Abstract
A large amount of wastewater with a high dye content is discharged from the textile printing and dyeing industry. Synthetic dyes, which are essentially exogenous chemicals, predominantly exhibit the property of poor biodegradability. Consequently, they are capable of persisting stably within the environment over protracted time spans. The high-chroma dye wastewater not only results in severe water pollution but also breaks ecological balance, thereby rendering it a pivotal and formidable facet in the realm of industrial wastewater treatment. Consequently, the treatment of printing and dyeing wastewater prior to its discharge is of utmost necessity. This article offers a relatively comprehensive exposition of the treatment methods for dye wastewater, with a specific focus on the adsorption method, the photocatalysis method, and their respective characteristics. Nano-TiO2@adsorbent composites, which integrate the advantages of adsorption and photocatalysis, have been widely studied for the treatment of dye wastewater. This paper provides a broad overview of the classifications, the adsorption-photocatalytic mechanism, and influencing factors of nano-TiO2@adsorbent composites. Nano-TiO2@adsorbent composites integrate the processes of adsorption, catalysis, and degradation, thereby significantly improving the efficiency of photocatalytic degradation for organic pollutants by titanium dioxide catalysts. Furthermore, the suggestion for the research and development of photocatalyst @textile composite materials for dye wastewater treatment is put forward in this article.
Introduction
Water resources constitute the bedrock of the natural ecosystem, and the tenet that “water is life” has become the consensus of mankind. 1 As early as 1927, Hoover Herbert, an American scholar, proposed that the scientific utilization of water could bring a bumper harvest of wealth and happiness to life. For human beings, water resources are of utmost significance for ensuring food security and disease prevention, involving all aspects of life such as agricultural irrigation, drinking, cooking and cleaning. In addition, water resources manifest a pivotal role in the realm of socio-economic development. In 2010, the United Nations declared that the safe and clean drinking water was an essential human right. Public health, urbanization and industrialization are all affected by water resources. 2 For wildlife, water serves not merely as a vital resource but also as an irreplaceable habitat. Therefore, water resources are deemed indispensable for the attainment of global sustainable development.
Despite the extensive distribution of water resources all over the world, the scarcity of freshwater has emerged as a formidable global challenge. 3 The Food and Agriculture Organization of the United Nations (FAO) and the World Water Council predict that 67% of the global population will encounter the water scarcity issue by 2050. 4 This scarcity poses a significant contradiction to the socio-economic development. The rational utilization of water resources has emerged as an indispensable prerequisite for sustainable development. 5 With the ever-increasing requirements of economic development, the scale of industrialization is expanding day by day. A large amount of industrial wastewater containing harmful substances is being discharged. This not only inflicts severe damage on the ecological environment but also poses a grave threat to human health due to the emergence of novel diseases.6,7 Consequently, the effective purification of industrial wastewater is of utmost significance for the harmonious coexistence between humanity and nature.
The textile industry, which is intricately intertwined with human lives, occupies a pivotal position in the global economic landscape. The data presented in Figure 1, sourced from The Sustainable Angle organization, reveals that the water consumption is substantial within the textile industry every year. 8 More importantly, the textile industry consumes approximately 10,000 tons of dyes annually during the printing and dyeing process. 9 Owing to incomplete adhesion of dyes to fabrics, a portion of the dyes is discharged into the wastewater. The proportion of discharged dyes ranges from 2% in the case of basic dyes to 50% in the case of reactive dyes, rendering textile wastewater highly chromatic and recalcitrant to biodegradation.10,11 Dye molecules impede the transmission of light and disrupt the process of photosynthesis. This leads to a decrease in the dissolved oxygen content in the water body and an increase in biological oxygen demand, thereby breaking the balance of the ecosystem. The majority of synthetic dyes are toxic in nature. After being discharged with wastewater, they induce water eutrophication and inflict damage upon aquatic organisms and their habitats. Synthetic dyes present in water are easily assimilated by fish and other organisms. When they infiltrate the human food chain, the human body will be affected by toxic substances and cause many diseases. The detrimental effects of several common textile dyes are listed in Table 1.
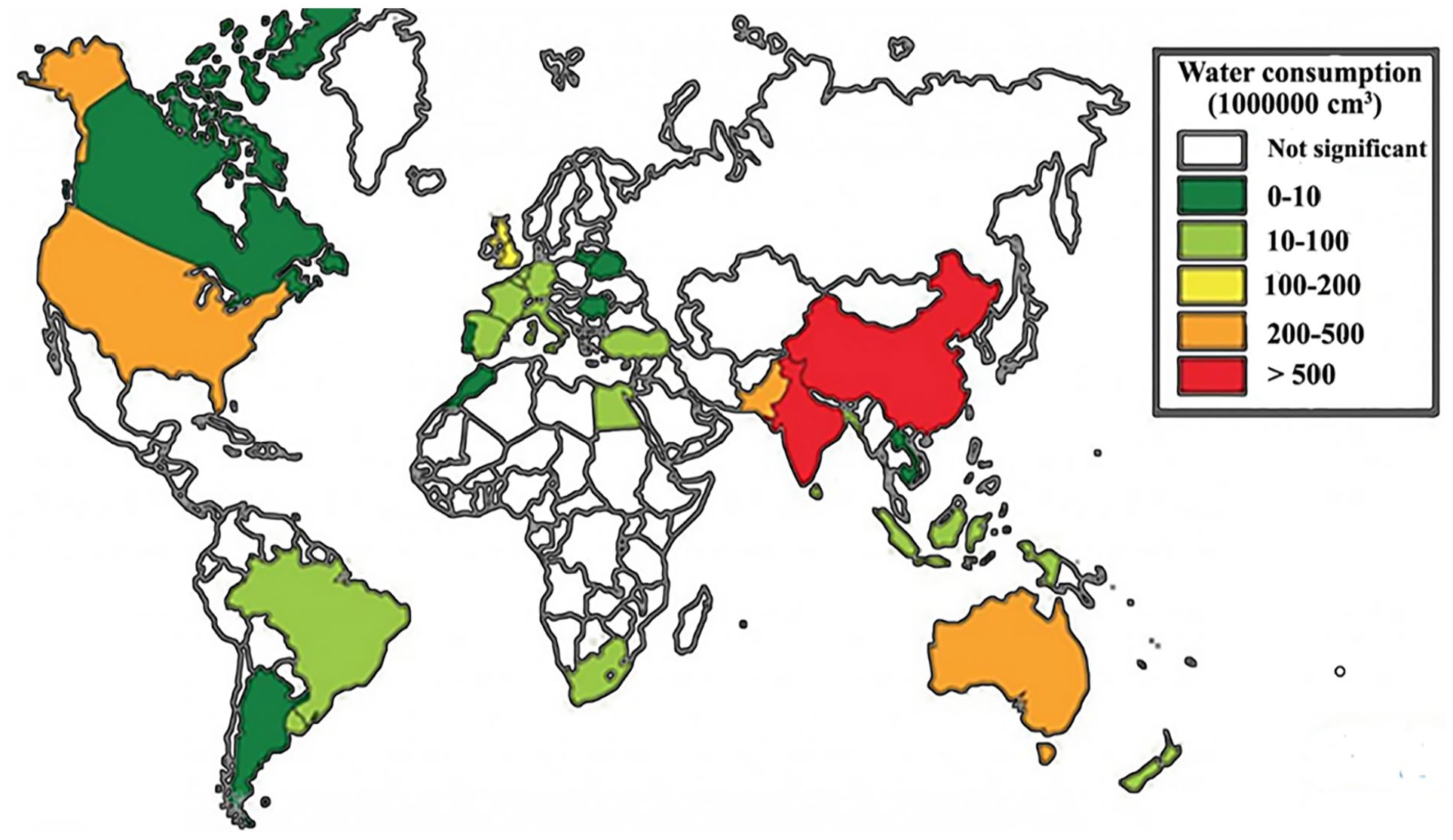
Annual water consumption of major textile industries in the world. 8
Harmful effects of several synthetic dyes commonly used in textile industry.

The color of clothing is required to be both esthetically appealing and resistant to the effects of washing. Consequently, the molecular structure of synthetic dyes is complex and stable, which mainly consists of two key components: the chromogenic group mainly responsible for fabric dyeing, and the auxochrome group, such as halogen, —SO3H, —NH2, —OH, —COOH, and —CH3. The auxochrome group can improve the water solubility of dyes and augment the affinity between fibers and dyes by receiving electrons or releasing electrons. 24 Since Perkins unexpectedly discovered aniline violet in 1856, there have been over 10,000 types of synthetic dyes worldwide, with an annual production of approximately 700,000 tons. According to the particle charge upon dissolution in water, synthetic dyes can be divided into cationic dyes (alkaline dyes), anionic dyes (acidic, direct, and reactive dyes), and nonionic dyes (dispersed dyes). Dyes may also be classified based on their chromogenic groups, that is, azo dyes, anthraquinone dyes, arylmethane dyes, indigoid dyes, sulfur dyes, phthalocyanine dyes, and nitro and nitroso dyes, as shown in Figure 2. 25 In the dye market, azo dyes account for 60%–70% of synthetic dyes due to their bright colors and high color fastness. They have nearly 2000 varieties and the largest production, and are widely used in the printing and dyeing industry.26,27
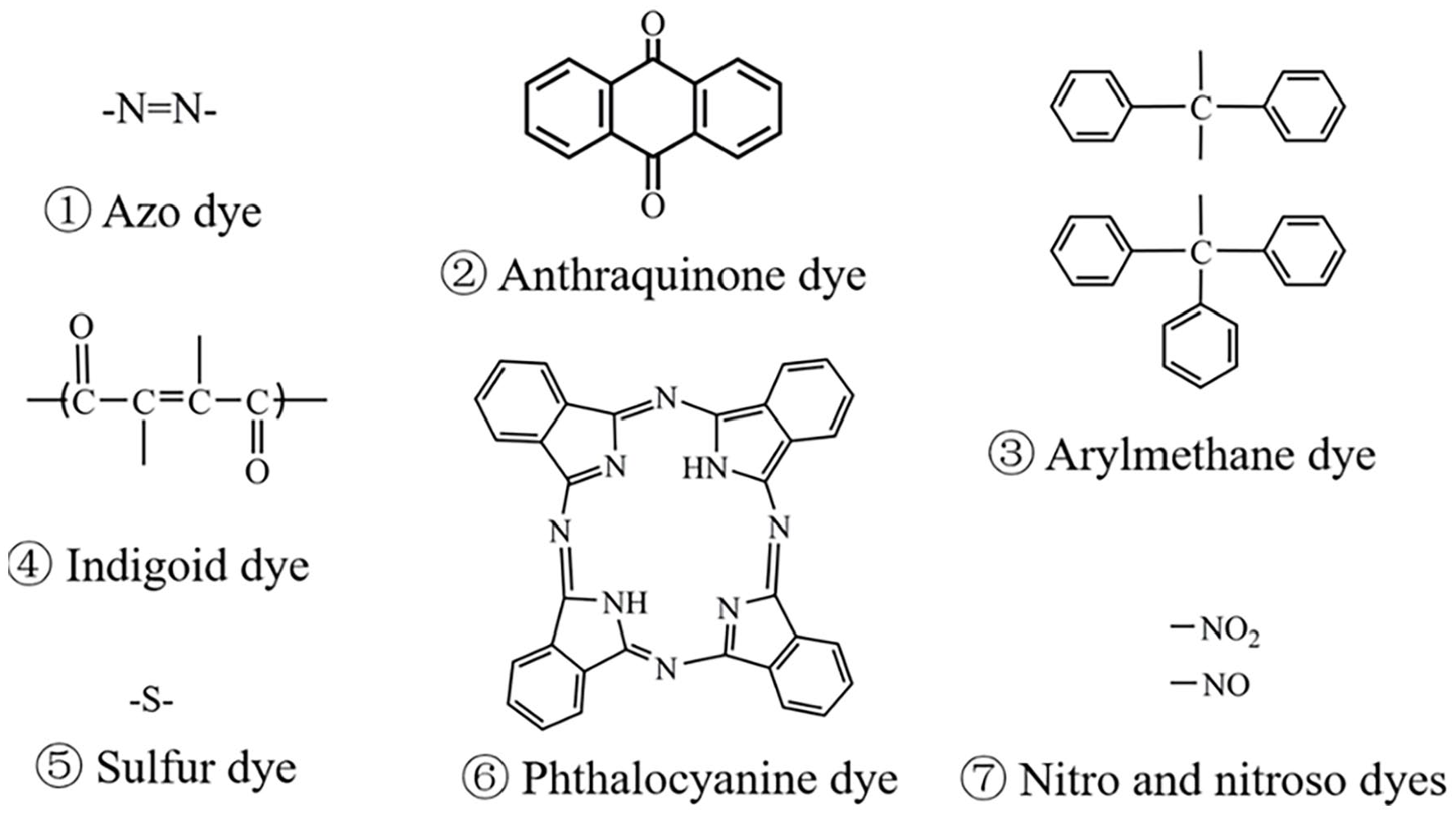
Classification of dyes and their chromophore structure.
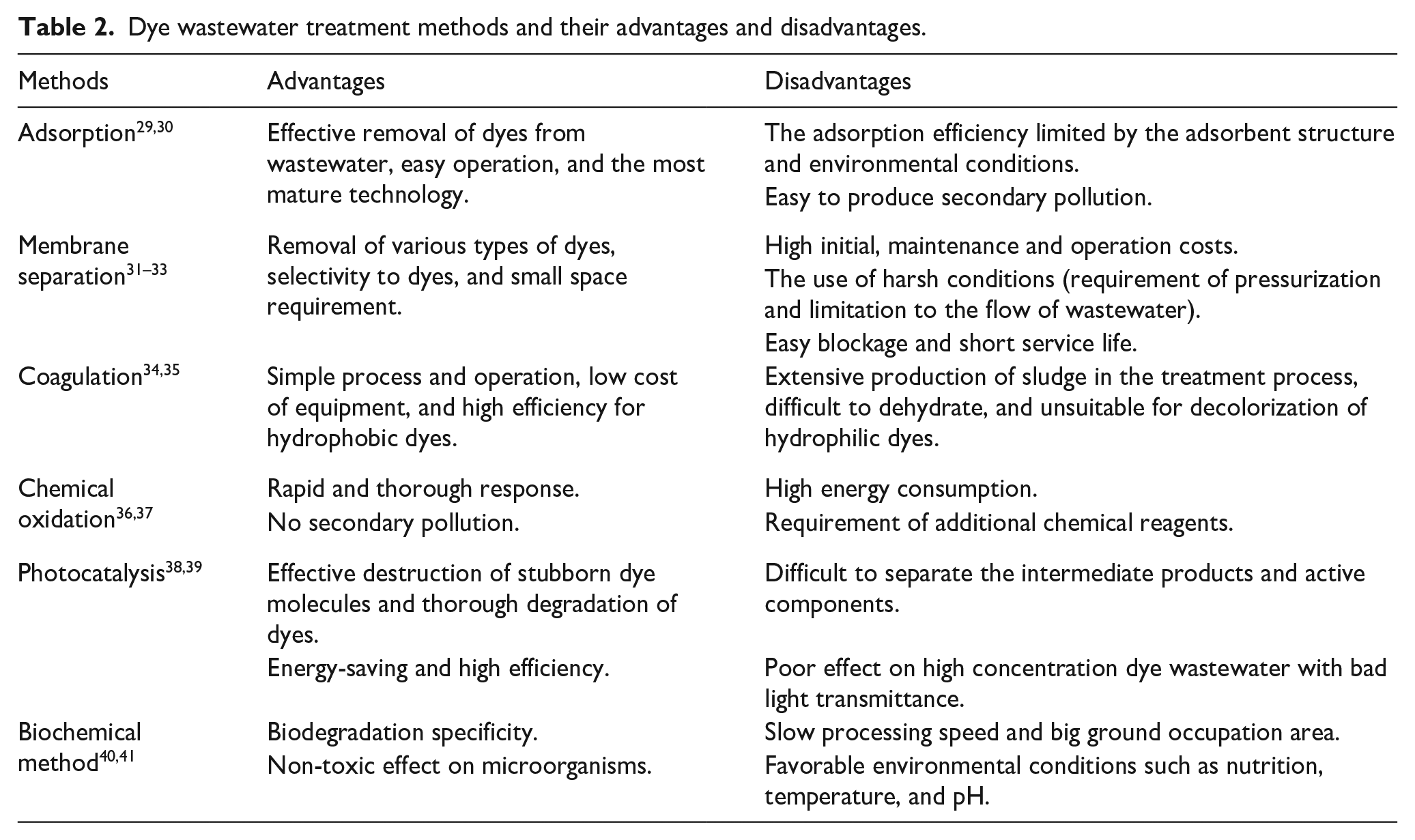
Synthetic dyes, in most cases, are exogenous chemicals with poor biodegradability, persisting in the environment for an extended period. Usually, it is difficult to completely remove the dyes from wastewater by traditional treatment methods. Therefore, specialized treatment methods are necessary. Based on the principle of dye removal, the methods can be classified into physical methods (such as adsorption, coagulation, membrane separation, etc.), chemical methods (such as oxidation, catalysis, reduction, etc.) and biological treatment methods.16,28 Each of these methods has its own advantages and limitations, as illustrated in Table 2.
Dye wastewater treatment methods and their advantages and disadvantages.
Adsorption
Adsorption process is the deposition of substances at the interface of two phases. The substance accumulated on the interface is called adsorbate, and the solid on which substances are adsorbed is called adsorbent. 42 It can be seen from Table 2 that adsorption method is one of the effective and convenient technologies for removing dyes from wastewater. 43 Its process is superior to other wastewater treatment technologies in terms of design flexibility, simplicity and operational simplicity. 44 Almost all types of dyes can be removed by this method. 45 Colorless and odorless high-quality water can be produced by adsorption without causing the formation of harmful substances. However, the adsorption method only transfers the dyes in the treated wastewater, which must be regenerated or destroyed later. Otherwise, secondary pollution will be caused easily. In addition, the cost of adsorbents is also an important consideration in the use of adsorption method. Locally available, efficient and cheap raw materials as well as their processed products can be used to remove dyes from wastewater. With the emergence and continuous development of such kind of adsorbents, the adsorption method has become an economically feasible dye removal method all over the world. 46
Types of adsorbents
There are quite a few kinds of adsorbents. The main types of adsorbents classified by the chemical composition are shown in Figure 3.
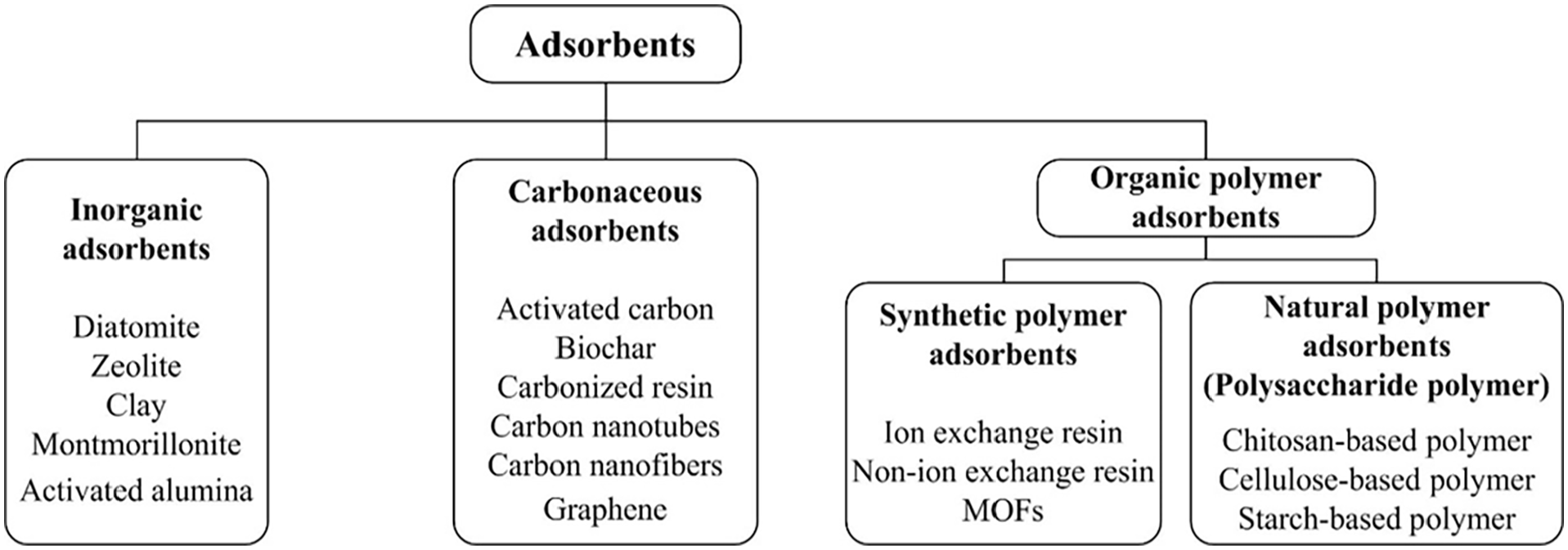
Classification of adsorbents.
Inorganic adsorbents
Inorganic adsorbents represented by diatomite, zeolite and clay are significantly lower in price than activated carbon. These materials have the characteristics of high specific surface area and high porosity.47–49 Therefore, the dyes in the solution can be enriched by physical adsorption. Diatomite is a soft, light-weight natural sedimentary rock mainly composed of micro-amorphous silica (SiO2·nH2O). 50 A large number of silicon hydroxyls are distributed on the surface and the inner surface of the pores. These silicon hydroxyls dissociate H+ in the aqueous solution, so that the diatomite particles exhibit a certain surface negative charge. Generally, zeolite is composed of silicon-oxygen tetrahedron and aluminum-oxytetrahedron, and clay is composed of silicon-oxygen tetrahedron and aluminum-oxyoctahedron, respectively. They have a constant structural negative charge due to the isomorphic substitution of Al3+ for Si4+. 51 Therefore, the adsorption capacity is obviously affected by the type of dyes. Inorganic adsorbents show good adsorption performance for cationic dyes, but poor or even no adsorption for non-ionic and anionic dyes. Activated alumina is also a widely-studied inorganic adsorbent.52,53 Generally, anionic surfactant is used to chemically modify alumina, and the obtained activated alumina is used for the adsorption of cationic dyes.
Carbonaceous adsorbents
Carbonaceous adsorbents mainly include activated carbon (AC),54–56 biochar,57,58 carbonized resin, 59 carbon nanotubes,60,61 carbon nanofibers,6,62,63 and graphene.59,64 AC, biochar, and carbonized resin are relatively traditional carbonaceous adsorbents, especially AC is widely used in dye wastewater treatment. These materials mainly rely on their porous structure and high specific surface area to realize the adsorption of dyes. In addition, oxygen-containing groups are present on their surfaces. The dyes can be adsorbed on the surfaces via hydrogen bonds between the oxygen-containing groups and the dye molecules. In order to further improve the adsorption performance of carbon materials, the structure was optimized, and carbon nanomaterials, carbon nanotubes, and carbon nanofibers, appeared. Carbon nanomaterial adsorbents have greater advantages in specific surface area and pore structure, thus exhibiting better adsorption performance compared with traditional carbonaceous adsorbents. In recent years, the newest carbon allotrope, graphene, has emerged, which is a monolithic two-dimensional carbon material. Graphene has randomly distributed sp 2 carbon atom hybrid aromatic ring regions, which can be combined with aromatic dyes through π-π stacking, but the strength of this interaction is much lower than that of electrostatic interaction. 65 Carbonaceous adsorbents can adsorb water-soluble dyes, but their adsorption selectivity is poor due to the lack of functional groups. And there is a problem of high cost, which limits the large-scale use of carbonaceous adsorbents.
Organic polymer adsorbents
At present, organic polymer adsorbents are more popular on the market. Among them, resin-based synthetic polymer adsorbents with different structures can be obtained by adjusting the synthetic procedures. Due to the different synthetic monomers and functional groups carried, there are thousands of types of such adsorption resins. Therefore, their application range is wider than that of inorganic and carbonaceous adsorbents. Ion exchange resins and non-ionic exchange resins sold on the market are their representatives.66–69 Many scholars in the world have conducted in-depth research on the adsorption behavior of resin-based synthetic polymer adsorbents for various dyes, and tried to use them as substitutes for AC and inorganic adsorption materials. However, synthetic polymer adsorbents also have their disadvantages, that is, high cost and poor environmental protection in the production and processing. Most importantly, since the adsorbents are synthetic polymer compounds in nature, it is difficult to degrade them. Therefore, the waste resins that exceed the number of uses will become secondary solid waste. Metal-organic frameworks (MOFs) are polymer materials obtained by connecting metal clusters with organic ligands. These materials have high specific surface area and porosity, so they show high adsorption capacity for dyes.70–72 However, due to the excellent hydrophilicity of MOFs, few reports indicate their long-term stable existence in aqueous solutions, especially for metal nodes. So the use of MOFs as conventional adsorbents needs further exploration.
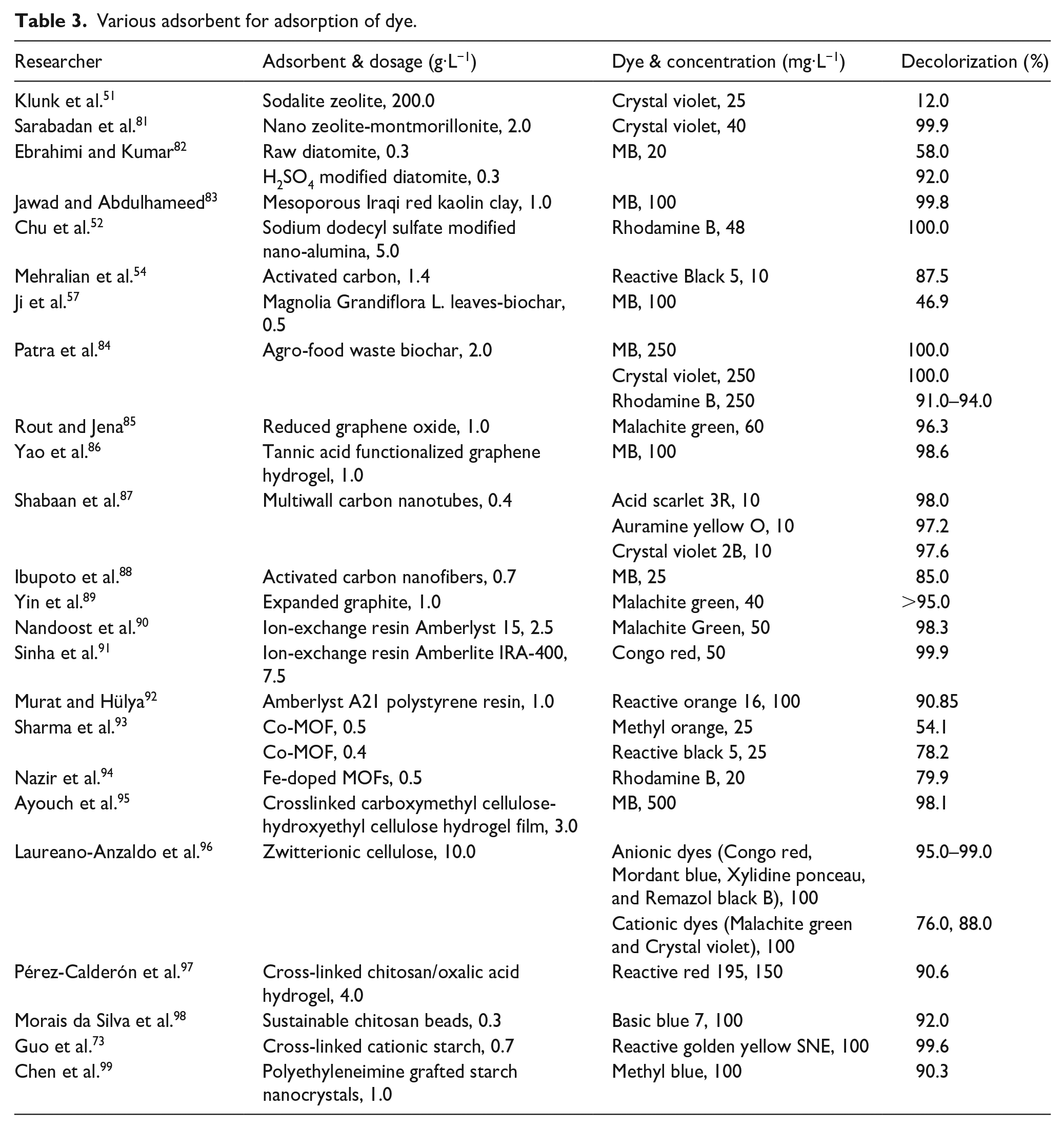
Natural polymer adsorbents mainly use some polysaccharides as the matrix, represented by-based, cellulose-based, and starch-based adsorbents.73–79 Similar to synthetic polymer adsorbents, these materials can carry various functional groups through further surface chemical modification to obtain different derivatives, thereby expanding their application range. They can not only adsorb and separate substances like inorganic adsorbents by ion exchange and the like-dissolves-like principle, but their adsorption mechanisms also include chelation, charge gravitation, van der Waals force, dipole force, and hydrogen bond. Not only that, because these materials are derived from nature and low in price, they are usually biocompatible and biodegradable materials that are renewable. The production process is environmentally friendly, which will not cause secondary pollution and reduce the impact on the environment. Therefore, natural polymer adsorbents have been paid more and more attention by scholars since their emergence, and are constantly developing toward the direction of eventually replacing synthetic polymer adsorbents. 80 The relevant researches on the dye adsorption performance of these types of adsorbents is shown in Table 3.
Various adsorbent for adsorption of dye.
Factors affecting adsorption performance
The adsorption process of adsorbents on dyes is influenced by many factors, such as pH, adsorption time, temperature, adsorbent dosage, initial dye concentration, and so on. Optimizing the adsorption conditions can improve the utilization efficiency of adsorbents and enhance the treatment effect of dye wastewater.
Effect of solution pH
Solution pH is one of the most important factors affecting the adsorption capacity of adsorbent in wastewater treatment. In solutions with different pH, the ionization of dye molecules and adsorbents varies, so the adsorption performance of adsorbents for dyes is significantly different. A relatively simple study is conducted to determine the optimal solution pH by comparing the adsorption capacity or dye removal rate of adsorbents for dye solutions with different pH. Senthamarai et al. investigated the effect of solution pH on the adsorption of MB by surface modified Strychnos potatorum seeds. They measured the removal rates of MB within the pH range of 2–10, and obtained the maximum removal rate at the optimal pH of 8.0. 100 Özcan et al. 101 studied the effect of solution pH ranging from 1 to 11 on the adsorption of Acid Blue 193 by BTMA bentonite, and found that the maximum removal rate of the dye was at acidic pH 1.5.
In addition, the surface charge of the adsorbent under different pH conditions was analyzed by measuring its zero point charge (pHpzc). When the solution pH is higher than pHpzc, the surface of the adsorbent is negatively charged, which is beneficial for the adsorption of cationic dyes by the adsorbent. On the contrary, the surface of the adsorbent carries positive charges, which is conducive to the adsorption of anionic dyes. 102 In the previous optimization experiment on the adsorption of reactive golden yellow SNE dye by cross-linked cationic starch, the pHpzc of cross-linked cationic starch was 6.8 measured by the pH drift method. In the subsequent adsorption experiment, the solution pH used was 5.0. 73
Effect of temperature
Temperature is another important parameter that affects the adsorption capacity of adsorbents. Generally, the adsorption process of dyes is analyzed by comparing the adsorption capacity of adsorbents at different temperature conditions to determine whether it is endothermic or exothermic. If the adsorption capacity increases with the increase of temperature, the adsorption is an endothermic process. This may be due to the enhancing mobility of the dye molecules and the increasing number of active sites of the adsorbent at higher temperature. The adsorption capacity increases with the decrease of temperature, indicating that adsorption is an exothermic process. This may be because the adsorption force between dye species and active sites on the surface of adsorbent is enhanced at lower temperature. While the adsorption capacity increases with the decrease of temperature, indicating that the adsorption is an exothermic process. This may be due to the enhanced adsorption force between dye species and surface active sites of adsorbents at lower temperature. 103
The influence of temperature can also be studied by isothermal adsorption and adsorption thermodynamics. The linear formulas of Langmuir and Freundlich isotherm adsorption models are shown as equations (1) and (2), 104 and the thermodynamic related formulas are expressed as equations (3)–(5). 105




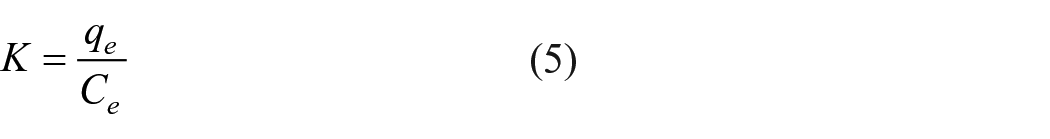
where
When the adsorption process follows Langmuir isothermal adsorption model, if
Effect of time
Time is an important factor affecting the mass transfer rate of dyes during the adsorption process. 107 At the beginning of adsorption, the adsorption rate of dye molecules by adsorbent is very fast. As time goes by, the adsorption rate gradually decreases and eventually reaches adsorption equilibrium. This is mainly due to the gradual reduction of active sites on the surface of the adsorbent as the adsorption reaction proceeds, and finally the adsorbent reaches the saturation state of adsorption capacity. Lebkiri et al. investigated the effect of adsorption time on the adsorption efficiency of crystal violet dye. The concentration of crystal violet dye rapidly decreased within 0–30 min after contact between anionic polyamide and dye solution. Beyond this time, the decrease in dye concentration slowed down and reached adsorption equilibrium within 180 min. 108 Salman et al. 109 found that the removal efficiency of chitosan coated cotton fiber composites for Remazol Brilliant Red F3B dye reached 75% within 90 min, and then the removal efficiency slowly increased, reaching equilibrium in about 150 min. It is worth noting that the time required to reach adsorption equilibrium is a key parameter in the practical application of adsorbents. The shorter the time to reach equilibrium, the higher the utilization efficiency of the adsorbent material in wastewater treatment. 110
Effect of initial dye concentration
Generally, the adsorption capacity of the adsorbent rises with the increase of initial dye concentration. Márquez et al. studied the adsorption capacity of the natural clayey composite for Basic Navy Blue 2RN dye and Drimaren Yellow Cl-2R dye with initial concentrations ranging from 10 to 60 mg·L−1. They found that the adsorption capacity raised from 0.7 to 5.2 and from 0.8 to 5.7 mg·g−1, respectively. 111 Dahlan et al. 112 reported that the adsorption capacity of modified metal-organic frame-5 increased with higher initial MB dye concentration. This phenomenon indicates that when the dye concentration is low, the active sites on the adsorbent surface cannot be fully utilized. With the increase of the initial dye concentration, the utilization rate of active sites of adsorbents increases, manifested as an increase in adsorption capacity. Nevertheless, when the active sites are almost completely occupied, that is, the adsorption capacity reaches saturation, further concentration increase cannot significantly improve the adsorption capacity.
Effect of adsorbent dosage
The dye removal rate generally increases with the increase of the adsorbent dosage. An increase in the adsorbent dosage leads to an increase in both the specific surface area and active sites, which is beneficial for the adsorption of dyes. 113 Lebkiri et al. 108 reported that increasing the dosage of anionic polyacrylamide from 0.007 to 0.039 g resulted in an increase in dye removal efficiency from approximately 60%–97%. However, increasing the adsorbent dosage can lead to a decrease in adsorption capacity. Due to the decrease in the amount of the dye adsorbed by the adsorbent per unit weight, the utilization efficiency of active sites decreases. Sadaf and Bhatti 114 varied the biosorbent dose from 0.05 to 0.3 g·50 mL−1 and the highest adsorption capacity of the biosorbent was found when the dosage was 0.05 g·50 mL−1. Therefore, in practical applications, the selection of adsorbent dosage should take into account both dye removal rate and adsorbent utilization efficiency.
Photocatalysis
In 1972, Japanese scientists Fujishima and Honda discovered the Hondo-Fujishima effect. In the experiment, titanium oxide single crystals placed in water were irradiated with light, and they found that water was decomposed into O and H. 115 Since then, photocatalysis has attracted people’s attention. By 1977, Frank and Bard in the University of Texas at Austin conducted a series of researches on the photocatalytic oxidation of cyanide around TiO2, ZnO, CdS, Fe2O3, and WO3 with a particle size of less than 1 μm, 116 making it possible to degrade sewage by photocatalysts. Photocatalysis has broad development prospects in environmental treatment, energy development and so on.
Photocatalysis generally refers to the photocatalytic reaction, which is usually composed of light absorption, photocatalysis, and the chemical changes resulting from the reaction. In 2007, the International Union of Pure and Applied Chemistry (IUPAC) defined photocatalysis as follows. Under the irradiation of ultraviolet, visible or infrared light, the photocatalyst absorbs light to change the rate of chemical reaction, the initial state, and the reaction components involved.
Photocatalyst usually refers to a compound that can absorb radiant light and be excited under the condition of light radiation, so that the reactants in the system undergo chemical transformation. While itself acts as a catalyst during the chemical reaction, it does not directly participate in the reaction or be consumed. 117 Nano-semiconductor photocatalysts are semiconductor materials with the surface effect of nano materials. Interest in nano-semiconductor photocatalysts has grown exponentially due to their potential and opportunities in various fields, including the management of environmental pollution, biosensors, medicine (cancer and virus destruction), and the pharmaceutical industry. The advantage of nano-semiconductor photocatalysis technology is that it can use renewable and pollution-free solar energy. So it can be a good alternative to energy-intensive treatment methods. Traditional pollution treatment methods transfer pollutants from one medium to another, potentially turning the pollutants into more difficult-to-degrade pollutants. The nano-semiconductor photocatalysis converts pollutants into non-toxic substances, such as CO2 and H2O, with mild reaction conditions and moderate reaction time. It can be applied to aqueous phase, gaseous phase and solid phase, which is a supplement and improvement to the existing technology. Therefore, the advantages of nano-semiconductor photocatalysts not only minimize the running cost, but also achieve the ideal effect in the most efficient way.
Nano-semiconductor photocatalysts do not have continuous electronic states like metals, but they have a band gap extending from the top of the highest occupied molecular orbital (HOMO), namely the valence band (VB), to the bottom of the lowest unoccupied molecular orbital (LUMO), namely the conduction band (CB). The size of this band gap is called energy gap (
The properties of excellent semiconductor photocatalysts are as follows. The core element of a semiconductor photocatalyst can reversibly change its valence state to accommodate holes without decomposing the semiconductor. For example, Ti3+ can be transformed into Ti4+ in non-stoichiometric TiO2. The element should have more than one stable valence in the semiconductor, so that it is cannot be decomposed due to the formation of holes, that is, photocorrosion. Zn2+ in ZnO and Cd2+ in CdS will be photocorroded due to the formation of holes. 118 The photoinduced holes should be highly oxidizing to generate hydroxyl radicals. The photogenerated electrons should have sufficient reducibility to produce superoxide from oxygen. In addition, semiconductors must have an appropriate band gap, and be highly stable to chemical corrosion, non-toxic and generally low in cost. 119
Nano-TiO2 photocatalyst
Nano-TiO2 is the earliest and most widely used semiconductor photocatalyst. In Japan, TiO2 is used in exterior wall coatings to play the role of self-cleaning of walls. It is also used in pavement asphalt and has a certain degradation effect on automobile exhaust. This is mainly related to its high photocatalytic activity, low cost, nontoxicity, good thermal stability, and chemical stability.
Structure and properties of nano-TiO2
Nano-TiO2 is an n-type semiconductor and its crystal structure is mainly classified as anatase, rutile, and brookite. The unit structures of the three crystal forms are shown in Figure 4. Among them, the rutile nano-TiO₂ is composed of irregular and slightly orthorhombic octahedra. The anatase nano-TiO₂ exhibits obvious orthorhombic crystal distortion and lower symmetry than the rutile nano-TiO₂. The brookite nano-TiO₂ is orthorhombic. 120 The nano-TiO2 with different crystal structures shows different physical and chemical properties.
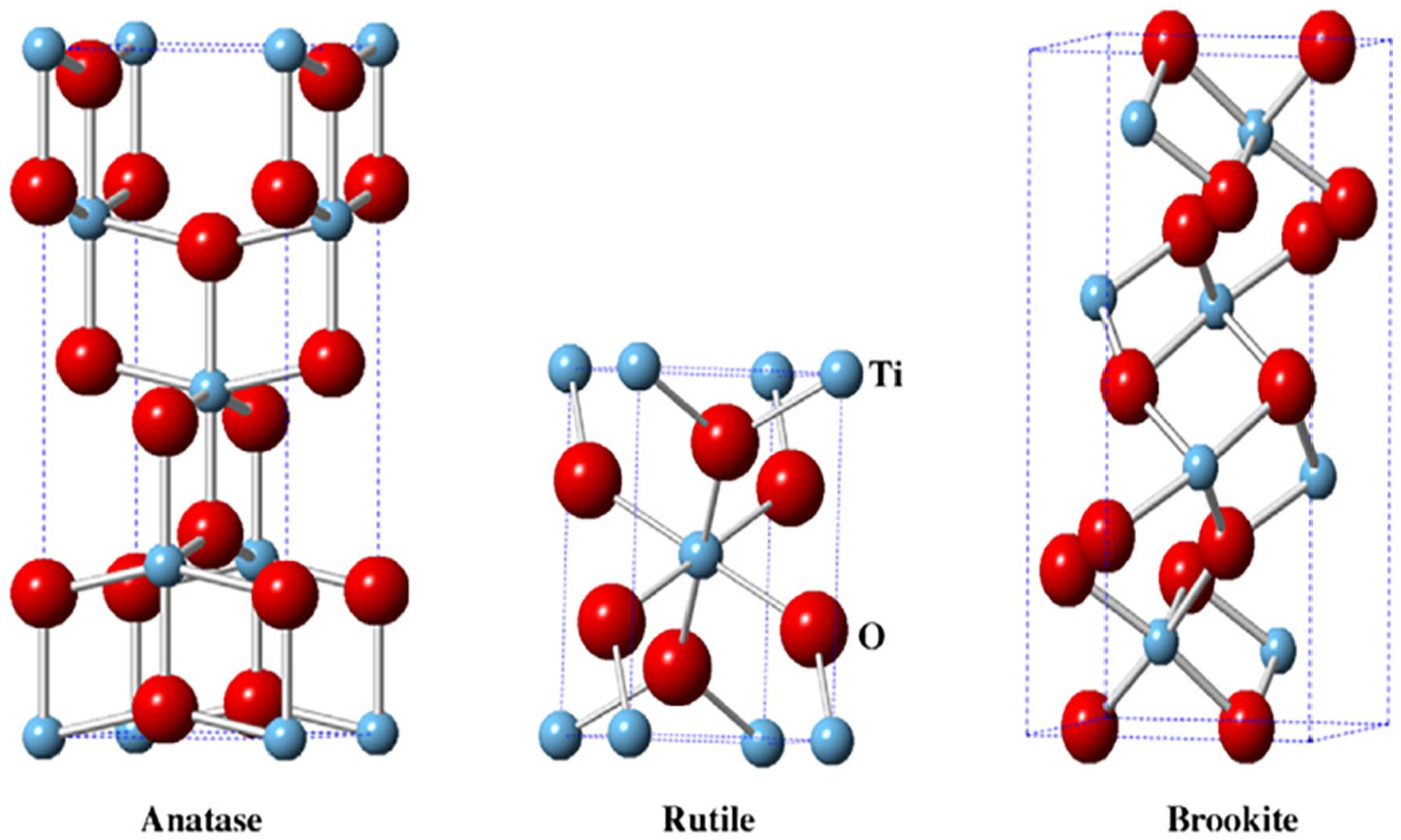
Crystal structure of anatase, rutile, and brookite. 121
Rutile type is the most common form of nano-TiO2 and is very stable at high temperature. Anatase nano-TiO2 is relatively stable at low temperature. Bentonite nano-TiO2 is unstable and has no photocatalytic activity. 121 In General, the photocatalytic activity of rutile nano-TiO2 is significantly higher than that of rutile nano-TiO2. This is because the band gap of rutile nano-TiO2 is 3.0 eV, which is smaller than that of anatase nano-TiO2 (3.2 eV). So rutile nano-TiO2 is not easy to cause oxidation-reduction reactions between oxygen and water to generate superoxide anion radicals (·O2−) and hydroxide radicals (·OH). 122 Moreover, there are many defects in the crystal lattice of anatase nano-TiO2, which can generate more h+ to capture e−. While the structure of rutile nano-TiO2 is too stable and there are few defects. This leads to the rapid recombination of photogenerated e− and h+ and reduces its photocatalytic activity. In addition, the formation of rutile nano-TiO2 requires high-temperature calcination treatment above 700℃. During the calcination process, the agglomeration of nano-TiO2 may increase its grain size and sharply decrease its specific surface area, resulting in a decrease in its photocatalytic activity. 123
Photocatalytic mechanism of nano-TiO2
When nano-TiO2 is excited by photons with
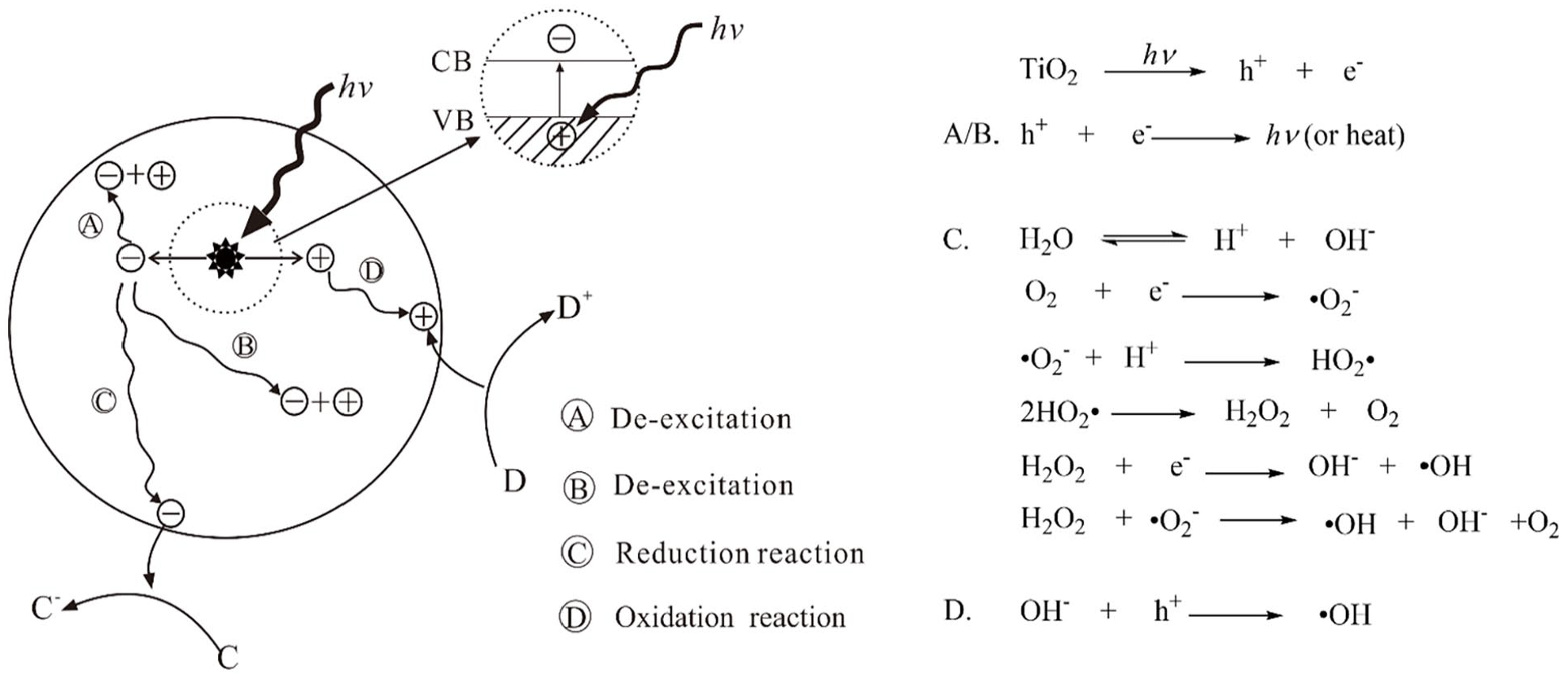
Photocatalytic reaction mechanism of the nano-TiO2.
Preparation of anatase nano-TiO2
In recent years, the main methods to prepare anatase nano-TiO2 include vapor-phase hydrolysis, vapor-phase oxidation, sol-gel method, precipitation method and hydrothermal method.124–128 Among them, the hydrothermal method for preparing nano-TiO2 has gradually become one of the commonly used methods for nano-TiO2 preparation due to its advantages of complete crystal grain development, small original particle size, and less particle agglomeration. The basic operation of hydrothermal method is as follows. The TiO2 precursors, such as titanium chloride, titanium sulfate, and butyl titanate, are added into a high-pressure reactor lined with corrosion-resistant and high-temperature resistant materials. Then, the reactor is heated to the desired temperature at a certain heating rate and kept at a constant temperature for a period of time. After the reaction is completed, TiO2 nanoparticles are obtained by depressurization, washing, and drying. The reaction temperature required for hydrothermal method is relatively low, generally within the range of 100℃–240℃. Therefore, this method effectively avoids the formation of rutile phase during high-temperature calcination, promotes the formation of anatase phase, and inhibits crystal grain agglomeration. Wang 129 prepared mesoporous TiO2 nanoparticles using the hydrothermal method in an ethanol/water mixed solution using butyl titanate as a precursor. She investigated the effect of hydrothermal temperature on the photocatalytic activity of TiO2, and found that the particle size of the prepared TiO2 was about 9 nm. The photocatalytic activity of TiO2 powder prepared at 180℃ was the highest, far exceeding that of commercial titanium dioxide (P25).
Photosensitization modification of nano-TiO2
However, an inherent defect of nano-TiO2 is its relatively broad
Photosensitive modification methods of nano-TiO2.
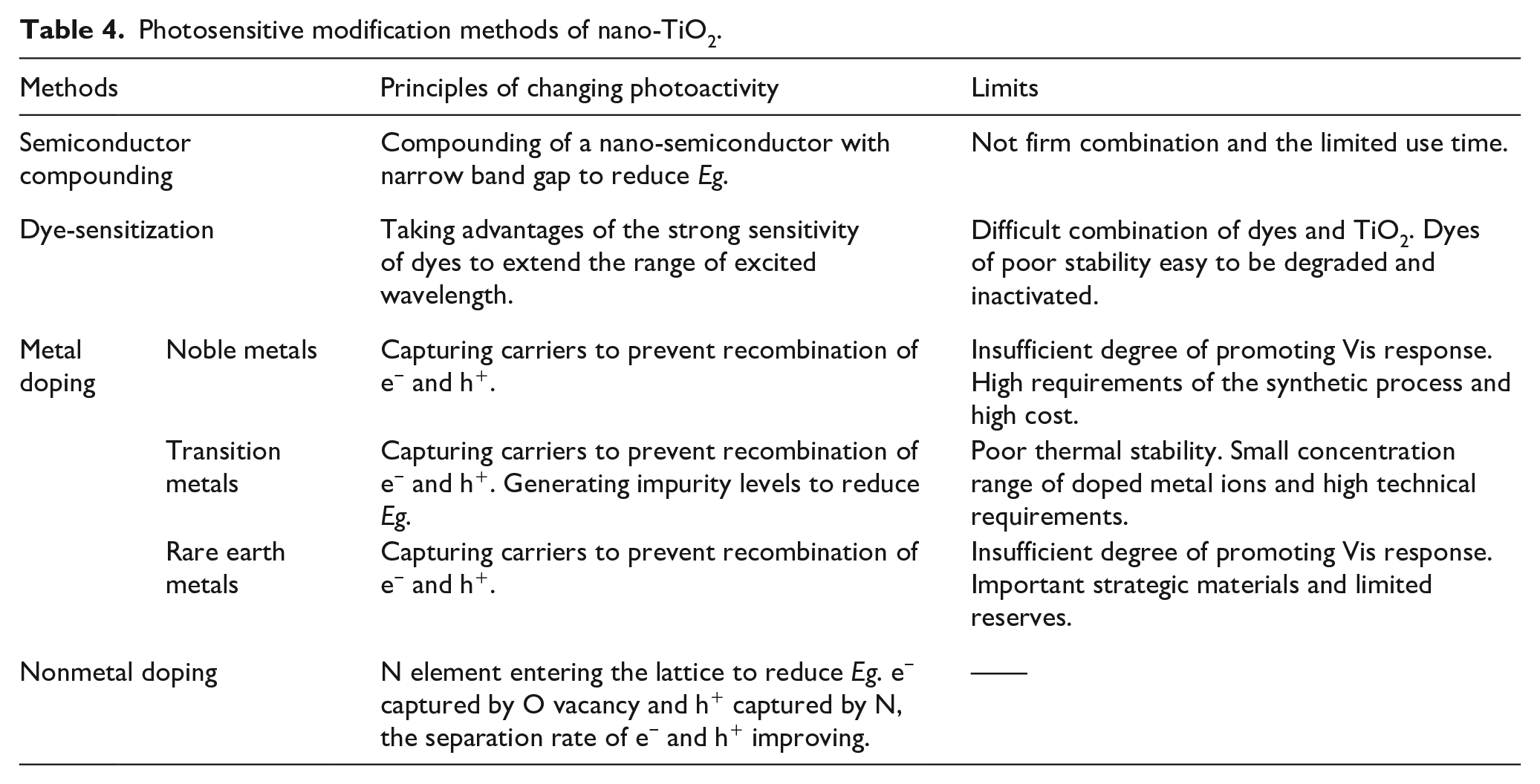
Semiconductor compounding
Semiconductor compounding refers to the compounding of a nano-semiconductor material with narrow
Dye-sensitization
The dye-sensitization of nano-TiO2 was originally aimed at the photosensitization modification of solar cells.135,136 At present, this method has also been applied to photocatalytic degradation experiments. The dyes used for photosensitization modification should have strong photoactivity and are loaded on the surface of nano-TiO2 by physical or chemical adsorption. This method can improve the efficiency of photocatalytic reaction by broadening the range of excitation wavelength. Common dye sensitizers include phthalocyanine, chlorophyllin, and eosin. Through dye-sensitization, nano-TiO2 can effectively realize Vis-responsive photocatalysis and improve the efficiency of photochemical energy conversion. However, it is also faced with the problem of difficulties in combining dyes and semiconductors. Moreover, the dyes themselves are organic substances that may also be degraded and inactivated in the photocatalytic process, so their stability is poor. In conclusion, it is difficult to introduce this technology into practical application.
The metals used for modification mainly fall into three categories: noble metals such as Au, Ag, Pt, and Pd; transition metals like Fe, Mn, and Co; and rare earth metals including Ce, Sm, Pr, and Er. Doping noble metals on the surface of nano-TiO2 can make the material quickly capture the e− on CB to prevent the recombination of e− and h+, thereby improving the photoactivity. However, the degree of promoting Vis response by this method is not high, and the synthetic process needs high requirements and cost. Different from noble metals, transition metals are mainly doped into nano-TiO2 in the ionic state. There are many studies on Fe3+ doping, and the photocatalytic effect of the synthesized nano-Fe-TiO2 is better.137,138 Doping nano-TiO2 with transition metal ions is equivalent to introducing an impurity level into the band gap of TiO2. On the one hand, the transition metal ion can capture the photogenerated carriers generated by excitation of TiO2. On the other hand, it can produce an impurity level to the CB of TiO2, so that the
Nonmetal doping
In 2001, Asahi et al.
141
conducted the photocatalytic degradation experiment of MB dyes for the first time using N-doped TiO2 as photocatalyst, and published the research results in the journal Science. This study found that the introduction of N narrowed the
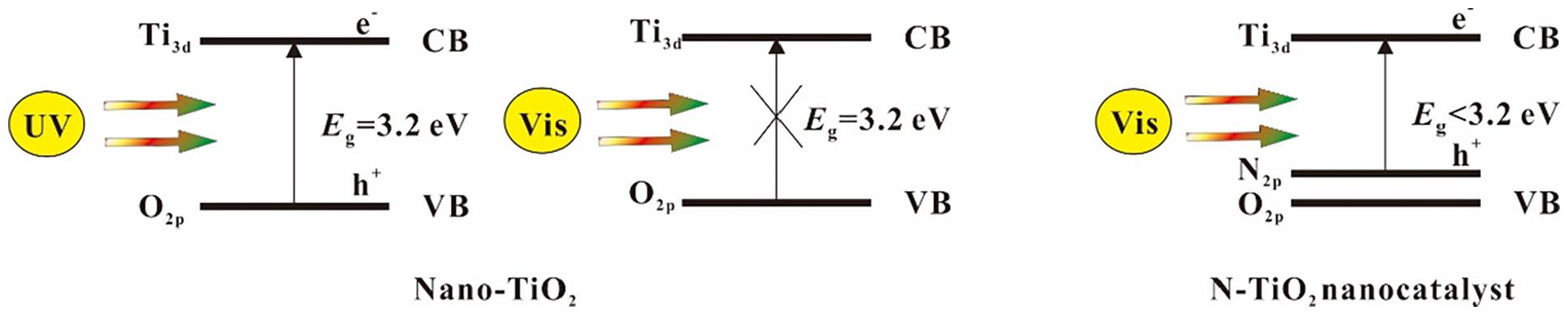
Excitation of nano-TiO2 and N-TiO2 nanocatalyst.
Research progress of nano-TiO2@adsorbent composites for degradation of dyes
In view of the social problem of dye wastewater pollution in textile printing and dyeing industry, nano-TiO2@adsorbent composites with the adsorbent as the carrier and nano-TiO2 as the photocatalytic material have been developed. This class of materials has the integrated function of adsorption and photocatalytic degradation of dye wastewater under the condition of light radiation. While adsorbing dyes, the composites can complete the degradation of dyes and achieve the goal of killing two birds with one stone.
Feasibility of combining adsorption with photocatalysis
From the previous analysis, it can be found that the adsorption method is mature and easy to operate, and can effectively remove dyes in wastewater. However, the adsorption process is only an enrichment process for pollutants in wastewater and there is no degradation process because the adsorbent itself does not have the ability to degrade dyes. Adsorbents and dyes are only combined in physical or chemical ways, and the quantity and properties of organic pollutants have not changed in this process. The dyes are only captured and enriched on the surface of adsorbents. The adsorbent is limited by its saturated adsorption capacity. After reaching its saturated adsorption capacity during the adsorption process, the adsorbent will face problems such as regeneration, replacement, or recovery. In this process, if any link is not handled properly, it is easy to cause secondary pollution.
Nano-TiO2 photocatalytic technology can destroy and degrade organic pollutants with complex and stable structures that are difficult to biodegrade. It shows obvious advantages of high efficiency, energy saving, non-selectivity, and thorough degradation of pollutants during use. So it has been concerned by scholars in the industry of textile dyeing and finishing since it came out. However, due to the small particle size, nano-TiO2 is not conducive to separation from the solution and easy to agglomerate. This limits its practical application. In addition, both nano-TiO2 and TiO2 semiconductor films have limited ability to adsorb organic pollutants and weak decolorization ability. For textile printing and dyeing wastewater with high concentration and poor light transmittance, photocatalytic reaction is difficult to occur. Therefore, this technology is difficult to apply alone in the treatment of textile printing and dyeing wastewater, and needs to be used in combination with other methods or for advanced treatment of dye wastewater.
Since the traditional adsorption method is the most mature method for treating textile dye wastewater, and the newly-emerging nano-TiO2 photocatalytic degradation technology has obvious advantages, the adsorbent is used as the carrier of nano-TiO2 photocatalyst. This adsorbent carrier can solve the problems of difficult separation and easy agglomeration of the nano-TiO2 photocatalyst. Besides, it can make the dye molecules be potentially adsorbed near the position where the photocatalytic reaction is carried out, which is conducive to the final in-situ photocatalytic degradation of the adsorbed dyes by nano-TiO2. The adsorbent carrier can also adsorb the reaction intermediates produced in the process of photocatalytic degradation, thus making the potentially toxic intermediates more likely to be completely mineralized. Moreover, the application of some types of adsorbents can improve the separation rate of h+–e− and improve the overall photoactivity. In this reaction system, the adsorbent is responsible for the enrichment of dyes in wastewater by virtue of its strong adsorption, and nano-TiO2 is responsible for the photocatalytic degradation of dyes enriched on the surface of the adsorbent and remaining in the solution. The adsorbent completes the precursor steps of dye wastewater treatment, namely adsorption and decolorization. Nano-TiO2 realizes the ultimate goal of dye wastewater treatment, that is, degradation of dyes. The organic combination of adsorption, catalysis and degradation processes enables the composites to efficiently treat dye wastewater with high concentration and large chroma.
Types of adsorbent carriers and applications
Nano-TiO2@inorganic adsorbent composites
The inorganic adsorbents used to prepare nano-TiO2@adsorbent composites mainly include zeolite and fly ash. Chong et al. 146 adopted an improved sol-gel method to mix titanium butanol with ethanol, and obtained TiO2–zeolite nanocomposites after a series of reactions. Under UV irradiation, the composites exhibited a higher apparent pseudo-first-order rate constant for reactive black 5 dyes with lower concentration compared with commercial nano-TiO2. This implied that the TiO2–zeolite nanocomposites followed the photocatalytic degradation behavior promoted by adsorption, which was helpful for the removal of water pollutants with trace amounts in the wastewater treatment stage. Moreover, the studies of Liao et al., 147 Vicentini et al., 148 and other teams confirmed that the nano-TiO2–zeolite composites had a synergistic effect of adsorption and photocatalysis when treating dye wastewater. Duta and visa prepared fly ash–TiO2 composites by hydrothermal synthesis using fly ash with large specific surface area and effective adsorption of dyes and P25-TiO2 as raw materials. 149 Under UV radiation, the composites exhibited a synergistic effect of adsorption and photocatalysis on the industrial benzoic acid blue dye, indicating that the adsorption effect can promote the photocatalytic reaction.
It can be seen from the above studies that the composites prepared by nano-TiO2 and inorganic adsorbents mainly rely on the porous structure and large specific surface area of the inorganic adsorbents to complete the adsorption of dyes. The final treatment effect of the composites on dyes is affected by the adsorption capacity and the adsorption rate of inorganic adsorbents. In addition, the photocatalytic reaction needs to be carried out under UV radiation, so there are problems of low efficiency and high energy consumption in practical applications.
Nano-TiO2@carbonaceous adsorbent composites
As a representative of carbonaceous adsorbents, AC is widely used as a carrier for gas and water remediation because of its good adsorption performance. Studies have confirmed the existence of synergistic effect of adsorption and photocatalysis by using the mechanical mixture or complex of nano-TiO2 and AC. Slimen et al.
150
added AC to the precursor of TiO2, dried and calcined to prepare anatase nano-TiO2/AC composites. The addition of AC increased the specific surface area of the photocatalyst, prevented the agglomeration of TiO2 particles, and led to the blue-shift of the absorption spectrum due to the quantization of the energy band structure. The degradation rate of MB by the composites under Vis radiation is twice that of P25-TiO2. Therefore, the nano-TiO2/AC composite had good Vis photocatalytic performance and was a promising photocatalyst for the degradation of organic pollutants. Ragupathy et al.
151
prepared nano-TiO2 loaded cashew nut shell AC by the sol-gel method. Due to the optimization of parameters such as specific surface area, porosity, hydroxyl number, and
Carbon nanomaterials have attracted extensive attention in the field of water treatment due to their hollow layered structure, the interaction of π-π bond and superior specific surface area. Carbon nanofiber (CNF) is a common carbon nanomaterial and CNF webs have been studied for photocatalytic degradation of dyes. Xu et al.’s 152 team successfully prepared nano-TiO2@CNF composites by hydrothermal method and blended spinning method, respectively. The samples obtained by the hydrothermal method at 900℃ and the blended spinning method at 1100℃ had the highest photoactivity under UV radiation, and the samples obtained by the hydrothermal method had higher degradation efficiency of Rhodamine B. But the blended spinning method can inhibit the transformation of TiO2 from anatase to rutile in the carbonization process and the obtained samples had higher reuse rate. Kim et al. 153 dissolved polyacrylonitrile and the TiO2 precursor in N, N-dimethylformamide and prepared nanofiber webs by electrospinning. The webs were immersed in silver nitrate solution, and finally Ag-TiO2/CNF composites were obtained by calcining the webs at high temperature. Under Vis radiation, the degradation rate of MB treated with Ag-loaded composites for 3 h was 17 times faster than that without Ag. As the receptors of e−, Ag nanoparticles captured photogenerated e− and reduced the recombination of h+–e− pairs, so as to improve the photocatalytic degradation rate. While the surface-exposed CNFs acted as the center for matrix physisorption due to their high specific surface area. Therefore, the Ag-TiO2/CNF composites had dual effects of photocatalysis and adsorption on MB and the composites are expected to be candidate photocatalytic materials that can be excited by sunlight.
Graphene is a new kind of carbon nanomaterial. The specific surface area of the monolayer graphene with a hexagonal honeycomb network structure is theoretically 2600 m2·g−1. 154 The sp2 bonded carbon lattices have many e− and delocalized π bonds, which not only improves the structural stability but also enhances the conductivity. Therefore, graphene has attracted more and more attention in the field of photocatalysis. A large number of studies have been carried out on the modification of TiO2 by graphene oxide (GO) or reduced graphene oxide (rGO) for application in the photocatalytic degradation of organic pollutants. Appavu and Thiripuranthagan 155 synthesized N, S co-doped TiO2/rGO nanocomposites by one-step hydrothermal method. The photocatalytic degradation effects of the nanocomposites on congo red, MB and reactive orange 16 were better than those of commercially available nano-TiO2, TiO2/rGO nanocomposites and N, S co-doped nano-TiO2. The nanocomposites containing 5% rGO had high specific surface area, improved the absorption of Vis, reduced the charge transfer resistance, and generated more potential oxide species, thus showing the highest photoactivity. Therefore, this nanocomposites with Vis photoactivity had a good prospect in the field of photocatalysis. Zhang et al. 156 prepared N doped nano-TiO2 coatings on the surface of rGO by sonochemical method and hydrothermal method, which significantly improved the photoactivity of Rhodamine B and a series of organic compounds by about 17.8 times. This was mainly due to the fact that N doping and rGO effectively improved the charge separation rate and provided rich catalytic active sites to improve the reaction activity. The adsorption capacity of the composites for pollutants, the life of h+–e− pairs, the absorption capacity of light, and the absorption rate of Vis is improved. The composites were suitable for the purification of industrial wastewater containing refractory organic pollutants.
Based on the above studies, it can be found that nano-TiO2@carbonaceous adsorbent composites adsorb the dyes through the large specific surface area of the carbonaceous adsorbent, and the recombine of h+–e− pairs is reduced due to the excellent electrical conductivity of the carbonaceous adsorbent itself, thereby improving the photocatalytic degradation efficiency. However, due to the lack of functional ionic groups, carbonaceous adsorbents have no selectivity for dyes, and they are too expensive to popularize.
Nano-TiO2@organic adsorbent composites
In the past 10 years, the research on nano-TiO2@adsorbent composites has begun to involve organic adsorbents. The organic adsorbents used to prepare composites mainly include chitosan, cellulose and starch. Chitosan is a natural polymer polysaccharide with good biocompatibility. Its combination with nanosemiconductors has been developed and applied in the field of wastewater treatment and biology. Karthikeyan et al. 157 directly mixed the chitosan solution with TiO2, then stirred and dried to synthesize the chitosan-TiO2 nanocomposites. The decolorization rates of Rhodamine B and Congo red by the nanocomposite under Vis radiation were significantly higher than those in the dark, and the decolorization rates were higher than 50%. Moreover, the nanocomposite also had a good antibacterial effect on Gram bacteria. In addition to the existence of particles, the combination of chitosan and nano-TiO2 was also studied in the form of composite films. Norranattrakul et al. prepared chitosan/nano-TiO2 composite films by solution casting method with the chitosan film as the polymer matrix of nano-TiO2. 158 The sample of 1 wt% chitosan without crosslinking had high photoactivity. The removal rates of three reactive dyes under the UV radiation were significantly higher than those under the dark condition, and the removal process of dyes conformed to L-H model. The photocatalytic process occurred both on the surface of nano-TiO2 and in solution. Farhadian et al. synthesized NST/chitosan composites by the sol-gel method using nano-TiO2 co-doped with N and S elements (NST). 159 Under Vis irradiation for 20 min, the degradation rate of tetracycline by NST/chitosan composites was as high as 91% and the degradation rate was increased by about 2 times. Therefore, the composite can be used as an effective catalyst for the degradation of organic matter in wastewater.
Cellulose is another organic adsorbent used to develop nano-TiO2@adsorbent composites.
160
Its ordered crystalline structure can be completely dissolved in inorganic acid. Hamad et al.
161
dissolved microcrystalline cellulose in HNO3 and H3PO4, respectively. The solutions were added to TiO2 precursor for impregnation next and the cellulose-TiO2 composites, CNT and CPT, were prepared by the single-step sol-gel method. Under UV radiation, the photocatalytic degradation rates of orange G by CNT and CPT were higher than that of P25-TiO2. This was mainly due to the fact that CNT was the microporous material with high specific surface area and CPT was the mesoporous material. In addition, the interaction between the cellulose chains and the surface groups on TiO2 precursors made
In view of the ideal results on the researches of nano-TiO2@chitosan composites and nano-TiO2@cellulose composites in adsorption-photocatalytic degradation of organic matter, our research group has synthesized nano-TiO2@starch based adsorbent composites through improved sol-gel method by using modified starch as adsorbent and carrier of nano-TiO2 for ionic dyes. 165 Under the simulated sunlight irradiation, the adsorption-photocatalytic degradation of N-TiO2@starch based adsorbent composites had obvious synergistic effect on dye treatment. During the process of adsorption-photocatalytic degradation, the TOC of dye solution decreased significantly. Moreover, the N-TiO2@starch based adsorbent composites were reusable. Figure 7 shows the mechanism for adsorption-photocatalytic degradation of ionic dyes by N-TiO2@starch based adsorbent composites.165,166
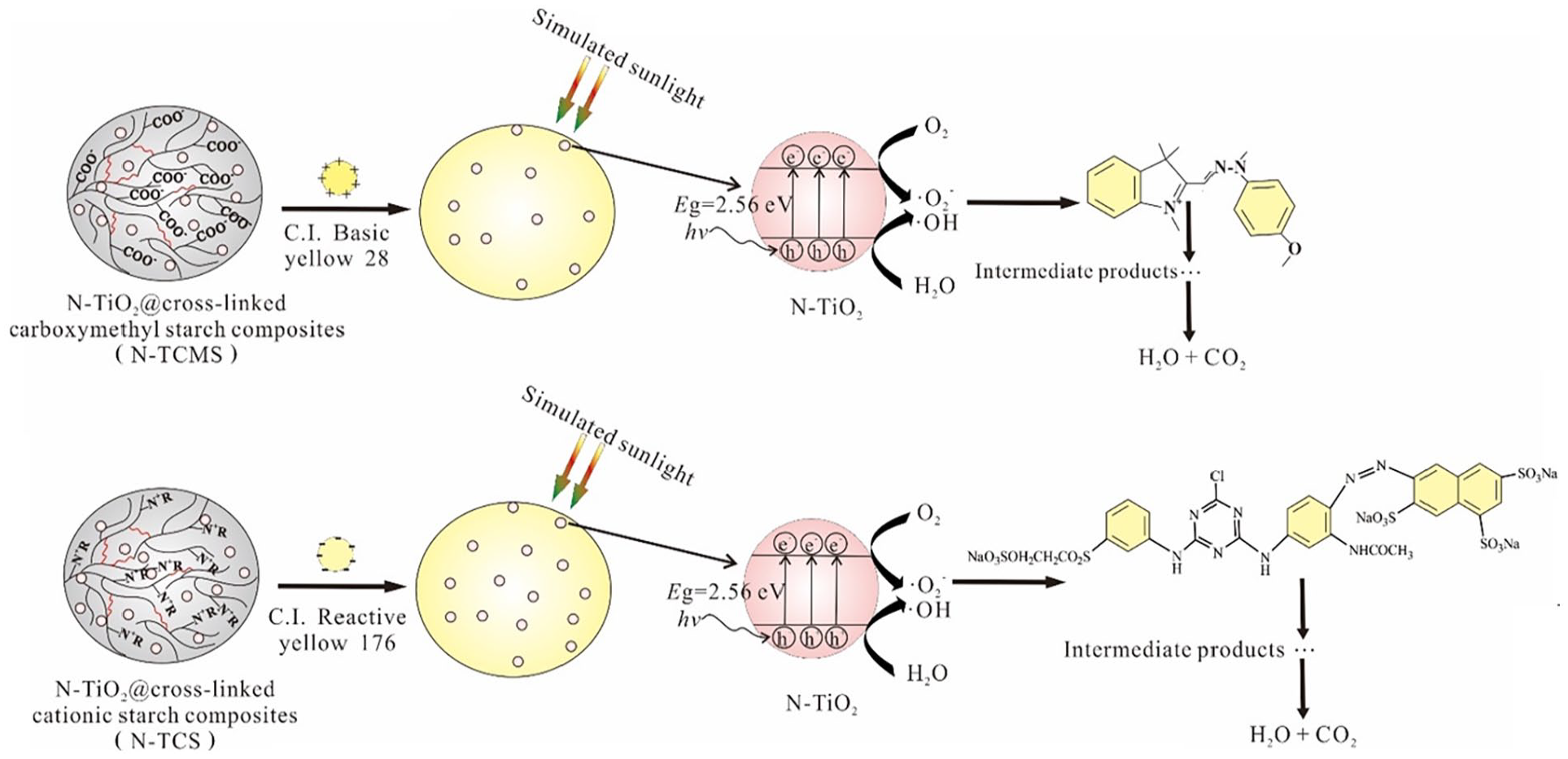
Figure of mechanism for adsorption-photocatalytic degradation of ionic dyes by N-TiO2@starch composites.
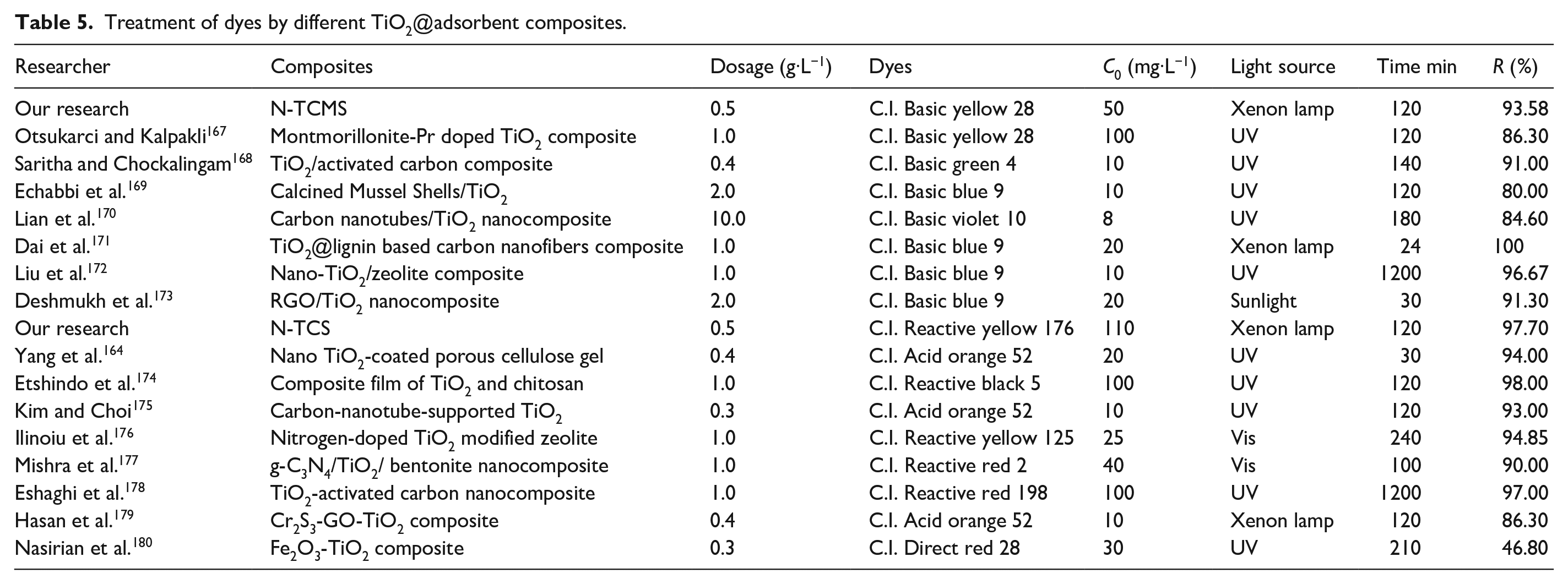
Table 5 shows the removal rates (
Treatment of dyes by different TiO2@adsorbent composites.
Factors affecting the adsorption-photocatalytic degradation reaction of dyes
Several factors influence the adsorption-photocatalytic degradation reaction of dyes by nano-TiO2@adsorbent composites, namely the initial dye concentration, contact time, temperature, pH, adsorbent particle size, total pore volume, surface area, adsorbent/dye ratio, and mode of treatment (Figure 7). These factors require consideration because optimization of these parameters can significantly increase the uptake ability of an adsorbent.
Initial dye concentration
The initial concentration of dyes has an important effect on the adsorption and photocatalytic degradation processes. The increase in the dye concentration intensifies the competition between dye molecules in the solution to occupy the limited adsorption active sites of the adsorbent. When the dose of the adsorbent is constant, the active sites will be exhausted and the adsorbent will reach saturation due to the fixed number of adsorption active sites. Therefore, the removal rates of dyes decrease with the increase of the initial concentration.181,182 For the photocatalytic degradation reaction, the increase of the dye concentration will lead to the weakening of the light transmittance of the solution, the reduction of photons reaching the surface of the photocatalyst, and the decrease of the photocatalytic degradation rate of the dye. 183 This is also one of the reasons for combining adsorption and photocatalysis for the treatment of high-concentration dye wastewater. The removal of dyes by nano-TiO2@adsorbent composites is the synergistic effect of the adsorbent and TiO2, so with the increase of the initial concentration of dyes, the removal rate of dyes by the composites decreases. Kanakaraju et al. 184 used 1.3 g·L−1 of Cu-TiO2-fly ash composites to adsorb-photocatalytically degrade methylene orange (MO) with concentrations of 5–25 ppm under Vis radiation, and found that the highest removal rate (99.91%) was obtained by using 5 ppm of MO. The increase of the initial dye concentration not only affects the final dye removal efficiency, but also reduces the dye removal speed. Chong et al. 146 performed adsorption-photocatalytic degradation of reactive black 5 dye in the concentration range of 1–20 ppm by TiO2-zeolite nanocomposites, and demonstrated that at a low concentration of 1 ppm, the nanocomposites exhibited the highest apparent pseudo-first-order rate constant.
Solution pH
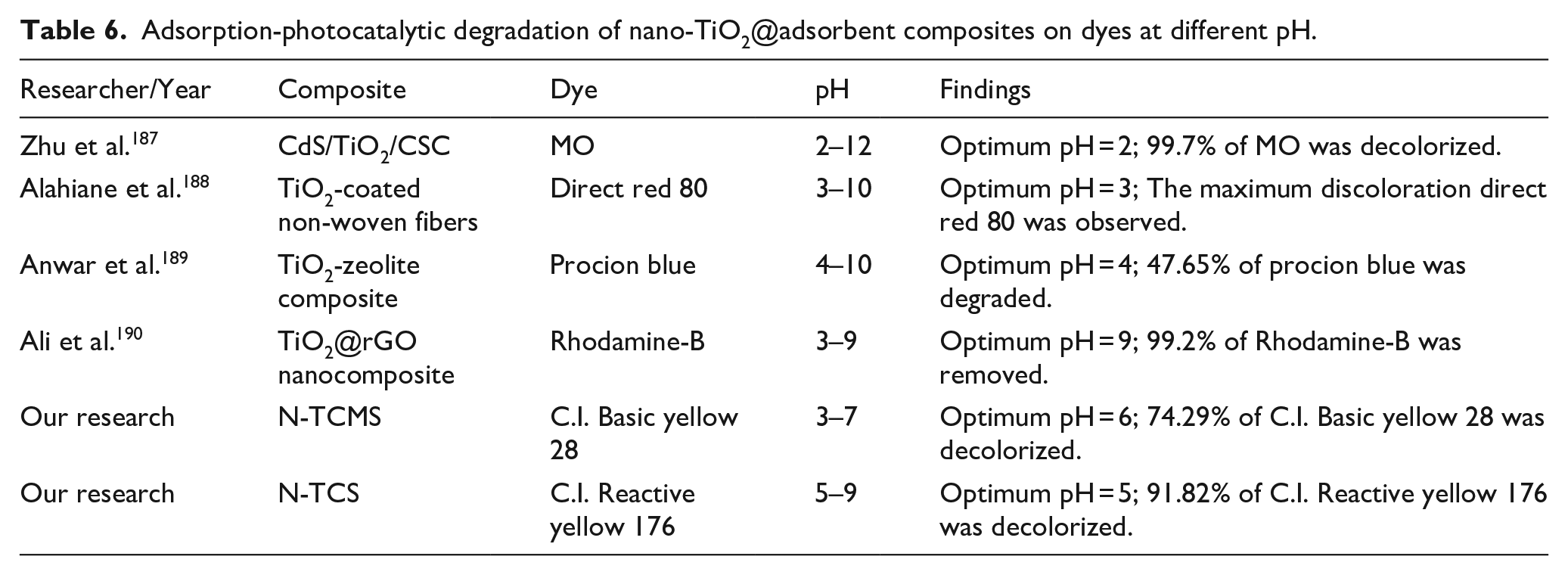
The pH of the solution is an important operating variable during dye wastewater treatment. The change of pH value of the solution will cause the change of the surface charge of the adsorbent and photocatalyst. Therefore, the pH of the solution affects the dye adsorption-photocatalytic degradation efficiency by nano-TiO2@adsorbent composites. The point zero charge (pHpzc) refers to the pH value of the solution when the surface charge of the composite is zero. The surface charge of composites is usually analyzed by pHpzc. When the solution pH is above pHpzc, the surface of the composite is negatively charged. On the contrary, the surface of the composite is positively charged.185,186 The pHpzc of the CdS nanocrystals/TiO2/crosslinked chitosan composite (CdS/TiO2/CSC) prepared by Zhu et al. 187 is 6.5. When the pH of the solution is less than 6.5, its surface is positively charged. And it was confirmed by experiments that in acidic solution, the decolorization effect of MO was the best due to the electrostatic attraction between the positively charged composite and MO anions. The pHpzc of N-TCMS and N-TCS measured by the pH drift method was 4.5 and 8.2, respectively. Therefore, the adsorption-photocatalytic degradation experiment of C.I. Basic yellow 28 by N-TCMS was carried out under the condition of solution pH > 4.5, and the adsorption-photocatalytic degradation experiment of C.I. Reactive yellow 176 by N-TCS was carried out under the condition of solution pH < 8.2. The experimental results are exhibited in Figure 8.165,166 Table 6 shows the optimal pH of nano-TiO2@adsorbent composites for dye adsorption-photocatalytic degradation.
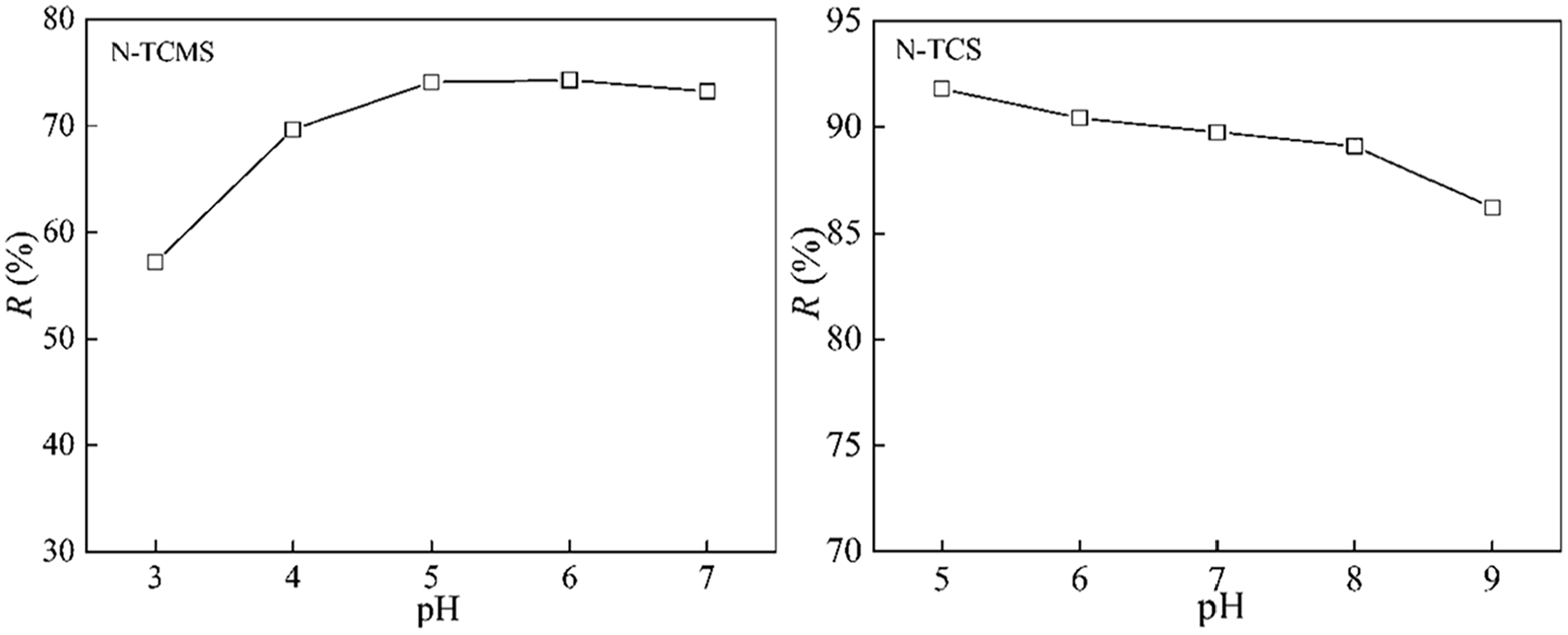
The effect of pH on the removal of dyes by N-TiO2@starch composites.
Adsorption-photocatalytic degradation of nano-TiO2@adsorbent composites on dyes at different pH.
Composite dosage
The dose of the composite is an important contributing factor. On the one hand, the dose plays a decisive role in the adsorption capacity of the adsorbent, and on the other hand, the dose affects the photocatalytic efficiency of the photocatalyst. Visa et al. 191 utilized TiO2-fly ash composites with different doses from 0.1 to 0.8 g·50 mL−1 to determine the influence of the dose on MB adsorption-photodegradation. They found that the removal rate of MB increased with the amount of the composite increasing and the optimal dose was 0.4–0.5 g·50 mL−1. Nouri et al. 192 used various doses of TiO2/calcium alginate composites to remove basic blue 41 and reported that increasing the composite dose from 0.05 to 0.1 g·100 mL−1 increased the basic blue 41 removal rate from 69% up to 96%. The study of Ragupathy et al. 151 obtained the same conclusion. The adsorption-photocatalytic degradation rates of brilliant green and MB by the TiO2/cashew nut shell AC composites reached the maximum when the dosage of the composites increased from 0.1 to 0.2 g·L−1. This promotion in adsorption-photocatalytic degradation is mainly attributed to two aspects. Firstly, the adsorption sites of the adsorbent increased as the dosage of the composite rose, and the adsorption capacity of the composite on dyes improved. Secondly but not least, the number of active sites of TiO2 increased, so the photogenerated ·OH increased and the oxidation capacity of the composite improved. Under the synergistic effect of the above two factors, the removal effect of dyes was enhanced.
Reaction time
In the practical application of dye wastewater treatment, time consumption is an important index to evaluate the efficiency of treatment technology. The treatment of dyes by nano-TiO2@adsorbent composites is the result of the synergistic effect of adsorption and photocatalytic degradation. Therefore, theoretically this technology should consume less time than pure adsorption or photocatalytic technology. Li et al. 193 studied the synergistic adsorption-photocatalytic performance of TiO2-MnTiO3/hollow activated carbon fibers heterojunction photocatalysts (TiO2-MnTiO3/HACFs) on MB under Vis, and found that the adsorption effect of HACFs played a major role in the first cycle of MB treatment. This is mainly due to the fact that the photocatalytic rate is affected by the activation and migration rate of photogenerated e− and h+, so the reaction rate of photocatalysis is much slower than that of adsorption. The final decolorization time of MB by TiO2-MnTiO3/HACFs was shorter than that of HACFs and P25. Liu et al. 194 found that under the condition of illumination, within the first 20 min of MB treatment by the TiO2-graphene composite (TiO2-GR), the concentration of MB decreased sharply due to the synergy of absorption and catalysis. At the same time, the authors emphasized that the remarkable adsorption-photocatalytic activity of TiO2-GR should first be mainly attributed to the adsorption performance of TiO2-GR. The TiO2/crosslinked carboxymethyl starch composite prepared by our research group had a very fast adsorption rate for C.I. Basic yellow 28, which basically reached the adsorption equilibrium under the dark condition for 20 min. It had obvious adsorption-photocatalytic degradation synergy under the UV radiation condition and the dye removal rate was significantly higher than that of adsorption. 165
In conclusion, in the treatment of dye wastewater with nano-TiO2@adsorbent composites under the illumination condition, due to the synergistic effect of adsorption-photocatalytic degradation in the initial stage and the fast adsorption rate, the chromaticity of the dye decreases rapidly. As the reaction proceeds, the adsorption active sites decrease, the adsorption weakens and the dye removal rate decreases. Overall, the dye removal rate increases gradually with time, and the dye removal rate by nano-TiO2@adsorbent composites is higher than that of pure adsorption and photocatalysis technology in the same reaction time.
Irradiation source
The radiation source plays a decisive role in the excitation of electron-hole pairs, so it has a significant influence on the photocatalytic effect of photocatalyst. In the relatively simple studies, UV and Vis were directly used as radiation sources to study the changes of dye solution concentration after the same time of radiation. Liu et al. 194 determined that the residual concentrations of MB solution were 6% and 13% respectively after 20 min of adsorption-photocatalysis by TiO2-GR under UV and Vis radiation conditions. As we all know, the intensity of UV is stronger than Vis, so the removal rate of dye is higher under the condition of UV radiation. In addition, more research has been done to study the influence of light intensity by using UV light sources with different power or light irradiance. You-ji and Wei 195 studied the removal rates of RhB by TiO2-zeolite nanocomposites under the irradiation of UV light source with different light irradiance. Within a certain range, the reaction rate increases significantly with the increase of light irradiance intensity. Liu et al. 196 tested the degradation of methyl orange (MO) by TiO2/cellulose under UV radiation in a range of 4.3–41 mW/cm2 and found that the degradation rate of MO was positively correlated with the intensity of UV. Li et al. 197 experimentally confirmed that the power of the UV lamp was almost linear with the apparent constant of MO degradation.
Based on the above research, it can be concluded that the light radiation intensity of the radiation source can determine the number of electron-hole pairs generated by affecting the energy provided to the photocatalyst in the composite material. Generally speaking, the higher the intensity of light radiation, the more electron-hole pairs produced by photocatalyst, resulting in a faster degradation rate and higher removal efficiency of dyes.
TiO2 loading capacity or adsorbent carrier amount
Catalyst loading capacity is an important parameter in the photocatalytic water treatment process. The optimum catalyst loading capacity enables the maximum photoactivity while preventing unnecessary catalysts consumption. Jin et al. 198 evaluated the influence of TiO2 loading and GO carrier amount on the adsorption-photocatalytic performance by studying the adsorption-photocatalytic degradation of low concentration acetamide by TiO2/GO with different mass ratios of TiO2 to GO under UV irradiation. The experiment showed that the apparent photocatalytic rate increased first and then decreased with the increase of mass ratios of TiO2 to GO. 2:1-TiO2/GO had the highest removal rate for acetamide with an initial concentration of 500 ppb, but its removal rate was much lower than that of pristine TiO2 NWs. Min Xiaobo et al. 199 proved that the MB removal efficiency of the various photocatalysts followed the order of 0.02TiO2@HKUST-1 > 0.03TiO2@HKUST-1 > HKUST-1 > 0.04TiO2@ HKUST-1 > TiO2 > 0.05TiO2 @HKUST-1. Liao et al. 147 prepared a modified zeolite supported nanocrystal TiO2 composite (MZTC) and evaluated its photocatalytic efficiency. 147 The degradation rate of MB increased with the amount of loaded TiO2, and MZTC-2.5 showed the highest photocatalytic activity. However, when the amount of loaded TiO2 was up to 5%, the degradation rate was lower.
In summary, firstly, the photocatalytic activity of the composite photocatalyst is related to its structure and carrier properties. The formation of a heterostructure between the photocatalyst and the carrier can significantly enhance the charge separation, thereby reducing h+–e− recombination. 200 The photocatalytic activity will be enhanced by the synergistic effect of the photocatalyst and the carrier.
Conversely, if the heterostructure formation is absent and the electron transfer ability of the carrier is poor, the photocatalytic activity of the composite photocatalyst will decrease. Secondly, the photocatalytic activity of the composite photocatalyst generally increases first and then decreases with the increase of photocatalyst loading. The main reason for the initial increase is that as the number of photocatalysts increases, the number of active sites also increases. The main reason for the subsequent decrease is the aggregation phenomenon of excessive photocatalyst, which leads to a reduction in exposed active sites.
Inorganic ions
High concentrations of inorganic ions, including inorganic anions and inorganic cations, are commonly present in wastewater. Alahiane et al. 188 evaluated the impacts of SO42−, Cl−, NO3−, HCO3−, CH3COO−, and HPO42− on photocatalytic degradation efficiency. On the one hand, inorganic anions may adsorb onto the surface of the catalyst, which leads to a decrease in the number of holes and ·HO radicals, thereby reducing the reaction rate. On the other hand, the influence of these anions on photocatalytic degradation efficiency also depends on the intensity of oxidation performance of free radicals newly generated after the interaction among the anions and ·HO radicals. If the oxidation of newly generated free radicals is stronger than that of ·HO radicals, photocatalysis will be enhanced. Conversely, the formation of less reactive radical species results in diminished photocatalytic performance. Soulaima Chkirida et al. 201 studied the effects of Mg2+, Na+, and K+ ions in solution on adsorption and photocatalytic performance. Cations had little effect on adsorption. The impact of a single cation on photocatalytic efficiency was almost insignificant, but mixed cations could significantly reduce the COD removal rate to 70%. This is mainly due to the jointly strong adsorption of mixed ions on the surface of TiO2 and their capture of h+ and ·HO radicals during the photocatalytic process.
The process of the adsorption-photocatalytic degradation reaction
In General, the adsorption-photocatalytic process of nano-TiO2@adsorbent composites for dyes can be divided into three primary sequential steps. In the first step, the dye molecules are initially adsorbed on the surface of the composite photocatalyst rapidly. In the second step, nano-TiO2 catalysts irradiated by the light source generate h+ and free radicals (mainly ·HO). In the third step, dye molecules on the surface of composite photocatalysts are oxidized by h+/·HO. The rate of the photocatalytic reaction largely depends on the amount of dyes adsorbed on the photocatalyst surface and the reaction rate between h+/·HO and the adsorbed dye molecules. 202
It is particularly noteworthy that the initial adsorption and the final photocatalytic oxidation are mutually reinforcing. Dye molecules adsorbed on the surface of the composites are easy to be oxidatively degraded by photocatalysts due to their high concentration and short distance from photocatalysts. Following the degradation of the adsorbed species, the active adsorption sites on the surface of the composites are exposed again, enabling continuous capture of additional dye molecules in the solution. Therefore, adsorption and photocatalysis exhibit a synergistic effect.
Conclusion
For nano-TiO2@adsorbent composites, the types of adsorbents include inorganic adsorbents, carbonaceous adsorbents, and organic adsorbents. Adsorbents complete the preliminary driving step of dye wastewater treatment, namely decolorization and adsorption. The decolorization process increases the transparency of dye wastewater, while the adsorption process increases the probability of dye molecules coming into contact with nano-TiO2. The above phenomena are beneficial to the in situ photocatalytic degradation of adsorbed dyes by nano-TiO2, thereby improving the photocatalytic effect. In addition, some adsorbent materials combined with nano-TiO2 can generate heterostructures and increase charge separation, thus improving the reaction efficiency. The combination of adsorption, catalysis, and degradation processes greatly improves the efficiency of photocatalytic degradation for organic pollutants by nano-TiO2 catalysts. Theoretically, as long as the structure of the adsorbent material remains stable, the composite material can “automatically” degrade dyes in wastewater continuously. Therefore, the composite material is an efficient water remediation material for treating textile printing and dyeing wastewater. During the treatment of dye wastewater, it is necessary to optimize reaction conditions such as light source, solution pH, wastewater concentration, composite material dosage, and so on.
Prospect
Currently, most nano-TiO2@adsorbent composites are in powder form, prone to loss and difficult to recycle. Membrane materials, on the other hand, are costly and require high operating conditions, thus limiting their use. It is worth mentioning that environmental purification textiles are attracting researchers’ attention. Textiles, as a kind of low-cost and widely used fiber material, are characterized by unique structures and properties. First of all, textiles exhibit porosity and high specific surface area, with numerous potential binding sites (hydroxyl groups, amino groups, etc.) in the molecular structure of fibers, providing favorable conditions for the adsorption of pollutants and the loading of functional compounds. Secondly, fibers with wide sources, various types, and good flexibility, are easy for industrial production. Last but not least, textiles are textile-able and can be flexibly designed their weaves and structures. Photocatalyst@textile composite materials can be obtained by electrostatic spinning technology, post-finishing technology, and fiber surface modification technology. According to different needs, textiles can be designed as woven fabrics, knitted fabrics, and non-woven fabrics, and processed into shapes. It is expected that photocatalyst@textile composite materials are not only used for wastewater treatment. Their main characteristic is to control, eliminate or kill compounds or harmful organisms that pollute the natural environment and human living spaces, so as to protect the ecological environment and benefit human health. Therefore, the research on photocatalyst@textile composite materials can be carried out in the future. On the one hand, this study can address issues related to limitations in the use of composite materials, mainly in terms of recycling and cost. On the other hand, functional textiles can be developed and the added value of textiles can be improved.
Footnotes
Correction (April 2025):
In this article, sentence “Metal doping”, “Semiconductor compounding”,“Dye-sensitization”, and “Nonmetal doping” are in a parallel relationship. They belong to “Photosensitization modification of nano-TiO2” was inadvertently added to the “Methods” section and has now been removed.
