Abstract
Ammonium is a water pollutant that harms the environment, particularly fisheries. Rice straw is also an economic waste of rice, with millions of tons produced per year around the world. In this paper, physical and chemical procedures for the modification of rice straw were studied. The kinetic and isothermal adsorption trends were investigated, including the ammonium removal efficiency, the contact time of the adsorbent, the amount of adsorbent, and the initial concentration of NH4+. The effect of temperature and pH on the adsorption process was discussed. The removal efficiency of NH4+ recorded 43, 53.7, and 69.5%, with maximum adsorption values of 2.9, 3.5, and 4.5 mg/g at temperatures of 25 ± 5, 35 ± 5, and 45 ± 5°C, respectively, at pH 7.5. The biochar obtained from rice follows the pseudo-second-order equation for ammonium adsorption kinetics (R2 = 0.98). The adsorption isotherm follows Freundlich’s model (R2 = 0.99) and Langmuir’s model (R2 = 0.98).
Introduction
In many nations, eutrophication is a water contamination issue in lakes, rivers, water repositories, and seas. Our study aims to tackle these issues by investigating the nature and reuse of wastewater, which reduces sewage release and enhances water deficiency conditions. Investigations of sewage medication have also been performed. The effect of ammonium was observed in groundwater, aquaculture, and surface water, on the other hand, causing eutrophication (Huo et al., 2012). The level of ammonium in surface water and groundwater is lower than 3 mg/l. This level is unacceptable in other areas, which require levels lower than 0.05 mg/l (e.g. in aquaculture) (Vu et al., 2017). Current treatment methods of water and wastewater such as coagulation and filtration are not masterful to remove ammonium effectively (Moradi, 2011). Various methods have been used to remove ammonium from wastewater, including precipitation, crystallization, biological removal, adsorption, and ion exchange. The adsorption and ion exchange techniques have a high efficiency to eliminate ammonium. These techniques need a small area for application and have acceptable efficiency in comparison to other methods (Abdehagh et al., 2013; Ma et al., 2012; Namasivayam and Sangeetha, 2006; Vargas et al., 2012; Zhu et al., 2005). Identifying new productive materials that may be used for adsorption and ion exchange is an important research topic (Li et al., 2016; Liu et al., 2010; Taheran et al., 2016).
However, adsorption onto the surface of activated carbon is the most widely used method in water treatment (Liu et al., 2013; Zhu et al., 2012). Activated carbon can remove many of the impurities that occur in water and wastewater (Sarker and Fakhruddin, 2015).
Recently, the use of adsorbents from nature, such as agricultural waste, as alternative resources to replace traditional adsorbents. These resources are economical and environmentally friendly solutions. The majority of previous studies have investigated the adsorption of heavy materials and gases, but few studies emphasized nutrients, such as sawdust for removing phenol (Jadhav and Vanjara, 2004), textile sludge biochars for removing oil (Sohaimi et al., 2017), sewage sludge for removing methylene blue from aqueous solutions (Fan et al., 2017), the removal of cadmium (II) (Guo et al., 2017; Wang et al., 2017; Zama et al., 2017), and atrazine and imidacloprid removal (Mandal et al., 2017). This method has been applied to many natural materials, such as wheat straw (Ma et al., 2011); activated carbon (Björklund and Li, 2017; Gierak and Łazarska, 2017); clinoptilolite (Martins et al., 2017); light-expanded clay aggregate (Sharifnia et al., 2016); wood and rice husks biochar (Cope et al., 2014; Kizito et al., 2015); peanut shells, corn cobs, and cotton stalks (Liu et al., 2016; Vu et al., 2017); six types of wetland plants (i.e. Canna indica, Pennisetum purpureum Schum, Thalia dealbata, Zizania caduciflora, Phragmites australis and Vetiveria zizanioides) (Cui et al., 2016); palladium (II) after recrystallization (Mozaffari et al., 2017; Xue et al., 2017); Al-eggshell for adsorption phosphate (Zhang et al., 2017); and treated ginger waste (Ahmad and Kumar, 2010). Literature reviews exhibit the possibility to reuse and develop agricultural wastes as cheap materials for adsorption. However, the conversion of agricultural waste into charcoal is not simple, as this process depends on several factors to activate the above materials.
Adsorption is a cheap and effective process for removing ammonium. The adsorption of ammonium has been studied through the wastewater treatment process by various adsorbents like activated carbon, biochar, zeolite, and corncob-biochar (Gupta et al., 2015; Ivanova et al., 2010; Lebedynets et al., 2004; Vassileva et al., 2009).
A previous research approved that the surface chemistry plays a more important role than the Brunauer–Emmett–Teller surface area of adsorbents and the pore structure in adsorbing inorganic compounds from water. The modification strategy is a current method to change the surface chemistry and promote the adsorption efficiency of adsorbents. The oxidation process with H2O2, KMnO4, H3PO4, or HNO3 has been used for modifying the surface characteristics (Abou El-Maaty, 2014; Halim et al., 2013; Moreno-Castilla et al., 2000).
Rice straw (RS) is an environmental burden in many countries. Thus, in this paper, we examine methods of RS activation for work in the adsorption environment. The internal composition of RS is similar to that of wood, which qualifies RS for conversion to good charcoal. We are interested in the adsorption of ammonium by RS for use as a cheap material to recycle water in fish farms with ammonium; it is also worth mentioning that most of these fish farms are near rice farms due to their common necessity of water, as in Egypt. Therefore, the activation of RS for adsorption has been studied using kinetic and isothermal models; on the other hand, the influence of various factors, such as temperature and pH, on the adsorption process was examined to obtain optimum conditions based on laboratory results and mathematical models (e.g. Lineweaver–Burk, Elovich, pseudo-second-order and pseudo-first-order models for kinetic studies and Freundlich and Langmuir models for isothermal studies).
The overall aim of this study was to determine adsorption characteristics of ammonium with a concentration beginning from 2 mg/l (to simulate wastewater of fish farming in Egypt) on biochars obtained from RS in different environmental conditions (including contact time, pH, ammonium concentrations, and adsorbent dose) to determine the kinetics and equilibrium adsorption parameter. These help to achieve insights in the utilization of biochar obtained from RS to adsorb ammonium and improve water quality in fish farms, which operate through water recycling by simple and easy technique.
Methods and materials
Activation of the material (adsorbents)
Biochar synthesis and activation
The biochar was produced from RS that was collected from Kafr El Sheikh city, Egypt and transported to Don State Technical University in Rostov-on-Don city, Russia. The RS material was washed to remove dust by tab water. The sample was dried at a temperature of 105°C until weight stabilization was achieved. The RS was cut to a size range of 1–1.5 cm and then soaked with 1.0 M of NaOH solution at a solid-to-liquid ratio of 1.0 g to 2.5 ml for 2 h. The following procedures were used by Gao et al. (2015) and Liu et al. (2016). The RS after soaking with NaOH was washed four times with tap water, then adjusted pH to 7–8 for the sample by washing with distilled water for three times (Jindo et al., 2014; Körner et al., 2001), and subsequently dried along with storage RSI (chemical activation).
Thermal analysis and thermal activation
Thermal analysis of the rice straw (RSI) has been performed using a NETZSCH-SAT-449 system under the following conditions: raising the temperature of a sample to 600°C with heating rate 30°/min under a static air atmosphere and crucible DSC/TG pan Al2O3. Furthermore, thermal activation was applied on the RSI by heating under slow pyrolysis at 500–600°C in the oven for 1 h and obtaining the RSII in 0.16 mm particles.
Adsorbate
Ammonium chloride was obtained from Aquatest company in Rostov-on-Don city, Russia (M= 53.5 g/mol). Five solutions with different concentrations of ammonium (3, 8, 12, 18, and 25 ppm) were prepared by dissolving 0.0446, 0.1188, 0.1783, 0.2673, and 0.371 g of ammonium chloride (NH4Cl), respectively, in 5 l of distilled water.
Experimental adsorption procedures
Adsorption kinetics
The adsorption kinetics of ammonium on biochar obtained from rice straw (RSII) were investigated by adding different doses of RSII (0.25, 0.5, and 1 g) to 250 ml of an ammonium chloride solution at a rate of 2 g/l (Yao et al., 2012). The initial concentration of ammonium is 12 ppm was added in different flasks, which placed in a shaker at 120 r/min at room temperature (25 ± 0.5°C) for a suitable amount of time to ensure mixing adsorbent with the solution. Samples were removed from the flasks for analysis at 5, 10, 15, 20, 30, 40, 60, 90, and 120 min. The samples were passed through 0.22 µm membrane filters to measure residual ammonium concentrations using an Expert 001 pH meter ionomer. All experimental results were measured in triplicate, by calculating average values. Pseudo-first-order, pseudo-second-order, Elovich, and Lineweaver–Burk models were used to analyze and describe the experimental data related to adsorption kinetics. The equations of kinetic models and their linear forms are given below:
1. Pseudo-first order (Macías-García et al., 2016)

The linearized form is
1. 2. Pseudo-second order


The linearized form is
1. 3. Elovich (Thilagan et al., 2013)


The linearized form is
1. 4. Lineweaver–Burk (Compostela, 1988)


The linearized form is

The ammonium removal efficiency (

Adsorption isotherm
The same sampling, mixing, filtration, and analysis procedures described for adsorption kinetics were used to identify the adsorption isotherms of ammonium in the RSII. Instead of different time periods, 0.5 g of sorbents was mixed with 250 ml of ammonium solutions with varying initial concentrations: 3.15, 8, 12.1, 18, and 25 mg/l. The Langmuir and Freundlich models were used to analyze the experimental data for the adsorption isotherm (Langmuir, 1918; Yao et al., 2011). The supposition of the Langmuir isotherm is based on sorption occurring at specific homogeneous sites of adsorbent heat (as adsorption for all sites is constant); however, the Freundlich isotherm is assumed to be a heterogeneous surface (multilayer adsorption) with a nonequal distribution of adsorption heat over the surface (Novak et al., 2010; Wu et al., 2017; Yusof et al., 2010). The Langmuir and Freundlich equations can be written as shown below:
1. Langmuir model

The linearized form is
2. Freundlich model


The linearized form is

During the experiments, the pH was monitored at all steps to create optimal conditions for ammonium absorption, which depends directly on the pH. The pH in all experiments was 7.5 ± 0.5, for the reasons mentioned in section effect of pH.
Results and discussion
Raw material characterization
Physicochemical properties of RS waste
The chemical configuration of RS depends on the planting time, climate, type of rice, source of water, and geographical conditions (Agbagla-Dohnani et al., 2003). The quality parameters and main characteristics of the initial RS are presented in Table 1.
Chemical analysis of the raw material.

aW is ash content for dry weight.
bA is moisture.
cSt is total sulfur.
dVΣ is total pores.
RS has a high concentration of silica and can be categorized as attractive material that contains silica. In addition, in the thermal analysis (based on temperature), silica is responsible for the type of ash that is formed (Elwan et al., 2006), with amorphous ash exhibiting reduced adsorption. Sodium hydroxide was selected to dilute the silica in the raw material, which uses the first step in the modification of RS. The reaction of sodium hydroxide and silicon oxide can be written by the following chemical equation

Results of the TG–DTG analysis of RS
Four temperature zones were obtained from the thermogravimetric analysis curve (TG–DTG) of raw RS (from room temperature to 120°C, from 120 to 370°C, from 370 to 500°C, and from 500 to 600°C), based on weight loss, as illustrated in Figure 1.

TG–DTG curve for RS. DTG: Differential thermogravimetry; TG: Thermogravimetry.
These zones were explained according to a previous study (Mall et al., 2006). The second zone recorded a high percentage of weight loss in comparison to the other zones. The weight loss was approximately 6.46% during the heating of RS up to 120°C, due to the loss of moisture in the RS. A significant weight loss of approximately 50.12% was recorded in the second zone from 120 to 370°C, which was due to the degradation of the cellulose-related component that forms carbon. Lignin was decomposed in the third zone (370–500°C), with a mass loss of 13.8%, indicating better stability than cellulose and hemicellulose. The last zone was from 500 to 600°C, with a slight loss of mass that stopped at 600°C. The final mass was approximately 29% of the initial weight, indicating that all organic compounds had been completely converted. Finally, the appropriate temperature to activate RS to RSII is 500–600°C.
Effect of pH
The ammonium adsorption of RSII was investigated by creating three pH groups. The first group used RSII without adjusting the pH, which was physically and chemically activated (burn RSI after soaking with NaOH, and without washing to produce RSII−1), while pH ranges were recorded in these groups from 9 to 10.2. The second group is pH 7, which was obtained by washing RS after soaking it with a solution of NaOH prior to a burning process. This pH range was achieved after washing RS with tap water and distilled water for three times. The third group was achieved by adding HCl to generate a pH from 2 to 6. In Figure 2, with a low pH from 2 to 5.5, the percentage of removed ammonium was low, while the process achieved the highest percentage of ammonium removal at 12%. We noted that when the pH increases from 5.5 to 8 the removal efficiency of NH4+ reaches to 57%, but when the pH increases from 9 to 10.2 the removal efficiency of NH4+ reaches to 15%.

Effect of pH on ammonium ion removal efficiency (initial concentration of NH4+ (
Decreased adsorption at different pH values is caused by ammonium ion effects with free hydrogen ions (H+) in the solution. The low efficiency of the adsorption process at a lower pH could relate to high protonation of functional groups (C = O, COO−) (HA + B ↔ A− + HB+) on the biochar surfaces, which impart a partial positive charge that repels the polar attraction of NH4+ ions in aqueous solutions. In the solution, there is competition between H+ and NH4+ ions adsorbed on the surface of an adsorbent. This competition is in favor of H ions when decreasing the pH in the solution.
Here, the ammonium adsorption in water followed the ion exchange mechanism. High pH values > 10 lead to the conversion of most ammonium ions into ammonia, which cannot be adsorbed. Therefore, all steps of adsorption processes were carried out with pH values of 7–8 (Huang et al., 2010; Jindo et al., 2014; Novak et al., 2010).
Adsorption studies: Kinetics and isotherm
Adsorption kinetics of RSII
The relationship between the rate of adsorption and the contact time shows the kinetics of adsorption. The process of NH4+ adsorption by RSII with different contact times and different concentrations is shown in Figure 3. Adsorption kinetics of RSII occurs in three main stages: fast, slow, and equilibrium. The first stage is the fastest stage (up to 15 min), the adsorption rate increased quickly, where half of the adsorption capacity is obtained. The NH4+ adsorption began to slow down in the second stage, which occurred from 15 min to the equilibrium stage of adsorption at 60 min. We also noticed that the adsorption rate increased with an increasing initial concentration of the solution at all stages of the study.

Adsorption kinetics of NH4+ onto RSII (temperature = 25 ± 2°C, pH = 7.5,
The speed of the reaction can be explained for RSII, where some anionic groups with negative charges are located on the surface of the adsorbent such as –COO−. These groups rapidly interact with ammonium ions. At the end of this reaction with the anionic groups, the NH4+ goes into a slow process, which represents adsorption on the internal skeleton of the RSII. So, the NH4+ adsorption on RSII consisted of two reaction processes. Also increasing the concentration of ammonium leads to congestion of cations in the solution, which increases the chances of adsorption as well as its speed.
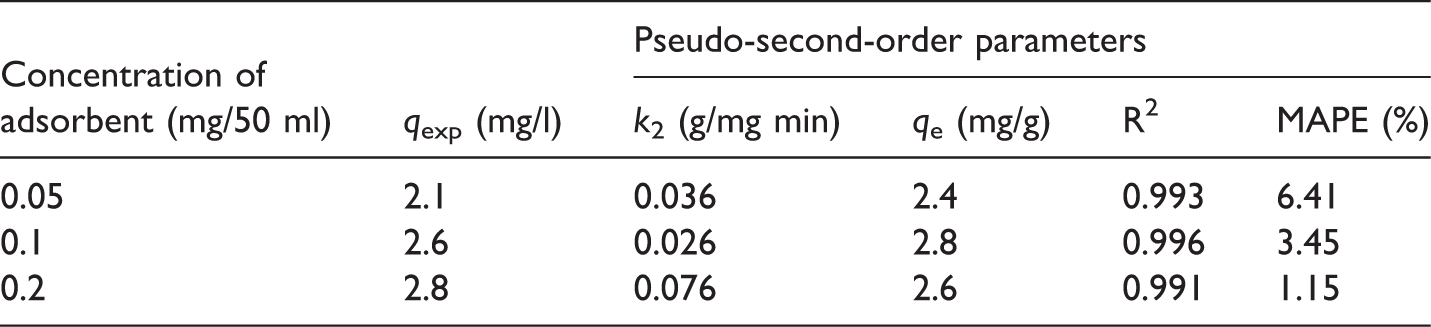
The kinetic NH4+ adsorption data were fitted with four kinetic models. Table 2 shows the model parameters with R2 and experimental values for five initial concentrations of the NH4Cl solution. As shown in Table 2, the results showed that the Elovich, pseudo-first-order, and pseudo-second-order models are suitable for describing the adsorption kinetics, with all R2 values being larger than 0.87. However, the pseudo-second-order model had the best fit, based on R2 values and decreases of 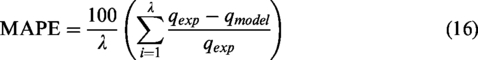
where
Coefficient of determination (R2) for the Lineweaver–Burk, Elovich, pseudo-first-order, and pseudo-second-order kinetic models and model parameters.

aInitial concentration of ammonium in aqueous solution.
bExperimental results for the adsorption capacity at the equilibrium.
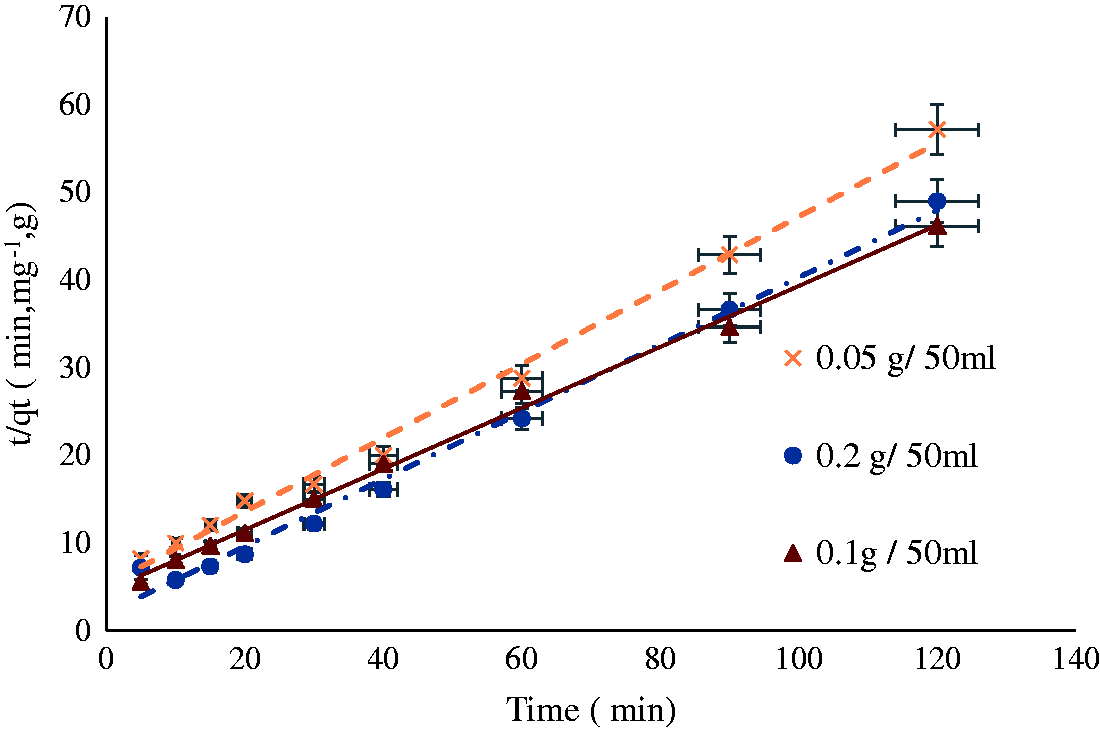
Pseudo-second-order plot (
Parameters of the pseudo-second-order model, with an initial concentration of NH4+ = 12 ppm at different concentrations of adsorbent RSII.
MAPE: mean absolute percentage error.
Adsorption isotherm of RSII
The experimental data for isotherm adsorption were placed in the Langmuir and Freundlich models and R2 values were extracted for both models. The adsorption isotherm of RSII for NH4+ is described in Figure 5 using the Langmuir and Freundlich models. The parameters of these models with correlation coefficient R2 were presented in Table 4. The results show that the Freundlich and Langmuir models accurately describe the adsorption of NH4+, with R2 values larger than 0.98 for all models, but the Freundlich model coincides with the experimental data better than the Langmuir model, with R2 = 0.99. In addition, the n constant for the Freundlich isotherm measures exchange intensity or surface heterogeneity and also indicates the ease of adsorption when n > 1. In this study, the value of n was larger than 1, which suggested that the adsorption conditions fit with the Freundlich model.

Ammonium adsorption equilibrium prediction on RSII from the Langmuir and Freundlich models (contact time = 120 min, temperature = 25 ± 2°C, and pH = 7.5).
Isothermal parameters for the Langmuir and Freundlich models of adsorbent NH4+ on RSII.

Effect of temperature on ammonium removal efficiency
The influence of three different temperatures on the adsorption of ammonium on RSII was investigated. In the experimental study, the concentration of adsorbent of RSII was 0.1 g/50 ml with the initial concentration of ammonium (

The pseudo-second-order equation model at different temperatures (
Conclusions
This study examined the potential use of biochar obtained from RS for NH4+ adsorption from aqueous solution. We investigated the factors that influence the process of adsorption: physical and chemical methods of modifying RS, contact time, adsorbent dose, initial concentration of adsorbate, and pH solution. The rice straw RSII achieved a removal efficiency of 43% of ammonium at room temperature (25°C), with an initial concentration of NH4Cl of 12 mg/l and a pH of 7.5 ± 0.5. The adsorption equilibrium of NH4+ was achieved after a contact time 90 min, with the best range of pH 7–8 yielding an improved adsorption performance. The kinetic data for the adsorption of NH4+ fit with the pseudo-second-order kinetic model. The isothermal data coincided with Freundlich’s model, which achieved R2 = 0.99 and Langmuir’s model (R2 = 0.98).
Overall, these results suggest that biochar obtained from rice straw (RSII) can be used effectively for ammonium removal from aqueous solutions.
Footnotes
Acknowledgements
Authors are thankful to Kafrelsheikh University and Don State Technical University for supporting our work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
