Abstract
Tat-NR2B9c, a clinical-stage stroke neuroprotectant validated in rats and primates, was recently deemed ineffective in mice. To evaluate this discrepancy, we conducted studies in mice subjected to temporary middle cerebral artery occlusion (tMCAO) for either 30 or 60 min according to the established principles for dose-translation between species. Tat-NR2B9c treatment reduced infarct volume by by 24.5% (
Introduction
The peptide neuroprotectant Tat-NR2B9c, also termed NA-1, is a clinical-stage therapeutic
1
developed from our discovery that the NMDA receptor-interacting protein postsynaptic density protein-95 (PSD95) is a hub for excitotoxic signaling and a stroke therapeutic target.2,3 Tat-NR2B9c binds the second PDZ domain of PSD95 (EC50 7 nM)
2
with high specificity and binding selectivity and perturbs synaptic GluN2B:PSD95:nNOS complexs2,4–9
The preclinical efficacy of Tat-NR2B9c as a stroke neuroprotectant has been validated by us and others in several stroke models in rats.2,4,10,14–16 and in comprehensive studies in non-human primates that provided strong evidence of efficacy.17,18 These led to the “ENACT” trial, 1 an international, multi-center, randomized placebo controlled trial that provided encouraging human efficacy data substantially similar to those seen in primates. Thus, it was surprising to us that two groups that tested Tat-NR2B9c in mice reported a lack of efficacy in their hands.19,20 Both groups used a dose of 3 nMole/g, which is effective in rats.2,4,10,14–16 We had never validated Tat-NR2B9c in mice since our research did not require genetically modified animals and mouse models reportedly exhibit high variability between labs even using the same strain, duration of MCAo, and survival period. 21 However, the discrepant data reported in mice vs. other species raises at least three possibilities: that the conclusions of rat, primate and human studies are incorrect, that the conclusions of published mouse studies were incorrect, or that Tat-NR2B9c is ineffective in mice.
Although Tat-NR2B9c is already in clinical testing, we felt it relevant to understand the discrepancy between reported mouse data and that in other species. Such warning signs are too often ignored in drug development, leading to further risks in subsequent development.
According to FDA guidance 22 and literature23,24 on dose translation between species, mice require approximately double the rat dose for dose-equivalence. Thus, we conducted mouse studies to evaluate the effect dose and of tMCAO duration on the ability of Tat-NR2B9c to reduce infarct size in mice.
Materials and methods
Experimental animals
All experiments and procedures were approved by the University Health Network animal care committee, were conducted in accordance with the guidelines of the Canadian Council of Animal Care and are reported herewith in accordance with ARRIVE guidelines. 25 Male C57 Black 6 mice weighing between 21.9–29.7 g (age 8–10 weeks) were used (Charles River, Ontario, Canada). They were kept at 21℃ and 65% humidity with a regulated 12-hour light–dark cycle and with free access to food and water.
Study design
In each study animals were randomly allocated to three treatment groups (0.0, 3.0, 10.0 nMole/g Tat-NR2B9c) or to sham treatment. The individual performing the experimental procedures, administering treatments and performing the analyses was blinded to the treatment assignments. Tat-NR2B9c was prepared at the indicated doses and administered intravenously via the tail vein using a pump (KD Scientific, Holliston, MA, model # 780200) in a volume of 1 µL/g over 5 min beginning at reperfusion.
Experimental procedures
Drug
Tat-NR2B9c/NA-1 (NoNO Inc., Etobicoke, Canada; GMP grade Lot #165I1107) was prepared in PBS at a concentration of either 3 mM/mL or 10 mM/mL.
Experimental outcomes
Infarct volumes (in mm3) were derived after the animals were sacrificed 24 h after tMCAO onset and the brains harvested and frozen to facilitate sectioning into 8 × 1 mm coronal sections. The slices were stained for 20 min at 37℃ with 2% TTC (2,3,5-triphenyltetrazolium chloride: Sigma Aldrich, St. Louis, MD, USA) in 0.9% Saline and photographed with a scanner. The areas of the infarct, ipsilateral hemisphere and contralateral hemisphere were measured using NIH Image (Software version 1.48) and volumes were derived by integrating the edema-corrected infarct areas (area of contralateral hemisphere minus area of healthy tissue in ipsilateral hemisphere). Neurological scores were compared at 3 h and 24 h.
Statistical methods
All analyses were based on intent-to-treat (ITT), analyzing all animals randomized to the study. An animal was considered randomized if it received any amount of study drug. Prespecified exclusions from the ITT analyses were death during tMCAO, SAH during filament removal, and either >20% rCBF during MCAO or ≤ 50% rCBF after reperfusion as measured by laser Doppler. In the 60-min tMCAO experiment, animals that were excluded prior to randomization were replaced. For the 30 min MCAO experiments, 10 animals/group (total 30 animals) were anticipated to suffice to detect a 40% reduction in infarct volume at 0.8 power. In the 60-min MCAO experiments, a smaller effect size was anticipated (30%) and thus 20 animals/group (total 80 animals) were required. One-way ANOVA was used to compare means with a Bonferroni correction for multiple comparisons.
Results
Since the literature on dose translation between species suggests that mice require approximately double the rat dose for dose equivalence,22–24 we examined the impact of Tat-NR2B9c at both the 3 nM/g dose previously reported to be ineffective in mice19,20 as well a higher dose of 10 nM/g. These were tested in two separate studies that differed only in the duration of tMCAO. In the first, tMCAO was for a 30-min duration, whereas in the second, tMCAO lasted 60 min. The rationale for the shorter tMCAO duration was to address the possibility that a 60-min tMCAO can in some circumstances produce a malignant and rapidly progressing stroke that is difficult to neuroprotect. 21
Exclusions and mortalities
Animals were only excluded from analysis if they met the prespecified exclusion criteria (Methods section). In the 30-min tMCAO experiment, two of the 30 animals (both in the 10 nmol/g group) died upon induction of tMCAO. In the 60-min tMCAO experiment, 6 of 86 animals died (3 in the 0 nmol/g, 2 in the 3 nmol/g and 1 in the 10 nmol/g groups) during tMCAO. One animal in the 3 nmol/g did not reperfuse adequately (rCBF < 50% after suture removal), but was not excluded from the analysis as it received study drug and was thus considered to be randomized.
Infarct volumes
The suture occlusion of the MCA reduced rCBF to < 20% of baseline in all cases (Figure 2(a) and (b)). Consistent with previous reports,19,20 the dose of 3 nM/g of Tat-NR2B9c had no impact on infarct volume in either the 30 min or 60 min tMCAO experiments. However, treatment with 10 nM/g reduced infarct volumes by 24.5% ( Effect of Tat-NR2B9c on 24-h infarction volume in male C57BL/6 mice. (a) After 30 min tMCAO. N = 8–10 animals/group. Bars represent means ± SE. * (a, b) Effectiveness of suture occlusion on rCBF during MCAO in (a) 30 min tMCAO and (b) 60-min tMCAO. Each symbol represents the mean rCBF ± SE. (c, d) 3-h and 24-h neuroscores. (c) 30-min tMCAO; 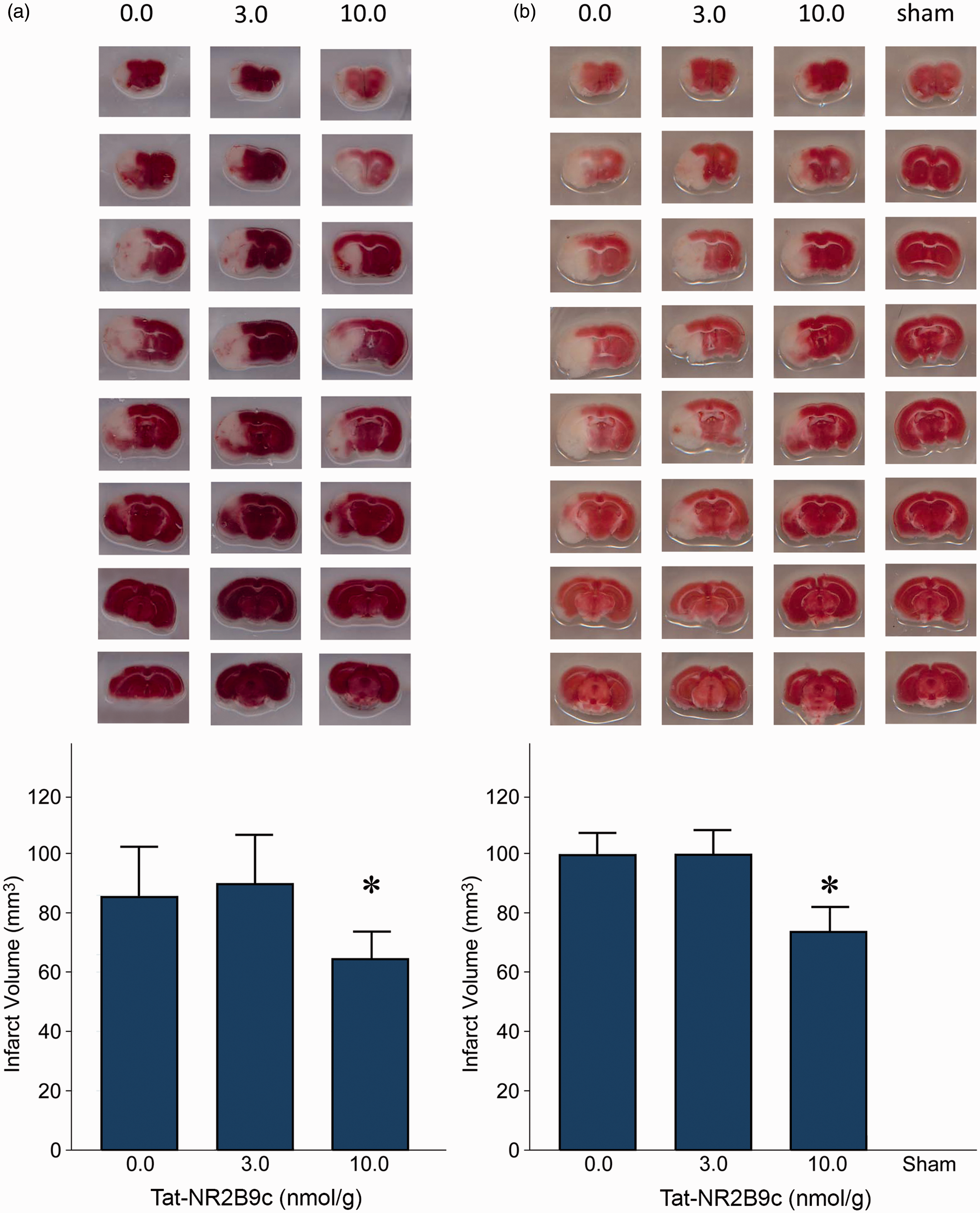
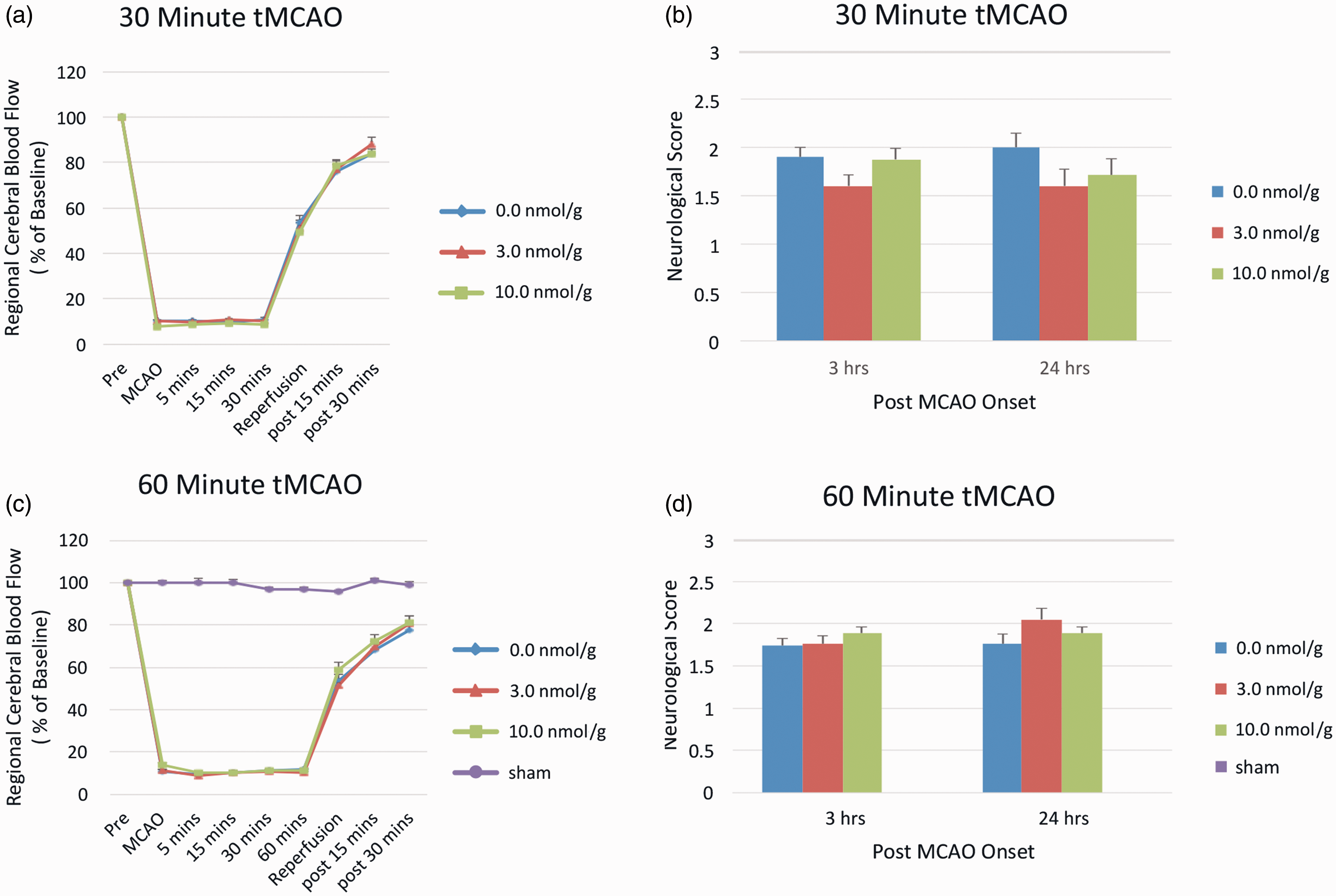
Neurological scores
There were no differences in neurological scores between groups in either the 30 min or 60 min tMCAO experiment (Figure 2(c) and (d)).
Discussion
The validity of PSD95 as a signaling hub and therapeutic target is at this stage generally accepted,29,30 Also, the agent Tat-NR2B9c has received significant internal and external validation.5–9,12,31–35 We had not used mice previously to direct the development of Tat-NR2B9c due to our concerns about the sensitivity of the mouse to rapid infarct extension, sometimes over minutes of MCAo 21 that could lead to significant variability and malignant infarcts. However, since Tat-NR2B9c is already in clinical trials, we felt it relevant to understand the discrepancy between reports of its ineffectiveness in mouse MCAO19,20 and the larger body of published literature in higher species.
The authors reporting negative studies19,20 tested Tat-NR2B9c in mice only at 3 nM/g, a dose identical to that usually used by our group in rats 4 and saw no therapeutic effect. According to Reagan-Shaw and colleagues: “One often-ignored explanation for drug ineffectiveness is the inappropriate translation of a drug dose from one animal species to another”. 23 We therefore conducted a dose–response study in male C57BL/6 mice using methods substantially similar to those reported by the authors,20,36 except that the highest dose was aligned with dosing requirements for mice. Also, Tat-NR2B9c was administered at reperfusion instead of 1 h after reperfusion because of our concern that the rapid progression of infarction in mice 21 would risk leaving no tissue to protect if the intervention is given too late.
We found that when established principles for dose-translation between species are followed, the discrepancy between reported mouse data and our findings of efficacy in higher species is largely resolved. However, while our data explain previously published negative findings with a simple dose–response experiment, they show a rather modest effect size, less than would be expected from previous rat studies. For example, a 90-min tMCAO in rats4,37 or primates 18 reduces the infarction volume by more than 50%. The small effect size of Tat-NR2B9c in mice may be due to our relative inexperience conducting mouse tMCAO experiments, to the high variability inherent in mouse tMCAO models between laboratories, 21 to a rapid malignant progression of the stroke, 21 or that mice are just less sensitive to neuroprotection by Tat-NR2B9c as compared with higher species. This may be due to differences in metabolism, pharmacokinetics, or the neurobiology of NMDAR-PSD95-nNOS signalling. Due to differences in protocols between our and the reported mouse studies, it is also possible that giving the agent at reperfusion (our experiment) instead of at 120 min 20 after a 60-min tMCAO onset or 30 min into a permanent MCAO 19 was more effective.
One caveat is that the groups reporting negative mouse data with Tat-NR2B9c obtained it from their own sources. Since Tat-NR2B9c is in clinical trials, we have significant experience in its production. The purity and potency of the agent can be highly variable, depending on the source and synthesis. Specifically, the peptide content in some manufacturing processes can range from as little as 20% and up to 80%. This wide range would certainly impact on the choice of dose needed to obtain a biological effect.
In conclusion, the disparity between previously reported negative experiments that failed to show a treatment effect of Tat-NR2B9c in mouse tMCAO is simply explained by adherence to the principles of dose translation between species. We also recommend that a dose–response study be conducted whenever an agent is tested in any new species, since its impact on that species biology is at that point still unknown.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Canada Research Chair (Tier 1) in Translational Stroke Research (M.T.). Tat-NR2B9c was a kind gift of NoNO Inc.
Acknowledgement
We thank Dr Badru Moloo for the administrative animal resource support throughout the experiments.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael Tymianski is CEO of NoNO Inc., a biotechnology company founded by the inventors of Tat-NR2B9c to enable its translation to clinical trials.
Authors’ contributions
LMT performed all animal surgeries, drug dosing and evaluations of outcomes. HC maintained blinding with blinded drug preparation, randomization and treatment allocation concealment. MT and LMT planned and analyzed the experiments and wrote the manuscript.
