhage may be surrogate for continued brain parenchymal bleeding.3–6 The pattern of contrast extravasation may consist of a single focal spot, multiple focal spots, or diffuse pattern in and around the hematoma. The hematoma stabilizes (termination of expansion) after reaching a certain size which varies considerably between individuals. Hemostasis is initiated by local activation of hemostatic pathways and mechanical tamponade.7,8 The process of stabilization is time dependent and occurs within first 24 h. Several studies9,10 have studied the time course of hematoma expansion and stabilization. Hematoma expansion occurred in 73% of 218 intracerebral hemorrhage patients who were studied within 3 h from symptom onset in a pooled analysis using placebo arm of factor seven for acute hemorrhagic stroke (FAST) trial and Cincinnati study. 11 In one study recruiting 98 patients with intracerebral hemorrhage within 3 h of symptom onset, 12 hematoma growth ≥33% was observed in 28% within the first hour and in 37% by 20 h after baseline evaluation. Kazui et al. 13 demonstrated the highest rate of hematoma expansion occurred within the first 3 h from symptom onset with 36% of patients showing hematoma expansion compared to 16% within 3 to 6 h and 15% rate within 6 to 12 h in 204 patients with spontaneous intracerebral hemorrhage.
Acute hypertensive response
Acute hypertensive response is a common systemic response to occurrence of intracerebral hemorrhage. While acute hypertensive response may occur more frequently and of a greater magnitude in those with chronic hypertension, the response is specifically a transient elevation above normal and premorbid values that occurs within the first 24 h of symptom onset.14,15 Acute hypertensive response is a transient response with spontaneous reduction of blood pressure in patients with intracerebral hemorrhage within few days.16–18 The presence of reversible intracerebral hemorrhage specific etiology for this phenomenon such as damage to the areas of the brain involved in regulation of blood pressure including insula, cingulate cortex, amygdala, prefrontal area, or compression over brainstem from intracerebral hemorrhage and increased intracranial pressure (ICP) has been proposed.15–21
Autonomic dysregulation which is a combination of increased sympathetic activity and decreased baroreceptor reflexes is an important contributor to acute hypertensive response.22–23 The decreased baroreceptor sensitivity manifests as prominent blood pressure variability. 24 Impaired baroreceptor reflexes have been identified in patients with intraventricular hemorrhage 25 and those with involvement of the insular cortex. 26 Direct compression and impairment of insular cortex and circuits connecting within amygdala and cingulate gyrus 27 or activation of sympathetic excitatory neurons of the rostral ventrolateral medulla due to distortion 28 may be involved. The transient occurrence of this phenomenon suggests possible functional adaption during the following few days after primary insult to vulnerable areas of the brain.27,29 Brain angiotensin II 30 and nitric oxide 31 have been implicated in inhibiting sympathetic nerve activity and thus control of blood pressure may be altered in intracerebral hemorrhage. 32 In experimental models of intracerebral hemorrhage, serum norepinephrine is elevated at 6 h after cerebral hematoma formation and reach peak levels at 24 h. 33 Serum aldosterone, epinephrine, and norepinephrine are elevated in patients with intracerebral hemorrhage who have severe neurological deficits with a gradual return to normal values by day 3 to day 7 of hospitalization. 34 Urinary catecholamines, normetanephrine, and metanephrine are significantly elevated with maximum urinary catecholamine metabolite levels detected between day 3 and 10 after symptom onset.35,36 However, the relationship between elevated serum norepinephrine levels and systolic blood pressure elevation in intracerebral hemorrhage patients is not strongly correlated. 37
Acute hypertensive response has gained unique prominence in patients with intracerebral hemorrhage due to very high prevalence (seen in almost 75% of patients)10,38 and association with hematoma expansion and increased mortality. 39 Elevation of systemic blood pressure at the time of hematoma is associated with increased rates of hematoma expansion. 40 Presumably, the higher systemic blood pressure predisposes to continued intraparenchymal hemorrhage by transmission of higher pressure to the damaged small arteries. Acute hypertensive response may increase angiotensin II-mediated oxidative stress and activation of matrix metalloproteases in cerebral vessels in region of intracerebral hemorrhage leading to increased bleeding.41,42
Interaction between acute hypertensive response and pathophysiological processes involved in intracerebral hemorrhage
The pattern of systemic blood pressure response was assessed in rats with renovascular hypertension and normotensive rats in a rat model of collagenase-induced intracerebral hemorrhage. 40 Following induction of intracerebral hemorrhage, both the normotensive and hypertensive rats demonstrated an elevation in systemic blood pressure. However, the magnitude of elevation was much greater in hypertensive rats (mean arterial pressure 139 mm Hg versus 90 mm Hg at 30 min post occurrence). The hematoma volume (quantified at 24 h using a spectrophotometric assay) was 60% greater in hypertensive rats who also demonstrated prominent acute elevation in blood pressure. Elevated blood pressure was associated with increased hematoma volume, brain swelling, and apoptosis in the perihematomal areas. The increased parameters of brain damage correlated with more severe neurological deficits (assessed by modified limb placing tests) at three weeks. There may be additional interaction between elevated systemic blood pressure and hemostatic system. The role of hemostatic factors in achieving stabilization of hematoma is supported by higher rates of hematoma expansion and continuing over a longer period after symptom onset in patients using oral anticoagulant 43,44 and antiplatelet medications. 45 Furthermore, administration of hemostatic factors such as intravenous recombinant factor VII reduces the rate of hematoma expansion. 46
The systemic and tissue (heart, aorta, lung, and kidney) renin angiotensin system activity increases in hypertensive state. 47 The renin angiotensin system regulates Angiotensin II, a potent vasoconstrictor, which increases blood pressure and cerebral blood flow. Recently it has been shown that Angiotensin II enhances prothrombotic activity and promotes in vivo thrombosis.48,49 There is a possibility that secondary pathologic processes such as edema, apoptosis and necrosis, and inflammatory cells in the perihematoma region may be influenced by acute hypertensive response. 50 The injury may be initiated in three steps: (1) mechanical disruption of the neurons and glial; 1 (2) mechanical deformation causing oligemia, neuro-transmitter release, mitochondrial dysfunction, and membrane depolarization.51–53 (temporary metabolic suppression (hibernation phase) to cellular swelling and necrosis depending on severity of mitochondrial dysfunction); and, (3) a secondary cascade initiated by products of coagulation and hemoglobin breakdown, in particular thrombin, which activate microglia by 4 h after injury.54–57 There is an inflammatory response characterized by the infiltration of peripheral neutrophils and macrophages and the activation of brain-resident microglia and astrocytes. 58 After intracerebral hemorrhage, blood-derived inflammatory monocytes 59 constitute the major blood-derived inflammatory cells peaking at three days and lasting up to seven days. Subsequently, these cells produce pro-inflammatory cytokines such as tissue necrosis factors. Circulating levels of renin, aldosterone, and angiotensin II are elevated in the early phase of hypertension. 60–62 Angiotensin I receptors are located in neurons, microglia and astrocytes and vascular endothelial cells of the blood–brain barrier and modulate local inflammation.63–65 and may play a role in modulating the inflammatory response in intracerebral hemorrhage. There is some evidence that systemic blood pressure elevation may lead to secondary increase in norepinephrine concentrations in subfornix. 66 Noradrenaline acting at beta-adrenoceptors induces expression of interleukin-1 beta in rat cortex67,68 and the β2-adrenoceptor agonists have anti-inflammatory action 69 and reduce apoptosis in the brain. 70 In ischemic stroke models, beta-adrenoreceptor antagonists (selective beta-adrenoreceptor 1 antagonists) provide neuroprotective effects.71,72
Definition of acute hypertensive response
Acute hypertensive response is defined as systolic blood pressure ≥140 mm Hg consistent with the 2003 World Health Organization/ International Society of Hypertension statement.39,73 However, the relationship between acute hypertensive response and hematoma enlargement 10 and mortality 39 is evident at higher systolic blood pressures. In one study, the mortality rate was 44% in patients with intracerebral hemorrhage and initial systolic blood pressure >196 mm Hg group compared with 18% in those with ≤196 mm Hg. 39 Systolic blood pressure ≥200 mm Hg was seen in 46% and 26% of patients with or without hematoma enlargement in another study. 10 Systolic blood pressure on admission of ≥200 mm Hg independently predisposed subjects to enlargement in multivariate analysis. The Intensive blood pressure reduction in acute cerebral hemorrhage trial (INTERACT) 74 reported that initial systolic blood pressure ≥181 mm Hg was observed in 47% of the 404 patients recruited to the study. Early intensive blood pressure reduction resulted in the most prominent reduction in hematoma expansion in intracerebral hemorrhage patients with an initial systolic blood pressure ≥181 mm Hg.10,39,74 Therefore, a systolic blood pressure value of 180 mm Hg or greater may be a better definition for acute hypertensive response from both clinical relevance and use as treatment threshold.75,76 Systolic blood pressure is preferentially used to define acute hypertensive response because the present evidence supports the association between systolic blood pressure and hematoma expansion 10 without any clear relationship with diastolic blood pressure. Systolic blood pressure is more sensitive representation of variability and changes in systemic blood pressure values obtained in patients with intracerebral hemorrhage. 77
Treatment of acute hypertensive response
Summary of randomized clinical trials evaluating treatment of acute hypertensive response in patients with intracerebral hemorrhage.
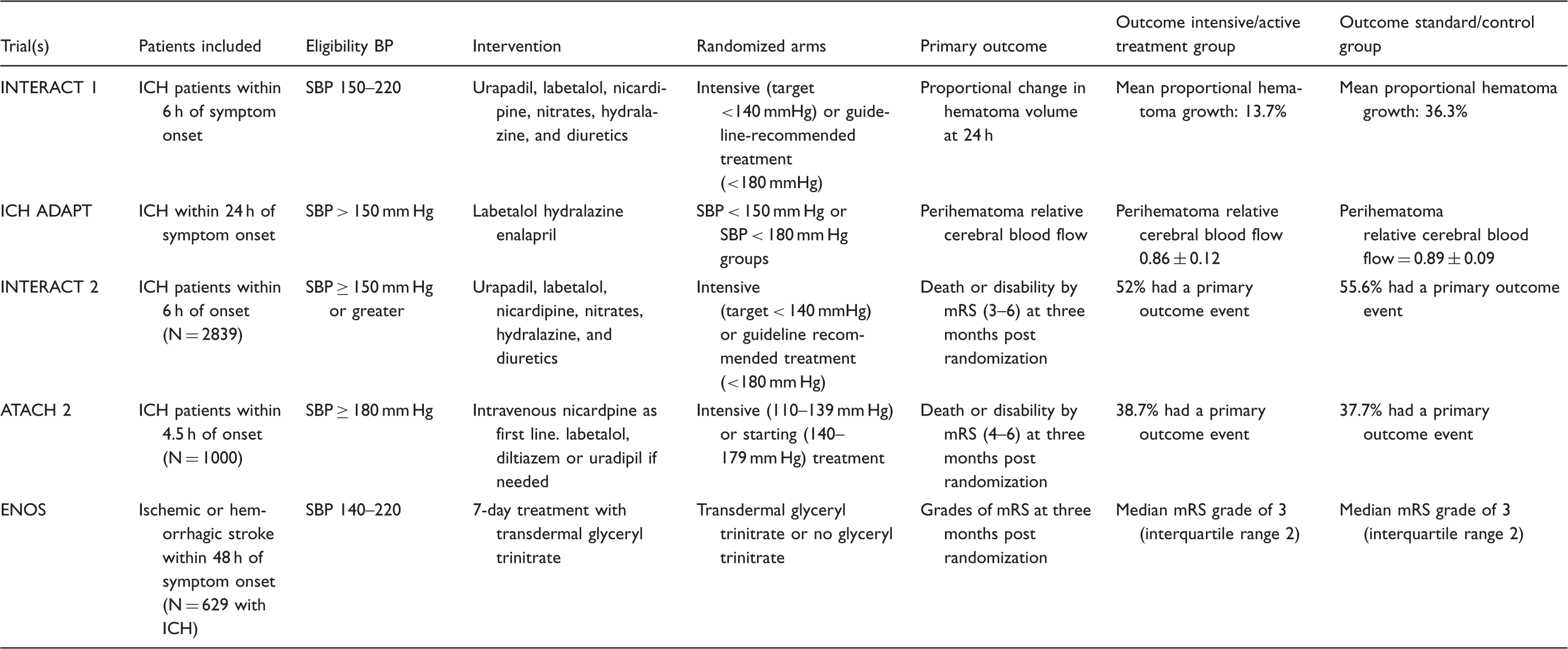
INTERACT: intensive blood pressure reduction in acute cerebral hemorrhage trial; ATACH: antihypertensive treatment of acute cerebral hemorrhage; FAST: Factor seven for acute hemorrhagic stroke; ENOS: efficacy of nitric oxide in stroke; SAE: serious adverse event; SBP: systolic blood pressure; mRS: modified Rankin scale; CT: computerized tomography; ICH: intracerebral hemorrhage.
Pilot studies
The pilot studies have provided data regarding safety of intensive or aggressive systolic blood pressure lowering in patients with intracerebral hemorrhage and preliminary data supporting reduction of hematoma expansion with such strategies.
The intensive blood pressure reduction in acute cerebral hemorrhage trial (INTERACT) randomized patients who had acute intracerebral hemorrhage diagnosed within 6 h of onset, and elevated systolic blood pressure (150–220 mm Hg) to early intensive lowering of blood pressure (target systolic blood pressure 140 mm Hg; n = 203) or standard guideline-based management of blood pressure (target systolic blood pressure 180 mm Hg; n = 201). 74 At 1 h post randomization, mean systolic blood pressure was 153 mm Hg in the intensive group and 167 mm Hg in the guideline group (difference 13.3 mm Hg); from 1 h to 24 h post randomization, 146 mm Hg and 157 mm Hg (difference 10.8 mm Hg) in the intensive and guideline groups, respectively. Mean proportional hematoma growth was 36.3% in the guideline group and 13.7% in the intensive group (P = 0.04) at 24 h. Relative risk reduction of 36% and absolute risk reduction of 8% in hematoma growth (defined as ≥33% or ≥12.5 cubic centimeter (cc) from baseline value) were seen in the intensive group compared with the guideline group. There was no difference in the rates of adverse events between the two groups. A more detailed analysis of 296 patients who had all 3 computed tomography scans available for the hematoma. 79 found an adjusted mean absolute increase in hematoma volumes at 24 were 2.40 cc centimeter in the guideline group compared with −0.74 cc in the intensive group, respectively (an overall difference of 2.80 cc). In INTERACT I, there was a significant association between achieved on-treatment systolic blood pressure levels in the first 24 h and both the absolute or proportional hematoma expansion in hematoma volume. 80 Lowest rates of in hematoma expansion occurred in the one-third of participants with the lowest on-treatment systolic blood pressure levels (median: 135 mm Hg).
The Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH) study recruited 60 patients having intracerebral hemorrhage with an elevated systolic blood pressure of at least 170 mm Hg who were seen within 6 h of symptom onset 81 and administered intravenous nicardipine to reduce systolic blood pressure to a target of: (1) 170 to 200 mm Hg in the first cohort of patients; (2) 140 to 170 mm Hg in the second cohort; and (3) 110 to 140 mm Hg in the third cohort. The rates of neurological deterioration and serious adverse events were lower than expected based on pre specified thresholds. In secondary analysis, the magnitude of systolic blood pressure reduction and not just the initiation of targeted to treatment goal specified was associated with lower rates of hematoma expansion and death and disability at three months. 82 The rates were 21% vs. 31% for hematoma expansion, and 35% vs. 48% for poor three-month outcome in patients with systolic blood pressure reduction of >54 mm Hg compared with those with ≤54 mm Hg at 2 h post recruitment. Using 24-h median area under the curve (AUC) (calculated as the area between the hourly systolic blood pressure measurements over 24 h and the baseline systolic blood pressure) to provide a comprehensive systolic blood pressure profile also suggested that benefit was greater with larger systolic blood pressure reduction frequencies were 32% vs. 17% for hematoma expansion, and 46% vs. 38% for poor three-month outcome (P > .05 for all).
In the intracerebral hemorrhage acutely decreasing arterial pressure trial (ICH ADAPT) 83 randomized patients with intracerebral hemorrhage within 24 h of symptom onset and systolic blood pressure> 150 mm Hg to an intravenous antihypertensive treatment protocol targeting a systolic blood pressure of <150 mm Hg (n= 39) or< 180 mm Hg (n= 36). Mean systolic blood pressure 2 h after randomization was significantly lower in the< 150 mm Hg target group (140 ± 19 vs. 162 ±12 mm Hg) despite a similar baseline systolic blood pressure. The perihematoma relative cerebral blood flow in the patients in < 150 mm Hg target group (0.86 ± 0.12) was not significantly different compared with those in the < 180 mm Hg group (0.89 ± 0.09). There was no relationship between the magnitude of blood pressure change and perihematoma relative cerebral blood flow in either of the groups.
Phase III studies
INTERACT 2
The INTERACT 2 randomized 2839 intracerebral hemorrhage patients and systolic blood pressure measurements of ≥150 and ≤220 mm Hg within 6 h of symptom onset
84
to either intensive treatment with a target systolic blood pressure level of < 140 mm Hg within 1 h, or standard (guideline-recommended) treatment, with a target systolic blood pressure level of <180 mm Hg using a standardized, blood pressure lowering protocols using available intravenous medications.. The agents included alpha-adrenergic antagonist (urapidil (n = 645)), calcium-channel blocker (nicardipine or nimodipine (n = 349)), combined alpha- and beta-blocker (labetalol (n = 285) and nitroglycerin (n = 268)), diuretics (furosemide (n = 268), nitroprusside (n = 197), hydralazine (n = 132)), and other medication (n = 129). The median time from symptom onset to start of treatment was 4.0 h and 4.5 h in subjects randomized to intensive and standard treatments, respectively. The mean systolic blood pressure at 1 h was 150 mm Hg in the intensive systolic blood pressure reduction group (with 462 patients [33.4%] achieving the target systolic blood pressure of < 140 mm Hg) as compared with 164 mm Hg in the standard-treatment group (a difference of 14 mm Hg). The rate of death or major disability defined by a modified Rankin scale (mRS) score of 3 to 6 at three months post-randomization was determined for 2794 participants (see Figure 1). The proportion of patients with death or major disability was 719 of 1382 (52%) in the group randomized to receive intensive treatment compared with 785 of 1412 (55.6%) in the group randomized to receive standard treatment (odds ratio [OR] with intensive treatment, 0.87; 95% confidence interval [CI], 0.75 to 1.01; P = 0.06). When modified Rankin scale grades were analyzed as an ordinal scale, significantly lower modified Rankin scale scores were observed in patients randomized to intensive blood pressure reduction (common OR, 0.87; 95% CI, 0.77 to 1.00; P = 0.04). Nonfatal serious adverse events occurred in 23% and 24% of the subjects randomized to the two groups.85,86 For sensitivity purposes, the primary outcome in INTERACT 2 was analyzed after adjustment for randomization strata and prognostic baseline variables (age, region, National Institutes of Health Stroke Scale score, time from onset of the intracranial hemorrhage to randomization, volume and location of the hematoma, and presence or absence of intraventricular hemorrhage).
87
The adjusted OR was 0.87 with a 95% CI 0.75–1.04, P = 0.122, which was reduction in significance of the effect of randomization to intensive systolic blood pressure reduction suggesting a confounding effect of certain covariates. The OR (95% CI) for unadjusted ordinal regression analysis was 0.87 (0.77–1.00), P = 0.044, but decreased to 0.89 (0.78–1.02), P = 0.11 after adjustment for covariates.
The distribution of modified Rankin scale scores according to treatment groups in large randomized clinical trials.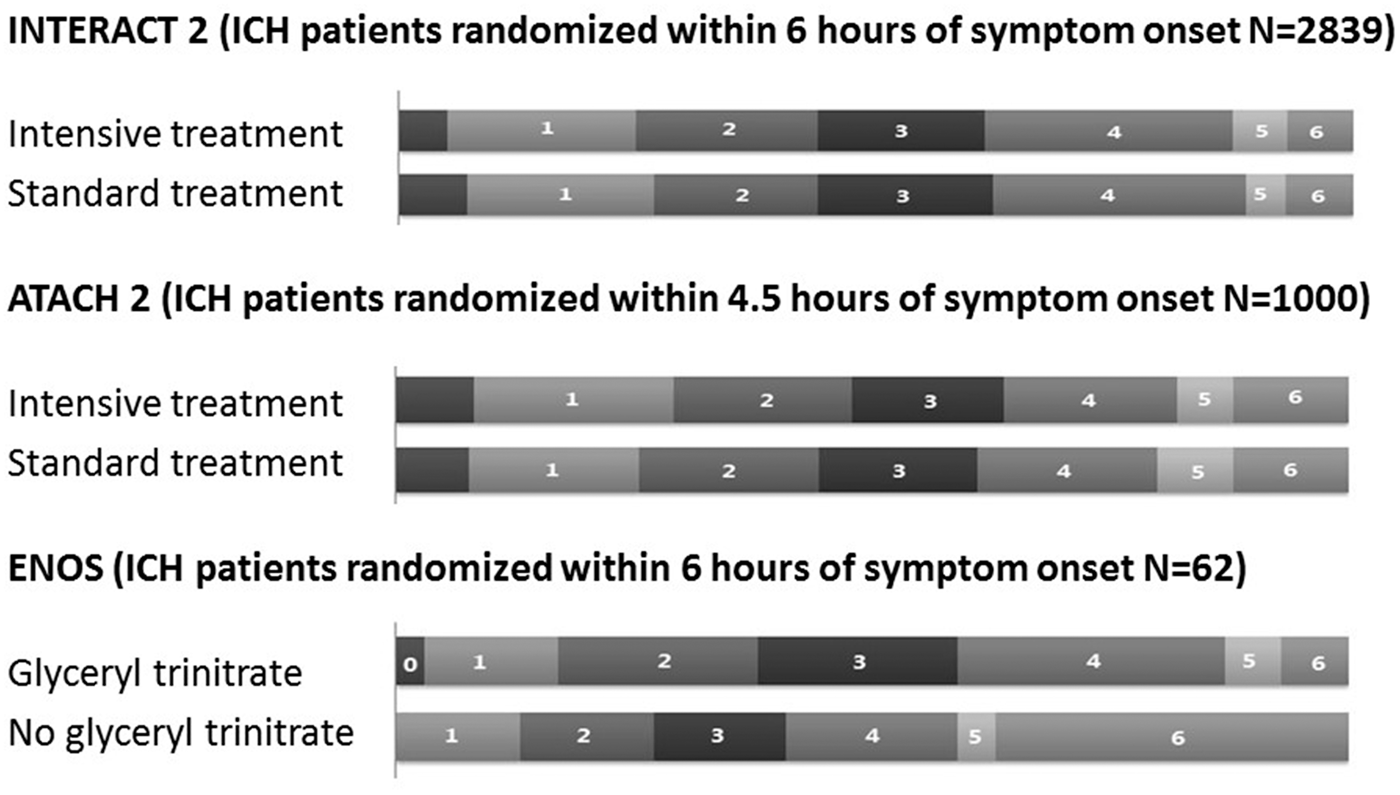
In a follow-up analysis, 88 associations of systolic blood pressure reduction (baseline minus average of achieved systolic blood pressure) during three time periods post randomization (15–60 min, 1–24 h, and 2–7 days) on poor outcome (death or major disability) at 90 days were analyzed. Compared with minimal reduction at 15–60 min (<10 mmHg), OR were 0.80 for moderate (10–20 mmHg) and 0.65 for large (≥20 mmHg) reductions (P trend < 0.01). Similar associations were also observed for systolic blood pressure reductions during 1 to 24 h (P < 0.01) and two to seven days (P = 0.02). Optimal recovery from intracerebral hemorrhage was observed in hypertensive patients who achieved the greatest systolic blood pressure reductions (≥20 mmHg) in the first hour and maintained for seven days. Patients were further stratified by baseline systolic blood pressure into two groups: <180 and ≥180 mmHg. There was no significant difference in associations between systolic blood pressure reduction and poor outcome between patients with baseline systolic blood pressure levels above or below 180 mmHg for any of the three time periods
In another follow-up analysis, 89 intensive blood pressure lowering produced comparable benefits on modified Rankin scale grades at 90 days in five subgroups defined by baseline systolic blood pressure of <160, 160–169, 170–179, 180–189, and ≥190 mm Hg. The means of achieved post-randomization blood pressure at 1, 6, 12, 18, and 24 h post-randomization, and the means of these five measurements were correlated with disability. Analyses of achieved blood pressure showed linear increases in the risk of disability for achieved systolic blood pressure above 130 mm Hg, while modest increases were also observed for achieved systolic blood pressure below 130 mm Hg. Compared with achieved systolic blood pressure of 130 mm Hg, adjusted OR for poor outcome were 1.21 at 140 mm Hg, 1.42 at 150 mm Hg, 1.57 at 160 mm Hg, 2.19 at 170 mm Hg, and 3.04 at 180 mm Hg. However, the pre randomization systolic blood pressure was lower in patient who achieved systolic blood pressure of 130 mm Hg compared with those who achieved a mean systolic blood pressures. Compared with achieved systolic blood pressure of 130 mm Hg, modest increase in the risk of death or major disability was seen at 120 mm Hg (OR 1.30).
ATACH 2
The ATACH-2 trial randomized 1000 patients with supratentorial intracerebral hemorrhage and systolic blood pressure >180 mm Hg within 4.5 h of symptom onset. 90 The goal of treatment was to reduce and maintain the hourly minimum systolic blood pressure to 140–179 mmHg in the standard treatment group and 110–139 mmHg in the intensive treatment group through the period of 24 h after randomization. After randomization, intravenous nicardipine infusion was the first line antihypertensive agent. If systolic blood pressure was greater than the target, despite infusion of the maximum nicardipine dose for 30 min, a pre-specified second agent, intravenous labetalol, diltiazem or uradipil was used. Enrollment was stopped following a pre-specified interim analysis because of futility. 91 The mean interval between symptom onset and randomization was 182.2 (±57.2) and 184.7 (±56.7) min for intensive and standard treatment groups, respectively. The mean baseline systolic blood pressures were 200 ± 27.1 mm Hg for the intensive and 201.1 ± 26.9 mm Hg for the standard treatment group. The mean minimum systolic blood pressures during the first 2 h were 128.9 (±16) mmHg for the intensive and 141.1 (±14.8) mmHg for the standard treatment group. Primary treatment failure (defined as inability to reach the target systolic blood pressure of <140 mmHg and < 180 mmHg for intensive and standard groups, respectively, within 2 h of randomization) occurred in 61 (12.2%) and 4 (0.8%). Secondary treatment failure (defined as hourly minimum systolic blood pressure remaining greater than the upper limit of the target range for two consecutive hours during 2–24 h after randomization) occurred in 78 (15.6%) and 7 (1.4%) in the intensive and standard treatment groups, respectively.
The proportion of patients with death or disability (modified Rankin scale score of 4–6) at three months was ascertained for 961 subjects. Death or disability was observed in 186 (38.7%) and 181 (37.7%) of subjects in intensive and standard treatment groups, respectively. In the primary analysis using multiple imputation method for missing data, the relative risk (RR) was 1.04 (95% CI: 0.85, 1.27) adjusted for age, initial Glasgow coma scale, and presence/absence of intraventricular hemorrhage. There was no difference between groups in the ordinal distribution of the modified Rankin scale score at three months (common OR 1.07, P = 0.56). The proportion of subjects with hematoma expansion of ≥33% in volume on the computed tomography scan obtained at 24 h after randomization (compared with baseline scan) was 18.9% and 24.4% in patients randomized to intensive and standard treatment, respectively (adjusted RR 0.78, 95% CI 0.58–1.03)
The proportion of treatment-related serious adverse events within 72 h was low (1.6% and 1.2% in intensive and standard treatment groups, respectively). However, the proportion of patients with any serious adverse events during the three months post-randomization was higher in the intensive treatment group (25.6% vs. 20%; adjusted RR; 1.30; 95% CI: 1.00, 1.69). The rate of renal adverse events within seven days after randomization was significantly higher in the intensive treatment group than in the standard-treatment group (9.0% vs. 4.0%, P = 0.002).
The Spot Sign score in restricting intracerebral hemorrhage growth (SCORE-IT) 92 was a prospective observational study nested within the ATACH-2 trial which included 133 patients with a computed tomographic angiography performed within 8 h from onset. In the 53 patients with spot sign (one or more focus of contrast extravasation within the hematoma), 23% and 32% of those randomized to intensive and standard treatment had hematoma expansion. After adjustment for potential confounders, intensive treatment was not associated with a significant reduction of hematoma expansion (RR 0.83, 95% CI 0.27–2.51, P = 0.74) or death or disability (RR1.24, 95% CI 0.53–2.91, P = 0.62).
Efficacy of nitric oxide in stroke trial
In the efficacy of nitric oxide in stroke (ENOS) trial, 93 4011 patients with ischemic or hemorrhagic stroke, who had a systolic blood pressure of 140–220 mm Hg were randomly assigned to seven days of treatment with transdermal glyceryl trinitrate or no glyceryl trinitrate within 48 h of symptom onset. When adjusted for stratification and minimization variables, the distribution of modified Rankin scale between the glyceryl trinitrate group (1993 patients assessed) and the no glyceryl trinitrate group (2002 patients assessed) did not differ (common OR 1·01, 95% CI 0·91–1·13). A total of 629 participants with intracerebral hemorrhage and mean baseline blood pressure of 172.1/93.4 mm Hg were enrolled in the trial; 94 of which 310 participants were randomly assigned to receive glyceryl trinitrate and 319 participants to no glyceryl trinitrate. The blood pressure was significantly lower by 7.5/4.2 mm Hg in patients who received glyceryl trinitrate (P = 0.02/0.05, respectively); At day 90, the median modified Rankin scale was 3 (interquartile range, 2) in both the glyceryl trinitrate and no glyceryl trinitrate groups and did not differ in an adjusted analysis (common OR, 1.04; 95% confidence. interval, 0.78–1.38; Adjusted common OR, 1.04 (95% confidence interval, 0.78–1.38; P = 0.81) in comparison by ordinal logistic regression adjusted for age, sex, premorbid modified Rankin scale, history of previous stroke, history of diabetes mellitus, total anterior circulation syndrome, systolic blood pressure, feeding status, time to randomization, and allocation to continue or stop pre stroke antihypertensive drugs. When adjusted for baseline value, treatment with glyceryl trinitrate was associated with a smaller hematoma volume (mean difference, −4.3 cm3; P = 0.06) in 181 patients who had repeat brain imaging at one week. At day 90, glyceryl trinitrate was associated with an improved functional outcome assessed using the modified Rankin scale, manifest as a shift to less dependency in 61 of 629 patients who were randomized within 6 h of symptom onset (adjusted common OR, 0.19, 95% CI 0.06 0.59, P = 0.004) by ordinal logistic regression adjusted for factors mentioned above.
Comparison of INTERACT 2 and ATACH 2
Intensive systolic blood pressure reduction as applied in INTERACT II resulted in benefit of small magnitude (absolute benefit of 3.6% on rate of severe disability or death) with no effect on hematoma expansion.
95
Antihypertensive treatment in the intensive systolic blood pressure reduction group did not achieve the target systolic blood pressure value of less than 140 mm Hg in a large proportion of patients. The ATACH-2 trial
90
was designed to determine the efficacy of rapidly lowering a systolic blood pressure in patients in an earlier time window after symptom onset than evaluated in previous trials.79,84,95 The trial was based on evidence that hematoma expansion and subsequent death or disability might be reduced with very early and more aggressive reduction in systolic blood pressure80,82 in those at higher risk due to presence of high systolic blood pressure (≥1707 to ≥200 mm Hg
13
) at presentation. All patients in ATACH-2 were randomized and treated within 4.5 h of symptom onset compared with an estimated 41% who were randomized ≥4 h after symptom onset in INTERACT-2. All the patients in ATACH 2 trial were randomized with initial systolic blood pressure ≥180 mmHg compared with only 48% of 2839 subjects in INTERACT-2.
84
The mean minimum systolic blood pressure in the first 2 h after randomization was 128.9 and 141.1 mmHg in intensive and standard treatment groups in ATACH-2, respectively. In INTERACT-2, the mean systolic blood pressure was 150 and 164 mmHg in the first 1 h in intensive and standard treatment groups, respectively (see Figure 2). ATACH 2 trial design had postulated that more rapid intensive systolic blood pressure reduction and exclusion of patients with no requirement for IV antihypertensive medication would increase the likelihood of demonstrating, a larger magnitude of therapeutic benefit, but the results did not confirm this hypothesis.
The systolic blood pressure profiles in patients according to treatment group in INTERACT 2 and ATACH 2 trial. Note that the graph derived from INTERACT 2 is based on average systolic blood pressure and that derived from ATACH 2 is based on minimum systolic blood pressure.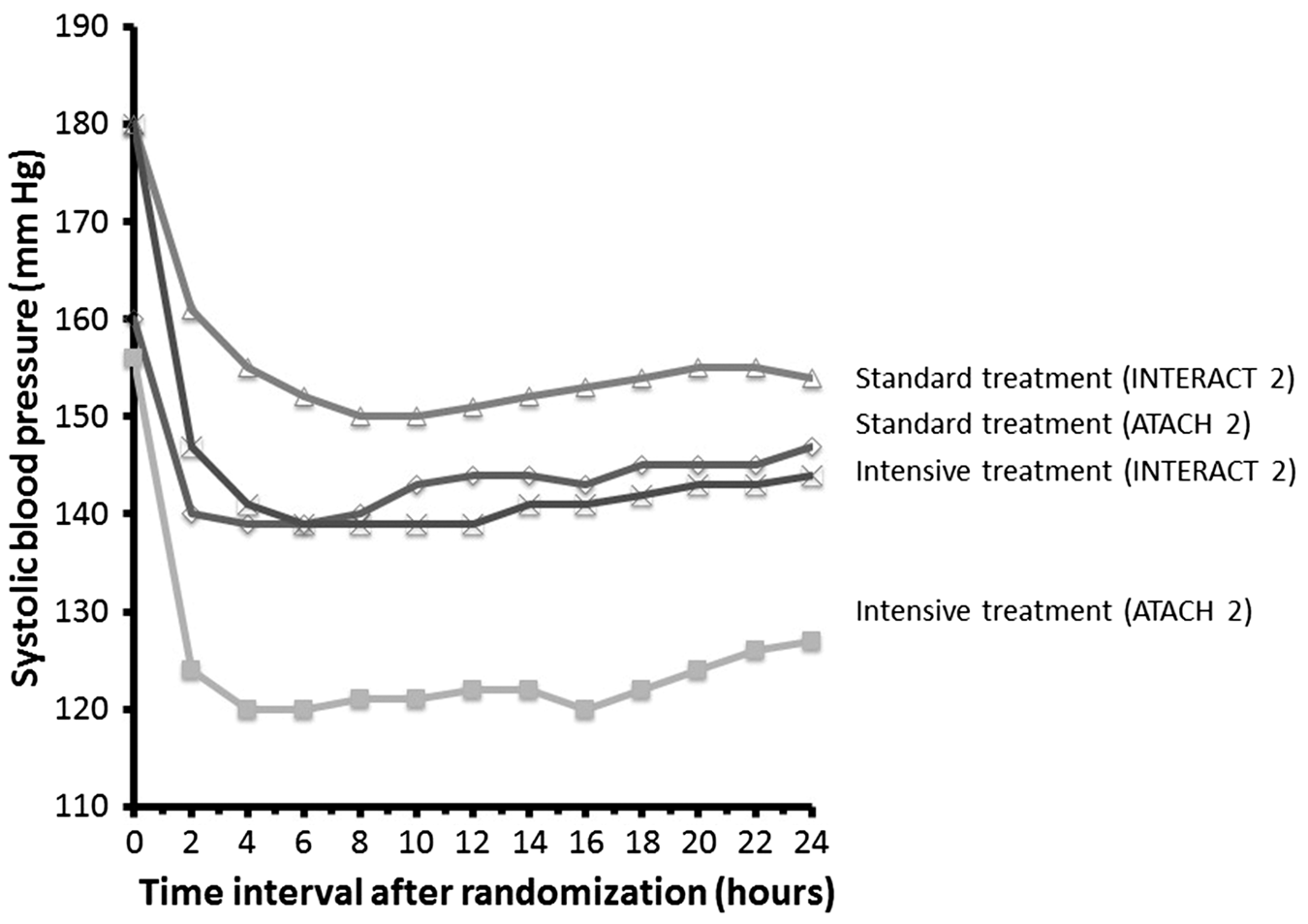
Incorporation of the results in clinical practice
There are three issues that relevant to management of acute hypertensive response: treatment threshold, at what blood pressure value should antihypertensive treatment be initiated; time window, what is the time period after symptom onset when antihypertensive treatment should be initiated; and treatment goal, what is the target blood pressure to be achieved with antihypertensive treatment.
INTERACT 2 included patients with systolic blood pressure measurements of ≥150 Hg. 84 Only 1873 of 2839 (66%) subjects required any intravenous antihypertensive medication; 10% and 57% of subjects randomized to intensive or standard systolic blood pressure reductions, respectively, did not require any intravenous antihypertensive medication. ATACH 2 included patients with systolic blood pressure >180 mmHg and a total of 4/500 and 1/500 patients randomized in the intensive and standard treatment groups did not require any IV antihypertensive medication. Therefore, using a treatment threshold of systolic blood pressure of 180 mm Hg or greater will ensure inclusion of patients who will require pharmacological reduction of systolic blood pressure and are less vulnerable to spontaneous reduction in systolic blood pressure.
The treatment target was one of the primary focus of both INTERACT 2 and ATACH 2. The mean systolic blood pressure at baseline was 150 mm Hg in the intensive systolic blood pressure reduction group (with 462 patients [33.4%] achieving the target systolic blood pressure of <140 mm Hg) at 1 h post randomization. Therefore, the systolic blood pressure profile in intensive treatment group in INTERACT 2 was a reflection of systolic blood pressure values closer to 140 mm Hg rather than lower systolic blood pressure values. A post hoc analysis of INTERACT 2 also demonstrated that patients with post randomization average systolic blood pressure of 130 mm Hg at selected time points within 24 h of randomization had the lowest odds of death and disability at three months. 84 Based on INTERACT 2 results, American Stroke Association guideline states “acute lowering of systolic blood pressure to 140 mm Hg in intracerebral hemorrhage patients with admission systolic blood pressure between 150 and 220 mm Hg who have no contraindication for blood pressure lowering is safe and may improve functional outcome”. 75 The ATACH 2 results suggest that compared with standard treatment (systolic blood pressure mean minimum systolic blood pressures during the first 2 h 141.1 (±14.8) mmHg), intensive systolic blood pressure reduction (128.9 (±16) mmHg) did not show any incremental clinical benefit and was associated with a higher rate of renal related adverse events. Therefore, the treatment goal between systolic blood pressure between 130 and 150 mm Hg may be best supported by current evidence and is relatively consistent with American Stroke Association guidelines.
The time window for treatment of acute hypertensive response is short to reduce hematoma expansion which occurs within the first few hours after symptom onset. The analysis from FAST trial suggested that treatment with recombinant activated factor VII (rfVIIa) can reduce the rate of hematoma expansion when initiated within 4 h from symptom onset. 46 The benefit was augmented when analysis was restricted to patients in whom treatment was initiated within 2.5 h after symptom onset. 96 Compared with placebo treated group, the magnitude of hematoma growth reduction was 7.3 ml if factor VII was initiated within 2.5 h and 3.8 ml when initiated within 4 h after symptom onset. In ATACH 2, the trial initially recruited patients within 3 h after symptom onset, but the recruitment window was extended to 4.5 h 91 based on new data suggesting that the occurrence of hematoma expansion was equally prevalent among patients who presented between 0 and 3 h after symptom onset and those who presented between 3 and 4.5 h after symptom onset. 91 Data from the ATACH 1 82 also supported the hematoma expansion and the rate of death or disability was lower in patients even when systolic blood pressure was reduced within 4.5 h after symptom onset. A time-dependent loss of benefit of intensive reduction in the systolic blood-pressure level in patients treated between 3 and 6 h after symptom onset is possible, although it was not observed in the subgroup analysis of INTERACT 2. 95 In the ENOS, 94 the adjusted OR was 1.04 in patients with intracerebral hemorrhage randomized within 48 h of symptom onset and reduced to 0.19 when only patients within 6 h of symptom onset were analyzed. Therefore, a treatment window of first 6 h for achieving therapeutic benefit is supported by current evidence.7,9,10,91,96
It should be noted that neither INTERACT 2 nor ATACH 2 included adequate number of patients with large intracerebral hemorrhages, ICP elevation, and compromised cerebral perfusion pressure. Therefore, the possibility of precipitating global or regional cerebral hypoperfusion with intensive systolic blood pressure reduction in such patients may still be a concern.
Incorporation of acute hypertensive response treatment in the multifaceted management of patients with intracerebral hemorrhage
It is important to recognize that treatment of acute hypertensive response is one of the multiple medical interventions that influence outcome of patients with intracerebral hemorrhage. Treatment of acute hypertensive response has been subsequently incorporated as 1 of the 26 quality indicators related to 18 facets of care to quantify the appropriateness of medical care. The items were selected due to its high prevalence and emphasis in guidelines from professional organizations. 97 The intracerebral hemorrhage specific intensity of care quality metrics 97 included treatment with intravenous antihypertensive medication within 24 h after emergency department arrival for elevated blood pressure (systolic blood pressure ≥180 mm Hg) as a quality parameter. The performance was scored as 1, if systolic blood pressure target goal is reached within 2.5 h of the second of the two consecutive measurements (systolic blood pressure ≥180 mm Hg) or not applicable, and 0, if systolic blood pressure target range is not achieved. The quality metrics recommended adequate systolic blood pressure control with an infusion for 24 h after initiation of treatment (24–27 h after symptom onset), to provide adequate systolic blood pressure control during the time that hematoma expansion will mostly occur. Early termination of antihypertensive treatment may lead to poor control of systolic blood pressure, with subsequent vulnerability to delayed hematoma expansion.
Conclusions
Although acute hypertensive response is a common occurrence, the current trials have shown small or no treatment benefit with aggressive reduction of systemic blood pressure in patients with intracerebral hemorrhage. In general, there is a consensus that blood pressure should be reduced using intravenous antihypertensive medication in patients with acute hypertensive response. There is a lack of agreement at this point regarding both the treatment threshold and target for blood pressure treatment. The question remains whether further trials focusing on a selected group of patients with intracerebral hemorrhage can identify a greater magnitude benefit with intensive reduction of blood pressure. However, at this point, such a group of patients have not been identified. The possibility that efficacy of intensive reduction of blood pressure may be higher among patients with greater severity of disease (Glasgow coma scale score of 5 to 12) needs further evaluation. Both ATACH-2 and INTERACT 2 had a limited representation of such patients and rates of death or disability were low (regardless of intervention) due to high proportion of patients with high Glasgow coma scale score and small intraparenchymal hemorrhages. The efficacy may be higher among patients with a higher rate of hematoma expansion such as those with contrast extravasation within the hematoma on computed tomographic angiography. 98 Although no significant difference in rates of hematoma expansion between patients treated with intensive and standard treatment was noted in SCORE IT, 92 the number of patients in the analysis was small. It is possible that therapeutic target for systolic blood-pressure lowering may vary between individual patients on the basis of individual cerebral hemodynamics, and optimal benefit may require individualized goals for treatment. 99 Although real-time bedside ascertainment of regional cerebral blood flow and regional ICP measurements is not possible at this time, technological advancements in future may permit individualized treatment. Alternate consideration is that intensive reduction of blood pressure as a sole intervention may not be enough to reduce death or disability rates and must be evaluated in conjunction with another intervention such as surgical evacuation or aspiration of intraparenchymal hematoma 100 or intraventricular thrombolysis and cerebrospinal fluid drainage. 101
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the National Institute of Neurological Disorders and Stroke and the National Cerebral and Cardiovascular Center; ATACH-2 ClinicalTrials.gov number.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
