Abstract
The properties of graphene enriched silk depend on the amount of graphene oxide (GO) adsorption on silk. GO dipping parameters include the GO solution pH, initial GO concentration, dipping time and temperature, and the type of substrate. The effects of GO dipping parameters on the sorption process are studied to achieve the maximum GO adsorption on the silk surface for the preparation of an economical graphene/silk-based textile product. In addition, equilibrium isotherms, kinetics, and thermodynamics of GO adsorption on the silk surface in a batch sorption process are examined to understand the adsorption mechanism. The Freundlich isotherm best describes the adsorption of GO onto the silk. A pseudo-second order kinetic model best describes the kinetics of GO adsorption. Thermodynamic studies reveal that GO adsorption is spontaneous and exothermic.
Introduction
Graphene is a flat, single layer of closely packed sp2-hybridized carbon atoms in a 2D honeycomb crystal lattice and is a fundamental structural feature of all other dimensionalities of graphitic materials such as fullerenes (0D), carbon nanotubes (1D), and graphite (3D). Geim and Novoselov successfully isolated single layers of graphene by micro-mechanical exfoliation in 2004. 1 This revolutionary discovery has opened a new era in the fields of physics, chemistry, biotechnology, and material science. This materials exhibit excellent electrical conductivity, mechanical properties, thermal conductivity, optical transparency, easy chemical functionalization, and low coefficients of thermal expansion (CTE) behavior.2,3 Graphene is prepared by three major routes: sonication, vapor deposition, and chemical methods. The chemical methods were used as a novel method of large-scale production of graphene. 4 Using a synthetic chemical method, a strong oxidizing agent reacts with graphite (precursor) to produce graphene oxide (GO). Oxygenated functionalities, such as epoxides (bridging oxygen atoms), carbonyls (C=O), and hydroxyls (-OH) are introduced into the graphite structure, not only to expand the layer separation, but also to make the material hydrophilic in nature. GO is an sp3-hybridized carbon atom network that is electrically insulating in nature. Various methods, such as thermal, electrochemical, ultraviolet, and chemical, can be used to restore the sp2-hybridized carbon atom network from the sp3-hybridized GO structure to give reduced graphene oxide (rGO). 2 Textile fibers have the advantage of possessing high surface area, are lightweight and flexible, and have adequate mechanical properties. Furthermore, the presence of functional groups in textile materials facilitate the adsorption of GO through electrostatic interaction, van der Waals forces, hydrogen bonding, π-π interactions, hydrophobic interactions, and so forth. 3
Prior to this research work, a comparative GO mass loading study was carried out to identify the most suitable textile substrate. Cotton, viscose, silk, wool, nylon, polyester, and acrylic fibers were coated with a 1% GO solution using up to five dipping cycles. Cotton yielded the highest GO add-on per unit surface area, followed by silk, viscose, nylon, wool, polyester, and acrylic after five dipping cycles. However, the cotton surface fractured due to prolong exposure under acidic conditions. Moreover, silk showed the second lowest mass-specific electrical resistance after cotton. Therefore, silk was chosen as an adsorbent for GO to prepare an economical electroconductive textile.
Silk is a natural polyamide fiber widely used for apparel, home furnishings and biomaterials due to its excellent mechanical properties, softness, breathability, biodegradability, and biocompatibility.5,6 Unlike cellulosic fibers, silk fibers interact with GO via hydrogen bonding and electrostatic interaction, which increase the GO mass loading.7-9 Graphene enriched silk was explored for use in numerous applications involving electrical conduction,7,10 water repellency, 7 UV-blocking, 7 flame retardancy, 11 antibacterial activity, 12 tissue scaffolds, 6 , glucose biosensors, 10 H2O2 detection, 10 flexible electrodes, 10 wearable biomedical devices, 10 and others.
However, limited information is available regarding the effect of process conditions such as pH, concentration, dipping time, and dipping temperature on the adsorption capacity of GO sheets onto the surface of silk. Furthermore, no study has investigated the GO adsorption behavior and mechanism of adsorption onto silk. In this study, process conditions were optimized on the basis of GO adsorption capacity. Silk was used as the adsorbent and GO treated silk was prepared by the dip coating method. For a detailed understanding of GO adsorption behavior and mechanism on silk, adsorption kinetics, isothermal, and thermodynamic studies were conducted. All studies were carried out in loose fiber form to avoid the structural influences of yarn or fabric on the adsorption process. This study identified the most suitable GO dipping conditions for the maximization of GO mass loading without deteriorating the GO surface chemistry.
Experimental
Materials
Graphite powder (particle size of 44 microns) was purchased from Alfa Aesar-England. Sulfuric acid (H2SO4, 98%), phosphoric acid (H3PO4), potassium permanganate (KMnO4), and hydrochloric acid (HCl, 37%) were acquired from Rankem-India. Sodium dithionite (Na2S2O4), hydrogen peroxide (H2O2, 30%), and ethanol (C2H5OH) were purchased from SD Fine Chem Limited-India. All chemicals used in this work were analytical reagent-grade. Milli-Q grade (resistivity of 13 M?.cm) deionized water (DI) was used throughout the experiments. Degummed silk (Rajesh Silk Fabrics, India) was used as adsorbent for current the adsorption study.
Preparation of GO Solution
Graphene oxide (GO) is chemically exfoliated from graphite by the improved Hummers’ method. 13 The obtained GO suspension is filtered through a PTFE membrane and washed with a 5% hydrochloric acid solution (3 times) and deionized water (4 times). During each washing cycle, the GO suspension is centrifuged at 8000 rpm for 10 min to remove un-exfoliated GO. 13 The centrifuged GO is then dried at room temperature (RT) for 24 h to prepare GO in powder form. GO stock solutions of 2 g/L are prepared by dissolving 2 g of dried GO powder in 1 L of deionized water. The 2 g/L stock solution is diluted with deionized water to prepare five different concentrations of GO solution (100, 200, 350, 500, and 1000 mg/L).
Preparation of GO Adsorbed Silk
GO enriched silk (GO-silk) is prepared by the dip coating method. In this method, 0.4 g of silk fiber is immersed in 40 mL of 1000 mg/L GO solution for 2 min at 40 °C. The initial pH of the GO solution is maintained at 2. After treatment, the treated silk is removed from the solution and excess solution is squeezed out. The residual concentration of the GO solution is determined by UV-Vis spectrophotometry.
Characterization
Scanning Electron Microscopy
The surface morphology of GO-silk is analyzed with a JSM-6510 LV (JEOL, Japan) scanning electron microscope (SEM) in which the sample is sputtered with a thin film of gold before being tested. For the SEM analysis, a 20 kV acceleration voltage is applied under high vacuum.
X-ray Diffraction
X-ray diffraction (XRD) spectra of the graphite powder, synthesized GO, silk, and GO-silk are recorded using a Empyrean X-ray diffractometer (PANalytical B.V., The Netherlands) with Cu-K radiation (λ= 1.5406 Å). These spectra are recorded at 25 °C with a step size of 0.0170° (2Θ).
UV-Visible Spectroscopy
The UV-Visible spectrum of graphene oxide (30 mg/L concentration) is recorded from 200 to 800 nm using a LAMBDA 365 UV-Vis Spectrometer (Perkin Elmer, USA).
Fourier Transform Infrared Spectroscopy
Fourier transform infrared spectroscopy (FTIR) analyses is carried out to confirm the oxidation of graphite and to examine bond formation between GO and silk from 400 to 4000 cm−1, using an ALPHA-II FTIR spectrometer (Bruker, USA) in transmittance mode with attenuated total response (ATR). FTIR spectra are recorded with a diamond crystal at 64 scans and 2 cm−1 resolution.
Raman Spectroscopy
Raman spectra of graphite, GO, silk and GO-silk are recorded with an inVia Raman spectrometer (Renishaw PLC, UK). Raman spectra are recorded from 1000 to 2000 cm−1 with a 785 nm laser.
Batch Adsorption Studies
The extent of GO adsorption on silk is determined from the residual concentration of GO in the solution after treatment. For determination of residual concentration of GO in the solution, a standard plot of GO concentration vs absorbance at 229 nm is prepared using the UV-Vis spectrophotometer. For treatment, a solution of requisite concentration is diluted from stock solution, and a test solution having a pH ranging from 2 to 7 is prepared by addition of 0.01 N HCl and 0.01 N ammonia solution. Silk is dipped in the aqueous GO solution and then removed. Residual GO concentration in the solution is determined from the standard plot. Each batch experiment is repeated five times and their average values are used for the calculation. The null hypothesis is assumed and expressed as “no significant difference exist between two mean uptake adsorption capacities.” Statistical significance tests are conducted with the two-tailed Student's
The adsorption capacity
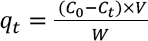
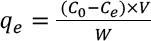
Adsorption Kinetics
A chemical kinetics study is carried out to find the time to attain equilibrium and the mechanism of adsorption of GO on silk. Pseudo-first order, pseudo-second order, intra-particle diffusion and liquid film diffusion models are fitted with experimental data to explain the kinetics and rate controlling step for the adsorption of GO on silk. The kinetic experiments are conducted at two different concentrations (550 and 1000 mg/L) to evaluate the effect of the initial concentration on kinetic parameters.
The linearized form of the Lagergren pseudo-first order kinetic model is given in Eq. 3.
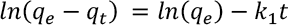
Where
The pseudo-second order model is shown in Eq. 4.
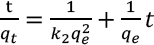
The calculated value of
The transfer of adsorbate to adsorbent from the solution phase involves three steps: solution moves to the external surface of the adsorbent or film diffusion, moves from the external surface to the porous structure of the adsorbent or intra-particle diffusion, and later, control by adsorption and desorption on the active sites of the adsorbent or equilibrium diffusion mechanism. 15 Liquid film diffusion is plotted to determine the rate controlling stage of the adsorption of GO onto silk. The mathematical form is given in Eq. 5.
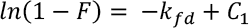
To study the role of intraparticle diffusion mechanism in the adsorption process of GO on silk, the Weber-Morris model is used (Eq. 6).
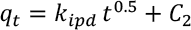
Intraparticle diffusion rate constant
Validity of Kinetic Model
Root-mean-square error
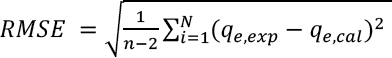
Isothermal Studies
The adsorption isotherm expresses the variation in the amount of adsorbate on the adsorbent surface as a function of concentration at constant temperature. Aqueous GO dispersions at five concentrations (100, 200, 350, 550, and 1000 mg/L), are diluted from a stock solution and used in adsorption isotherm experiments. The other process conditions are as follows: the amount of silk is 0.4 g, dipping time is 2 min, pH is 2, and temperature is 40 °C and 70 °C. The most common adsorption isotherm models (i.e., Langmuir, Freundlich, and Dubinin- Radushkvich (D-R)) are used for analyzing the adsorption equilibrium of GO nanosheets on the silk surface.
The Langmuir isotherm (Eq. 8) assumes that a fnite monolayer adsorption occurs on energetically homogeneous surface without any interaction between two adsorbed molecules.
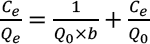
The efficiency of GO adsorption on silk is investigated by applying a dimensionless separation factor (RL) which can be expressed as in Eq. 9.
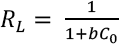
The Freundlich isotherm assumes an infinite multilayer adsorption on a heterogeneous surface. Its logarithmic form is expressed in Eq. 10. 14
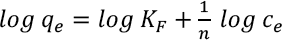
The D-R isotherm model is based on the concept that volume filling of the adsorbate occurs on the microporous sites of adsorbent, as represented in Eq. 11.
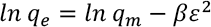
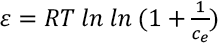
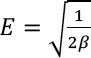
Thermodynamics Studies
Thermodynamic parameters are evaluated from the batch adsorption experiment in the temperature range of 30 °C to 70 °C. Adsorption thermodynamic enthalpy (ΔH°)and entropy (ΔS°) are determined from the Van't Hoff plot between
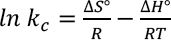
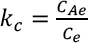
The change in Gibbs free energy upon adsorption Δ
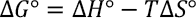
Results and Discussion
SEM
SEM images of GO treated silk at 2500 and 10,000 magnifications are shown in Figs, 1a and b, respectively. These SEM images show that large amounts of GO sheets are uniformly present on the silk surface. However, the connectivity between the GO nano-sheets was inadequate due to single dipping in the GO solution. A multi-layer configuration of the GO sheets was observed on the silk surface (Fig. 1b).

SEM image of GO-silk.: (a) GO-silk X2500 and (b) GO-silk × 10,000.
XRD
XRD analysis of graphite and intercalated GO is shown in Fig. 2a. The XRD pattern of graphite shows a characteristic strong and intense diffraction peak at 2Θ = 26.4°, corresponding to a basal spacing d002 = 0.337 nm. The diffractogram of intercalated GO shows a strong and sharp peak at 2Θ = 9.2 °, corresponding to a basal spacing d001 = 0.994 nm. The inter-layer spacing of GO is larger than graphite due to the attached functionalized oxygen-containing groups (e.g., hydroxyl, epoxy carbonyl, and carboxyl groups) on the basal planes and edges of the GO structure. This increase in basal spacing confirms the oxidation of graphite and transformation of sp2 hydrophobic carbons into disordered sp3 amorphous carbons network 19
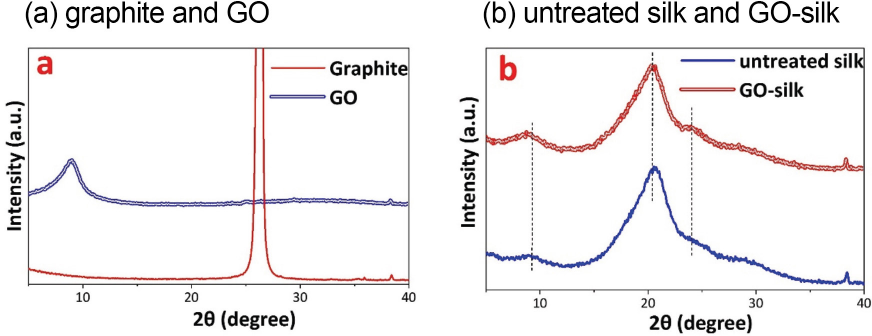
XRD pattern. (a) Graphite and GO, and (b) untreated silk and GO-silk.
The XRD spectra of untreated silk and GO-silk are represented in Fig. 2b. The XRD spectrum of untreated silk shows three 2Θ peaks at 9.1°, 20.4°, and 24.2°. These peaks are associated with the silk ιI crystal structure., The characteristic GO peak (2Θ = 9.2°) is not observed in the diffractogram of GO-silk. This was due to the small amount of GO adsorbed on the silk by a single dipping in a dilute GO solution. It also implies that GO nano-sheets were uniformly deposited on the silk fiber surface and that the introduction of these GO nano-sheets did not influence the crystal structure of silk 12
UV-Visible Spectra
The chemical exfoliation of GO was verified by UV-Vis spectroscopy in the range of 200 to 800 nm (Fig. 3a). The UV-Vis spectrum of GO shows a peak centered at 229 nm which corresponds to π→π* transitions of aromatic C=C bonds. 21 This confirms the successful chemical exfoliation of graphite and functionalization of GO with oxygen functionalities.
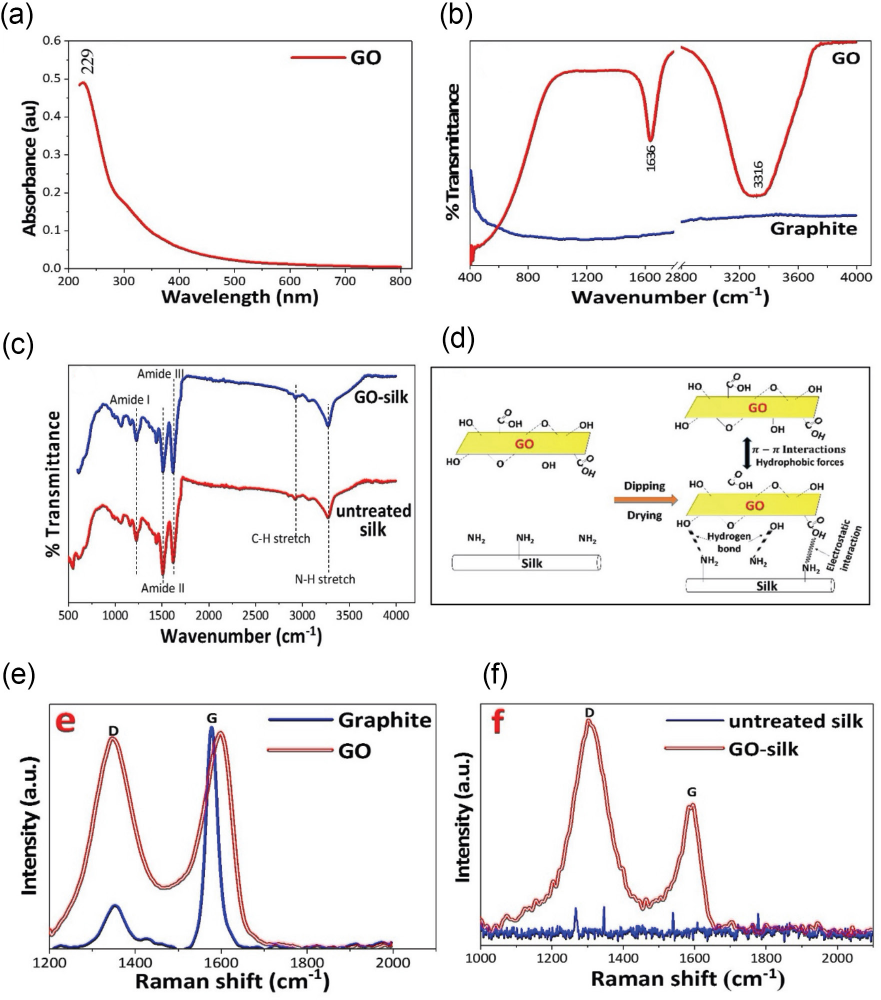
(a) UV-Vis spectra of GO, (b) FTIR spectra of graphite and GO, (c) FTIR spectra of untreated silk and GO-silk, (d) interaction between GO and silk, (e) Raman spectra of graphite and GO, and (f) Raman spectra of untreated silk and GO-silk.
FTIR
Fig. 3b shows the FTIR spectra of graphite and GO. In the graphilte sample, no significant peak was observed due to the absence of any functional group. The FTIR spectrum of GO shows multiple signs of oxygen-derived functional groups present in the structure. The intense peak at 3316 cm−1 corresponds to the O-H stretch of carboxylic acid, due to the presence of absorbed water molecules. The intense peak at around 1636 cm−1 is attributed to C=O group stretch. These peaks confirmed the presence of GO and the successful chemical oxidation of graphite. 22
FTIR spectra of untreated silk and GO-silk is represented in Fig. 3c. Untreated silk exhibited characteristic adsorption band at 1700-1600 cm−1 (amide-I), 1560-1500 cm−1 (amide-II), and 1300-1200 cm−1 (amide-III). 6 After adsorption of GO onto silk, intensity of all the vital peaks increased and moved to a higher region. The band position at 3273 and 2926 cm−1 corresponded to amide A (N-H stretch), and the C-H stretch moved to 3259 and 2915 cm−1 after the deposition of GO nano sheets on silk. The peak positions of amide III, II, and I at 1228, 1510, and 1619 cm−1 moved to 1226, 1508, and 1617 cm−1, respectively. These changes suggest that GO interacts with silk via hydrogen bonding. Further multilayer deposition of GO occurs on silk by electrostatic interaction, π-π interactions, and hydrophobic forces. 7 The possible interaction between GO and silk is represented in Fig. 3d.
Raman Spectra
Raman spectroscopy is commonly used to identify the structural fingerprints of carbon-based material. Raman shifts for graphite and GO are shown in Fig. 3e. Raman spectra of carbon nanomaterials are found in the range of 1200 to 1800 cm−1. Both graphite and GO show intense peaks at 1350 and 1600 cm−1 corresponding to the D and G bands, respectively. The D band is associated with defects and disorder (sp3-hybridized) in carbon network caused by the graphite edges due to strong oxidation. The G band corresponds to the first-order scattering of the E2g mode due to the in-phase vibration of the sp2-hybridized C-C bonds in the graphite lattice.
23
The Raman spectra of graphite and GO shows typical D band peaks at 1349 and 1343 cm−1. The small intensity D band in graphite appear due to the defects and disorder in the hexagonal graphitic layers and presence of amorphous carbon species. The D band intensity in the GO spectrum increased due to the transformation of in-plane sp2 hydrophobic carbons into disordered sp3 amorphous carbons network by the introduction of oxygenated surface functionalities during oxidation. The G band appears at 1576 and 1599 cm−1 for graphite and GO, respectively. The G band in GO is blue shifted to 1599 cm−1 due to the reduction in size of the in-plane sp2 domains due to the strong oxidation of graphite.
24
The intensity ratio of D band to G band determines the quality of the product. Compared with graphite, the band intensity ratio (
Untreated silk and GO-silk samples are characterized by Raman spectra, which is an efficient tool to verify the GO deposition on silk. As shown in Fig. 3f, the D and G bands are absent in untreated silk. However, after GO adsorption onto silk, GO-silk shows two prominent peaks at 1304 and 1596 cm−1, which are assigned to D band and G band, respectively. The intensity of the D band was higher than the G band and the band intensity (
Effect of pH on Adsorption
pH influences the surface charges of the adsorbent and adsorbate in solution. The effect of initial pH on the adsorption process was determined by dipping 0.4 g of silk in 40 mL of 350 mg/L GO solution for 2 min at 40 °C and varying the solution pH from 2 to 7. Study below pH 2 was not performed due to the instability (coagulation) of GO. 26 Since higher pH solutions resulted in GO precipitation and the formation of hydroxyl complexes, solutions up to pH 7 were investigated for GO adsorption. 27
Fig. 4 shows the adsorption capacity of GO on silk in the pH range of 2 to 7. A maximum adsorption capacity of 11.07 mg/g was achieved at pH 2. Adsorption capacity then decreased with increasing pH. There was a sharp decrease in adsorption capacity going from pH 3 to 5 (10.64 mg/g to 6.04 mg/g). On further increasing the pH from 5 to 7, no significant difference was observed in the adsorption capacity (6.04 mg/g to 5.49 mg/g). Effect of pH on adsorption capacity may be due to the nature of surface charges of the adsorbent and adsorbate in solution. GO is anionic in the pH range 2 to 7. However, silk is cationic at pH < 3.5 and anionic at pH greater than 4.11 This may be reason for more GO nanosheets deposition on silk via electrostatic attraction at pH range 2 to 4. Further increase in pH changes the character of the silk from catatonic to anionic, leading to repulsion between anionic GO and anionic silk and consequent reduction in the adsorption. Moreover, with an increase in pH, the C/O ratio of GO (ratio of backbone carbon to oxygenated functional groups) increased and the available functional groups on GO edges decreased, leading to decreased adsorption. 26
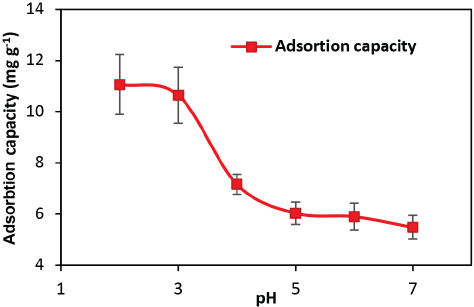
Effect of pH on GO adsorption.
Effect of GO Concentration on Adsorption
The effect of GO concentration on the adsorption was investigated with varying GO concentrations from 100 to 1000 mg/L. At pH 2, 10 min of dipping time at 40 °C and 70 °C were used. Fig. 5 represents the effect of GO concentration on the adsorption process. At the initial phase (for 100 to 350 mg/L), there was a sharp increase in adsorption capacity, which was due to the more available functional groups of silk as compared to available GO in the solution. Further increasing the GO concentration from 350 to 550 mg/L, the adsorption capacity tended to level of due to the levelling of the available functional groups of silk and GO. It seems that up to a 550 mg/L monolayer deposition of GO occurred on the silk due to the electrostatic interaction between the surface functionalities of silk and GO. Beyond 550 mg/L GO concentration, the GO attaches either with already adsorbed GO on silk surface or with free functional group that may still be present on silk, leading to multilayer deposition of GO on silk. This was further validated by the best fitting Freundlich isotherm. The adsorbed amount of GO per unit mass of silk (
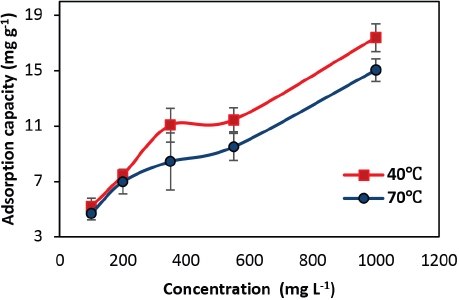
Effect of GO concentration on GO adsorption.
A similar trend is observed at 70 °C, where significant improvement in GO adsorption is observed with varying initial GO concentration from 100 to 1000 mg/L. However, the maximum adsorption capacity was lower as compared to that at 40 °C (15.04 mg/g). At both 40 °C and 70 °C, the variability between the data decreased as the concentration of GO increased.
Effect of Temperature on Adsorption
The adsorption of GO on silk was examined as a function of temperature, keeping the other process parameters constant, at a GO solution concentration of 550 mg/L, pH 2, and 5 min of dipping time. Bath temperature was varied from 30 °C to 70 °C and the data is represented in Fig. 6. On increasing the temperature from 30 °C to 70 °C, the adsorption capacity of GO on silk decreased significantly from 13.50 to 10.53 mg/g respectively. This may be due to a decrease in the number of active functional groups due to degradation or deoxygenation of GO with increased temperature. The change in GO solution color from brown to dark brown with the increase in temperature strengthened the above assumption. Drastic reduction in adsorption capacity was observed with increased temperature from 60 °C to 70 °C. The decrease in adsorption capacity with the rise in temperature indicates the exothermic nature of the process.
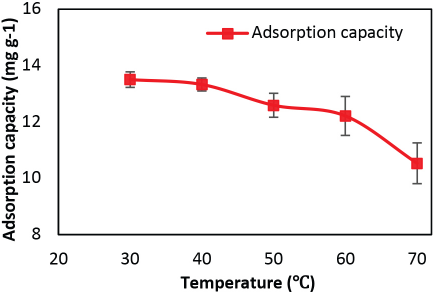
Effect of temperature on GO adsorption.
Effect of Dipping Time on Adsorption
The effect of dipping time on the adsorption process was determined by keeping other parameters (pH, initial concentration, and temperature) constant and varying the dipping time from 0.5 to 10 min. Silk (0.4 g) was dipped in 40 mL of 550 and 1000 mg/L GO solution at 40 °C. Fig. 7 indicates that at the initial phase (0.5 to 2 min), the adsorption was rapid as all the active sites of silk were free to interact with GO. The adsorption progressively decreased and reached equilibrium with time. The quantities adsorbed at equilibrium for initial GO concentrations of 550 and 1000 mg/L were 13.33 and 17.09 mg/g respectively (Fig. 7). Equilibrium was attained after 3 min of dipping time for both initial GO solution concentrations. At 1000 mg/L, equilibrium is attained after 2 min of dipping time. At the high concentration, large amounts of GO nano-sheets overcame resistance to the mass transfer of GO on the silk surface, resulting in higher probability of interaction between the active sites of GO and silk.
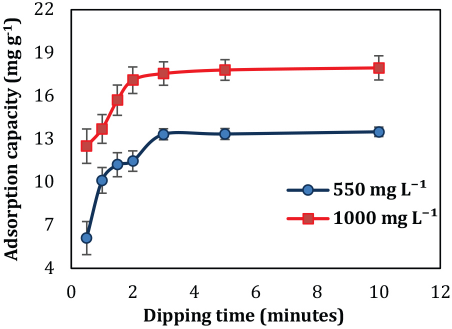
Effect of dipping time on GO adsorption.
Kinetic Studies
Pseudo-first order and pseudo-second order kinetic models were used to explain the adsorption kinetics of GO on silk. The kinetic measurements were conducted for varying GO concentration (550 and 1000 mg/L) at 40 °C and pH 2.
The pseudo-first order kinetic model was fitted through a straight line plot between
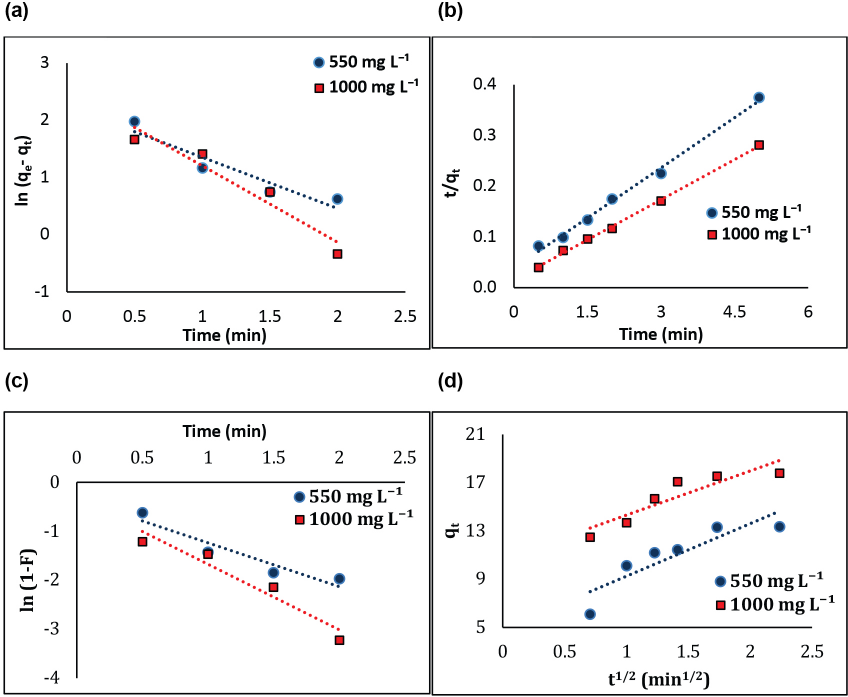
Kinetic plots at different GO concentrations. (a) Pseudo-first order, (b) pseudo-second order, (c) liquid film diffusion, and (d) intra-particle diffusion.
Kinetic Parameters for the Pseudo-First Order and Pseudo-Second-Order Model
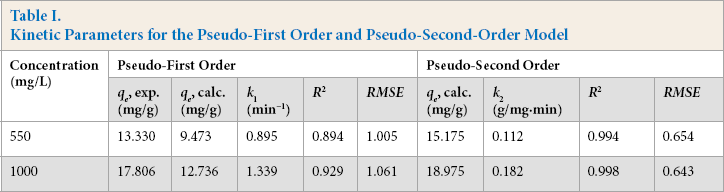
The higher value of the regression coefficient
The liquid film diffusion model was applied to understand the rate controlling stage of the adsorption of GO onto silk. A linear plot between
Kinetic Parameters for the Liquid Film Diffusion and Intra-Particle Diffusion Model
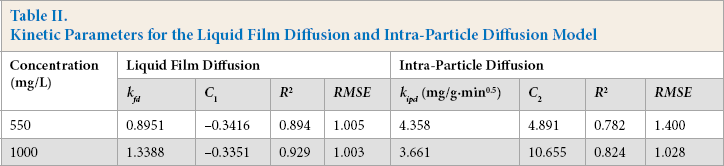
The Weber-Morris intra-particle diffusion model was used to determine whether the intra-particle diffusion was the sole rate limiting step. According to the model, if a plot of
Adsorption Mechanism of GO on Silk
Based on the kinetic studies, it can be assumed that the adsorption process is multi-linear and two or more steps control the process. At the initial phase, a sharp increase in the adsorption capacity occurred due to the external surface adsorption or the instantaneous adsorption stage, where a large number of GO nano-sheets interacted with silk surface functionalities via hydrogen bonding and electrostatic forces. At this point, monolayer deposition of GO nano-sheets occurred on the silk surface. The second linear phase is the gradual adsorption stage, where intra-particle diffusion may be the rate-limiting step, but the inadequate value of the regression coefficient at both concentrations suggest that intra-particle diffusion was not involved. This stage is governed by multilayer deposition, where GO nano-sheets attached either with already adsorbed GO nano-sheets (via π-π interactions or hydrophobic interactions) or the unoccupied functional groups of silk (Fig. 3d).
Isothermal Studies
A detailed understanding of the adsorption isotherm provides crucial information about the adsorption properties, feasibility of the adsorption process, and the maximum adsorption capacity. The isotherm studies were conducted at 40 °C and 70 °C by varying the initial GO concentration from 100 to 1000 mg/L. The experimental data was ft with the commonly used Langmuir, Freundlich, and D-R models for determining the adsorption system.
The linear plot of the Langmuir isotherm at different temperatures for adsorption of GO on the silk is presented in Fig. 9a. Langmuir parameters
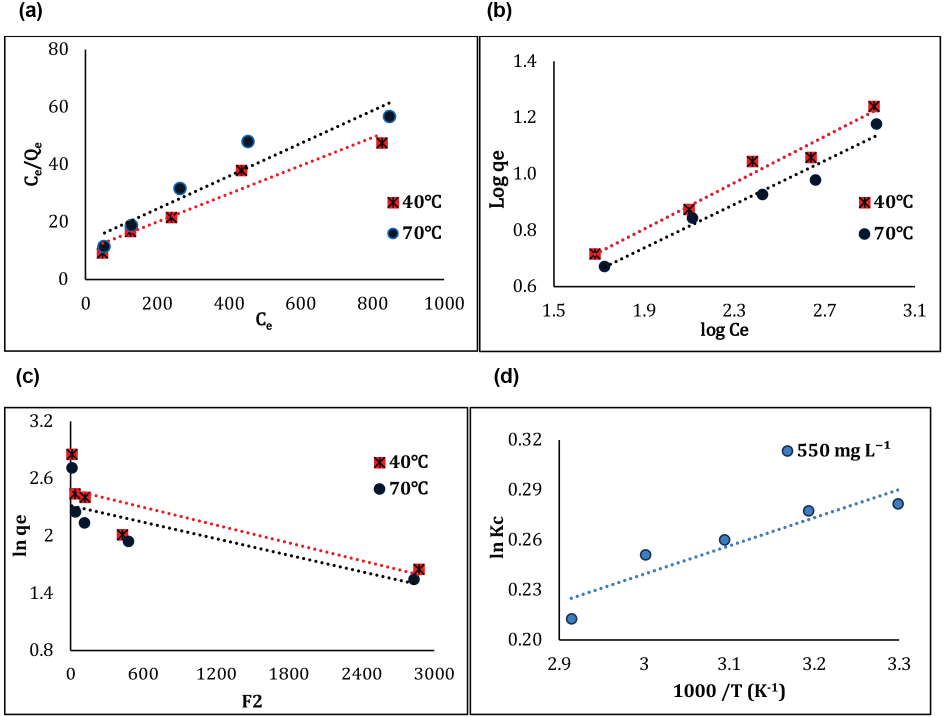
(a) Langmuir isotherm, (b) Freundlich isotherm, (c) D-R isotherm, and (d) Van't Hoff plots for GO adsorption.
Parameters of Langmuir, Freundlich and D-R Isotherms for GO Adsorption at 40 °C and 70 °C
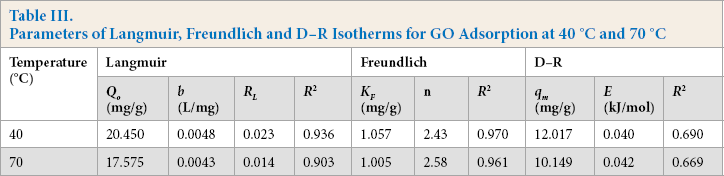
Freundlich parameters
The D-R isotherm plot for the adsorption of GO onto silk is shown in Fig. 9c. The D-R isotherm parameters and
Thermodynamics Studies
Adsorption thermodynamic enthalpy (Δ
Thermodynamic Parameters for the Adsorption of GO on Silk

Conclusions
This study revealed that silk can be used as an effective adsorbent media for application of graphene-based electro-conductive materials. The adsorption of GO from liquid solution is feasible and influenced by initial pH, initial concentration, and dipping temperature. Optimum adsorption was obtained at pH 2 and 40 °C for a GO dose of 1000 mg/L. Up to a 550 mg/L monolayer deposition of GO occurred on silk, due to the electrostatic interaction between the surface functionalities of silk and GO. Beyond 550 mg/L GO concentration, the GO attached on GO already adsorbed on the silk surface, or free functional groups still present on silk can lead to multilayer deposition of GO on silk. This is further validated by the best fitting of the Freundlich isotherm. The calculated maximum uptake capacity of silk as adsorbent for GO was 20.45 mg/g, which was appreciable in a single dipping cycle without surface modification. The pseudo-second order kinetic model explains the kinetics of GO adsorption onto silk. At the higher concentration, equilibrium was attained faster than at the lower concentration. Values of the free energy of adsorption (
Apart from biocompatibility and biodegradability, speedy adsorption of GO at low temperature in only single dipping cycle suggests that silk was a promising textile fiber for easy and economical preparation of graphene-based materials for various applications. Future research may envisage the statistical optimization of GO dipping variables with the response surface methodology to maximize the GO add on.
