Abstract
17β-estradiol reduces cell death after global and focal ischemia and subarachnoid hemorrhage in rodents. Presently, we tested whether estrogen improves outcome after intracerebral hemorrhage (ICH) in male rats. Rats were implanted subcutaneously with 0.05, 0.25, or 0.50 mg pellets of estrogen (21-day release) or subjected to a sham procedure. Two weeks after implantation, they were given a striatal ICH via an infusion of collagenase. The three estrogen groups had significantly smaller lesions at a 7-day survival. Some rats had core temperature measured with an implanted telemetry probe, which also measured whole-body movements. Estrogen did not affect temperature nor activity levels after ICH. A second study with 0.25 mg pellets, administered once or twice, showed persistent histologic protection (30 days) and some functional benefit (e.g., elevated beam). A spectrophotometric hemoglobin assay showed that the 0.25 mg dose significantly reduced hemorrhagic blood volume at 12 hours after ICH. Regardless, estrogen did not lessen cerebral edema at 2 days after ICH and functional benefits were not consistently found on all tests (e.g., cylinder task). In summary, estrogen pretreatment reduces injury after ICH, in part by reducing bleeding. Estrogen may thus lessen injury and improve outcome after ICH in humans.
Introduction
Cerebral ischemia and related conditions (e.g., hypoxia, trauma) are leading causes of morbidity and neurological impairment. Indeed, stroke (focal ischemia), which predominantly affects the elderly, is the third leading cause of death and the leading cause of morbidity in North America. With many women receiving hormone replacement therapy (Lemaitre et al, 2002) and the possibility of treating others, including men, with estrogen, its effects on stroke occurrence (e.g., lower incidence of stroke in premenopausal women (Ayala et al, 2002)) and outcome must be clarified. Many experimental studies find that estrogen reduces injury after global (Jover et al, 2002; Shughrue and Merchenthaler, 2003; Sudo et al, 1997) and focal (McCullough et al, 2001; Simpkins et al, 1997) cerebral ischemia in rodents. In addition, estrogen reduces injury and mortality after experimental subarachnoid hemorrhage (SAH) in rats (Yang et al, 2001). This contrasts with clinical data showing that women have higher mortality and a less favorable recovery after severe SAH in comparison with men (Johnston et al, 1998; Kongable et al, 1996). Surprisingly, the effect of estrogen on intracerebral hemorrhage (ICH), which accounts for approximately 15% of all strokes (Mayo et al, 1996), is unknown.
An ICH causes cell death by direct tissue destruction, neurotoxicity, inflammation and swelling, impairments in blood flow, as well as by delayed neurodegenerative events (e.g., apoptosis) (Broderick, 1994; Del Bigio et al, 1996; Diringer, 1993). Estrogen is thought to reduce cell death after cerebral insults by a number of mechanisms (Hurn and Macrae, 2000; Stein and Hoffman, 2003) including, for example, improvements in cerebral blood flow and antiapoptotic effects. Thus, estrogen would likely benefit animals suffering an ICH.
Given the paucity of data on intracerebral hemorrhagic stroke and estrogen, we studied whether continuous 17β-estradiol treatment administered before a striatal ICH reduces injury and improves functional outcome in male rats. We used the bacterial collagenase model of ICH (Rosenberg et al, 1990). Estrogen was administered as a subcutaneous continuous release pellet in three doses—0.05, 0.25, and 0.50 mg (21-day release) starting 2 weeks before ICH with euthanasia at 7 days after ICH. In a second study, estrogen (0.25 mg) was administered either once or twice, and long-term (30 day) outcome was assessed. Motor function was assessed by several tests that assessed walking, spontaneous limb usage, and skilled reaching. Intraparenchymal bleeding was determined by a spectrophotometric hemoglobin assay at 12 hours after ICH (Choudhri et al, 1997). Cerebral edema was measured in some groups at 2 days after ICH. We hypothesized that estrogen pretreatment reduces injury and improves functional outcome in large part by reducing bleeding after the collagenase-induced ICH.
Methods
Animals
All procedures were in accordance with the Canadian Council on Animal Care guidelines and were approved by the Biosciences Animal Policy and Welfare Committee at the University of Alberta. A total of 144 young adult male (∼280 g on first surgery day) Sprague–Dawley rats, obtained locally, were used in this study. Of these, five were excluded because of technical problems (blocked cannula) and one was excluded because of marked hydrocephalus. All animals had food and water freely available and were individually housed after the start of the experiment. All surgical procedures were performed with aseptic techniques.
There are four separate experiments. In the first, rats were administered either of three doses of estrogen and assessed at a 7-day survival. In the second study, treated and untreated rats were allowed to survive for 30 days after ICH. A third experiment determined the volume of intraparenchymal bleeding at 12 hours after ICH. The fourth experiment measured cerebral edema at 2 days after ICH.
Estrogen Pellet Implantation
Animals were briefly anesthetized using isoflurane (4% induction; 1.5% to 2% maintenance in 70% N2O, 30% O2), and a small incision was made in the back of the neck. Animals randomly received 17β-estradiol pellets (Innovative Research of America Inc., Sarasota, FL, USA) implanted subcutaneously, or no pellet (sham surgery; ICH+0). The pellets contained 0.05 mg (ICH+0.05), 0.25 mg (ICH+0.25), or 0.50 mg (ICH+0.50) of 17β-estradiol that was supposedly time released over 21 days (experiment 1). In experiment 2, rats were either administered a second pellet at 1 week after ICH (ICH+0.25+0.25), one pellet plus a subsequent sham operation (ICH+0.25+0), or two sham pellet surgeries (ICH+0+0). We used 17β-estradiol pellets from Innovative Research of America because (1) these pellets provide a convenient delivery method minimizing stress (versus repeated injections), (2) they were supposed to provide a sustained release of estrogen according to the manufacturer, and (3) these pellets are commonly used in estrogen-stroke research (e.g., Alkayed et al, 2000; Jover et al, 2002).
The four groups of rats in experiment 1 are: ICH+0 (n=18), ICH+0.05 (n=18), ICH+0.25 (n=18), and ICH+0.50 (n=20). The three groups in experiment 2 are: ICH+0+0 (n=10), ICH+0.25+0 (n=10), and ICH+0.25+0.25 (n=10). Experiment 3 had two groups: ICH+0 (n=8) and ICH+0.25 rats (n=8). Experiment 4 had two groups: ICH+0 (n=9) and ICH+0.25 (n=9).
Core Temperature Telemetry Probe Implantation
Eleven days after the pellet implantation/sham operation a subset of rats in experiment 1 (ICH+0, n=3; ICH+0.05,. n=3; ICH+0.25, n=3; ICH+0.50, n=3) were implanted with core temperature telemetry probes (model TA10TA-F40, Transoma Medical, St Paul, MN, USA). Animal cages rested on receivers (RPC-1, Transoma Medical) that sampled core temperature twice per minute via the A.R.T. (v. 2.2) program (DeBow and Colbourne, 2003). Briefly, rats were anesthetized with isoflurane (∼20 mins) and a 2 cm incision was made in the abdomen into which the sterilized probe was implanted. The muscle was sutured closed and then infiltrated with Marcaine (Sanofi Canada, Markham, ON, Canada). The skin was then closed and treated with topical antibiotic. These probes remained in situ until euthanasia.
In addition to measuring temperature, these telemetry probes also provide a relative measure of activity, which is detected as movement (i.e., signal strength changes) of the probe over the receiver every 30 secs (Colbourne et al, 1998). The average activity count for the full day before ICH served as a baseline. The 24-hour periods after ICH were also averaged and analyzed.
Intracerebral Hemorrhagic Stroke Surgery
On day 14 after estrogen pellet/sham surgery, rats were anesthetized with isoflurane and placed in a stereotaxic frame. A small hole was made in the skull 3 mm right and 0.2 mm anterior to bregma. A 26-gauge needle (Hamilton syringe, Hamilton, Reno, NV, USA) was lowered to 5.5 mm below the surface of the skull and 1.0 μL of sterile saline containing 0.2 U of bacterial collagenase (Type IV-S, Sigma, Oakville, ON, Canada) was infused over 5 mins. The needle stayed in place for 5 mins after the infusion was complete. The burr hole was sealed with a small metal screw followed by infiltration of the scalp wound with Marcaine, and then stapled closed. This is the ICH model of Rosenberg and colleagues (Rosenberg et al, 1990) with minor modifications.
Behavioral Tests
Rats in experiment 1 were tested on the limb-use asymmetry test (Hua et al, 2002) and the horizontal ladder test (Metz and Whishaw, 2002) on day 7 after ICH. Rats in experiment 2 were tested on these tests on days 7 and 28 after ICH. In addition, they were tested on an elevated beam (Feeney et al, 1982) on these days. These rats were also tested on the staircase test from days 14 to 18, which measured independent forelimb reaching success for food pellets (Montoya et al, 1991).
The asymmetry test involved placing rats in a transparent cylinder (44 cm in height, and 20 cm in diameter) for 10 mins while videotaping them from underneath. From the videos, the number of independent forelimb contacts with the wall was determined. The percent of left forelimb use (contralateral to lesion) was expressed as: (contralateral touches/ipsilateral+contralateral touches+co-usage) × 100.
The horizontal ladder test allowed determination of limb placement during walking. The ladder was 1 m long and had variably spaced bars from one to 3 cm apart. Rats were videotaped crossing the ladder three times. We calculated the total number of falls (limb slips through bars) over the 3 trials as well as the total steps made in the middle 0.5 m segment of the test. From this we determined each limb's fall rate.
Rats traversed an elevated beam four times on two test days. The beam was 1 m long (2.5 cm wide) and had a dark goal box at the end. Rats were scored according to Feeney (Feeney et al, 1982) with the addition of a 0 score to represent a fall. Briefly, this scale ranged from 0 (fall) to 7 (success with 2 or fewer foot slips). The median score on each test day was analyzed.
The staircase test required rats to be pretrained (before pellet implantation). For this, rats were food deprived to 90% of baseline weight and then trained daily over 2 weeks (two 10-mins trials per day separated by 3 to 4 hours). After training, the rats were returned to free feeding. Rats were also under temporary food restriction during testing. The post-ICH results were expressed as a percentage of baseline, which was the average of the last 10 training trials.
Lesion Volume Analysis
Rats were euthanized 7 (experiment 1) or 30 days (experiment 2) after ICH. Rats were killed with an overdose of sodium pentobarbital (80 mg/kg intraperitoneal) and transcardially perfused with 0.9% saline followed by 10% neutral-buffered formalin. Forty micrometer coronal sections were taken every 400 μm starting from +1.7 mm to bregma and extending posterior −4.8 mm to bregma. Slides were stained with haematoxylin and eosin, and digitized at 1200 DPI with a flatbed scanner. These images were analyzed with Scion Image J 4.0 (Scion Corporation) by an observer who was blinded to group identity as previously performed (DeBow et al, 2003; MacLellan et al, 2004). In cases where the area of injury was not entirely clear, we examined the slides with a light microscope (e.g., for degenerating cells, eosinophillic cells, pallor, presences of inflammatory cells, etc.). We determined the volume of lost tissue, which was defined as the volume of the normal hemisphere (excluding ventricle)—volume of remaining ‘normal’ tissue on the damaged side (excluding lesion, lesion cavity and ventricle). The volume of each hemisphere was calculated as: average (area of the coronal section−area of ventricle−area of damage) × interval between sections × number of sections.
Intracerebral Blood Volume Analysis (Experiment 3)
The volume of extravasated blood was determined in ICH+0 and ICH+0.25 rats at 12 hours after ICH. This was determined with the spectrophotometric hemoglobin assay of Choudhri et al (1997) with minor modifications (MacLellan et al, 2004). Briefly, rats were overdosed with sodium pentobarbital 12 hours after ICH and transcardially perfused with 100 mL of 0.9% saline. The brain was extracted and the olfactory bulbs and cerebellum were discarded. The brain was then homogenized (Brinkman Instruments, ONT, Canada) in a test tube containing distilled water (total volume of 3 mL). After centrifuging (15,800 g for 30 mins) this solution, six aliquots of the supernatant (100 μL each) were each reacted with Drabkin's reagent (400 μL; Sigma) for 15 mins. The absorbance readings minus background reading at 540 nm were determined with a spectrophotometer (model DU-8, Beckman Coulter Ltd., London, UK). The data from each rat's six samples were averaged. Using a previously determined curve (MacLellan et al, 2004), from known blood volumes, we determined the amount of blood in each rat's brain.
Cerebral Edema (Experiment 4)
Cerebral edema was measured in ICH+0 and ICH+0.25. rats at 2 days after ICH. Briefly, rats were euthanized, decapitated, and the brain removed, divided into hemispheres, and sectioned into a 4 mm chunk encompassing the lesion, and weighed (MacLellan et al, 2004). These tissue samples were then reweighed after a 24-hour bake at 100°C (water content=wet weight−dry weight/wet weight × 100).
Plasma Estradiol and Testosterone Analyses
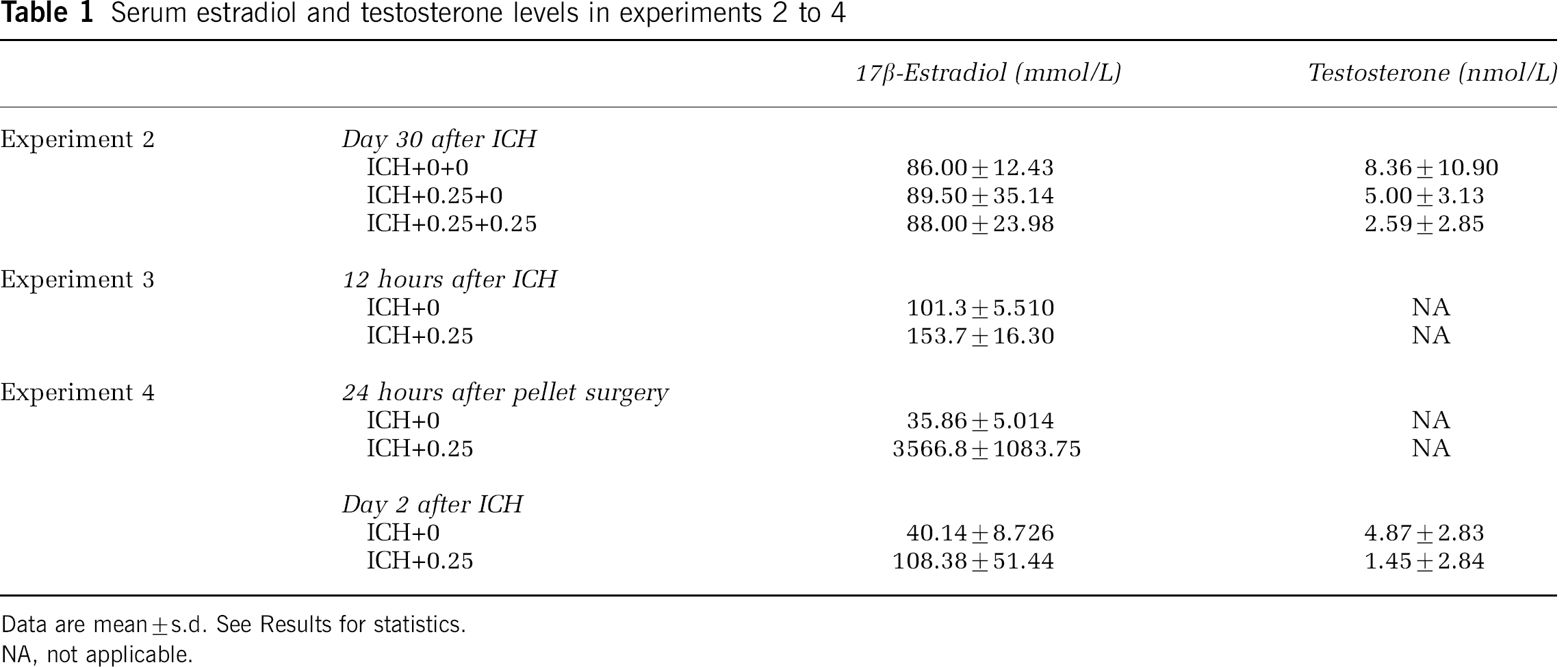
Cardiac blood samples were taken at euthanasia in experiments 2 (30 days after ICH), 3 (12 hours after ICH), and 4 (day 2 after ICH). In addition, samples were collected from the tail on the day after pellet implantation in the fourth experiment. Samples were placed in a refrigerator for approximately 2 hours for clotting to occur. The samples were then centrifuged (15,800 g for 10 mins) to obtain the serum. This was stored at −20°C, and later analyzed via an electrochemiluminescence immunoassay for the quantitative determination of estradiol levels and a radioimmunoassay for testosterone levels by the University of Alberta Hospital blood services staff. Testosterone was also assayed in experiments 2 and 4. Unfortunately, the day-after-pellet samples were not analyzed for testosterone levels in experiment 4 due to an experimenter error.
Data Analysis
All data presented are mean±s.d., except for the elevated beam scores, which are given as medians. Most data were analyzed by ANOVA followed by planned comparisons. However, in some cases nonparametric tests (Mann–Whitney test; Wilcoxon Signed Ranks test) were used because of heterogeneity (e.g., estrogen data in experiment 4) or because a rating scale was used (beam walking test).
Results
Experiment 1
The 0.05 mg (P=0.021), 0.25 mg (P<0.001), and 0.50 mg (P=0.001) pellets significantly (one way ANOVA with planned comparisons) reduced tissue loss at day 7 after ICH compared with untreated ICH (Figure 1A). The 0.25 mg dose reduced injury significantly more than the 0.05 mg pellet (P=0.015), but not significantly more than the 0.50 mg group (P=0.138).
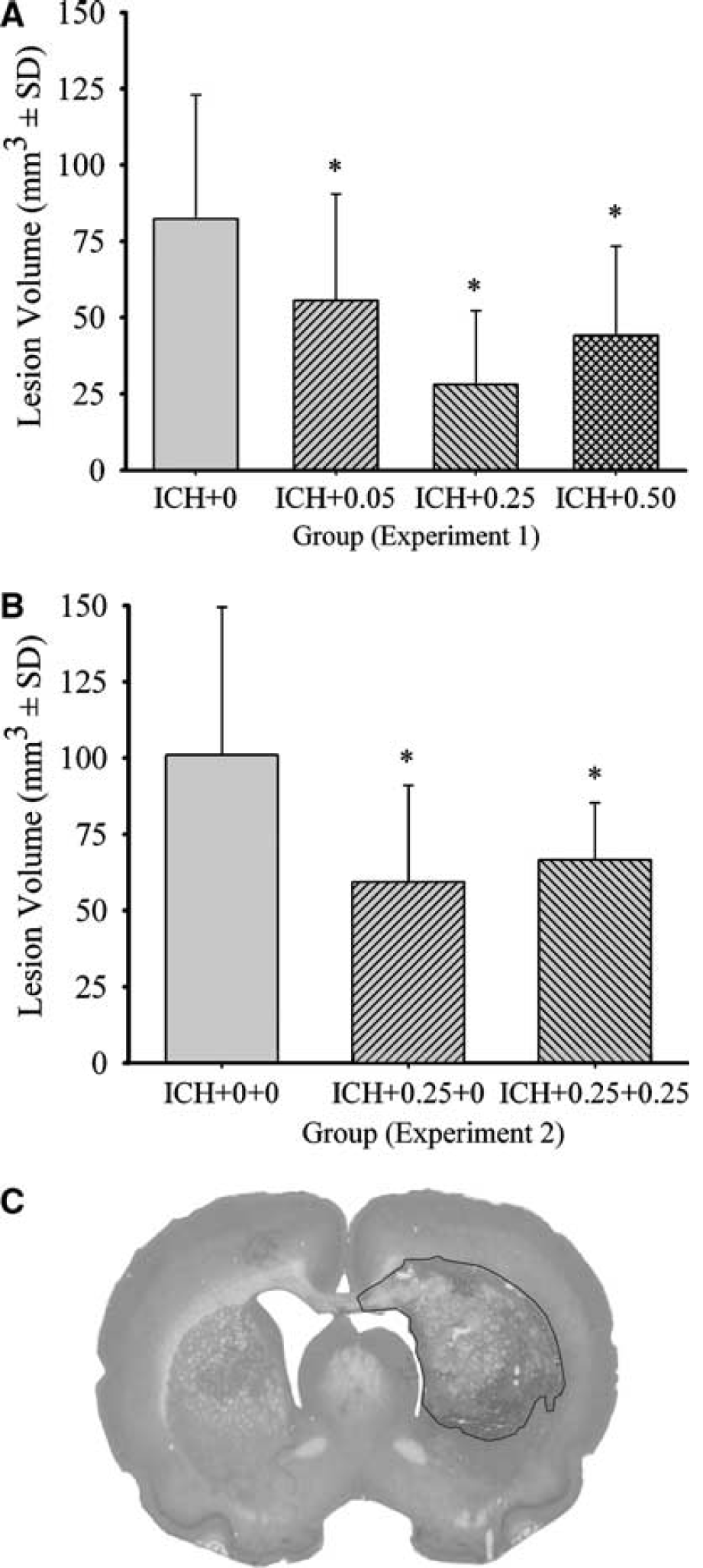
Average volume of tissue lost (mm3±s.d.) in experiments 1 (
Two rats died in the ICH+0 group, of which one died on day 1 and the other was euthanized on day 2. One rat in the ICH+0.50 group died of apparent isoflurane overdose. One rat in the ICH+0.05 group was euthanized on day 1. Reasons for euthanasia included: substantial loss of body weight, failure to eat and drink, exceptionally severe neurologic symptoms, or other signs of impending death (e.g., pulmonary edema). These rats were not behaviorally tested nor were they included in the lesion volume analysis.
A mixed ANOVA on error rate in the ladder task revealed a significant side effect (right versus left) because more falls occurred with the left, contralateral forelimbs (P<0.001) and hindlimbs (P=0.001). The contralateral forelimb data are shown in Figure 2A. The group main effects and interactions were nonsignificant (P≥0.082), indicating that estrogen did not affect these behavioral deficits. The touching asymmetry found in the cylinder task was not affected by estrogen treatment (P=0.606; Figure 3A).
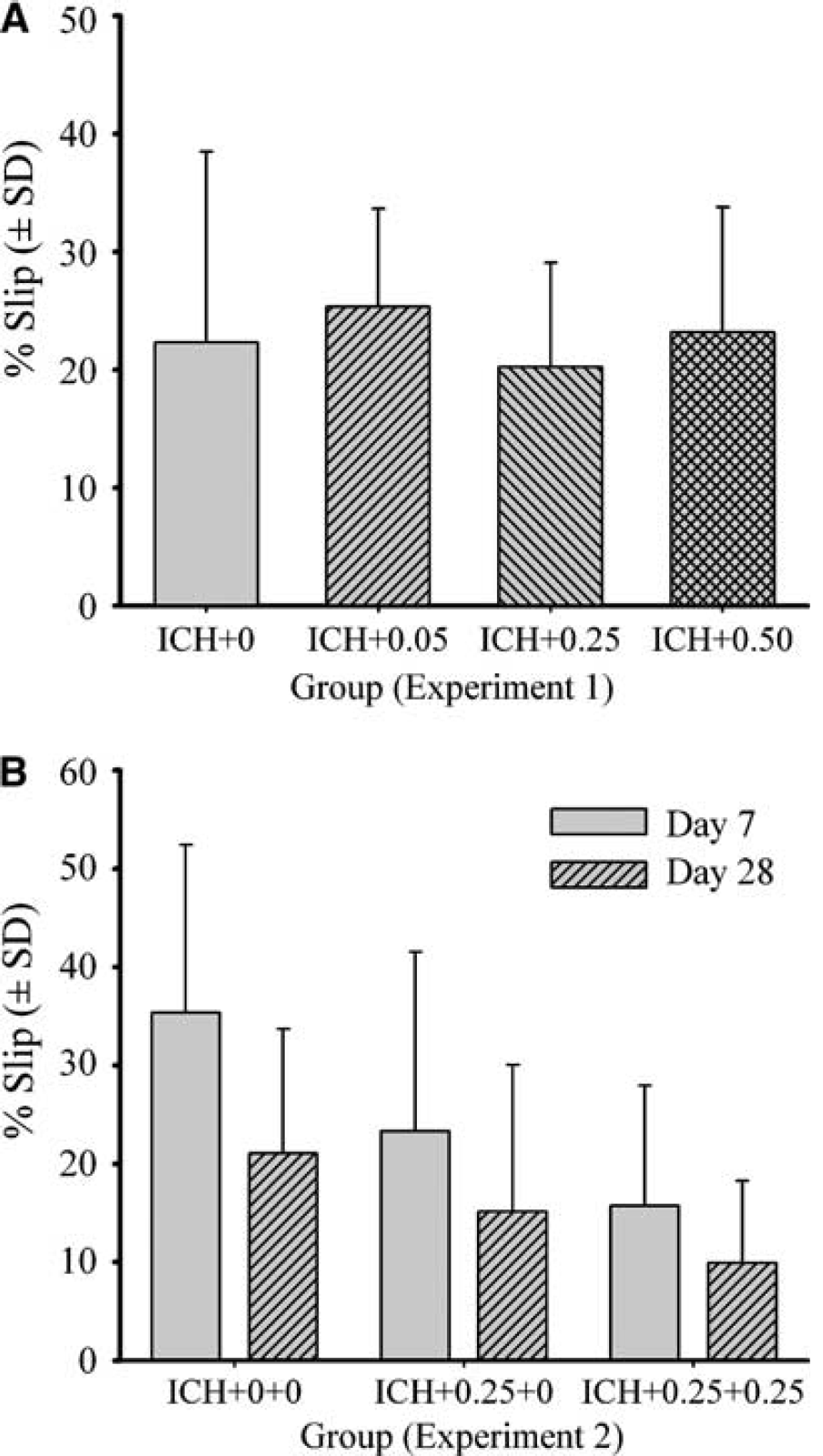
Error rate (mean±s.d.) in the horizontal ladder test on day 7 after ICH in experiment 1 (
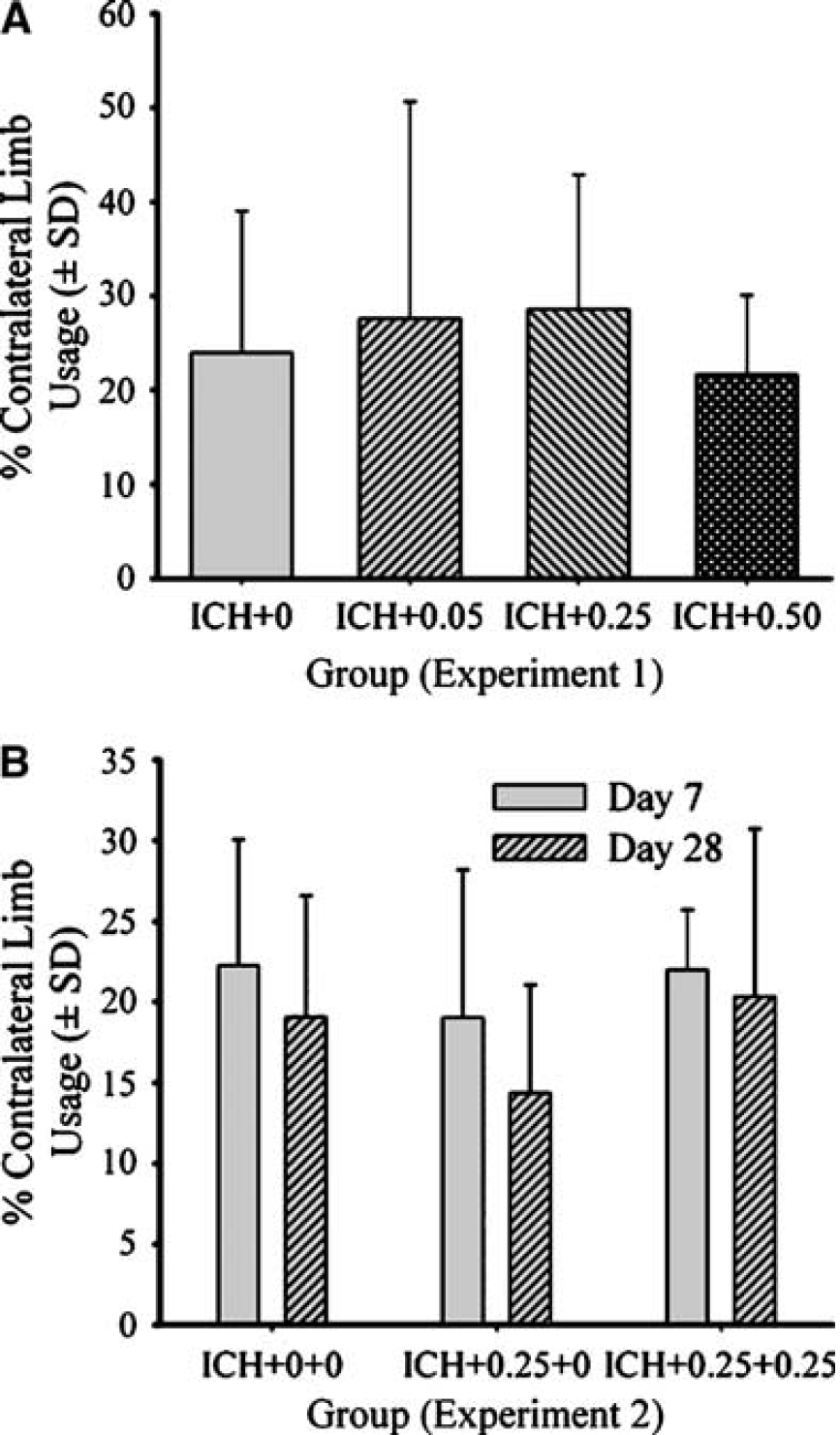
Percent contralateral forelimb usage (mean±s.d.) for wall contacts in the limb-use asymmetry test for experiments 1 (
There were no notable differences among groups over the 7 days of post- ICH temperature monitoring (Figure 4). Thus, the reduction in hemorrhagic lesion volume was not because of a change in core temperature after ICH. For the average activity counts (Figure 5), there was a significant day effect (P=0.003), but the group main effect and group × day interaction were nonsignificant (P≥0.949). These groups were transiently hypoactive for days after ICH, but there were no notable differences among the groups.
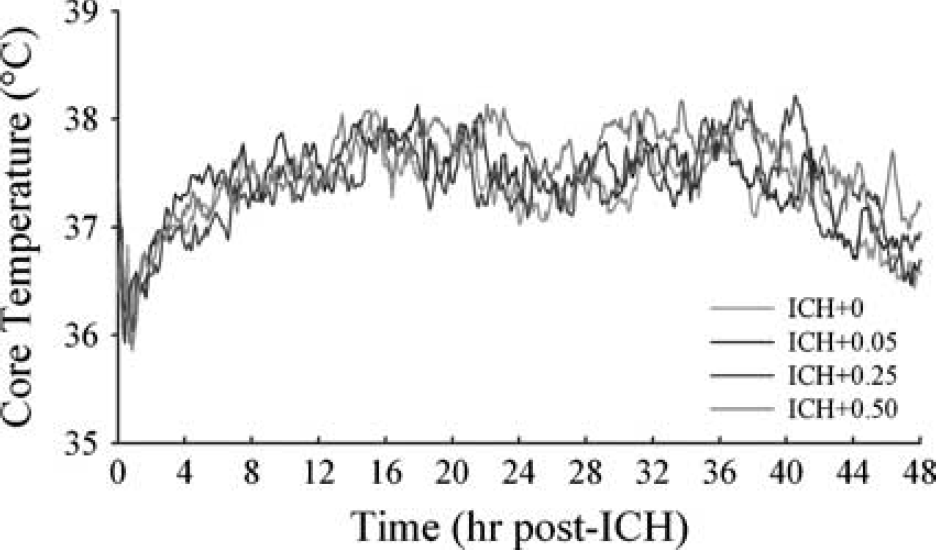
Core temperature (°C) after ICH. Temperature was measured for 7 days after ICH, but only 2 days are shown. Groups were similar throughout the 7-day monitoring period. Temperature was sampled once every 30 secs and presented as 5-mins averages.
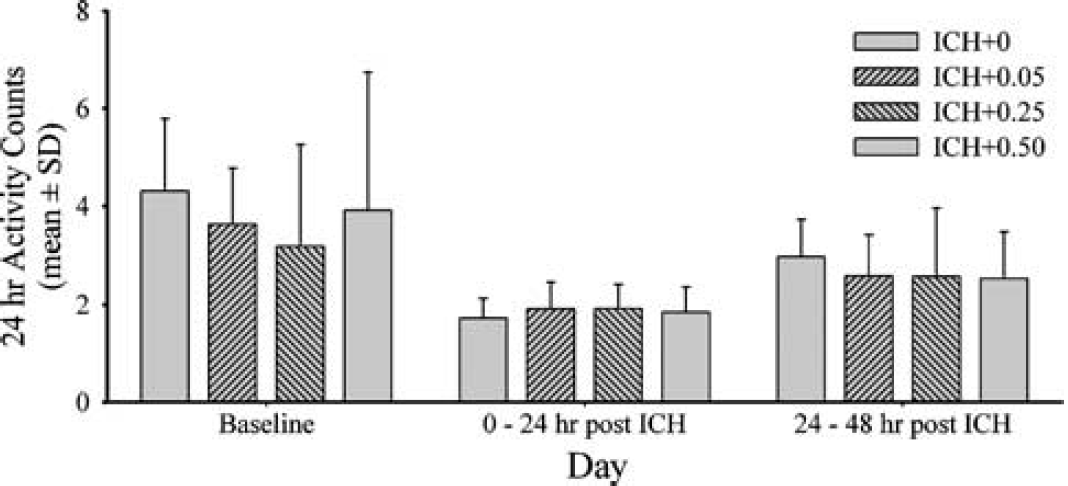
Activity counts (summated every 30 secs and averaged over 24-hour periods; mean±s.d.) as measured with core temperature telemetry probes (via movement of probe over receiver). The ICH caused hypo-activity (versus baseline) that eventually returned to normal. There were no group differences. Two post- ICH days of data shown as group trends were similar afterwards.
Experiment 2
Both the ICH+0.25+0. (P=0.014) and the ICH+0.25+0.25 (P=0.038) groups had significantly less injury than the ICH+0+0 group at a 30-day survival (Figure 1B). The estrogen groups were not significantly different (P=0.648).
A between–within ANOVA on the % error rate in the horizontal ladder test (Figure 2B) revealed that there were significant group (P=0.016) and day main effects (P=0.007). The group × day interaction was not significant (P=0.555). The ICH+0.25+0.25 group made significantly less errors (P=0.005) whereas the difference between the ICH+0+0 and ICH+0.25+0 groups did not reach statistical significance (P=0.083). A between–within ANOVA on the % contralateral forelimb touches in the cylinder test (Figure 3B) revealed that there were no significant group (P=0.275), day (P=0.051), or group × day main effects (P=0.723). Group differences were found on the elevated beam task (Figure 6) on day 7 (P=0.041), but not day 28 (P=0.126). On day 7, both estrogen-treated groups, which themselves were not different (P=0.533), were significantly better at traversing the beam compared with the untreated ICH group (P≤0.037). Reaching performance in the staircase test (Figure 7) with the ipsilateral and contralateral forelimbs were analyzed with mixed ANOVAs. In both cases there was a significant day main effect (P≤0.013). The group effects were not significant (P≥0.082) nor was the interaction (P≥0.323). Two rats (one ICH+0.25+0 and one ICH+0.25+0.25) were excluded from this analysis for failing to reach a criterion of 7 pellets per side over the last three training days.
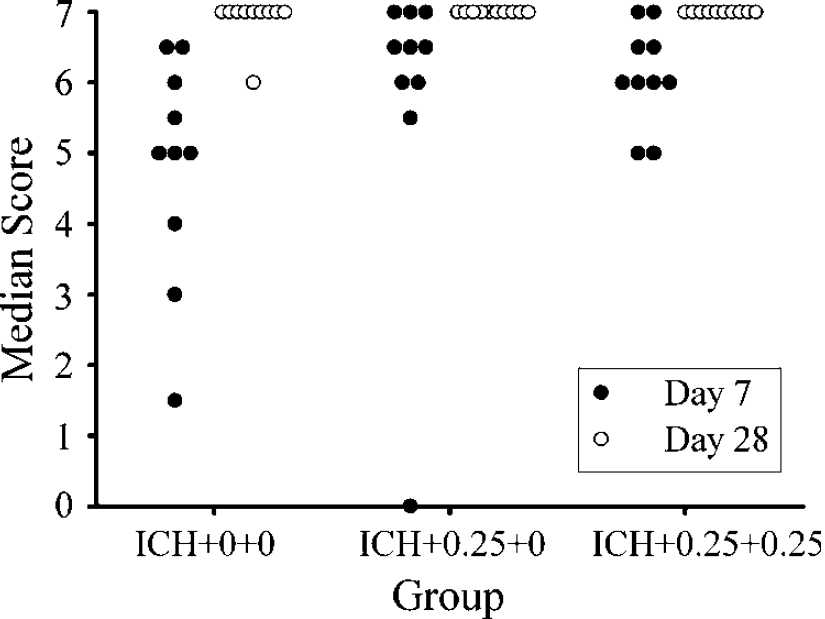
Median scores in the elevated beam test at 7 and 28 days after ICH (experiment 2). Both estrogen groups had significantly better scores at 7 days, but not at 28 days. Almost all rats obtained a perfect score of 7 on day 28 (i.e., complete recovery).
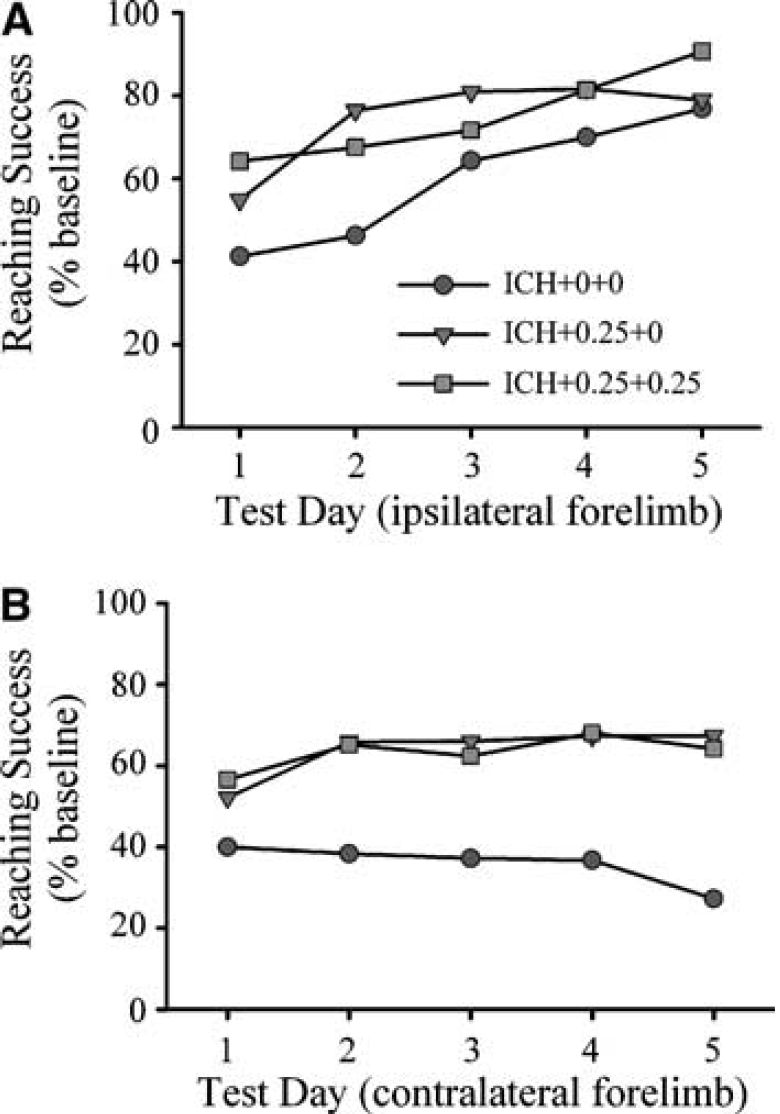
Reaching performance with the ipsilateral (
Serum estradiol and testosterone levels (Table 1) were not significantly different among groups (P≥0.179) at the time of euthanasia, which is after the pellets were expected to be depleted.
Serum estradiol and testosterone levels in experiments 2 to 4
Data are mean±s.d. See Results for statistics.
NA, not applicable.
Experiment 3
The 0.25 mg pellet significantly (Figure 6; P=0.050) reduced the volume of blood in the brain by 48% at 12 hours after ICH versus untreated ICH as determined by the spectrophotometry hemoglobin assay. This was not assessed with the other pellet doses. Serum estradiol was also determined at 12 hours after ICH in these rats (Table 1). The ICH+0.25 group had significantly higher estradiol levels than the ICH+0 group (P=0.015) (Figure 8).
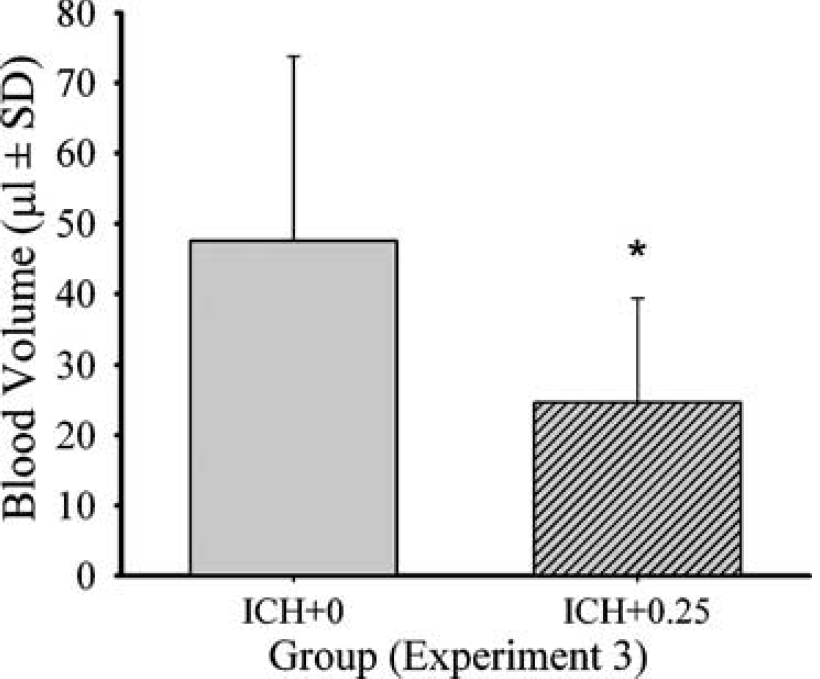
Bleeding volume (mean±s.d.) at 12 hours after ICH as determined by a spectrophotometric assay. The ICH+0.25 group had a significantly smaller hematoma than the ICH+0 group. An ∗ denotes significant difference from the ICH+0 group.
Experiment 4
One rat in the ICH+0 group died during the ICH surgery. Both the ICH+0 and the ICH+0.25 groups had significantly more water content in the ipsilateral than in the contralateral hemisphere (P≤0.001). The groups were not significantly different (P≥0.393) (Figure 9).
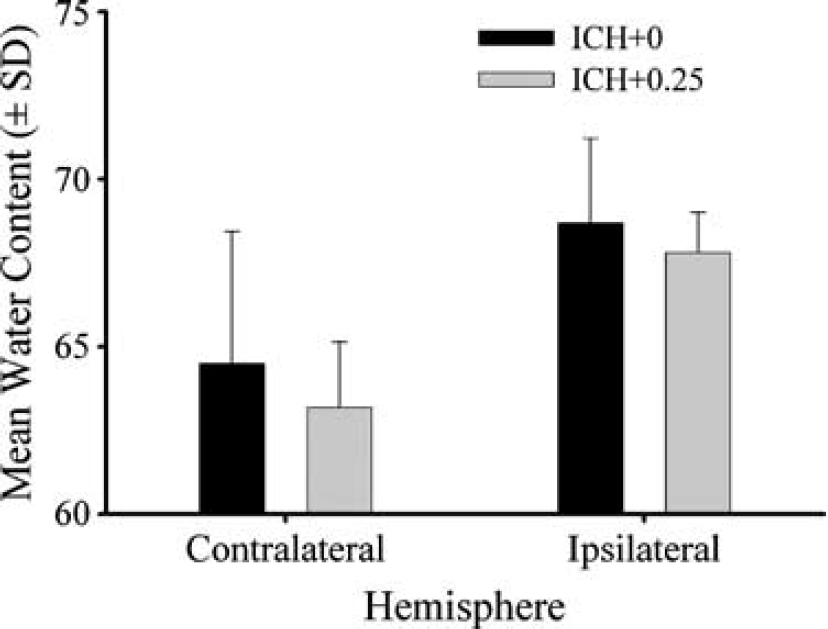
Mean (±s.d.) cerebral water content in the ipsilateral (right; ICH lesioned) and contralateral hemispheres at 2 days after ICH in untreated and estrogen-treated groups.
Estradiol and testosterone data are given in Table 1. Estrogen data were analyzed by a nonparametric test because of extreme heterogeneity. Estrogen levels were significantly (P≤0.002) higher on the day after pellet implantation and at euthanasia (day 2 after ICH) in the estrogen-treated group. Unlike the untreated rats (P=0.204), the estrogen levels were substantially higher on the day after pellet implantation in the ICH+0.25 rats (P=0.012). Testosterone levels at the time of euthanasia were significantly lower in the ICH+0.25 group (versus ICH+0; P=0.038). Testosterone and estrogen levels were negatively related (r=−0.531, P=0.042).
Discussion
This is the first study showing that estrogen administered pre- and post-insult reduces brain injury after ICH in rats. The optimal dose of estrogen appears to be the 0.25 mg pellet, which released estrogen over 21 days (2 weeks pre- to 1 week after ICH). Other doses also reduced injury and it should be noted the 0.50 mg pellet was not significantly different from the 0.25 pellet group. Importantly, the 0.25 mg dose resulted in sustained histologic protection (30 days after ICH). However, a second treatment with estrogen starting at 7 days after ICH did not further reduce injury. This cytoprotective effect of estrogen was, at least partly, because of a substantial reduction in bleeding after collagenase infusion. The temperature profiles after untreated ICH and ICH treated with estrogen were similar. Thus, hypothermia, which reduces cell death after global and focal ischemia (Colbourne et al, 1997; Dietrich et al, 1996) as well as ICH (MacLellan et al, 2004), was not a factor in this study. In addition, the activity profiles after ICH were similar among groups, indicating that exercise, which can lessen injury after ICH (DeBow et al, 2003; Lee et al, 2003), did not influence the present findings. Finally, estrogen did not have a marked effect on cerebral edema. Surprisingly, estrogen's marked cytoprotective effect did not consistently translate into improved functional outcome, although some benefits were observed.
Estrogen significantly reduced the bleeding into the brain at 12 hours after collagenase infusion. It is likely that estrogen caused bleeding to cease sooner, thus minimizing the insult. Notably, numerous studies show that 17β-estradiol promotes coagulation (Garcia-Manzano et al, 2002; Jaimez et al, 2001; Rosendaal et al, 2002; Scarabin et al, 1997). However, other possibilities exist. For instance, estrogen may directly affect collagenase activity. Estrogen may also reduce endothelial cell injury. Regardless, the most parsimonious explanation for reduced bleeding is the hypercoagulability state that estrogen is well known to produce.
Given the multiple protective effects of estrogen discussed earlier, we suspect that reduced bleeding, while very important, is only partly responsible for the observed cytoprotection. Notably, the ICH+0.25 group had a 48% reduction in bleeding at 12 hours whereas there was a 66% reduction in tissue lost at day 7. However, our study design did not allow us to directly ascertain how much the effect of reduced bleeding accounts for the lessened structural damage. The estrogen-mediated reduction in bleeding not only reduces the primary structural damage (e.g., dissection of blood through tissue), but should also mitigate the secondary effects of ICH, such as inflammation, edema and apoptosis, owing to reducing the primary insult. Thus, it is surprising that estrogen did not reduce edema at 2 days after ICH. However, estrogen may still promote a faster resolution to cerebral edema and a time course study is required to test this hypothesis.
It is not uncommon to find a mismatch between histologic and functional outcome measures (Corbett and Nurse, 1998). Indeed, several therapies improve functional outcome after ICH but fail to decrease the lesion size (Del Bigio et al, 1999; Peeling et al, 2001). Nonetheless, it is surprising that the impressive and persistent reduction in the loss of tissue after ICH with estrogen did not substantially and consistently improve functional recovery. Perhaps the tissue salvaged did not function normally or the residual damage was sufficient to substantially impair performance on some of the behavioral tests. It is also possible that the salvaged tissue did not normally support performance on these tests (e.g., spontaneous limb usage in the cylinder). This is unlikely, however, given the statistically significant correlations between striatal lesion volume and performance on these tests (DeBow et al, 2003; Hua et al, 2002; MacLellan et al, 2004). The failure to improve functional outcome in the first study was hypothesized to be indicative of a transient histologic effect. In other words, we expected the salvaged tissue to function abnormally and thus eventually die. Furthermore, numerous treatments only transiently protect after global (Colbourne et al, 1999; Dietrich et al, 1993) and focal ischemia (Valtysson et al, 1994) and studies using estrogen to treat global and focal ischemia have not assessed long-term outcome (e.g., 1-month survival). Our results of experiment 2, however, do not support this hypothesis because a lasting reduction in injury was found and some functional benefit was also observed.
There are several additional limitations with our study that should be considered. First, the effects of endogenous estrogen in female rats should be examined on ICH outcome. Similarly, ICH outcome should be assessed in aged, reproductively senescent females and in those administered estrogen replacement therapy. Second, we found that estrogen reduced levels of testosterone (experiment 4), which may affect outcome. However, a study using the middle cerebral artery occlusion model suggests that testosterone does not mediate the protective effects of estrogen, at least in that model (Toung et al, 1998). Nonetheless, further study (e.g., castrating males) in ICH models is warranted. Third, while we have identified several protective pre-ICH doses of estrogen, it remains to be determined whether estrogen would protect when administered after the onset of ICH. Fourth, other dosing regimens and delivery methods (e.g., Silastic capsule delivery, injections) would likely improve outcome and should be studied. The latter is especially important because the estrogen pellets we used were found to induce an extremely high level of estrogen on the day after the pellet implantation. While the levels stabilize, it is possible that the supraphysiologic levels of estrogen affects outcome. Fifth, while we showed that a second dose of estrogen (at 1 week after ICH) did not improve outcome beyond that provided by the first pellet, the optimal duration of treatment remains to be determined. Finally, additional behavioral tests or earlier assessment may have revealed additional functional benefits of estrogen treatment.
In summary, estrogen administration led to significantly reduced bleeding and lesion volume after collagenase-induced ICH in rats. Further study is required to determine if other mechanisms contribute to estrogen's beneficial effects on an ICH. Despite several limitations, our data are encouraging in that they show that estrogen limits brain injury because of a severe ICH and our study has identified the most likely mechanism, which is a reduction in bleeding. Estrogen may thus serve as a broad-spectrum treatment for stroke as it appears to reduce injury after ICH, SAH, as well as global and focal cerebral ischemia. Additionally, estrogen may reduce bleeding and injury in ischemic stroke patients who undergo spontaneous or t-PA-induced hemorrhagic transformation.
Footnotes
Acknowledgements
The authors thank D Clark, L Davies, M Iseke, D Lee, C MacLellan, and T Rajapakse for technical assistance.
