Abstract
Fatty acids are an interesting class of educts for chemical processes as they are available from renewable resources. For obtaining high-purity fatty acids, efficient separation techniques are needed. An interesting option is adsorption. In the present work, therefore, the adsorption of oleic acid on hydrotalcite, a readily available adsorbent, is studied. The focus is on studying the influence of the composition of the solvent on the adsorption. Adsorption isotherms are reported for oleic acid in pure isopropanol and mixtures of isopropanol and water at temperatures between 278 and 308 K. Even though the solubility of oleic acid in isopropanol is higher than in mixtures of isopropanol and water, the highest capacity of the adsorber is found for pure isopropanol. The reasons are discussed. No significant influence of the temperature was observed.
Introduction
Fatty acids are interesting educts for the chemical industry as they are available from renewable resources and offer opportunities for new synthesis routes, e.g. for producing polymers (Baumann et al., 1988; Biermann et al., 2000). Oleic acid (OA) is one of the most important fatty acids, as many fats and oils contain high amounts of this compound (Baumann et al., 1988), however, along with other fatty acids. For obtaining pure fatty acids, efficient separation techniques are needed. Separating fatty acids is a difficult task, for which different methods have been studied. Besides crystallization or vacuum distillation (Hamilton, 1978; Haraldsson, 1984; Mularczyk and Drzymala, 1989), an interesting option is adsorption (Jamal and Boulanger, 2010). Adsorption of OA has been studied by Cren and Meirelles (2005, 2012), who have shown that strong anion-exchange resins can be used for removing OA from ethanolic solutions containing water. In the present study, hydrotalcite is used as adsorber. Hydrotalcite is an anion-exchange material from the class of layered double hydroxides (LDH) (Rives and Ulibarri, 1999). These materials are anionic clays which have a double layer structure. One layer is similar to brucite (Mg(OH)2) (Rives and Ulibarri, 1999) but some Mg2+ ions are replaced with Al3+ ions. This layer is therefore positively charged. This charge is compensated by the second layer, also called interlayer, that contains anions as well as water (Rives and Ulibarri, 1999; Vaccari, 1998). The anions can be e.g.
Different adsorption mechanisms of substances on hydrotalcite have been described in the literature: Hydrotalcite can be used simply as anion-exchanger (Ulibarri et al., 2001) on which the counterions in the interlayer are exchanged upon adsorption of an anionic species, like R-COO−. But the binding, especially of R-COO−, may not only be of physical nature: in the so-called intercalation (Borja and Dutta, 1992; Carlino, 1997; Kanoh et al., 1999; Meyn et al., 1990; Wiesen et al., 2015), the adsorbent binds the acids in the lattice as R-COOH. Elongated solutes like carboxylic acid anions or carboxylate ions form either mono- or bimolecular layers with a perpendicular orientation to the adsorbent surface (Borja and Dutta, 1992; Itoh et al., 2003; Kanoh et al., 1999). Long carboxylic acids can show kinks (Borja and Dutta, 1992; Meyn et al., 1990). Carlino (1997) suggests that they adsorb parallel to the hydroxide layer as ‘flat stacks’ for low concentrations of carboxylic acids. The formation of bimolecular films (Borja and Dutta, 1992) is reported for carboxylic acids in ethanolic solutions, whereas for carboxylic salts in aqueous solutions monomolecular films are found (Meyn et al., 1990). Kanoh et al. (1999) have studied the temperature dependence of the intercalation of sodium stearate in aqueous solutions in a hydrotalcite and found that with increasing temperature the bilayer changes to monolayers. For methanolic solutions, the adsorption of sodium stearate on hydrotalcite does not depend on temperature (Kanoh et al., 1999). One problem of the adsorption of long carboxylic acids on hydrotalcite is the accessibility of the interlayer. It can be increased by swelling the adsorbent in components like glycerol (Hansen and Taylor, 1991; Wiesen et al., 2015). LDH materials can also be modified prior to adsorption by calcination (Carlino, 1997; Crepaldi et al., 2002; Ulibarri et al., 2001; Wiesen et al., 2015). The water and carbonate ions are removed from the interlayer by applying high temperature, which leads to a change in the structure of the metal hydroxide layer to a mixed metal oxide (Bergaya et al., 2006; Crepaldi et al., 2002). Upon adsorption, the interlayer is rebuilt (Carlino, 1997; Ulibarri et al., 2001; Wiesen et al., 2015). Wiesen et al. (2015) show that calcinated hydrotalcites are efficient adsorbents for fatty acids. However, the advantage of calcination for the adsorption goes together with an increased effort for desorption and regeneration, which requires calcination.
In the current work, the adsorption of OA on an untreated hydrotalcite with carbonate ions as anions is studied. As solvent, isopropanol (iPOH) and mixtures of iPOH and water are used. The focus of the study is on the influence of the solvent composition on the adsorption, which has not been studied before for the adsorption of fatty acids on hydrotalcite. The outcome of the study is unexpected: even though iPOH is a much better solvent for OA than water (OA is practically insoluble in water, whereas iPOH is a good solvent for OA), we find that the adsorption of OA on the hydrotalcite is highest for pure iPOH.
Experimental section
Material
OA was obtained from Fluka (standard for gas chromatography (GC) natural, >0.995 g/g). iPOH was obtained from Sigma-Aldrich (p.a. quality, ≥0.998 g/g). Hydrotalcite EXM 2152 with the chemical structure [Mg0.75Al0.25(OH)2](CO3)0.5⋅0.5H2O was provided by Clariant-Produkte (Deutschland) GmbH (Wiesen et al., 2015). The hydrotalcite is stable for pH values of 3 < pH < 12 (Miyata and Kumura, 1973). Trimethylsulfoniumhydroxide (TMSH) (0.2 M in methanol), used for GC analysis, was obtained from Macherey-Nagel. As internal standard for the GC anisole (≥0.99 g/g) from Roth was used.
Adsorption experiments
The adsorption experiments were carried out in capped 2 mL vials. About 25 mg of hydrotalcite powder was put in the vials and the amount was determined by weighing. It is known with an accuracy of 0.1 mg. Stock solvent mixtures of iPOH and water were prepared gravimetrically. The accuracy of the mass fraction of water is better than 0.1 mg/g. A second stock solution containing OA and the stock solvent was prepared with an accuracy of the OA mass fraction of 0.2 mg/g. Both stock solutions were added in different ratios to the vials containing the adsorber using a fully automated liquid weighing laboratory robot. Details on the set-up are described in Werner et al. (2014). The initial OA concentrations were between 0.1 mg/g and 5 mg/g and are known with an accuracy of better than 0.3 mg/g. The pH of the mixture was about 4.5 and was not adjusted. The total volume of liquid in the vial was about 0.7 mL. For all weighing steps carried out in the present study calibrated laboratory balances with resolutions of 1 mg and better were used. The mass of the OA + iPOH + water solution added to the vial is known with an accuracy of 0.2 mg. The vials were shaken in a thermostated oven (Memmert, ICP 600, temperature accuracy of 1 K) on a shaker (Vortex, Genie 2T) for at least 3 h to reach equilibrium. Shaking times of 20 h showed no difference in load capacity. After reaching equilibrium, the adsorber was separated from the liquid phase by centrifugation (Heraeus Multifuge X1R) and filtration through syringe filters (Whatman, 4 mm, 0.2 µm polytetrafluoroethylene (PTFE)).
The experiments were carried out with pure iPOH and different mixtures of iPOH as solvent (iPOH mass fractions: 0.6, 0.7, 0.8, 0.9 and 1 g/g) at 298 K and for the solvent with 0.8 g/g iPOH also at 278 K, 288 K, and 308 K.
GC was used for analysis of the OA content of the supernatant. The liquid samples, which were obtained after centrifugation and filtration, were prepared by adding 10 µL anisole as internal standard and 100 µL TMSH for derivatization of the fatty acid (Macherey-Nagel, 2012) to about 200 µL sample. The masses of the sample, anisole, and TMSH were determined by weighing with an accuracy of ±0.1 mg. The derivatization is necessary as the boiling temperature of OA is too high for the GC. Through methylation of the OA to its methyl ester, the boiling temperature is decreased from 360 ℃ to 230 ℃ (Butte, 1983; Christie, 1993; Macherey-Nagel, 2012; Müller et al., 1990). To ensure that all OA in the sample is derivatized, the TMSH was added in excess. Details on the GC analysis are given in the Supplementary Material.
Data evaluation and modeling
The equilibrium loading of the adsorber with OA 

Results and discussion
The experiments were performed with initial mass fractions of OA in the solution of up to
Figure 1 shows the experimental results including estimates of the experimental uncertainty and their fit by the Langmuir model, cf. Table 1. The observed loadings are low, not only the data shown in Figure 1, but also the maximum loadings from the Langmuir model, cf. Table 1, which do not exceed 10 mg/g. The error bars presented in Figure 1 result from error propagation as explained above. They are large as the loading is basically obtained from a difference of two small numbers, cf. Equation (1). In some cases the error bars obtained for results for one solvent composition exceed the differences between the results for different solvent compositions. Nevertheless, by comparing the results for the entire isotherm, the influence of the solvent composition is clearly discernible.
Adsorption isotherms of OA on hydrotalcite at 298 K. Experimental data for different solvent compositions Fitted parameters OA: oleic acid.
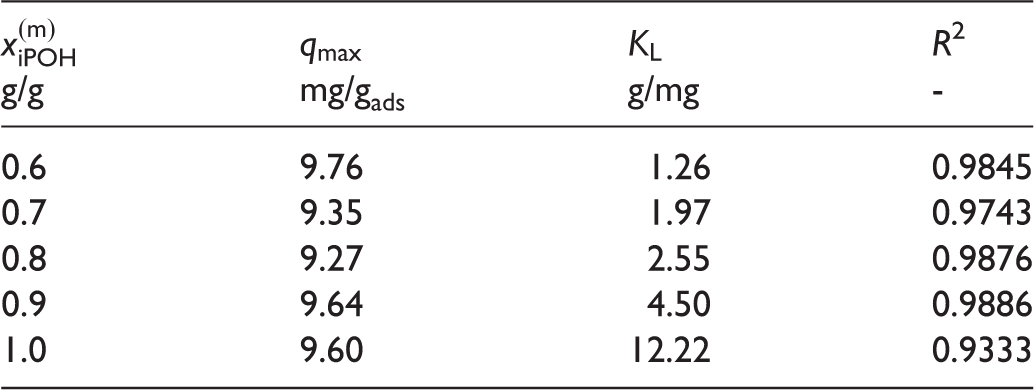
The loading of the adsorbent with OA is favored by high concentrations of iPOH in the solvent. The highest loadings are observed for pure iPOH. This finding is unexpected as iPOH is a much better solvent for OA than water. The positive influence of iPOH on the adsorption must therefore result from a positive influence on the interactions of OA with the adsorbent. An effect which could explain these findings is the swelling of the adsorbent (Wiesen et al., 2015): upon replacing water by iPOH in the interlayer, the interlayer thickness is expected to increase, leading to a better accessibility of the adsorbent surface for the large OA molecules. Furthermore, the adsorbent surface is hydrophilic. Therefore, when water is present in the solvent, it is expected to accumulate in the interlayer. As water is a poor solvent for OA, the concentration of OA in the interlayer will be low leading to poor adsorption. Adding iPOH to the solvent moderates this adverse effect.
Another issue is that of the role of the anion exchange. OA is only a weak acid (Kanicky and Shah, 2002) and only partially dissociated at the conditions studied in the present work. The carbonate ions compete with the OA anions for the positively charged sites on the adsorbent. It can be expected that in this competition the small carbonate ions are in better position, which is in line with the finding of low OA loadings. The observed negative influence of water on the adsorption could, hence, also be explained by shielding of the positive sites by the highly polar water.
For an iPOH concentration in the solvent of Adsorption isotherms of OA on hydrotalcite for solvent composition 
Conclusion
In this paper, we have studied the adsorption of OA on hydrotalcite. The solvents are iPOH and different mixtures of iPOH and water. The focus is on studying the influence of the solvent on that adsorption. The OA loading of the hydrotalcite was below 10 mg/g for all conditions studied in the present work, which is probably too low to be attractive for industrial separations. Even though iPOH is a better solvent for OA than water, it has a positive influence on the adsorption of the OA. This must result from a positive influence of iPOH on the interaction of OA and the adsorbent and is a consequence of the replacement of water in the interlayer by iPOH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the MAGNENZ project with financial support of BMBF.
Supplementary Material
The Supplementary material for this article is available on the Journal site.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
