Abstract
Glutamate is accumulated in abundance during the early period of experimental hematoma, and the activation of N-methyl-D-aspartate (NMDA) receptors by glutamate can result in an influx of calcium and neuronal death in cases of intracerebral hemorrhage (ICH). Memantine, which is known to be a moderate-affinity, uncompetitive, NMDA receptor antagonist, was investigated with regard to its ability to block the glutamate overstimulation and tissue plasminogen activator (tPA)/urokinase plasminogen activator (uPA)/matrix metalloproteinase (MMP)-9 modulation in experimental ICH. Intracerebral hemorrhage was induced via the infusion of collagenase into the left basal ganglia of adult rats. Either memantine (20 mg/kg/day) or PBS was intraperitoneally administered 30 min after the induction of ICH, and, at daily intervals afterwards, for either 3 or 14 days. Hemorrhage volume decreased by 47% in the memantine group, as compared with the ICH-only group. In the memantine group, the numbers of TUNEL+, myeloperoxidase (MPO)+, and OX42+ cells decreased in the periphery of the hematoma. Memantine resulted in an upregulation of bcl-2 expression and an inhibition of caspase-3 activation. Memantine also exerted a profound inhibitory effect on the upregulation of tPA/uPA mRNA, and finally decreased the MMP-9 level in the hemorrhagic brain. In modified limbplacing test, the memantine-treated rats exhibited lower scores initially, and recovered more quickly and thoroughly throughout the 35 days of the study. Here, we show that memantine causes a reduction of hematoma expansion, coupled with an inhibitory effect on the tPA/uPA and MMP-9 level. Subsequently, memantine was found to reduce inflammatory infiltration and apoptosis, and was also determined to induce functional recovery after ICH.
Introduction
The early period of intracerebral hemorrhage (ICH) is characterized by perihematomal glutamate accumulation (Qureshi et al, 2003). The glutamatemediated overstimulation of N-methyl-
Neuroprotective agents that work by high-affinity binding to NMDAR, most notably MK-801 (Parsons et al, 1999), result in the virtually complete blockage of receptor activity; thus, these drugs tend to cause unacceptable clinical side effects, including hallucinations, drowsiness, and coma (Lipton and Chen, 2004). Memantine is a specific, moderateaffinity, uncompetitive, open-channel NMDAR antagonist, with strong voltage dependency and rapid blocking/unblocking kinetics (Parsons et al, 1999; Möbius, 2003). Memantine reduces overstimulation of the NMDAR induced by abnormally high glutamate concentrations, thereby preventing neuronal calcium overload, and restoring normal receptor-signaling functions (Wilcock, 2003). Clinical and preclinical data indicate that memantine exerts neuroprotective effects in a variety of neurological disorders, including Alzheimer's disease, vascular dementia, and cerebral ischemia (Block and Schwarz, 1996; Wilcock, 2003; Culmsee et al, 2004).
Tissue plasminogen activator (tPA), a serine protease with known fibrinolytic activity, is another important molecule with regard to glutamate toxicity. It enhances N-methyl-
In this study, we attempted to determine whether the inhibition of NMDAR by memantine may exert antiapoptotic and antiinflammatory effects during ICH. Given the relation between NMDAR and tPA, we also attempted to assess endogenous tPA/uPA expressions and matrix metalloproteinase (MMP)-2/9 levels, as well as hematoma expansion.
Materials and methods
Induction of Intracerebral Hemorrhage and Memantine Treatment
All of the procedures were performed after receiving institutional approval, in accordance with the NIH Guide for the Care and Use of Laboratory Animals. One hundred and ten male Sprague–Dawley rats (Daehan Bio, Seoul, South Korea), each weighing 200 to 220 g, were used in the following experiments. The rats were separated into two groups: the ICH-only group (n = 55) and the memantine-treated ICH group (n = 55). Experimental ICH was induced via stereotaxic intrastriatal administration of type IV bacterial collagenase (Sigma), as described in other studies (Rosenberg et al, 1990; Jeong et al, 2003; Chu et al, 2004; Jung et al, 2004; Park et al, 2005). In brief, after intraperitoneal injections of 1% ketamine (30 mg/kg; Sigma, St Louis, MO, USA) and xylene hydrochloride (4 mg/kg; Sigma, St Louis, MO, USA), the rats were placed in a stereotaxic frame (David Kopf instruments, Tujunga, CA, USA), and burr holes were made. A 30-gauge Hamilton syringe needle was then inserted into the striatum. Intracerebral hemorrhage was induced via the administration of 1 µL of solution containing 0.23 CDU of type IV collagenase (Sigma) over a period of 5 min. After the completion of the collagenase infusion, the craniotomies were sealed with bone wax, and the rats were allowed to recover.
Memantine (20 mg/kg/day; Lundbeck), dissolved in 2 mL of 0.9% saline, was administered intraperitoneally, with the first treatment given 30 min after the induction of ICH, and then once daily afterward for 3 days. Saline was also administered to the ICH-only group during the same period. Physiological parameters, including mean arterial blood pressure, blood gases, and glucose concentration, were also measured throughout the experiment.
Behavioral Testing
Behavioral testing (n = 15 for each group) was conducted weekly for a period of four weeks, using modified limbplacing tests (MLPT), which were monitored by two individuals who had been masked to the treatment status of the rats (Jeong et al, 2003; Chu et al, 2004; Jung et al, 2004; Park et al, 2005). This test consists of two limbplacing tasks, which assess the sensorimotor integration of the forelimb and the hindlimb, by monitoring the subject's responses to tactile and proprioceptive stimulation. First, the rat is suspended 10 cm above a table, and the stretch of the forelimbs towards the table is observed and evaluated: a normal stretch is scored as 0 points; abnormal flexion is scored as 1 point. Next, the rat is positioned along the edge of the table, with its forelimbs suspended over the edge, and is then allowed to move freely. Each forelimb (forelimb – second task, hindlimb – third task) is gently pulled down, and retrieval and placement is evaluated. Finally, the rat is placed near the table edge, to assess the lateral placement of the forelimb. The three tasks are scored in the following manner: normal performance is scored as 0 points; delayed (at least 2 sees) and/or incomplete performance is scored as 1 point; no performance is scored as 2 points. A total score of 7 points indicates maximal neurological deficit, and a score of 0 points denotes normal performance.
Spectrophotometric Assay of Hemorrhage Volume
At 3 days after the induction of ICH, hemorrhage volume was quantified with a previously described spectrophotometric assay (Choudhri et al, 1997; Asahi et al, 2000b; Sumii and Lo, 2002; Park et al, 2005). Hemispheric brain tissue was acquired from normal rats which had been subjected to complete transcardial perfusion for the removal of intravascular blood. Incremental volumes of homologous blood (0 to 200 µL) were added to each hemispheric sample, along with PBS, to a total volume of 3 mL, followed by 30 sees of homogenization, 1 min of sonication on ice, and 30 min of centrifugation at 12,000 r.p.m. Drabkin's reagent (1.6 mL; Sigma) was then added to 0.4-mL aliquots, and allowed to stand for 15 min at room temperature. Optical density (OD) was then measured and recorded at 540 nm with a spectrophotometer (Molecular Devices). These procedures showed a linear relationship between the hemoglobin concentrations in the perfused rat brain and the volume of added blood. Measurements from the perfused ICH brains (n = 6 in each group) were then compared with this standard curve, allowing us to obtain data regarding hemorrhage volume (in µL).
Measurements of Brain Water Content
At 3 days after the operation, the rats were anesthetized and killed via decapitation (n = 12 in each group). The rats' brains were removed immediately, and divided into two hemispheres along the midline, after which the cerebellum was removed from each brain. The brain samples were then immediately weighed on an electronic analytical balance to obtain wet weights, and then dried in a gravity oven at 100°C for 24 h, to obtain dry weights. Water contents were expressed as a percentage of wet weight: the formula used to calculate the water contents was as follows: (wet weight—dry weight)/(wet weight) (Chu et al, 2004; Jung et al, 2004; Culmsee et al, 2004).
Tissue Preparation and Immunohistochemistry
On day 3, the rats (n = 6 for each group) were reanesthetized, and perfused through the heart with 50 ml cold saline and 50 ml of 4% paraformaldehyde, in 0.1 mol/L of phosphate-buffered saline. After 24 h of fixation in 4% paraformaldehyde, the rats' brains were cryoprotected with 30% sucrose for 24 h, then sliced into 30-µm sections with a cryostat (Leica CM 1900), as has been described elsewhere (Jeong et al, 2003; Chu et al, 2004; Jung et al, 2004). We then conducted immunostaining with antibodies specific for myeloperoxidase (MPO; 1:200, DAKO) and Ox-42 (1:500, Chemicon). We then performed the TUNEL procedure for the in situ detection of deoxyribonucleic acid (DNA) fragmentation, as has been described elsewhere (Gavrieli et al, 1992; Matsushita et al, 2000; Chu et al, 2004; Jung et al, 2004). Coronal sections cut through the center of the hemorrhagic lesion were analyzed by counting marker-specific cells throughout the whole section (3 sections per each antibody staining, 1 mm width). Total counts in these sections were then converted to cell densities, to perform quantitation and comparison between the groups.
Western Blotting Analysis
At 1 day after the induction of ICH, the rats were killed via decapitation, and the brain was immediately extracted (n = 4 in each group). The homogenates from the ipsilateral hemisphere were centrifuged for 10 min at 13,000 r.p.m. and 50 µg of the obtained proteins was separated on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto nitrocellulose membranes. These membranes were incubated in blocking buffer (5% skim milk in Tris-buffered saline containing Tween 20 (TBST) (50 mmol/L Tris, pH 7.5, 0.15 mmol/L NaCl, 0.05% Tween 20)), and incubated again with anti-Bax (1:200; Santa Cruz), and anti-Bcl-2 (1:200; Santa Cruz), anti-Bcl-xl (1:200; Santa Cruz) in TBST overnight at 4°C. The incubated membranes were then treated with secondary antibody conjugated with horseradish peroxidase (HRP) in TBST for 1 h at room temperature. Anti-β-actin antibody (Santa Cruz, USA) was used as a control. The blots were developed via enhanced chemiluminescence (Pierce, Rockford, IL, USA), digitally scanned (GS-700, Bio-Rad, Hercules, CA, USA) and analyzed (Molecular Analyst®, Bio-Rad). Relative optical densities were obtained by comparison of the measured values with the mean values from the ICH-only group.
Reverse transcriptase-polymerase chain reaction (RT-PCR)
At 1 day after the induction of ICH, RT-PCR was performed for tPA, uPA, and PAI-1, using a slightly modified version of a previously described method (Asahi et al, 2005). Total RNA was then isolated from the homogenate (50 mg) of the hemorrhagic hemisphere with TRI reagent (Sigma), and RT-PCR was conducted using the First strand cDNA Synthesis kit for RT-PCR (Roche) (n = 4 in each group). The following primer sets were utilized to amplify tPA, uPA, and PAI-1 (25 cycles of 95°C, 58°C, and 72°C for 40 sees each); tPA: 5'-CAC AGC TTT ACC ACA TCC-3’ (sense) and 5'-CAG ATA GCA CCC AGC AGG AA-3’ (antisense); uPA: 5'-CTTCAGCGACGAAACTCTGG-3’ (sense) and 5'-TCTGGGTCAGCAGCACACA-3’ (antisense); PAI-1: 5'-ATCAACGACTGGGTGGAGAG-3’ (sense) and 5'-AGCCTGGTCATGTTGCTCTT-3’ (antisense). The products of this PCR (10 µ***I) were then subjected to electrophoresis on 1% agarose gel containing 0.03 µg/ml ethidium bromide, and we quantified the transcript levels via analysis of scanned photographs of gels, using the appropriate imaging software (Molecular Analyst®, Bio-Rad). mRNA expression levels were normalized to GAPDH, and relative optical densities were obtained by comparison of the measured values with the mean values of the ICH-only group.
Measurement of Caspase-3 Activity
Caspase-3 activity was evaluated using the ApoAlert kit (Clontech) on day 1, as was described previously (Lee and Lo, 2003). Lysed brains (n = 4 for each group) were incubated for 1 h in reaction buffer containing 10 mmol/L dithiothreitol at 37°C. Fluorescent intensities were measured with a plate reader (FL600, Bio-Tek, excitation 380 nm, emission 460 nm).
Gel Zymography of Matrix Metalloproteinases
Gel zymography was performed on day 1, as described previously (Asahi et al, 2000a). Similarly prepared protein samples (as in western blot analysis; n = 4 in each group) were loaded and separated on 10% Tris-glycine gel, using 0.1% gelatin as substrate. After electrophoretic separation, the gels were incubated in renaturing buffer (2.5% Triton X-100 in distilled water) at room temperature for 30 min, with gentle agitation. The renaturing buffer was then decanted and replaced with developing buffer (50 mmol/L Tris Base, 40 mmol/L Tris-HC1, 200 mmol/L NaCl, 5 mmol/L CaCl2, 0.2% Brij 35). After 30 min of equilibration with the developing buffer, the gels were incubated with fresh developing buffer for 18 h at 37°C. After developing, the gels were stained with 0.5% Coomassie Blue R-250 for 30 min, then appropriately destained. Positive control standards for MMP-2 and MMP-9 were obtained from human fibrosarcoma cells (HT1080), which secretes pro-MMP-9 (92 kDa), pro-MMP-2 (66 kDa), and active MMP-2 (62 kDa) (Maquoi et al, 2002). Matrix metalloproteinases were quantified as fold increase compared with ICH-only, as measured by OD.
Statistical Analysis
All data in this study are presented as means ± standard deviations. The obtained data were analyzed by repeated measures of analysis of variance, and unpaired Student's t-tests, if the data were distributed normally (Kolmogorov–Smirov test, P 0.05). In other cases, we used Mann–Whitney U-tests. In all cases, we specified which test was used. A two-tailed probability value of < 0.05 was considered to be significant.
Results
Physiological Parameters
All animals survived the surgery. The physiological parameters, including mean arterial blood pressure, blood gases, serum glucose, and body temperatures, were not significantly different in any experimental groups before, during, or after ICH.
Memantine Treatment Induced Functional Recovery
Although both the ICH-only and the ICH-memantine groups exhibited more than five points on MLPT on the first day after the induction of ICH, the ICH-memantine group exhibited a less profound degree of deficit than did the ICH-only group (P < 0.05, Student's t-test; Figure 1A). The ICH-memantine group continued to recover, with the difference between the two groups being statistically significant until at least 4 weeks after the induction of ICH (P < 0.01, t-test). At week 4, the memantine-treated group scored less than one point on the MLPT.
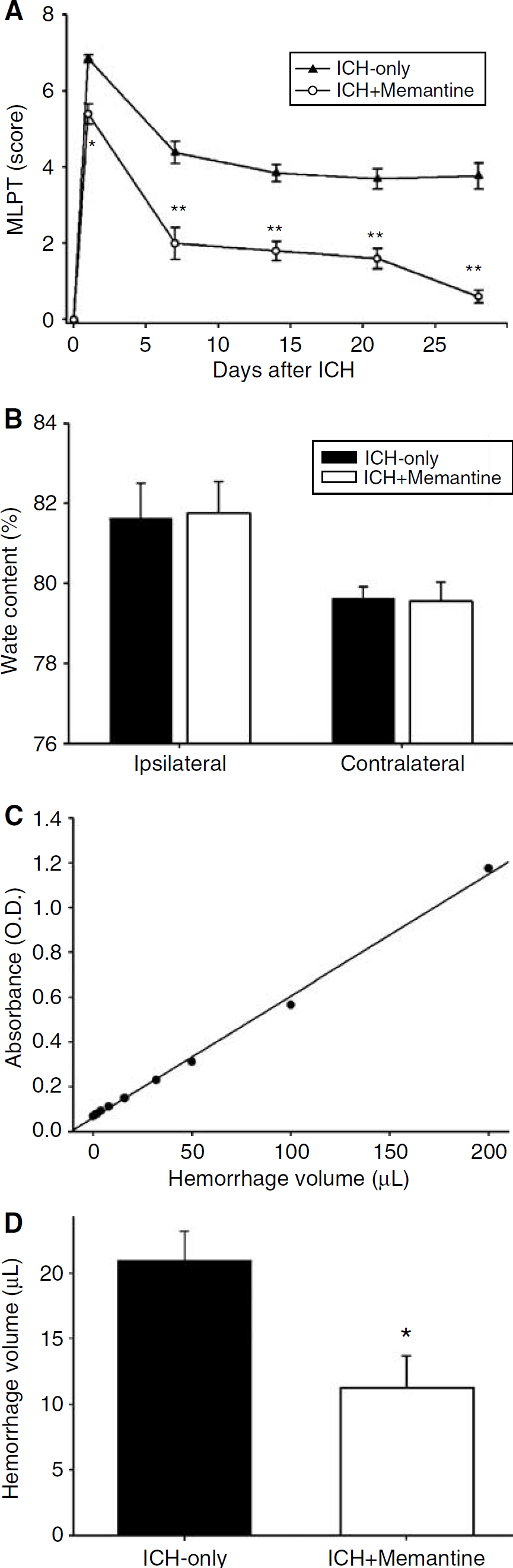
Functional recovery and hematoma volume. The memantine-treated rats exhibited less initial neurological deficits and better functional recovery than did the rats in the ICH-only group (
Memantine Treatment Suppressed Hematoma Expansion
In the ICH-only group, the brain water content of the lesioned (left) hemisphere was found to be 81.77% ± 0.78%, and in the ICH-memantine group brain water content was measured to be 81.98% ± 0.50% (P = 0.62, t-test). The brain water content in the nonlesioned (right) hemispheres of the ICH-only group was determined to be 79.58% ± 0.46%, and 79.65% ± 0.24% in the ICH-memantine group (P = 0.82, t-test). Brain water content was not significantly different between the two groups (Figure 1B).
However, memantine resulted in a 47% reduction in hemorrhage volume as compared with that of the ICH-only group, 3 days after the induction of ICH. The hemorrhage volumes were 21.0 ± 4.5 µL in the ICH-only group, and 11.2 ± 5.0 µL in the ICH-memantine group (Figure 1C and 1D; P = 0.028, t-test).
Memantine Decreased Apoptosis and Inflammatory Infiltration
TUNEL+ cells were detected within the hemorrhage itself, and were also detected in the periphery of the hemorrhage (Figure 2A). In quantitative analysis, the ICH-memantine group evidenced a reduced number of TUNEL+ cells (31 ± 6 cells/mm2) as compared with the ICH-only group (337 ± 25 cells/mm2; P < 0.01, t-test; Figure 2B). The ICH-memantine group also evidenced a reduced number of OX42+ microglial cells (ICH-memantine: 69 ± 5 cells/mm2; ICH-only: 89 ± 10 cells/mm2; P < 0.01, t-test) and MPO+ neutrophils (ICH-memantine: 129 ± 21 cells/mm2; ICH-only: 224 ± 16 cells/mm2; P < 0.01, t-test), as compared with the ICH-only group, 3 days after the induction of ICH (Figure 2B).
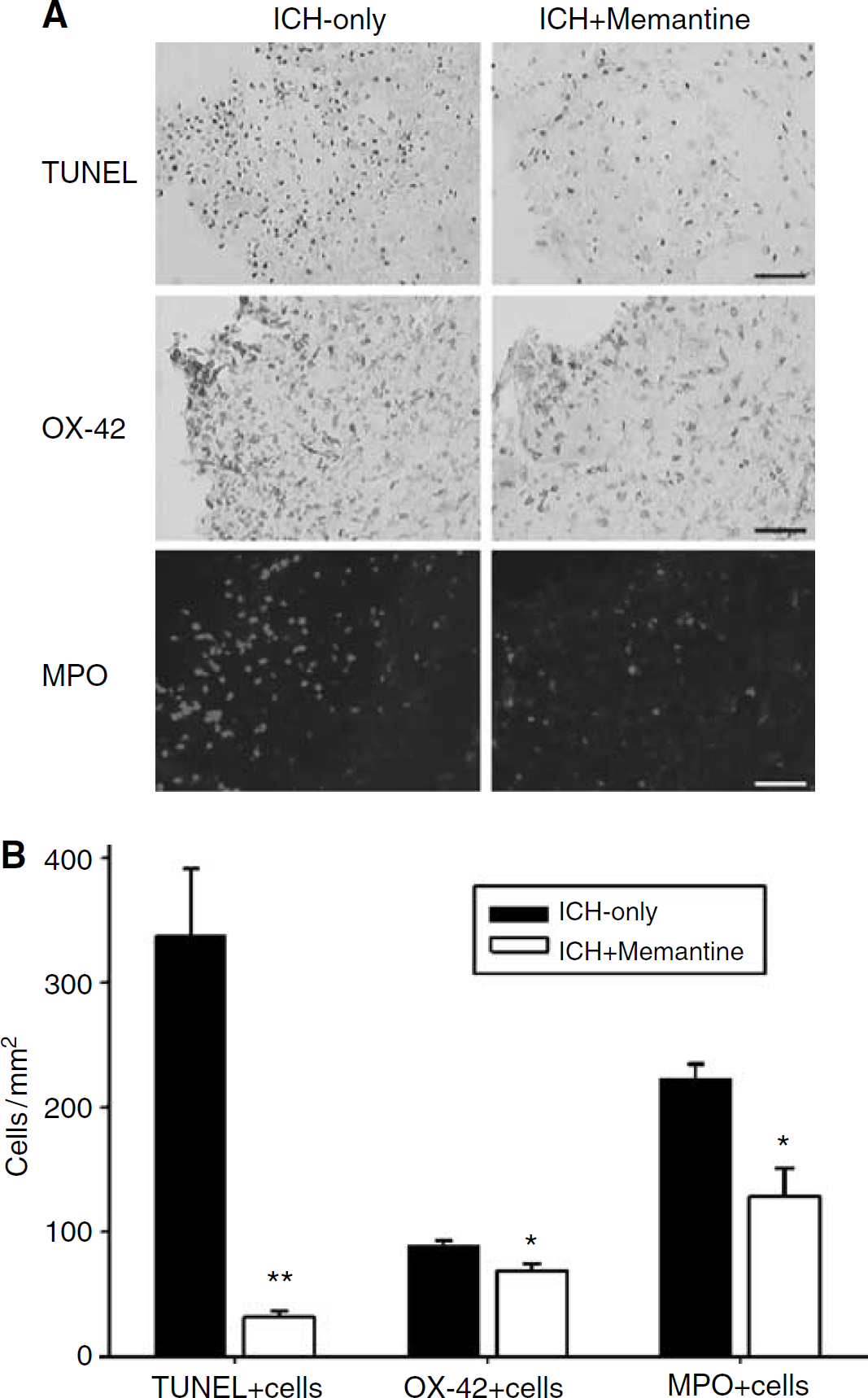
Histologic analysis of apoptosis and inflammation. Memantine caused a decrease in the number of TUNEL+, OX42+, and MPO+ cells in the perihematomal regions (
Memantine Enhanced Bcl-2 Expression and Reduced Caspase-3 Activation
Western blotting for Bax, Bcl-2, and Bcl-xl revealed that the memantine treatment had resulted in an upregulation of Bcl-2 expression (Figure 3A). The relative OD of Bcl-2 expression was 5.5 (± 1.0) times as high in the ICH-memantine group as in the ICH-only group (P < 0.01, t-test; Figure 3B). Meanwhile, Bax and Bcl-xl expression remained unchanged in both groups.
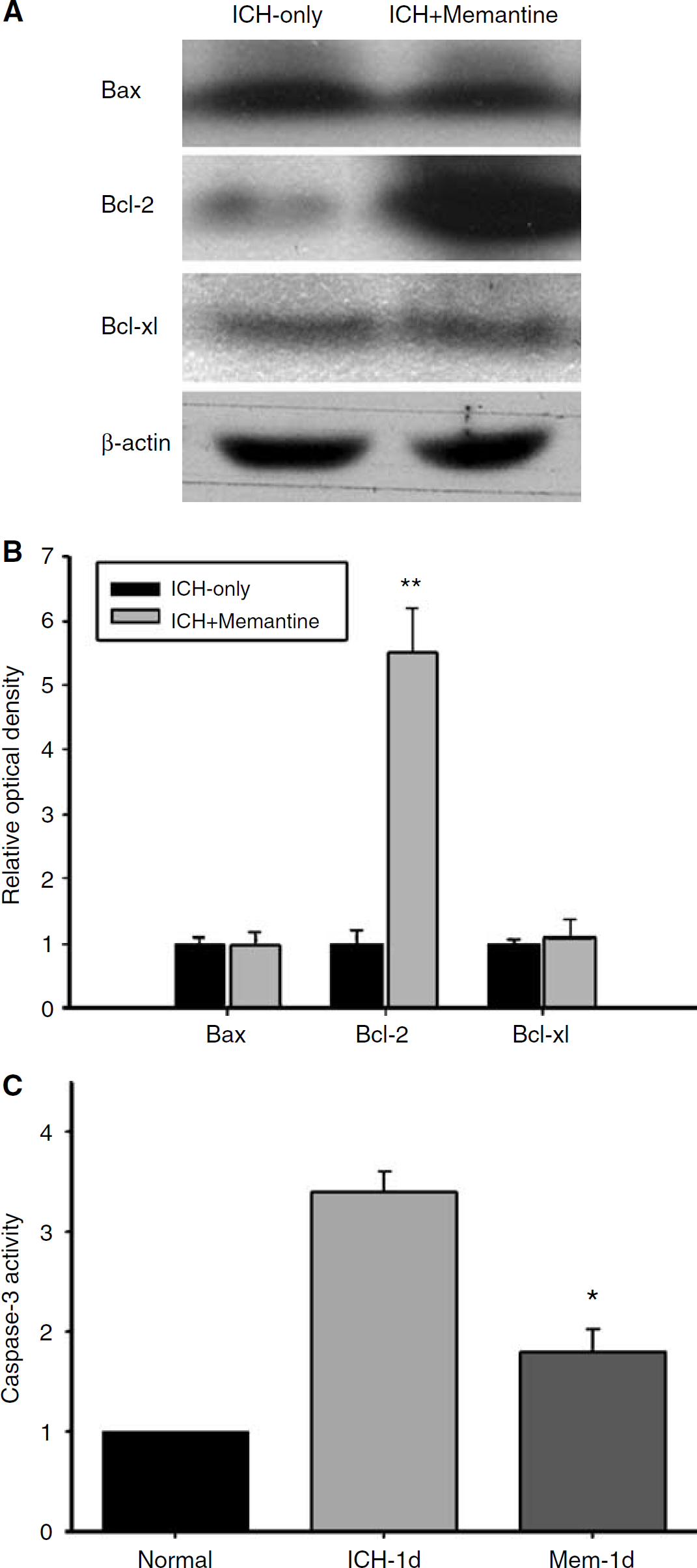
Western blotting for a poptosis-related molecules. We documented an upregulation in Bcl-2 expression in the ICH-memantine group (
Caspase-3 activity was reduced in the ICH-memantine group. At 1 day after the induction of ICH, the caspase-3 activity in the ICH-memantine group was significantly lower than that of the ICH-only group (47% decrease; P < 0.01, t-test; Figure 3C).
Memantine Reduced Tissue Plasminogen Activator/uPA Expression and Matrix Metalloproteinase-9 Level after Intracerebral Hemorrhage
The RT-PCR analysis of endogenous proteolytic enzyme expression indicated that memantine resulted in a reduction of tPA, uPA, and PAI-1 expression in the hemorrhagic rat brains. Hemorrhagic brains expressed the mRNA of these molecules fairly abundantly, whereas the normal brains seldom expressed them (Figure 4A). Memantine exerted a profoundly inhibitory effect on the ICH-associated upregulation of tPA and uPA. The memantine group exhibited a 90% decrease in tPA mRNA, an 87% decrease in uPA mRNA, and a 73% reduction in the levels of PAI-1 mRNA, as compared with the ICH-only group (P < 0.01, t-test; Figures 4A and 4B).
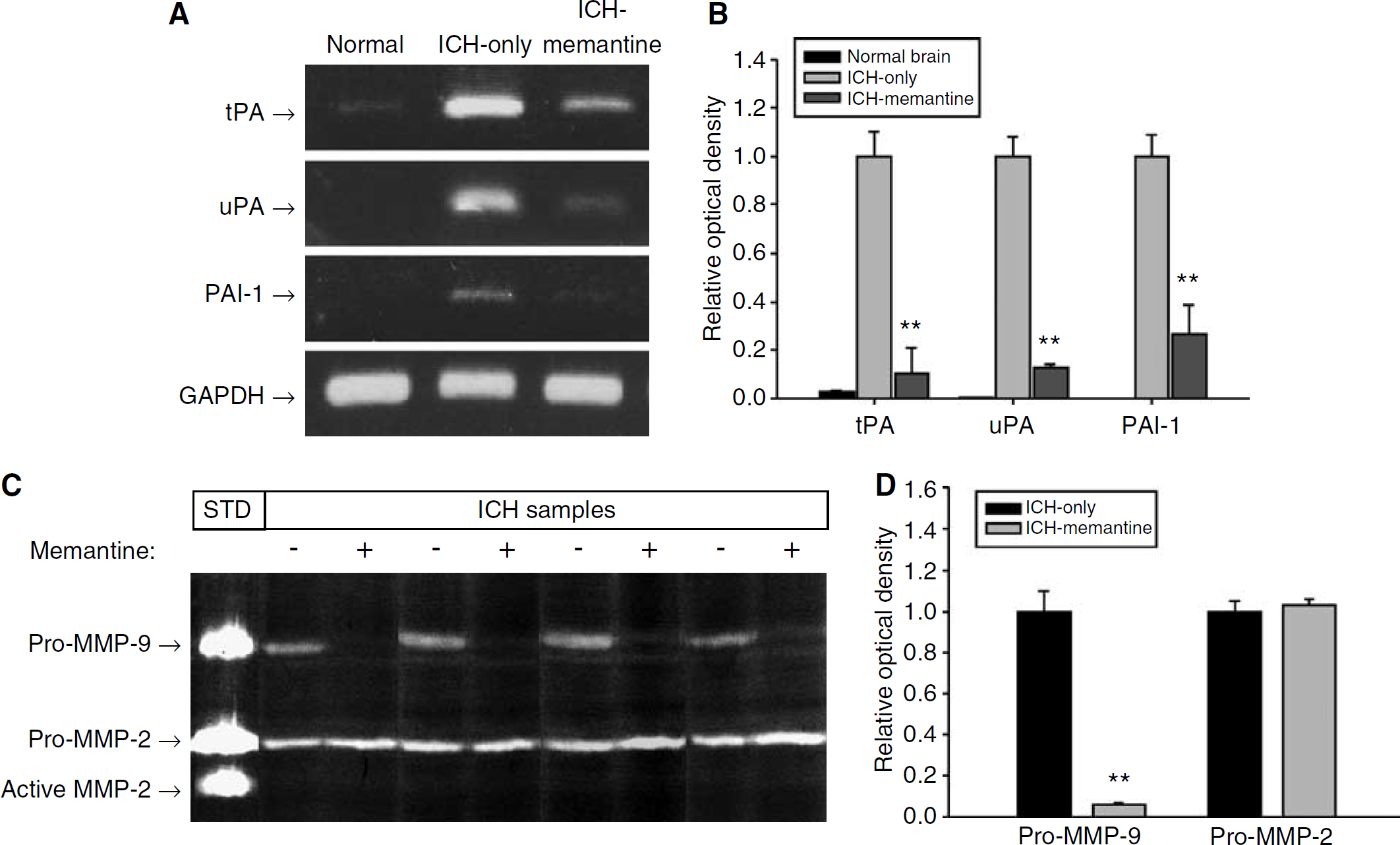
Expression profiles of tPA, uPA, PAI-1, and MMPs. Reverse transcriptase-polymerase chain reaction revealed that memantine had caused reductions in the expression of tPA, uPA, and PAI-1 in the hemorrhagic brains (
In the zymography of MMPs, the ICH-only group showed both bands of pro-MMP-9 and pro-MMP-2 (Figure 4C). On the contrary, memantine-treated group showed markedly decreased levels of pro-MMP-9, as compared with the ICH-only group. Relative OD analysis revealed a 94% decrease in pro-MMP-9 induced by memantine treatment (P < 0.01, t-test; Figure 4D). Pro-MMP-2 was not affected by memantine treatment (P = 0.451).
Discussion
In this study, we attempted to characterize the neuroprotective and antiinflammatory effects of memantine in a rat model of experimental ICH. We also targeted the possible proteolytic enzyme modulation effect induced by NMDAR antagonism. Our results indicated that memantine treatment enhanced functional recovery, and memantine was also associated with both antiinflammatory and antiapoptotic effects. In addition, memantine was found to reduce hemorrhagic expansion, and to exert a profound inhibitory effect on tPA/uPA upregulation and MMP-9 increment in the hemorrhagic rat brains. The neuroprotective effects of memantine in cases of ICH have never been reported previously. In addition, the observation that memantine inhibited endogenous tPA/MMP-9 and suppressed consequent hematoma expansion might have valuable implications with regard to the treatment of ICH.
Glutamate excitotoxicity in ICH had also never been fully investigated. Glutamate accumulates transiently in extracellular fluids in the perihematomal region during the early period of ICH, beginning only 30 min after the inception of ICH (Qureshi et al, 2003). Glutamate stimulation of the metabotropic glutamate receptors results in a vertiginous increase in neuronal tPA synthesis (Shin et al, 2004). Tissue plasminogen activator is generated naturally both by neurons and microglia, and plays an important role in stroke pathophysiology (Gravanis and Tsirka, 2005). As our results show, endogenous tPA can be induced in experimental ICH models. Previous reports suggest that tPA cleaves the NR1 subunit of the NMDAR, and potentiates NMDA toxicity via the amplification of NMDA calcium currents (Nicole et al, 2001; Gravanis and Tsirka, 2005). Tissue plasminogen activator and uPA have both been known to constitute exacerbating factors in thrombin or hemoglobin-induced cell death (Figueroa et al, 1998; Wang et al, 1999). Tissue plasminogen activator appears to be associated with microglial activation subsequent to the induction of ICH, eventually culminating in BBB breakdown and brain edema (Thiex et al, 2004).
Thus, this cascade of reactions, which involves glutamate, NMDAR, and tPA, constitutes a vicious cycle which culminates in catastrophic neurotoxicity (Liu et al, 2004). As the results of our present study indicate, memantine may serve to disrupt this vicious cycle, due to its ability to ameliorate NMDAR overstimulation, and due to its inhibitory effects on tPA/uPA and MMP-9. The enhanced functional recovery observed in the memantine group appears to be attributable to a combination of these effects.
Hematoma volume is a principal determinant of mortality and functional outcome after ICH (Broderick et al, 1993; Mayer et al, 2005), and early hematoma growth is an important factor in neurological deterioration (Brott et al, 1997). Early hematoma growth can occur even in the absence of coagulopathy, and results from continued bleeding or rebleeding at multiple sites, within the first few hours after the onset of ICH (Mayer, 2003; Mayer et al, 2005). In our study, memantine treatment was determined to reduce hematoma expansion.
One of the most important factors involved in the hematoma expansion is MMP-9. Matrix metalloproteinase-9 level have previously been shown to increase in experimental ICH models (Rosenberg and Navratil, 1997; Wang and Tsirka, 2005). Plasma MMP-9 concentrations were showed to be significantly higher in patients exhibiting early hematoma enlargement (Silva et al, 2005). Matrix metalloproteinase-9 levels are amplified by tPA, via the LRP lipoprotein receptor (Wang et al, 2003; Lo, 2004), and tPA/uPA system is epistatic to MMP-9 activation (Liu et al, 2005). In addition, MMP inhibitors can cause the reduction of tPA-induced hemorrhagic transformations in experimental models of ischemia (Lo et al, 2003). In our study, decreased levels of MMP-9 appears to be partially attributable to the decreased level of tPA, because tPA upregulates MMP-9 mRNA and protein (Wang et al, 2003). Consequently, the decreased hematoma expansion in memantine treatment is likely due to the decreased tPA and MMP-9 level.
Excessive MMP activity generates a host of deleterious effects, including cell death, inflammation, and edema (Lo et al, 2003). Matrix metalloproteinase-9 plays a key role in ICH brain injury as well as in cerebral ischemia, and blocking MMP-9 activity shows therapeutic efficacy in experimental ICH (Wang and Tsirka, 2005). In our study, we showed that blocking glutamate excitotoxicity ameliorated excessive MMP-9, which suggests that glutamate toxicity exists at the upstream of MMP-9 in experimental ICH. Targeting glutamate in ICH may provide more fundamental therapeutic approaches with regard to the excessive MMP-9.
Nevertheless, further studies are warranted because of the following reasons. First, memantine failed to reduce the cerebral edema in the present study, although MMP inhibitors have been shown to reduce cerebral edema in ICH secondary to bacterial collagenase in rat brain (Rosenberg and Navratil, 1997). Second, collagenase-induced ICH is a not so physiologic hemorrhage that the effect of memantine on tPA/uPA and MMP-9 should be verified again in more physiologic ICH conditions like human situations. Third, memantine was administered 30 min after ICH induction, which is too early for use clinically. A time window study should be performed to confirm the effect at later time points.
In this study, we presented evidences that memantine treatment can enhance functional recovery, and exerts a set of pleiotropic neuroprotective effects, including the reduction of hematoma expansion and the inhibition of apoptosis and neuroinflammation. The memantine-induced inhibitions of endogenous tPA/uPA synthesis and subsequent inhibition of MMP-9 may provide us with new insights into the treatment of ICH with NMDAR blockers.
Footnotes
Acknowledgements
Memantine used in this study was kindly provided by Lundbeck.
