Abstract
Intracerebral hemorrhage (ICH) is the most severe form of stroke. Catheter-delivered thrombolysis with recombinant tissue-type plasminogen activator (rtPA) for the drainage of ICH is currently under evaluation in a phase III clinical trial (MISTIE III). However, in a pig model of ICH, in situ fibrinolysis with rtPA was reported to increase peri-lesional edema by promoting N-methyl-D-aspartate (NMDA)-dependent excitotoxicity. In the present study, we engineered a non-neurotoxic tPA variant, OptPA, and investigated its safety and efficacy for in situ fibrinolysis in a rat model of ICH. Magnetic resonance imaging analyses of hematoma and edema volumes, behavioral tasks and histological analyses were performed to measure the effects of treatments. In vitro, OptPA was equally fibrinolytic as rtPA without promoting NMDA-dependent neurotoxicity. In vivo, in situ fibrinolysis using OptPA reduced hematoma volume, like rtPA, but it also reduced the evolution of peri-hematomal neuronal death and subsequent edema progression. Overall, this preclinical study demonstrates beneficial effects of OptPA compared to rtPA for the drainage of ICH.
Introduction
Spontaneous intracerebral hemorrhage (ICH) is the most severe form of stroke occurring in up to five million persons every year worldwide. The evolution and the degradation of the hematoma injure the brain through several mechanisms, including a raise in intracranial pressure (ICP) and delayed neuronal death around the lesion.1,2 Subsequent peri-hematomal edema (PHE) formation worsens patients’ prognosis. 3 To mitigate this development, it has been proposed to remove coagulated intra-parenchymal blood as early as possible. However, surgical evacuation with standard craniotomy has failed to significantly improve the outcome of patients.4,5 The last published guidelines indicate that for most patients with supratentorial ICH, the efficacy of surgery remains unknown. Nevertheless, supratentorial hematoma evacuation in deteriorating patients may be considered as a life-saving measure. 6 There is thus a growing interest for less invasive approaches for patients with supratentorial ICH. One promising possibility involves inserting a small catheter into the core of the hematoma, allowing the infusion of a fibrinolytic agent to liquefy coagulated blood thereby facilitating its drainage out of the brain. This so called in situ fibrinolysis strategy has been positively evaluated in the MISTIE phase II trial, which confirmed that a brain hematoma could be removed using the recombinant form of tissue-type plasminogen activator (rtPA). 7
Despite these promising findings, there are some concerns about the safety profile of rtPA for in situ fibrinolysis, because of its pro-neurotoxic action when present at high concentrations in brain parenchyma. 8 Indeed, beyond its beneficial fibrinolytic effect, tPA also promotes N-Methyl-D-Aspartate (NMDA) receptor signalling and subsequent excitotoxic neuronal death,9,10 which leads to an increase of the PHE in animal ICH model.11,12 This phenomenon could be prevented by co-infusion of an NMDA receptor antagonist, MK-801, 13 or a delayed infusion of the type-1 plasminogen activator inhibitor (PAI-1). 14 Interestingly, we previously reported that the lysine binding site (LBS), contained in the kringle 2 domain of tPA, is required for its interaction with the NMDA receptor leading to subsequent neurotoxicity.15,16
Based on these observations, we engineered a variant of tPA devoided of neurotoxic properties, named Optimized tPA (OptPA), and set out to demonstrate its safety and its efficacy to promote the drainage of hematomas without increasing PHE in a rat model of ICH.
Materials and methods
Amino acids are numbered from the N-terminal serine of the mature Homo sapiens tPA sequence (UniProtKB: P00750).
Statement of animal well-being
Experiments were carried out in accordance with the European Communities Council Directive (86/609/EEC) regarding the care and use of animals for experimental procedures and were approved by the local ethical committee CENOMEXA (referral N° 02656.01). All efforts were made to limit animal suffering. None of the experimental procedures induced animal mortality. All experiments were performed following the ARRIVE guidelines (www.nc3rs.org.uk), including randomization of treatment as well as analysis blind to the treatment. Rats and mice were housed in a 12 h/12 h-light/night cycle with water and food ad libitum at the University Center for Biological Resources of Caen, FRANCE.
Chemicals
NMDA was obtained from Tocris (Bristol, UK); Spectrofluor 444FL was from American Diagnostica (ADF Biomedical, Neuville-suroise, France); and Dulbecco’s modified Eagle’s medium (DMEM), poly-D-lysine, cytosine β-D-arabinoside and hygromycin-B were obtained from Sigma-Aldrich (L’Isle d’Abeau, France). Lipofectamine 2000, GeneTailor Site-Directed Mutagenesis System, fetal bovine and horse sera and laminin were obtained from Life Technologies (Cergy Pontoise, France). rtPA (alteplase) was obtained from Boehringer-Ingelheim (Paris, France).
Generation and production of the tPA variants
Building and characterization of some rtPA variants are described in the literature.12,17 Mutations in the 275th position are known to reduce tPA sensitivity to plasmin without affecting its fibrinolytic activity.18–20 Substitution of tryptophan in position 253 by an arginine (W253R) is known to avoid the increase of NMDA receptor-mediated excitotoxicity by tPA. 12 Here, human wild type tPA pcDNA5/FRT vector was constructed using a PCR strategy from the pUC19 – tPA vector. By directed mutagenesis, the W253R/R275S-tPA plasmid was generated. Final constructs were systematically sequenced. HEK-FlpIn cells (Life Technologies) were grown in RPMI-1640 medium supplemented with 10% fetal bovine serum and 2 mM glutamine. At 90% confluence, cells were transfected in the presence of Lipofectamine 2000 reagent. Positive clones were isolated by hygromycin B selection. The quality of the transfection was assessed by quantitative RT-PCRq. To produce high yields of tPA variants, stable transfected HEK-293 cells were grown in a laboratory-scale bioreactor (CELLine AD 1000; Dominique Dutscher SAS, Brumath, France). rtPA variant secreted in the supernatants was purified using a nickel–nitrilotriacetic acid metal-affinity chromatography matrix (Qiagen, Courtaboeuf, France), subsequently conditioned in an NH4HCO3 0.5 M buffer and finally stored. Immunoblottings were made using a polyclonal sheep antiserum raised against human tPA (1:5000), prepared at the National institute for agronomic research (INRA, Clermont-Theix, France), followed by incubation with the appropriate peroxydase-conjugated secondary antibody. Immunoblots were revealed with an enhanced chemoluminescence ECL Plus immunoblotting detection system (Perkin Elmer-NEN, Paris, France). For in vivo studies, all the rtPA variants were conditioned in HEPES 0.3 M pH = 7.4.
Activity measurement
OptPA and rtPA were incubated in the presence of a fluorogenic substrate (5 µM) (Spectrofluor FL444). The reaction was carried out at 37℃ in 50 mM Tris (pH 8.0) containing 150 mM NaCl in a total volume of 100 µL. The amidolytic activity was measured as the change in fluorescence emission at 440 nm (excitation at 360 nm).
Euglobulin fractions, containing β-globulins and γ-globulins, were separated by isoelectric precipitation from a pool of human plasma. The euglobulin solution (100 µL) was supplemented with 20 mM calcium chloride and each of the tPA mutants at doses ranging from 30 UI/ml to 80 UI/ml. The time to clot lysis was recorded by OD measurements at 405 nm and 37℃. Tests were performed in duplicate (from three independent experiments). Results are expressed as the time needed to obtain 50% clot lysis (mean ± SD).
In vivo excitotoxic lesion
Excitotoxic lesions were induced under isoflurane-anesthesia in male Swiss mice (25–30 g, n=33). Striatal injections (coordinates: 0.5 mm posterior, + 2.0 mm lateral, −3.0 mm ventral to bregma) of NMDA (n = 10), NMDA + rtPA (n = 11) or NMDA + OptPA (n = 12), in a total volume of 1 µl (10 mM NMDA, 45 mM rtPA and/or 45 mM OptPA), were performed while the animals were positioned in a stereotaxic frame. Injections were made using adapted needles (calibrated at 15 mm/ml; assistant ref 555/5; Hoechst, Sondheim-Rhoen, Germany) Needles were kept in place until 5 min after injection. After 24 h, brains were scanned by magnetic resonance imaging (MRI, Pharmascan 7 T, Bruker, Germany). T2-weighted images were acquired using Multi-Slice Multi-Echo sequences: TE/TR 51.3 ms/1700 ms with 70 × 70 × 350 µm3 spatial resolution. Lesion sizes were quantified on these images using ImageJ (NIH software v1.45 r, National Institutes of Health, Bethesda, MD, USA).
ICH induction in rats
ICH was induced by a stereotaxic intra-striatal injection of heparin-free heterologous blood, obtained by intra-cardiac sampling from a naïve rat. Male Sprague-Dawley rats (280 g) were anesthetized with isoflurane (2%) in 30%/70% oxygen/nitrous oxide. Using a stereotaxic frame, a craniotomy was made and 100 µl of blood was injected through a 25-gauge needle over 5 min with a flow rate of 20 µl per minute (WPI micro-pump) at the following coordinates from bregma: +0.2 mm anterior, + 3.2 mm lateral, + 5.8 mm depth. The needle was then removed, the craniotomy closed with surgical wax and the scalp sutured. Animals were allowed to recover. Since animals with a small lesion volume typically do not show clear functional impairment, we excluded animals with a hematoma volume lower than 20 mm3 after the first MRI acquisition.
In situ fibrinolysis
Three hours after ICH induction, rats were anesthetized again to proceed to in situ fibrinolysis21. In the two treated-groups (rtPA and OptPA), a double catheter (2 × 25 G) was inserted using a stereotaxic frame, at the coordinates previously defined for hemorrhage induction. The fibrinolytic agent (2 µl of either rtPA or OptPA, 1 mg/ml) was then injected in a randomized and blinded manner, through the catheter, followed by the aspiration of the liquefied blood 30 min later. This injection-drainage cycle was repeated three times every 30 min. Three groups were composed as follows: no in situ fibrinolysis (hereafter named None, n = 8), in situ fibrinolysis using rtPA (n = 8) and in situ fibrinolysis using OptPA (n = 7). The needle was then removed, the craniotomy closed and the scalp sutured. The animals were then allowed to recover. Independently, nine extra rats were subjected to ICH and treated or not as detailed above (n = 3 per group) for immunohistological assessment at + 24 h.
MRI acquisitions
To evaluate the hematoma volume and the occurrence of PHE, MRI acquisitions were performed on a 7 T Pharmascan MRI system (Bruker, Germany). Rats were anesthetized with 2% isoflurane in an 80%/20% NO2/O2 gas mixture. T2-weighted images were obtained using a rapid acquisition with relaxation enhancement (RARE) sequence, with TR/TE 5200/58.2 ms and 83 × 83 × 500 µm3 spatial resolution.21,22 Hematoma (hypointense signal) and PHE (hyperintense signal) volumes were measured using ImageJ 1.45 h. MRIs were acquired 2 h after blood injection to assess the occurrence of ICH, and then repeated at + 24 h, + 7days and + 15 days. Quantitative analyses were conducted blinded to the received treatment.
Immunostainings
At + 24 h, deeply anesthetized rats (n = 3 per group) were trans-cardiacally perfused with 150 ml of cold heparinized saline followed by 250 ml of fixative (PBS 0.1 M; pH 7.4 containing 2% paraformaldehyde and 0.2% picric acid). Brains were cryoprotected (sucrose 20% in veronal buffer; 48 h; 4℃) before freezing in tissue-Tek (Miles Scientific, Naperville, IL, USA). Cryomicrotome-cut transversal sections (10 nm) were collected on poly-lysine slides and stored at −80℃ before processing for Fluoro-Jade C (FJC) staining. Brain sections were sequentially immersed in basic alcohol solution (1% NaOH in 80% ethanol) for 5 min, in 70% ethanol for 2 min, and in 0.06% potassium permanganate solution for 10 min. Slides were then transferred to 0.0001% working solution of FJC for 15 min including DAPI. After the processes of washing and drying, slides were cleared in xylene and cover-slipped with non-fluorescent mounting media. Images were digitally captured using a Leica DM6000 microscope-coupled coolsnap camera and visualized with Metavue 5.0 software (NIH).
Behavioral tasks
ICH-induced neurological deficits were assessed using a corner test, as previous described by Okauchi et al. 23 Briefly, rats were allowed to proceed into a corner for which the angle was 30°. To exit the corner, animals could turn to either the left or the right, and the direction was recorded. This task was repeated 10 times for one session at 2 days, 7 days and 15 days, and the percentage of right turns was calculated. In naïve animals, the score was 45%, while impairment led to a higher percentage of right turns. Animals with an unusual lateral preference in the corner test at baseline (≥80%) were excluded.
Statistical analyses
All statistical analyses were performed with R software, 24 using the PMCMR Package. 25 Comparisons between two groups were performed using the Mann–Whitney test. Multiple comparisons in case of independent groups were tested using the Kruskall–Wallis rank sum test followed by a Dunn’s post hoc analysis (p-values were corrected using Bonferroni’s adjustment). Multiple comparisons in case of unreplicated blocked data were tested using the Friedman rank sum test followed by a Conover’s post hoc analysis (p-values were corrected using Bonferroni’s adjustment).
Results
OptPA is a non-neurotoxic fibrinolytic agent
Human wild-type tPA (wt-tPA) and the W253R/R275S-tPA mutant (OptPA) were generated and subcloned in the pcDNA5/FRT expression vector for stable transfection and production in HEK-293 cells (Figure 1(a)). Hereafter, recombinant tPA (rtPA) means either the wt-tPA control produced in HEK cells or the recombinant wt-tPA contained in alteplase. OptPA contains two single point mutations, a substitution of arginine 275 by serine in order to prevent its processing by plasmin (sc-tPA to tc-tPA),16–26 and a second substitution of tryptophane 253 in the kringle 2 domain by arginine to prevent its interaction with NMDA receptor
16
(Figure 1(b)). As expected, purified OptPA was produced and released by HEK-293 cells as a single-chain conformation and was less sensitive to cleavage by plasmin (Figure 1(c)). As previously reported,
26
the R275S substitution also led to a reduction of the capacity of OptPA to activate plasminogen into plasmin in the absence of fibrin, i.e. out of the blood clot, with an intrinsic activity reduced by around 60% when compared to rtPA (p = 0.029) (Figure 1(d), n = 4). Then, the fibrinolytic activity of OptPA was measured in a clot lysis assay (Figure 1(e), n = 3), in which fibrin clots had been generated by adding 20 mM of calcium to a pool of human plasma depleted of α-globulins. At all the doses tested (from 30 to 80 UI/ml), OptPA displayed a fibrinolytic activity similar to rtPA. Thus, the fibrin contained in the blood clot restored the fibrinolytic capacity of OptPA to a level similar to rtPA. We then tested the capacity of OptPA to promote excitotoxicity in mice after intrastriatal injection of NMDA, either alone or combined with either rtPA or OptPA (Figure 1(f)). After 24 h in comparison to NMDA alone, NMDA + OptPA treatment did not increase the excitotoxic lesion size (p = 0.566), while NMDA + rtPA resulted in a much larger lesion (p = 0.024).
Production and characterization of OptPA (W253R/R275S-tPA). (a) Detail of the pcDNA5/FRT plasmid encoding for rtPA and W253R/R275S-tPA (OptPA). The two substitution sites (W253R and R275S) are identified by a hashtag (#). (b) OptPA is an rtPA with two-point mutations: W253R in the kringle 2 domain and R275S at the site of cleavage to produce a two-chain rtPA. (c) Immunoblot of the purified proteins expressed in HEK cell system. Both rtPA and OptPA were submitted to plasmin treatment (stochiometric ratio 1:1). Only rtPA was fully converted in its two-chain form after 2 h of plasmin incubation. OptPA was less sensitive to plasmin action. (d) OptPA had a reduced activity toward short peptides mimicking plasminogen (41% ± 14, n = 4) when compared to rtPA (108% ± 25) (Mann–Whitney test p = 0.029). (e) OptPA had a similar fibrinolytic activity when compared to rtPA (95% ± 3%, n = 3). ANOVA statistical testing showed a strong dose-response effect for both rtPA and OptPA (p < 0.0001). (f) In vivo, rtPA (p = 0.024) but not OptPA (p = 0.566) increased lesion volume induced by NMDA 24 h after injection of the drugs into the striatum of Swiss mice (mean volume lesion in mm3 ± SD: 5.03 ± 2.68 for NMDA alone, n = 11; 8.31 ± 3.17 for NMDA + rtPA, n = 10 and 5.70 ± 2.88 for OptPA, n = 12). Kruskall–Wallis rank sum test followed by a Dunn’s test, *p < 0.05; ns = non-significant.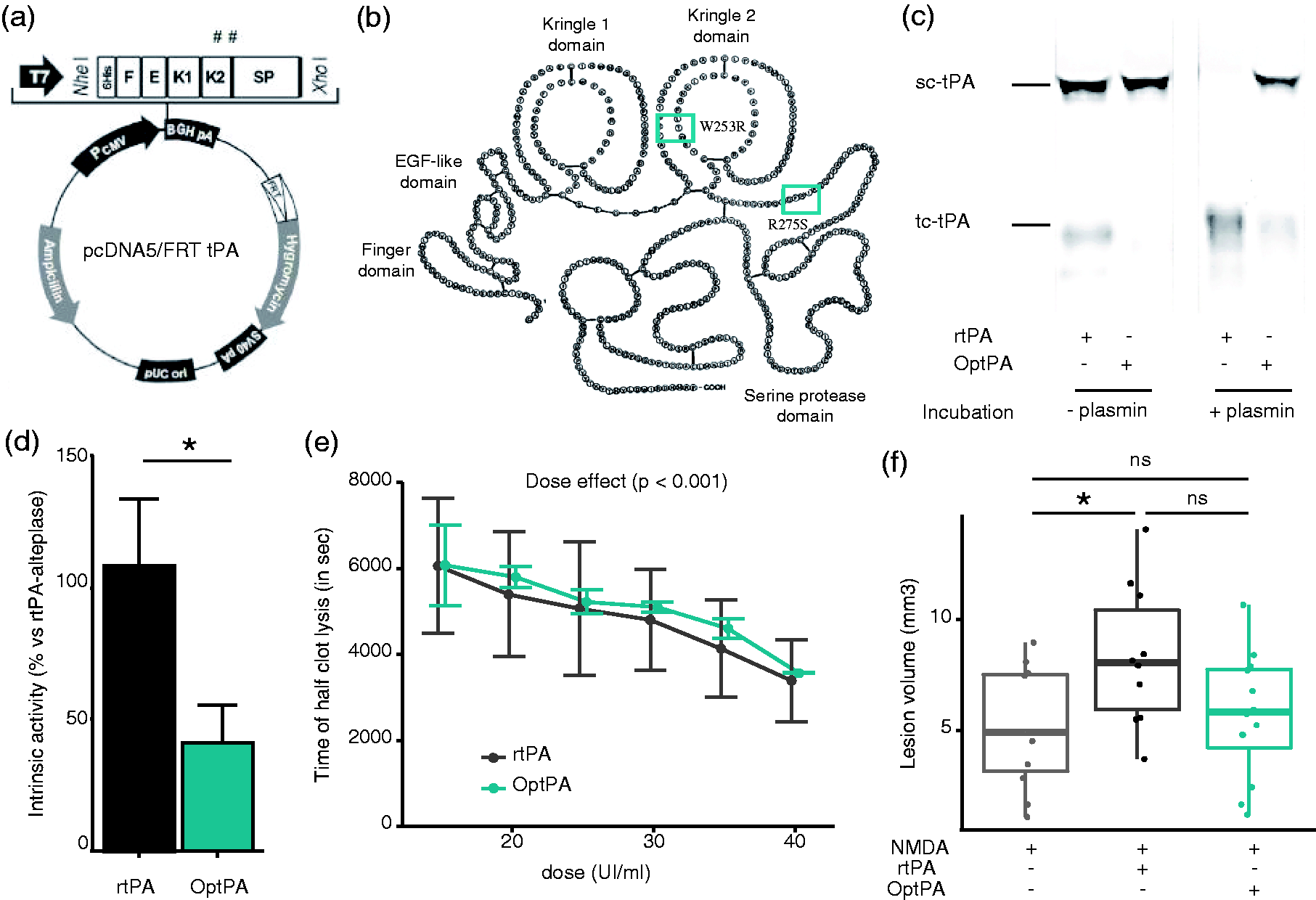
Fibrinolysis with OptPA to treat ICH reduces hematoma volume without increasing edema volume
We evaluated the safety and efficacy of in situ fibrinolysis using OptPA after heterologous blood-induced ICH in rats. Three drainages were made at + 3 h, + 3 h 30 and + 4 h as illustrated in Figure 2(a). T2-weighted MRI identified the hematoma as an area with hypointense signal and edema as hyperintense signal around the hematoma (Figure 2(b)). Two hours after blood injection, deep-seated hematomas of 34.33 ± 12.98 mm3 were observed in the striatum. In the absence of fibrinolysis, the hematoma volume spontaneously decreased after 24 h (p = 0.023) and seven days (p = 0.023) (Figure 2(c)). PHE appeared as early as 2 h after the injury, increased by 76% after 24 h (p = 0.047) and disappeared after seven days (p = 0.023) (Figure 2(d)). The efficacy of in situ fibrinolysis using either rtPA (n = 8) or OptPA (n = 7) was then evaluated in the same setting (Figure 3(a)). Both rtPA and OptPA led to clot lysis allowing the drainage of the blood out of the brain, with a significant decrease in hematoma volume at 24 h (respectively p = 0.023 and p = 0.047) (Figure 3(b)). Intergroup comparisons of the variation of the hematoma volumes between 2 h and 24 h revealed that OptPA significantly reduced the hematoma volume when compared to the non-treated group (p = 0.016) but not the rtPA treatment probably as a consequence of the fast natural resorption of the clot in the non-treated group (31% reduction of the hematoma after 24 h) combined with smaller baseline hematoma volumes in the rtPA group (34.3 µl for the non-treated group; 31.8 µl for the rtPA group and 34.9 µl for the OptPA group) (Figure 3(c)).
Characterization of a model of spontaneous intracerebral hemorrhage in rats induced by injection of heterologous blood; imaging of the lesion; and volumetric assessment. (a) Experimental design of the ICH model and treatment with either rtPA or OptPA. (b) Representative image-based lesion volumetry. White arrows indicate perilesional edema visualized by MRI. (c) Physiological reduction of the hematoma after ICH induction. The hematoma volume decreased between + 2 h and + 24 h (34.33 ± 12.98 mm3 at + 2 h vs. 23.60 ± 10.08 mm3 at + 24 h, p = 0.023) and also between + 24 h and + 7 days (18.87 ± 6.77 mm3 at + 7 days, p = 0.023). (d) Physiological evolution of the edema volume. Edema volume increased during the first 24 h after ICH induction (5.11 ± 3.0 mm3 at 2 h vs. 9.03 ± 5.70 mm3 at + 24 h, p = 0.047), and dramatically decreased between + 24 h and 7 days (1.13 ± 2.13 mm3, p = 0.023). Friedman rank sum test followed by a Conover’s test with Bonferroni correction. *p < 0.05. In situ fibrinolysis with OptPA to treat ICH in rats led to a significant reduction of the hematoma volume without increasing edema volume. (a) Lesion volumetry from MRI of ICH in rats without treatment or with in situ fibrinolysis (rtPA and OptPA) at + 2 h, + 24 h, Day 7 and Day15. (b) Intragroup evolution of the hematoma volume. The hematoma volume decreased significantly in the first 24 h following ICH induction in the three groups to reach 23.60 ± 10.08 mm3 for non-treated animals, 15.89 ± 8.99 mm3 for rtPA-treated animals and 15.55 ± 6.83 mm3 for the OptPA group. (c) Decrease of the hematoma volume was significantly higher due to the fibrinolytic action (−19.31 ± 4.69 mm3 for the OptPA-treated group vs −10.73 ± 3.71 mm3 for the non-treated group (p = 0.016) and − 15.91 ± 4.26 mm3 for the rtPA group (p = 0.156)). (d) Edema volume increased significantly between 2 h and 24 h in the non-treated condition only (+3.93 ± 3.06 mm3, p = 0.047), while there was a non-significant trend of increase in the rtPA group ((+1.52 ± 2.04 mm3, p = 0.328) and a non-significant evolution of edema in the OptPA group (−0.72 ± 1.96 mm3, p = 1.000). For all groups, edema at seven days was significantly decreased compared to 24 h. (e) Analysis of the variations in edema volumes between 2 h and 24 h showed that OptPA treatment led to a significant stabilization of the edema volume when compared to the non-treated group (p = 0.039) that was not observable in the rtPA group (p = 0.169). Friedman rank sum test followed by a Conover’s test for Figure 3(b) and (d), Kruskall–Wallis rank sum test followed by a Dunn’s test with Bonferroni correction for Figure 3(c) and (e). *p < 0.05; ns = non-significant.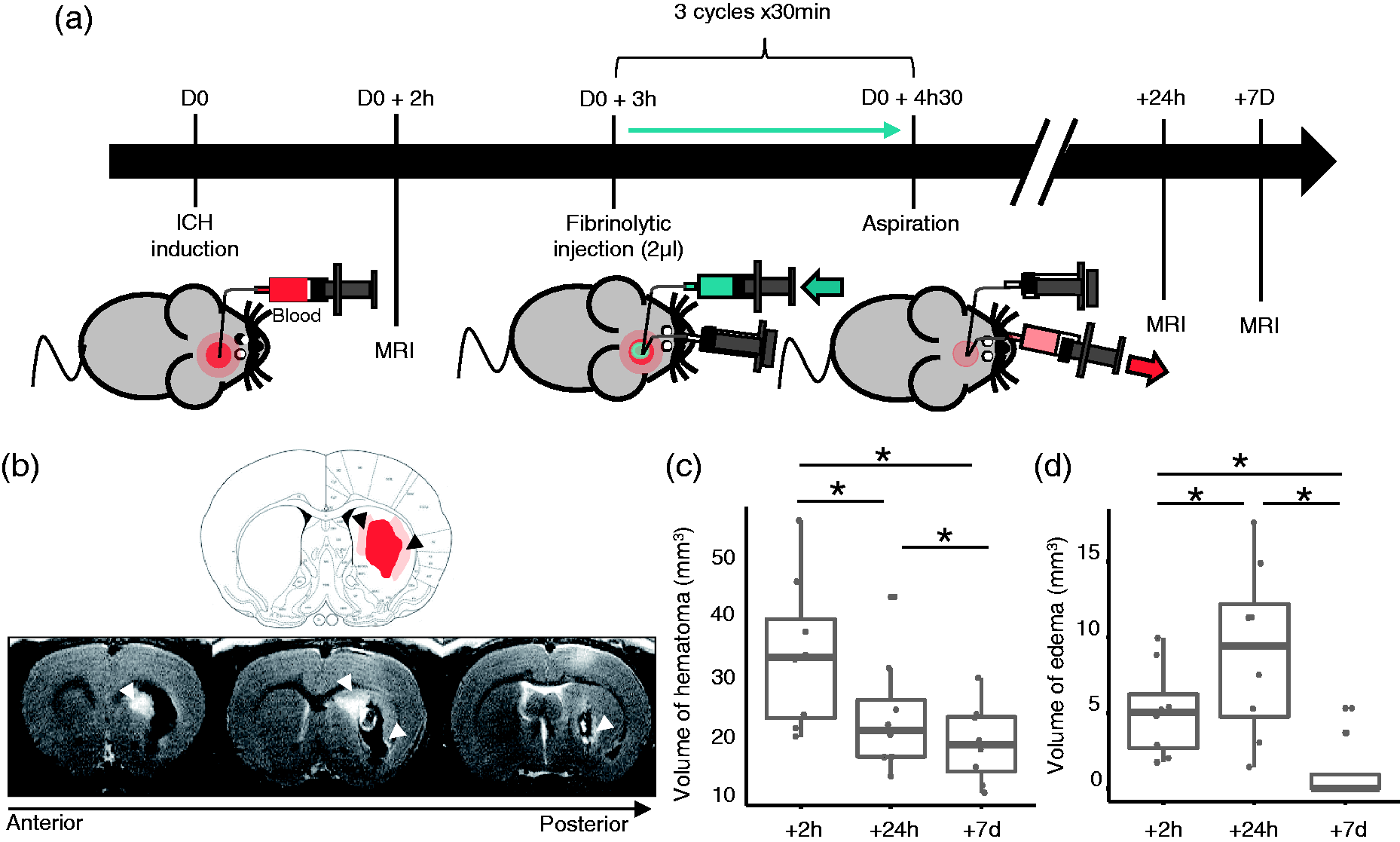
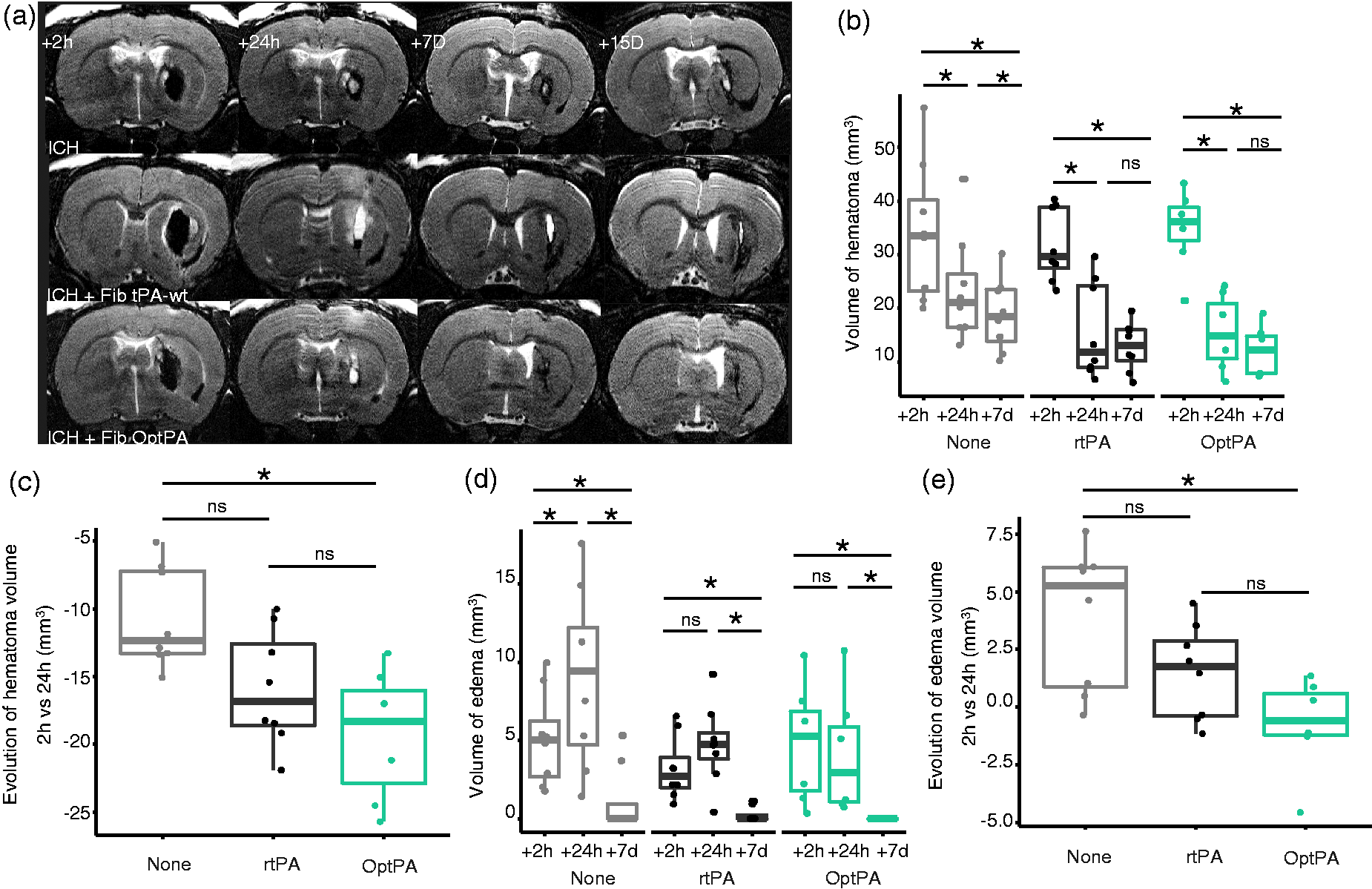
Both rtPA (p = 0.328) and OptPA (p = 1.000) treatments did not enhance edema formation during the first 24 h (Figure 3(d)). Intergroup comparisons also showed that although OptPA prevented the time-dependent increase of edema volume during the first 24 h when compared to the non-treated group (p = 0.039), rtPA did not (p = 0.311) (Figure 3(e)). This observation was confirmed by measurement of the extent of dying neurons surrounding the hematoma using Fluoro-Jade C staining (Figure 4(a)). Although, in situ fibrinolysis using rtPA led to a similar amount of dying neurons as in non-treated ICH animals (p = 0.925), OptPA-mediated in situ fibrinolysis led to a significant reduction in the number of dead neurons (p = 0.045) (Figure 4(b)).
A reduced amount of degenerating neurons was observable in edematous tissue after OptPA treatment. (a) Illustrative photomicrographs of Fluoro Jade C (FJC) on ICH brains without treatment (None) or after in situ fibrinolysis with rtPA or with OptPA. “#” indicates the hematoma, and scale bar = 100 µm. (b) Quantification of neuronal death from staining with FJC in the peri-hematoma area shows that OptPA treatment significantly reduced the number of dying neurons (428.64 ± 341.16 cells/mm2, -31.9% vs non-treated ICH animals, p = 0.005) 24 hours after ICH induction (N = 3 animals, n = 18 pictures/animal), but that the rtPA treatment did not (597.11 ± 442.29 cells/mm2 vs 631.6 ± 358.41 cells/mm2, p = 0.925). Kruskall-Wallis rank sum test followed by a Dunn’s test. *p < 0.05; **p < 0.01; ns = non-significant.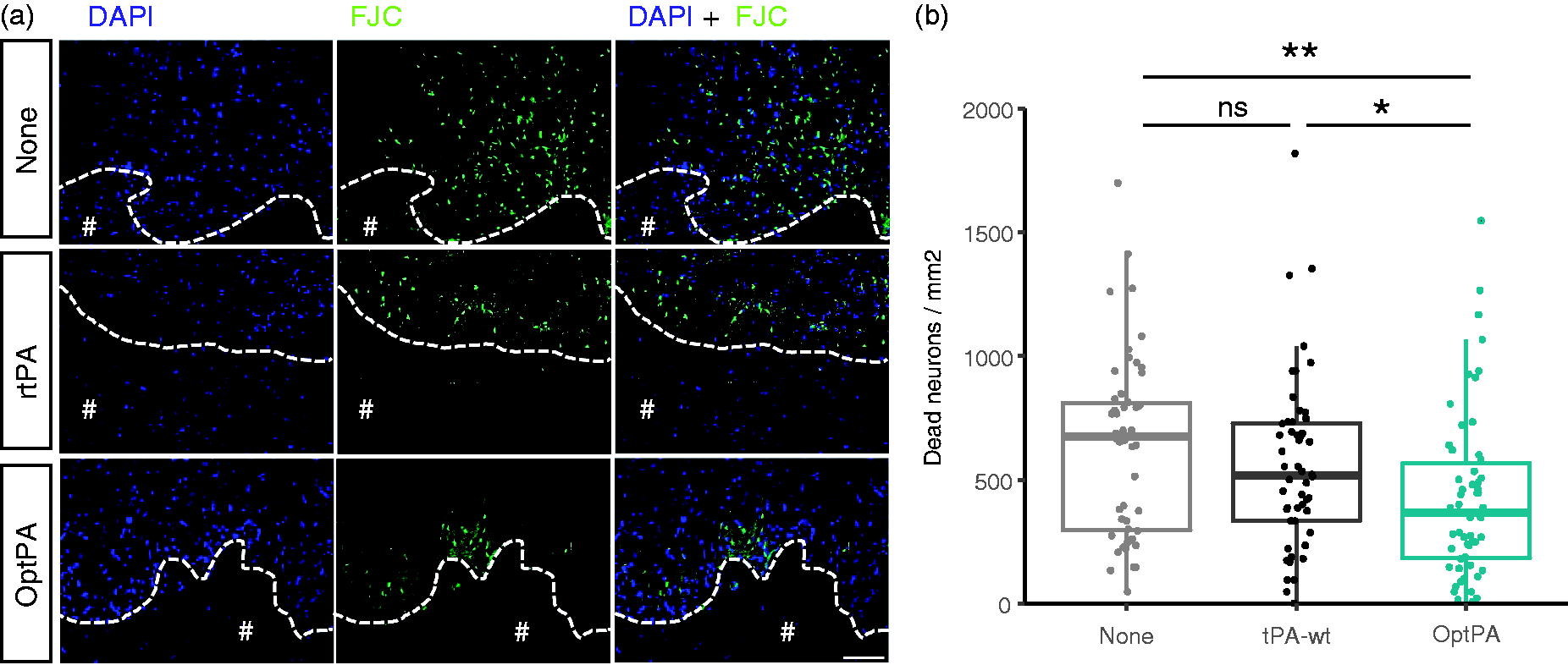
In situ fibrinolysis improves functional outcome
The corner test was executed as previously described
23
at day 2, day 7 and day 15 after injury as shown in Figure 5(a). All animals showed a strong and significant deficiency at day 2 irrespective of the treatment received (Figure 5(b)) (corner turns: 95.0 ± 5.3% for non-treated ICH group, 85.0 ± 19.3% for rtPA treatment and 87.1 ± 21.4% for OptPA treatment). In situ fibrinolysis improved functional outcome from day 7 onwards, for both rtPA and OptPA (p = 0.058 vs. baseline). Intergroup comparisons at day 7 showed that OptPA-treated animals recovered when compared to the non-treated group (p = 0.030), but rtPA-treated animals did not (p = 0.115).
In situ fibrinolysis led to significant behavioral recovery seven days after ICH induction. (a) Experimental protocol to test the effect of in situ fibrinolysis on behavioral outcome using a corner test. (b) All the animals showed a strong behavioral deficit two days after ICH induction (p < 0.001 for every groups; + 58.7 ± 9.9 right turns in non-treated animals, + 40.0 ± 20.7 right turns in rtPA-treated animals, + 37.1 ± 26.9 right turns in OptPA-treated animals). In situ fibrinolysis using both rtPA and OptPA increased the recovery of ICH rats (recovery observed from day 7; difference between baseline and day 7: + 22.5 ± 26.0 %, p = 0.058 for rtPA- and + 18.6 ± 17.7 %, p = 0.058 for OptPA-treated animals versus + 46.3 ± 10.6%\, p = 0.014 for non-treated animals). (c) Intragroup comparison showed a benefit of in situ fibrinolysis with better recovery in the OptPA group when compared with the rtPA group (+46.2 ± 10.6 right turns for the non-treated group between Baseline and 7 days (n = 8) vs. + 22.5 ± 26 for the rtPA group (n = 8, p = 0.11) and + 18.6 ± 17.7 for the OptPA group (n = 7, p = 0.03 vs. non-treated animals). Friedman rank sum test followed by a Conover’s test and a Bonferroni correction for Figure 5(b) and Kruskall–Wallis rank sum test followed by a Dunn’s test with a Bonferroni correction for Figure 5(c). *p < 0.05; ***p < 0.001; ns = non-significant.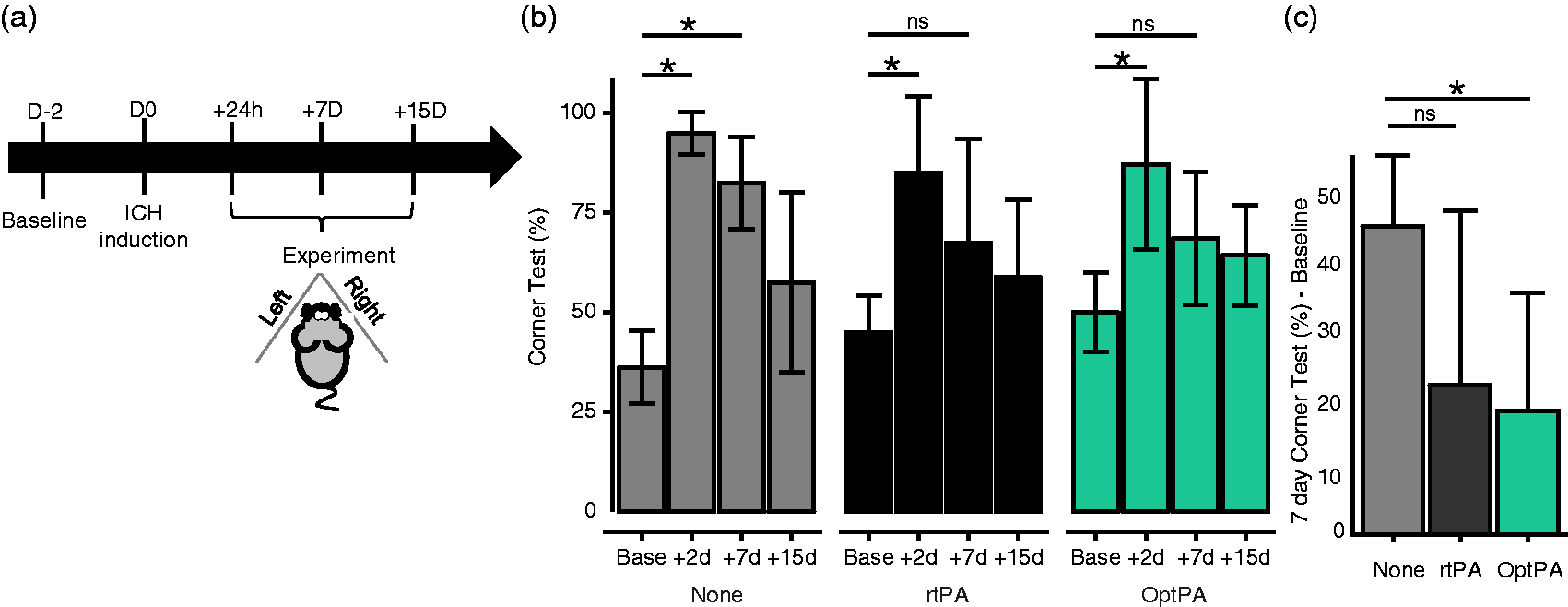
Discussion
ICH not only causes primary brain injury through direct mechanical effects of the hemorrhage (deleterious mass-effect 27 ), but also leads to secondary brain injury through processes that evolve over hours to days and which may, therefore, be preventable. 2
During the first hours, excitatory amino acids (AA), among them glutamate, are secreted and osmotic forces take place locally due to swelling of brain cells in response to ICH13,28–30. Then, a second stage is characterized by the initiation of multiple inflammatory processes31,32 (complement initiation, metalloproteinase (MMP) maturation, cell recruitment) and by the release of clot components. Mainly, both the “conductor” thrombin in the early hours 13 and iron released from haemoglobin degradation during the subsequent days 33 cause secondary injury and peri-hematomal tissue damage, including breakdown of the blood–brain barrier (BBB). A growing body of evidence suggests that such secondary processes contribute to neurological deterioration after ICH.34–36 Consequently, PHE rises as a valuable marker of secondary injury that could provide a useful surrogate end-point for experimental and clinical studies of novel therapeutic agents to prevent secondary injury following ICH. The question of whether PHE adversely influences neurological outcome has become more relevant as new molecular targets and novel therapies have emerged. However, studies that investigated whether PHE is an independent predictor of neurological outcome have produced rich debates.1,37–39 Urday et al. 40 carefully reviewed the last trials and publications. Their conclusions were that PHE has clinical relevance both as a therapeutic target, owing to its augmentation of the mass effect of a hemorrhage, and as a surrogate marker for novel interventions that target secondary injury.
The aim of our current study was to identify a drug that could be most adapted for in situ fibrinolysis after cerebral hemorrhage, which includes both intracerebral and intraventricular hemorrhage indications. The last results of the clinical trials MISTIE (phase II) and CLEAR (phase III) show that in situ fibrinolysis associated with drainage of the liquefied blood out of the brain is clearly a good strategy for clinical improvement of ICH patients.3,41–42 Nevertheless, although beneficial (i.e. the fibrinolytic effect), this effect of rtPA is counterbalanced by side effects such as bleedings and PHE growth. The results of CLEAR III show that in situ fibrinolysis with rtPA decreases the risk of death but fails to improve functional outcome in IVH. 42 Thus, here our objective was to find a drug displaying beneficial effects in draining the hematoma deep-seated in the brain, without triggering neurotoxicity and PHE growth. The treatment with OptPA was overall beneficial when compared to the non-treated group, whereas treatment with rtPA (our recombinant form or alteplase) was not.
Two major ICH models exist for studies in rodents: the injection of collagenase degrading the collagen of the vascular walls, or a direct injection of autologous or heterologous blood within the cerebral parenchyma. 43 The collagenase-induced ICH model has the benefit of having a real vascular rupture, but it also destructs cerebral tissues, including vessel walls, phenomena that are not compatible with in situ fibrinolysis without continuous secondary bleeding. Thus, we decided here to use a heterologous blood injection model to trigger ICH. In this study, rtPA and OptPA were used in the same concentration as used in the MISTIE trial. Three different dosages were firstly considered in the MisTIE trial, 0.3 mg, 1.0 mg and 3.0 mg per injection. Finally, only the 1.0 mg was selected as the best dose to achieve a therapeutic effect with high safety. In our present study, we decided to also use 1.0 mg/ml of OptPA, which was enough to trigger in situ fibrinolysis as confirmed in the in vitro assays (Figure 1; 2 µl was injected three times, i.e. a total dose of 6 µg was injected in a 30 -µl hematoma). Further studies would help to determine the maximum tolerated dose for OptPA to achieve the best therapeutic effect with a high safety.
Several limitations may be raised about this study. First, the sample size was not fully adequate for extensive behavioral analyses. Further studies in rodents (specifically designed to show behavioral improvement with acceptable statistical power) and in large animals will help to confirm the beneficial effect of OptPA in behavioral tasks. Another limitation of our study is possibly the lack of sham group; a choice we did to fit with the design of a clinical trial, such it was done for the MISTIE trial, 7 with the objective to compare two treatments, OptPA in one hand and rtPA in another hand. Finally, our study focused on striatal hematomas, additional studies should be performed in order to confirm previous observations in intraventricular hemorrhage (IVH), 22 and to prove that the combination of OptPA plus minimally invasive surgery can also be used for deep-seated hematomas which are inaccessible using interventional strategies.
Footnotes
Funding
This work was supported by grants from the INSERM (French National Institute for Health and Medical Research), the TC2N (Trans Channel Neuroscience Network), the FP7 Eranet-NEURON 3-275-006, and the Regional Council of Lower Normandy.
Acknowledgements
We thank Prof HR Lijnen for kindly giving us the pUC19 – tPA vector, and Prof R Dijkhuizen for giving us some advices.
Declaration of conflicting interests
DV and JP are co-authors of a patent protecting OptPA licensed to Op2Lysis.
Authors’ contributions
RG, JP, DV and TG designed the experiments. RG and JP performed the experiments. RG, JP and MN analyzed the data. RG, TG, MN, DV, and JP wrote the manuscript.
