Abstract
Cerebral ischemia stimulates N-methyl-D-aspartate receptors (NMDARs) resulting in increased calcium concentration and excitotoxicity. Yet, deactivation of NMDAR failed in clinical studies due to poor preclinical study designs or toxicity of NMDAR antagonists. Acamprosate is an indirect NMDAR antagonist used for patients with chronic alcohol dependence. We herein analyzed the therapeutic potential of acamprosate on brain injury, neurologic recovery and their underlying mechanisms. Mice were exposed to cerebral ischemia, treated with intraperitoneal injections of acamprosate or saline (controls), and allowed to survive until 3 months. Acamprosate yielded sustained neuroprotection and increased neurologic recovery when given no later than 12 hours after stroke. The latter was associated with increased postischemic angioneurogenesis, albeit acamprosate did not stimulate angioneurogenesis itself. Rather, increased angioneurogenesis was due to inhibition of calpain-mediated pro-injurious signaling cascades. As such, acamprosate-mediated reduction of calpain activity resulted in decreased degradation of p35, increased abundance of the pro-survival factor STAT6, and reduced N-terminal-Jun-kinase activation. Inhibition of calpain was associated with enhanced stability of the blood–brain barrier, reduction of oxidative stress and cerebral leukocyte infiltration. Taken into account its excellent tolerability, its sustained effects on neurologic recovery, brain tissue survival, and neural remodeling, acamprosate is an intriguing candidate for adjuvant future stroke treatment.
INTRODUCTION
Excitotoxicity is one key element in formation of brain injury after cerebral ischemia.1,2 The complex cell signaling resulting in excitotoxic brain injury after cerebral ischemia involves stimulation of N-methyl-D-aspartate receptors (NMDARs) due to excessive extracellular glutamate release and subsequent increase in intracellular calcium.1–3 The latter gives rise to activation of multiple intracellular pro-injurious signaling pathways such as activation of calpain and c-Jun N-terminal kinase. However, stimulation of NMDAR is not exclusively followed by cell injury or cell death but can also increase cell survival upon activation.4,5
The variety of cell cascades involved in excitotoxic ischemic brain injury gives rise to numerous potential targets for drug intervention,1,2 thus leading to a great deal of preclinical studies. The latter repeatedly showed both neuroprotection and functional recovery in rodent models of cerebral ischemia,6–8 albeit the majority of these studies focused on short-term observations only. Translational approaches into the clinic, however, have failed until recently. 6 The reason for that is multifactorial, including narrow therapeutic time windows, significant side effects, and insufficient quality standards. 2 In light of this, the application of a clinically approved drug together with long-term observation periods might circumvent some of the aforementioned obstacles.
Acamprosate is clinically used for prevention of relapse in patients experiencing alcohol dependence and is effective as well as safe. 9 During alcohol dependence, NMDARs are upregulated resulting in a hyperglutamatergic state, which might lead to potential excitotoxic tissue injury during periods of withdrawal.10,11 In this context, acamprosate inhibits NMDAR stimulation, albeit these effects are indirect only.12–14 In addition to the aforementioned effects during alcohol dependence, experimental evidence suggests a therapeutic role for acamprosate in neurologic disorders such as multiple sclerosis. 15
Since activation of the glutamatergic system and stimulation of NMDAR are decisive in the development of cerebral ischemia as stated afore, a single study analyzed the therapeutic potential of acamprosate in a rat model of cerebral ischemia. 16 Although limited and descriptive in its nature, this study remained the only one demonstrating neurologic recovery on day 3 after stroke after pretreatment with acamprosate. Consequently, the present work systematically analyzed the therapeutic effect of acamprosate in a mouse model of cerebral ischemia, elucidating neurologic recovery and underlying mechanisms for an observation period of 3 months.
MATERIALS AND METHODS
Experimental Paradigm
For all experiments, male C57BL6 wild-type mice (Harlan, Horst, The Netherlands; 25 to 27 g) were used that had free access to food and water. Experiments were approved by local authorities (i.e., LANUV and government of Lower Saxony, Germany) and performed according to EU guidelines for care and protection of animals. Experiments were blinded to both experimenters and analysts and were in compliance with the ARRIVE guidelines (Animal Research: Reporting in Vivo Experiments) for how to report animal experiments. Mice were subjected to 30 minutes of transient focal cerebral ischemia as described below followed by intraperitoneal treatment with either acamprosate (Sigmal-Aldrich, Taufkirchen, Germany; solved in NaCl) or standard saline (control) during reperfusion, at 3, 6, 9, 12, and 24 hours after stroke. Referring to previous studies,15–20 mice received a modified injection protocol with a single intraperitoneal injection of acamprosate at a dose of 400 mg/kg bodyweight (BW). To exclude toxic side effects of acamprosate after high dosage bolus application, analysis of potential weight loss and blood count analysis was performed 1 day before stroke induction as well as on days 7 and 14 after stroke (Table 1). The therapeutic potential of acamprosate as an adjuvant therapeutic means next to thrombolysis was further assessed by additional treatment with either recombinant tissue plasminogen activator (rt-PA) (10 mg/kg BW) or standard saline during reperfusion. Animals were allowed to survive 2 or 84 days after stroke, respectively. Histologic neuroregeneration was assessed by means of differentiation analysis of bromodeoxyuridine (BrdU)-positive cells, which was given daily via single intraperitoneal injections on days 8 to 40 (50 mg/kg BW). Animals used for statistical analysis, including survival periods are given in Supplementary Table 1. Only animals that did not survive the observation period were excluded from the study.
Weight and blood count analysis after treatment with acamprosate
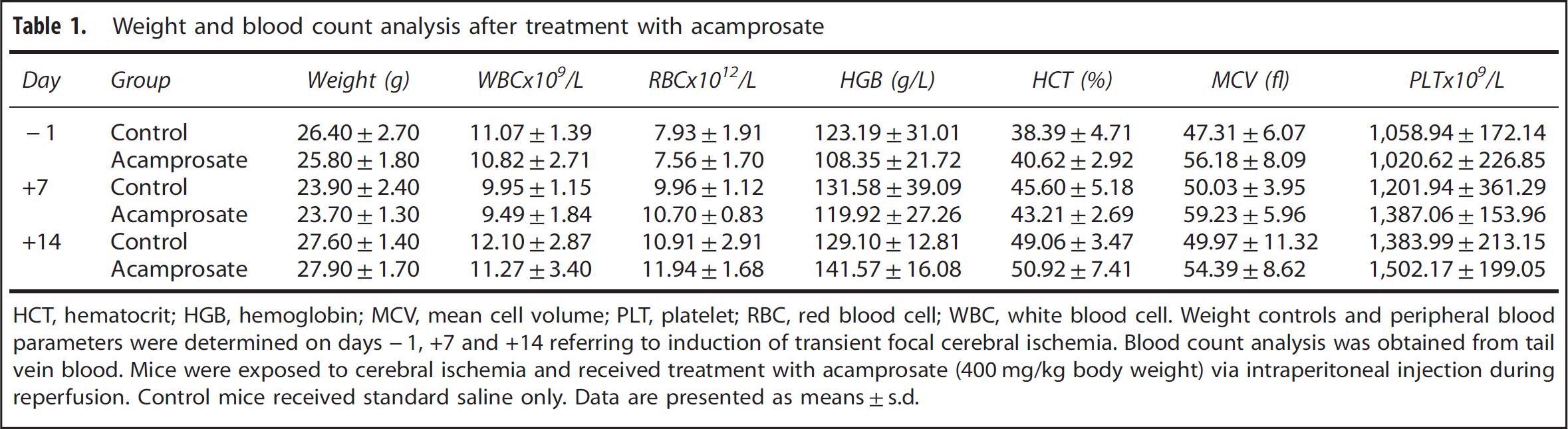
HCT, hematocrit; HGB, hemoglobin; MCV, mean cell volume; PLT, platelet; RBC, red blood cell; WBC, white blood cell. Weight controls and peripheral blood parameters were determined on days − 1, +7 and +14 referring to induction of transient focal cerebral ischemia. Blood count analysis was obtained from tail vein blood. Mice were exposed to cerebral ischemia and received treatment with acamprosate (400 mg/kg body weight) via intraperitoneal injection during reperfusion. Control mice received standard saline only. Data are presented as means ± s.d.
Induction of Transient Focal Cerebral Ischemia Experimental stroke was induced using the thread occlusion model as previously described. 21 As such, mice were anesthetized with isoflurane (1.5%) and nitrogen (68.5%) at 30% oxygen. The silicon-coated nylon filament (180 μm tip diameter; Doccol, Sharon, MA, USA) was inserted into the left common carotid artery. After having reached the left middle cerebral artery, the thread was kept in situ for 30 minutes under constant Laser Doppler flow (Perimed, Sweden). Blood flow was observed for an additional 15-minute period at the end of surgery to guarantee reperfusion.
Assessment of Postischemic Brain Injury and Neuroregeneration
Infarct volumes were analyzed on day 2 after stroke induction using 2,3,5-triphenyltetrazolium chloride (TTC, 2%) staining of 2 mm thick brain sections. Analysis of infarct volumes was performed using ImageJ software (www.imagej.nih.gov). The latter was also used for measurement of brain edema as the relative increase in ipsilateral hemispheric volume in comparison with the contralateral hemisphere.
Long-term neuroprotection was measured by means of determination of neuronal density for which NeuN staining was performed on day 84. Therefore, mice were killed and transcardially perfused with saline and 4% paraformaldehyde. 22 Cryostat sections of 20 μm each were generated and used for immunohistochemical analysis of neuronal density and neuroregeneration. Quantitative analysis within four regions of interest at anteroposterior+0.14 mm, mediolateral ± 1.5 to 2.25 mm and dorsoventral − 2.5 to 3.25 mm was performed on three sections per animal and staining. Stainings were performed using the following primary antibodies: mouse monoclonal anti-NeuN antibody (1:1,000; Millipore, Livingston, UK), monoclonal mouse anti-BrdU antibody (1:400; Roche, Basel, Switzerland), monoclonal rat anti-BrdU antibody (1:400; Abcam, Cambridge, UK), monoclonal rat CD31 (1:200, BD Biosciences, Heidelberg, Germany), and goat polyclonal anti-doublecortin antibody (1:50; Santa Cruz Biotechnology, Heidelberg, Germany). After an incubation period of 18 hours at 4°, sections were repeatedly washed and incubated for 1 hour at room temperature (RT) with the following secondary antibodies: goat anti-mouse Alexa 488 (1:100; Jackson ImmunoResearch, Newmarket, UK), goat antimouse Cy-3 (1:400; Dianova, Hamburg, Germany), goat anti-rat Alexa 594 (1:400; Dianova), and donkey anti-goat Alexa 488 (1:250; Invitrogen, Darmstadt, Germany).
Analysis of Neurologic Recovery
Neurologic recovery was analyzed during an observation period of 3 months in mice that had been treated with either acamprosate or standard saline at 12 hours after stroke. Four well-established behavioral tests were used, all of which have been described by our group. 21 These tests were the corner turn, foot fault, rota rod, and balance beam test. Before beginning of actual tests on days 7, 14, 28, 56, and 84, mice were trained 2 days before induction of stroke. The corner turn test was performed by placing the mouse into an apparatus consisting of two vertical boards forming an angle of 30°. The laterality index (number of right turns/10) was calculated after 10 trials per test day. For the rota rod test, an accelerating velocity (4 to 40 r.p.m.) was set with a maximal velocity achieved after 260 seconds. Maximal testing time was 300 seconds with two runs per test day. The time until the animal dropped was used for statistical analysis. To perform the foot fault test, mice were placed on an elevated steel grid. The relative percentage of foot fault errors (i.e., when animals misplaced their forelimbs) for the right forelimb (referring to the total amount of right forelimb steps) was calculated. The balance beam test was exerted using a long beam with constantly reduced width that is 60 cm elevated from the ground in a horizontal manner. Mice were supposed to reach the platform at the end of the beam. With a maximal testing time of 60 seconds, animals had to reach the platform. The test was performed twice per time point.
Analysis of Blood–Brain Barrier Integrity
Determination of extravasated Evans blue served as readout parameter for determination of blood–brain barrier (BBB) leakage at 48 hours after stroke induction as previously described. 23 Two hours before killing, Evans blue (2%; 2 mL/kg BW) was intravenously injected and a photometric analysis of Evans blue extravasation with λexc. = 620 nm and λem. = 680 nm was performed in homogenates of left ischemic hemispheres.
Analysis of Oxidative Stress
Oxidative stress was indirectly analyzed at 48 hours via measurement of thiobarbituric acid reactive substances such as malondialdehyde within ischemic hemispheres. Malondialdehyde is known to react with thiobarbituric acid yielding a chromogenic compound, which can be photometrically measured at 532 nm.24,25
Flow-Cytometry Analysis
Detection of CD45+ high leukocytes in both the blood and the brain was performed as previously described with slight modifications.22,26 Mice were killed 2 days after stroke and blood samples were collected and the brain was removed. Homogenates of left ischemic hemispheres were used to measurements, which were performed using a rat anti-CD45 antibody (BioLegend, Fell, Germany) for 30 minutes at 4°. Absolute cell numbers were measured using countbright counting beads (Invitrogen, Grand Island, NY, USA).
Determination of Calpain Activity
Calpain activity was measured 48 hours after stroke in tissue lysates obtained from ischemic hemispheres as previously described.25,27 Left ischemic hemispheres were homogenated in in lysis buffer (100 mmol/L Tris-HCl, 145 mmol/L NaCl, and 10 mmol/L EDTA at pH 7.4) and used for fluorimetric analysis by means of a fluorescence microtiter plate reader at 37° with Aexc. = 355 nm and Aem. = 460 nm. Substrate was Suc-Leu-Leu-Val-Tyr-AMC (50 μmol/L; Bachem, Weil am Rhein, Germany) with or without addition of 10 mmol/L Ca2+. Values of calpain activity are given in picomol per min per milligram protein.
Western Blotting
Immunoblots were performed at 48 hours after stroke induction in left ischemic hemispheres, which were complemented with lysis buffer (50 mmol/L Tris, pH 8.0, 150 mmol/L NaCl, 1% Triton X-100, protease inhibitors). Lysates were used for SDS-PAGE electrophoresis (40 μg protein for each sample) and loaded onto 12% polyacrylamide gels. The following primary antibodies were used: rabbit polyclonal anti-p35 (Abcam), goat polyclonal anti-p25 (Thermo Scientific, Braunschweig, Germany), rabbit polyclonal p-Akt (Abcam), rabbit polyclonal Akt (Abcam), rabbit polyclonal anti-phospho-c-Jun (Santa Cruz Biotechnology), rabbit polyclonal anti-c-Jun (Abcam), rabbit polyclonal anti-pSTAT6 (Abcam), and a rabbit polyclonal anti-beta actin antibody (Abcam). Secondary antibodies that were labeled with peroxidase included a goat anti-rabbit (Santa Cruz Biotechnology) and rabbit anti-goat (Thermo Scientific) antibody. Membranes obtained after blotting and staining were scanned and used for densitometric analysis. The latter was performed using Image J software for which relative densities of single bands were determined after rectangular selection followed by generation of profile plots for each single lane. These plots were used for calculation of relative densitometric values in reference to actin, for example.
Statistical Analysis
For analysis of multiple groups, a one-way analysis of variance (ANOVA) followed by the Tukey's post hoc test was performed. On the contrary, statistical comparison between two groups included the Student's t-test. All data are given as means ± standard deviation (s.d.) with a P-value of < 0.05 regarded statistically significant.
RESULTS
Acamprosate Is Neuroprotective and Offers a Wide Therapeutic Window
Acamprosate is well tolerated, except for mild side effects such as headache, flatulence, nausea, and diarrhea. 28 Since the latter might be relevant for stroke outcome, we first determined possible side effects after application of acamprosate during an observation period of 2 weeks. Single application of a relatively high dose of 400 mg/kg BW, however, did not result in significant side effects in terms of weight loss or blood count parameters (Table 1).
Since previous experiments on acamprosate in experimental stroke were restricted to prestroke treatment and a survival period of 3 days, 16 we next analyzed the therapeutic effects of acamprosate in terms of therapeutic window and in combination with rt-PA-induced tissue toxicity. Assessment of the therapeutic time window revealed that single intraperitoneal injections successfully induced reduction of infarct volume on day 2 when given no later than 12 hours after stroke (Figure 1A). Moreover, acamprosate reduced brain edema under the same experimental paradigm (Figure 1B). Consequently, further experiments were performed using the 12-hour injection paradigm only. In light of a potential clinical use, some mice received intravenous injection of either NaCl or rt-PA during reperfusion followed by the aforementioned intraperitoneal injection of acamprosate or NaCl 12 hours after stroke. Infarct volume analysis at 48 hours again showed reduction of brain injury in mice treated with acamprosate, which also diminished rt-PA-induced brain toxicity (Figure 1C). Finally, longevity of acute neuroprotection was further elucidated via determination of neuronal density 2 months after stroke induction. Treatment with acamprosate resulted in sustained neuroprotection as compared with controls (Figure 1D).
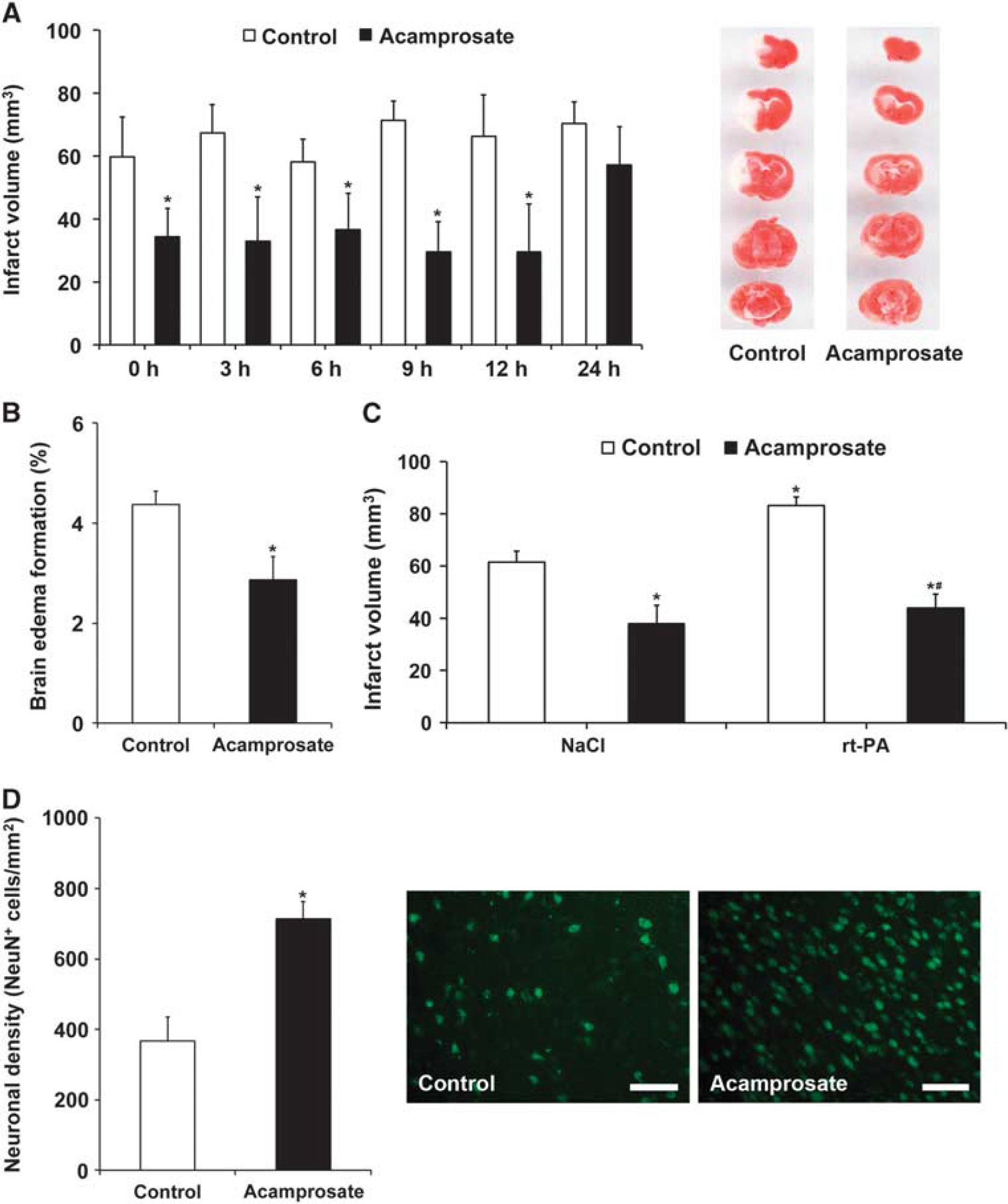
Acamprosate is neuroprotective and ameliorates recombinant tissue plasminogen activator (rt-PA)-induced toxicity. (
The aforementioned histologic effects of acamprosate were further elucidated with respect to functional relevance. Consequently, neurologic recovery was analyzed for an observation period of 3 months (Figure 2). Using the corner turn, rota rod, foot fault, and balance beam test, mice receiving treatment with acamprosate showed significantly enhanced test performance compared with controls. These results suggest that acamprosate induces sustained postischemic neuroprotection and neurologic recovery during an observation period of at least 3 months.
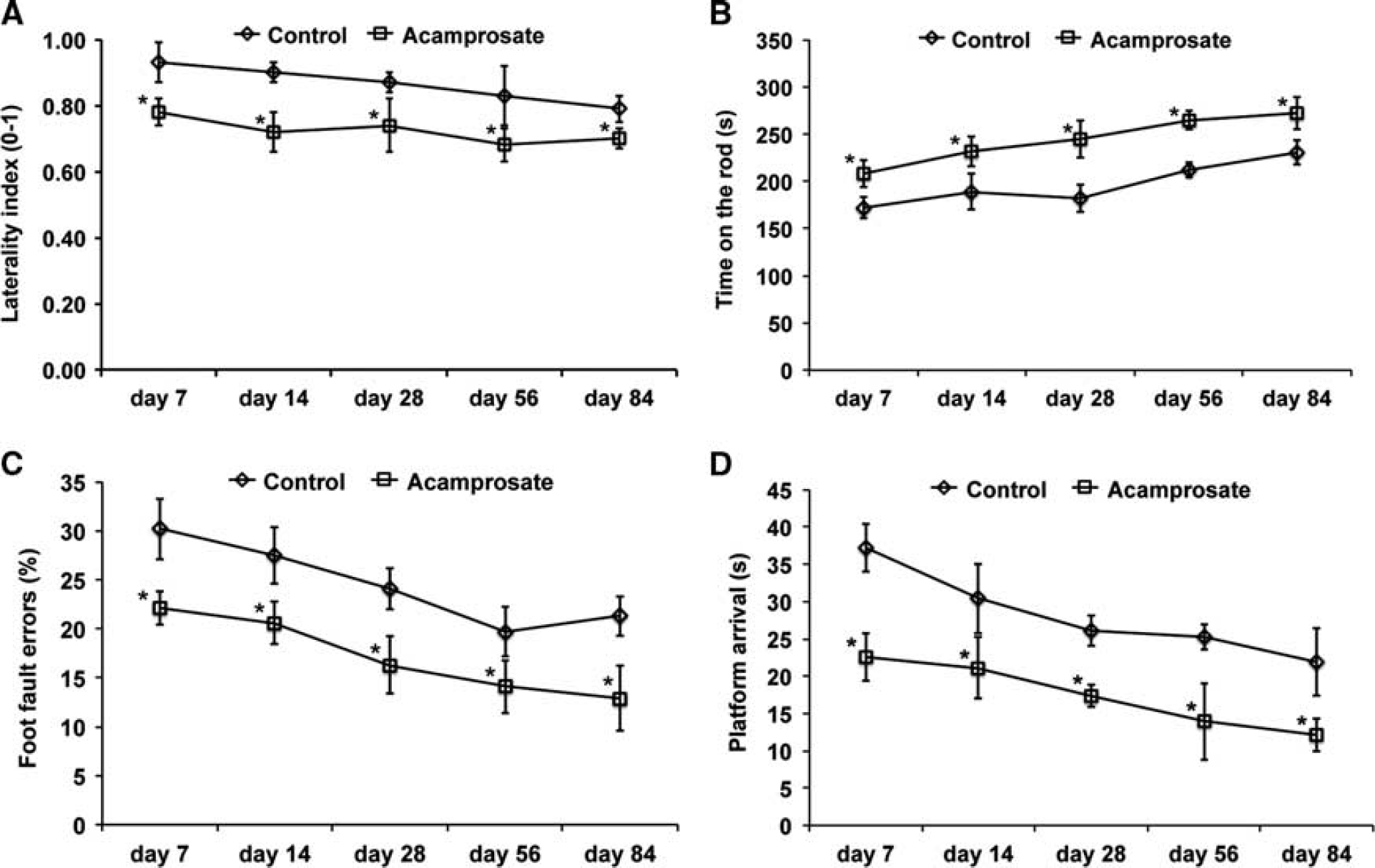
Acamprosate induces neurologic recovery. Mice were treated with either acamprosate (400 mg/kg) or NaCl (control) 12 hours after stroke. Behavioral tests included the corner turn (
Treatment with Acamprosate Results in Increased Postischemic Neuroregeneration
Since acamprosate induces sustained neuroprotection, we wondered whether such a treatment is associated with enhanced postischemic neuroregeneration. Analysis of (formerly) proliferating BrdU+ cells within the lesion site yielded increased numbers of BrdU+ cells in the treatment group compared with controls (Figure 3A), albeit numbers of BrdU+ cells in the subventricular zone itself did not significantly differ between each other (46.1 ±9.7 in controls versus 39.8±12.4 BrdU+ cells per mm2 in the treatment group). Differentiation analysis of BrdU+ cells in the lesion site revealed significantly increased coexpression of the immature neuronal marker doublecortin (Dcx; Figure 3B) and the mature neuronal marker NeuN (Figure 3C), albeit absolute numbers of the latter were low. Since both neurogenesis and angiogenesis mutually affect each other, 29 further analysis of angiogenesis revealed an increase in BrdU+ cells coexpressing the endothelial marker CD31 in mice treated with acamprosate (9.2 ±2.1% compared with 3.4 ±0.7% in controls). Acamprosate, however, did not induce postischemic angioneurogenesis per se. Rather, sustained neuroprotection due to acamprosate treatment results in prevention of secondary cell death of new-born cells. In this context, treatment of sham-operated mice (n = 8 per group) did not increase angioneurogenesis 4 weeks after surgery when compared with controls (data not shown).
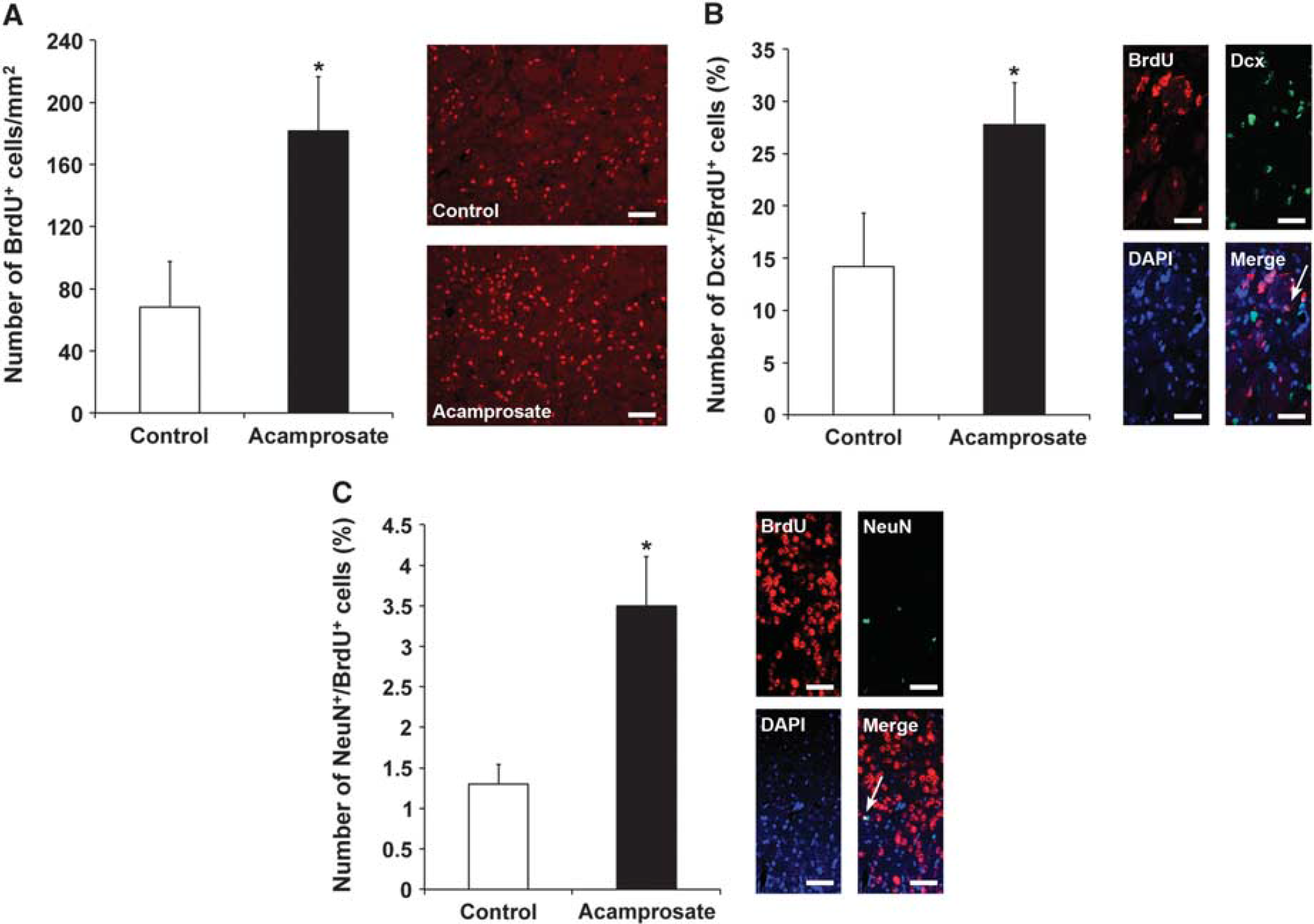
Treatment with acamprosate induces postischemic neuroregeneration. Mice were treated with either acamprosate (400 mg/kg) or NaCl (control) 12 hours after stroke. At 84 days after stroke, the number of bromodeoxyuridine (BrdU+) cells within the lesion site was analyzed in both acamprosate-treated mice and controls (
Acamprosate Stabilizes Blood–Brain Barrier and Reduces
Postischemic Oxidative Stress and Inflammatory Response The aforementioned observations after treatment with acamprosate are not restricted to reduction of infarct volume and brain edema, but have a significant impact on both integrity of the BBB and the extracellular milieu in the lesion site. As such, extravasation of Evans blue 2 days after stroke induction was significantly reduced due to acamprosate treatment (Figure 4A). Likewise, acamprosate diminished oxidative stress and reduced the postischemic inflammatory response in the lesion site when compared with controls (Figures 4B and 4C). On the contrary, effects on systemic peripheral immune responses were not observed after treatment with acamprosate (Figure 4D).
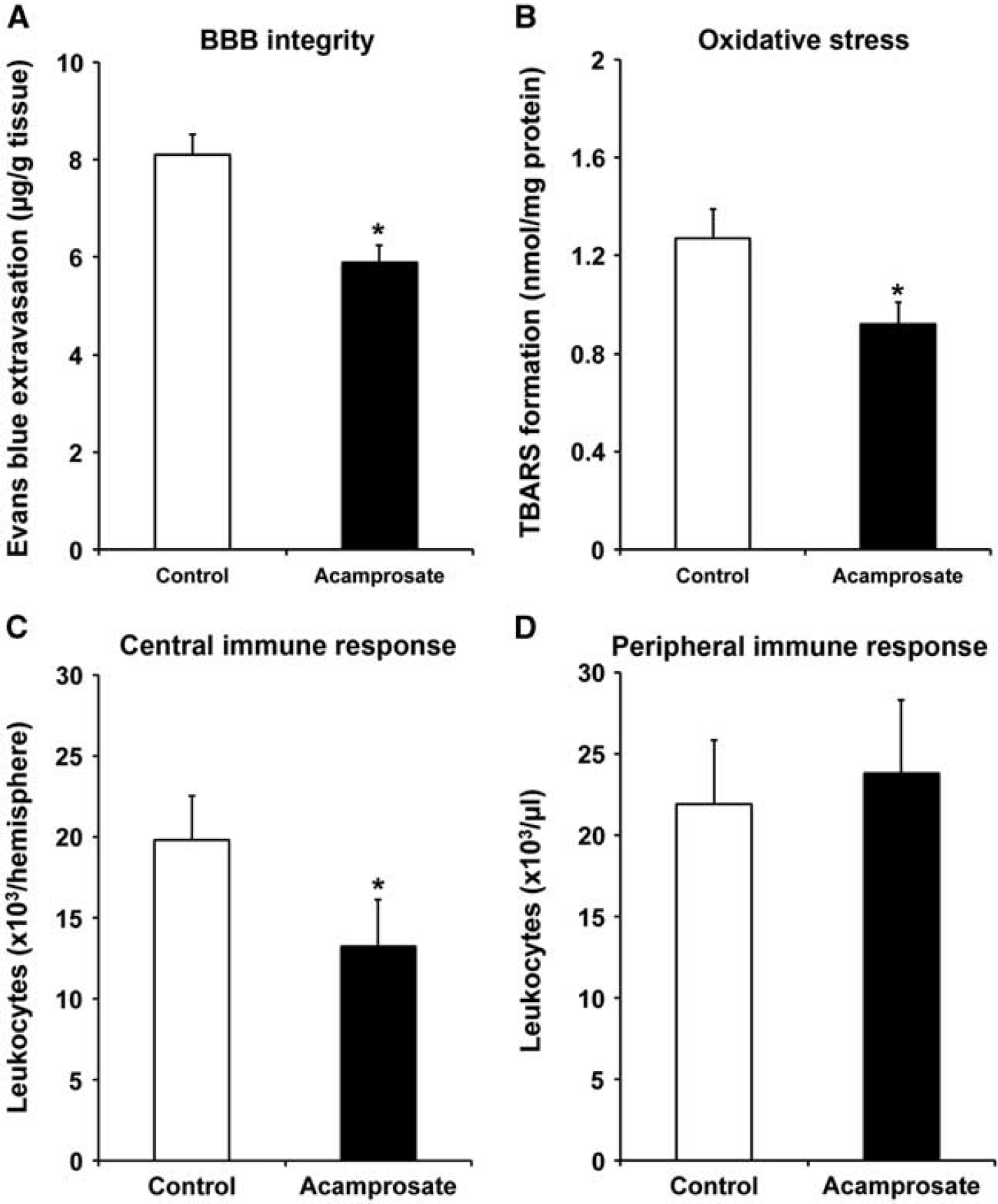
Acamprosate stabilizes blood–brain barrier (BBB) and reduces both oxidative stress and central immune response. Acamprosate (400 mg/kg) was given 12 hours after stroke induction. Controls received standard saline only. Mice were killed 48 hours after stroke induction for measurement of readout parameters. Integrity of BBB was assessed via determination of Evans blue (
Acamprosate Reduces Poststroke Calpain Activity and Modulates Excitotoxic Cell Signaling
Activation of NMDAR induces an increase in intracellular calcium concentration with subsequent activation of calpain and further excitotoxic signaling cascades.1–3 We therefore wondered whether treatment with the indirect NMDAR antagonist acamprosate affected calpain activity and further excitotoxic signaling path-ways. Indeed, calpain activity was significantly reduced 2 days after stroke induction when compared with controls (Figure 5A). Stimulation of NMDAR increases calpain activity and subsequent cleavage of p35 into p25, which is known to induce neuronal cell death. 1 Consequently, treatment with acamprosate resulted in decreased protein cleavage of p35, resulting in enhanced p35 and reduced p25 protein abundance (Figures 5B and 5C).
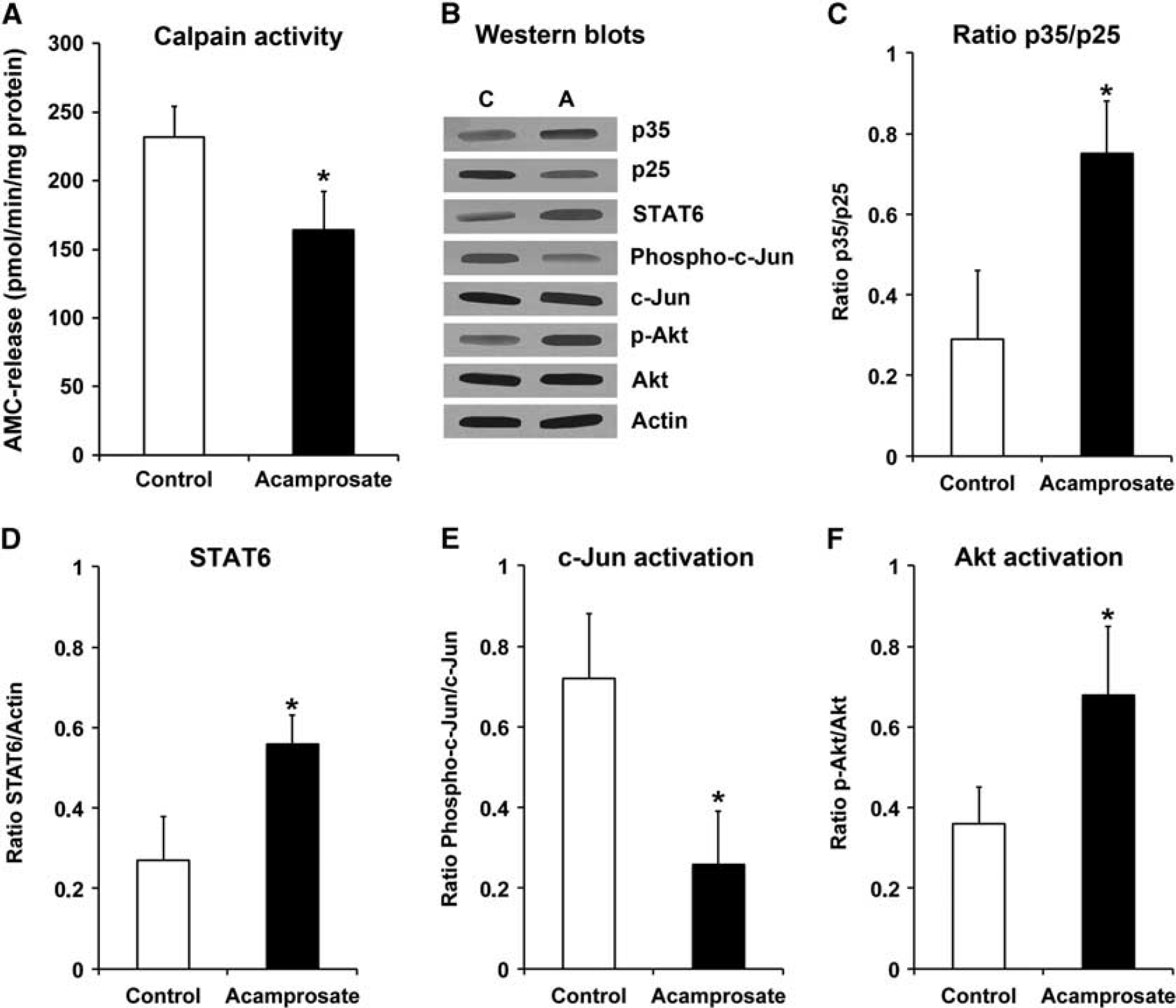
Acamprosate inhibits post-stroke calpain activity and modulates excitotoxic cell signaling pathways. Mice received treatment with either acamprosate (400 mg/kg) or NaCl (control) 12 hours after stroke. Animals were killed 48 hours after stroke induction. (
Analysis of the calpain substrate signal-transducer-and-activator-of-transcription-6 (STAT6) revealed increased protein abundance of STAT6 (Figures 5B and 5D), which was followed by decreased phosphorylation (i.e., activation) of the STAT6 downstream target c-Jun (Figures 5B and 5E). Finally, acamprosate increased the phosphorylation (i.e., activation) of the pro-survival signal Akt (Figures 5B and 5F). These results suggest that acamprosate induces neuroprotection via indirect blockage of NMDAR and prevention of increased intracellular calcium, thus leading to inhibition of calpain-dependent pro-injurious signaling pathways.
DISCUSSION
The present study analyzed the therapeutic potential of acamprosate in a model of transient focal cerebral ischemia. Our study shows that treatment with acamprosate no later than 12 hours upon stroke induction results in sustained neuroprotection and neurologic recovery. Furthermore, acamprosate-induced neuroprotection critically depends on inhibition of calpain-dependent pro-injurious signaling cascades.
Despite recent advances of recanalizing therapeutic strategies,30,31 additional therapies for achievement of sustained neurologic recovery are still in order. Consequently, elucidating mechanisms of cerebral ischemia and application of distinct inhibitors interfering with pro-injurious signaling cascades is mandatory, if such an approach is meant to be successful in future clinical trials. In this context, the use of a clinically approved drug with minor side effects such as the indirect NMDAR inhibitor acamprosate is intriguing. 9 The latter is clinically used for patients with chronic alcohol dependence, which is associated with upregulation of NMDAR and downregulation of γ-aminobutyric acid type A (GABAA), resulting in a hyperglutamatergic state. 32 Treatment with acamprosate, however, is not only limited to inhibition of NMDAR but also involves modulation of GABAA transmission due to inhibition of presynaptic GABAB receptors. 33
Excessive release of glutamate and the aforementioned hyperglutamatergic state is decisive for the development of excitotoxicity during cerebral ischemia. However, interfering with stimulation of NMDAR and subsequent excitotoxic ischemic brain injury remains controversial. Both stimulation and inhibition of NMDAR can induce cell death or cell survival, depending on concentrations of NMDA and NMDAR inhibitors.4,5 Consequently, a more differentiated hypothesis suggests that effects on NMDAR interference depend on the site of the NMDAR to be modified; i.e, stimulation of synaptic NMDA receptors induce cell survival while stimulation of extrasynaptic NMDAR induce cell death. 1
Excitotoxicity results in an increase in intracellular calcium concentrations, and acamprosate is known to inhibit the latter via indirect inhibition of NMDAR. 14 Until recently, only one study used acamprosate in a model of experimental stroke. 16 The study, however, included an observation period of 3 days only where rats received intraperitoneal injections of acamprosate 24 hours and 30 minutes before stroke induction. Given the high mortality of controls (55%) in a model of cerebral ischemia including transient ligation of the common carotid artery and arterial hypotension with blood withdrawal, the authors claim increased survival rates and better neurologic recovery of rats treated with acamprosate. The present study herein describes for the first time neuroprotection against stroke when acamprosate is given in a therapeutic experimental paradigm, i.e., 12 hours after stroke induction. Interestingly, neuroprotection sustained throughout the observation period of 3 months and was associated with both increased neurologic recovery and enhanced neuroregeneration.
Taken into account that cerebral ischemia itself triggers endogenous neurogenesis, 34 treatment with acamprosate in nonischemic sham-operated mice did not result in increased (angio-)neurogenesis, suggesting that acamprosate does not stimulate (angio-)neurogenesis per se. Rather, increased postischemic angioneurogenesis in acamprosate-treated mice is a consequence of acute long-lasting neuroprotection in these mice, although both angiogenesis and neurogenesis might cross-fertilize each other. This is also supported by the fact that treatment with acamprosate does not affect postischemic cell proliferation within the subventricular zone, which itself is not exposed to cerebral ischemia directly. In other words, acamprosate supports survival of new-born cells within an otherwise hostile poststroke microenvironment. These new-born cells might then contribute to neuroregeneration via indirect effects such as constant secretion of trophic factors as has been described before. 27
Acute long-lasting neuroprotection due to treatment with acamprosate results in increased BBB integrity, reduction of infarct volume and brain edema as well as decreased central immune cell infiltration. Importantly, acamprosate reduces rt-PA-induced brain toxicity, again suggesting its potential feasibility under clinical settings. All of these parameters are related to acamprosate-induced inhibition of excitotoxicity with subsequent prevention of an increase of intracellular calcium. The latter is a key factor of NMDA-induced excitotoxicity, among which the activation of calcium-dependent calpains is crucial, resulting in tissue injury and cell death.35,36 As such, reduced calpain activity after treatment with acamprosate results in regulation of calpain substrates, which are involved in cell survival signaling cascades. A direct causal conclusion between acamprosate-induced calpain inhibition and BBB integrity and other aforementioned parameters cannot, however, be drawn from the present data set, nor was it the scope of the study. Reduced poststroke oxidative stress and decreased cerebral leukocyte infiltration are rather most likely secondary events to acute inhibition of excitotoxicity, thus further maintaining neuroprotection by acamprosate. In this sense, enhanced BBB integrity as observed in the study is most likely a secondary event due to acamprosate-induced inhibition of excitotoxicity as well; a direct impact of calpain inhibition on poststroke BBB integrity is unknown to the best of the authors' knowledge.
The methodological approach chosen in the present study by using whole left ischemic hemispheres that contain both nonischemic and ischemic areas do not allow a conclusion as to the extent of contribution of single cell signaling cascades to neuroprotection (or aggravation of poststroke brain injury). Yet, cleavage of p35 as part of cdk5 cell signaling into pro-injurious p25 is significantly reduced by acamprosate. 1 Being a substrate of calpain, 1 reduced p35 cleavage is most likely a direct consequence of acamprosate-induced calpain inhibition, thus preventing neuronal death. Likewise, degradation of the pro-survival factor STAT6,37,38 which is another calpain substrate independent of cdk5 signaling, was reduced. The latter thus results in inhibition of the pro-injurious STAT6-downstream c-Jun N-terminal kinase pathway.39,40 In line with inhibition of pro-injurious cell signaling pathways after acamprosate treatment, phosphorylation (i.e., activation) of the pro-survival pathway Akt was increased. Although calcium-induced activation of Akt signaling upon stimulation (and not inhibition) of the NMDAR itself has been described, 1 it is likely that acamprosate modulates Akt signaling by an independent yet unknown mechanism. Unveiling of the latter was, however, beyond the scope of the present work. Although these aforementioned mechanisms are well-known key players, they might not be exclusive in light of the plethora of mechanisms involved in mediating stroke injury. Nevertheless, it is reasonable to assume that these signaling pathways are at least critically involved in acamprosate-induced neuroprotection that deserve further attention for future preclinical and putative clinical studies alike.
Footnotes
TRD and JRP contributed to development of study design, performance of research, writing of manuscript, financial support. BK and JS contributed to performance of research. EK and MB contributed to writing of manuscript. DKH contributed to development of study design, writing of manuscript, and financial support.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
