Abstract
This study investigated using walnut shells (WSs), a commonly found adsorbent, to remove tylosin, a common antibiotic, from wastewater. Characterization of the WS was carried out using Brunauer, Emmet, and Teller (BET), Fourier transform infrared spectrophotometer (FT-IR), scanning electron microscope (SEM), and X-ray diffraction (XRD). To determine the adsorption conditions in the batch system, solution pH, adsorbent amount, and contact time factors were studied. In the continuous system, flow rate and adsorbent amount were investigated. In adsorption studies carried out in a batch system, the optimum pH was determined as the original pH of the antibiotic solution (pH 7.5). Adsorption equilibrium was established in 30 minutes for a 10 g/L adsorbent amount. Kinetic and isotherm parameters were calculated using data obtained from experimental studies. The kinetic studies were evaluated with pseudo-first-order, pseudo-second-order, and intraparticle diffusion kinetic models. The tylosin's adsorption results on WS show that it fits the pseudo-second-order kinetic model. The isotherm studies on the adsorption of tylosin with WS were performed using the Freundlich, Langmuir, and Dubinin–Radushkevich (D-R) isotherm models. The tylosin's adsorption results on WS show that it fits the Langmuir isotherm model. In the adsorption studies carried out in the column system, the adsorbent amount and flow rate parameters were 30 g/L and 0.2 mL/min, respectively. In studies carried out in batch and column system environments, 78% and 100% removal were achieved, respectively. This method was successfully applied to determine tylosin in wastewater samples.
Introduction
Drugs are used globally for their effectiveness in various treatments (Das and Sengupta, 2023). Antibiotics are widely used to treat and prevent bacterial infections in humans, animals, and poultry for treatment and growth promotion (Angenent et al., 2008). Macrolides are considered one of the most prominent classes of antibiotics. They are widely used in human medicine because they are active against gram-positive and some gram-negative bacteria (Paíga et al., 2019). There has been a significant increase in the rate of antibiotics detected in rivers, lakes, and aquatic environments. When drugs are not removed from these water bodies, drug residues end up in drinking water facilities (Das and Sengupta, 2023).
Recently, the increasing consumption of drugs, especially antibiotics, is a concern for both environmental protection and human health (Sharma et al., 2019). Used drugs are excreted from human and animal bodies and are transferred to wastewater in metabolized or unchanged form. Antibiotics contained in animal or human waste can enter the environment through municipal wastewater treatment or land irrigation with recycled water. Similarly, animal and poultry manure may contain antibiotics that remain largely unchanged. Antibiotics in this manure have the potential to migrate to the point of application to agricultural lands and to adjacent surfaces or shallow groundwater (Topp et al., 2016). In these ways, antibiotics can reach the surface and groundwater (Rivera-Utrilla et al., 2013).
Tylosin is a macrolide antibiotic produced by
Antibiotic resistance in environmental microorganisms can rise as a result of antibiotic contamination in the environment (Topp et al., 2016). A significant public health issue is brought on by the rise in antibiotic resistance (Angenent et al., 2008). Antibiotic elimination is not possible with the current general treatment plant techniques. Antibiotics are removed via various techniques, including oxidation, flocculation, coagulation, biosorption, and extraction (Shim et al., 2019). Adsorption is one technique that has recently shown promise for antibiotic elimination because of its ease of use, high efficiency, and affordability (Jung et al., 2013).
Adsorption is a technique that can be used to remove pollutants that cannot be easily biodegraded from water (Vijayaraghavan and Yun, 2008). In adsorption methods, the amount of substance bound to mass depends not only on the type of mass and substance but also on physicochemical factors such as concentration, temperature, mass amount, and solution pH (Volesky, 2003). When the studies are examined, diatomaceous earth (Stromer et al., 2018), nano-hydroxyapatite modified biochar (Li et al., 2020), iron and manganese oxide-modified corn straw (Yin et al., 2018), chitosan/cellulose nanocomposite microspheres (Luo et al., 2019), goethite modified biochar (Guo et al., 2016) were used as biomass in the removal of tylosin from wastewater.
In this study, the removal of tylosin, a macrolide antibiotic commonly used to protect animal health and promote animal growth, from wastewater by adsorption method was investigated. WSs found as waste in the environment were used for the removal of this antibiotic. For this purpose, quantitative determinations were carried out in high-performance liquid chromatography (HPLC). In addition, adsorption studies will be examined in continuous and batch systems, and the advantages of these two methods will be discussed. Thus, a new biomass usage was provided for the removal of environments contaminated with tylosin antibiotics.
Material and method
Materials
WSs used as an adsorbent in the study were collected from the Bafra/Samsun region. WSs were washed two times with pure water to remove their impurities and then dried in an oven at 50 °C. The dried WSs were ground with a grinder and sieved with a 150 µm sieve.
Chemicals and instrumentation
A stock solution of tylosin (Figure 1) at a concentration of 100 mg/L was prepared, and other concentrations to be used in the study were prepared by diluting ultrapure water from these solutions. The mobile phase was prepared at a mixing ratio of 40–60% acetonitrile to ultrapure water (20 mM ortho-phosphoric acid) and adjusted to pH 2.8 by buffering with 1 M NaOH solution. NaOH (1.0–0.1 mol/L) and HCl (1.0–0.1 mol/L) solutions were used to modify the pH of the solutions. The antibiotics and all other chemicals used in the study were obtained from Sigma-Aldrich. Wastewater is a mixture of chemical and metallic waste materials collected from different factories operating in various production types such as paper, fertilizer, textile, and metal in the organized industrial zone of Çorum/Turkey (Bilgin et al., 2018).
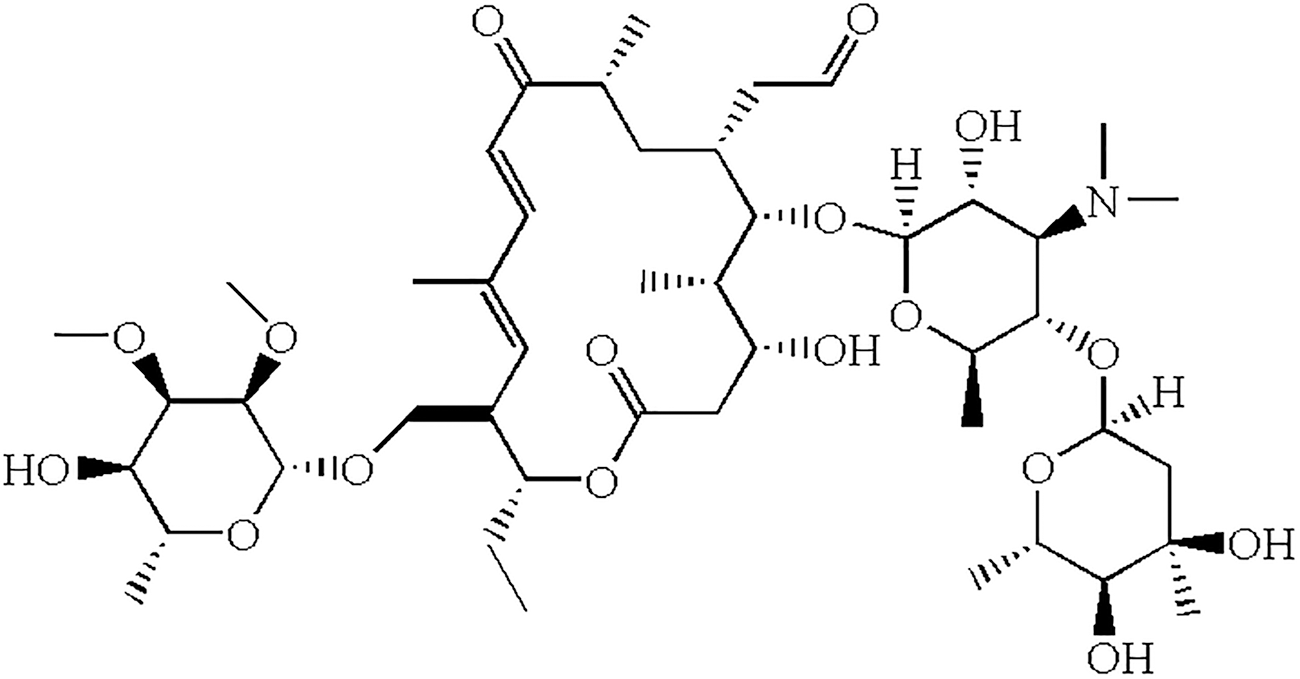
Molecule structure of tylosin.
The study determined quantitatively using the Shimadzu (Kyoto, Japan) brand HPLC device. The system includes a pump (LC-20 AD), detector (SPD-M 20A), column oven (CTO 20 AC), and degassing unit (DGU 20 A). Depending on the device, Terra RP C-18 (250 mm × 4.60 mm ID, 5 µm) column was used, and the column temperature was set to 35 °C. The solution flow rate was 1 mL/min (Paíga et al., 2019; Prats et al., 2001). The average pore radius and total pore volume were determined using a Quantachrome brand IQ-Chemi model BET device. Thermo Scientific brand Nicolet 6700 model FTIR was used to analyze molecular structures. Images of adsorbent surface morphologies and dimensions were obtained using an FEI-brand Quanta 450 FEG model SEM. XRD analyses were performed with a Rigaku SmartLab brand X-ray diffractometer in the reading range of 5–80° (2
Adsorption studies
This study investigated adsorption conditions in batch and continuous systems to remove Tylosin from the solution medium using WS. For the batch system, optimum conditions were determined for pH, adsorbent amount, time, and adsorbate concentration. Investigations were conducted on the column system's adsorbent quantity and flow rate characteristics. Calculations were performed using data from experiments using a few kinetic and isotherm models.
The antibiotic solution's pH was varied from 2.5 to 9.5 to examine how pH affects adsorption capacity. The adsorbent quantity was worked in the range of 2–20 g/L in the batch system and 10–40 g/L in the column system to examine the impact of the adsorbent amount on adsorption capacity. Contact time was studied in the 5–90 minutes range, and antibiotic concentration was studied in the range of 2–60 mg/L.
The pseudo-second-order, pseudo-first-order, and the intra-particle diffusion equation kinetic models were used to assess the data derived from the experimental findings. Under ideal adsorption conditions, the impact of salt content on adsorption effectiveness was examined using solutions with a 0.01–0.15 mol/L NaCl concentration. At a rate of 0.1–4.0 mL/min, the impact of flow rate on adsorption in the column system was explored. Adsorption capacity and adsorption efficiency (%) were determined using the formulas in Table 1:
Adsorption effiency and capacity.

Results and discussions
Characterization
The surface area of the adsorbent before adsorption was found to be as high as 49.920 m²/g. According to this result, we can say that there are many active sites on the surface of the adsorbent and that there is a large area where pollutants can bind. After adsorption, the surface area decreased to 5.954 m²/g. This decrease shows that tylosin covers the pores and the surface of the WS during the adsorption process. Before adsorption, the pore volume is relatively high at 10.330 cc/g. This high volume indicates that the adsorbent offers a sizeable void space in its internal structure and can take many contaminants into its pores. After adsorption, the pore volume decreased to a meager value of 0.049 cc/g. This dramatic decrease indicates that tylosin molecules almost fill the pores of WS. Before adsorption, the pore diameter was measured as 18.750 Å. This value indicates that the adsorbent has a mesoporous structure and can adsorb medium-sized molecules. After adsorption, the average pore diameter decreased to 16.944 Å Table 2.
BET analysis data of WS before and after adsorption.

This decrease in pore diameter indicates that the pores are filled during adsorption, and the pollutants bind to the pore walls, causing the diameter to partially narrow. FTIR analysis was performed to interpret and evaluate the presence and effectiveness of the functional groups of the adsorbent (WS) used in the studies. Figure 2 shows FTIR analysis before and after adsorption (Table 3). FTIR analysis values and corresponding functional groups are given.

FTIR spectrum of WS before adsorption and after adsorption.
FTIR analysis values and corresponding functional groups.

When we look at the peaks at 3350–3340 cm−1, we can say that the density of OH groups increased significantly after adsorption. When the peaks in 2a and 2b are compared, it can be said that tylosin molecules adsorbed onto WS due to the difference in the peaks. The peaks observed at 1031 cm−1 (3a) and 1033 cm−1 (3b) can be interpreted as the bending vibration peaks of the C–O side groups. The difference between these peaks indicates that the adsorption has been successful.
SEM micrographs of WS were captured before (Figure 3(a)) and after adsorption (Figure 3(b)). The SEM analysis data showed that the surface morphology displayed a high surface area, porous, and indented structure. The SEM images of WS upon adsorption (Figure 3(b)) revealed that a significant portion of the binding sites of WS was coated in tylosin molecules.

SEM pictogram of WS (a) before adsorption (scale bar-5 μm, ×5.000) (b) after adsorption (scale bar-10 μm, ×1.000).
According to the XRD analysis results of WS in Figure 4(a), distinct peaks were observed at 15.74°, 21.68°, and 34.42° angles. Peaks at these angles indicate the presence of amorphous and crystalline structures on the surface of the WS (Grubb and Jelinski, 1997). In the XRD analysis of WS after tylosin adsorption given in Figure 4(b), distinct peaks were observed at similar angles (15.86°, 21.62°, and 34.67°). However, there were significant decreases in the intensities of these peaks. The decrease in peak height, especially at the 34.67° angle, can be explained by the fact that tylosin molecules fill the active sites on the surface of the WS and partially mask the crystal structure. It can be said that adsorption creates disorder in some crystal phases on the surface of the WS.
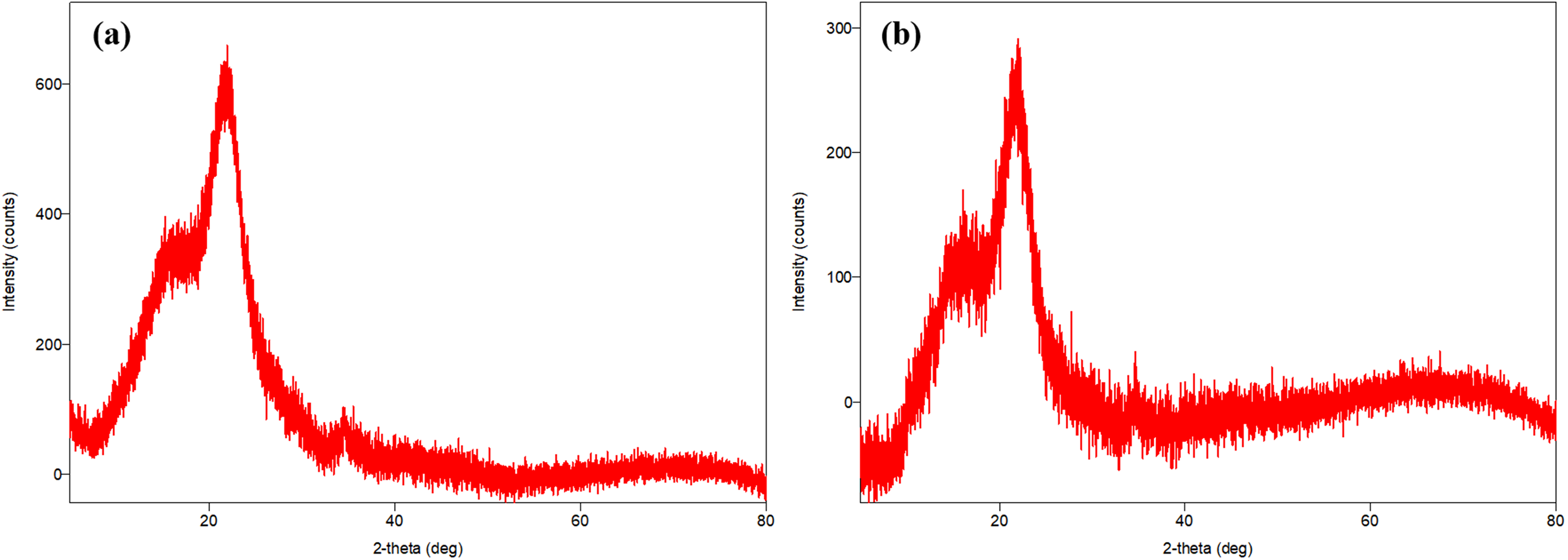
XRD patterns of (a) WS, (b) tylosin after adsorption WS.
Adsorption studies in the batch system
We examined the ideal circumstances for adsorption research in the batch system. To this end, we conducted real wastewater studies and investigations into solution pH, adsorbent quantity, contact time, adsorbate concentration, and salt effect.
Effect of pH
The effect of solution pH is a critical parameter in adsorption studies. The effect of pH on tylosin adsorption onto WS was investigated in the pH 2.5–9.5 range. Adsorption capacity values against changing pH were plotted in Figure 5(a).
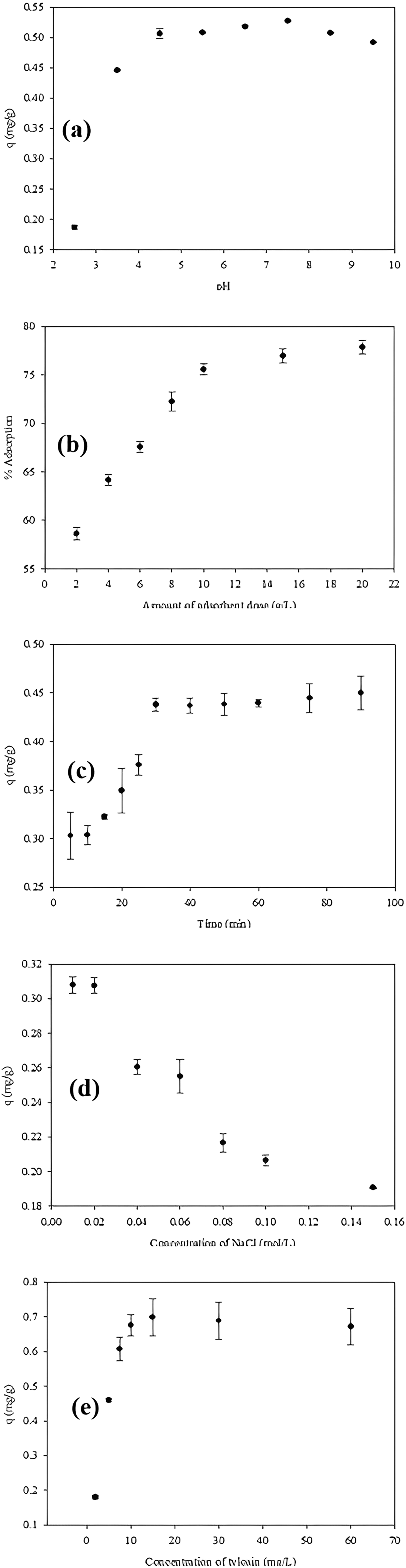
(a) pH effect, (b) adsorbent dose, (c) interaction time, (d) salt effect, (e) adsorbate concentration on adsorption.
Adsorption capacity increased in the pH range of 2.5–4.5, remaining almost constant after pH 4.5. The pH dependence of adsorption can be explained by the electrostatic attraction force between the functional groups on the surface of the adsorbent and the tylosin molecules. Since the negative charge density on the surface of the adsorbents increases with increasing pH, the probability of tylosin ions binding to the adsorbent also increases. At low pH values, tylosin ions become difficult to bind to the surface due to the positive charge density on the surface of the adsorbents, and the adsorption efficiency is low.
Effect of adsorbent dose
The effect of adsorbent amounts on adsorption in the batch system was evaluated in the range of 2–20 g/L. The % removal graph against increasing WS amount is shown in Figure 5(b).
When the amount of adsorbent was increased from 2 g to 10 g/L, the adsorption efficiency rose from 58% to 78%. From 10 g/L on, it was nearly steady. The increased amount of adsorbent allows tylosin ions to adhere to a larger surface area, which explains the current scenario. The adsorbent surface reaches saturation, and the adsorption efficiency stabilizes after a specific quantity.
Effect of interaction time
The effect of contact time on the adsorption of tylosin antibiotic with WS was investigated between 5 and 90 min. The graph from the experimental data is given in Figure 5(c), and the adsorption capacities are evaluated.
The adsorption capacity improved in the first 30 minutes and stayed nearly constant as the contact time increased. The adsorption equilibrium was established in under 30 minutes.
Effect of salt
In real wastewater conditions, there is a significant amount of salt. Therefore, the effect of ionic strength (also known as salt effect) on the adsorption of tylosin in WS is an important parameter to investigate. It was investigated using antibiotic solutions containing 0.01–0.15 mg/L NaCl under ideal adsorption conditions.
As shown in Figure 5(d), a slight decrease in adsorption capacity was observed in the removal of antibiotic with increasing NaCl concentration.
Effect of adsorbate concentration
In adsorption studies, the extent to which contaminants are removed from an aqueous solution depends largely on the initial sorbate concentration. Adsorption of the antibiotic tylosin initially increased with increasing dye concentration, then reached saturation at a certain point in Figure 5(e).
Kinetic models
To describe the adsorption mechanism, numerous kinetic models, including mass transfer and chemical reaction processes, have been created in adsorption studies. The most widely used kinetic models to explain the adsorption mechanism are the pseudo-first-order (Lagergren et al., 1898), pseudo-second-order (Ho and McKay, 1998), and the intraparticular diffusion (Weber and Morris, 1963) kinetic models (Tables 4 and 5).
Kinetic model equations.

Kinetic model parameters.

To learn more about the dynamics and control mechanism of the adsorption processes, the time-dependent data for antibiotic adsorption with WS were assessed using pseudo-first-order, pseudo-second-order, and intra-particle diffusion kinetic models. Table 6 displays the equation constants and
Calculated kinetic model parameters.

When Figure 6(a), Figure 6(b), and Figure 6(c) are examined, the adsorption is in accordance with the pseudo-second-order kinetic model. In addition, the R2 values (0.999) given in Table 5 also support the fact that the process is by the pseudo-second-order kinetic model. The q values calculated from this model are also by the experimental values. On the other hand, the
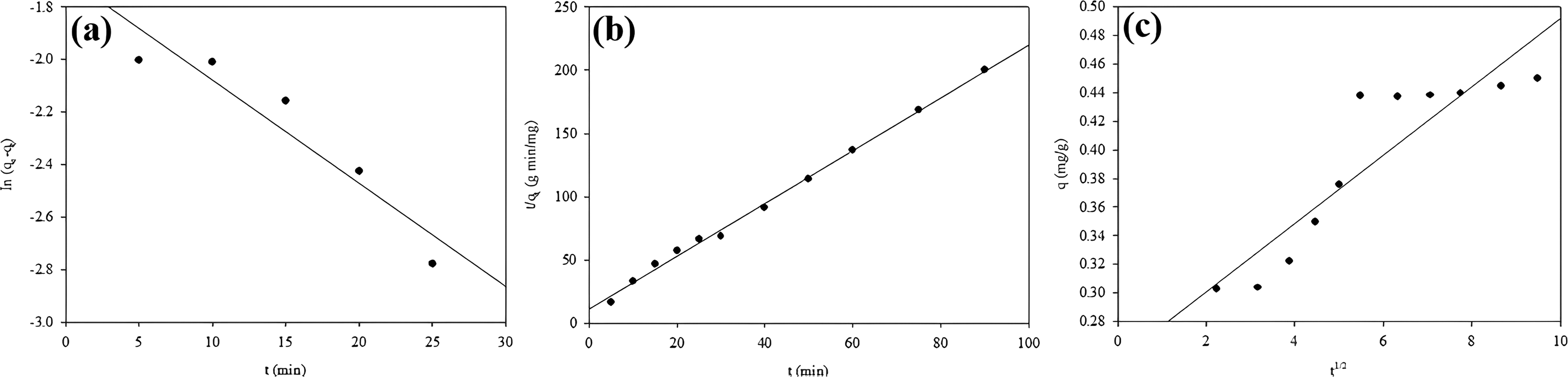
(a) Pseudo-first-order kinetic model, (b) pseudo-second-order kinetic model, (c) intra-particle diffusion model.
Isotherm models
Adsorption isotherms are essential because they describe the interaction between sorbate and adsorbent. They help to find the relationship between the amount of sorbate adsorbed on the adsorbent and the sorbate concentration in the liquid at equilibrium. The literature contains various equilibrium isotherm models. In this study, the data obtained from the experimental study of sorbate concentration for tylosin adsorption were applied to Freundlich (Freundlich, 1907), Langmuir (Langmuir, 1918), and D-R (Dubinin and Radushkevich, 1947) isotherm models.
Experimental data for the adsorption of the antibiotic tylosin onto WS were calculated using the isotherm model equations in Tables 7 and 8.
Isotherm model equations.

Isoterm model parameters.

The
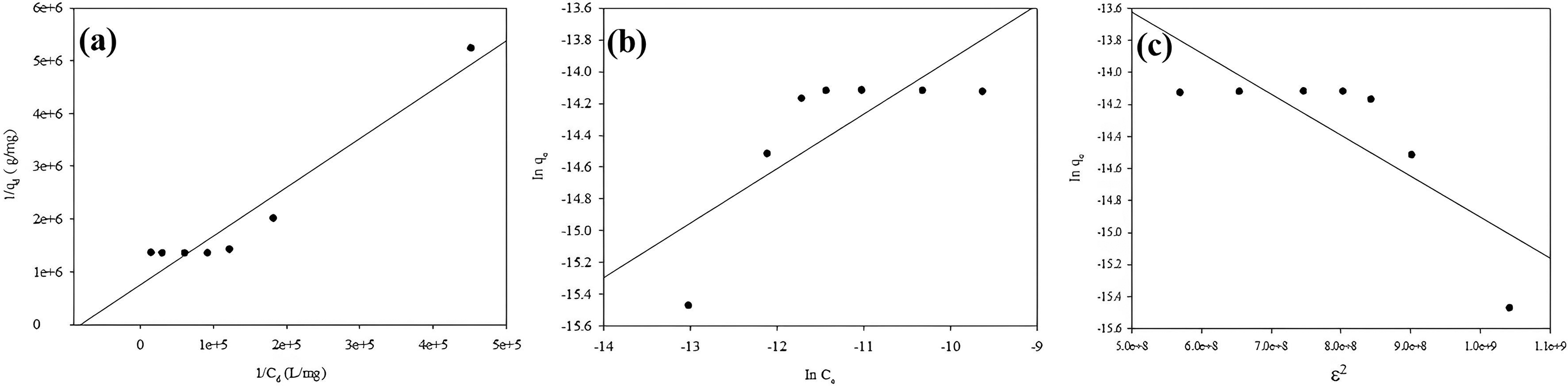
(a) Langmuir isoterm model, (b) Freundlich isoterm model, (c) D–R.
Calculated isotherm model parameters.

Adsorption studies in the column system
The adsorption process is separated into batch and continuous systems based on the application methods. In industrial-scale applications, studies of continuous system adsorption are more straightforward and useful. As a result, WS's adsorption potential in the continuous system was also studied. For this reason, the ideal WS concentration and flow rate for tylosin adsorption in the continuous system were established.
Adsorbent dose at column system
In order to investigate the effect of the amount of adsorbent in the column on the adsorption efficiency, WS was used in amounts ranging from 10 to 40 g/L. Adsorption efficiencies are shown in Figure 8(a). As the amount of WS in the column increased, the adsorption efficiency remained constant after a certain point. After 30 g/L of WS, the increase in the amount of adsorbent in the column did not cause any increase in adsorption efficiency.
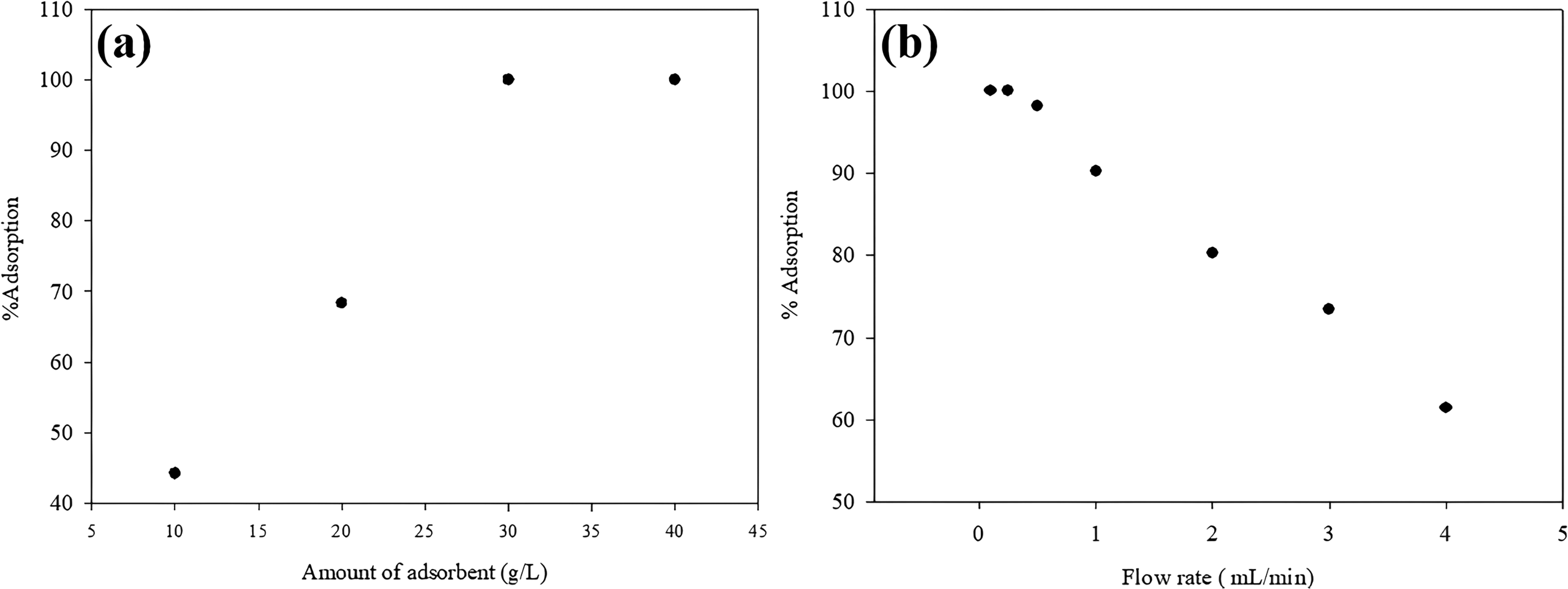
(a) Adsorbent dose (b) flow rate in column system.
Flow rate
To determine the most suitable flow rate for tylosin adsorption in the continuous system, solutions with the optimum adsorbent amount and pH were passed through the column at 0.1–4.0 mL/min. Figure 8(b) shows the effect of flow rate on adsorption yield.
As the flow rate increased, a significant decrease in adsorption efficiency was observed. According to these results, the optimum flow rate was determined as 0.2 mL/min.
Wastewater studies
The wastewater environment contains many different types of pollutants. To investigate the effect of adsorption efficiency by other pollutant ions in the environment, an adsorption study was carried out with wastewater containing tylosin antibiotics under optimum adsorption conditions. Under these optimum conditions, the adsorption efficiency of tylosin antibiotic in wastewater was 72.26%. In adsorption studies, this efficiency was found to be 78.16%. Although the efficiency in removing tylosin in wastewater decreased slightly, a high removal was achieved.
The adsorbents prominent in the literature for removing tylosin antibiotics from the solution medium are given in Table 10. Regarding % removal efficiency, nano-hydroxyapatite-modified biochar (Li et al., 2020) and WS stand out as the most successful adsorbents. However, establishing the adsorption equilibrium of nano-hydroxyapatite-modified biochar over a very long time, such as 72 hours, may limit its practical use. In addition, the fact that it does not require any process to prepare WS natural adsorbent provides a great advantage. In the literature, batch system adsorption studies are generally encountered in removing tylosin. This study investigated both batch system and continuous system adsorption studies of tylosin. Due to their ease in industrial applications, column systems are essential in adsorption studies. In this respect, the study is one of the leading examples examining the column system adsorption efficiency for industrial applications in removing tylosin from water. In this respect, it is thought to contribute significantly to the literature. In the adsorption study using WS, 100% antibiotic removal was achieved.
Comparison of walnut shells and other adsorbents in tylosin removal.

Using WS in batch and column systems provides high removal efficiency in short contact times and may offer a lower-cost solution. These results indicate that WS may be an effective alternative for tylosin removal in both batch and column systems, especially for low cost and shorter processing times.
Conclusions
This study examined adsorption parameters in batch and continuous systems to remove the antibiotic tylosin from an aqueous media using WS. pH, amount of adsorbent, mixing duration, initial concentration of antibiotics, and salt effect were studied in batch systems; adsorbent amount and flow rate effects were studied in continuous systems. The adsorbent was characterized using BET, FTIR, SEM, and XRD.
The optimum pH was determined as the original pH of the antibiotic solution (pH 7.5). The fact that there is no need for pH adjustment for solution pH, which is the most important parameter of adsorption studies, and that it is suitable for working in a wide pH range provides a great advantage. The adsorption equilibrium was reached in the batch system in a mere 30 minutes or less. This is a cost-effective and time-efficient situation. Adsorption equilibrium was reached in the batch system in just 30 minutes, which is very short. This is cost-effective and time-efficient.
The optimum conditions in the column system were found to be 30 g/L and 0.2 mL/min. Under these conditions, 100% removal was achieved. Removal of 78% was achieved under optimum adsorption conditions in the batch system. It can be concluded that WS is a highly effective and low-cost adsorbent in the removal of tylosin from wastewater.
Footnotes
Author contribution
B.A and İ.T.S. carried out the experiment. B.A. and İ.T.S. wrote the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Projects Department of Hitit University (Project no: FEF19001.20.004).
