Abstract
In this work, lignosulfonate intercalated Mg2Al layered double hydroxide was fabricated by coprecipitation method, which was used as a functional adsorbent for removing pyridine from wastewater. The X-ray powder diffraction and Fourier transformed infrared were carried out to investigate the structure of the product. In the removal process, the as-prepared lignosulfonate intercalated Mg2Al layered double hydroxide sample exhibited good adsolubilization property for pyridine, with maximum capacity of 400.8 mg g−1 in initial pyridine concentration of 400 mg/l and the removal percentage achieved about 87.9%. In addition, the influence of pH, time, and initial concentration of pyridine on the adsorption capacity was also examined. Moreover, the adsorption kinetic followed the pseudo-second-order model. Furthermore, after regeneration, the adsorbent can still show high adsorption capacity even for 10 cycles of desorption–adsorption. It hoped that lignosulfonate intercalated Mg2Al layered double hydroxide can be used as adsorbent for the removal of organic pollutants in wastewater.
Introduction
Pyridine, as one class of the most important nitrogenous heterocyclic compounds (Bai et al., 2011; Mohan et al., 2004; Padoley et al., 2008), has been widely used as raw material in petrochemical, pharmaceutical, antiseptic, paint, pesticide, and some other organic chemical industries (Lataye et al., 2006; Sabah and Celik, 2002). Then, a large amount of wastewater containing pyridine was also produced as by-product. However, the release of pyridine into the environment is very toxic for aquatic organisms and human itself, because it can cause diseases of heart, respiratory tract, and nervous system and even lead to teratogenic and carcinogenic effects. Thus, the removal of pyridine from wastewater is an important research topic worthy of detailed study. At present, many researchers have devoted themselves to develop effective and economical methods to deal with the wastewater containing pyridine and the common methods used for the removal of pyridine from wastewater mainly including photocatalysis (Zhao et al., 2004), membrane separation (Mandal and Bhattacharya, 2006), biological degradation (Lee et al., 1994; Rhee et al., 1996; Sims and Sommers, 1986), ozonation (Andreozzi et al., 1991), and so on. However, these methods often involve complex process, high cost, and even may lead to secondary pollution during removal process. Compared with these methods, adsorption is a preferential choice because of its high efficiency, easy operation, and reversibility for treating and purifying wastewater and widely applied to remove organic compounds from water effluents (Liao et al., 2013; Sabah and Celik, 2002). Many materials such as metal oxides, silica, mesoporous carbons, sepiolite, and montmorillonite (Khajeh et al., 2013) have been used as adsorbents for the wastewater treatment.
The capacity for pyridine during adsorption process was considerably dependent upon many factors, such as mesoporous structure, surface area, pore size with its distribution, mechanical stability, and surface chemical properties. Furthermore, pyridine is an organic compound; according to the principle of similarity, it should more easily dissolve in organic system. However, it is hard to find a suitable organic adsorbent with the properties of large surface area, abundant of mesoporous and mechanical stability. Based on the above, it is a good choice to synthesize an inorganic–organic hybrid that can be used for the adsolubilization of pyridine, which combination of the inorganic and organic advantages.
Layered double hydroxides (LDHs), also called hydrotalcite-like compounds or anionic clays, have been extensively investigated and applied in anionic exchange, catalyst, polymerization, photochemistry, and electrochemistry (Carpani et al., 2004; Hulea et al., 2006; Ma et al., 2010; Malherbe et al., 1998; Toraishi et al., 2002), owing to their special chemical and physical properties. The LDHs are composed of positively charged brucite-like layers with interlayer region containing charge compensating anions and solvation molecules, then, the generic formula for the LDHs can be written as [M2+1−xM3+x (OH)2]x+(An−)x/n·mH2O, where M2+ and M3+ take up the octahedral holes in a hydrotalcite-like layer, An− are situated in the hydrated interlayer galleries and gallery anions (or interlayer anions) of charge n, x is the molar ratio of [M2+]/([M2+] + [M3+]), and m is the molar amount of intercalated water (Evans and Slade, 2006). LDHs have a large amount of splendid properties, such as memory effect, interlayer anion exchange capacity, large surface area, thermal stability, and so on. It is worth noting that the intrinsic hydrophilic surface property of the LDHs can be changed into hydrophobic by inserting a variety of anionic species, especially organic anions, into the interlayer region assembling organic LDHs. Moreover, the interlayer region of LDHs can be enlargement by intercalated large surfactant anions into the interlayer space, resulting in an increasing adsorption capacity (Babakhani et al., 2014; Chuang et al., 2008; Cursino et al., 2013; Lei et al., 2014; Mohanambe and Vasudevan, 2004; Moyo et al., 2011; Naik and Vasudevan, 2011; You et al., 2002; Zaghouane-Boudiaf et al., 2011). Furthermore, the adsorbate of hydrophobic organic is dissolved in the three-dimensional hydrophobic organic phase, rather than adsorbed on the outside surface of LDHs. This adsorption process is intituled “adsolubilization” (Kunio and Syuji, 1998). Lignosulfonate (LS), or lignin, is a natural molecule and the main by-product of the paper industry with low cost, usually used as corrosion inhibitor and dispersing agent of phenolic resins (Huang et al., 2015). There is a strong affinity between LS and the aromatic compound pyridine because there are two aromatic benzene rings in the LS molecule (see Scheme 1), but it has certain solubility in water. At the same time, LS can be intercalated into the interlayer of LDHs because it is a kind of anionic surfactant. Therefore, LS intercalated LDH is used for adsolubilization of pyridine, which can solve the problems such as the adsorption capacity of pyridine and the solubility of LS.
The structural formula of lignosulfonate building unit.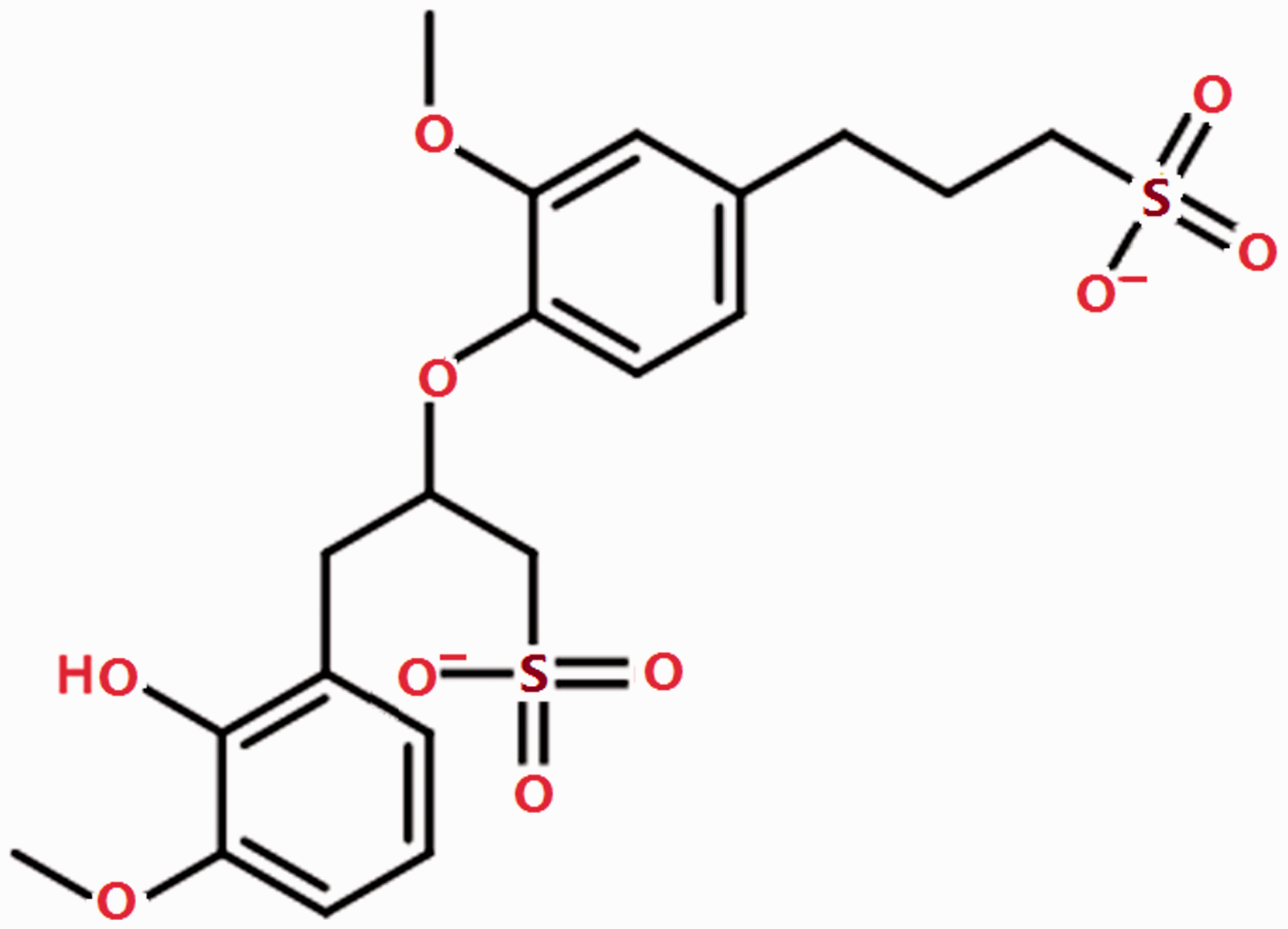
In this work, Mg2Al-LDH intercalated with LS was used as adsorbent for removal of pyridine in aqueous solution. The adsorption performance of the hybrid LDH material for pyridine was systematically researched. The influence of pH, contact time, adsorbent dosage, and adsorption–desorption cycles on the adsorption process was investigated. The results demonstrated that the Mg2Al-LS-LDH has excellent performance for the removal of pyridine and a great potential for practical application in wastewater treatment.
Experimental section
Materials
Mg(NO3)2·6H2O, Al(NO3)3·9H2O, and NaOH were purchased from Beijing Chemical Co., Ltd. Sodium lignosulfonate (SLS) was received from Aladdin Industrial Co., Ltd. Pyridine was obtained from Sinopharm Chemical Reagent Co., Ltd. All the reagents were analytical grade chemicals and used without any purification.
Preparation of adsorbents
The modified Mg2Al-LS-LDH was synthesized by a conventional coprecipitation method. Solution A: 0.03 mol of Mg(NO3)2·6H2O and 0.015 mol of Al(NO3)3·9H2O was dissolved in 200 ml of deionized water (Mg2+:Al3+ molar ratio = 2:1). Solution B: 2 m of NaOH solution was obtained by dissolving NaOH in deionized water. Fifty milliliters of solution B was dropwise added into 200 ml of solution A in a flask. After stirring for 1 min, 50 ml of SLS (0.15 m) solution was added into the flask. The resulting suspension was stirred at 358 K under pH 9 for 10 h. Upon cooling, the resulting slurry was separated by filtration. At the end, the Mg2Al-LS-LDH was dried at 335 K for 24 h.
For comparison, the CO3-LDH was also synthesized by a conventional coprecipitation method. Solution A: 0.03 mol of Mg(NO3)2·6H2O and 0.015 mol of Al(NO3)3·9H2O were dissolved in 200 ml of deionized water (Mg2+:Al3+ molar ratio = 2:1). Solution B: 0.072 mol of NaOH and 0.03 mol of Na2CO3 were dissolved in 200 ml of deionized water. Solution B was dropwise added into solution A in a flask. The resulting suspension was stirred at 358 K under pH 9 for 10 h. Upon cooling, the resulting slurry was separated by filtration. At the end, the CO3-LDH was dried at 335 K for 24 h.
Adsolubilization tests
A batch of experiments was implemented in conical flasks. A given mass of lignosulfonate intercalated Mg2Al layered double hydroxide (LS-LDH) (30–140 mg) was added into 100 ml of pyridine solution. The range of initial concentration of pyridine solution was from 25 to 400 mg l−1. The value of pH (3, 4, 6, 8, 10, 12) was adjusted by added droplets of HCl or NaOH solution. Twenty-seven hours was discovered to be enough to reach adsolubilization equilibrium. After adsorbing, the suspending liquid was filtered with 0.22 µm organic membrane filter. The pyridine concentration of the leftover was detected by high-performance liquid chromatography (HPLC) with the detection wavelength of 254 nm. The adsorption capacity (Qe) of pyridine was calculated by the following equation
Regeneration tests
To explore reutilization performance of the adsorbent, the regeneration tests were carried out. The powder of LS-LDH after the first adsorption was immersed in 100 ml acetone by ultrasonic treatment for 30 min. The recycled powder was rejoined in pyridine aqueous solution with the same concentration. This process on adsorption–regeneration cycle was repeated 10 times. Remaining pyridine concentration after each adsorption cycle was detected in the same way as above.
Characterization
Powder X-ray diffraction (XRD) data were collected on a Shimadzu Model XRD-6000 powder diffractometer, using Cu KR radiation (30 mA and 40 kV) in the 2θ range of 1–70°, with a scanning rate of 10° min−1. Fourier transform infrared (FT-IR) spectra were received using a standard KBr pellet technique (1 mg of sample in 100 mg of KBr) on a Bruker Vector 22 Fourier transform spectrometer in the range between 400 and 4000 cm−1 with 2 cm−1 resolution. The morphology of LS-LDH was obtained by scanning electron microscope (SEM, Zeiss SUPRA 55). Elemental analysis was performed using VarioEL III Elementar CHNS analyzer. The LS content was calculated on the basis of sulfur content. Inductively coupled plasma atomic emission spectroscopy (ICPS-7500) was also used to measure the ratio of Mg, Al, and S elements in the LDH. Solutions were prepared by dissolving a small amount of LS-LDH in dilute HNO3 solution. The low-temperature N2 adsorption–desorption experiments were carried out using a Quantachrome AS-IC-VP system. Samples were outgassed at 353 K for 8 h. The specific surface areas were calculated using the Brunauer–Emmett–Teller (BET) method based on the N2 adsorption isotherms. The HPLC was recorded on an Agilent 1260 type instrument. The mobile phase consisted of methanol–water (60:40), and the detection wavelength was 254 nm. The parameters of HPLC method are as follows: C18 column was used, flow rate was 1 ml min−1, LoD was 0.0045 mg l−1, and QoD was 0.015 mg l−1. A calibration curve of pyridine solution was performed before each test, which ensured the linear correlation of absorbance and concentration (correlation coefficient of R2 > 0.99).
Results and discussion
Structure of the adsorbents
Figure 1 shows the XRD patterns of CO3-LDH, LS-LDH, and LS in the range of 2θ = 1–70°. In each LDH case, the reflections can be indexed to a hexagonal lattice with R-3 m rhombohedral symmetry, which often is used for the description of LDH structures (Bookin et al., 1993). The value of d003 represented the thickness of the brucite layer plus the interlayer space, displaying the size and orientation of the intercalated anions. The series of (00 l) peaks at 2θ = 10.6, 21.2 and 31.8° in Figure 1(b), 2θ = 1.97 and 2.92° (a lower 2θ cannot be seen in the XRD pattern) and 2θ = 1.19, 2.38 and 3.56° in the inset of Figure 1 are the characteristics of layered phase with three forms of intercalation structure; the d003 basal spacings of 0.86 and 9.08 nm (2θ = 1.97°/2 ≈ 2.92°/3 = 0.97°) that are similar with a previous report (Huang et al., 2015) for the long chain LS anions exited in the interlayer of LDH both in “flat” and “vertical” forms, and there is another d003 basal spacing of 7.36 nm (2θ = 1.19°) indicating the third “tilted” form. Compared with CO3-LDH (Figure 1(a), d003 = 0.78 nm), the expansion of the basal spacing of LS-LDH, indicating that the LS was successfully intercalated into the galleries of LDH. The (110) reflection (2θ = 61°) showed no obvious shift compared with CO3-LDH, displaying that no significant change occurred in the LDH host layers after incorporation of the SLS. Compared with SLS (Figure 1(c)), LS-LDH gave priority to the characteristic peaks of LDH phase.
XRD patterns of CO3-LDH (a), LS-LDH (b), and SLS (c), partial enlargement of LS-LDH pattern is shown in the inset. LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide; SLS: sodium lignosulfonate; XRD: X-ray diffraction.
The FT-IR spectra of CO3-LDH, SLS, and LS-LDH hybrid are shown in Figure 2(a). For CO3-LDH (a), the strong and sharp adsorption band at about 1370 cm−1 belongs to the ν3 mode (asymmetric stretching) of (a) FT-IR spectra of CO3-LDH (a), SLS (b), and LS-LDH (c) and (b) SEM image of LS-LDH. FT-IR: Fourier transform infrared; LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide; SEM: scanning electron microscope; SLS: sodium lignosulfonate.
Effect of pH on pyridine adsorption
The adsorption capacity of hybrid LS-LDH is remarkably dependent on the solution pH value because the solution pH effects not only on the surface properties of LS-LDH but also the pyridine species presented in water solution. The dissociation constant (pKa) value of pyridine is 5.2, indicating that pyridine in water can exist as protonated form (pyridinium, Py-H+) and neutral form (pyridine) when pH<5.2 and pH > 5.2, respectively. Adsorption of pyridine by LS-LDH was investigated at different initial pH values in the range from 3.0 to 12.0 at 328 K, pyridine concentration of 100 mg l−1 for 27 h, and an initial LS-LDH concentration of 0.6 g l−1. The adsorption behavior was demonstrated in Figure 3(a). Obviously, the amount of pyridine absorbed was increased with pH value of the solution from 71 mg g−1 (pH 3.0) to 140 mg g−1 (pH 6.0), while the adsorption capacity of the LS-LDH decreased with pH value from 140 mg g−1 (pH 6.0) to 98 mg g−1 (pH 12.0). This phenomenon in the adsorption profile can be explained as the zeta potential on the surface of LS-LDH was dependent on the pH value of solution (Figure 3(b)). As can be seen, the isoelectric point (pHpzc) of LS-LDH is 5.4, indicating that the adsorbent surface is positively charged at pH < 5.4 and negatively at pH > 5.4, respectively. Therefore, according to the electrical double layer theory, there was nearly no diffusion layer on the surface of LS-LDH when the pH value of solution changed from 5.0 to 6.0, which is benefit for the adsorption of pyridine. Furthermore, pyridine exists as neutral form and the surface of LS-LDH was positively charged at pH = 5.0–6.0. It has been reported that pyridine is preferentially adsorbed on a positively charged surface owing to the more electronegative N atom (Ahmed et al., 2014). Therefore, these advantages resulted in the highest removal percentage of pyridine at pH = 5.0–6.0. Meanwhile, the adsorption capacity decreased at low pH (3.0–4.0), which can be attributed to the thick diffusion layer and the electrostatic repulsion between Py-H+ and LS-LDH. When the pH is high (8.0–12.0), the surface of the LS-LDH is negative and the diffusion layer is thick that are disadvantage for pyridine to approach the surface of LS-LDH, resulted in a little adsorption.
(a) Plot of the pyridine adsorption capacity in aqueous solutions at different pH by LS-LDH and (b) plot of the zeta potential of LS-LDH composite at different pH values. LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.
Effect of time on pyridine adsorption
Contact time is one of the key parameters in adsorption process. The adsorbed amount of pyridine on LS-LDH and CO3-LDH versus time was studied at an initial pyridine concentration of 100 mg l−1 at T = 328 K and pH = 6, while the contact time varied from 1 to 25 h. The results are demonstrated in Figure 4. It was observed that the pyridine uptake increased sharply at the first 14 h with adsorption capacity of 128 mg g−1 on LS-LDH and 94.6 mg g−1 on CO3-LDH, followed by a slow increase step and gradually reached equilibrium in the next step of 14–25 h with the adsorption capacity of 141.5 mg g−1 on LS-LDH and 106.8 mg g−1 on CO3-LDH. Moreover, LS can dissolve in water which has no absorbability on pyridine.
Adsorption profile of pyridine with contact time in terms of adsorption capacity (Vsol = 100 ml, adsorbent mass = 60 mg, initial pyridine concentration C0 = 100 mg l−1 and pH = 6).
Kinetic model parameters for the adsorption of pyridine by LS-LDH.

LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.



Isotherm parameters for pyridine adsolubilization on LS-LDH.

LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.
Effect of adsorbent dosage on pyridine adsorption
Figure 5 reveals the influence of the amount of LS-LDH on pyridine adsorption. First of all, with the adsorbent dosage, the pyridine removal increased monotonously before reaching a maximum removal. The main reason is the increasing of adsorption sites with the adsorbent dosage. When the amount of LS-LDH increased from 0.3 to 2 g l−1, the removal percentage increased from 67.2 to 87.9%. However, the removal percentage of pyridine changed slightly, though adsorbent dosage increased incessantly, displaying that it was very hard to thoroughly remove pyridine from the solution by just only increasing the amount of adsorbent because of the lower Ce of pyridine (Figure 5(a)). Figure 5(b) shows the adsorption curves of different LS-LDH dosages versus contact time. It is clearly illustrated that the adsorption capacity reduced with the dosage of adsorbent.
(a) Plot of Qe versus Ce with different dosages of LS-LDH and (b) effect of adsorbent dosage on pyridine adsorption ability (Vsol = 100 ml, initial pyridine concentration C0 = 100 mg l−1 and pH = 6). LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.
Effect of initial concentration of pyridine on adsorption
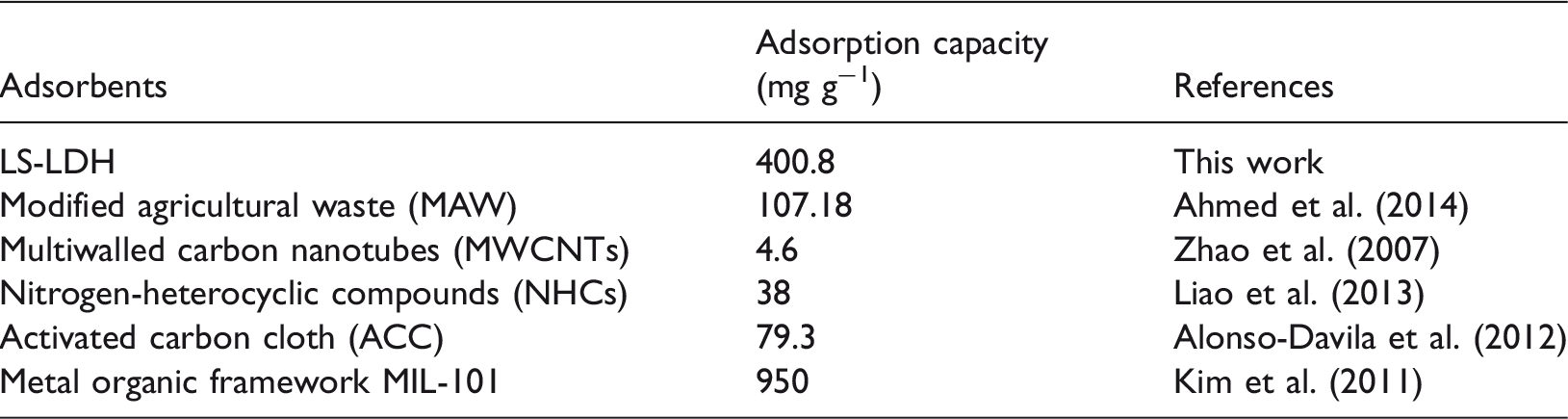
Figure 6(a) depicted the effect of initial pyridine concentration (25 mg l−1 ≤ C0 ≤ 400 mg l−1) on the adsorption ability of LS-LDH. It clearly showed that the adsorption capacity of LS-LDH increased with the initial pyridine concentration, which can be attributed that there is much more contact probability between adsorbents and pyridine in high concentration. Figure 6(b) showed the adsorption capacity of LS-LDH significantly increased with the initial pyridine concentration or Ce, indicating that the adsorption process was partitioning and the nitrogenous heterocyclic compound pyridine was dissolved in three-dimensional hydrophobic interlayer rather than adsorbed on the outside surface of LS-LDH. Similar adsolubilization process was also observed in our previous work (Zhao et al., 2011, 2015). The adsorption capacity can reach to 400.8 mg g−1 at pH = 6. In Table 3, the adsorption capacity of pyridine by several materials is listed. Compared with other materials, the adsorption capacity of pyridine on LS-LDH is larger. Although the capacity of metal organic framework MIL-101 has 950 mg g−1, it is too expensive.
(a) Effect of initial pyridine concentration on the adsorption ability of LS-LDH and (b) plot of Qe versus Ce with different initial pyridine concentration (Vsol = 100 ml, adsorbent mass = 60 mg, and pH = 6). LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide. Adsorption capacities of materials for pyridine removal from wastewater. LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.
According to the N2 adsorption isotherm, the BET surface area of LS-LDH was 74.47 m2 g−1. And, the minimum molecular diameter of pyridine was about 0.42 nm that is calculated by using Materials-Studio. If the surface of LS-LDH was fully occupied, the most maximum massive of pyridine is ca. 71 mg g−1. The pyridine uptake on the surface of LS-LDH is only about 17.6% of the total uptake (400.8 mg g−1). Therefore, we believe most pyridine molecules entered into the interlayer space of LS-LDH. The LS anion has strong gather tendency resulting in many hydrophobic phases in the interlayer region. Moreover, these LS molecules tend to form surfactant micelles in solution, which were expected to have the capacity of dissolving hydrophobic organic compounds (Zhao et al., 2011). Therefore, it can be concluded that the pyridine molecules could be captured and solubilized in the hydrophobic interlayer space owing to adsolubilization. Figure 7 shows that it can coexist with some types of interactions between pyridine and LS-LDH, including π–π conjugated effect (area A in Figure 7), nonpolar attractions (area B), hydrogen bonds (area C), and electrostatic interactions (area D). The crystal structure of LS-LDH after the adsorption experiment was characterized by XRD (namely LS-LDH-a, Figure 8). Compared with the LS-LDH, the d003 value for the LS-LDH-A was 0.90 nm, larger than that of the LS-LDH (d003 = 0.86 nm), indicating that some pyridine molecule has been intercalated into the space of the LS-LDH based on the adsolubilization effect, which resulted in the slight stretch of LS molecular in the interlayer of LS-LDH. Furthermore, the FT-IR spectrum of LS-LDH-a (Figure 9(b)) shows the characteristic band of pyridine at 806 cm−1, but that of LS-LDH (Figure 9(c)) has not, which is assigned to the ν (C–N) stretching vibrations (Figure 9(a)), further confirming the above conclusion.
The mechanism of pyridine adsolubilization into a LS-LDH. LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide. XRD patterns of (a) LS-LDH and (b) LS-LDH-a. LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide; XRD: X-ray diffraction. FT-IR spectra of (a) pyridine, (b) LS-LDH-a, and (c) LS-LDH. FT-IR: Fourier transform infrared; LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.


Recycling of adsorbents
The recycling experiment is a critical requirement in practical applications. The adsorbed pyridine can be eliminated by desorption with the help of acetone, which can be concentrated and reused. Therefore, the LS-LDH can be reconverted as adsorption material for reuse. Continuous adsolubilization and regeneration cycles for pyridine with the LS-LDH adsorbent were repeated up to 10 times under the same experimental condition in this work (Figure 10). Obviously, the removal percentage of pyridine can be achieved 84% at the first regeneration cycle. However, the adsolubilization capacity of the adsorbent decreased progressively with a cycle of regeneration. This phenomenon could be explained by slight loss of LS-LDH with the extraction of pyridine in the acetone treatment process. Despite all these, the pyridine removal percentage still remained higher than 55% after 10 recycles, displaying a good reusability of the LS-LDH.
Recycling experiment with 10 cycles of removing pyridine by LS-LDH. LS-LDH: lignosulfonate intercalated Mg2Al layered double hydroxide.
Conclusions
The LS-LDH was synthesized by coprecipitation method. The composite was investigated as an adsorbent for the adsorption of pyridine in aqueous solution and a series of factors were carefully discussed. The adsorption behavior of pyridine by LS-LDH was apparently influenced by pH and the adsorption capacity can reach to 400.8 mg g−1 at pH = 6. The removal percentage achieved was about 87.9%. Moreover, the adsorption kinetics followed the pseudo-second-order model and the experimental equilibrium was well fitted to the linear model. Importantly, the LS-LDH could be easily regenerated by acetone, and it still remained a well adsorption capacity after 10 adsorption–desorption cycles. Thus, it can be concluded that the LS-LDH could be used as potential adsorbents in wastewater treatment for the removal of pyridine.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 973 Program (no. 2014CB932104), the National Natural Science Foundation of China, Program for New Century Excellent Talents in Universities, the Fundamental Research Funds for the Central Universities (ZZ1501 and YS1406) and the Program for Changjiang Scholars, Innovative Research Team in University (no. IRT1205).



