Abstract
Vasogenic edema in the corpus callosum is a characteristic finding in high-altitude cerebral edema (HACE). Furthermore, microhemorrhages have been found at autopsies in brains of HACE victims. The objective of this study was to determine if microhemorrhages also occur in nonlethal HACE. Consequently, magnetic resonance imaging (MRI) was performed in patients who had suffered from HACE and in patients who had suffered from severe acute mountain sickness (AMS) by applying imaging techniques highly susceptible to blood or blood remnants. Two experienced neuroradiologists independently evaluated the exams blinded to clinical data. The MRI was performed 2 to 31 months after the event. The MRI of the HACE patients revealed multiple hemosiderin depositions in the brain—predominantly found in the corpus callosum—indicative of microhemorrhages. These changes were not present in the three AMS patients. In summary, hemosiderin deposits detectable by MRI predominantly in the corpus callosum indicate that microhemorrhages occur in nonlethal HACE, which may serve as a novel diagnostic MRI sign for HACE even many months after the event.
Introduction
High-altitude cerebral edema (HACE) is a life-threatening form of acute high-altitude illness that can occur in nonacclimatized individuals exposed rapidly to high altitudes (Bärtsch and Roach, 2001). It is characterized by severe truncal ataxia and clouded consciousness, and may be accompanied by variable focal symptoms that may ultimately result in death within 24 to 48 h because of brain herniation (Dickinson et al, 1983). It rarely occurs below 3,500 to 4,000 m and affects 0.5% to 1.5% of mountaineers between 4,000 and 5,500 m (Hackett et al, 1976). In most cases, acute mountain sickness (AMS), characterized by headache, anorexia or nausea, dizziness, and insomnia, precedes the development of HACE. Lumbar spinal fluid pressure is increased (Singh et al, 1969) and magnetic resonance imaging (MRI) is compatible with vasogenic brain edema. In one study, seven out of nine patients with HACE presented with T2-hyperintensities confined to the genu and splenium of the corpus callosum. All four patients available for follow-up fully recovered and the MRI changes resolved entirely (Hackett et al, 1998).
In postmortem exams, multiple microhemorrhages were found in the brain of HACE victims, suggesting that a disruption of the blood—brain barrier occurs in lethal HACE (Dickinson et al, 1983). However, there was no such evidence in AMS (Bailey et al, 2006). We hypothesized that microhemorrhages may also occur in severe, nonlethal HACE and that, therefore, the accumulation of insoluble iron (III) oxide-hydroxide in the form of hemosiderin indicative of microhemorrhage can be found in individuals who had survived HACE.
To test this hypothesis, we performed a magnetic resonance (MR) exam of the brain parenchyma of three individuals who had been diagnosed with HACE within the past 2 years, with a specific focus on microhemorrhages, using a conventional T2*-MRI sequence and a highly sensitive susceptibility weighted imaging (SWI) technique that combines magnitude and phase information with high resolution. Three individuals with comparable altitude exposure who had developed severe AMS but without any signs of HACE served as controls.
Materials and methods
Patients
Patient A developed HACE at 3,860 m in 2006 in the Andes. She is a 66-year-old woman with a history of well-controlled hypertension who was treated with an ACE inhibitor and indapamide. She had no previous exposure to altitudes above 3,000 m and flew to 3,825 m after staying 2 days at 2,300 m. She had symptoms of AMS 8 h after arrival, was somnolent with ataxia and persistent symptoms of AMS on the following day when her blood pressure was normal (160/70 mm Hg), and fell unconscious 44 h after arrival at high altitude. She was flown to Lima where her blood pressure was 220/120 mm Hg, cerebral edema was diagnosed by CT scan, and other underlying illnesses were excluded. After a slow recovery, she was transferred to Erlangen, Germany, 7 weeks after the onset of HACE, where an MRI of the brain was obtained. With the exception of a minor impairment of memory, she recovered completely following further medical treatment.
Patient B developed HACE at 5,700 m in 2005 on Kilimanjaro. He is a 26-year-old healthy mountaineer who had often developed AMS above 3,000 m. Without acclimatization, he ascended within 3 days to an altitude of 5,700 m. He developed ataxia and tunnel vision shortly before arrival in the camp and became unconscious during the night. He was given dexamethasone, treated in a portable hyperbaric chamber, carried down to 4,700 m, and transported to a hospital (1,540 m), where he completely recovered within 4 days.
Patient C developed HACE at 4,400 m while trekking to the base camp on the South side of Mt. Everest in 2004. He is a 40-year-old healthy mountaineer who had rarely climbed at altitudes above 3,000 m. On day 2, he had transient headache and developed anorexia, and felt weak on day 5 at 4,000 m but continued his ascent to 4,400 m, where he slept through the next day and developed severe ataxia on day 7. Except for anorexia, somnolence, and weakness, he had no other symptoms. He was carried down to Pheriche (4,240 m) where he was treated with supplemental oxygen and dexamethasone. On day 9, he was flown to Kathmandu (1,355 m), where he recovered within 1 day.
Patient D developed severe AMS while climbing Kilimanjaro. He is a 40-year-old healthy mountaineer who had no medical problems trekking to 5,700 m and sleeping at 5,400 m on a previous occasion. In 2005, he ascended to 4,600 m at Kilimanjaro in 4 days. On the next day, he developed headache, nausea, and dizziness and felt ‘drunk,’ but he was encouraged by his guide to continue and successfully forced himself to reach the summit (5,895 m) without assistance. He recovered without receiving medication during descent to 3,000 m on the same day. His AMS scores on the summit were calculated retrospectively and determined to be 9 and 3.73 points for Lake Louise (LL) and Environmental Symptoms Questionnaire Cerebral Symptoms (AMS-C) scores, respectively.
Patients E and F are healthy 26 and 47-year-old mountaineers who had participated in our studies performed in August 2005 at the Margherita hut (4,559 m), which involved a 2-day ascent from 1,170 m in Alagna. They had the highest AMS scores in a group of 34 subjects but did not suffer from impaired consciousness or severe ataxia. Their LL scores were 10 points and the AMS-C scores 2.72 and 2.49 points, respectively. They received acetaminophen for headache and recovered within 1 to 2 days.
Patient recruitment: To evaluate the new findings in patient A, patients B to D were recruited through advertising via the internet. Patients B to F gave informed consent for the MRI examination following ethics approval of the University of Göttingen.
Clinical Diagnosis of Acute Mountain Sickness and High-Altitude Cerebral Edema
Acute mountain sickness was assessed using the LL (Roach et al, 1993) and AMS-C (Sampson et al, 1983). Clinical AMS (moderate to severe) was diagnosed as a combined LL score (self + clinical assessment) ≥ 5 points and AMS-C ≥0.7 points as previously described (Bailey et al, 2006). In patient D, this score was obtained retrospectively according to subject memory. High-altitude cerebral edema was diagnosed retrospectively when severe ataxia necessitating assistance for walking and/or impaired consciousness had been present. In subjects A, B, and C, HACE had also been diagnosed by the attending physicians who had examined the patients immediately after evacuation by helicopter or airplane. None of the subjects had taken any drugs for prevention of AMS or HACE during ascent.
Early HACE is characterized by truncal ataxia and somnolence or other signs of clouded consciousness (Bärtsch and Roach, 2001). Based solely on history, it is difficult to distinguish ataxia from dizziness, which is considered a symptom of AMS (Roach et al, 1993). To identify mountaineers who unequivocally suffered from HACE, we requested those with severe ataxia that called for assistance to make the retrospective diagnosis of HACE. With this definition, we cannot rule out that subject D who felt ‘drunk’ on Kilimanjaro had mild, beginning HACE that, however, resolved rapidly during descent without treatment.
Magnetic Resonance Imaging
The MRI protocol consisted of conventional T2-weighted images. Additionally, we used a conventional T2* sequence because of its high sensitivity to blood degradation products. Furthermore, SWI, a high-resolution sequence (Haacke et al, 2004), known to be even more sensitive to blood remnants, was applied in the two less-affected HACE subjects and all AMS subjects.
Magnetic resonance images were acquired on a clinical 3-Tesla whole-body scanner (Magnetom TIM TRIO, Siemens Medical Systems, Erlangen, Germany) using a 12-channel-phased-array receive-only head coil. Magnetic resonance sequences were obtained with the following parameters:
T2 contrast: axial; repetition time (TR)/echo time (TE) 4,710/107; flip angle 148°; number of slices/thickness 20/4 mm; field of view 220 mm; acquisitions/time of acquisition 2/3:15 mins.
T2* contrast: axial, TR/TE 503/20; flip angle 20°; number of slices/thickness 20/6 mm; field of view 220; acquisitions/time of acquisition 2/1:42 mins.
SWI: primary axial three-dimensional data set; TR/TE 28/20; flip angle 15°; number of slices/thickness 72/1.2 mm; field of view 230; acquisitions/time of acquisition 1/6:38 mins.
Postprocessing of Magnetic Resonance Imaging Data
The conventional sequences were displayed in a two-dimensional mode on a Leonardo® Workstation (Syngo, Siemens Medical Systems, Erlangen, Germany), and the SWI data sets were visualized in three dimensions. The scans were qualitatively evaluated by two experienced neuroradiologists independently (MK, KK) who were blinded to the medical history and the clinical data.
Results
The routine MR exam of the most severely affected HACE subject (subject A), including fluid-attenuated inversion recovery (FLAIR) and diffusion weighted imaging (DWI) images, still displayed the morphological characteristics consistent with HACE 6 weeks after event: high signal intensity affecting the corpus callosum—in particular, the splenium (SCC)—on FLAIR images and abnormal DWI with increased water diffusivity confirmed by an increase in the apparent diffusion coefficient (Figure 1).
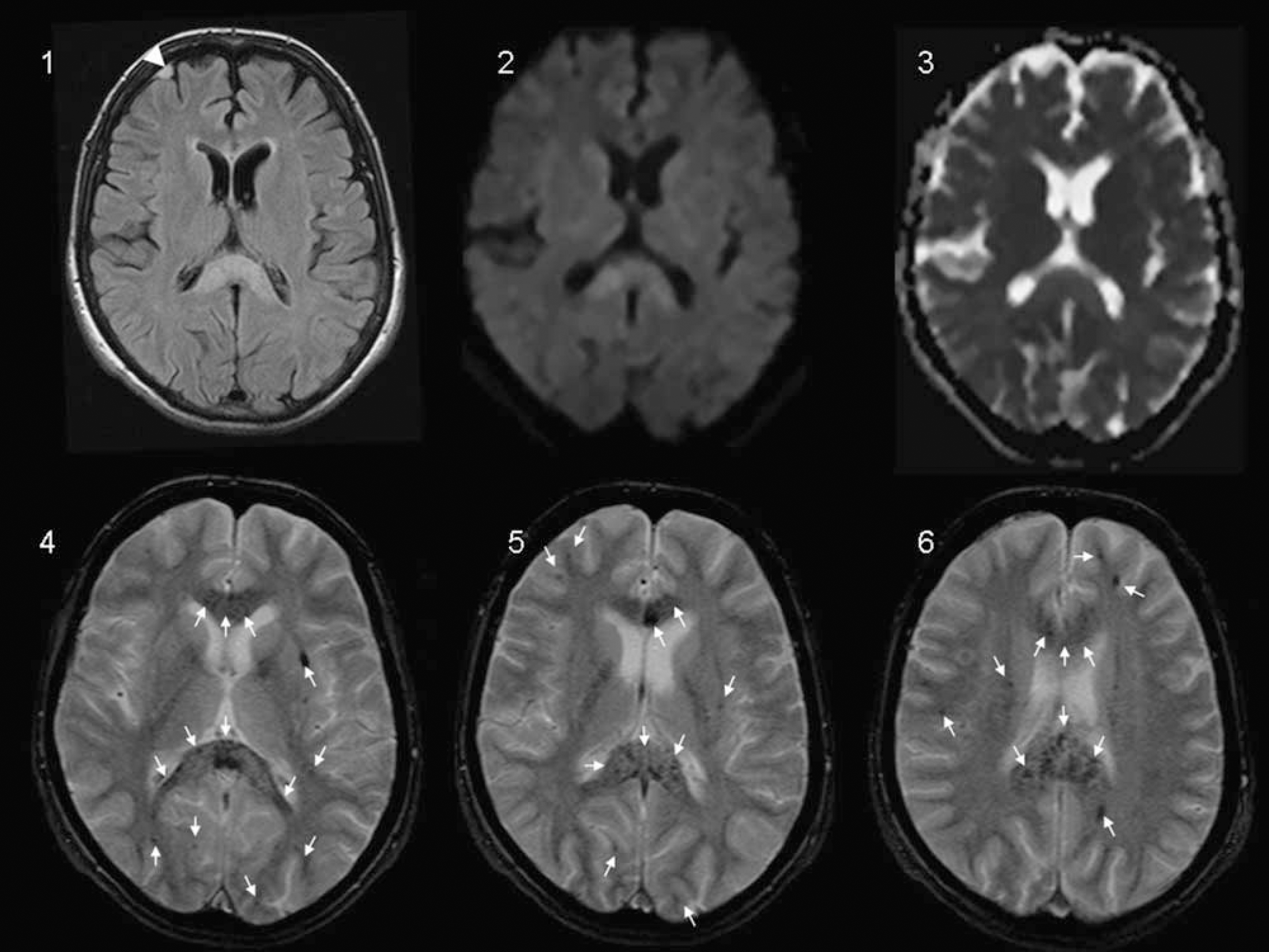
Magnetic resonance images of patient A: FLAIR (1) shows an areal lesion of the corpus callosum—confirmed by DWI (2), with increased values in apparent diffusion coefficient (ADC) (3), indicating increased water diffusivity compatible with vasogenic edema; in combination, the characteristic MRI pattern consistent with HACE (1 to 3) that persists 6 weeks after the event. A novel finding relates to the multiple patchy hypointensities that correspond to multiple microhemorrhages (arrows) displayed on the T2*images (4 to 6). The right frontal meningioma (visible on FLAIR (1); arrowhead) is an incidental finding.
The sequences included in the MR protocol allowed the exclusion of other central nervous system pathologies—for example, ischemic stroke, chronic inflammatory process, or tumor.
T2 and T2*
The conventional T2-weighted images revealed microbleeds that were detectable only in the most severely affected subject (patient A). However, T2*-weighted images with a standard resolution of 6 mm significantly displayed blood or blood remnants in the corpus callosum in all three HACE subjects, whereas the control subjects were not affected (Figure 2). Additionally, in patient A, there were posthemorrhagic lesions in the deep white matter in the frontal, parietal, and occipital lobes, as well as in the insular region remote from the corpus callosum.
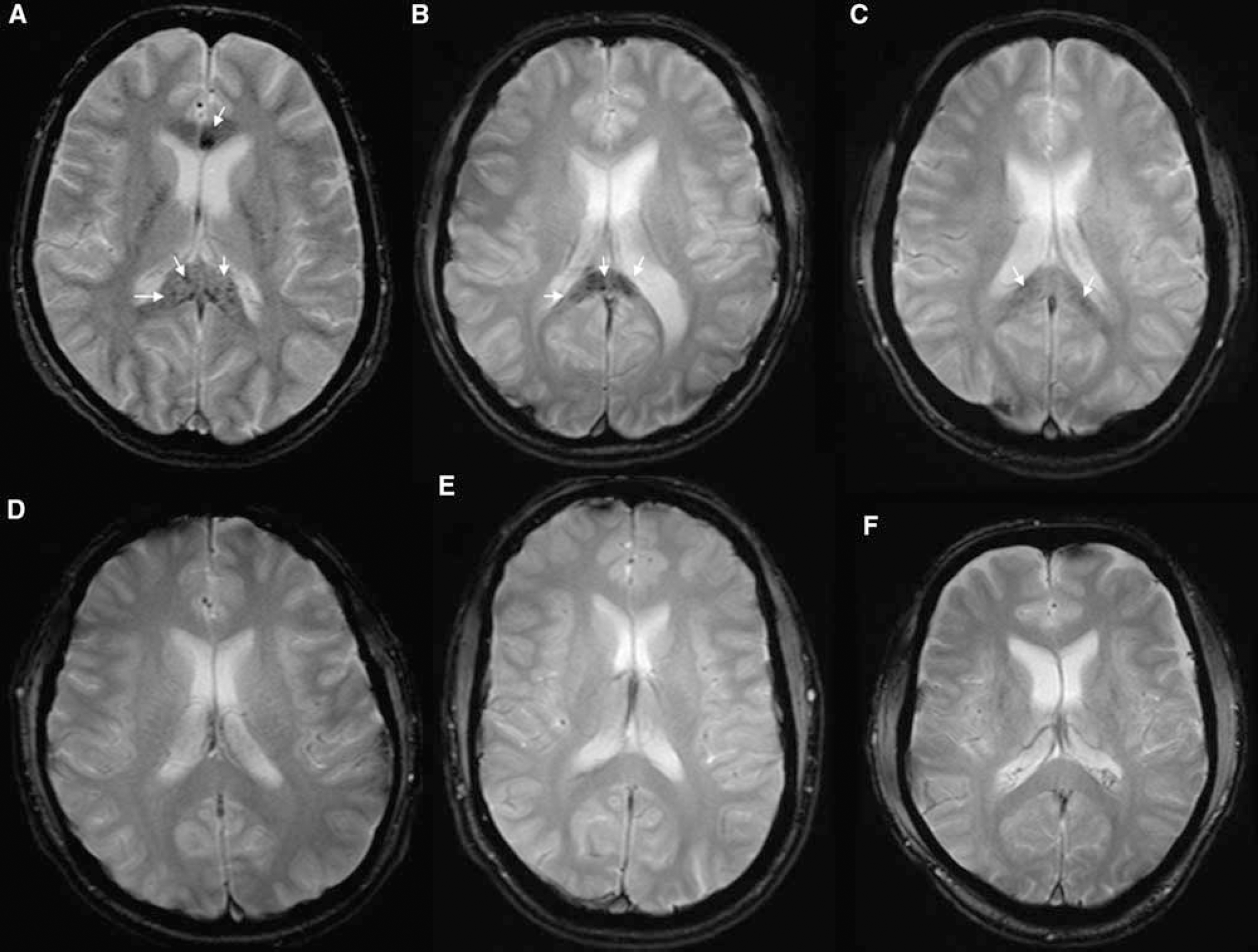
The susceptibility-weighted T2* images show the differences between the normal MR images of AMS patients (
High-Resolution Susceptibility Weighted Imaging Three-Dimensional Data
The SWI data (Figure 3) confirmed the results of the T2*-weighted images with the exception that more lesions in additional localizations could be detected and their delineation was clearer: the corpus callosum was affected in the SCC, the genu (GCC), and also in the corpus corporis callosi. Additionally, lesions were detectable in the left central cerebral area for patient C. Patient B even had a higher lesion load affecting the frontal, parietal, and occipital lobes bilaterally; the lesions in the white matter were less areal or confluent as in the corpus callosum and had a more petechial character.
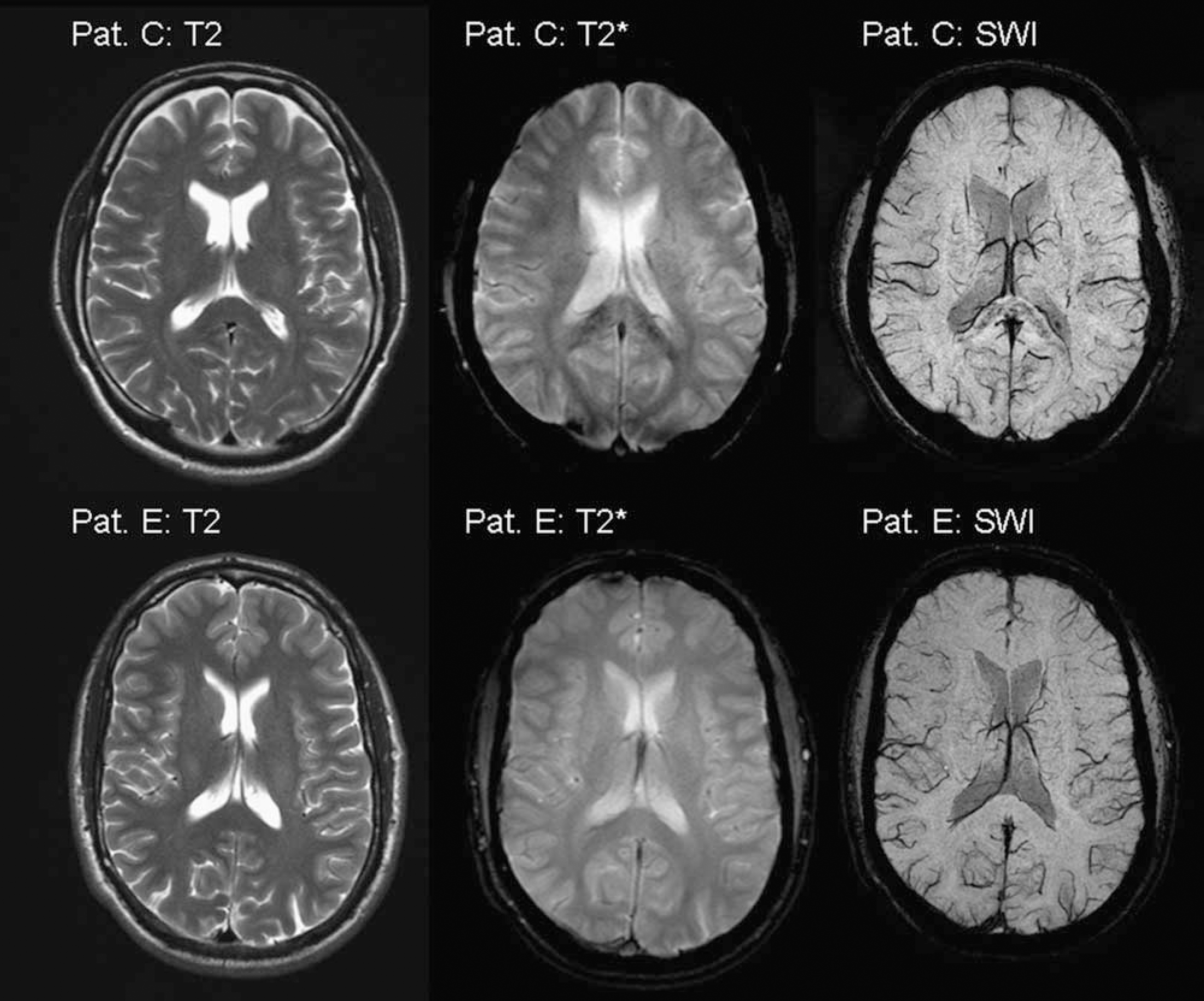
On T2-weighted images, there is no evidence of previous microbleeds, whereas, T2*-weighted images show hemosiderin deposits in the HACE patient (patient C) in the splenium. Even in high-resolution susceptibility weighted (SW) images with its upgraded ability in detecting blood and blood remnants, there was no evidence of hemorrhages in the AMS subjects (patient E).
Besides the previously described callosal regions, genu and splenium, there was also a predilection for the corpus—more apparent in the multiplanar reconstruction on sagittal slices (Figure 4).
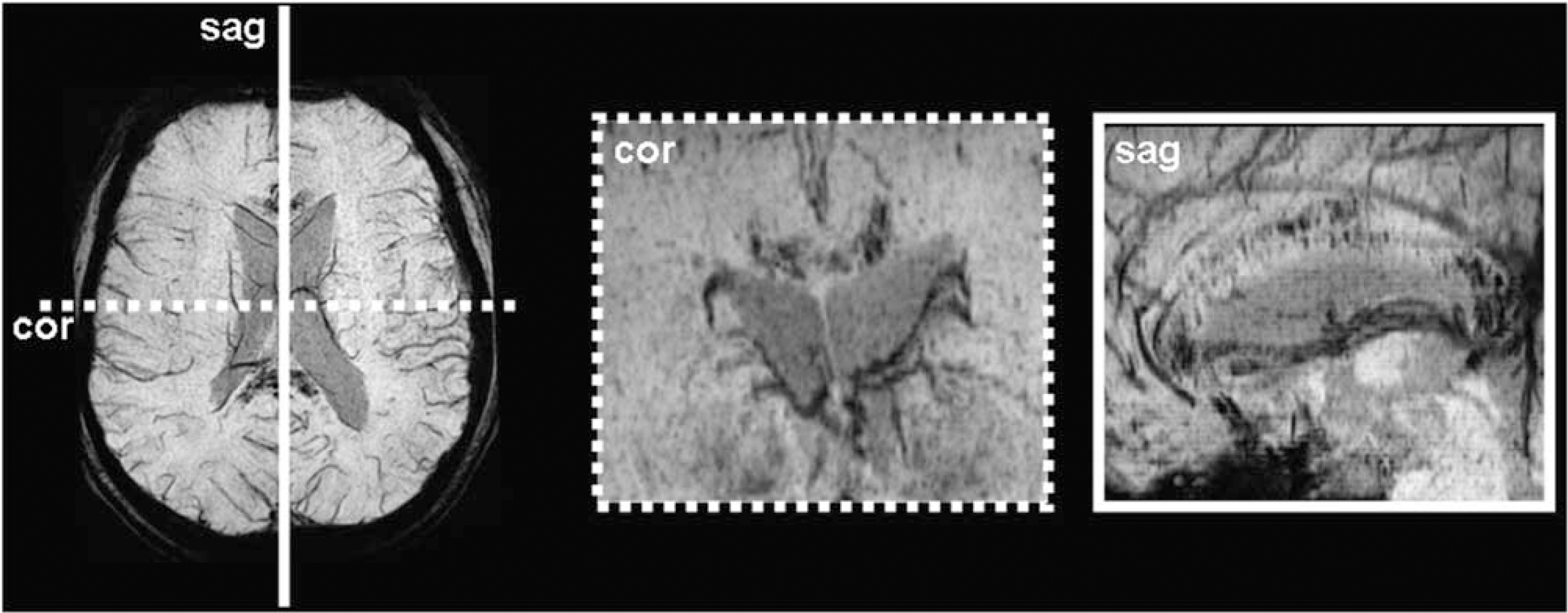
The multiplanar reconstruction shows the predilection for the corpus callosum and also shows that the genu, splenium, and corpus are mainly affected (patient B).
Discussion
The new finding of this study is that hemoglobin degradation products are detectable in the brain parenchyma of individuals who had suffered from HACE 2 to 31 months previously, whereas cerebral hemorrhages could not be detected in individuals with comparable altitude exposure who only suffered from severe AMS. Previous MR studies (Hackett et al, 1998; Wong et al, 2004) of nonlethal HACE have not reported microbleeds. However, these studies did not use susceptibility-weighted MR sequences, and therefore microhemorrhages may have escaped detection. Our observation is compatible with a previous report of vasogenic edema in HACE (Hackett et al, 1998) and with autopsy findings of microhemorrhages in the brains of subjects with lethal HACE (Houston and Dickinson, 1975; Dickinson et al, 1983). Our data prove that microhemorrhages also occur in nonfatal HACE. In addition, our findings show that HACE is associated with a disruption of the blood—brain barrier and that microhemorrhages may be a characteristic finding that distinguishes AMS from HACE. Because high-altitude pulmonary edema is also associated with extravasation of red blood cells from pulmonary capillaries (Swenson et al, 2002), there might be a pathogenic link between the two life-threatening high-altitude edemas.
The microhemorrhages are predominantly located in the corpus callosum, which has previously been identified as the predilection site for HACE (Hackett et al, 1998). The reason why the corpus callosum is so vulnerable to capillary ‘stress-failure’ remains to be resolved. Blood is supplied to the corpus callosum by small and very short perforating arteries that lack adrenergic tone, potentially making it susceptible to hypoxic vasodilatation and autoregulatory failure with resultant overperfusion (Hackett, 1999). However, the localization of edema and microhemorrhages does not explain truncal ataxia, the leading symptom of HACE. Symptoms due to dysfunction of the corpus callosum have neither been routinely investigated nor described in HACE. Our patients have recovered completely from HACE without any apparent clinical sequelae. However, we cannot exclude minor subclinical dysfunction linked to the corpus callosum, because adequate testing has not yet been performed. A test of ‘disconnection syndrome’ (e.g., associative agnosia) may prove of future interest to examine callosal perturbation more specifically. Reports of personality changes (Jeong et al, 2002) and irreversible dementia (Usui et al, 2004) following recovery from HACE indicate a disturbance of the telencephalon.
Recent MRI investigations show that mild vasogenic edema is a normal reaction to hypoxia (Kallenberg et al, 2007; Schoonman et al, 2007). Several factors may account for the rare progression to clinically relevant cerebral edema with microhemorrhages at high altitude. Hypoxemia is usually more severe in HACE than in AMS, and this may induce a greater hemodynamic and cellular or biochemical response. It has been suggested that in the setting of hypoxic cerebral vasodilatation, autoregulation may become transiently impaired, forcing an increase in cerebral capillary hydrostatic pressure to promote extracellular (vasogenic) edema and brain swelling subsequent to mechanical disruption of the blood—brain barrier. Furthermore, hypoxia-induced increase in the accumulation of vascular endothelial growth factor (Tissot van Patot et al, 2005; Xu and Severinghaus, 1998) and in reactive oxygen species (Bailey et al, 2006) may be enhanced and has a greater effect on the blood—brain barrier. Finally, more severe hypoxia may have direct weakening effects on basal membrane structures as shown in animal models of lung capillaries (Miserocchi et al, 2001). There is, however, no direct relationship between ambient PaO2 and the above-discussed mechanisms that may contribute to the pathophysiology of HACE. Tissue hypoxia is most likely the determining factor that shows a large interindividual variability at a given altitude due to differences in ventilation, gas exchange, cardiovascular adjustments, and metabolism.
We cannot exclude that some of the microhemorrhages may have been present before the altitude exposure. This possibility is, however, very unlikely, because microhemorrhages in the corpus callosum have not been reported as incidental findings (Vernooij et al, 2007). Furthermore, the localization associated with usual causes for micro bleedings is very different from the pattern found in our subjects. Chronic hypertensive encephalopathy, the most frequent cause of cerebral (micro-)hemorrhages is typically located in the deep central gray matter and the basal ganglia (Blitstein and Tung, 2007; Tsushima et al, 2003). It is, however, possible that the systemic hypertension that was found after evacuation to low altitude contributed to the greater number of microhemorrhages in the corpus callosum of subject A. Amyloid angiopathy affects mainly the frontal and parietal lobes involving the cortex and subcortical white matter (Blitstein and Tung, 2007; Greenberg et al, 1993). Cavernous malformations are usually located in the deep white matter (Lonjon et al, 1993). Microhemorrhages due to vasculitis are typically localized in the supratentorial white matter (Blitstein and Tung, 2007). In addition, there was no clinical evidence of antiphospholipid antibody syndrome or vasculitis, and no history of a severe traumatic brain injury. In summary, the localization of microhemorrhages, predominantly in the splenium and the genu of the corpus callosum, are new findings that have not been reported previously.
Our observations appear to be characteristic for HACE, because microhemorrhages have not been reported from asymptomatic individuals after exposure to extreme altitudes above 7,000 m (Garrido et al, 1993, 1995; Fayed et al, 2006): In contrast to our findings of T2 hypointensities, unspecific white matter lesions with high signal on T2-weighted images have been found. The lack of hemosiderin deposits in these mountaineers suggests that none of them suffered from HACE, although we cannot exclude the possibility that minor findings may have escaped detection, because no blood or hemosiderin-sensitive sequences such as T2* were applied (Figure 3). However, more extended microhemorrhages can be detected on the standard T2-weighted images as shown at least in the most affected of our HACE patients (patient A).
Because it is impossible to draw a clear line between severe AMS and beginning HACE, we cannot exclude that mild early HACE, which may have been present in subject D, is not associated with microhemorrhages. These may occur only in more severe HACE that was present in subjects A and B, who were both unconscious, or in HACE lasting 1 or more days (subjects A to C). Further investigations need to address the question whether there is a threshold for the occurrence of microhemorrhages in the corpus callosum with regard to altitude or duration and severity of illness.
Our observations are consistent with the severe vasogenic edema previously documented in HACE (Hackett et al, 1998). The disruption of the blood—brain barrier and microhemorrhages in the corpus callosum may be a distinct morphologic characteristic that distinguishes more severe HACE from AMS. The microhemorrhages remain detectable over several years. Thus, these microbleeds, which result in the accumulation of hemosiderin, may prove a novel diagnostic feature of HACE and provide an exciting opportunity for retrospective diagnosis.
