Abstract
Intracerebral hemorrhage (ICH) is a stroke subtype with high rates of mortality and morbidity. The immune system, particularly complement and cytokine signaling, has been implicated in brain injury after ICH. However, the cellular immunology associated with ICH has been understudied. In this report, we use flow cytometry to quantitatively profile immune cell populations that infiltrate the brain 1 and 4 days post-ICH. At 1 day CD45hi GR-1+ cells were increased 2.0-fold compared with saline controls (
Introduction
Intracerebral hemorrhage (ICH), a bleeding into the brain parenchyma, can occur in neonates, children, or adults either spontaneously or as a result of trauma (Qureshi et al, 2001; Kase and Caplan, 1994). ICH is the stroke subtype with the highest rates of mortality and morbidity; only 38% of patients survive for 1 year (Xi et al, 2006; Foulkes et al, 1988; Qureshi et al, 2001). Breakdown of the blood-brain-barrier (BBB) is well documented in ICH, leading to extravasation of plasma proteins and marked perihematomal interstitial and vasogenic edema (Xi et al, 2006; Liu and Sturner, 1988; Wagner et al, 1996).
There are numerous reports indicating that various immune components are associated with the hemorrhagic milieu after ICH, including proinflammatory cytokines, complement components, microglial activation, and neutrophil infiltration (Xi et al, 2006; Wang and Dore, 2007; Wagner, 2007; Jenkins et al, 1989; Gong et al, 2004). However, there is very little work detailing the types of leukocytes that enter the brain after ICH. Furthermore, studies that do exist are largely limited to immunohistochemical techniques. A more precise assessment of these leukocytes, including determining the frequency of central nervous system (CNS) infiltrating immune cells, is needed to understand the role of the immune system in the pathogenesis of ICH-induced brain injury.
In this report, we are the first to use flow cytometry to quantitatively study the leukocytes that enter the brain after experimental ICH in mice. We report that ICH leads to an increase in immune cell populations, including CD4 T cells and total blood-derived leukocytes, with concomitant alterations in physiologic and behavioral parameters. These data suggest a role for CNS inflammation in ICH.
Materials and methods
Animals
Unless otherwise noted, all materials and reagents were purchased from Sigma (St Louis, MO, USA).
All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC). Male C57BL/6J mice (approximately 20 to 30 g) were purchased from Jackson Laboratories (Bar Harbor, NE, USA). Animals were housed in standard laboratory housing and allowed
ICH Model
Mice were anaesthetized with 2% isoflurane in 24% oxygen and 74% nitrous oxide, administered through an anesthesia mask (Kopf, Tujunga, CA, USA). Deep sedation was monitored throughout the procedure by the absence of pain reflexes in the toes and pupils. Body temperature was held at 37°C with a feedback-controlled heating blanket.
Our experimental ICH procedure was based on a previously described method by Yang et al (2006), with some modifications. Mice were placed in a stereotaxic frame (Kopf). A skin incision was made along the midline of the dorsal surface of the skull, exposing the bregma. A 1 mm cranial burr hole was drilled 2.5 mm lateral and 0.5 mm anterior to the bregma. Approximately 30 μL of autologous blood was collected by clipping a small portion of the distal tail. Further bleeding was prevented by cauterization of the tail. Blood was drawn into a 50 μL Hamilton syringe with a 26-gauge needle. The needle was inserted 4 mm ventral through the cranial hole into the brain and 20 μL of blood or sterile saline was infused more than 15 mins. After infusion, the hole in the skull was filled with dental cement and the incision was closed. Mice were allowed to survive for 1- or 4-days post-ICH, at which time they were euthanized with isoflurane and decapitation.
Brain Water Content Measurement
Mice were euthanized and brains were removed and immediately weighed on an analytical balance. Brains were desiccated by heating at 85°C for 16 h and weighed again. Whole-brain water content was measured as a marker of edema. Brain water content was calculated according to the following formula: Brain water content = (wet weight—dry weight)/wet weight (Yang et al, 2006).
Motor Deficit
Motor deficits were assessed using the rotarod task (Jeong et al, 2003). The amount of time mice were able to stay on the top of the rotating rod (32 r.p.m., constant speed) was measured before surgery and again before euthanasia. The percent change from presurgery to the time before euthanasia was calculated. This value was used in the comparison between the blood infused and saline-infused groups.
Isolation of Central Nervous System Immunologic Cells
Cells were isolated as described previously (Kang et al, 2002). Briefly, excised mouse brains were pushed through a nylon cell strainer (BD Biosciences, Franklin Lakes, NJ, USA) with a 100-μ pore width and incubated at 37°C for 45 mins in 250 μg/mL collagenase type 4 (Worthington Biochemical Corp., Lakewood NJ, USA). Immunologic cells were then concentrated via continuous Percoll gradient (Pharmacia, Piscataway, NJ, USA) centrifugation at 10,000 r.p.m. (Sorvall SS-34 rotor) for 30 mins. The resulting layer of immunologic cells was then collected and placed into a 50 μL falcon tube. RPMI (1 ×) was added until the final volume was 50 mL and the solution was then centrifuged at 1500 r.p.m. (Sorvall Legend RC centrifuge) for 10 mins. The resulting cell pellet was resuspended in fluorescence-activated cell sorting buffer (1% bovine serum albumin and 2% sodium azide).
It is important to note that the hematoma was filtered out of cell suspensions before flow cytometric analysis. Therefore, flow cytometric analysis was not influenced by cells potentially residing within the hematoma itself. Further, it is unlikely that cells would remain viable 1 or 4 days post-ICH within a blood clot. Thus the cell populations observed are highly likely to only be leukocytes that infiltrated into the parenchyma from the periphery or from the hematoma during retraction of the clot.
Flow Cytometric Analysis
Cells isolated from the CNS were stained with anti-CD4 PE, anti-CD8 FITC, anti-CD3 APC, anti-CD45 PE-Cy7, and anti-Gr-1 APC-Cy7 on ice for 45 mins. Samples were then washed twice with fluorescence-activated cell sorting buffer, resuspended in cold phosphate-buffered saline, and fixed in 1% paraformaldehyde. Samples were then analyzed on a BD LSRII instrument (BD Biosciences).
Raw data were displayed with side scatter along the
Statistical Analysis
Data were analyzed with the statistical package in Microsoft Excel using Student's
Results
To verify our ICH model, we fixed and sectioned some of the brains to inspect the hematoma. Figure 1A shows a representative brain, cut in 2 mm serial sections. Visual examination of the brain reveals a mild to moderate hemorrhage with the hematoma centered in the anterolateral portion of the left hemisphere extending into the striatum. There is also some observable peri-hematomal edema.
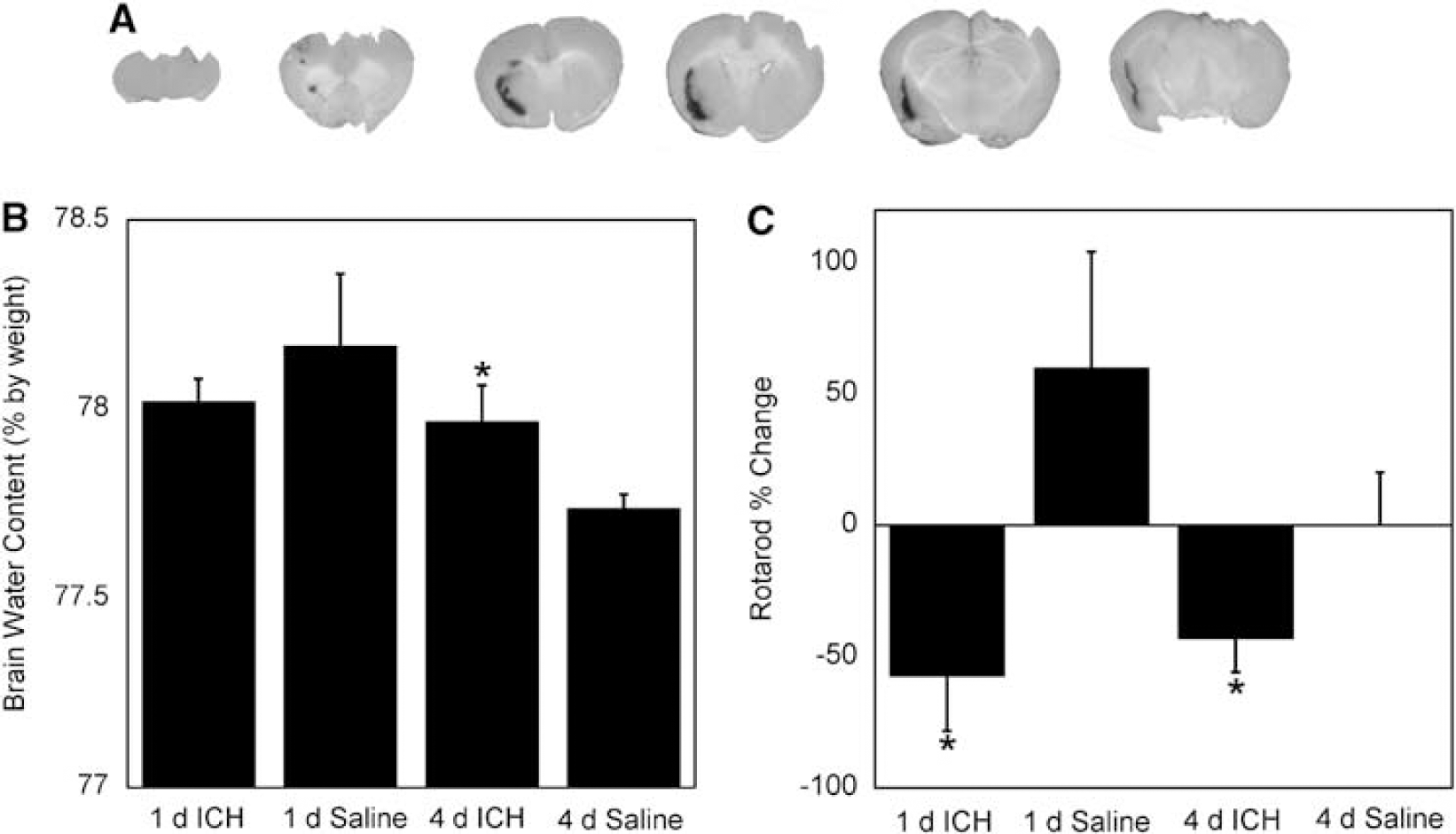
Physiologic and behavior changes associated with intracerebral hemorrhage (ICH). (
Figure 1B shows the results of whole-brain water content measurements, used as a marker of edema. At 4-day ICH, animals had a small, but statistically significant, increase in brain water content compared with saline controls (78.0 versus 77.7%;
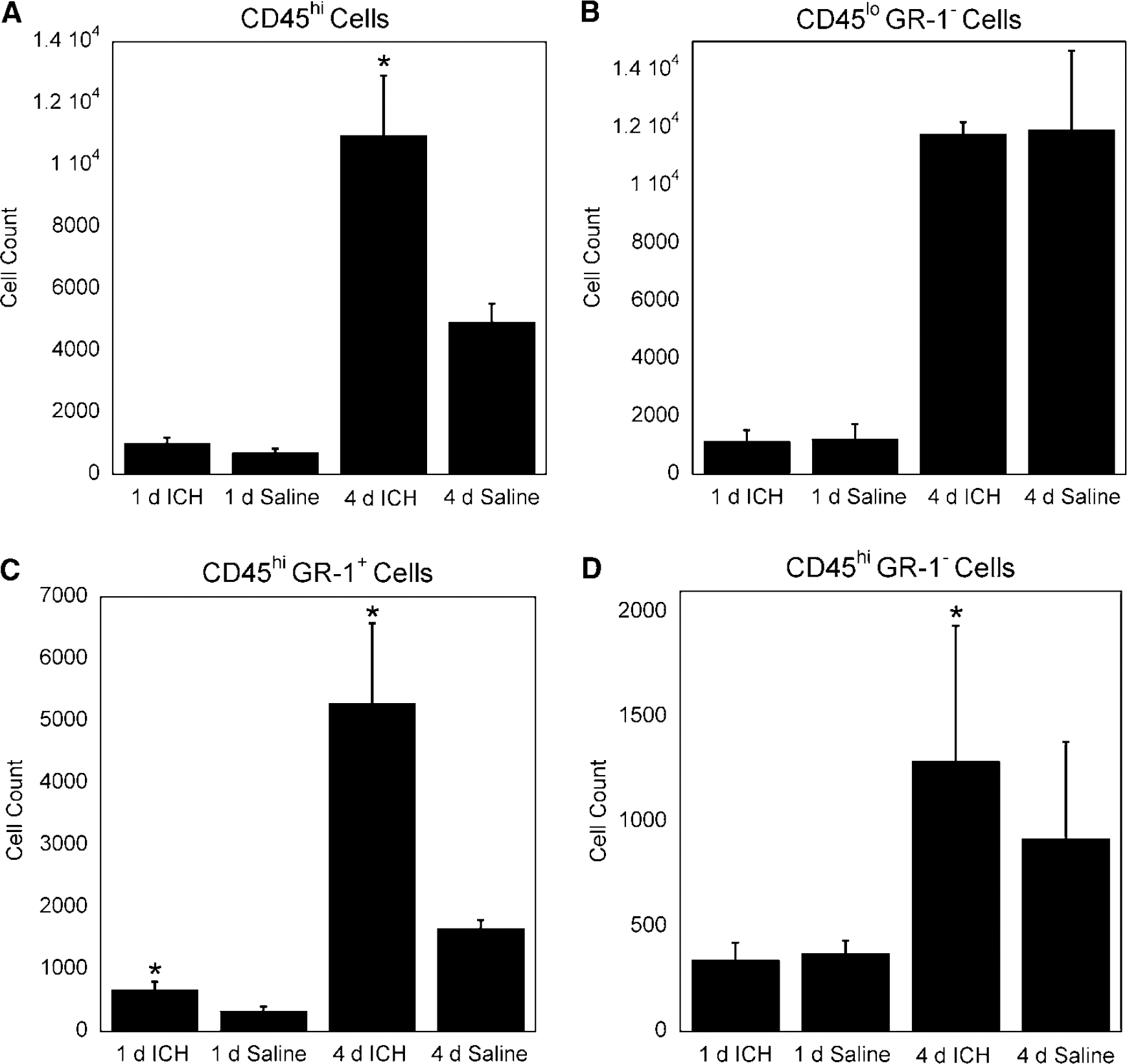
An immune response after ICH in humans and animal models has been noted (Xi et al, 2006; Gong et al, 2000; Xue and Del Bigio, 2000, 2005). We wished to characterize the immune response in our ICH model 1 and 4 days post-ICH because important clinical sequelae begin to appear between 1 and 5 days post-ICH (Qureshi et al, 2001). Using flow cytometric analysis we have showed that similar leukocyte populations were present in the brains of both saline and ICH mice; however, the numbers specific inflammatory cell types differed in ICH mice as compared with saline controls. At 1 day, we observed an increase in CD45hi GR-1* cells (
Discussion
In this study, we have examined leukocyte trafficking into the CNS after ICH. Our goal was to use a quantitative method to define the inflammatory cell infiltrate after ICH and to associate this leukocyte infiltration with pathophysiological data. There is a paucity of flow cytometric data in the literature defining the specific inflammatory cell types that infiltrate into the parenchyma during brain inflammation. However, multiple sclerosis has been characterized with regard to leukocyte infiltration. In contrast to our findings with ICH, CD8+ T cells traffic into the brain, in addition to other cell types, during multiple sclerosis (Johnson et al, 1999).
Leukocytes, particularly T cells, traffic into the CNS under normal physiologic conditions. However, under these conditions, cells rarely enter the brain parenchyma and do not remain in the CNS for long periods of time unless they experience antigen and become activated (Ransohoff et al, 2003; Hickey, 1999; Engelhardt and Ransohoff, 2005). Immune trafficking into the brain parenchyma as a part of the stroke pathologic assessment has gone relatively unstudied. A previous report has used histochemical and immunohistochemical methods to show the presence of neutrophils, microglia, and CD8a+ cells in the brain after experimental ICH in the rat (Xue and Del Bigio, 2000). However, our use of flow cytometry extends these observations and enables one to rapidly identify and quantify these cell types, as well as their ratios to one another.
There are two possible sources of leukocytes found in the brain parenchyma after ICH. Some cells will migrate from the hematoma into the perihematomal tissue and others will enter through an open BBB (Xi et al, 2006). As noted previously, we observed similar populations, but different numbers of immune cells among ICH and saline mice. This is likely because of the fact that the saline treatment produces a small amount of brain injury as a result of the needle tracks, microhemorrhages, and mass effect from the volume of the injection.
We chose 1 and 4 days time points for this analysis based on the amount of erythrocyte lysis and subsequent inflammatory stimuli. In rodent models of ICH, erythrocyte lysis is low at 1 day and high at 4 days (Wagner et al, 2003). Because erythrocyte products may contribute to inflammation, this is a logical choice of time points. Our observation of increased T cells at 4 days but not 1 day suggests recruitment from the periphery as opposed to the hematoma.
A significant increase in CD45hi GR-1+ cells at 1 and 4 days was observed in ICH mice compared with saline controls (Figures 2 and 3C). A CD45hiGR-1+ cell population is mostly comprised of neutrophils. An increase in neutrophils is not surprising at these time points, as these cells are a part of the innate immune response and typically are among the first inflammatory cells to migrate to the site of injury (Fagan et al, 2004). These cells perform pattern recognition responses as well as promote inflammation (Fagan et al, 2004). Our observed CD45lo<5 GR-1− profile is characteristic of a microglia population (Mack et al, 2003; Figures 2 and 3B). Like neutrophils, microglia are part of the innate immune system. Similar numbers of microglia in ICH and saline mice suggest that this cell type does not proliferate specifically in response to ICH.
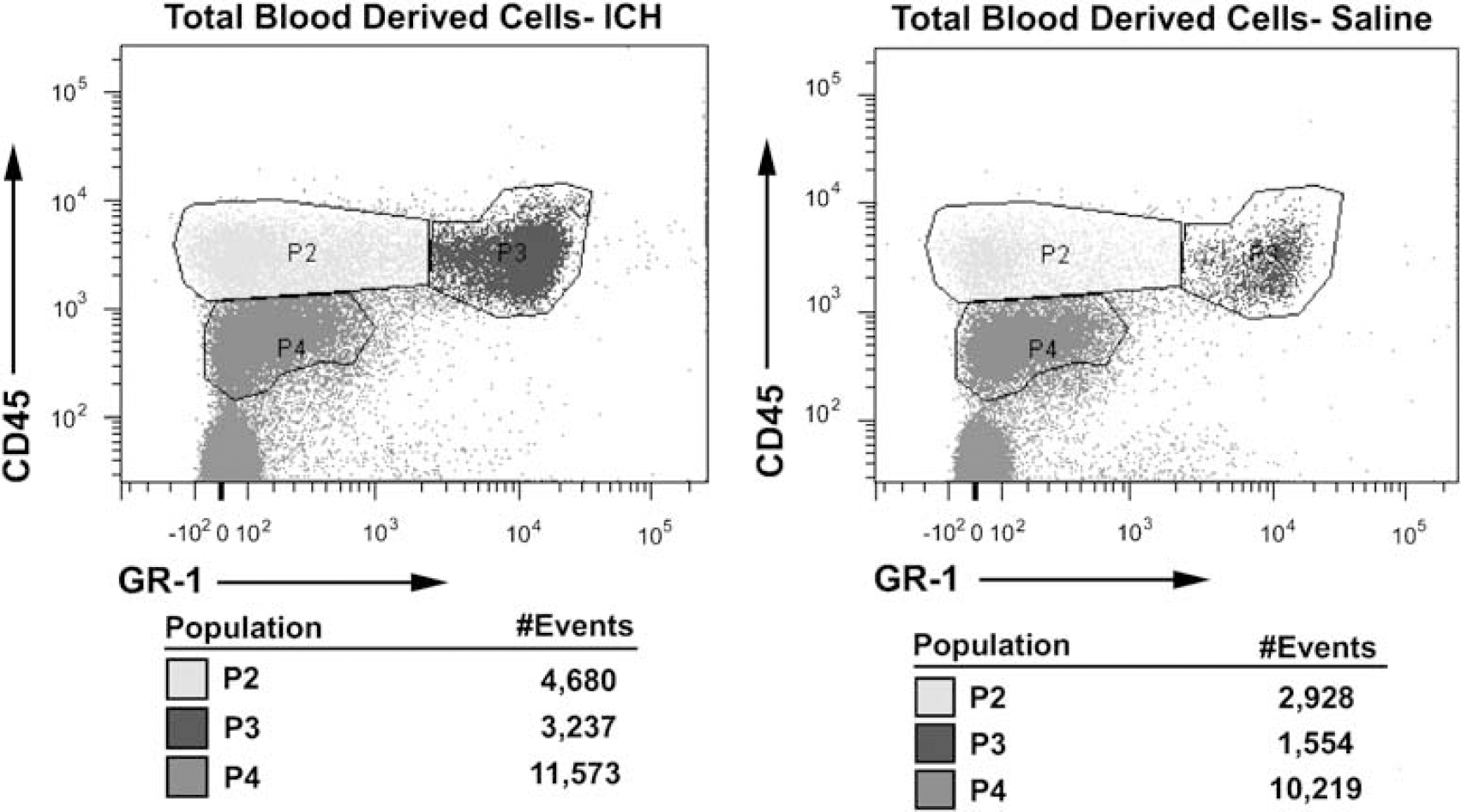
Representative scatter plots showing analysis of CD45hi cells at 4 days in a mouse receiving intracerebral hemorrhage (ICH) and a saline control. An overall increase in CD45hi inflammatory cell infiltrate was observed at 4 days post-ICH, compared with saline-treated mice (
Assessment of blood-derived inflammatory cells in the CNS 1 and 4 days after intracerebral hemorrhage (ICH). Brain infiltrating inflammatory cells were stained with anti-CD45, a pan marker for blood-derived cells, and anti-GR-1 antibodies. GR-1 is a pan surface marker for granulocytes. (
On analysis of T lymphocyte subsets, we did not observe a difference in CD8+ cells between ICH and saline mice at either time point (Figure 4C). Conversely, we observed a significant increase in CD4 + cells in ICH-treated mice at 4 days but not 1 day (Figure 4B). These CD4+ cells are likely to be CD4 + T cells as they stain positively for CD3 and express CD45hi that is characteristic of this lymphocyte subset. CD4 + T cells are part of the adaptive immune system and may take 5 days or more to become antigen specific in the CNS (Bailey et al, 2006). Increased numbers of CD4+ T cells in ICH is interesting, as traditional helper T cells do not normally recognize syngeneic protein as antigen.
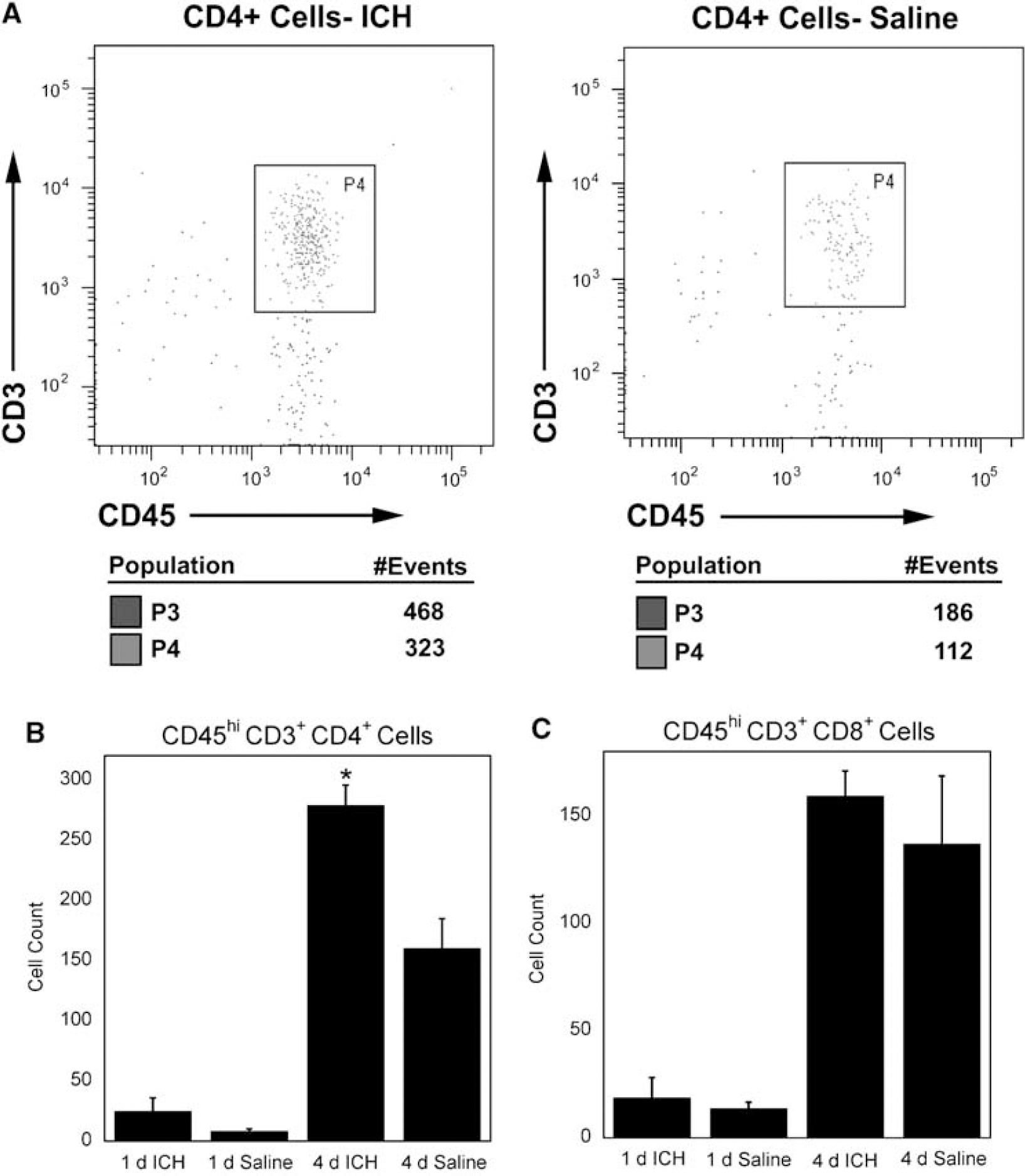
CD4+ and CD8+ cells. (
This suggests that the increased numbers of CD4 + cells in ICH could be the result of bystander activation or the recognition of endogenous antigens that have become newly exposed to the immune system as a result of ICH. Another possibility is that this CD4 + cell population contains regulatory T cells. CD4 + regulatory T cells have been shown to recognize both foreign and syngeneic antigens (Larosa and Orange, 2008; Pacholczyk et al, 2007; Fontenot and Rudensky, 2004). Therefore, it is currently unclear what functional role that the observed increase in CD4 + cells might play in the pathophysiology associated with ICH. However, there may be a chemospecific response occurring that is specific to the extravascular blood.
Using our model, at 4 days, we also observed a significant increase of CD45hi GR-1− cells (
In summary, we have found infiltration of neutrophils into ICH brains at 1 day, and infiltration of several leukocyte populations at 4-day post-ICH. The observation that CD4 + T cells migrate into the brain in this murine model is significant because it implies a role for the adaptive immune system in ICH. This study therefore shows the need for future research designed to adequately define the contribution of specific helper T-cell subsets in ICH.
Footnotes
Acknowledgements
This work was supported in part by a grant from the NIH: R01NS050569 (JFC).
The authors state no disclosures or conflict of interest.
