Abstract
The vascular basement membrane contributes to the integrity of the blood-brain barrier (BBB), which is formed by brain capillary endothelial cells (BCECs). The BCECs receive support from pericytes embedded in the vascular basement membrane and from astrocyte endfeet. The vascular basement membrane forms a three-dimensional protein network predominantly composed of laminin, collagen IV, nidogen, and heparan sulfate proteoglycans that mutually support interactions between BCECs, pericytes, and astrocytes. Major changes in the molecular composition of the vascular basement membrane are observed in acute and chronic neuropathological settings. In the present review, we cover the significance of the vascular basement membrane in the healthy and pathological brain. In stroke, loss of BBB integrity is accompanied by upregulation of proteolytic enzymes and degradation of vascular basement membrane proteins. There is yet no causal relationship between expression or activity of matrix proteases and the degradation of vascular matrix proteins in vivo. In Alzheimer’s disease, changes in the vascular basement membrane include accumulation of Aβ, composite changes, and thickening. The physical properties of the vascular basement membrane carry the potential of obstructing drug delivery to the brain, e.g. thickening of the basement membrane can affect drug delivery to the brain, especially the delivery of nanoparticles.
Keywords
Introduction
Basement membranes are layers of complex extracellular matrix (ECM) proteins that provide support for epithelial and endothelial cells, hence separating these from the underlying tissue. 1 In the central nervous system (CNS), the vascular basement membrane separates the endothelial cells from neurons and glial cells and contributes to vessel development and formation and maintenance of the blood-brain barrier (BBB).2,3 The BBB is formed by thin non-fenestrated brain capillary endothelial cells (BCECs) sealed together by tight junctions that protect the CNS by limiting the passage of, e.g., naturally occurring molecules, microbiological pathogens, and the majority of administered pharmaceuticals circulating in the blood.3–5 The restrictive function of the BCECs is sustained by pericytes embedded in the vascular basement membrane and astrocyte endfeet.3,6,7 Together BCECs, pericytes, and astrocytes synthesize and deposit proteins extracellularly thereby forming the vascular basement membrane.8–11
Overview of the composition of the extracellular matrix in the brain.

Structurally, the vascular basement membrane consists of two principally different entities, i.e. the endothelial and parenchymal basement membranes, both of main importance for maintaining the integration of BCECs with pericytes and astrocytes.10,14,22 These three cell types adhere to the basement membrane by specific members of the integrin or dystroglycan receptor families, thereby maintaining the cells in position and adding to the mechanic-physical properties of the BBB.12,23 Dystroglycan consists of a highly glycosylated extracellular α-subunit and a transmembrane β-subunit. The α-dystroglycan form is responsible for the linkage to the basement membrane proteins whereas β-dystroglycan links α-dystroglycan to the actin cytoskeleton. 24 In the adult mouse brain, the expression of dystroglycan is found in endothelial cells and perivascular astrocytes and their endfeet. 25 Integrins are heterodimer transmembrane glycoproteins consisting of α- and β-chains, which bind to many proteins of the vascular basement membrane and activate several signalling pathways. 26 Different integrin isoforms are involved in the binding of the BCECs, pericytes, and astrocytes to the vascular basement membrane. Particularly different β1-integrins are reported to be expressed by BCECs (α1β1, α3β, α6β1, and αvβ1),23,27–29 pericytes (α4β1), 30 and astrocytes (α1β1, α5β1, α6β1). 28 The interaction of endothelial β1-integrins with collagen IV of the basement membrane is correlated to expression of the tight junction protein claudin-5 and to the BBB integrity in vitro. 31 Thus the interaction through the integrin receptors both provide physical support and regulate signalling pathways, whereby the BCECs can adapt to changes in the microenvironment. 23
Soluble factors found in the vascular basement membrane in the brain.

Major changes in the molecular composition of the vascular basement membrane can be observed in acute and chronic neuropathological settings,35–38 which probably add significantly to disease pathogenesis. In the present review, we cover the molecular composition of the vascular basement membrane, how this basement membrane is affected in stroke and Alzheimer’s disease (AD), and how physical properties of the basement membrane ultimately can act as a second barrier beyond the BBB, thus potentially obstructing drug delivery to the brain.
Molecular composition of the vascular basement membrane
The assembly of the vascular basement membrane is dependent on an initial self-assembly of laminin into a sheet followed by binding to nidogen and HSPGs. To complete the formation of the vascular basement membrane, nidogen and HSPGs are linked to collagen IV, hence allowing the latter to form a secondary stabilizing polymer network (Figure 1).39,40 Laminins are cross-shaped and the most abundant non-collagenous basement membrane protein. The different laminins all consist of an α-chain, a β-chain, and a γ-chain. Five α, four β, and three γ chains exist and can combine to form 16 different laminin isoforms.34,41 The biological role of the laminins is largely defined through their α-chain interaction with integrins expressed by BCECs, pericytes, and astrocytes.34,42 The laminins of the vascular basement membrane consist of either α1, α2, α4, or α5 in combination with β1 and γ1 chains resulting in a composition of the laminin isoforms 111, 211, 411, and 511.
14
Additionally, laminin 421 is also present along the cerebral vasculature.8,18,43 The laminin self-assembly results in the formation of polymers. This polymerization is a two-way process with an initial temerature-dependent step followed by a calcium-dependent step. Calcium is thus crucial for the polymerization of laminin.
44
Illustration showing the architecture of the vascular basement membrane. The basement membrane is composed of different extracellular matrix (ECM) proteins, with the majorities being collagen IV, various laminin isoforms, perlecan, and nidogen-1. Laminin and collagen IV can self-assemble into 3D networks that are interconnected by nidogen and perlecan. Adopted from Hallmann et al.
14
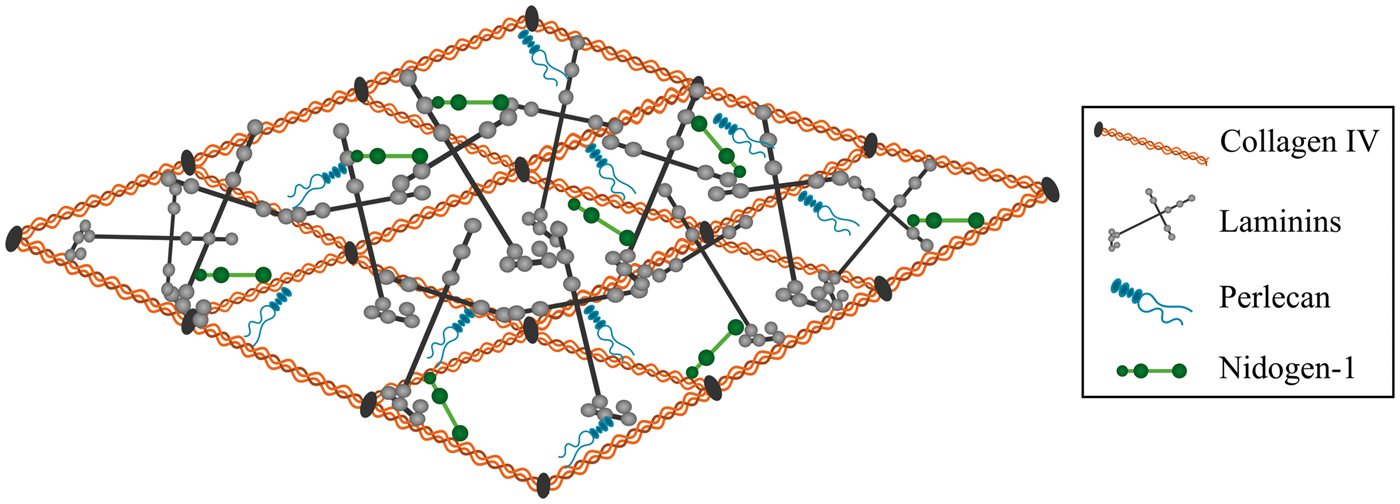
Collagen IV consists of polypeptide chains derived from six different α chains, which can combine and form three different collagen IV isoforms. The collagen IV isoform predominantly expressed in the brain consists of two α1 and one α2 chain that fuse to form the collagen IV [α1(IV)]2α2(IV) isoforms.1,44–46 Inside cells, the three α-chains assemble forming triple helical molecules, termed protomers, which oligomerize outside the cells into a supramolecular network. 47 The self-assembly of the collagen IV protomers is associated with both lateral (side-by-side) as well as C- and N-terminal non-covalent bindings of the collagen IV monomers. 48 The assembly of the collagen IV network outside the cells is dependent on chloride, which facilitates the modulation of salt bridge interaction within the trimeric non-collagenous 1 domain of the C-terminal. 47
The laminin and collagen IV networks are held together by nidogen. 44 Two isoforms of nidogen, nidogen-1 and -2 (entactin-1 and -2), have been identified and consist of three globes (G1, G2, G3) connected by segments of variable lengths. G1 and G2 are located within the N-terminus and G3 at the C-terminus.49,50 Nidogen-1 is important for connecting and stabilizing the self-assembled layers of laminin and collagen IV.49,51 In addition to laminin and collagen IV, nidogen-1 also binds fibulin and perlecan.52,53 Nidogen-2 is also able to bind basement membrane components. However, the mRNA expression of nidogen-2 is significantly downregulated already at early postnatal age, implying its significance during embryogenesis. 50
Three different HSPGs, perlecan, agrin, and collagen XVII, are found within the vascular basement membrane with agrin and perlecan being the most abundant. 54 Perlecan has a multi-domain protein core with three glycosaminoglycan (GAG) chains at its N-terminus. It is integrated into the collagen IV/laminin network and plays an important role in the maintenance of basement membrane integrity and binding of growth factors.55,56 There are several spliced isoforms of agrin (i.e. z+ and z0), each with different functions. The isotype of agrin present in the vascular basement membrane is referred to as z0, as this form is devoid of an amino acid insertion at the COOH terminal.57,58 The HSPGs are able to tether and accumulate growth factors to protect them from degradation and modulate paracrine signalling of surrounding cells.59,60 Some of the heparin-binding and perlecan core protein binding factors found within the vascular basement membrane are listed in Table 2.
Cellular contribution to the basement membrane along the vascular tree
The distinct types of vessels denoting the cerebral vascular tree, i.e. major arteries penetrating the pial surface, arterioles, capillaries, postcapillary venules, venules, and veins, mutually vary in terms of vascular wall thickness and composition of basement membrane proteins. Especially, the distribution of various laminin isoforms along the brain vasculature has received much attention.10,14,34
Pial arteries consist of a monolayer of endothelial cells and its basement membrane surrounded by smooth muscle cells (tunica media), connective tissue (tunica adventitia), and a coverage of pial cells. The pial arteries penetrate the brain parenchyma and subsequently branch into smaller arterioles surrounded by the pial cells with their underlying basement membrane, the astroglial basement membrane, and astrocyte endfeet. Together the pial and astroglial basement membrane is referred to as the parenchymal basement membrane (Figure 2).61,62 Thus, in the large parenchymal arteries and arterioles, the vascular basement membrane consists of two principally different entities, i.e. the endothelial and parenchymal basement membranes distinguished by their distribution of laminin isoforms. The endothelial basement membrane contains the laminin isoforms 411 and 511 derived from endothelial cells, whereas the parenchymal basement membrane of the penetrating arteries and arterioles contains laminin isoforms 111 and 211 derived from pial cells and astrocytes, respectively (Figure 2).10,34 Laminin 411 is unequivocally expressed in the endothelial basement membrane independent of vessel type. Oppositely, the expression of laminin 511 is correlated to both vessel maturation and type.8,34
The differential expression of laminins along the cerebrovascular tree. The illustration shows the lining of the parenchymal (light blue) and endothelial basement membrane (dark blue) along the cerebrovascular tree with a cross section of the pial vessel, penetrating arteriole, capillary, and postcapillary venule. Laminin α1 and α2 are expressed by pial cells of the pia mater (yellow line) and astrocytes, respectively, and these can be found in the parenchymal basement membrane of the penetrating arterioles. As the contribution from the pial cells diminishes, the parenchymal basement membrane of the capillaries and postcapillary venules no longer has laminin α1. Laminin α4 is uniformly expressed in the endothelial basement membrane along the cerebrovascular tree. Oppositely, laminin α5 is expressed in the endothelial basement membrane of the pial vessel, penetrating arterioles, and capillaries but has patchy distribution along the postcapillary venules. In capillaries, there is no clear separation of the endothelial and parenchymal basement membranes, and the basement membrane thus appears as a single entity that envelops the pericyte. In postcapillary venules, the parenchymal and endothelial basement membranes line the border of the virtual perivascular space (PVS). E: Endothelial cells, P: pericytes, A: Astrocyte endfeet.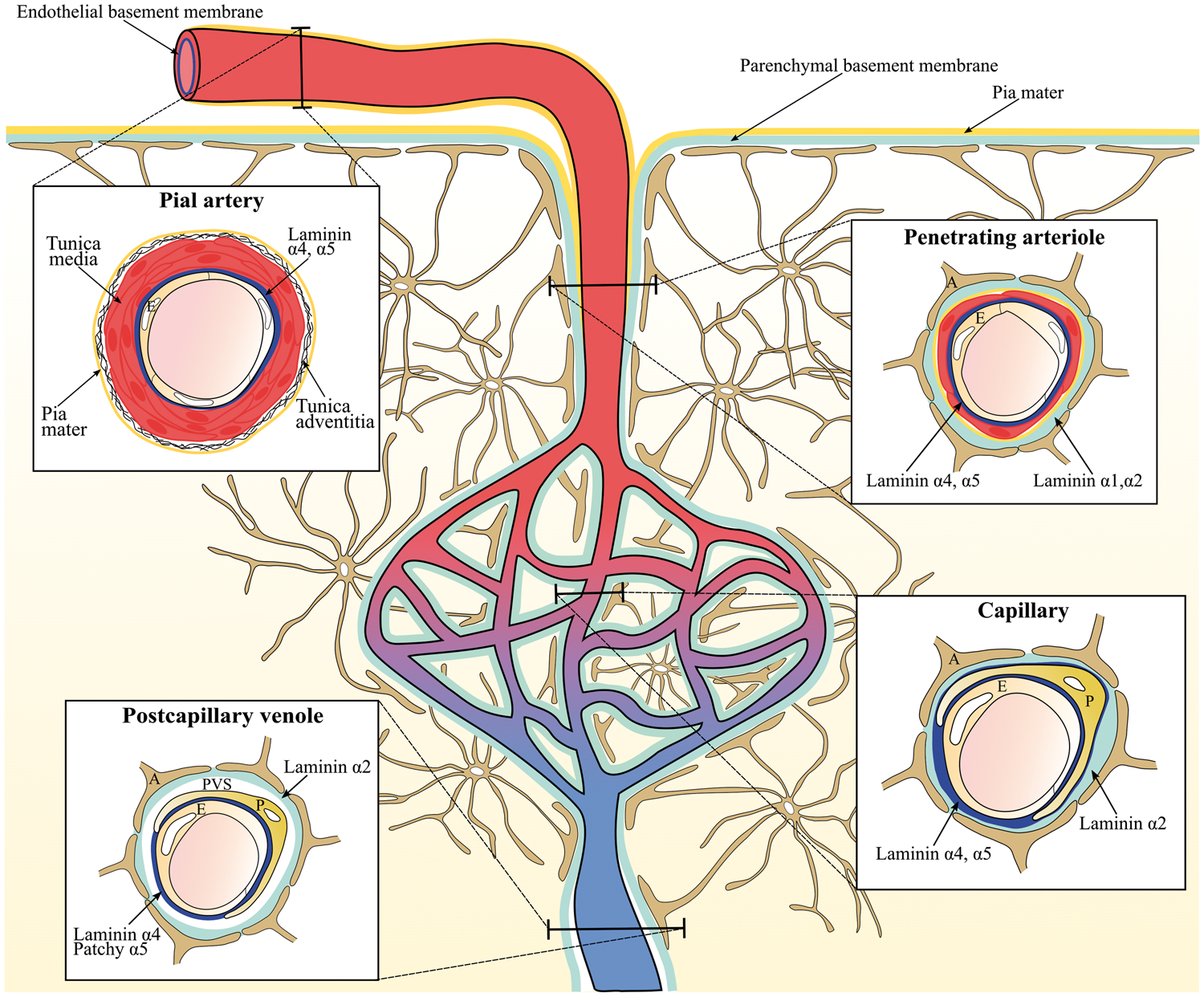
As the arterioles diminish, smooth muscle cells are substituted with pericytes, which are embedded in the endothelial basement membrane ensheathing the endothelial cells. Also, the pial sheet disappears as the smooth muscle cells are lost along the arteriole branches. 62 Thus, in the capillaries, the parenchymal and endothelial basement membrane appear as a single entity, which contains laminin 411 and 511 derived from endothelial cells and laminin 211 from astrocytes. The capillaries do not contain laminin 111 since they no longer receive a contribution from the pial cells (Figure 2).10,14,62 In postcapillary venules, the endothelial and parenchymal basement membranes are separated by a virtual perivascular space, which is especially apparent in cerebral inflammation where leukocytes accumulate in a perivascular cuff before infiltrating the brain parenchyma.33,61 Oppositely to arteries, arterioles, and capillaries, the endothelial basement membrane of the postcapillary venules exerts a patchy distribution of laminin 511, which has been correlated with sites where lymphocytes fail to extravasate. 10
The contribution of the pericytes to the vascular basement membrane protein composition is considered significant due to the unique location of pericytes embedded in the endothelial basement membrane. 63 For instance, recent data indicate that pericytes secrete both laminin 411 and 511 (Figure 2).9,22 Their secretion of basement membrane proteins is particularly prominent during development and angiogenesis. 64 Additionally, the close relation between the BCECs and pericytes is considered important for induction of basement membrane protein synthesis by BCECs. 63 The gene expression of collagen α1, laminin α4, laminin α5, agrin, and perlecan by murine BCECs is nonetheless unaffected when co-cultured with pericytes, indicating that interactions between BCECs and pericytes are not mandatory for expression of these basement membrane proteins. 8
The function of the vascular basement membrane proteins
Looking at the function of the different basement membrane proteins, it should be emphasized that many of these proteins are also expressed in tissues and cells outside the CNS. Many genetic knockout models of specific basement membrane proteins exert loss of function with structural deformities in tissues outside the CNS that preclude their development further than the embryonic stage.65,66
The significance of different laminin isoforms in the brain was pursued by the creation of different murine laminin isoform knockouts. Knockout of the laminin α2 chain (LAMA2−/−) causes the autosomal recessive muscle disorder, laminin 2 deficient congenital muscular dystrophy. These mice show retarded growth and die at the age of 4–5 weeks. 65 The LAMA2−/ − mice also have a defect in the BBB as verified by the presence of inflammatory cells in the brain parenchyma, changed organization of tight junction proteins, reduced pericyte coverage, and extravasation of albumin. Together, these results suggest that α2-containing laminins from astrocytes are of great importance for both development and function of the BBB. 67 The significance of astrocytic laminin was further investigated in γ1 null mice where the γ1 chain of laminin was specifically depleted in astrocytes resulting in faulty assembly of laminin 211. The γ1 laminin chain knockout mice exhibit weakened vascular integrity causing haemorrhages in small arterioles of the basal ganglia, thalamus, and hypothalamus. The weakened vasculature correlates well to a disrupted interaction between astrocytes and vascular smooth muscle cells. 68 Depletion of astrocytic laminin is also associated with increased BBB permeability, confirmed by extravasation of albumin, IgG, and 500 kD FITC-Dextran, and decreased levels of the tight junction proteins, occludin and claudin-5 in brain capillaries. The astrocytic laminin also affects pericyte differentiation by maintaining the pericytes in a non-contractile state. 11 The non-contractile form of pericytes is the primary cell type, which stabilizes BBB integrity. 69 The ablation of astrocytic laminin and resulting changes in pericyte differentiation could be the reason for the weakened vascular integrity observed in the astrocytic laminin depleted mice. 11 Laminin 411 and 511 produced by the BCECs also proved important for the vascular integrity and function of the BBB. In the experimental autoimmune encephalomyelitis (EAE) mouse model of multiple sclerosis, extravasation of inflammatory cells across the BBB selectively occurred at sites with sparse expression of laminin 511 in the endothelial basement membrane. 10 Supporting this observation, deletion of endothelial laminin α4 also reduced migration of T cells across the BBB in the EAE model.70,71
Studies of collagen IV [α1(IV)]2α2(IV) knockout mice reveal that collagen IV is unnecessary for deposition of other basement membrane components during early embryonic development. However, the deletion causes embryonic lethality due to an impaired stability of the basement membrane at later embryonic age. 66 Instead of creating a complete knockout of collagen IV, exon 39 of the collagen IV α1 chain was spliced directly to exon 41 creating the mouse model referred to as Col4a1Δex 40 . These mice develop haemorrhages in deep regions of the brain resembling the pathology of small vessel disease, which indicates that collagen IV is of great importance for maintaining proper function of the brain’s microvasculature.46,72
Knockout of nidogen-1 in mice causes neurological defects presented by episodes of involuntary movements, which resemble seizures and when lifted at its tail the knockout mice exhibit a twisted posture. Immunolabeling of other basement membrane proteins reveals that the vascular basement membrane is sufficiently formed in the absence of nidogen-1. However, brain capillaries had a thinned vascular basement membrane compared to the controls. 51
Knockout of either perlecan or agrin is embryonic lethal. 54 Perlecan deletion does not affect the formation of the vascular basement membrane; instead, the stability is affected causing, e.g., invasion of neuronal cells into the ectoderm during embryogenesis. 73 Mice that completely lack the z+ splice form of agrin, also have a severe reduction of all other agrin isoforms including z0. The mice have slightly smaller brains compared to their age-matched control littermates and die at birth due to neuromuscular defects. 74 Furthermore, the accumulation of agrin in the vascular basement membrane occurs at the developmental time when blood vessels become impermeable indicating that agrin supports BCECs during the formation of the BBB.57,75 This has been further supported by a murine study showing that the localization of the adherens junction proteins VE-cadherin, β-catenin, and ZO-1 in the BCECs is stabilized by the presence of agrin. 58 This stabilizing effect of agrin is suggested to occur by binding to endothelial αvβ1 integrin since agrin previously has been shown to interact with αvβ1 integrin.58,76
Together, the basement membrane proteins play a significant role in the maintenance of the tightness of the BBB. This has been confirmed by various in vitro studies in which either immortalized or primary cell cultures were grown in transwell inserts mimicking the BBB. 77 Growing primary porcine BCECs on filters coated with laminin, collagen IV, and fibronectin or a mixture of one-to-one of these proteins significantly increased the trans-endothelial electrical resistance (TEER) of BCEC monolayers. The greatest increase in TEER was observed when BCECs were grown on collagen IV, collagen IV/fibronectin, and fibronectin/laminin coated inserts. 16 It should be noted that the ECM proteins employed in this study are neither specific for the CNS nor the endothelial basement membrane. Thus the increase in TEER does not necessarily correlate to in vivo conditions, where, e.g., fibronectin is not an integral component of the normal endothelial basement membrane. However, the importance of the basement membrane for maintaining barrier properties gains support by a recent study showing that porcine BCECs grown on pericyte and/or astrocyte generated basement membranes increase both the expression of the tight junction proteins occludin and claudin-5, and the electric resistance across porcine BCECs. 78
Changes of the vascular basement membrane in stroke
Overview of the changes in basement membrane protein composition seen in stroke.
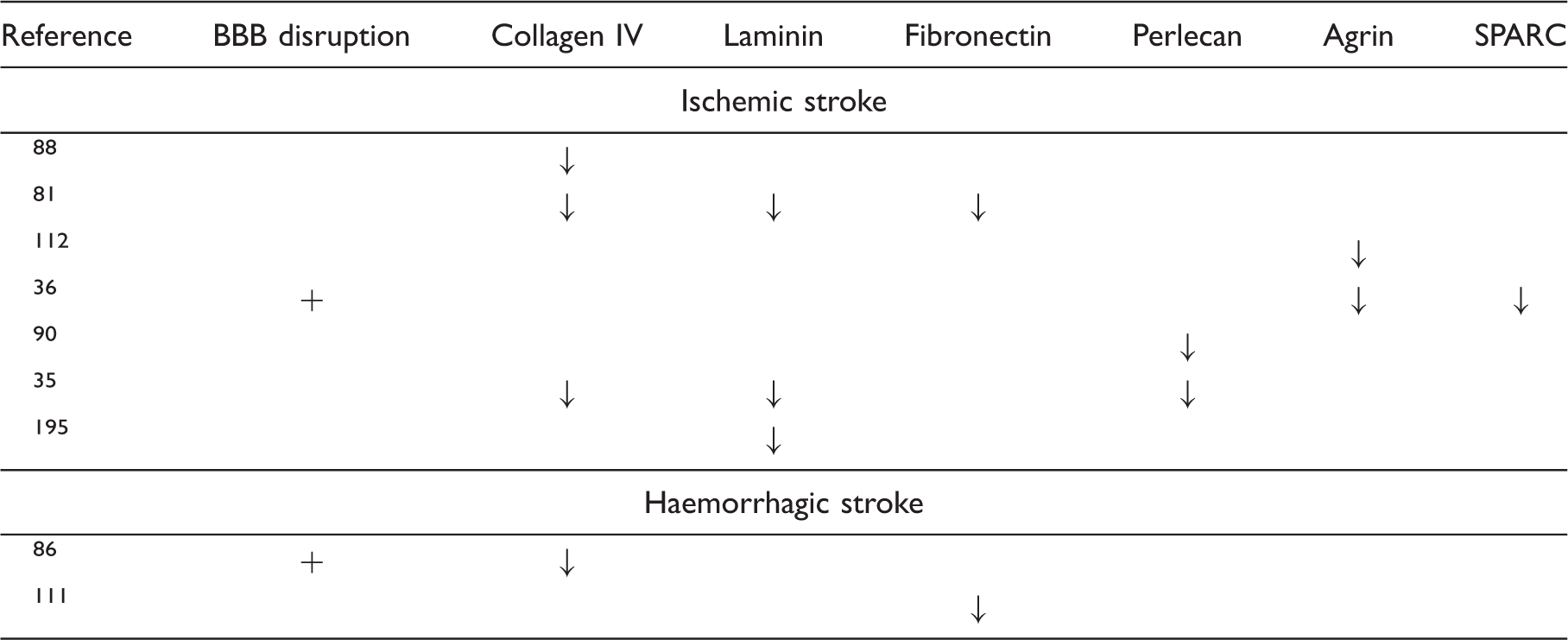
As previously mentioned, the Col4a1Δex 40 mutation predisposes mice to haemorrhagic stroke, thus underlining the important role of collagen IV for vascular integrity.46,72 Likewise, mutations or degradation of other basement membrane proteins are suggested to predispose to stroke. 72 The HSPG perlecan is lost in the ischemic core within hours following stroke in non-human primates. 35 In the ischemic core, the cysteine proteases cathepsin B and L are both upregulated and hypothesized to be responsible for the degradation of perlecan since perlecan is the most sensitive basement membrane protein to proteolysis by these enzymes in vitro.35,89,90 The degradation of perlecan causes the generation of the protein fragments C-terminal domain V (DV) and LG3 of perlecan. Especially the production of perlecan DV is increased after ischemic stroke, and the generation of this fragment is proposed to play a protective role, since deficiency in perlecan leads to worsened stroke outcomes in a rodent model, while administration of perlecan DV improves the outcome.55,89 Degradation fragments of other basement membrane protein such as collagen IV may also influence the outcome of stroke, as collagen IV fragments have anti-angiogenic potential. 91 Even though these fragments have been studied in relation to tumor angiogenesis92,93 they might also be involved in stroke pathogenesis.
Following stroke, the plasminogen activator system and matrix metalloproteinases (MMPs) are also significantly upregulated. The plasminogen activator system is an enzymatic cascade, which is normally involved in fibrinolysis and thus clot degradation, matrix turnover, synaptic regulation, and plasticity in the developing and adult brain.94,95MMPs, a family of zinc endopeptidases, are produced in a latent form and contain a pro-peptide responsible for maintaining the latent form of MMP and a hemopexin-like domain, which can be recognized by tissue inhibitor of metalloproteinases (TIMPs). 96 The activity of these protease systems is highly regulated and depends on the developmental stage or the type of injury. A rapid increase in synthesis of urokinase plasminogen activator (uPA) and plasminogen activator inhibitor-1 (PAI-1) is observed following ischemic stroke.97,98 uPA is involved in the activation of plasminogen to plasmin, which contributes to fibrin degradation. In vitro studies reveal that plasmin is able to degrade both laminin and fibronectin and activate pro-MMPs. Consequently, the increased levels of plasmin seen in stroke may contribute to the degradation of the vascular basement membrane proteins.97–101 However, since PAI-1 is also upregulated,97,98 this might potentiate the effect of uPA and thus the involvement of plasmin in basement membrane degradation in vivo is not fully elaborated.
Several studies have shown an upregulation of pro-MMP-2 and -9 levels in the brain after stroke.97,98,102 Nonetheless, discrepancies in the time course of upregulation of the MMPs exist among species and models.98,102–104 Different enzymes or cytokines can activate MMPs through cleavage of the pro-domain or by a cysteine switch mechanism. Enzymes that are directly or indirectly involved in the activation of pro-MMP-2 include membrane-type 1-MMP (MT1-MMP), MT3-MMP, uPA, and plasmin, which are all upregulated following stroke.98,105 Additionally, the increased production of inflammatory cytokines, reactive nitrogen, and oxygen species produced after stroke, can trigger the secretion of MMP-9 and activation of pro-MMP-9.80,106–108 Both MMP-2 and -9 have a fibronectin binding domain allowing them to bind the basement membrane proteins and specifically digest fibronectin, collagen IV, and laminin in vitro.35,80,109 However, the active sites of MMP-2 and -9 do not accommodate the structure of native collagen IV or laminin, thus these basement membrane proteins have to be denatured to fit the active sites of the MMPs. 110 Yet, an increased activation of MMP-2 and -9 could lead to digestion of the basement membrane proteins.35,87,88,109,111 Supporting this observation, scavenging the oxidative responses and thus inhibition of MMP-9 attenuates the loss of fibronectin 24 h after haemoglobin injection into the rat caudate nucleus. 111 Furthermore, a significant reduction in the levels of agrin and SPARC is observed after global cerebral ischemia. MMP-2 and -9 are suggested to play a key role in this degradation.36,112 Although it might seem that MMPs are solely harmful, they probably have a dual role after haemorrhagic stroke. Thus, their expression is increased 1–3 and 7–14 days after stroke. Inhibition of MMPs during the acute phase improves the integrity of the BBB, whereas inhibition in the long-term probably interferes with the subsequent angiogenesis and remodeling. This indicates that the MMPs are involved in a pathological process in the acute phase on days 1–3 and conversely participate in a recovery process in the long-term phase around days 7–14. 113 Even though the different protease systems are involved in changes of the BBB integrity following stroke, there is no direct evidence that the active forms of these proteases contribute to cerebral vascular basement membrane degradation in vivo.
Changes of the vascular basement membrane in Alzheimer’s disease
Oppositely to the changes of the basement membrane seen in ischemic and haemorrhagic stroke, the changes in AD include deposition of Aβ, basement membrane thickening, and changes in the basement membrane protein composition.
AD is a neurodegenerative disorder characterized by continuous shrinkage of brain tissue and a gradual decline in memory and other cognitive functions. 114 The pathological hallmarks of AD are extracellular amyloid plaques primarily composed of amyloid beta (Aβ) peptide and intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein. 114 Emerging evidence also indicates that vascular dysfunction is a third solid hallmark in AD.115–117
For years, the main hypothesis about the neuropathology of AD has been the ‘amyloid cascade hypothesis’, which states that the primary event in AD pathogenesis relates to the accumulation of Aβ. This notion is based on the fact that ∼1% of AD cases can be explained by genetic mutations in amyloid precursor protein (APP) or particular enzymes, secretases, which are involved in the sequential proteolysis of APP generating Aβ.
118
However, the amyloid cascade hypothesis is challenged because a subset of the elderly has Aβ plaques without developing clinical symptoms of AD, suggesting that other factors could be involved in AD pathogenesis.
119
The cause of the remaining ∼99% AD cases of apparently non-genetic, sporadic nature remains unknown. Yet, different risk factors have been identified with aging being the most evident, and an increasing amount of evidence also suggests that neurovascular dysfunction contributes to the neurodegeneration observed in AD prior to Aβ deposition.
120
Increased risk of AD is also related to diabetes, obesity, hypercholesterolemia, hypertension, atherosclerosis, and stroke, all factors that also contribute to pathological changes of the vasculature.
121
For instance, changes in the composition of the vascular basement membrane are observed during normal aging and in conditions like diabetes and atherosclerosis.122–127 Thus the changes of the vascular basement membrane observed in AD may be initiated by another underlying vascular condition. Three distinct changes of the vascular basement membrane can be seen in AD, i.e. (i) deposition of Aβ, (ii) basement membrane thickening, and (iii) changes in the basement membrane protein composition (Figure 3).
Schematic illustration of the vascular basement membrane at the blood-brain barrier (BBB) in health, stroke, and Alzheimer’s disease (AD). Normally the vascular basement membrane is well organized. In stroke, the basement membrane (continued) Figure 3. continued. proteins are degraded by mechanisms that may involve different protease-systems, e.g. matrix metalloproteinases (MMPs) such as MMP-2 and -9, urokinases of the plasminogen activator system, and the cysteine proteases cathepsin B and L. Stroke is also accompanied by decreased BBB integrity with leakage of serum proteins, increased production of inflammatory cytokines and reactive oxygen species, and invasion of inflammatory cells. Oppositely, in AD, accumulation of amyloid beta (Aβ) as cerebral amyloid angiopathy (CAA) in the basement membrane of both larger vessels and capillaries is observed. Furthermore, basement membrane thickening and changes in the protein composition of the basement membrane with increased deposition of, e.g., perlecan can also be seen in AD. BM: Basement membrane.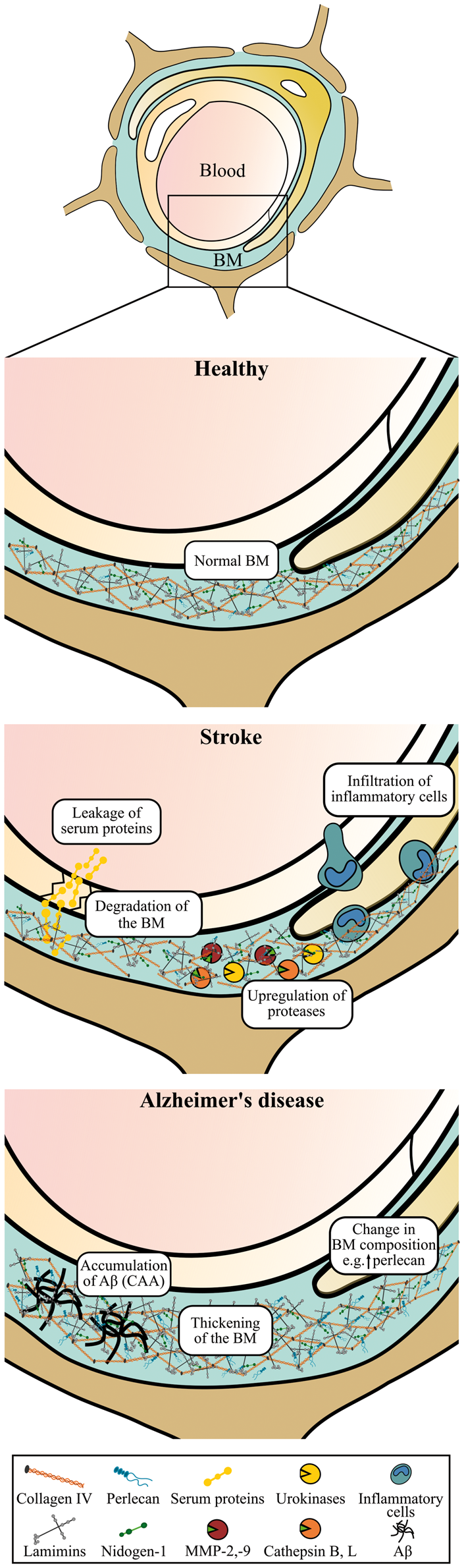
i) The resulting pathology occurring from the deposition of Aβ in the vasculature is referred to as cerebral amyloid angiopathy (CAA). Aβ can accumulate within the walls of pial and parenchymal arteries, arterioles, capillaries, and veins, and it is identifiable in approximately 80% of AD cases. A recent study, analyzing postmortem brain samples from 134 AD patients, found that the distribution of senile plaques and CAA could be categorized into four types. Type 1 cases had predominantly senile plaques with or without CAA in the pial vessels; type 2 cases had senile plaques and CAA in both the pial vessel walls and penetrating arteries; type 3 cases had senile plaques and CAA in predominantly capillaries and arteries; and type 4 cases had a more vascular phenotype, in which Aβ deposition was more dominant in CAA than in the senile plaques. Hence, the presentation of CAA in AD is quite heterogeneous. 128
CAA in the pial and parenchymal vessel can also be found in non-demented individuals, and the identification of CAA thus does not positively correlate with AD or with the severity of AD. A study analyzing autopsies from 100 demented and non-demented individuals identified a small but significant correlation between the presence of CAA in capillaries and AD (Figure 3), suggesting that there might be different mechanisms involved in the development of CAA in capillaries and in the pial and larger parenchymal vessels. 129 Genetics denotes an import factor for this difference since CAA in capillaries is associated with a higher frequency of individuals with the APO ɛ4 allele, and thus impact the heterogeneous presentation of CAA.128,130,131
In capillary CAA, Aβ is deposited as small bumps lining the basement membrane of the capillary wall (Figure 3), whereas in larger vessels the Aβ deposition is prominent in the tunica media in closer proximity to the smooth muscle cells. 130 The distribution of Aβ in the vessel walls correlates with the perivascular drainage of solutes from the brain. Fluorescent tracers injected into the brain parenchyma initially spread along the extracellular spaces and along the endothelial basement membrane of the capillaries, and then along the basement membrane of the smooth muscle cells of the tunica media. 132 Aβ1-40 isoform is the main form that accumulates in vessels, which contrasts the isoform found within amyloid plaques of the brain parenchyma denoted by the Aβ1-42 isoform. 133 The cause of the Aβ deposition in the vessel wall remains unknown but it is linked to both a decreased clearance of Aβ across the BBB and thickening of the basement membrane.133,134
Overview of the changes in basement membrane protein composition seen in Alzheimer’s disease.

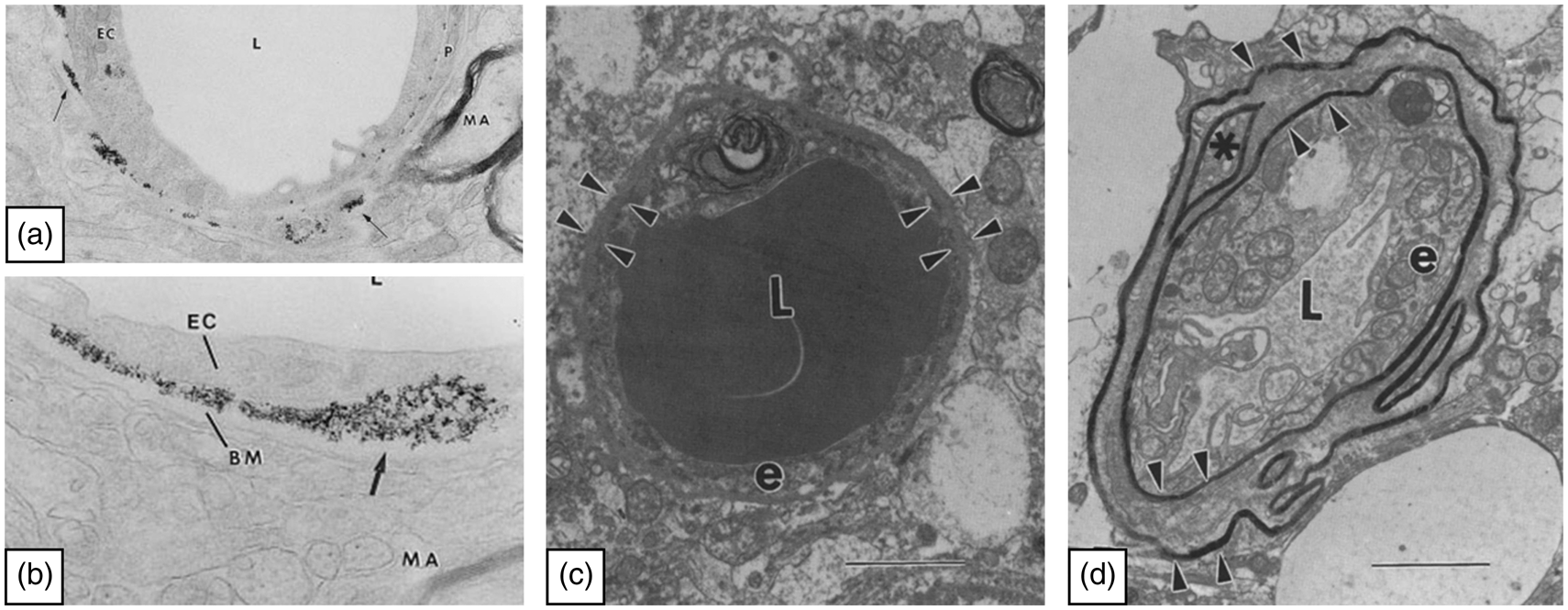
Images showing the restrictive capabilities of the basement membrane and its thickening in Alzheimer’s disease. (a, b) The images illustrate the distribution of the iron particles Feridex along the basement membrane, after experimental opening of the blood-brain barrier using mannitol. The images are adapted from Muldoon et al. 150 Permission American Society for Neuroradiology. L: Lumen, EC: endothelial cell, BM: basement membrane, MA: myelinated axon. (c, d) Images of capillaries from a patient with AD in a region with only few amyloid plaques (c) and a region severely affected by plaques (d). The arrowheads outline the basement membrane as a thin line in the region with only few plaques and in the regions severely affected by plaques the basement membrane have regional thickenings. The images are adapted from Perlmutter. 166 L: Lumen, e: endothelial cell. Permission Springer Nature.
iii) The distribution of individual basement membrane proteins also appears altered in AD, although the reported changes are inconsistent (Table 4). Most studies observing basement membrane thickening also detect increase in collagen IV or report on the deposition of collagen fibrils in the thickened basement membrane.38,135–137 However, in some animal models of AD, the amount of collagen IV decreases over time, which may indicate that remodelling of the basement membrane is a continuous process in AD.37,140,141 Also, this suggests that the vascular basement membrane is being remodelled early and not later in the progression of AD as indicated above, as the preclinical murine models only allow for study up until approximately two years. Supporting the notion that the remodelling of the basement membrane is a continuous process, the basement membrane proteins such as laminins, collagen IV, and nidogen can disrupt the formation of Aβ1-40 and Aβ1-42 fibrils.142,143 Thus, the secretion of these proteins may be a reactive phenomenon to prevent Aβ fibrillation and strengthen the vessel wall by increasing the basement membrane thickness. 139 In addition to the changed expression of collagen IV, the amount of both perlecan and fibronectin is also increased in both capillaries in AD transgenic mouse models and in postmortem brain samples from AD patients.37,38,140 Such changes could accelerate the accumulation of Aβ in the vessel wall as the disease progresses, since perlecan accelerates Aβ fibril formation and stabilizes newly formed fibrils. 144
Although some findings are contradictory, the different studies covered above together suggest that the initial changes in the basement membrane composition with increased deposits of collagen IV and laminin occur to protect against increased levels of Aβ fibrils in the AD brain. This would gradually lead to vessel wall rigidity and reduced cerebral blood flow, hence creating the basis for accumulation of Aβ and subsequent CAA and capillary sprouting.115,145 The reduced cerebral blood flow was further correlated to the accumulation of Aβ in the AD brain. 146 As the disease progresses, the basement membrane may gradually change and favour the deposition of perlecan and fibronectin on behalf of laminin and collagen IV, which will increase the propensity towards Aβ fibrillation,37,144 hence justifying the idea of the basement membrane being responsive before Aβ starts to deposit, but certainly without beneficial long-term effects.
Drug delivery strategies to obtain blood to brain transport can be hampered by the vascular basement membrane
The BBB constitutes a barrier to most pharmacological drugs, allowing only small drugs to enter the brain. 147 A variety of drug-delivery strategies were developed to enable BBB passage, some of which are reviewed in following paragraphs with emphasis on additional permeability restraints of the vascular basement membrane to soluble molecules and nanoparticles. The vascular basement membrane provides a route for transport of soluble proteins like Aβ from the interstitial fluid of the brain parenchyma. These proteins can diffuse alongside the vascular basement membrane and gradually enter the surface of the brain, or they can transverse through the vascular basement membrane and leave the brain via BCECs. 2 These transport routes are also followed when compounds like pharmaceuticals and large proteins are subjected to brain delivery following intrathecal-, intracerebroventricular-, convection-enhanced-, or intranasal delivery. 148 The vascular basement membrane does not constitute a barrier to waste products leaving the brain under normal physiological conditions. 2 In stroke, degradation of the vascular basement membrane is observed and in AD, Aβ can accumulate in the basement membrane hence contributing to the formation of CAA.35,149 Thus, both conditions may compromise routes for diffusion along the vascular basement membrane.
The basement membrane can also form a significant barrier situated beyond the BBB to some nanoparticles passaging through BCECs. This was evidenced in vivo in an experimental model with hyperosmolar opening of the tight junctions of the BCECs in where nanoparticles, of varying size (20–150 nm) and surface charge, were injected into the circulation. All the injected nanoparticles were able to passage the BCECs but some of the nanoparticles failed to transverse through the vascular basement membrane. The gathering of the nanoparticles at the vascular basement membrane was proposed to relate to the physical properties of the iron particles rather than their size suggesting that negative surface charge of the nanoparticles is of importance for interactions between nanoparticles and proteins of the vascular basement membrane (Figure 4). 150
Relatively few studies have investigated the possible barrier properties of the vascular basement membrane to medication and reagents. Three recent studies examined the entry of nanoparticles to the brain parenchyma using focused ultrasound, which induces a transient opening of the BBB that allows for paracellular transfer in between BCECs.151–153 While these studies detected passaging across the BBB of nanoparticles varying in size from 50 nm to 240 nm,151–153 they did not elucidate whether nanoparticles were distributed inside the brain parenchyma, and it was not possible to reveal their distribution further with respect to possible localization inside the basement membrane.
As previously mentioned, the vascular basement membrane also provides a reservoir for soluble factors. This can also include important vascular growth factors and neurotrophic factors with therapeutic potential being continuously expressed and secreted in the adult brain.154,155 A strategy to further allow such growth factors to enter the brain is to transfect BCECs with cDNA injected into the circulation.156–158 Thus, the binding capacity of the vascular basement membrane must be taken into consideration when designing delivery strategies across the BBB, as the growth factors could get trapped there.
Generation of therapeutic antibodies targeted to the diseased brain constitutes promising candidates for the treatment of neurodegenerative disorders. The creation of bispecific antibodies targeting both transferrin receptors (TfRs) of BCECs and a neuronal target of the diseased brain parenchyma shows great potential.159,160 In the studies conducted by Yu et al., 160 bispecific antibodies were designed to target TfRs and the β-secretase, which is involved in the cleavage of amyloid precursor protein and subsequent generation of Aβ. These bispecific antibodies led to decreased Aβ deposition in the brain parenchyma making it plausible that bispecific antibodies are indeed able to pass the brain’s vascular basement membrane and reach their target within the brain parenchyma.159,160
Nanoparticles like liposomes are also tested for their transport across the intact BBB. Liposomes are small vesicles consisting of a double layer of phospholipids, which are usable for delivery of drugs either encapsulated in an aqueous core, in a lipid layer or adhered to the surface of the liposomes. 161 When designing liposomes, the charge of the surface denotes an important parameter since anionic sites are located both at the surface of the BCECs and in the vascular basement membrane. 162 An understanding of the physical and chemical interaction between the drug carrier and the basement membrane is important to predict if the particle will pass or accumulate within the basement membrane. 163 Liposomes only diffuse within the ECM provided the surface charge of the particles are maintained within a well-defined window, largely independently of size, 164 which is in good accordance with experiments performed on iron particles mentioned above. 150 The potential of nanoparticles for passaging the BBB and basement membranes was recently reviewed with respect to experimental approaches for studies using in vitro models of the BBB. 165 The filtering effect of the ECM on the nanoparticles could not be demonstrated when laminin or collagen IV alone were used to form the matrix, 164 which emphasizes the importance of the complex nature of the vascular basement membrane in constituting multiple intertwined proteins.
Perspective
The vascular basement membrane is differently affected in acute and chronic pathological settings.
Stroke is accompanied by degradation of basement membrane proteins and a general loss of BBB integrity. Simultaneously, an increase in the expression of different protease systems capable of cleaving basement membrane proteins is also observed. However, a clear causal relationship between increased protease activity and protein degradation in vivo has not been firmly established, although suggestive temporal and topographical correlations have been shown by several groups. How the major changes in molecular composition and thickening of the basement membrane in AD occur is not well established. Thus, the mechanisms underlying the structural changes of the basement membrane deserve further investigation. Halting the composite changes of the basement membrane in neurodegeneration could possibly ease the delivery of macromolecular drugs and nanoparticles into the brain, and curtail the changes in acute stroke could limit barrier leakage and haemorrhagic consequences.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
