Abstract
In this research work, removal of Cochineal Red A dye from aqueous solution was studied using layered double hydroxide materials prepared by a coprecipitation method at variable pH. The effects of the synthesis pH, the nature of the divalent cations and the total metal salt concentration on the formation of layered double hydroxides and their application to the removal of Cochineal Red A dye from aqueous solution were investigated. The structure of the materials prepared was characterized by X-ray diffraction and Fourier transform infrared. It was found that the nature and content of the bi- and trivalent metal ions in the layered double hydroxide influenced the adsorption. Ni–Fe had 0.96 to 1.70 times more adsorption capacity before reaching its equilibrium adsorption than Zn–Fe prepared at pH 7–9 with 2.2 M of total metal salt concentration. No difference was observed in the amount of Cochineal Red A dye adsorbed onto Zn–Fe prepared at both pH 7–9 and pH 10–11 in the range 1.1–3.3 M of total metal salt concentration. The results show that the increase in the precipitation pH leads to decreasing adsorption capacity. The Cochineal Red A dye adsorption follows the Temkin model for the Ni–Fe and Zn–Fe prepared at pH 7–9 and follows the Langmuir model for the Zn–Fe synthesized at pH 10–11 with high coefficient correlation. The adsorption kinetics data fitted the pseudo-second-order model. A thermodynamic study indicates that the adsorption process is both spontaneous and endothermic in nature.
Introduction
Dyes are widely used in many industries, such as the textile, cosmetic, plastic, paper, food and petroleum industries.1–4 Many dyes can be discharged into water effluent and can cause a serious hazard to aquatic organisms due to their toxicities and their carcinogenicities.4–6
Cochineal Red A, azo dye, is widely used in different industrial fields, including food, cosmetics, drugs, textile and pharmaceutical industries.7,8
It leads to the number of environmental and health hazards. It may cause occupational asthma, anaphylaxis, urticaria, angioedema, bronchospasm or aggravation of atopic dermatitis and food allergies. It may increase hyperactivity in affected children and adversely affect those who are sensitive to aspirin.9–11 Therefore, it needs to be removed before the wastewater can be discharged.
Various methods, such as chemical, biological and photochemical degradation, coagulation, sedimentation, adsorption, membrane processing and using combined treatments involving different methods, have been applied for the removal of dyes from wastewater.12–16 Among these methods, adsorption is one of the most effective and attractive methods for dye removal due to its low operating costs, high removal efficiencies, operating flexibility, insensitivity to toxicants and the option of recycling and reusing adsorbents.17–20
Different adsorbents, such as activated carbon, bentonite clay, mesoporous silica and carbon nanotubes, have been used successfully for the adsorption of dyes from aqueous solutions. 12 Recently, layered double hydroxides (LDHs), a class of anionic clays, have attracted great interest due to their potential applications in catalysis, photochemistry, electrochemistry, polymerization, magnetization, biomedical science and in environmental processes.21–25
LDHs, also known as hydrotalcite-like materials or anionic clays, are represented by the general formula [M2+1−xM3+x(OH)2][Am−x/m,nH2O], where M2+ is a divalent metal cation, such as Mg2+, Zn2+ and Cu2+, M3+ is a trivalent metal cation, such as Al3+, Fe3+ and Cr3+, Am− is the exchangeable interlayer anion and x is defined as the ratio between divalent and trivalent cations with different values between 0.2 and 0.33. 26
The objectives of this study were to synthesize LDHs by a coprecipitation method at variable pH with different synthesis conditions which are as follows: synthesis pH, metal salt concentration and the nature of the divalent cations and to evaluate the adsorption performance of the materials so prepared for their capacity to remove the acid dye Cochineal red A from aqueous solution. As is known, the synthesis pH has a high impact on the formation of the layered structure in LDHs, the nature of the bi- and trivalent metal ions influences the structural properties of the LDH and the effect of metal salt concentration is an important factor for the precipitation of LDH materials. 25
From the literature, no information for the removal of Cochineal red A by the materials synthesized in this work is available.
Results and discussion
Characterization of materials
Figure 1 shows the X-ray diffraction (XRD) patterns of the materials synthesized.

XRD patterns of synthesized materials.
XRD patterns for Zn–Fe and Ni–Fe are shown in Figure 1(a). It can be seen that the diffraction patterns for Ni–Fe have peaks at lower 2θ as (003) and (006), which are the characteristics of hydrotalcite-like compounds. The XRD patterns show additional peaks that are associated with the second-phase sodium chloride (NaCl) which can be attributed to the reaction of Na+ with Cl−. It should be noted that the pH of the solution evolves continuously throughout the synthesis, favouring the presence of non-LDH phases. The pattern shown at 33.85° and 44.85° was associated with either Ni(OH)2 or Fe(OH)3. Because the as-synthesized Ni–Fe exhibits poor crystalline structure, a lower diffraction intensity was observed. 27 The XRD patterns of the Zn–Fe show that no hydrotalcite structure is formed under the synthesis conditions used (as described in the ‘Experimental’ section on the sorbent synthesis). It is composed of one major crystalline phase identified as ZnO due to the synthesis conditions used; based on Zn solubility curves, precipitation of ZnO starts at pH 9. 28
As shown in Figure 1(b) and (c), there is no difference in the structural properties of the materials obtained. So, it can be concluded that the total metal salt concentration in this range does not affect the structural properties of the materials prepared.
Chang et al. considered a broader range of concentrations of metallic salt solutions which varied from 0.003 to 0.3 M for the preparation of Zn–Al LDH. Differences in the structural properties of the materials obtained were observed.
As shown in Figure 1(d), the increase in the precipitation pH leads to the formation of mixed hydroxides. An excess of hydroxide ions leads to the complete precipitation of the metal hydroxides. 25
The infrared spectra of the materials prepared are shown in Figure 2. The broad bands observed at 3350 and 3390 cm−1 were attributed to the stretching mode of hydrogen-bonded hydroxyl groups from both the hydroxide layers and interlayer water. 29 The bands around 1550 and 1650 cm−1 for all spectra as shown in Figure 3 represent the H–O–H bending vibration of water.16,30 Bands at lower wavenumbers are due to LDH lattice vibrations (M–O, O–M–O). 24 The bands recorded at 1360 and 1370 cm−1 can be assigned to carbonate ions formed by the adsorption of atmospheric CO2. 31 As can be seen from Figure 3, the intensity of the infrared bands increases with increasing synthesis pH.
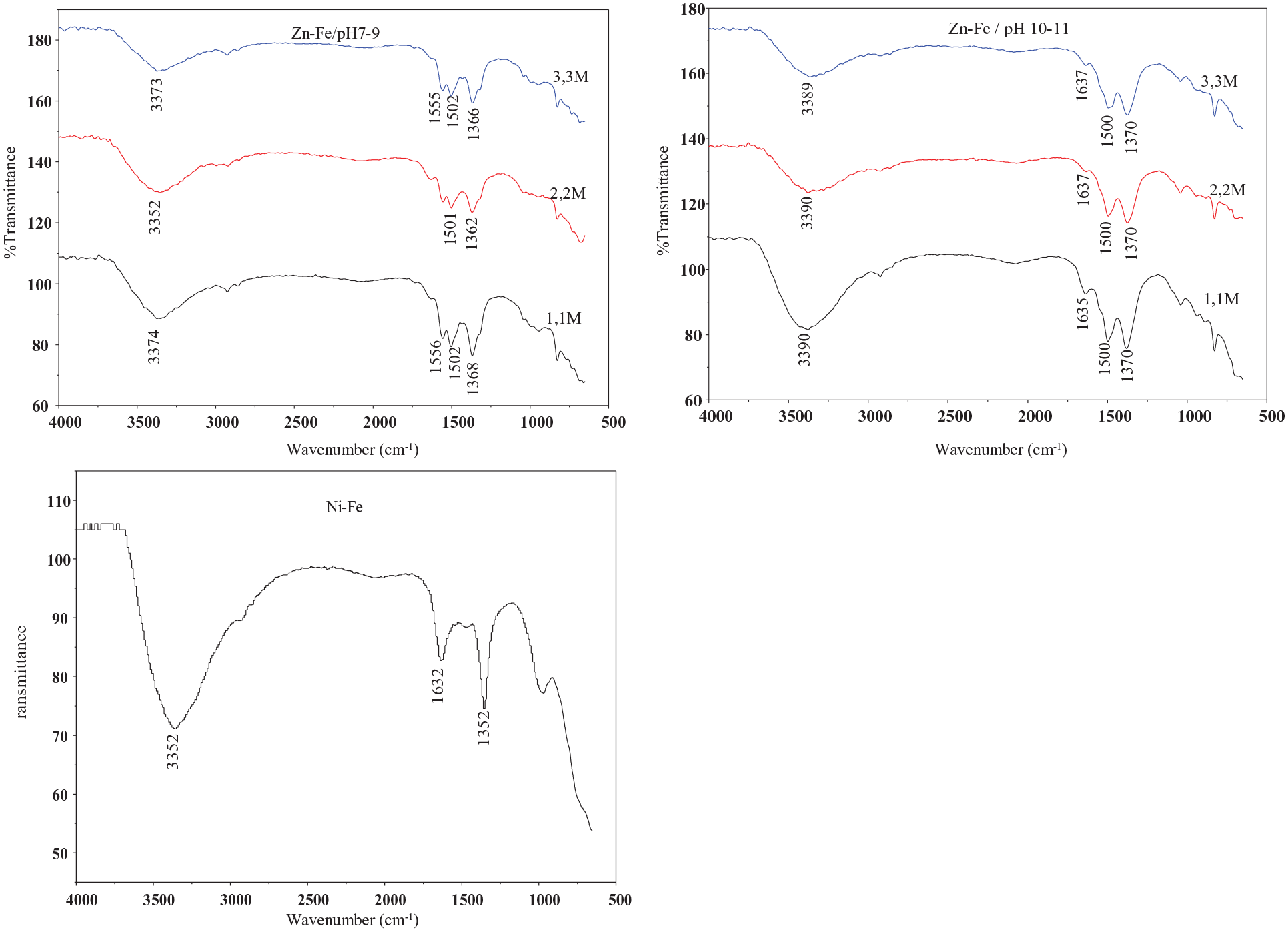
Infrared spectra of synthesized materials.

Effect of time in the adsorption of Cochineal Red A on synthesized materials.
Study of Cochineal Red A removal using the synthesized materials
Contact time effect on dye adsorption
Figure 3 shows plots of the observed concentration of dye versus contact time in the presence of the synthesized adsorbents. It is observed that the adsorbed amount of Cochineal Red A dye at time
The equilibrium adsorption capacities for Cochineal Red A dye reach its maximum of 30.64 and 30.55 mg g−1 for the Ni–Fe and Zn–Fe (2.2 M) prepared at pH 7–9, respectively. The nature and content of the bi- and trivalent metal ions in the LDH influence the adsorption. Rajib Lochan Goswamee et al. 33 have found that the adsorption capacity of Cr2O72− in an Mg–Al LDH with a higher Al3+ content in the structure is higher than for the product with a lower Al3+ content. In this study, the nature of the bivalent metal ions influences the adsorption. As shown in Figure 3, Ni–Fe had 0.96 to 1.70 times more adsorption capacity before reaching its equilibrium adsorption than Zn–Fe prepared at pH 7–9 with 2.2 M of total metal salt concentration. As confirmed by XRD analysis, differences in the structural properties of each material were observed. While no difference was observed in the amount of Cochineal Red A dye adsorbed in the range 1.1–3.3 M of metal salts concentration.
As can be seen from Figure 3, the Zn–Fe LDHs synthesized at pH 7–9 are faster than those prepared at pH 10–11. adsorption equilibrium was reached after 80 min with Zn–Fe prepared at pH 7–9 and after 180 min with those prepared at pH 10–11. In addition, the amounts of dye adsorbed by Zn–Fe synthesized at pH between 7 and 9 are much larger than those synthesized at pH between 10 and 11. These results led us to deduce that the increase in the precipitation pH leads to a bad adsorption capacity. This is due to the excess of hydroxide ions which leads to the complete precipitation of metal hydroxides.
Kinetic modelling
Figure 4 shows the pseudo-first-order kinetic model, a plot of

Pseudo-first-order kinetic for the adsorption of Cochineal Red A on synthesized materials.

Pseudo-second-order kinetic for the adsorption of Cochineal Red A on synthesized materials.
The kinetic parameters for all experimental data determined using pseudo-first-order and pseudo-second-order models are reported in Table 1.
Pseudo-first- and pseudo-second-order kinetics parameters of Cochineal Red A dye adsorption onto synthesized materials.

Large differences between experimental values of the equilibrium adsorption capacities and those calculated from the pseudo-first-order model were observed (Table 1). So, the first-order kinetic model is not adequate to describe the adsorption process. Due to its good correlation with the experimental results, the pseudo-second-order model has been used to describe the adsorption process. Consequently, the adsorption of dye on synthesized LDHs is a chemical adsorption rather than physical. 34
Equilibrium isotherms
The adsorption isotherms of Cochineal Red A dye on the materials synthesized are shown in Figure 6.
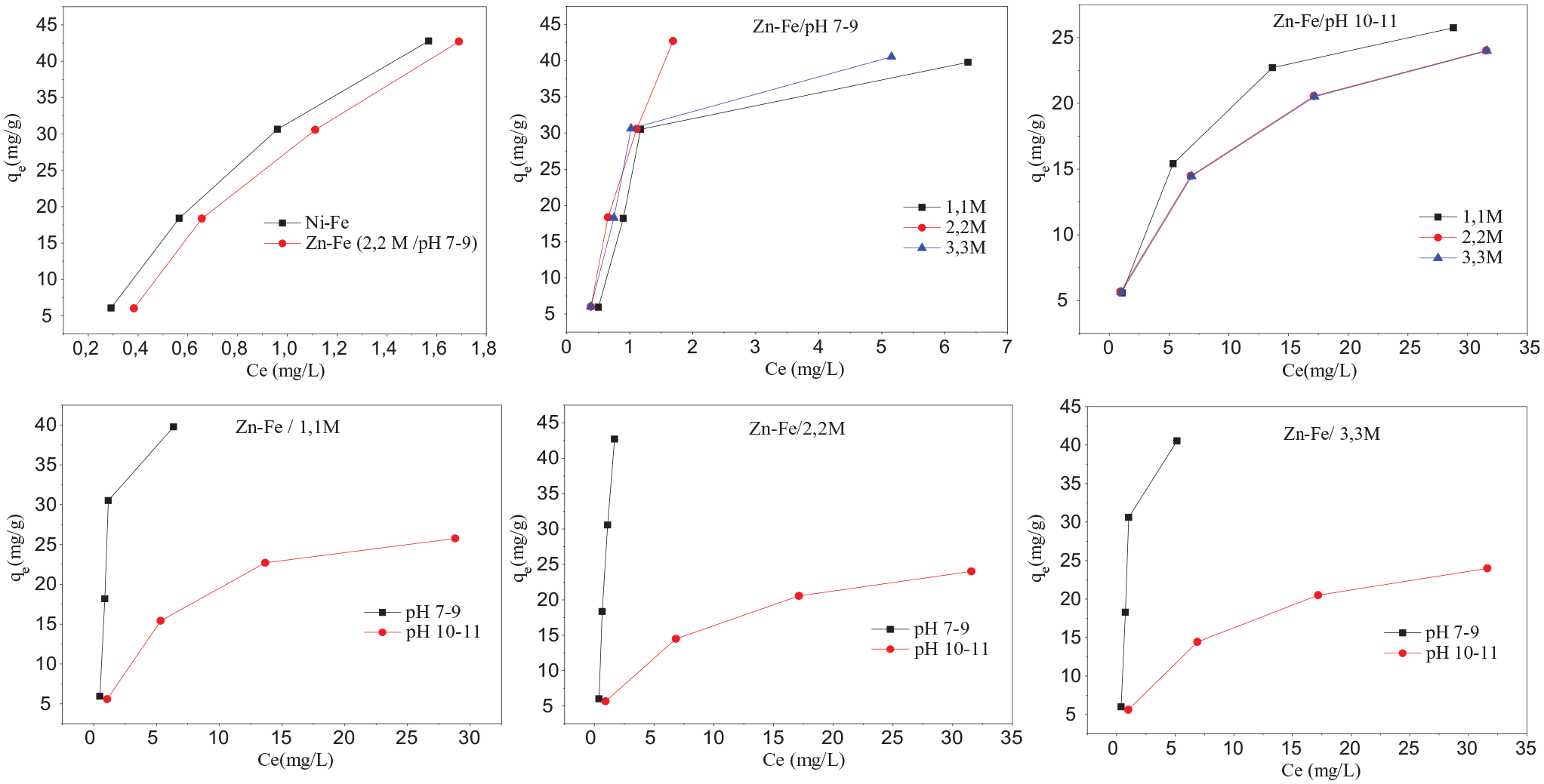
Cochineal Red A adsorption isotherms on the synthesized materials.
The results showed a significant difference in the adsorption ability of Zn–Fe prepared at pH 7–9 and those prepared at pH 10–11. The increase in precipitation pH leads to a bad adsorption capacity while no difference in the adsorption ability of Ni–Fe and Zn–Fe (pH 7–9/2.2 M) was observed. The nature and content of the bi- and trivalent metal ions in the LDH influence the adsorption. The total metal salt concentration does not affect the adsorption ability in the range 1.1–3.3 M.
As shown in Figure 7, the Langmuir, Temkin and Freundlich models were applied to describe the adsorption behaviour at equilibrium. The parameters obtained for all models are listed in Table 2.

Plots for the adsorption of Cochineal Red A by the synthesized materials. (a) Freundlich. (b) Temkin. (c) Langmuir.
Temkin, Freundlich and Langmuir isotherm constants for the adsorption Cochineal Red A dye on synthesized materials (T = 298 K).

From the correlation coefficient values (
Thermodynamic parameters of adsorption
As given in Table 3, the values of ΔH° for Ni–Fe and Zn–Fe (2.2 M/pH 7–9) are determined as 25.38 and 46.83 (kJ mol−1), respectively.
Values of thermodynamic parameters for the adsorption of Cochineal Red A dye onto Zn–Fe and Ni–Fe materials (

The positives values of ΔH° confirm that the adsorption process is endothermic. In addition, the positives values of the entropy changes (ΔS°) may be due to the increase in randomness at the solid solution interface during adsorption. This implies some structural changes in sorbate and sorbent, which leads to an increase in the disorderness of the solid solution system.
The negatives values of ΔG° indicate that the adsorption is spontaneous. The values of ΔG° became more negative with increasing temperature, showing that higher temperature facilitated the adsorption of dye on these materials.
Conclusion
The adsorption of Cochineal Red A dye on materials synthesized by a coprecipitation method at variable pH has been investigated. The effect of the synthesis conditions, such as pH, nature of the divalent cations and the total metal salt concentration on the formation of LDHs and their application to the removal of Cochineal Red A dye, was studied.
Our results show that the nature and content of bi- and trivalent metal ions in LDHs influence the adsorption.
No difference was observed in the amount of Cochineal Red A dye adsorbed onto Zn–Fe prepared at both pH 7–9 and pH 10–11 in the total metal salt concentration range of 1.1–3.3 M.
The increase in precipitation pH leads to decreasing adsorption capacity.
The adsorption of dye on all materials synthesized can be explained by a pseudo-second-order mechanism.
Among the three models tested, the Langmuir, Freundlich and Temkin models, the Temkin model was the most appropriate in describing the Cochineal Red A adsorption isotherm using Ni–Fe and Zn–Fe prepared at pH 7–9 in the range 1.1–3.3 M of total metal salt concentration with high correlation coefficient indicating the uniform distribution of binding energies between the molecules adsorbed and adsorbent. For Zn–Fe synthesized at pH 10–11, the experimental results are well described by the Langmuir model, indicating the monolayer adsorption of Cochineal Red A on these materials.
The thermodynamic study indicated that the dye adsorption on the materials synthesized is an endothermic and spontaneous process.
The synthesized materials Ni–Fe and Zn–Fe prepared at pH 7–9 with different total metal salt concentrations exhibited exceptional adsorption capacities indicating that these materials might be potentially efficient materials for the removal of Cochineal Red A dye from aqueous solutions.
Experimental
Sorbents synthesis
Zn–Fe and Ni–Fe LDHs were synthesized through a coprecipitation method at variable pH with [Ni]
A mixed aqueous solution of 0.14 mol of Zn(Cl)2, 0.07 mol of Fe(Cl)3 6H2O and 100 mL of distilled water was slowly added to a solution containing 200 mL of NaOH (2 M) until the pH was between 7 and 9 . The precipitate obtained was washed, dried at 70 °C overnight and then grounded into fine powder using an agate mortar.
Synthesis at other total metal salt concentrations in the range 1.1–3.3 M was synthesized as described above, where the molar ratio and synthesis pH were kept constants.
Synthesis at pH between 10 and 11 was performed in a similar manner for the different metal salt concentrations.
A Ni2+/Fe3+ LDH chloride was prepared by a method analogous to the Zn–Fe LDH described above, except that Ni(Cl)2 6H2O (20 g) and Fe(Cl)3 6H2O (11.37 g) were used as the source of nickel and iron(III) and the pH was between 9 and 10.
Sorbate
The molecular structure of Cochineal Red A (C20H11N2Na3O10S3) which is an azoic dye and often known as Acid Red 18 (trisodium salt), Food Red 102 and Ponceau 4R is given in Figure 8. 36

Cochineal Red A structure.
Characterization of the prepared materials
Powder XRD analyses were carried using an X’PERT-PRO diffractometer with CuKα radiation (λ = 0.154060 nm) at 45 kV and 40 mA from 5° to 80°. The scanning diffraction angle was set at the speed of 0.06° s−1, using a step size of 0.02° every 0.6 s and a slit width 0.6 mm. The Fourier transform infrared (FTIR) spectra of the samples in the 4000–400 cm−1 range were performed with an Alpha Bruker FTIR spectrometer. The pH values were determined using a Hanna HI 8424 portable pH meter. The pH meter was calibrated with standard 4.0 and 7.0 pH buffer solutions each time before the meter was used.
Study of Cochineal Red A removal with synthesized materials
Kinetic study and modelling
In this study, initially, the Cochineal Red A dye concentration was fixed at 50 mg L−1, and 400 mg of the prepared materials were shaken with 250 mL of dye solution. Experiments were carried out at room temperature and at constant stirring (500 r min−1). After selected time intervals, samples were taken from the solution and then centrifuged. The dye concentration was analyzed using a spectrophotometer (Optizen 3200) by measuring absorbance at λmax = 507 nm. The amount of dye adsorbed at time

where
The adsorption kinetic is an important aspect of processes for pollutant removal. The Lagergren first-order kinetic model (equation (3)) and the pseudo-second-order model (equation (4)) were used to describe the mechanism for process adsorption


where


Equilibrium adsorption measurements and modelling
The adsorption isotherms for each system of materials/adsorbate were studied by contacting 80 mg of material suspensions with Cochineal Red A in aqueous solution. In addition, 50 mL of dye solution ranging from 10 to 70 mg L−1 were stirred till equilibrium was reached and then centrifuged. The dye equilibrium concentration was determined by visible spectrophotometer at 507 nm.
The quantity of the dye adsorbed at equilibrium was calculated by the following expression

where
The Langmuir model, which is valid for monolayer adsorption onto surface containing a number of identical sites and there are no interactions between adsorbed molecules, is described in a linear form by the equation

where
The linear form of the Freundlich equation which is investigated for heterogeneous system is given by the following equation

where Ce is the equilibrium concentration of adsorbate (mg L−1), n is the amount of solute adsorbed, Qe is the amount of dye adsorbed at equilibrium (mg g−1) and Kf is Freundlich isotherm constant.
The Temkin isotherm assumes that adsorption is characterized by a uniform distribution of binding energies between the molecules adsorbed and adsorbent. The linear form of this later is given by the equation

where AT is Temkin isotherm equilibrium bending constant (L g−1) and B is a constant related to heat of adsorption (J mol−1).
Thermodynamic study
Thermodynamic parameters provide in-depth information about internal energy changes that are associated with adsorption.
The effect of temperature was studied for Ni–Fe and Zn–Fe (2.2 M/pH 7–9) at four constant temperatures (298, 308, 318 and 328 K), fixed initial dye concentration (
The thermodynamic parameters, such as enthalpy change (ΔH°), entropy change (ΔS°) and Gibbs free energy change (ΔG°), were calculated using the following equations


where
Free energy changes (ΔG°) of adsorption are calculated from

Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
