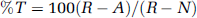Abstract
By isolating for the first time ever a peptide transporter from the blood—brain barrier (BBB) and developing an antisense that selectively targets the brain-to-blood efflux component, we were able to deliver a therapeutic concentration of the neurotrophic peptide pituitary adenylate cyclase-activating polypeptide (PACAP) 27 to brain in animal models of Alzheimer's and stroke. Efflux pumps at the BBB are major causes of BBB impermeability to peptides. PACAP is neuroprotective in vitro in femtomole amounts, but brain uptake of PACAP27 is limited by an efflux component of peptide transport system-6 (PTS-6). Here, we characterized, isolated, and sequenced this component of PTS-6, identifying it as β-F1 ATPase, and colocalized it with PACAP27 on BBB endothelial cells. Antisenses targeting the BBB inhibited PACAP27 efflux, thus increasing brain uptake of PACAP27. Treatment with antisense +PACAP27 improved cognition in a mouse model of Alzheimer's disease and reduced infarct size after cerebral ischemia. This represents the first isolation from BBB tissue of a peptide transporter and shows that inhibition of peptide efflux pumps is a potential strategy for drug delivery to brain.
The blood—brain barrier (BBB) is a major obstacle to the delivery of promising therapeutics to the central nervous system. The presence of brain-to-blood saturable transport systems (efflux pumps) has emerged as a major reinforcer of BBB impermeability (Taylor, 2002; Begley, 2004; Banks, 2005). For example, pituitary adenylate cyclase-activating polypeptide (PACAP), the newest member of the secretin/glucagon/vasoactive intestinal polypeptide (VIP) family of regulatory peptides, is a pleuripotent peptide with neuroprotective, endocrine, and vasodilatory effects (Vaudry et al, 2000). In vitro, PACAP is neuroprotective in femtomole concentrations (Arimura et al, 1994). In vivo, the 38 amino-acid form of PACAP (PACAP38) reverses ischemic damage even when given intravenously and 24 h after 4 vessel stroke (Uchida et al, 1996). However, the physicochemical and pharmacokinetic properties of PACAP38 make it difficult to work with. Use of the C-terminal-truncated 27 amino-acid form (PACAP27) is limited by an efflux component of peptide transport system-6 (PTS-6), which decreases brain uptake by approximately 10-fold (Banks et al, 1993). Reduction in brain uptake by efflux transporters of 4- to 10-fold is enough to render many otherwise promising central nervous system therapeutics ineffectual (Taylor, 2002; Löscher and Potschka, 2002; Begley, 2004; Thomas, 2004).
Inhibition of the efflux pumps should increase brain uptake of potential therapeutics and so could be used as a novel central nervous system drug delivery strategy. However, identification of most efflux pumps for peptides are unknown and, because of technical difficulties, no transporter, peptide or otherwise, has ever been originally isolated from BBB tissue. How to specifically inhibit efflux pumps once identified is also problematic. However, in the case of PACAP, inhibition of its efflux pump would increase retention of the endogenous PACAP produced in the brain as well as increase brain retention of the circulating endogenous and peripherally administered PACAP that crosses the BBB. Here, we present the first isolation of a peptide transporter from BBB material, identifying the efflux component of PTS-6 as β-F1 ATPase. We developed enzymatically resistant antisenses that selectively inhibited the PTS-6 efflux pump after peripheral administration. We show that inhibition of PTS-6 results in a dramatic increase in PACAP27 uptake by brain, improves learning in a mouse model of Alzheimer's disease, and reduces infarct size after middle cerebral artery occlusion in mice. These results show that BBB transporters can be isolated and identified, validate the inhibition of BBB efflux pumps as a therapeutic strategy, and also show how antisenses can be developed that target the BBB.
Results
We first used the in vitro BBB model of brain microvessels isolated from mouse brain to characterize I-PACAP27 binding. Figure 1A shows the relation between incubation time and the percent-specific binding of I-PACAP27 to mouse brain microvessels in the range of 1.0 to 20 mins (n = 3 per time). Specific binding reached a maximum by approximately 20 mins. Results fitted to a one-site binding hyperbolic model showed that 50% of maximal binding occurred by 3 ± 1.2 mins. On the basis of this, further studies were conducted at 2.5 mins incubation times. Specific binding was temperature dependent (Figure 1B; n = 3). A statistically significant difference occurred among the groups (F(2,6) = 15.10, P < 0.05) and specific binding of IPACAP27 at 4°C was significantly less compared with 25°C and 37°C (P < 0.05). Specific binding of IPACAP27 was affected by pH (Figure 1C; n = 3) as shown by analysis of variance (ANOVA; F(6,14) = 6.39, P < 0.005), but the range test showed that only the pH 6.1 value differed from the values of 7.2, 7.7, and 8.1. Subsequent experiments were conducted at pH 7.4 at room temperature for 2.5 mins incubation time.
Characterization of PACAP27 binding to brain microvessels. (
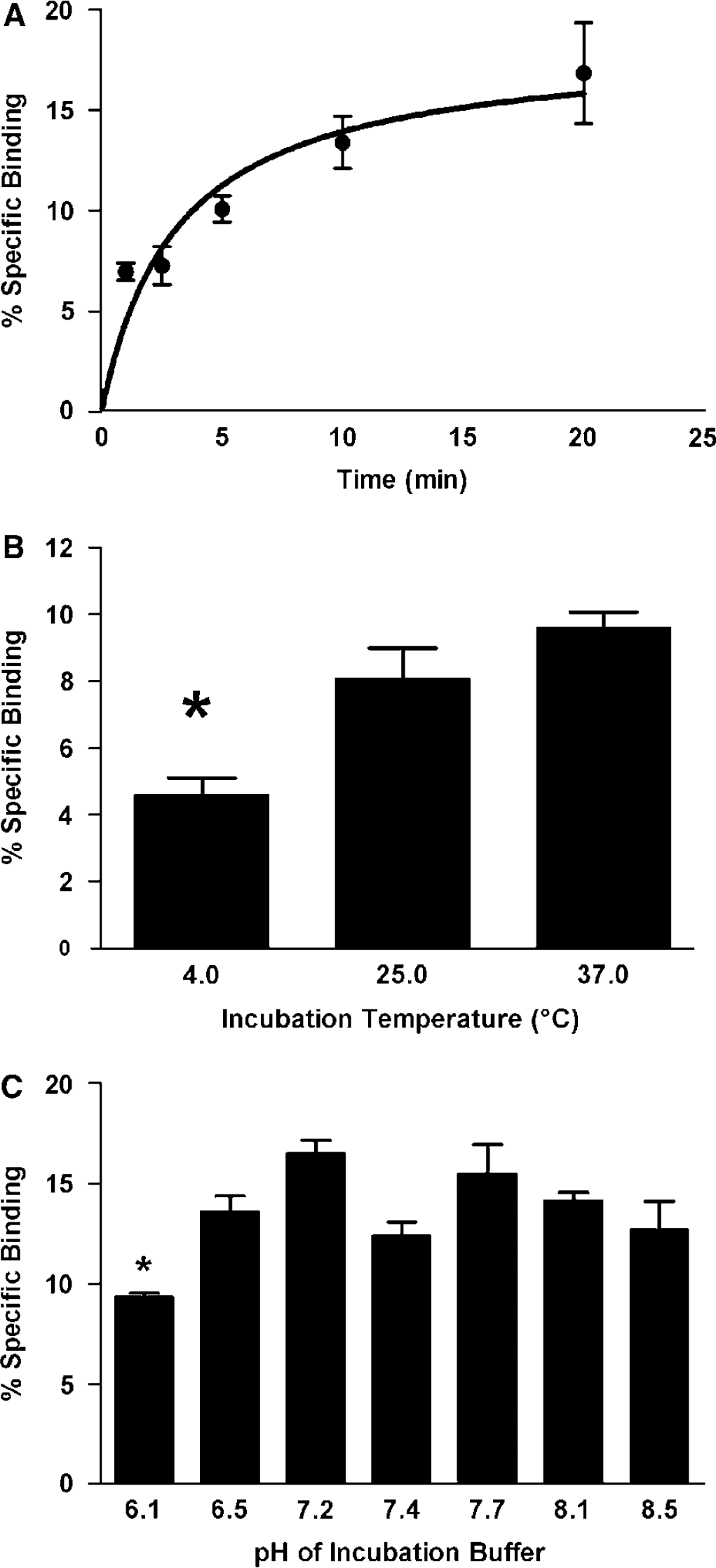
Figure 2A shows self-inhibition of PACAP27 binding by increasing concentrations of unlabeled PACAP27 (1 to 100 ng per tube; n = 2 to 3 per concentration) in the mouse brain microvessels. The results are expressed with the specific binding at 53 pmol/L of I-PACAP27 (no unlabeled PACAP27) set to 100% and nonspecific binding at 0% and each data point represents two to three replicates. An inverse relation existed between the log concentration of unlabeled PACAP27 and specific binding (n = 6, r = 0.982, P < 0.001, slope = −28.1, y intercept = 72.9). On the basis of the results in Figure 2A, an expanded dose—response curve of 9 concentrations (0.5 to 180 nmol/L; n = 3 per concentration) was used to calculate KD and Bmax (Figure 2B). The binding data were analyzed for best fit comparing a one-site and two-site hyperbolic-binding model with the Prism program. The results indicated a one-site model produced the better fit with a dissociation constant (KD) of 38.56 ± 8.94 nmol/L and binding maximum (Bmax) of 87.48 ± 8.69 fmol.
Kinetics of PACAP27 binding to brain microvessels. (
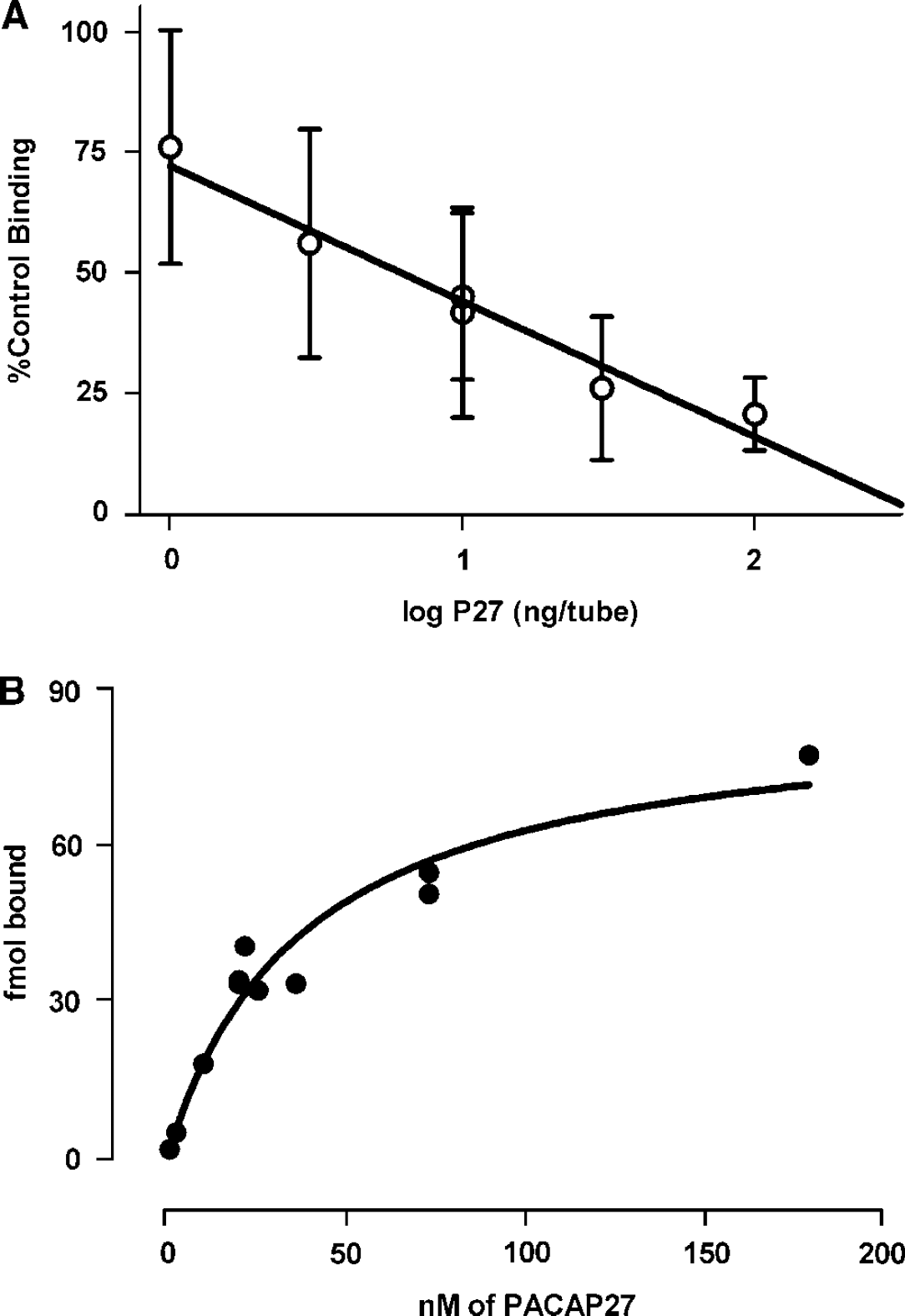
To determine the specificity of the PACAP binding site, mouse brain endothelial cells were incubated with PACAP27 in the presence of 50 nmol/L concentration of one of the two naturally occurring forms, PACAP27 and PACAP38, one of two PACAP antagonists, PACAP (6 to 27) and PACAP (6 to 38), or VIP; n = 6 per group. ANOVA showed a statistically significant effect among the groups shown in Figure 3A: F(5,30) = 11.94, P < 0.0001. The binding of PACAP27 to cells was not affected by VIP. The displacing affinity of the PACAP-related peptides were not statistically different, but had an arithmetic hierarchy of PACAP38 > PACAP27 > PACAP (6 to 38) > PACAP (6 to 27) for inhibiting PACAP27 binding to brain microvessels.
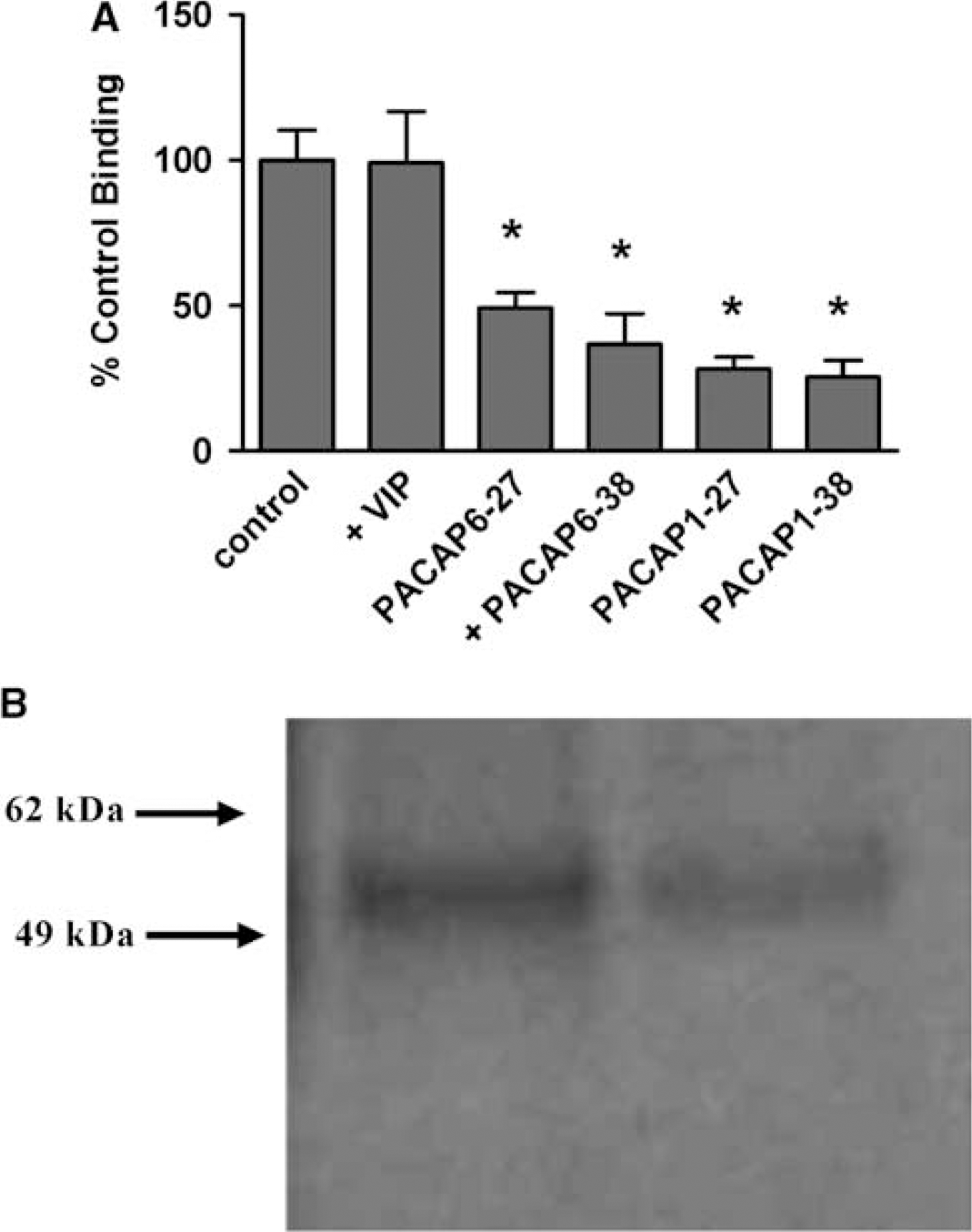
Characterization and Isolation of PTS-6. (
Autoradiography of I-PACAP27 crosslinked to isolated microvessel membranes showed a prominent band at 55 kDa (Figure 3B). Analysis by mass spectrometry showed this protein to be ATP synthase β-subunit, also known as β-F1 ATPase.
A single injection containing the three antisenses-directed against β-F1 ATPase administered by intracerebroventricular (icv) injection as a cocktail produced an inhibition in the transport of icv administered I-PACAP27 that began within 30 mins of administration and lasted 48 h (Figure 4A): F(10,54) = 8.74, P < 0.001, n = 6 to 7 per group. The icv antisense cocktail as assessed by t-test had no effects on the ligands of three other BBB efflux systems (Figure 4B, n = 8 per group for I and β-endorphin and n = 14 for Tyr-MIF-1) nor for I-PACAP38 (n = 17), but did produce a significant inhibition in I-PACAP27 efflux (t = 4.5; P < 0.005, n = 4 per group), where all ligands were also administered icv 24 h after the cocktail.
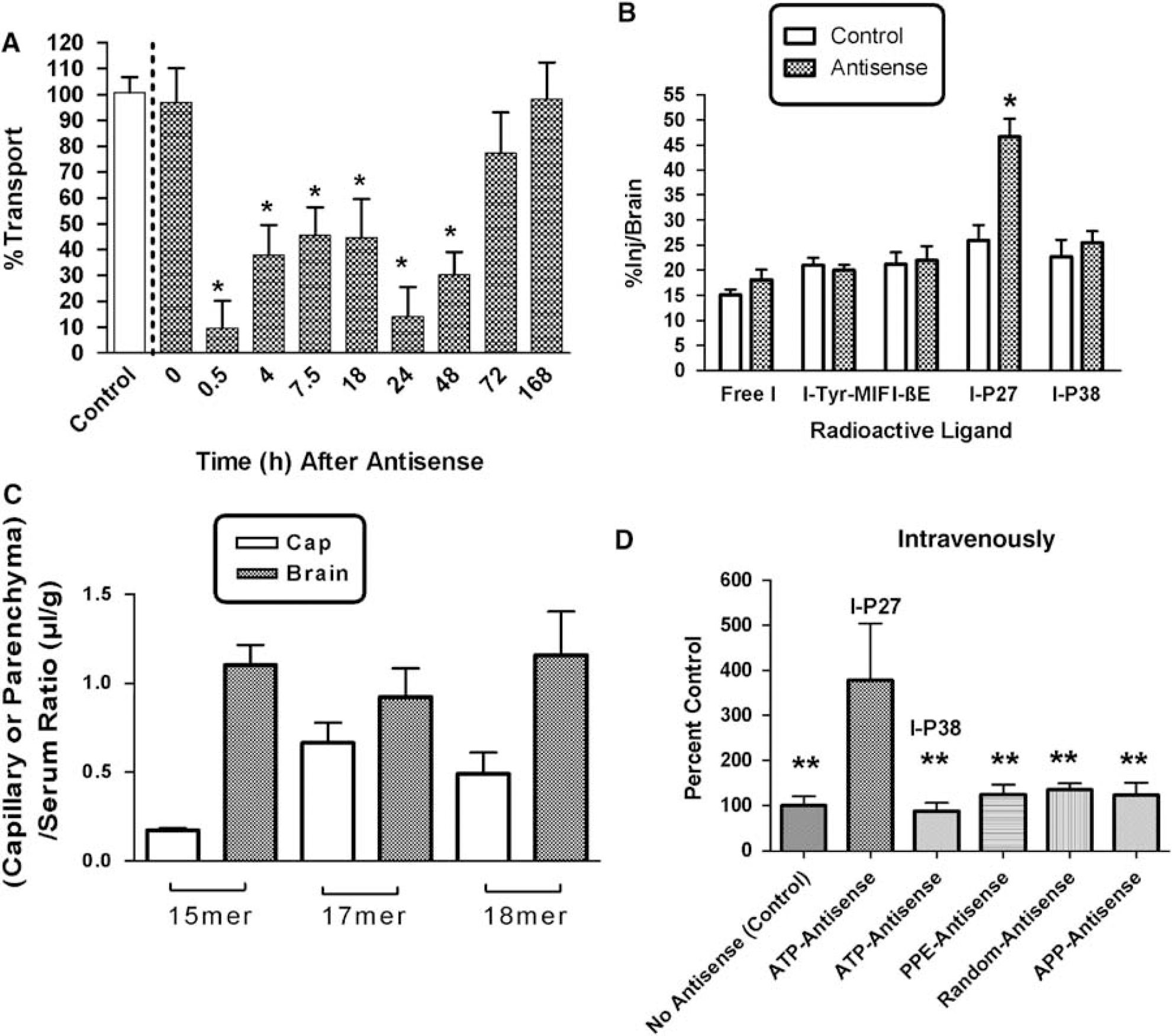
Antisense and PACAP. (
We then determined whether the antisense molecules were effective after i.v. administration. Results for capillary depletion (Figure 4C) showed that the 32P-labeled antisenses were all taken up by the capillary bed and crossed the BBB to enter brain parenchyma. A single injection of the antisense cocktail by tail vein 24 h before study produced a fourfold increase in the uptake of I-PACAP27 (Figure 4D), but not I-PACAP38, delivered by brain perfusion. The antisenses directed against preproenkephalin, amyloid precursor protein, or the random 42mer produced no statistically significant effect on uptake of I-PACAP27.
Merged images of PACAP27 and β-F1 ATPase are shown in Figure 5. Panels A—C show a capillary with exogenous PACAP27 added and panels D—F show a control capillary in which PACAP27 was not added. A and D show β-F1 ATPase immunoactivity (green), B and E show PACAP27 immunoactivity (red) and C and F show colocalization in merged images. As expected, no PACAP27 immunoactivity was seen (panels E and F) when PACAP27 was not preincubated. Panel C shows that PACAP27 and β-F1 ATPase immunoactivities colocalize.
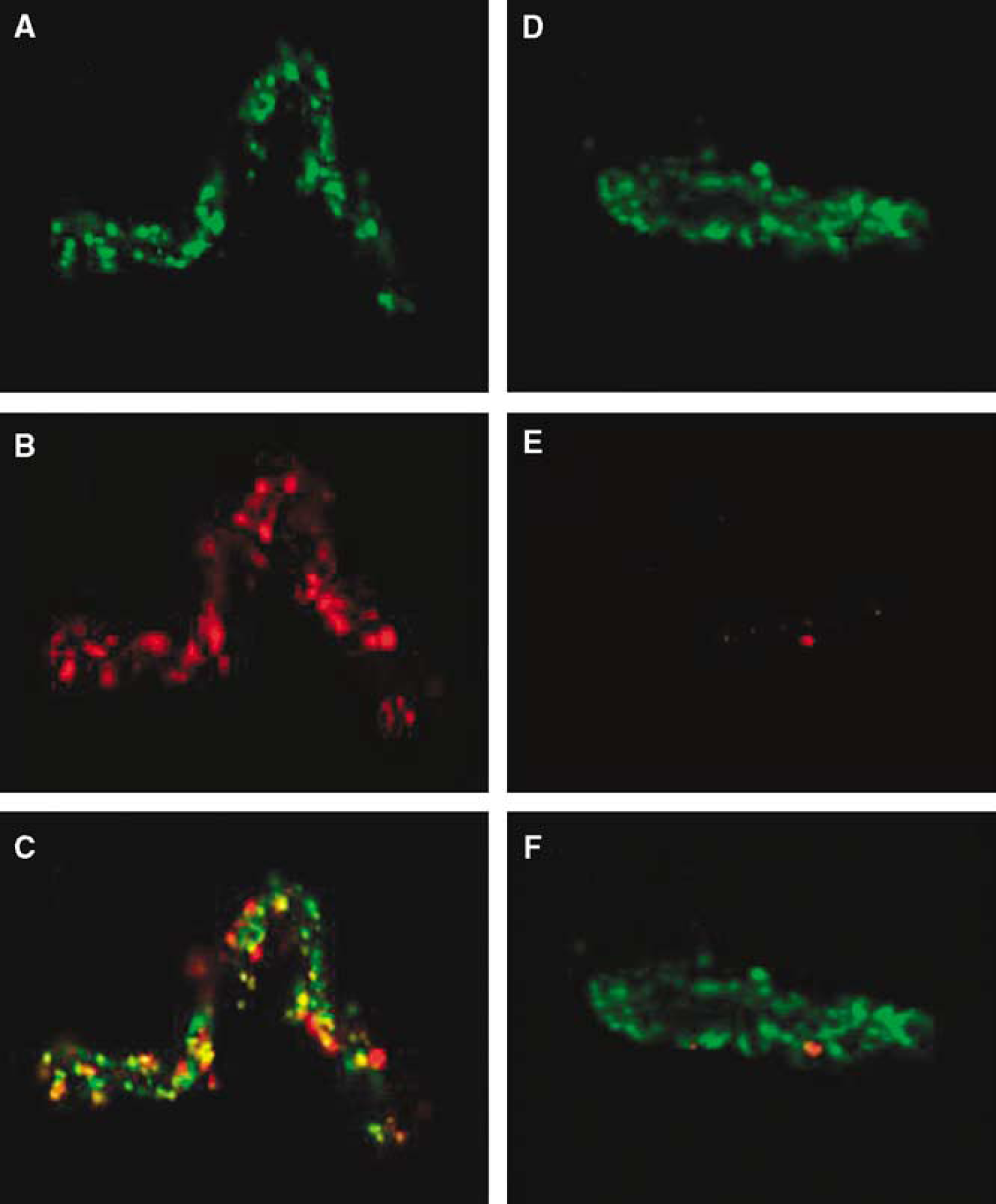
Colocalization of β-F1 ATPase and PACAP27 in mouse brain microvessels is shown by the yellow spots in (
To evaluate the effect of the PTS-6 antisenses and PACAP on ischemia in the mouse, the neurologic deficit score, the mortality, and the infarct volume after permanent middle cerebral artery occlusion (pMCAO) were determined. Two-way ANOVA for neurologic deficit score (Figure 6A) with +/–PACAP as one independent variable and +/–antisense as the other showed a significant effect (F(1,88) = 4.87, P < 0.05)) for the antisense treatment, but no effect of PACAP treatment or interaction. Newman—Keuls found no significant effects. Representative images of the anterior surface of a series of four 2,3, 5-triphenyltetrazolium chloride-stained coronal sections are shown in Figure 6B. Two-way ANOVA for infarct volume showed a significant effect (F(1,74) = 9.09, P < 0.005) for treatment with antisense, but not for PACAP or interaction (Figure 6C). Newman—Keuls post-test showed the saline—saline group to be significantly different from the saline—antisense and the PACAP—antisense-treated groups (P < 0.05). A decrease in mortality from 20% (6 deaths out of 30 mice) for saline—saline-treated mice to 10% (2 deaths out of 20 mice) for PACAP—antisense-treated mice 24 h after ischemia was not significant by Fisher's exact test with two-sided P-value.
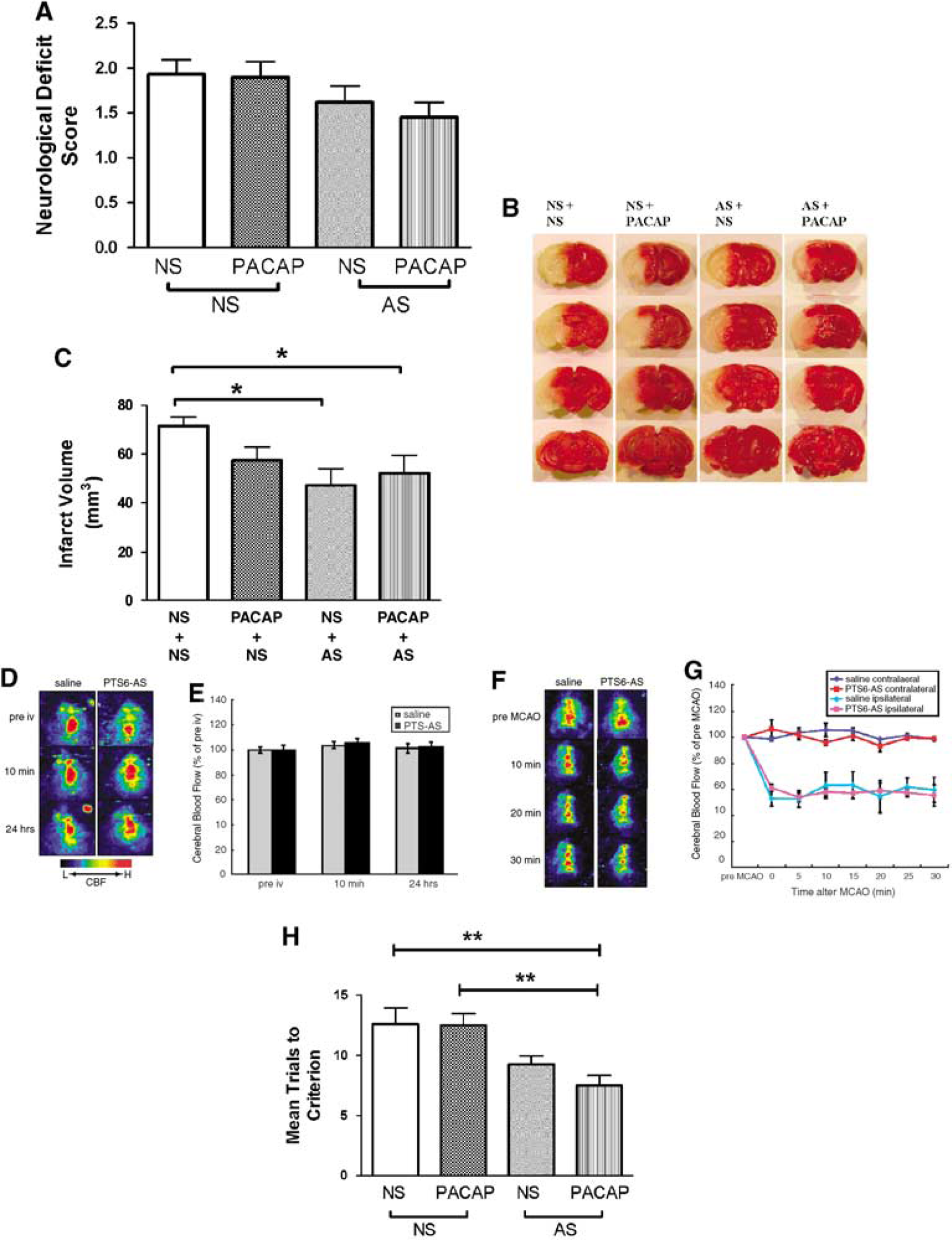
Effects of antisense cocktail directed against β-F1 ATPase on models of brain insult. Antisenses (AS) or saline (NS) were injected into the jugular vein at 24 h before ischemia. (
To rule out the possibility that the PTS-6 antisenses exerted their effects on ischemia by altering cerebral blood perfusion, we performed laser doppler perfusion imaging studies. The Cerebral blood flow (CBF) mean value for cortex at 10 mins and 24 h after intravenous (i.v.) injection of the antisenses were not significantly changed between PTS-6 antisenses and the saline-injected group (n = 6; Figures 6D and 6E). Moreover, during MCAO up to 30 mins, the CBF value on the contralateral side or ipsilateral the side of cortex were also not different between these groups (n = 3 to 4; Figures 6F and 6G).
We also tested the effect of PTS-6 antisenses and PACAP on learning in 12-month-old SAMP8 mice, an animal model of Alzheimer's disease (Morley et al, 2002). The two-way ANOVA showed a statistical effect for antisense (F(1,27) = 43.4, P≪0.01)], but not for PACAP or interaction (Figure 6H). Newman—Keuls showed that mice given antisense + PACAP learned more quickly than control mice or mice given only PACAP (P<0.01).
Discussion
Pituitary adenylate cyclase-activating polypeptide is a potent neuroprotectant whose delivery to brain is complicated by the BBB. PACAP is transported across the BBB by PTS-6 that likely represents a family of influx and efflux transporters (Banks et al, 1993). Therapeutic use of PACAP27 in particular is hampered by a brain-to-blood efflux transporter. Our finding of a pH and temperature sensitive saturable binding site for I-PACAP27 on isolated mouse brain microvessels is consistent with brain endothelial cells have both PACAP transporter (Uchida et al, 1996) and receptor (Knutsson and Edvinsson, 2002; Nonaka et al, 2005) activities. PACAP binds to three receptor subtypes: PAC1, VPAC1, and VPAC2 (Shioda, 2000; Vaudry et al, 2000). The PAC1 receptor has several splice variants and is specific for PACAP, whereas the other two receptors also bind VIP (Hasimoto et al, 1993; Spengler et al, 1993). As we found that I-PACAP27 binding was neither displaced by VIP nor showed an inhibition pattern classic for PAC1 (Salomon et al, 1993; Rodriguez-Henche et al, 1994; Salano et al, 1996; Shioda, 2000), we concluded that brain endothelial cells likely possess a novel-binding site.
Identification of the PACAP27-binding protein by mass spectrometry confirmed that it was not one of the previously identified receptors for PACAP. The identified protein, β-F1 ATPase, colocalized with PACAP immunoactivity on freshly isolated brain endothelial cells. β-F1 ATPase has been shown to act as either a binding protein or a transporter in other tissues for a number of other ligands unrelated to PACAP. Originally identified as an extra-membrane component of ATPase in mitochondria (ATP synthase β-subunit), it was subsequently found to be identical to the apolipoprotein A-1 receptor that transports high-density lipoproteins at hepatocytes (Martinez et al, 2003). It is also expressed in neurons where it acts as a receptor for enterostatin, a pentapeptide involved in feeding (Park et al, 2004). Cultured brain endothelial cells, but not aortic endothelial cells, express and secrete this protein and expression is increased by cholesterol, insulin, and retinoic acid (Weiler-Guttler et al, 1990; Mockel et al, 1994). However, the function of β-F1 ATPase in brain endothelial cells was unknown. Interestingly, two other lipoprotein-binding molecules have been found to act as efflux transporters for peptides.
P-glycoprotein is a member of the ATP-binding cassette family, binds Apo A-1, and transports some of the small opiate peptides and cyclosporin in the brain-to-blood direction (Begley, 2004). Low-density lipoprotein receptor-related protein-1 acts as an efflux transporter for amyloid β protein (Deane et al, 2004).
Inhibition of PTS-6 efflux should selectively increase PACAP in the brain both by decreasing the loss of PACAP endogenous to the brain and by increasing the retention of PACAP entering the brain from the blood. We showed that both of these events occurred in mice treated with a cocktail of phosphorothioate antisenses directed against β-F1 ATPase. Many reviews have stated that antisense molecules cannot cross the BBB. However, those statements in most reviews were not referenced and were based on untested assumptions about how the BBB and antisense would interact. Phosphorothioate oligonucleotides are enzymatically resistant (Jaeger and Banks, 2004) and many are taken up by and transported (Banks et al, 2001, 2006) across brain endothelial cells. Peptide nucleic acids, another class of antisense molecules, also effectively cross the BBB (Tyler et al, 1999). The antisenses developed here against PTS-6 were taken up by the capillaries and by brain tissue after the i.v. injection of their 32P-labeled versions. A single icv injection of the antisenses inhibited for approximately 48 h the transport of I-PACAP27 injected into the brain's lateral ventricle. Efflux systems for an inorganic ion efflux system (using iodide as the ligand), PTS-1 (using the tetrapeptide Tyr-MIF-1 as the ligand), and P-glycoprotein (using β-endorphin as the ligand) were not affect by the antisenses (Davson and Hollingsworth, 1973; Banks and Kastin, 1990; King et al, 2001). When injected iv, the antisenses increased brain uptake of I-PACAP27 perfused through the vasculature of the brain, whereas other antisenses were ineffective. I-PACAP38 influx or efflux was not affected by the antisense cocktail, showing that β-F1 ATPase acts as an efflux pump for PACAP27, but not PACAP38. This is consistent with previous pharmacokinetic work that suggested that the efflux systems for PACAP27 and PACAP38 were different because PACAP27 efflux is not modulated by a luteinizing hormone-releasing hormone, whereas PACAP38 efflux is modulated (Banks et al, 1993).
Pituitary adenylate cyclase-activating polypeptide is an extremely potent neuroprotectant. As noted in discussion, femtomole concentrations are effective in vitro and picomole doses are effective in vivo. More specifically, small doses of PACAP38 are protective against amyloid β-protein neurotoxicity in vitro (Onoue et al, 2002) and against ischemic damage in vivo (Uchida et al, 1996). Thus, we postulated that PTS-6 inhibition could allow both endogenous and exogenous PACAP27 to exert therapeutic effects in rodent models of Alzheimer's disease and stroke. Work based on PACAP38 indicates that very small amounts of PACAP are needed for neuroprotection (Banks et al, 1993). Thus, inhibition of PTS-6 alone could allow enough endogenous PACAP27 to enter or remain in brain to be therapeutically effective. Our results in the ischemic model support this idea, as treatment with antisense alone reduced infarct size. However, the Alzheimer's model required the combination of i.v. PACAP27 and antisense to improve learning. In the stroke model, the improvements cannot be ascribed to increased cerebral blood flow in either the normal or ischemic brain. We conclude that inhibition of PTS-6 can be effectively achieved with peripheral administration of antisense and that such inhibition allows endogenous and exogenous PACAP27 to attain therapeutic levels in animal models of central nervous system disease. In summary, we isolated the PACAP27 efflux component of PTS-6 from brain endothelial cells and identified it as β-F1 ATPase. PACAP27 and β-F1 ATPase immunoactivity colocalized on brain endothelial cells. Inhibition of β-F1 ATPase with specific antisenses reduced efflux of I-PACAP27 but not other substances including I-PACAP38 from brain and greatly increased influx into brain. Inhibition of PTS-6 resulted in a therapeutic effect in mouse models of stroke and Alzheimer's disease. We conclude that β-F1 ATPase is the efflux component of PTS-6 and its inhibition with peripherally administered antisense allows PACAP27 to exert its neuroprotective effects in models of Alzheimer's disease and stroke.
Materials and methods
Radioactive Labeling of PACAP
Iodination of 5.0 µg of PACAP27 (Bachem, Torrance, CA, USA) with 131I was performed by the lactoperoxidase method. Iodinated PACAP27 (I-PACAP27) was purified on a C18 column by RP-HPLC. Incorporation of 131I as determined by acid precipitation was >95% and specific activity was approximately 1.82 mCi/mg.
Brain Microvessel Isolation
Cerebral microvessels were isolated from mice by a modification of a method described by Gerhart et al (1988). All reagent volumes were proportionally adjusted for the quantity of tissue processed and unless otherwise noted, all reagents were of cell culture quality from Sigma Chemical Company (St Louis, MO, USA). All glassware was precoated with lactated Ringer's solution containing 1% bovine serum albumin to minimize sticking and to maximize recovery of microvessels. Male CD-1 mice from our in-house colony weighing 25 to 40 g were anesthetized with urethane (0.5 g/kg) before decapitation. This and all other animal studies were performed under protocols approved by the VA animal care and use committee, and in accordance with IACUC guidelines. For each microvessel isolation procedure, whole brains from 10 mice were dissected from surrounding structures, the pituitary and the pineal glands discarded, and the remainder placed in stock buffer (25 mmol/L HEPES, 1% dextran in minimum essential medium (Gibco Laboratories, Grand Island, NY, USA) pH 7.4) on ice. The tissue was homogenized with 10 strokes in a glass tissue grinder with a teflon pestle. The homogenate was then filtered through a series of nylon mesh membranes (300 µm, followed by 2×100 µm; Spectrum Scientific Corp., Houston, TX, USA), mixed with an equal volume of 40% dextran in stock buffer and centrifuged at 5000g for 15 mins at 4°C. The myelin layer was carefully removed. The pellet was resuspended in stock buffer and filtered through a 25 µm nylon mesh membrane (Bio-Design, Carmel, NY, USA). The microvessels were washed from the surface of the nylon mesh with stock buffer four times, collected, and centrifuged at 5000g for 15 mins at 4°C. The purity and quantity of each preparation was routinely checked by light microscopy.
Binding Study
Freshly isolated microvessels (30 µg protein) were resuspended in 45 µL of incubation buffer (129 mmol/L NaCl, 2.5 mmol/L KCl, 7.4 mmol/L Na2HPO4, 1.3 mmol/L KH2PO4, 0.63 mmol/L CaCl2, 0.74 mmol/L MgSO4, 5.3 mmol/L glucose, 0.1 mmol/L ascorbic acid, pH 7.4) containing 1% bovine serum albumin (BSA). Microvessel protein levels were determined with the Bio-Rad DC protein assay kit (Hercules, CA, USA). Incubation buffer, I-PACAP27 (4 µL of 0.8 nmol/L), and any additives as indicated below were mixed with the 45 µL suspension of cells to a final volume of 60 µL (to give a final I-PACAP27 concentration of 53 pmol/L) and incubated for 2.5 mins at room temperature unless otherwise specified. At the end of incubation, samples were assayed for protein-bound radioactivity by vacuum filtration through Gelman Sciences glass microfiber filters (Ann Arbor, MI, USA) pretreated with 0.5% polyethyleneimine. Filters were washed three times with 4mL of incubation buffer at 4°C. The radioactivity trapped on the filters was measured using a γ-counter. Nonspecific sticking of PACAP to the filters was determined in the absence of microvessels and averaged approximately 20% of total binding in the presence of microvessels. Total binding (%) was taken as the binding in the presence of microvessels. Nonspecific binding was defined as residual binding to microvessels in the presence of 1 µg per tube (4.7 µmol/L) of unlabeled PACAP. Specific binding was taken as total binding minus nonspecific binding. All experiments were performed in triplicate on at least three occasions and the results reported as means with their standard errors.
Inhibition of Specific Binding of PACAP27
Self-inhibition was tested by adding varying amounts of unlabeled PACAP27. Results are expressed relative to the specific binding in the absence of unlabeled PACAP27 (% control binding). The femtomole specifically bound was calculated by dividing the percent of specific binding by 100 and multiplying by the pmol/L (I-PACAP27 and any unlabeled PACAP27 combined) of the incubation solution and by the volume. This was plotted against the nmol/L in the media and the relation fitted to a one-site hyperbolic-binding model.
Inhibition of I-PACAP27 Binding by PACAP Related Peptides
The effects of 50 nmol/L of unlabeled PACAP27, PACAP38, the PACAP antagonists PACAP 6 to 27 and PACAP 6 to 38 (Bachem), and VIP (Sigma) on I-PACAP27 specific binding was determined. Results were expressed as percent of control (I-PACAP27 only).
Isolation of PACAP27 Transporter (PTS-6)
Microvessels prepared from 10 male CD-1 mice were lysed in water for 2 h at 4°C and centrifuged to obtain the membrane pellet. I-PACAP27 (105 c.p.m.) and the membrane fraction were incubated for 20 mins in a volume of 20 µL of incubation buffer at room temperature. Disuccinimidyl suberate at a concentration of 0.05 mol/L was used to crosslink the I-PACAP27 to membrane binding sites by incubating for 15 mins at room temperature. The crosslinked membranes were then subjected to hydrophobic fractionation using the MEM-PER kit (Pierce, Rockford, IL, USA). The hydrophobic fraction was loaded in duplicate onto a 10% Bis/Tris denaturing gel. One half of the gel was subjected to Western transfer to a nylon membrane and stained with Coomassie blue. The other half of the gel was air-dried and subjected to autoradiography. After visualization of the radioactive bands, the autoradiogram was aligned to the Coomassie-stained membrane and the corresponding band was cut out. The 55 kDa band was sent for protein identification to the Taplin Mass Spectrometry Facility (Harvard Medical School).
Effects of Antisense Oligonucleotides to ATP Synthase β Subunit on Brain-to-Blood Efflux
Three non-overlapping regions of the mouse ATP synthase β-subunit were used to construct three enzymatically resistant phosphorothioate oligonucleotide antisense molecules (Midland Certified Reagent Company, Inc. Midland, TX, USA): an 18mer to the 144-9 region: 5′-(_P = S)TC CAATGACATTCATGAT-3′; a 15mer to 328 to 332 region: 5′-(_P = S)CTGGTAGCCTACAGC-3′; a 17mer to the 249 to 353/4 region: 5′-(_P = S)ATCGATCCCTTCTTGGT-3′. The effect of these antisense oligonucleotides on PTS-6 efflux activity was determined after both their icv and i.v. administration.
To test the effectiveness of PTS-6 antisense oligonucleotides after their icv administration, 2-month-old male CD-1 mice kept on a 12/12 h light/dark cycle with food and water freely available were anesthetized on the day of study with 0.15mL of 40% urethane. The scalp was removed and a hole made into the lateral ventricle, 1.0 mm lateral and 1.0 mm posterior to the bregma, with a 26-gauge needle with a tubing guard that kept the depth of the holes constant (3.0 to 3.5 mm). Mice received an icv injection of 1.0 µL lactated Ringer's solution with 1% BSA containing 100 ng of each of the three antisense oligonucleotides. Immediately (t = 0) or at varying times (0.5, 2, 4, 8, 18, 24, 36, 48, or 72 h) after the antisense injection, mice received a second icv injection of 1.0 µL lactated Ringer's solution with 1% BSA containing 1.5 (105) c.p.m. of I-PACAP27. Mice were decapitated 10 mins after the icv injection of IPACAP27, the brains removed, and the level of residual radioactivity in brain determined by counting in a γ-counter. Mice that had been killed by an overdose of urethane were used to determine the level of residual radioactivity in brain in the total absence of transport (R) and mice that did not receive antisense were used to determine the level of residual activity in unimpaired transport (N). The percent of transport was calculated with the equation:
Specificity of the icv administered antisense oligonucleotides for I-PACAP27 efflux was tested by determining their effects on efflux of the PTS-1 ligand Tyr-MIF-1, the p-glycoprotein ligand β-endorphin, free iodine, which is a ligand for a perchlorate-sensitive active transporter and on PACAP38. Mice received an icv injection of lactated Ringer's solution with 1% BSA either containing or not containing (control) the three oligonucleotide antisenses. After 24 h, the mice received a second icv injection containing radioactive PACAP38, Tyr-MIF-1, β-endorphin, or free iodine were decapitated 10 mins later, and the level of residual activity in brain determined in a γ-counter. For comparison, I-PACAP27 transport was also studied 24 h after the icv injections. Results were expressed as the percent of the injected dose remaining in brain.
To test the effectiveness of PTS-6 antisenses after their i.v. administration, 2-month-old male CD-1 mice kept on a 12/12 h light/dark cycle with food and water freely available were given an injection by tail vein of 0.9% saline with or without 10 µg per mouse of each of the three antisenses. To determine the specificity of the PTS-6 antisenses on I-PACAP27 efflux, three other antisenses were also injected iv. These were a 10mer directed against the methionine enkephalin region of preproenkephalin (5′- (_P = S)TCA TGA AGC C-3′) (Banks et al, 2006), a 42mer directed at the amyloid-β mid-region of amyloid precursor protein (5′-(_P = S)GGCGCCTTTGTTCGAACCCACATCTTCAGCAAAGAACACCAG-3′; Kumar et al, 2000), and a random 40mer (5′-(_P = S)GATCACGTACACATCGACACCAGTCGCCATGACTGAGCTT). After 24 h, the mice were anesthetized with urethane and both jugular veins exposed. The thorax was opened, the descending thoracic aorta clamped, both jugular veins severed, and lactated Ringer's solution containing I-PACAP27 (250,000 c.p.m./ml) perfused through the left ventricle of the heart at the rate of 2 mL/min. I-PACAP38 was perfused in some mice treated with PTS-6 antisense. After 5 mins, the perfusion was stopped and 20mL of lactated Ringer's solution was infused through the left ventricle of the heart to wash out the vascular space of the brain. The level of radioactivity was determined in the brain and in an aliquot of the perfusion fluid and results expressed as the brain/perfusion ratio in units of microliter per gram.
Brain and BBB Uptake of Antisense
We used the capillary depletion method to determine whether the antisenses were taken up by the BBB (Triguero et al, 1990; Gutierrez et al, 1993). Each antisense was end-labeled by mixing 10 µg of the antisense with 1.5 µL of 10× kinase buffer, 1.5 µL of T4 polynucleotide kinase (New England Biolabs, Ipswich, MA, USA) and 10 µL of [γ32P]ATP. The mixture was then incubated in a 37°C water bath for 45 mins. After incubation, the kinase was inactivated by heating the sample to 65°C for 5 mins. Labeled antisense was removed from the reaction mixture by ethanol precipitation followed by centrifugation. For the initial ethanol precipitation, the labeled P-Olg was mixed with 80 µL of water, 10 µL of 3 mol/L sodium acetate (pH = 5.0), 2 to 5 µL of Pellet Paint Co-Precipitant (Novagen, La Jolla, CA, USA) and 300 µL of cold ethanol. This mixture was incubated at −70°C overnight. The labeled antisense was separated by centrifugation at 13,000 r.p.m. for 20 mins. After removing the supernatant, the pellet (containing the labeled antisense) was resuspended in 500 µL of cold ethanol and subjected to centrifugation at 13,000 r.p.m. for 15 mins. This step was repeated three times to remove any remaining unincorporated [γ32P]ATP. After the final wash, the pellet was resuspended in 100 µL of water and the level of radioactivity was determined by counting an aliquot of the sample in a scintillation counter. Mice were anesthetized and given an injection into the jugular vein of 0.2mL saline containing approximately 106 d.p.m. of the labeled antisense. At 10 mins later, arterial blood was obtained from the descending aorta, the thorax opened and the descending thoracic aorta clamped, both jugulars severed, and the blood washed out of the brain by injecting 20mL of lactated Ringer's solution into the left ventricle of the heart in 60 secs. The brain was removed and emulsified in a glass homogenizer (8 to 10 strokes) at 4°C in a ninefold volume of physiologic buffer (10 mmol/L HEPES, 141 mmol/L NaCl, 4 mmol/L KCl, 2.8 mmol/L CaCl2, 1 mmol/L MgSO4, 1 mmol/L NaH2PO4, and 10 mmol/L D-glucose adjusted to pH 7.4). Dextran solution was added to the homogenate to a final concentration of 26%. An aliquot was centrifuged at 5400g for 15 mins at 4°C in a swinging bucket rotor. The pellet containing the brain microvessels and the supernatant containing the brain parenchyma were carefully separated. Results were expressed as capillary/serum and parenchyma/serum ratios.
Colocalization of β-F1 ATPase and PACAP27 Binding to Brain Microvessels
Microvessels prepared from 10 male, 8-week-old CD-1 mice were resuspended in 100 µL of incubation buffer containing 1% BSA. The microvessels were divided into a control group and an experimental group of 25 µL each. PACAP27 (2.5 µg) was added to the experimental group and the volume in both tubes was increased to a total of 30 µL using the aforementioned incubation buffer. The microvessels were then incubated for 20 mins at room temperature with gentle mixing and tapping. Disuccinimidyl suberate (1.5 µL) at a concentration of 0.05 mol/L was used to crosslink PACAP27 to membrane-binding sites by incubating for 10 mins at room temperature. The crosslinked membranes were then washed in 1mL aliquots of the incubation buffer by inverting the tubes several times and centrifuging at 4000 r.p.m. for 3 mins at 4°C. The supernatant was carefully removed and the pellet washed three more times with incubation buffer. The washed and pelleted microvessels were resuspended in 100 µL of incubation buffer in preparation for confocal microscopy analysis.
The treated microvessels were resuspended and 25 µL was applied to each etched ring on glass slides (Gold Seal Products) that had been coated with poly-
Permanent Middle Cerebral Artery Occlusion Model
Peptide transport system-6 antisenses (10 µg per mouse of each of the three antisenses) or saline (200 µL) were injected via the jugular vein 24 h before pMCAO. Mice were anesthetized with 2.0% sevoflurane in N2O/O2, after which they were subjected to pMCAO by the intraluminal filament technique by use of a monofilament nylon suture as previously described (Ohtaki et al, 2006). PACAP27 (5 nmol/kg, 200 µL) or saline were injected into the jugular vein immediately after pMCAO. Four groups were studied: saline-preinjection+ saline-postinjection (saline—saline); saline—preinjection + PACAP27 postinjection (saline-PACAP27); PTS-6 antisenses-preinjection + saline-postinjection (antisense-saline); PTS-6 antisenses-preinjection + PACAP27-postinjection (PTS-6 AS-PACAP27). The neurologic score was evaluated at 2 h after pMCAO as followed by the established neurologic scoring system (Hattori et al, 2000): (0 = normal; 1 = consistent forelimb asymmetry but no apparent problem in ambulation; 2 = asymmetry in ambulation and reduced resistance to lateral push; 3 = circling movement and/or seizures; and 4 = morbidly ill and near death. At 24 h after tMCAO, the brains were removed and were sliced into four 2-mm coronal sections using a mouse brain matrix. Brain slices were then stained with 2% 2,3, 5-triphenyltetrazolium chloride (Wako, Tokyo, Japan) at 37°C for 30mins. The infarct areas were measured using NIH Image software and the infarct volumes were calculated by integration of the infarct areas.
Measurement of Cerebral Blood Flow
Cerebral blood flow was measured with a laser Doppler perfusion Imager (Peri Scan PIM2; PERIMED, Stockholm, Sweden). This noninvasive procedure provides a measurement of blood perfusion in superficial blood vessels of the brain without craniotomy. Mice were anesthetized with sodium pentobarbital sodium (60 mg/kg body weight). Additional doses were given as needed to maintain anesthesia. The skull was exposed by reflecting the scalp and was fixing it with cutting tape. The image of 30×30 mm area that covered the skull was obtained. The perfusion index values (a global indicator of blood velocity and perfusion) were expressed in units of volts, and the velocity/perfusion images were set to have a user-defined color scale ranging from 0 (showing black) to 10 (showing red) volts. Short- and long-term effects of i.v. infusions of PTS-6 antisenses on CBF were determined by measuring CBF before i.v. injection and at 10 mins and 24 h after injection. To evaluate CBF during MCAO in PTS-6 antisense and saline-treated groups, scans were performed at 24 h after injection (pre-MCAO) as baseline. The CBF was measured every 5 mins during MCAO up to 30 mins. For each evaluation, we generated a mean perfusion index for the elliptic region of interest on both cortex regions over the site supplied by the proximal segment of the MCA. Blood flow values were expressed as a percentage of the CBF baseline values.
Acquisition in the T-maze Foot Shock Avoidance
The effects on acquisition (learning) of i.v. PACAP27 and PTS-6 antisenses were tested in 12-month-old SAMP8 mice in the T-maze. The SAMP8 is a spontaneous mutant that develops age-related, amyloid-β protein-mediated cognitive impairments (Morley et al, 2002). The T-maze is a hippocampal-dependent learning task in which the animal must integrate multiple cues in a novel environment to learn a new task and the methodology previously escribed (Morley et al, 2000; Farr et al, 2000). A start box located at the bottom of the start alley is separated from the alley by a plastic guillotine door, which prevents movement down the alley until training begins. An electrifiable stainless steel rod floor runs throughout the maze to deliver a scrambled foot shock. The guillotine door is raised and a buzzer sounds simultaneously; 5 secs later foot shock is applied. The goal box that is entered on the first trial is designated ‘incorrect’ and the foot shock continued until the mouse enters the other goal box, which in all subsequent trials is designated as ‘correct’ for that particular mouse. At the end of each trial, the mouse is returned to its home cage until the next trial. The intertrial interval is 30 secs with a foot shock intensity of 0.35mA. The buzzer intensity is 55 dB. Mice were trained until they made one avoidance. Mice were given the antisenses (10 µg per mouse of each of the three antisenses in 200 µL) or saline 48 h before training by tail vein injection. They were then given PACAP27 (0.5 nmol/kg in 200 µL per mouse) or saline 24 h before training by tail vein injection.
Statistics
Means are reported with their n and standard errors. Two groups were compared by Student's t-test. More than two groups were compared by ANOVA, followed by Newman—Keuls post-test.
Footnotes
Acknowledgements
This study was supported by VA Merit Review and R01AA12743, R01NS41863, R21DA019396, and R01NS051334.