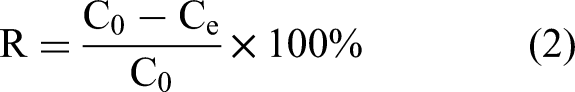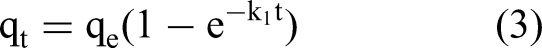Abstract
The reduction and resource recovery of waste incineration fly ash represent a central focus of current research initiatives. In this study, Ca-Al-layered double hydroxides (LDHs) were prepared via a coprecipitation method using waste incineration fly ash as a calcium source combined with aluminum chloride. Their Pb2+ adsorption capacities were compared with commercial LDHs and LDHs synthesized from chemical reagents. Results indicate that the optimal Ca/Al molar ratio for both fly ash-derived LDHs and reagent-synthesized LDHs was 2:1. The Pb2+ adsorption capacities of fly ash-derived LDHs (FCA2), reagent-synthesized LDHs (CA2), and commercial LDHs (CL) were 1149.78 mg·g−1, 1024.00 mg·g−1, and 1069.34 mg·g−1, respectively. After five regeneration cycles, FCA2 maintained stable adsorption performance, with Pb2+ removal efficiency exceeding 95%. The synthesized LDHs were characterized by SEM, XRD, FTIR and BET, revealing that the dominant adsorption mechanisms included ion exchange, surface complexation, and precipitation. This study not only proposes a method for high-value resource utilization of waste incineration fly ash but also provides insights for aquatic heavy metal pollution control and hazardous waste management.
Introduction
The rapid expansion of electroplating, battery manufacturing, metallurgy, and related industries has led to escalating discharges of heavy metal-laden wastewater (Chen et al., 2023; González et al., 2016). Among heavy metals, lead (Pb) has garnered substantial scientific concern owing to its persistent bioaccumulation potential and acute toxicity, with the battery sector identified as a predominant contributor to Pb-containing effluents (Li et al., 2023; Salman et al., 2021). Upon environmental release, Pb can biomagnify through trophic chains, ultimately accumulating in human tissues and inducing multiorgan dysfunction — including detrimental effects on the central nervous, renal, cardiovascular, and immune systems (Fiorito et al., 2022; Naseem and Tahir, 2001). These risks underscore the critical need for advanced remediation technologies to mitigate Pb contamination, thereby safeguarding ecosystem stability and human health.
Current mainstream technologies for Pb-contaminated wastewater treatment encompass chemical precipitation (Zhang et al., 2020), ion exchange (Ahmed et al., 1998), adsorption (Ijaz et al., 2024), membrane filtration (Choudhury et al., 2018), electrochemical treatment (González et al., 2016) and bioremediation (Idrees et al., 2023). Among these, adsorption technology has gained prominence due to its exceptional adsorption capacity, operational simplicity, and regenerability, rendering it a predominant choice in industrial applications.
In recent years, researchers have focused on a variety of low-cost adsorbents derived from solid wastes, including agricultural waste biomass (Asim et al., 2020; Suresh et al., 2023), fly ash (Lin et al., 2023), sludge waste (Nguyen et al., 2021), slag (Li et al., 2022a), gangue (Mei et al., 2023) and tailings (Shi et al., 2024). Among these, fly ash is classified as a hazardous waste. Currently, the primary disposal method for fly ash involves solidification and stabilization followed by landfill (Lan et al., 2024). However, due to the increasing production of fly ash from municipal solid waste incineration (MSWI) and the limited availability of land resources (Lin et al., 2022), resourceful treatment of fly ash has become increasingly necessary. At present, fly ash recycling mainly focuses on low-value applications such as brick production, ceramic granules, and co-processing in cement kilns (Deng et al., 2023; Shen et al., 2024). The preparation of adsorbents from fly ash represents a high-value utilization pathway.
Layered double hydroxides (LDHs) are a class of anionic layered materials featuring tunable lamellar architectures (e.g., layer spacing, metal species composition, cation ratios, and exchangeable interlayer anions) combined with exceptional chemical stability (Yang et al., 2024). These materials have been extensively employed in adsorption technologies due to their excellent properties (Ahmad et al., 2024; Fan et al., 2024; Farhan et al., 2024; Wang et al., 2024b). Waste incineration fly ash, characterized by high calcium and chlorine content, serves as a precursor for LDHs synthesis, offering dual environmental benefits: enhancing the economic value of recycled fly ash products while simultaneously mitigating environmental contamination through heavy metal immobilization, thereby reducing the material's inherent ecotoxicity.
In a related study, Wang et al. (2024a) investigated the synthesis of LDHs derived from fly ash. The researchers successfully fabricated calcium-aluminum LDHs via hydrothermal synthesis under optimized conditions: 20 h reaction time, pH 11, 120 °C temperature, and Ca/Al molar ratio of 2:1. Elemental analysis revealed that the synthesized material contained predominantly chloride, sulfate, and acetate anions in its interlayer structure. Notably, hydrothermal synthesis necessitates higher reaction temperatures and energy consumption compared to conventional coprecipitation methods for LDH fabrication. Furthermore, hydrochloric acid demonstrates superior performance over acetic acid in calcium extraction efficiency, heavy metal sequestration, and avoidance of foreign ion incorporation (Chen et al., 2024). Anion exchange capacity studies (Dong et al., 2022) established the following affinity sequence: CO32−>SO42−>Cl−> Br−>HCOO−>NO3−. This hierarchy explains why chloride-intercalated LDHs exhibit enhanced adsorption performance relative to those containing mixed sulfate-chloride-acetate anion complexes.
This study established an integrated process for LDH synthesis from MSWI fly ash. The protocol involved HCl-assisted acid leaching to extract calcium and associated metallic elements from fly ash, followed by coprecipitation synthesis utilizing calcium and chloride ions from the leachate supplemented with controlled aluminum chloride additions. The synthesized LDHs were systematically characterized through X-ray diffraction (XRD), scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM-EDS), and Fourier-transform infrared spectroscopy (FT-IR). Adsorption performance evaluation demonstrated exceptional Pb2⁺ removal capacity, with subsequent mechanistic analysis elucidating the adsorption kinetics, isothermal behavior, and underlying molecular interactions. Collectively, this work establishes a technological framework for synergistic resource recovery and heavy metal remediation from MSWI fly ash, advancing circular economy principles in hazardous waste management.
Material and methods
Materials
The MSWI fly ash used in this study was sourced from a municipal solid waste-to-energy plant in Shangrao City, Jiangxi Province, China. Calcium chloride (CaCl2), hydrochloric acid (HCl), sodium hydroxide (NaOH), nitric acid (HNO3), zinc sulfate heptahydrate (ZnSO4·7H2O), manganese sulfate monohydrate (MnSO4·H2O), and copper sulfate pentahydrate (CuSO4·5H2O) were procured from Xilong Scientific Co., Ltd (Guangdong, China). Aluminum chloride (AlCl3) and lead nitrate (Pb(NO3)2, (GR) were obtained from Damao Chemical Reagent Factory (Tianjin, China), while cadmium sulfate octahydrate (CdSO4·8/3H2O) was purchased from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). Unless otherwise specified, all reagents were analytical grade and used as received, with deionized water employed throughout the experimental procedures.
Synthesis of Ca-Al-LDHs
Calcium-aluminum layered double hydroxides (CaAl-LDHs) were fabricated through dual synthesis pathways: conventional coprecipitation employing analytical-grade chemical precursors and waste valorization utilizing MSWI fly ash.
Chemical Reagent-Based LDH Synthesis: A stoichiometric coprecipitation approach was employed. First, 44.40 g calcium chloride (CaCl2) was dissolved in 500 mL deionized water under vigorous stirring, followed by dissolution of 13.35 g aluminum chloride (AlCl3) in 500 mL deionized water. The solutions were combined (Ca/Al molar ratio = 4:1) and subjected to continuous magnetic stirring (250 rpm). During agitation, 2 mol·L−1 sodium hydroxide (NaOH) was titrated into the mixture at 3 mL·min−1 until pH ≥ 12. The resultant slurry was aged for 30 min at ambient temperature, vacuum-filtered, washed thrice with deionized water, and dried at 80 °C for 24 h. The product was sieved through 100-mesh (150 μm) screens and designated CA4. Similar procedures yielded CA3 and CA2 materials with Ca/Al ratios of 3:1 and 2:1, respectively.
Fly Ash-Derived LDH Synthesis: The MSWI fly ash acid leaching protocol employed 2 mol·L−1 hydrochloric acid (HCl) as the leaching agent under optimized parameters: 1:5 solid-to-liquid ratio (w/v), ambient temperature 25 ± 1 °C, and 1 h contact duration. Following this, the leachate composition was quantitatively analyzed via ICP-OES to determine Ca/Al ratios, enabling precise supplementation with aluminum chloride (AlCl3) to achieve target molar ratios of 2:1, 3:1, and 4:1. Subsequent LDH synthesis followed identical coprecipitation parameters established for reagent-based synthesis including pH control, aging time, and thermal treatment conditions yielding fly ash derived materials designated FCA2, FCA3, and FCA4.
Preliminary experiment
The adsorption experiments were systematically conducted using CaAl-LDHs with Ca/Al molar ratios of 2:1, 3:1, and 4:1. In a standardized protocol, 0.1 g of adsorbent was precisely weighed and transferred to a 250 mL Erlenmeyer flask, followed by addition of 100 mL Pb(NO3)2 solution (300 mg·L−1 Pb2+). The flasks were then secured in a thermostatic orbital shaker maintained at 25 ± 1 °C with 150 rpm agitation. Upon reaching adsorption equilibrium, 10 mL aliquots were withdrawn and filtered through 0.45 μm nylon membranes, with filtrates analyzed for residual Pb2+ concentration via inductively coupled plasma optical emission spectrometry (ICP-OES). The optimal-performing material was subsequently selected for comprehensive parametric adsorption studies.
The lead ion adsorption capacity and removal rate were calculated by the following equation:
Here, qe (mg·g−1) is the adsorption capacity, R(%) is the removal rate, Ce(mg·L−1) and C0 (mg·L−1) are the residual and initial Pb2+ concentration in the solution, m(g) is the mass of adsorbent material, and V (L) is the volume of lead solution.
Adsorption experiments
Adsorption kinetics experiments
The kinetic profiles of CA2, FCA2, and CL (commercial LDHs) adsorbents were evaluated under controlled conditions. Specifically, 50 mg of each material was dispersed in 100 mL Pb(NO3)2 solution (100 mg·L−1 Pb2+) within 250 mL Erlenmeyer flasks. The initial solution pH was adjusted to 5.5 ± 0.1 using 0.1 M HNO3/NaOH. The reaction systems were agitated in a thermostatic orbital shaker (25 ± 1 °C, 150 rpm) with aliquots sampled at prescribed intervals (5, 30, 60, 120, 180, and 360 min). Following membrane filtration (0.45 μm nylon), residual Pb2+ concentrations were quantified via ICP-OES. Each set of experiments was done in three repetitions and the mean values were calculated. Kinetic modeling was implemented through nonlinear regression analysis using pseudo-first-order (Equation 3), pseudo-second-order (Equation 4) rate equations and intraparticle diffusion model (Equation 5) as described below:
Pseudo-first-order kinetic equation:
Pseudo-second-order kinetic equation:
Intraparticle diffusion model:
In this equation, qt (mg·L−1) means the adsorbed amount at moment t; t (min) is the adsorption time; and k1 and k2 are both constants.
Adsorption isotherm experiments
Equilibrium adsorption studies were performed with CA2, FCA2, and CL adsorbents under systematically varied Pb2+ concentrations. Specifically, 50 mg of each material was immersed in 100 mL Pb(NO3)2 solutions spanning nine concentration gradients (50, 100, 200, 300, 400, 500, 600, 800, 1000 mg·L−1) within 250 mL Erlenmeyer flasks. The initial pH was standardized to 5.5 ± 0.1 using 0.1 M HNO3/NaOH solutions. Reaction systems were equilibrated in a thermostatic orbital shaker (25 ± 1 °C, 150 rpm) until adsorption saturation. Post-equilibrium samples were filtered through 0.45 μm nylon membranes prior to ICP-OES quantification of residual Pb2+ concentrations. Each set of experiments was done in three repetitions and the mean values were calculated. Isotherm modeling was conducted through nonlinear regression analysis employing Langmuir (Equation 6) and Freundlich (Equation 7) isotherm models as mathematically described below:
Langmuir isothermal adsorption equation:
Freundlich isothermal adsorption equation:
In the above equation, qm (mg·g−1) is the maximum adsorption capacity of the adsorbent, KL, KF, and n are all constants.
Effect of initial pH on adsorption
The pH-mediated adsorption performance of CA2, FCA2, and CL adsorbents was systematically investigated. In controlled batch experiments, 50 mg of each material was immersed in 100 mL Pb(NO3)2 solution (100 mg·L−1 Pb2+) contained in 250 mL Erlenmeyer flasks. The initial solution pH was adjusted to 1.5, 2.5, 3.5, 4.5, 5.5, and 6.5 using 0.1 M HNO3/NaOH, with pH ≤ 6.5 selected to prevent Pb(OH)2 precipitation based on solubility equilibrium calculations. Reaction systems were equilibrated in a thermostatic orbital shaker (25 ± 1°C, 150 rpm) until adsorption equilibrium. Post-equilibrium samples were filtered through 0.45 μm nylon membranes for ICP-OES determination of residual Pb2+ concentrations. Each set of experiments was done in three repetitions and the mean values were calculated.
Dosage variation experiment
The adsorbent dosage effect was investigated by dispersing CA2, FCA2, and CL materials in mass gradients of 0.05, 0.1, 0.2, 0.3, and 0.5 g into 250 mL Erlenmeyer flasks. Each flask received 100 mL Pb(NO3)2 solution (100 mg·L−1 Pb2+) with initial pH adjusted to 5.5 ± 0.1 using 0.1 M HNO3/NaOH. The reaction systems were equilibrated in a thermostatic orbital shaker (25 ± 1°C, 150 rpm) until adsorption equilibrium. Post-equilibrium samples were filtered through 0.45 μm nylon membranes prior to ICP-OES quantification of residual Pb2+ concentrations. All experimental trials were conducted in triplicate, with arithmetic means calculated from the replicate data sets.
Effect of co-existing ions
Multi-ion competitive adsorption studies were conducted by immersing 50 mg of CA2, FCA2, and CL materials in 250 mL Erlenmeyer flasks containing 100 mL synthetic wastewater with equimolar concentrations (100 mg·L−1) of Pb2+, Cd2+, Cu2+, Zn2+, and Mn2+. The initial pH was adjusted to 5.5 ± 0.1 using 0.1 M HNO3/NaOH. Reaction systems were equilibrated in a thermostatic orbital shaker (25 ± 1°C, 150 rpm) until adsorption equilibrium. Post-equilibrium samples were filtered through 0.45 μm nylon membranes prior to ICP-OES quantification of residual metal ion concentrations.
Adsorbent regeneration study
To evaluate the reusability of the adsorbents, regeneration experiments were conducted through desorption. Specifically, the saturated adsorbent (100 mg) was transferred to a 250 mL Erlenmeyer flask containing 100 mL 0.2 mol·L−1 NaOH solution. The system was agitated in a thermostatic orbital shaker (25 ± 1°C, 150 rpm) for 30 min to achieve desorption equilibrium. Subsequently, the regenerated material was filtered, washed thoroughly with deionized water, and oven-dried at 80°C for 12 h. The cycled adsorbent was then subjected to repeated Pb2+ adsorption tests under identical initial conditions (100 mg·L−1, pH 5.5). Regeneration efficiency was quantified by monitoring the adsorption capacity retention over five consecutive regeneration cycles.
Characterizations of LDHs
X-ray diffraction (XRD) spectra of the materials before and after adsorption saturation were analyzed by MiniFlex600 from Japan (Cu Kα, λ = 0.15406 nm). The surface ultrastructure of the samples before and after adsorption was characterized by scanning electron microscopy (SEM) and x-ray energy spectrometry (EDS) by Hitachi SU8010 from Japan. Fourier-transform infrared spectroscopy (FTIR) was measured in the range of 4000–400 cm−1 by Nicolet 6700 from Thermo Scientific, USA. The specific surface area and pore size distribution of the samples before and after adsorption were determined by micromeritics ASAP 2460.
Results and discussion
Preliminary experimental analysis
According to HJ 781-2016 standard, fly ash is digested with concentrated hydrochloric acid, concentrated nitric acid, hydrofluoric acid, and perchloric acid, while leachate is digested with concentrated nitric acid. Following digestion on an electric hotplate, ICP-OES analysis is employed to determine metal components in HCl-treated fly ash and its corresponding acid leachate (Table S1). The results demonstrate significant metal migration, with the majority of metallic species transferred to the acid leachate phase. This redistribution provides essential precursors for subsequent LDH synthesis.
Material composition analysis
Quantitative ICP-OES analysis revealed the Ca/Al molar ratios in synthesized LDHs (Table S2). The experimentally determined Ca/Al ratio was markedly lower than the theoretical stoichiometric value. This may be attributed to the relatively brief ageing period of the hydrated hydrotalcite, coupled with substantial leaching of calcium and aluminium salts during the three-stage washing process. Nevertheless, the final composite material remains within the characteristic range of a stable layered dihydroxide structure (The molar ratio of Al3⁺/Ca2⁺+Al3⁺ is 0.17–0.33). Heavy metal incorporation levels in fly ash-derived LDHs were systematically quantified (Table S3). X-ray diffraction analysis (Figure S1) confirmed successful formation of Ca-Al-LDH crystalline phases, with FCA2-4 exhibiting enhanced structural heterogeneity attributable to impurity metal co-precipitation from the fly ash matrix.
Adsorption performance comparison
The influence of Ca/Al molar ratios on Pb2+ adsorption capacity was systematically evaluated (Figure 1). However, due to significant discrepancies between the molar ratios of the synthesised materials and theoretical values, the actual molar ratios across the three material groups differed only marginally. Consequently, the final Pb2⁺ adsorption efficiencies proved comparable. Nevertheless, CA2 and FCA2 demonstrated superior synthesis yields relative to the other two groups, whilst the hydrotalcite adsorption efficiencies from all fly ash-derived groups exhibited greater stability. This equilibrium between yield and performance prompted the selection of CA2 and FCA2 for subsequent parametric adsorption studies, ensuring experimental reproducibility while maintaining high adsorption efficacy.

Effect of different Ca to Al molar ratio on the Pb2+ adsorption capacity of the materials.
Material characterization analysis
X-ray diffraction
The XRD spectra of CA2, FCA2, and commercial LDH (CL) are presented in Figure 2(a). CA2 and FCA2 exhibit nearly identical peaks, and FCA2 exhibits higher peak intensities, which are attributed to trace metal impurities inherited from fly ash precursors. Both materials exhibit characteristic LDH structural features, with their XRD patterns demonstrating close alignment to the reference diffraction profiles PDF#35-0105 and PDF#31-0245 from the International Centre for Diffraction Data (ICDD) database. Further XRD analysis was conducted on the synthesised hydrotalcite materials, with Table S4 presenting the lattice parameters for each group. The synthesised hydrotalcite exhibits a hexagonal crystal system, with lattice parameters being closely aligned across all groups. The impurities present in the structures of FCA2, FCA3, and FCA4 exerted negligible influence on the lattice constants. CL also has diffraction peaks typical of LDHs, consistent with the profile recorded on PDF#42-0065 in the database of ICDD, and its anion is predominantly sulfate. Although the anions in our synthesized material differ from those in the commercial hydrotalcite, the adsorption mechanism (Bai et al., 2025) indicates that the primary influence of different anions lies in their direct precipitation reaction with lead. Comparing the solubility product constants of lead chloride and lead sulfate—Ksp(PbCl2) = 1.7 × 10⁻5 and Ksp(PbSO₄) = 2.53 × 10⁻8—lead sulfate precipitates more readily. Therefore, the adsorption performance of the synthesized material, when compared with commercial hydrotalcite, better demonstrates its superior performance. XRD analysis indicates that both fly ash and chemical reagents successfully synthesized Ca-Al-LDHs. In addition, the characteristic diffraction peaks at 11°, 22° and 32° with narrow half peak widths and sharp peak shapes, indicating that our synthesized LDHs have good crystallinity(Liu et al., 2024).

(a) XRD of CA2, FCA2 and CL; (b) FTIR of CA2, FCA2 and CL.
FTIR analysis
The FTIR spectra of CA2, FCA2 and CL are shown in Figure 2(b). Broad absorption peaks at 3477 cm−1 and 3639 cm−1 are attributed to the O-H stretching vibrations of hydroxyl groups (Qiu et al., 2022)., while weaker features near 2922 cm−1 arise from C-O stretching modes and hydrogen-bonded interlayer water molecules (Li et al., 2022b). Adsorbed water deformation vibrations manifest at 1620 cm−1, accompanied by a prominent CO32− symmetric stretching band at 1479 cm−1 indicative of atmospheric CO2 absorption during coprecipitation (Qiu et al., 2022). Metal-hydroxyl (M-OH) stretching modes appear at 861 cm−1 (Xie et al., 2024), with lattice vibrations from M-O bonds spanning 426–795 cm−1 (Qiu et al., 2023). Notably, a distinct sulfate (SO42−) stretching mode at 1117 cm−1(Xu et al., 2023) confirms anion diversity in CL. No distinct chloride ion peak was identified in the synthetic sample, though the peak at 3639 cm⁻1 may reflect the interaction between Cl⁻ and OH groups. A review of multiple literature sources (Kong et al., 2021; Qiu et al., 2022; Wang et al., 2024a) confirms that this characteristic is present across numerous CaAl-Cl-LDH systems. This spectral profile collectively verifies successful LDH synthesis while elucidating environmental interactions during material fabrication.
Scanning electron microscopy (SEM-EDS) analysis
The SEM-EDS characterization of CA2, FCA2, and commercial LDH (CL) is presented in Figures 3-1, 3-2, 3-3. CA2 and FCA2 exhibit irregularly stacked lamellar architectures characteristic of Ca-Al-LDHs, with CA2 demonstrating enhanced structural regularity compared to FCA2 (Figure S2), which may be due to metallic impurity interference in the fly ash-derived FCA2. In contrast, CL displays well-defined rod-like layered morphology, suggesting anion-dependent crystallization behavior where sulfate intercalation promotes longitudinal growth. Elemental mapping analysis reveals homogeneous distribution of Ca, Al, Cl, and S across all LDHs, with chloride ions (Cl−) predominantly occupying interlayer galleries in CA2/FCA2, while sulfate (SO42−) dominates in CL consistent with XRD and FTIR observations. This spatial elemental homogeneity confirms effective structural integration of target components during synthesis.

SEM image and EDS analysis of CA2 (a) (b) (c).

SEM image and EDS analysis of FCA2 (d) (e) (f).

SEM image and EDS analysis of Cl (g) (h) (i).
N2 adsorption–desorption experiments
Figure 4 showed the N2 adsorption/desorption isotherms and corresponding pore size distribution curves of CA2, FCA2 and CL. The specific surface area, average pore size and total pore volume are shown in Table S10. Based on Figure 4(a), the N2 adsorption/desorption isotherms of all three groups of adsorbents exhibited typical H₃ hysteresis loops conforming to the International Union of Pure and Applied Chemistry (IUPAC) classification criteria. Their characteristic curves were all of Type IV, indicating the presence of mesoporous structures in all materials. The specific surface areas of CA2, FCA2 and CL were 11.3855, 15.5793 and 7.6734 m2·g⁻1, their average pore diameter were 20.5738, 24.9478 and 13.2252 nm. The average pore size distribution of the samples further confirms their mesoporous nature. Among the three materials, FCA2 exhibits the largest specific surface area while maintaining a high pore volume.

N2 adsorption/desorption isotherms (a) and corresponding pore size distribution curves (b) of CA2, FCA2 and CL.
Adsorption experiment results and discussion
Adsorption kinetics
The results of the kinetic fitting are shown in the Figure 5(a)-(f) (Table S5 and S6). All materials exhibited rapid adsorption kinetics, achieving >90% of total capacity within 60 min before progressing to equilibrium attainment within 120 min. At initial Pb2+ concentration of 100 mg·L−1, maximum adsorption capacities reached 193.31 mg·g−1 (CA2), 194.21 mg·g−1 (FCA2), and 191.77 mg·g−1 (CL) demonstrating comparable performance across synthetic and commercial LDHs. Nonlinear regression analysis revealed dual-mechanism adsorption behavior, with both pseudo-first-order and pseudo-second-order models showing strong correlation (R2 > 0.99) to experimental data. The close agreement between modeled (qe,cal) and experimental (qe,exp) equilibrium capacities suggests synergistic control by surface diffusion and chemical complexation mechanisms (Yu et al., 2021). This kinetic duality confirms the hybrid physical-chemical nature of Pb2+ sequestration in LDH systems.The intraparticle diffusion model fitted to the adsorption process of lead ions on three material groups (Figure 5(a)-(f), Table S6) can be divided into two stages. The first stage represents the rapid external diffusion process of Pb2⁺ from the solution towards the material surface, while the second stage constitutes a progressive adsorption process, indicating that Pb2⁺ begins to enter the pores of the adsorbent (Yu et al., 2023). The adsorption rate is governed by intra-particle diffusion. Furthermore, the non-zero C value in Table S6 indicates that the adsorption process involves mechanisms beyond particle-internal diffusion.

Adsorption kinetics of Pb2+, (a,d) CA2, (b,e) FCA2, (c,f) CL; adsorption isotherms of Pb2+, (g) CA2, (h) FCA2, (i) CL.
Adsorption isotherm
The equilibrium adsorption behavior of Pb2+ on CA2, FCA2, and CL was investigated through isothermal studies (Figure 5(g)-(i), Table S7). Nonlinear regression analysis using Langmuir and Freundlich models revealed superior correlation (R2 > 0.97) for the Langmuir model, indicating monolayer adsorption dominance over multilayer mechanisms (Freundlich R2 > 0.80). The Langmuir-predicted maximum capacities of 1135.33 mg·g−1 (CA2), 1151.56 mg·g−1 (FCA2), and 1165.95 mg·g−1 (CL) closely matched experimental values (1024.00mg·g−1, 1149.78mg·g−1 and 1069.34 mg·g−1), confirming chemisorption-controlled monolayer coverage (Yu et al., 2023). Comparative analysis with literature reports (Table S8) demonstrated exceptional Pb2+ uptake performance of fly ash-derived LDHs, surpassing conventional synthesized counterparts in adsorption capacity. Post-adsorption ICP-OES analysis (Table S9) confirmed negligible metal leaching (<0.5 mg·L−1), ensuring environmental safety without secondary contamination risks. This comprehensive evaluation positions fly ash-derived LDHs as high-performance heavy metal adsorbents with dual advantages of superior capacity and ecological compatibility.
Effect of initial solution pH
The pH-mediated adsorption performance of LDHs toward Pb2+ was systematically evaluated Figure 6(a). Given the pH-sensitive speciation of lead ions and precipitation risks at pH >7, experiments were conducted within the acidic range (1.5–6.5). Minimum adsorption capacity (<100 mg·g−1) occurred at pH 1.5, attributable to protonation-induced surface charge reversal causing electrostatic repulsion of Pb2+ (Feng et al., 2020). and partial LDH dissolution under extreme acidity (Mahmoudi et al., 2022). Adsorption efficiency increased sharply to peak values (>190 mg·g−1) at pH 2.5, maintaining stability through pH 3.5–6.5. This abrupt enhancement correlates with inhibited Pb2+ hydrolysis and optimized surface complexation kinetics in mildly acidic conditions. The robust pH tolerance demonstrated by fly ash-derived LDHs within environmentally relevant pH ranges validates their operational reliability for practical wastewater treatment.

Effect of pH on LDHs adsorption of Pb2+ and final pH (a), analysis of the point of zero charge for CA2, FCA2, and CL(b).
The inverse correlation between equilibrium pH and Pb2+ adsorption capacity (Figure S3). During adsorption, NO3− ions from Pb(NO3)2 participate in ion exchange with interlayer OH− groups in LDHs, releasing hydroxyl ions that transiently elevate solution pH. Simultaneously, Pb2+ sequestration through hydroxide precipitation (Pb(OH)2 formation) consumes free OH− ions via stoichiometric complexation. The net hydroxide depletion from these competing pathways dominates at higher adsorption levels, inducing progressive solution acidification that inversely correlates with Pb2+ uptake efficiency. Consistent pH depression patterns across diverse experimental conditions confirm this dual-mechanism regulation
The surface charge properties of the synthesized materials (CA2, FCA2, and CL) were evaluated by determining their Points of Zero Charge (PZC). The PZC values were determined using the pH drift method (Alnasrawi and Mohammed, 2023). Figure 6(b) shows the zero charge point (PZC) plots for materials CA2, FCA2, and CL. The plots indicate the zero charge point values, which correspond to the intersections of the curves with the x-axis (dashed line). These results indicate that all three materials possess strongly basic surfaces. The high PZC values suggest that the surfaces are positively charged across a wide pH range. This implies that these materials would preferentially interact with anionic species in solution under neutral to moderately alkaline conditions. However, the effective removal of pb2+ observed in our experiments suggests that the adsorption mechanism is not primarily governed by electrostatic attraction. The principal mechanisms likely include: surface complexation/coordination,ion Exchange and precipitation(Mureseanu et al., 2022; Zhao et al., 2011).
Dosage variation experiment
The influence of adsorbent dosage on Pb2+ removal efficiency is illustrated in Figure 7(a). FCA2 and CL exhibited gradual enhancement in Pb2+ uptake with increasing dosage (0.5–5 g·L−1), achieving >95% removal efficiency at 0.5 g·L−1. In contrast, CA2 demonstrated dosage-independent performance beyond 0.5 g·L−1, indicating rapid adsorption saturation at lower dosage thresholds. This divergence suggests distinct active site, fly ash-derived LDHs maintain available binding sites across wider dosage ranges compared to reagent-synthesized counterparts. The observed saturation behavior at 0.5 g·L−1 dosage implies optimized operational parameters for cost-effective wastewater treatment applications.

Effect of dosage on the adsorption of pb2+ (a), the effect of coexisting ions on the adsorption of pb2+ (b), LDHs adsorption lead regeneration (c).
Effect of co-existing ions
The anti-interference capability and ionic selectivity of fly ash-derived LDHs were evaluated through multi-ion competitive adsorption studies (Figure 7(b)). Under control conditions (CK: Pb2+-only system), FCA2 maintained exceptional Pb2+ removal efficiency (>90%) even in complex matrices containing Zn2+, Cu2+, Mn2+, and Cd2+. Notably, FCA2 exhibited significantly reduced affinity toward Zn2+ and Cu2+ compared to CA2 and CL, this phenomenon attributable to inherent zinc and copper enrichment in fly ash precursors (Table S3) that saturates exchange sites through isomorphic substitution. This site pre-occupation mechanism selectively inhibits competing cation adsorption while preserving Pb2+ sequestration capacity, demonstrating unparalleled heavy metal specificity under realistic wastewater conditions.
Adsorbent regeneration study
The reusability of CA2, FCA2, and CL adsorbents was systematically assessed through five successive adsorption-desorption cycles (Figure 7(c)). All materials demonstrated exceptional regeneration stability, maintaining Pb2+ removal efficiencies >95% throughout cyclic operations. This sustained performance highlights robust structural integrity and preserved active site functionality after repeated alkaline regeneration (0.2 M NaOH). The <5% capacity attenuation observed across cycles validates the economic viability of fly ash-derived LDHs for long-term wastewater remediation applications.
Adsorption mechanism
Heavy metal sequestration by LDHs is governed by multifaceted mechanisms including isomorphic substitution, hydroxide precipitation, surface complexation, interlayer anion exchange, and coordination bonding. To delineate the dominant pathways, pre-adsorption and post-adsorption samples were subjected to advanced characterization through scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), X-ray diffraction (XRD) and N2 adsorption/desorption (BET). These techniques collectively revealed structural modifications, functional group interactions, and crystallographic changes arising from Pb2+-LDH interfacial reactions, enabling comprehensive mechanistic interpretation at molecular to macroscopic scales.
CA2 maintained its lamellar LDH architecture with characteristic hexagonal platelet morphology post-adsorption, exhibiting lamellar thickening and sporadic surface nanoparticulate deposition.
Comparative SEM analysis of CA2 and FCA2 before/after Pb2+ adsorption (Figure 8(a)-(d)) revealed distinct structural modifications. CA2 maintained its lamellar LDH architecture with characteristic hexagonal platelet morphology post-adsorption, exhibiting lamellar thickening and sporadic surface nanoparticulate deposition. In contrast, FCA2 adsorption of the lamellae turned into an irregular loose stacking structure with a small number of small particles stacked on top of each other. Complementary EDS mapping (Figure S4) confirmed Pb-enriched zones co-localized with these surface features, verifying successful Pb2+ immobilization through surface precipitation and interlayer confinement mechanisms.

SEM images of CA2 (a-b) and FCA2 (c-d) before and after adsorption.
The XRD patterns pre- and post-Pb2⁺ adsorption (Figure 9(a)-(b)) demonstrate preserved Ca-Al-LDH crystallinity, with characteristic reflections maintaining angular positions . attenuation in peak intensity post-adsorption suggests surface-mediated Pb2⁺ complexation inducing localized lattice strain without structural collapse (Yu et al., 2023). Consistent d-spacing (Table S4) indicating preferential Pb2⁺ adsorption at surface sites rather than interlamellar regions. This crystallographic stability correlates with SEM-observed surface particulates (Figure 6), collectively verifying LDHs’ dual adsorption mechanism of surface chemisorption and interlayer confinement while maintaining framework integrity.

The XRD patterns of CA2 and FCA2 before and after adsorption (a-b), the FTIR patterns of CA2 and FCA2 before and after adsorption (c-d).
The Fourier transform infrared spectrum (Figure 9(c)-(d)) revealed critical functional group interactions during Pb2⁺ sequestration. The attenuated O-H stretching band at 3400–3600 cm⁻1 indicates hydroxyl group participation through surface complexation and hydroxide precipitation (Wang et al., 2024b), this can also consistent with observed pH depression during adsorption equilibration. As described in Section 3.2.2, the peak at 3639 cm⁻1 may reflect interactions between Cl⁻ and OH groups. The attenuation of this peak post-adsorption could also result from interlayer Cl⁻ anions reacting with Pb2⁺ in solution. Furthermore, the discussion on PZC in Section 3.3.3 also suggests that the adsorption mechanism may involve precipitation. Concomitant redshift of the carbonate vibration from 1479 cm⁻1 to 1454 cm⁻1, coupled with intensity reduction, confirms CO₃2⁻ ligand exchange with Pb2⁺ species (Khalil et al., 2023). Simultaneous attenuation of M-OH (877 cm⁻1) and M-O lattice vibrations (426–795 cm⁻1) provides direct spectroscopic evidence of metal-oxygen bond reorganization during chemisorption. These spectral signatures collectively validate multifaceted adsorption mechanisms governing heavy metal immobilization.
Figure 10 presented the N2 adsorption/desorption isotherms and corresponding pore size distribution curves for CA2, FCA2, and CL before and after adsorption. Desorption isotherms and their corresponding pore size distribution curves. Combined with Table S10, it can be observed that both the specific surface area and pore volume of the materials increased after adsorption. The specific surface areas of CA2, FCA2, and CL increased from 11.3855, 15.5793, and 7.6734 m2/g to 13.2014, 33.9111, and 10.5476 m2/g, while their pore volumes increased from 0.044292, 0.067809 and 0.022175 cm3/g to 0.046129, 0.099942 and 0.033154 cm3/g, with the most pronounced increase observed in FCA2. This may be attributed to the deposition of lead ions on the material surface and newly developed pores through chemical reactions such as ion exchange and surface complexation. The decrease in average pore size for CA2 and FCA2 after adsorption may result from the smaller dimensions of the new pores created by the adsorption reaction, The adsorption process not only captures lead ions but also generates additional adsorption sites within the material matrix.
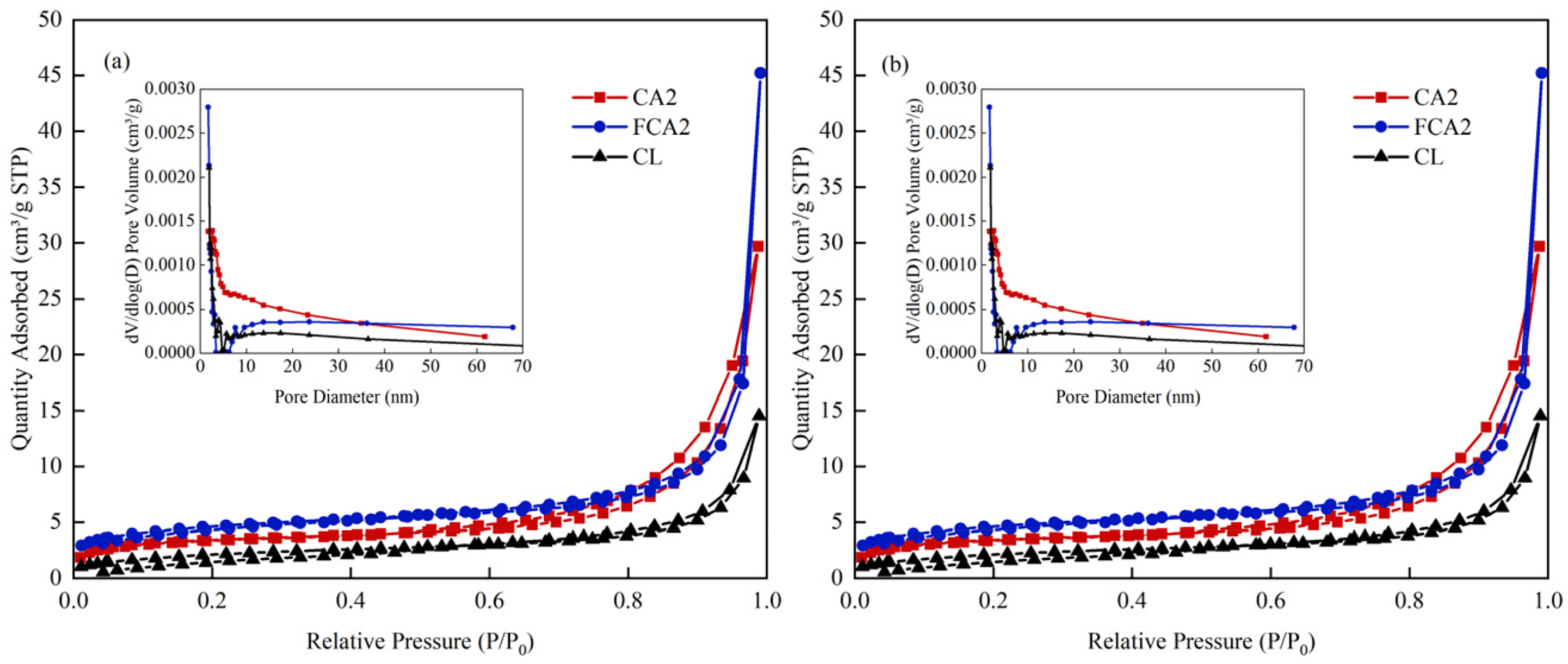
N2 adsorption/desorption isotherms and corresponding pore size distribution curves for Ca2, FCA2, and cl prior to adsorption (a) and after adsorption (b).
The Pb2⁺ sequestration process by fly ash-derived Ca-Al-LDHs operates through a synergistic interplay of ion exchange, surface complexation, and precipitation mechanisms (Figure 11). Interlayer Ca2⁺/Al3⁺ cations dynamically exchange with Pb2⁺ to maintain charge balance, while surface hydroxyl and carbonate groups coordinate Pb2⁺ via stable inner-sphere complexes, a process corroborated by FTIR-observed bond reorganization and XRD-confirmed structural retention. Concurrently, pH-dependent hydrolysis induces Pb(OH)2 precipitation, with carbonate coprecipitation further immobilizing Pb2⁺ as PbCO₃ crystallites on LDH surfaces, morphologically evidenced by SEM-identified surface particulates (Figure 8).This tripartite mechanism explains the material's pH-responsive adsorption maxima while maintaining crystalline integrity (Figure 9). This multifactorial adsorption paradigm explains the material's exceptional Pb2⁺ affinity and pH-responsive behavior observed in batch experiments.

The adsorption mechanisms of Pb2+ by LDHs.
Conclusion
This study established a closed-loop process utilizing HCl-mediated metal extraction from waste incineration fly ash followed by coprecipitation synthesis of Ca-Al LDHs with chloride intercalation. Comparative analysis with reagent-derived and commercial LDHs revealed superior Pb2⁺ adsorption capacity for fly ash-derived LDHs (1149.78 mg·g⁻1) versus conventional materials (1024.00–1069.34 mg·g⁻1). Crucially, the fly ash-LDH system exhibited negligible leaching of competing metal ions (<0.5 mg·L⁻1) during operation, mitigating secondary contamination risks. Multimodal characterization (SEM/XRD/FTIR) coupled with kinetic/isotherm modeling elucidated tripartite sequestration mechanisms: 1) Interlayer Ca2⁺/Al3⁺ and Pb2⁺ exchange maintaining charge balance; 2) Surface complexation through hydroxyl/carbonate coordination; 3) pH-dependent precipitation of Pb(OH)2/PbCO₃/PbCl2 phases. Post-adsorption structural retention enables direct material reuse.
This dual waste-to-resource strategy simultaneously addresses hazardous fly ash management and high-performance adsorbent development, exhibiting 30–40% cost reduction compared to conventional LDH synthesis while achieving 95% Pb2⁺ recovery efficiency across five regeneration cycles. The preserved LDH crystallinity post-contaminant loading further enables functionalized reuse in construction materials or agricultural amendments, pioneering circular economy applications for toxic industrial byproducts.
Supplemental Material
sj-docx-1-adt-10.1177_02636174251409011 - Supplemental material for Adsorption performance of calcium-aluminum layered double hydroxides synthesized from waste incineration fly ash for removal Pb(II) from water
Supplemental material, sj-docx-1-adt-10.1177_02636174251409011 for Adsorption performance of calcium-aluminum layered double hydroxides synthesized from waste incineration fly ash for removal Pb(II) from water by Zhou Zhang, Xilin Chai, Haiying Wei, Yubing Duan, Liqin Xu, Zhihui Yang and Runhua Chen in Adsorption Science & Technology
Footnotes
Acknowledgements
This work was supported by the Science and Technology Research Project of the Education Department of Jiangxi Province, China (GJJ201719), the Project of Shangrao City Science and Technology Bureau (2020I002, 2021F007).
CRediT authorship contribution statement
Zhou Zhang: Conceptualization, Data curation, Writing – original draft, Funding acquisition.
Xilin Chai: Supervision, Conceptualization.
Haiying Wei: Methodology.
Yubing Duan: Methodology.
Liqin Xu: Investigation, Visualization, Writing – review & editing.
Zhihui Yang: Supervision.
Runhua Chen: Conceptualization.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.