Abstract
Positron emission tomography with 15O-labeled gases (15O-PET) is important for in vivo measurement of cerebral oxygen metabolism both in clinical and basic settings. However, there are currently no reports concerning 15O-PET in mice. Here, we developed an 15O-PET method applicable to mice with spontaneous respiration of 15O-gas without a tracheotomy catheter. Sequential 15O-PET was also performed in a mouse model of chronic cerebral hypoperfusion with bilateral common carotid artery stenosis (BCAS) induced by placement of microcoils. 15O-gas with isoflurane was supplied to the nose of mouse with evacuation of excess 15O-gas surrounding the body. 15O-PET was performed on days 3, 7, 14, 21, and 28 after surgery. Cerebral blood flow (CBF), cerebral blood volume, oxygen extraction fraction (OEF), and cerebral metabolic rate of oxygen (CMRO2) were calculated in whole brains. A significant decrease in CBF and compensatory increase in OEF in the BCAS group produced CMRO2 values comparable to that of the sham group at three days post-operation. Although CBF and OEF in the BCAS group gradually recovered over the first 28 days, the CMRO2 showed a gradual decrease to 68% of sham values at 28 days post-operation. In conclusion, we successfully developed a noninvasive 15O-PET method for mice.
Keywords
Introduction
Since oxygen is a fundamental energy source for brain activity, use of Positron emission tomography (PET) for in vivo quantitative measurements of parameters related to cerebral oxygen metabolism such as cerebral blood flow (CBF), cerebral blood volume (CBV), oxygen extraction fraction (OEF), and cerebral metabolic rate of oxygen (CMRO2) in the presence of 15O labeled gases (15O–O2, 15O–CO2, and 15O–CO) is of great importance both in clinical1–4 and basic settings.5–7 Given the importance of small animal models such as mice and rats to many research fields, the development of noninvasive measurement methods to assess oxygen metabolism in small animals is needed.
In vivo measurements of cerebral oxygen metabolism by living rats can be achieved using PET and injectable 15O–O2 8–12 or 15O–O2 gas inhaled through a tracheotomy catheter.13,14 Both techniques are applicable to O-15 tracers other than O2. Although both techniques are valuable for evaluating cerebral oxygen metabolism in ischemic brains of normal and hypertensive rats, 15 to date there are no reports of methods that can be applied to monitor cerebral oxygen metabolism in live mouse brains. Both techniques described above could be adapted to mice, but they have several limitations, including inadequate specific activity of injectable 15O–O2, animal sacrifice for probe preparation, and invasiveness of the tracheotomy. Given the wider variety of mouse disease models, such a method could have broader applications compared to those that use rats as the animal model.
As such, we sought to develop a method that allows in vivo noninvasive measurement of cerebral oxygen metabolism in living mice. Here we adopted a bilateral common carotid artery stenosis (BCAS) mouse model of vascular dementia, wherein chronic cerebral hypoperfusion produces white matter injury, glial activation, and cognitive impairment.16,17 Previous Laser Doppler measurement of the time course of brain surface CBF measured in BCAS mice showed a transient drop and gradual recovery during the first month after the BCAS operation. 17 However, this study included no information related to oxygen metabolism. Thus, in this study, we first developed a noninvasive PET method to evaluate cerebral oxygen metabolism in mice with spontaneous respiration of 15O-gas, and evaluated the usefulness of this method for sequential evaluation of the cerebral oxygen metabolism in BCAS mice during the first 28 days after the operation.
Materials and methods
Animals
Male C57BL6/J mice (8–9 weeks old) were purchased from Japan SLC (Shizuoka, Japan) and housed under a 12 h light/12 h dark cycle with free access to food and water. The animal experiments in this study were conducted in accordance with guidelines for animal research on Human Care and Use of Laboratory Animals (Rockville, National Institute of Health/Office for Protection from Research Risks, 1996). The study protocol was approved by the Sub-committee for Laboratory Animal Welfare, National Cerebral and Cardiovascular Center Research Institute, Osaka, Japan (Permit Number: 15063 and 16006), and the Animal Experimentation Committee of the Kyoto University, Kyoto, Japan (Permit Number: 14–42). The animal experimental data reported in this study are in compliance with ARRIVE (Animal Research: Reporting in Vivo Experiments) guidelines.
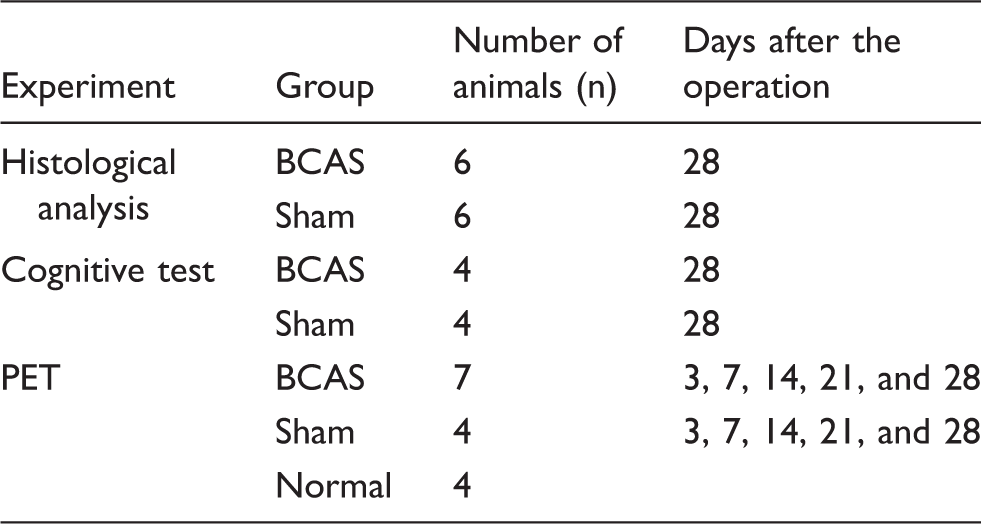
Animal numbers and experimental schedule for each experiment.
Histological staining
Mice (n = 6 for both the BCAS and sham groups) 28 days after the operation were intraperitoneally injected with 50 mg/kg pentobarbital and perfused transcardially with 0.1 M phosphate-buffered saline (PBS) followed by 4% paraformaldehyde in 0.1 M PBS. Brains were stored in the fixative for 3 h and then transferred to 15% sucrose in 0.1 M phosphate buffer for 24 h. Coronal sections (20 µm thick) at approximately bregma + 0.7 mm were cut with a cryomicrotome (Leica, Bensheim, Germany). For fluorescent staining of white matter, brain sections were incubated with FluoroMyelin Green fluorescent myelin stain (1:300 dilution, Molecular Probes, Cat. #F34651) for 1 h at room temperature. For immunostaining, the sections were soaked in 0.1% Triton X-100 for permeabilization followed by an overnight incubation at 4℃ with primary antibodies (a rabbit polyclonal anti-Iba1 antibody, 1:500 dilution, Wako, Cat. #019-19741; a rabbit polyclonal anti-GFAP antibody, 1:1,000 dilution, DAKO, Cat. #Z0334). Sections were then incubated with secondary antibodies (Alexa Fluor 594 and 488-labeled donkey anti rabbit IgG, 1:300 dilution, Molecular Probes, Cat. #A21207 and #A21206, respectively) for 1.5 h in the dark at room temperature. Fluorescence was visualized with an Olympus FluoView microscope equipped with a laser scanning confocal imaging system.
Novel object recognition test
Cognitive impairment of BCAS mice was assessed by a novel object recognition test given on day 28 after the surgery. Experiments were performed in a dimly illuminated room (30 Lx) and mice were habituated in a black box (30 cm × 30 cm × 30 cm) for three days (10 minutes a day) before the training. In the training session, two different objects (a yellow triangular prism and a blue quadrangular pyramid) were placed in the box, and the mice were allowed to interact freely with the objects for 10 minutes. Six hours after the training session, the mouse was allowed to explore in the same box, wherein one of the objects, the blue quadrangular pyramid that was used in the previous training, was replaced by a novel object, in this case a wooden ball. The exploratory preference as an indicator of recognition memory was defined as the ratio of the exploratory time spent exploring the blue quadrangular object in the training session to the time spent exploring the wooden ball in the test session, versus the total time spent exploring both objects.
PET experiments
Oxygen-15 was produced via the 14N(d,n)15O nuclear reaction with a 0.3% O2/N2 gas target for 15O–O2 and 15O–CO preparation, or with a 2.5% CO2/N2 gas target to prepare 15O–CO2. A 1.0, 2.8, or 0.7 μA current of deuterons was accelerated by an in-house CYPRIS-HM18 cyclotron (Sumitomo Heavy Industries, Ltd., Tokyo, Japan) to prepare 15O–O2, 15O–CO, or 15O–CO2, respectively. The bombarded target gas including the resulting 15O–O2 or 15O–CO2 was continuously transported to a 15O-gas purification module to obtain 15O–O2, 15O–CO, or 15O–CO2 gases with high radiochemical purity. Radiochemical purity of 15O-labeled gases was verified by gas chromatography analysis to be more than 99%. The 15O-gas was then transferred to a 15O-gas supply system located near the PET scanner.
PET data were acquired in list mode using a microPET Focus120 scanner (Siemens).
18
Three PET scans with 15O–O2, 15O–CO2, and 15O–CO were successively performed in mice from the BCAS and sham groups (n = 6 and 2, respectively) at 3, 7, 14, 21, and 28 days after the operation (Figure 1(a)). One and two mice in the BCAS and sham groups, respectively, were successively scanned by PET with 15O–CO, 15O–CO2, and 15O–O2 to assess randomization effects (Figure 1(b)). In a similar way, two nine-week-old mice in the normal group were also successively scanned by PET with 15O–O2, 15O–CO2, and 15O–CO, while another two mice were scanned in the reverse order (Figure 1). Scan durations were 10 min for 15O–O2 PET, 10 min for 15O–CO2 PET, and 8 min for 15O–CO PET, with continuous supplies of corresponding 15O-labeled gases for 10 min, 10 min, and the initial half of the period (4 min), respectively; 8 to 12 minute breaks separated PET scans to change cyclotron settings and allow for radioactivity decay in the body before the next PET scan.
PET procedures with animal numbers. Ten out of 15 mice were successively scanned with 15O–O2, 15O–CO2, and 15O–CO (a) while 5 out of 15 mice were scanned in the reverse order (b).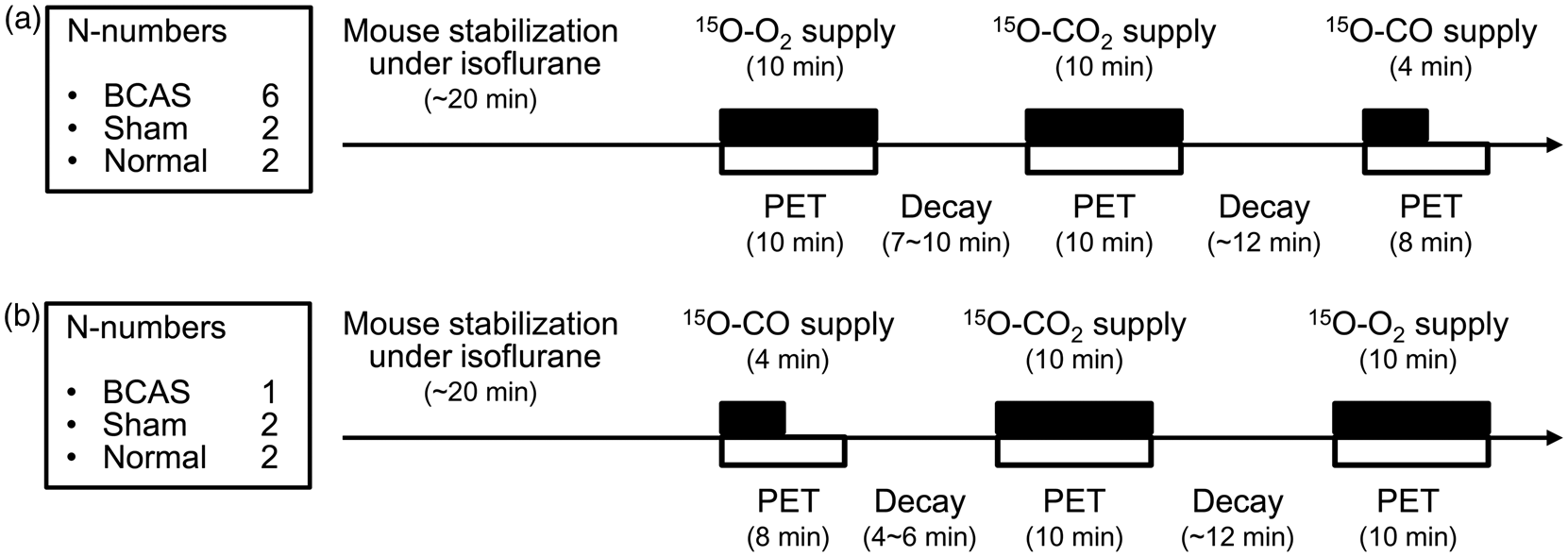
On the day of the PET experiment, the animal was placed in a prone position on a heating pad to maintain the body temperature in a newly designed animal case that allowed for spontaneous inhalation of 15O-labeled gas without tracheotomy (Figure 2(a)). The body temperature was maintained at 36–37℃ during the PET experiments. The animal case had two insertion slots near the head: one for spontaneous respiration of the gas mixture consisting of air, oxygen, and either nitrogen or 15O-gas with isoflurane (around 2%) at a total flow rate of 0.95–1.15 L/min; the second slot allowed evacuation of excess 15O-gas around the body to a gas waste tank placed outside the experimental room at a flow rate of 9–10 L/min, which was sufficient for radioactivity decay (Figure 2(b)). Before initiating the PET scans, the animal was conditioned on the PET bed with spontaneous respiration of a gas mixture consisting of air, oxygen, and nitrogen supplemented with isoflurane using the first slot. A second slot was used for high flow gas evacuation, and the animal was warmed for at least 5 min. We carefully confirmed that the animal was breathing naturally under each condition. When the animal did not reach stable conditions as evidenced by slight head rolling, which indicates an insufficient anesthesia level for the PET experiments, the animal was conditioned for a longer period (∼20 min) until stability and natural breathing was achieved. At the start of the PET scan, 15O-gas (0.3 L/min) was supplied to the animal instead of the cold nitrogen gas (0.3 L/min) used to condition the mouse on the animal bed. The radioactivity concentration of the 15O-gas supplied to the system from the cyclotron settled at 740–1140 MBq/min, which was delivered into the field of view at 655-1010 MBq/min, as calculated from the tube length. The maximum deadtimes during the scans were 2.3 ± 0.7%, 4.3 ± 1.0%, and 2.3 ± 0.5% for 15O–CO PET, 15O–CO2 PET, and 15O–O2 PET, respectively.
Settings of mouse and tubes on a microPET animal bed for 15O-gas PET with spontaneous respiration as shown in photographs (a) and a diagram of the 15O-gas supply system (b). During PET scans, 15O-gas was supplied to the system instead of cold nitrogen gas.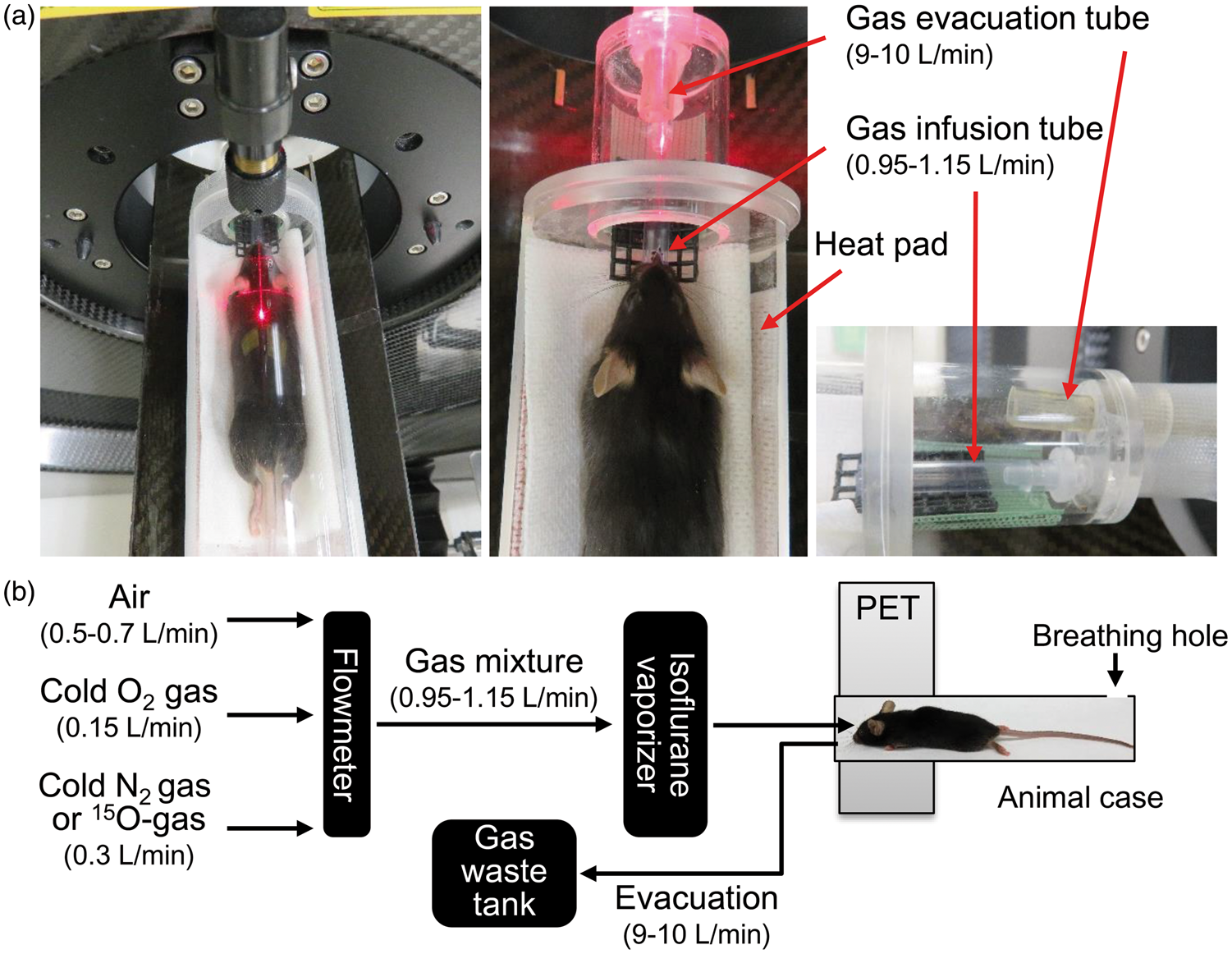
After the PET scans, the mice were kept at a warm temperature to allow recovery from the anesthesia and then returned to their regular housing.
Data analysis
PET data were reconstructed using Fourier rebinning
19
and a filtered back projection algorithm with a 1 min frame duration, 128 × 128 × 95 matrix size, and 0.43 × 0.43 × 0.80 mm3 voxel size, which included corrections for random and deadtimes without corrections for scatter and attenuation. Figure 3 shows representative volumes of interest (VOIs) drawn in the whole brain and the heart on summed images of 15O × CO2 PET and 15O × CO PET scans. Brain and heart VOIs were 29.62 ± 4.67 mm3 and 0.512 mm3, respectively (the shapes and volumes of the heart VOIs were identical for all measurements, and only the location was adjusted for a series of PET scans of each animal taken on a single day). The time activity curve (TAC) in the heart VOI was used as an input function for parameter calculations with an assumed recovery coefficient of 1.0. Parametric images of CBF, CBV, OEF and CMRO2 were calculated from each voxel-wise TAC using in-house software following an ordinary three step autoradiography method.
20
In brief, CBV was calculated from a 15O–CO image based on equation (1):
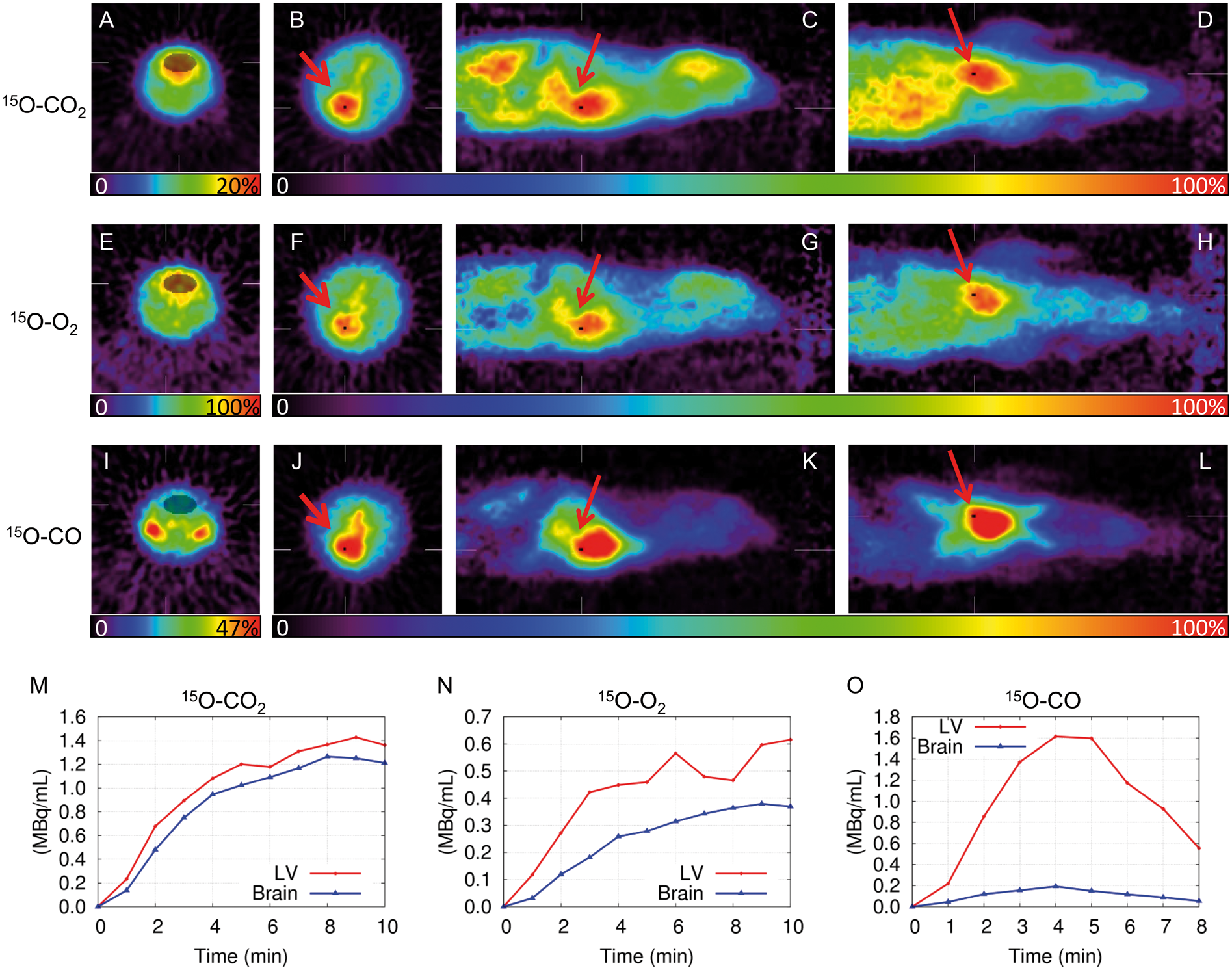
Representative PET images of head (A, E, I) and thoracic (B–D, F–H, J–L) regions of a BCAS mouse (21 days after operation) administered with 15O–CO2 (A–D), 15O–O2 (E–H), or 15O–CO (I–L). PET images are shown as summed images of all frames. Circles designate regions of interest manually placed on the images to calculate cerebral parameters in whole brains (A, E, I) and the hearts (B, F, and J in transverse, C, G, and K in sagittal, and D, H, and L in coronal planes). The time radioactivity curves from the mouse administered with 15O–CO2 (M), 15O–O2 (N), or 15O–CO (O) are also represented.

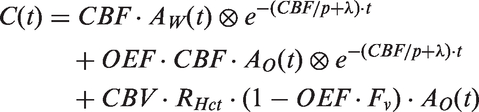

Since several constants were assumed for the calculations as mentioned above, we simulated the effects of variations in the recovery coefficient on experimental parameters and present the results in a Supplementary document.
Statistics
CBF (ml/min/g), CBV (ml/g), OEF, and CMRO2 (ml/min/g) values of the whole brain calculated from the parametric images are expressed as means ± standard deviations. The significance of differences for time-dependent changes in CBF, CBV, OEF, CMRO2, and post-operative weight between the BCAS and sham groups was assayed with repeated measures two-way ANOVA followed by multiple comparisons with statistical significance determined using the Holm-Šídák method (alpha = 5.000%) with GraphPad Prism 6 (GraphPad Software, Inc., CA, USA). A p value < 0.05 was considered statistically significant.
Results
Body weight after operation.

Data are represented as mean ± SD in grams.
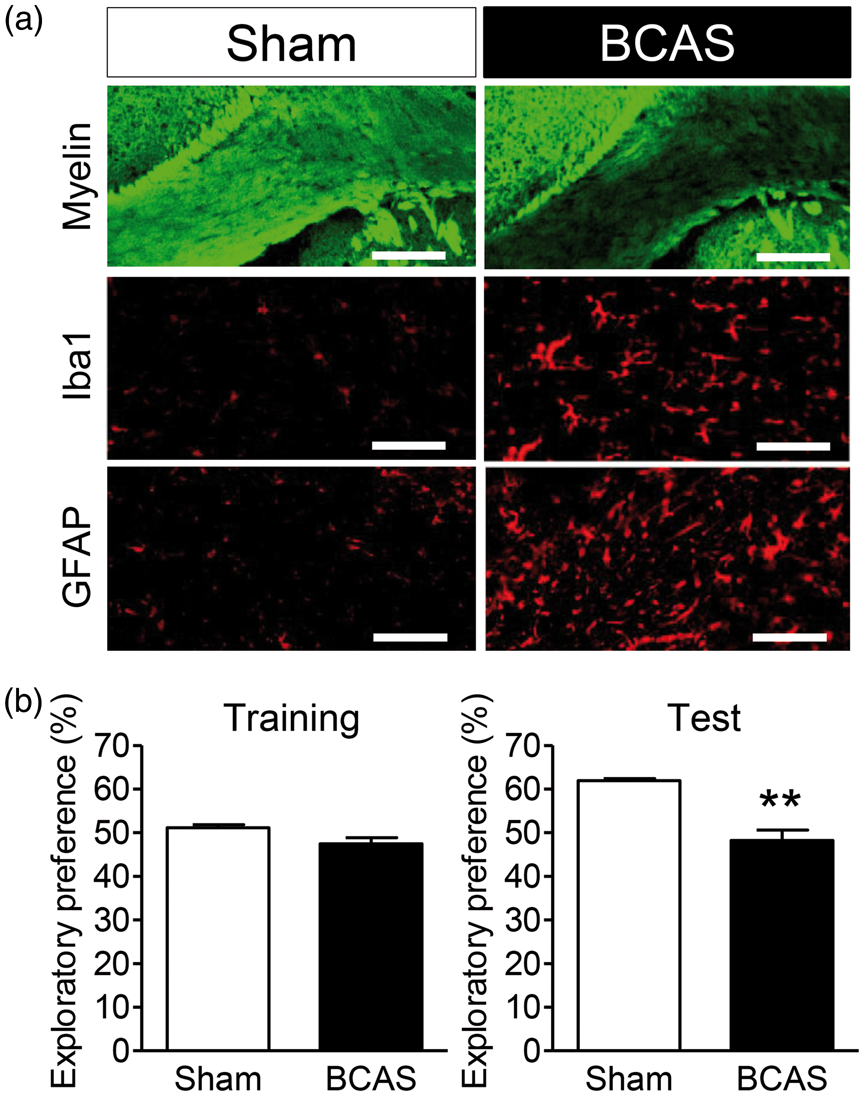
(a) Histological evaluation of the corpus callosum in the sham (left panels) and BCAS groups (right panels) 28 days after the operation. Fluoromyelin staining for myelin density (upper panels), and immunostaining of Iba1 (a marker of microglia/macrophages; middle panels) and GFAP (a marker of astrocytes; lower panels) are shown. The scale bar represents 200 µm in the upper panels and 100 µm in the middle and lower panels. (b) Effects of chronic cerebral hypoperfusion on recognition memory in a novel object recognition test conducted 28 days after the operation. The preferences for one of the objects in the training session (left) and the displaced/novel object in the test session (right) were measured for the sham and BCAS groups. Note that BCAS-operated mice exhibited a diminished ability to discriminate between novel and familiar objects in the test session. Values are mean ± SEM (n = 4). **p < 0.05 vs. the sham group.
The total time for the entire PET experiment, including 3 scans and 2 breaks, for each mouse during a single day was 47.8 ± 4.1 (43–74) min. Following an ordinary three step autoradiography method with the time radioactivity curve in the heart VOI used as an input function (Figure 3), values for CBF, CBV, OEF, and CMRO2 in whole brains were calculated.
The CBF, CBV, OEF, and CMRO2 of the normal group were 0.62 ± 0.05 ml/min/g, 0.051 ± 0.006 ml/g, 0.44 ± 0.06, and 0.063 ± 0.005 ml/min/g, respectively, with 8.7%, 12.4%, 13.3%, and 8.5% of coefficients of variance (%COV), respectively. The BCAS group showed the lowest CBF value at three days post-operation (0.27 ± 0.02 ml/min/g, 39% of the sham value: 0.69 ± 0.05 ml/min/g) and gradually increased thereafter (Figure 5(a)). Meanwhile, the CBF of the BCAS group was 90% of the sham value at 28 days post-operation (0.57 ± 0.05 and 0.63 ± 0.01 ml/min/g in BCAS and sham groups, respectively) (Figure 5(a)). The CBV values in both groups ranged from 0.039 to 0.051 ml/g with no significance between the groups during the experimental period (Figure 5(b)). The OEF of the BCAS group was significantly increased at three days post-operation (0.92 ± 0.12 in the BCAS group, which was 2-fold higher than the sham value: 0.46 ± 0.03), and gradually decreased to levels similar to the sham group such that by 21 days post-operation, there was no significant difference between the groups (Figure 5(c)). As CMRO2 is proportional to the product of CBF and OEF, the BCAS group showed a similar CMRO2 value to that of the sham group at three days post-operation (0.059 ± 0.005 and 0.067 ± 0.006 ml/min/g in BCAS and sham groups, respectively) (Figure 5(d)). The BCAS group showed a gradual decrease in CMRO2 at seven days after the operation such that the value was 85% that of the sham group (0.056 ± 0.005 and 0.067 ± 0.006 ml/min/g in BCAS and sham groups, respectively) and by day 28 was 68% of the sham group (0.044 ± 0.007 and 0.065 ± 0.004 ml/min/g in BCAS and sham groups, respectively) (Figure 5(d)). Meanwhile, the sham group values remained unchanged throughout the experimental period. Representative perfusion images in BCAS and sham mice over 28 days post-operation are displayed in a Supplementary figure.
Temporal changes in CBF (ml/min/g) (a), CBV (ml/g) (b), OEF (c), and CMRO2 (ml/min/g) (d) in whole brains of BCAS (closed circle symbol) and sham (open circle symbol) mice after the operation. Values are expressed as means ± sd. *p < 0.05 vs. the sham group on the same day.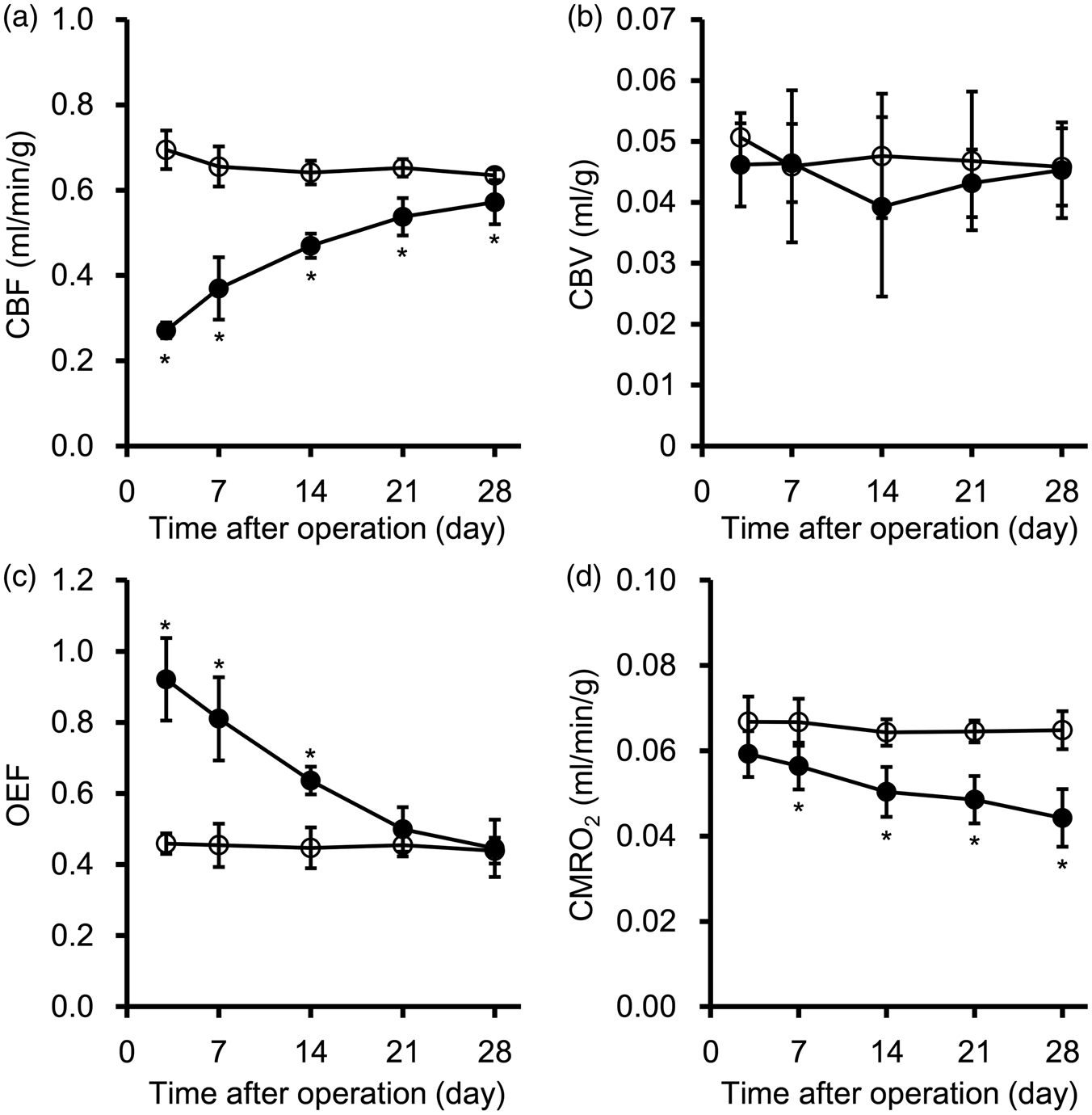
Discussion
Here we developed a 15O-gas supply system that allowed mice to spontaneously breathe 15O-gas with isoflurane anesthesia without the need for invasive procedures such as tracheotomy. Using this apparatus, we acquired in vivo measurements of cerebral oxygen metabolism in living mice with microPET. The complete non-invasiveness of this method enabled repetitive measurements using a single mouse such that temporal changes in cerebral oxygen metabolism in BCAS mice, which have chronic cerebral hypoperfusion and serve as a vascular dementia model, could be followed for one month after the operation. Moreover, since this method is straightforward and completely noninvasive, it should have applications for a broad range of research fields and can be used to elucidate functions related to cerebral oxygen metabolism in a variety of diseases, including cerebrovascular disease, Alzheimer’s disease, Parkinson’s disease, and Moyamoya disease.
Since the pathology of the above-mentioned diseases is characterized by a long progression period, a repeatable and noninvasive method to evaluate physiological parameters in animal models is required. Although nuclear medicine using PET was originally recognized as an optimal modality for such requirements, PET using 15O-gas for basic research with small animals such as rats remained problematic since an invasive tracheotomy was needed to administer the 15O-gas 13 . To avoid invasive procedures and to supply 15O-gas to the mice through normal nasal respiration, the flow rate of the 15O-gases and the deuteron current was limited to ensure stability of the 15O-gas purification system and an appropriate radioactivity dose for PET scanner performance. Since the total infusion flow rate was set through the addition of air and cold oxygen to deliver sufficient anesthesia and maintain animal homeostasis, we next checked the evacuation flow rate to prevent radioactivity from remaining around the animal’s body. Since preliminary tests indicated that higher levels of background radioactivity remained with lower evacuation flow rates, we used an evacuation flow rate that was ∼10-fold higher than the infusion flow rate. In fact, we achieved more than 30 of the heart to background ratios of the radioactivity in the summed images, which indicated that background noise should have minimal effects on the parameter calculations.
To further reduce the invasiveness of 15O-PET procedures, we avoided arterial blood sampling during the PET scan, particularly considering that mice have a small total blood volume. Thus, we defined a VOI in the heart to estimate input functions used to calculate parameters. Although precise evaluation of this estimation approach is needed because this is the first study to use the heart VOI in a 15O-PET assay of oxygen metabolism by mice, the parameters in this study were obtained using ordinary equations that are widely applied both in clinical and basic settings and are similar to those reported in previous studies using rats (Supplementary Table).8–13 Therefore, the OEF value obtained in this study appears to be adequate for mice. The recovery coefficient for the input function, which was obtained from the radioactivity concentration time course in the left ventricular cavity, was assumed to be 1.0 for calculations of CBF, CBV, OEF, and CMRO2. The true recovery coefficient might be less than 1.0 considering the possible underestimation of the radioactivity concentration due to the spatial resolution of the PET scanner, cardiac motion, and heart sizes, as well as spill-in of relatively lower radioactivity concentrations from surrounding tissues such as the myocardium and the lungs. The relatively large positron range of O-15 also diminishes the effective spatial resolution of the PET scanner, and in turn the magnitude of the recovery coefficient. From our simulation study (Supplementary document), the degree of overestimation for CBF, CMRO2, and CBV values increased as the recovery coefficient decreased. However, we used animals that had a similar range of body weights (Table 2) and normal cardiopulmonary functions so that at least the same recovery coefficient would be appropriate for the analysis. Regarding ambiguity of RHct, OEF and CMRO2 estimation was not affected by errors in RHct because the error was cancelled out in the calculation CBV × RHct (equation (3)), which is in contrast to CBV that is inversely proportional to RHct (equation (1)). Therefore, our main objective in this study, the comparison of cerebral oxygen metabolism between BCAS and sham animals, was not affected by RHct variations. Our sensitivity analysis revealed that errors in OEF estimation due to ambiguity of Fv ranged from 0.5 to 1.0 were within ±5% with true Fv = 0.835 (Supplementary document). A 1 min frame duration in the reconstructed images was considered to be sufficient in our study because changes in the TACs of the hearts and brains were slow with continuous inhalation of 15O-gases, and the autoradiography method used in the parameter estimation was based on the TAC integral rather than the curve shape. In addition, to avoid invasive procedures for successive 15O-PET scans on a single mouse, blood gas parameters could not be measured because 100 μl of arterial blood is required for measurements using a portable blood gas analyzer. 8 Instead, body temperature and the infusion flow rate were carefully observed to ensure that the animal remained stable under anesthesia during the PET experiments. The mice in the normal group showed %COV values of around 10%, which would support the stability of animal conditions during PET experiments and the reliability of our 15O-PET system considering that around 20% of %COV was reported in a previous study that used injectable 15O-O2 and normal rats. 11 Therefore, our study showed reasonable results in the differences between the BCAS and sham groups.
We used BCAS mice in this study to evaluate the usefulness of the novel 15O-gas PET method to assess a mouse model of diseases. BCAS mice reportedly show chronic hypoperfusion across the entire brain following placement of small coils that narrow both sides of the common carotid artery and induce cerebral impairments such as white matter lesions and cognitive dysfunction during later phases.16,17 In fact, the BCAS mice we prepared at the beginning of the study also showed disturbances in the myelin sheath as well as activation of microglia/macrophages and astrocytes (Figure 4(a)) that was followed by cognitive impairment 28 days after the operation (Figure 4(b)), which together support the use of BCAS mice as a reliable model animal for PET experiments. However, follow-up observations and repetitive measurements of parameters in a single subject animal would be needed to understand the pathological progress of BCAS mice over a long time period, and these would require that the experimental approach be noninvasive. The PET results showed a marked CBF decrease shortly after the operation and a gradual recovery in a manner similar to that seen for Laser Doppler measurements of brain surface CBF. 17 We also observed a compensatory OEF increase during the first two post-operative weeks that helped prevent a rapid CMRO2 decrease shortly after the operation. Consequently, the CMRO2 gradually decreased over one month to 68% of the sham value at 28 days after the operation in BCAS mice, which might reflect a mild and gradual impairment of neuronal and axonal functions over time that could lead to cognitive disability during later phases. Further studies of BCAS mice, in case with older age, that combine the novel 15O-gas PET technique developed in this study with molecular pharmacology-based approaches will open new avenues to elucidate the underlying mechanisms of vascular dementia. Information from such studies could be used to develop future treatments and preventative strategies for diseases related to defects in cerebral oxygen metabolism. Moreover, since the 15O-gas PET technique is simple and also applicable to rats, evaluation of the usefulness of this method using rats with focal ischemia is currently in progress.
Regarding the limitations of this study, we did not restrain the animal on the PET scanner bed to avoid an increase in background radioactivity on the abdominal side due to increased pressure from the PET scanner bed. Moreover, we did not use apparatuses to monitor respiration and heart rates to ensure natural airflow and respiration of 15O-gas. Instead, we confirmed that the animals had a normal body temperature and were breathing naturally before initiating the PET scans. Isoflurane was used during the three PET scans to maintain animal stability, but given the reported effects of isoflurane on CBF, glucose metabolism, and neural activity,22,23 alternate anesthetic agents could be beneficial for some studies, especially those concerning neurological deficits and/or metabolic abnormalities.
In this study, we successfully developed a completely noninvasive 15O-gas PET method using mice that spontaneously respired 15O-gas. This noninvasive method enabled temporal evaluations of several cerebral oxygen metabolic parameters, including CBF, CBV, OEF, and CMRO2 in a mouse model of cerebrovascular disease over the course of one month. This method should have broad-ranging applications that can be used in a variety of research fields that examine mouse models of central nervous system diseases.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by JSPS KAKENHI Grant Number 15K09912 and Intramural Research Fund (26-6-17) for Cardiovascular Diseases of National Cerebral and Cardiovascular Center. The funding bodies had no role in study design, data collection and analysis, decision to publish, or manuscript preparation.
Acknowledgments
The authors are grateful to Ms. Masako Kunimi and Mr. Masashi Kaji for their contributions to animal handling and cyclotron operation, respectively.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
TT, HS, and HI designed the research TT, JM, and NK performed the research. TT, MY, JM, HS, KK, SK, and HI analyzed the data. TT, HS, and KK wrote the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
