Abstract
Clay-based adsorbent materials are widely used to remove heavy metal ions from water. In this study, we have fabricated a novel composite material, consisting of rectorite and sludge-derived biochar (REC/SDBC), and characterized its effectiveness in removing lead from water. Our SEM images show that the REC is evenly dispersed on the surface of the SDBC. The composites have a mesoporous structure, and its BET surface is (16.5 m2/g) comparable with that of SDBC but larger than that of REC. We found that the maximum adsorption capacity for Pb(II) was 26.89 mg g−1 for REC/SDBC composites with a REC:SDBC ratio of 3:1, which was achieved within 120 min. This is higher than the adsorption capacities of REC (19.81 mg g−1) and SDBC (23.15 mg g−1) alone. Furthermore, we observed that the adsorption capacity was greater at pH 5.0 compared to other pH values. Our results also indicate that the pseudo-second-order model and the Langmuir isotherm model provide good fits for the rates and isotherms of Pb(II) adsorption onto REC/SDBC composites. The adsorption kinetic constant and the Langmuir adsorption constant were calculated to be 0.0065 g mg−1 min−1 and 0.319 L mg−1, respectively. X-ray photoelectron spectroscopy analysis confirmed that the reaction mechanism is mainly dominated by chemisorption, involving cationic exchange, surface complexation, and surface precipitation. The high adsorption capacity, combined with the cost-effectiveness and environmental-friendly nature of REC/SDBC composites, make them promising candidates for removing heavy metallic elements from wastewater.
This is a visual representation of the abstract.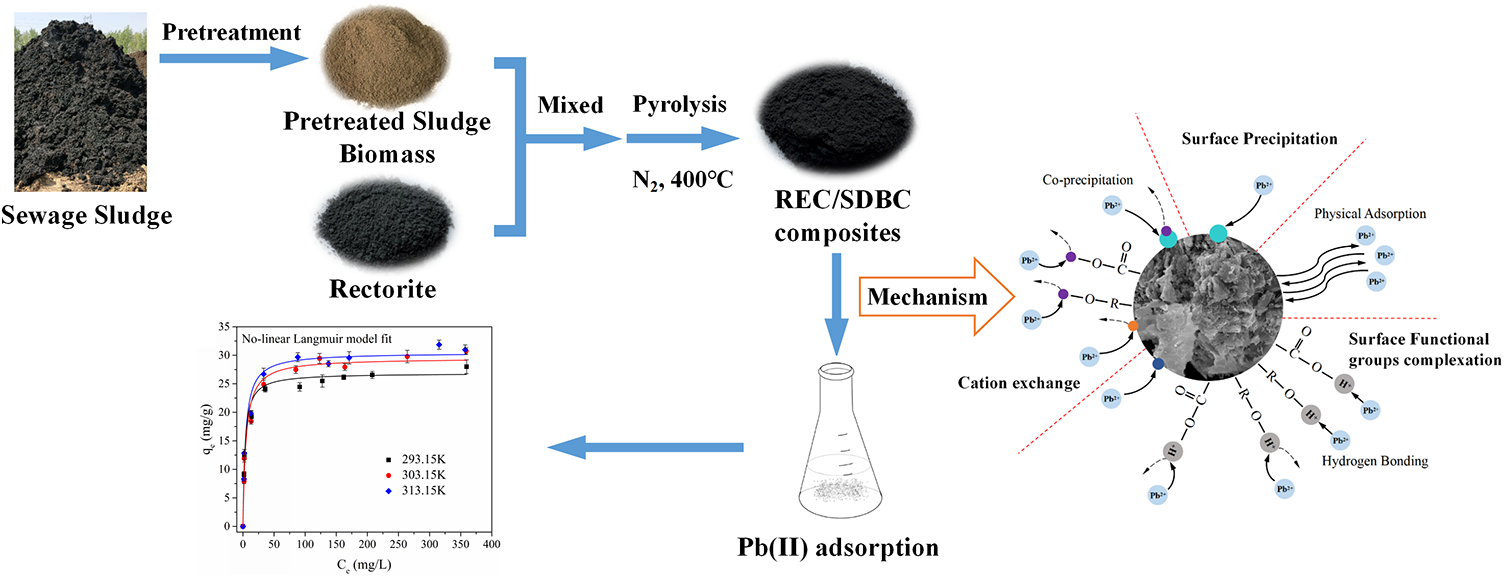
Introduction
A by-product of wastewater treatment plants is Sewage sludge (SS) and disposal of SS is a challenging issue because of its large quantities and considerable amount of hazardous contents including heavy metals, bacteria, fungi, and organic matter (Gao et al., 2020; Hoang et al., 2022; Xiao et al., 2018a). Traditional disposal methods, such as landfill, combustion, and anaerobic digestion, are expensive and may cause secondary pollution (Zhang et al., 2013). In order to promote resource utilization, the sludge needs to be treated in an environmentally sound manner (Fang et al., 2023). Pyrolysis is able to destroy or stabilize most microorganisms and toxic metals simultaneously (Liu et al., 2021), achieving the target of “reduce, reuse and safety” in SS disposal. Biochar, which refers to a carbonaceous solid byproduct of biomass pyrolyzed in the absence of oxygen, is an environmental-friendly substance with superior properties such as high surface-to-volume ratio and rich functional groups over the surface.
According to recent studies (Abdul et al., 2017; Edo et al., 2020; He et al., 2018; Xiao et al., 2018b; Zhou et al., 2017b), biochar is an ideal option for treating water in plants contaminated with organic or heavy metal pollutants. It has been shown to effectively remediate soil and has minimal impact on climate change through carbon sequestration, making it a more sustainable choice compared to traditional soil remediation methods (Cao et al., 2011; Woolf et al., 2010; Zhang et al., 2021). Additionally, biochar can be produced on a large scale using readily available materials and simple fabrication processes, from industrial facilities to individual farms (Damdib et al., 2023; Liu et al., 2015). As a result, the conversion of sludge to biochar through pyrolysis has become a popular research topic for sustainable sludge disposal. However, the relatively low functionality of biochar has limited its widespread application (Pan et al., 2021).
To address the issue of low efficiency in biochar, several engineering techniques have been utilized. These include surface activation through the use of chemical reagents such as alkali, acid, and oxidants (Song et al., 2018; Tang et al., 2018; Wang and Liu, 2018), as well as the loading of metal (metal oxides) and functional particles onto the surface of biochar to improve its adsorption capabilities (Abdul et al., 2017; Inyang et al., 2014; Kah et al., 2017; Liang et al., 2017). Rectorite (REC) is an interstratified clay that contains alternating layers of dioctahedral smectite and mica, but with a 1:1 expandable ratio (Zhang et al., 2010). Due to its ability to easily exchange organic and inorganic cations, such as Na+, K+, and Ca2+ (Wu et al., 2011), REC is highly effective in adsorbing heavy metals like lead (Pb). As a result, REC emerges as a promising and reliable functional particle for the modification of sludge-derived biochar (SDBC). Specifically, SDBC has been selected as the supporting structure for the loading of REC particles onto its surface, with the aim of enhancing its physiochemical properties and improving its adsorption efficiency for the removal of Pb(II).
To date, only a few studies were conducted regarding the adsorption of contaminants by biochar-clay composites (Chen et al., 2017; Li et al., 2017c; Wang et al., 2017a; Yao et al., 2014). Biochar-clay composites are the preferred adsorbents for removing pollutants due to their cost-effectiveness and high adsorption efficiency.
Heavy metals enter the environment mainly in water, soil, and the atmosphere and migrate between them, contaminating a wide range of areas. The most serious contamination of soil and water, among many heavy metals (Abdulkarim and Abu Al-Rub, 2004; Tang et al., 2020),is lead (Pb), which is highly toxic, abundant, nonbiodegradable, and can accumulate in nature. Consequently, heavy metal pollution, including that of lead, threatens ecosystem health (Bordoloi et al., 2017; Liu et al., 2019; Wang et al., 2017b; Wu et al., 2018). Even dilute solution of Pb is particularly hazardous (Liu et al., 2019; Renu et al., 2021). Consequently, it is essential to develop efficient methods to treat solutions and resolve heavy metal pollution. Considering the high toxicity of lead, the necessity of developing effective removal methods is especially more pronounced for Pb. In this regard, numerous methods have been proposed so far. For example, adsorbents derived from industrial or agricultural waste products have a simple and low-cost production process (Dias et al., 2017; Lyu et al., 2018; Wang et al., 2018; Wu et al., 2017; Zhou et al., 2017b). It has been reported that clay minerals possessed high adsorption capacity for Pb (Uddin, 2017). Nevertheless, these studies were mostly focused on montmorillonite-derived adsorbents. Rectorite has a similar structure with montmorillonite but is cheaper than montmorillonite. There is no comprehensive investigation into the synthesis of REC/SDBC composites for removing Pb(II) in aqueous solutions.
In this study, we aim to utilize the byproduct SS from wastewater treatment plants as a low-cost source of clay minerals for the development of cost-effective REC/SDBC composites for removing heavy metals from water. Our specific objective is to evaluate the physiochemical properties of these composites, with a focus on their performance and mechanistic pathways for adsorbing Pb(II) in aqueous solutions. To achieve this, we conducted a thorough characterization of the synthesized REC/SDBC composites; studied their kinetics, equilibriums, and thermodynamic parameters; and analyzed their chemical states before and after adsorption. Our goal is to investigate the potential of REC/SDBC composites for adsorbing Pb(II).
Materials and methods
Materials
In this article, REC was prepared by Hubei Mingliu Inc., China. The SS was gathered at the Hunan, China's Yuelu Wastewater Treatment Facility. The SS was dried at 60 °C for 48 h before being put through a 60-grit sieve (0.3 mm). Moreover, Pb(NO3)2 with an analytical quality was prepared from Sonopharm Chemical Reagent Co., China. A stock solution of Pb(II) (500 mg g−1, pH 2.0) was used in the experiment, the working solution with other Pb(II) concentrations was prepared by dilution. Every other chemical was of analytical quality and was used exactly as it was given (Electrical resistivity = 18.23 M Ωcm). Moreover, pure water was utilized to reduce aqueous solutions.
Preparation of REC/SDBC composites
Rectorite/SDBC composites were prepared using the method previously reported by Li et al. (2017c) and Chen et al. (2017). In summary, 2.4 g of REC powder was mixed with 60 mL of deionized (DI) water to create a stable REC suspension, which was then sonicated for 30 min. The REC suspension was then combined with 0.8 g of pretreated SS biomass material and stirred for 2 h. The resulting mixture was separated, and the feedstocks were dried at 60 °C. Next, the feedstocks were heated in a tube furnace at a rate of 5 °C/min for 2 h under a N2 environment. The resulting REC/SDBC composites had a REC to SS biomass mass ratio of 3:1 and were identified as such after being ground and passed through a 60-mesh sieve. Rectorite/SDBC composites with REC to SS biomass mass ratios of 5:1, 1:1, and 1:3 were also synthesized using the same procedure.
Characterizations
The morphology of the materials was assessed using FE-SEM (Zeiss SIGMA). Additionally, the elemental composition of REC/SDBC composites was determined using EDS (EDAX Genesis). Nitrogen adsorption at 77 K was measured using a BEL-SORP-Mini II analyzer. The surface area and adsorption–desorption isotherms of the specimens were calculated using the BET technique (Dias et al., 2018; Kah et al., 2017). The main elements (C, H, N, O) were quantified using a Vario EL cube elemental analyzer (Elemantar). The ash content was determined after 4 h of calcination at 600 °C, and the main metal contents of the samples were detected using XRF (Rigaku, ZSKprinus) (Li et al., 2018b). Heavy metal contents of the specimens were measured after acid digestion using ICP-MS (NexION 300X PerkinElmer). The crystallographic structure of the samples was characterized using XRD with Cu Kα radiation (D8 ADVANCE, Bruker AXS). The surface functional groups of the samples were identified using FT-IR (Nicolet 5700). The surface charge properties of the composites were measured using zeta potential measurements (Nano-ZS series Model ZEN3600, Malvern, UK). X-ray photoelectron spectroscopy (XPS) of the REC/SDBC composites before and after Pb(II) adsorption were obtained using a Thermo-Scientific Escalab 250Xi instrument with Al Kα irradiation of hv = 1486.6 eV as the X-ray source.
Batch adsorption experiments
Batch adsorption tests were conducted to investigate the performance and mechanism of Pb(II) adsorption using the prepared samples. The tests were carried out in 250 mL sealed conical flasks on a temperature-controlled rotary shaker at 150 rpm. A 100 mg of the samples (REC, SDBC, or REC/SDBC composites with different mass ratios of 5:1, 3:1, 1:1, and 1:3) were mixed with 100 mL of Pb(II) solution. The initial pH was adjusted to 5 ± 0.1 using 0.2 M HNO3 and NaOH solutions at a temperature of 20 ± 0.5 °C. To determine the kinetics parameters of adsorption in a 50 mg L−1 Pb(II) solution, 2 mL batches were sampled at different times (5–1440 min). The effect of pH on Pb(II) adsorption was studied at a pH range of 2–6, with the pH being adjusted using 0.2 M HNO3 and NaOH solutions. Adsorption isotherm tests were also conducted at 293.15, 303.15, and 313.15 K for 24 h using Pb(II) concentrations of 10–500 mg L−1. After the adsorption process was completed, 2 mL batches were sampled using a syringe and immediately filtered through a 0.22-μm membrane for Pb(II) concentration analysis.
A Purkinje General TAS-990 spectrometer (Beijing China) was used to measure the concentration of Pb(II) using the flame atomic absorption spectroscopy (AAS) method. To ensure accuracy, each test was repeated three times, and the average value was recorded to reduce relative errors to less than 5%. The adsorption capacity of Pb(II) is defined as equation (1), wherein 
Results
Characteristics of REC/SDBC composites
The physicochemical characteristics and elemental contents of SS, REC, SDBC, and REC/SDBC composites are presented in Tables 1 and 2, respectively. It can be observed that the yield value and ash content are high in SDBC and REC/SDBC composites, which can be attributed to the high inorganic mineral component present in these samples. This is due to the fact that REC is a type of clay mineral that remains stable at high temperatures, resulting in a higher yield value and ash content in REC/SDBC composites. The XRF results in Table 2 also indicate a significant amount of inorganic elements, such as silicon, aluminum, and Pb(II), in each sample. Additionally, it has been found that the amounts of C, H, N, and O in SDBC and the REC/SDBC composites are lower than those in the starting sludge (SS), yet the metal concentrations are comparatively higher. This phenomenon can be explained by the high-mineral biochar pyrolysis process, where the mineral enrichment outweighs the degree of carbonization (Li et al., 2018b; Park et al., 2018; Ren et al., 2018). The H/C ratio increases in SDBC and REC/SDBC composites, indicating a decrease in the degree of carbonization and the preservation of organic residues, such as polymeric CH2 and fatty acids (Chen et al., 2008; Chen et al., 2012).
Properties of REC and SDBC and REC/SDBC composites prepared in this work.

Elemental analysis of SS, REC, SDBC, and REC/SDBC composites.

The morphological characteristics of REC, SDBC, and REC/SDBC composites are shown in Figure 1 using FE-SEM. It can be observed that REC has a lamellar structure (Figure 1(a)). The majority of SDBC particles are amorphous and granular, with a rough and porous surface (Figure 1(b)). In Figure 1(c), it can be seen that REC in REC/SDBC composites is dispersed on the surface of SDBC, with partial exfoliation also present in the lamellar structure. The EDS spectra of REC/SDBC composites are shown in Figure 1(d). The main components of the composites are C, O, Si, and Al, which are consistent with the elemental contents listed in Table 2. The interaction between REC and SDBC, as well as the uniform dispersion of REC, contributes to the improved adsorption capacity of heavy metals by REC/SDBC composites.

SEM images of SDBC (a), REC (b), and REC/SDBC composites (c) and EDS spectra of REC/SDBC composites (d).
Figure 2 shows the adsorption–desorption isotherms for nitrogen molecules and the corresponding BJH pore size distribution of the REC, SDBC, and REC/SDBC composites. Table 1 displays the total pore volume, average pore width, and BET-specific surface area for each sample. According to the IUPAC classification, all N2 adsorption–desorption isotherms exhibit type IV behavior with an H3 hysteresis loop (Huang et al., 2018; Xu et al., 2018; Zhang et al., 2013), indicating the presence of a mesoporous structure in the samples. This is also reflected in the pore size distribution. The specific surface area and average pore diameter of the REC/SDBC composites are lower than those of SDBC due to the deposition of REC on the surface of SDBC.

BET N2 adsorption–desorption isotherms and the corresponding BJH pore size distribution plots (inset) of the obtained samples.
The XRD spectra of REC, SDBC, and REC/SDBC composites are shown in Figure 3. The peaks at 2θ = 3.95° and 7.99° are characteristic of REC (Chen et al., 2015), while the peak at 2θ = 20.04° is also attributed to REC (Yao et al., 2014). Sludge-derived biochar exhibits a characteristic peak at 2θ = 26.6°, indicating the presence of quartz as the main crystalline structure (Cao et al., 2011). A weak peak of Fe2O3 is also detected, suggesting a small amount of Fe2O3 in the SDBC samples (Li et al., 2018a). In the REC/SDBC composites, the peak at 2θ = 7.99° disappears and the intensity of the peak at 2θ = 3.95° decreases, indicating partial destruction of the ordered structure of REC (Wu et al., 2011). Additionally, the peak of quartz is observed but is weaker than in SDBC, indicating a decrease in quartz crystallinity in the composites.

X-ray diffraction(XRD) patterns of the REC, SDBC, and REC/SDBC composites. (a) 2θ ranging from 2–10°, (b) 2θ ranging from 10–90°.
In contrast to traditional biochar made from organic matter residues such as wheat straw (Yang et al., 2017), banana peel (Zhang et al., 2013), peanut shell (Bai et al., 2017), and pig manure (Dhyani et al., 2018), SDBC is a carbon-mineral composite that contains both mineral oxides and organic functional groups (Ren et al., 2018). While the surface functional groups of SDBC and REC/SDBC composites are similar, there are slight differences. Figure 4 shows the FT-IR spectra of the specimens, with bands at 3640 cm−1 and 3400 cm−1 present in all samples. These bands represent the stretching vibration of –OH in the organic components of SDBC (Park et al., 2018) and Si-OH in REC (Yao et al., 2014). In the composites, the peak at 3427 cm−1 is less pronounced compared to the sharp peak at 3390 cm−1 in REC. For SDBC, the peaks at 1614 cm−1, 1438 cm−1, 1029 cm−1, and 787 cm−1 can be attributed to C=C and C=O, CH2, C-OH, and –CH groups, respectively (Deng et al., 2017; Wang and Wang, 2018; Zhang et al., 2013). The observed peaks at 530 cm−1 and 468 cm−1 may be due to the presence of aluminum salts in SDBC (Deng et al., 2017). In REC, the peaks at 1024 cm−1 and 546 cm−1, 476 cm−1 are the result of stretching and bending vibrations of Si-OH, respectively. The peak at 918 cm−1 is likely due to the stretching vibration of Al-O-(OH)-Al (Wang et al., 2009). It should be noted that the peaks at 1629 cm−1 and 696 cm−1 in the FT-IR spectra are a combination of REC and SDBC. These results indicate that the REC/SDBC composites have been successfully fabricated and contain a variety of functional groups, which contribute to their effectiveness in removing Pb(II).

FT-IR spectra of the REC, SDBC, and REC/SDBC composites.
Adsorption kinetics
Before further examination of the adsorption performance of the as-prepared samples, SDBC, REC, and REC/SDBC composites with various mass ratios were selected to investigate the ability of Pb(II) removal. Figure 5 demonstrates the quantity of Pb(II) adsorbed by a series of specimens over time. The equilibrium adsorption capacities of Pb(II) are 24.06, 26.89, 25.95, and 24.53 mg g−1 for the REC/SDBC with mass ratios of 5:1, 3:1, 1:1, and 1:3, respectively. The Pb(II) adsorption capacities using REC/SDBC composites are invariably higher than that using SDBC (23.15 mg g−1) and REC alone (19.81 mg g−1). Besides, compared with the adsorption capacity of SDBC which increased continuously, REC/SDBC composites reached equilibrium mostly in 120 min. Especially when the mass ratio of REC/SDBC composites was 3:1, the adsorption capacity became as high as 26.89 mg g−1, which is close to the median adsorption capacities (36.23 mg g−1) of various clay minerals for Pb(II), ranging from 0.45 to 238.98 mg g−1 (Table S1, Supplementary Materials). Notably, the sorption capacity of the composite material is a little higher than the sum adsorption capacity of the individual components. For instance, the adsorption capacity for REC/SDBC composites with a mass ratio of 3:1 is 30% larger than the sum adsorption capacity of REC and SDBC with comparable mass in the composite (23.15 × 0.25 + 19.81 × 0.75). The preferential adsorption performance of Pb(II) by REC/SDBC composites originates from the synergism of REC and SDBC. Therefore, REC/SDBC composites with a mass ratio of 3:1 were selected for further investigation.

Adsorption kinetics of Pb(II) by REC, SDBC, and REC/SDBC composites with mass ratios of 5:1, 3:1, 1:1 and 1:3.
Pseudo-first-order (equation (2)) and pseudo-second-order (equation (3)) kinetic models were employed for describing the adsorption data in an attempt to analyze the adsorption process and mechanism of Pb(II) onto REC/SDBC composites (Wang et al., 2018; Wang and Wang, 2018; Xiao et al., 2018a):


Kinetic constants of REC, SDBC, and REC/SDBC composites for Pb(II) adsorption.

Effects of pH
The removal efficiency of Pb(II) by REC/SDBC composites is influenced by the surface charge and initial solution pH. This is because cation exchange, surface complexation, and surface precipitation are the primary mechanisms responsible for the adsorption of Pb(II) onto REC/SDBC composites. Figure 9(a) shows the impact of starting solution pH (between 2 and 6) on the removal of Pb(II) by REC/SDBC composites. It is evident that the adsorption behavior is pH-dependent. The uptake of Pb(II) increases continuously as the pH increases, with a relatively low adsorption capacity (less than 10 mg g−1) at pH 2–3. However, there is a sharp increase in adsorption capacity at pH 4, followed by a slower increase at pH above 5. This pH-dependent process can be attributed to the easier adsorption of more hydrolyzed Pb(II) compared to free Pb(II) (Li et al., 2017a; Ren et al., 2018) and the change in surface charge of the absorbent. Figure 9(b) shows that the surface of REC/SDBC composites is positively charged at pH < 3 but becomes more negatively charged as the pH increases. At pH 5, the decrease in zeta potential becomes much slower. This is mainly due to the deprotonation of surface functionalities reaching a maximum (Liang et al., 2017), which is consistent with the change in adsorption capacity. Therefore, as the pH increases, the electrical repulsion decreases, leading to an increase in adsorption capacity.
Adsorption isotherms
Figure 8 displays the findings of the investigation into the impact of the starting Pb(II) concentration on the adsorption performance of Pb(II) onto REC/SDBC composites. The composites’ adsorption capacity rises quickly at low concentrations before slowing down and progressively reaching equilibrium. Figure 8 depicts the simulation of the adsorption process using the Langmuir (equation (5)) and the Freundlich isotherm model (equation (6), with Table 4 listing the fitting parameters:


Kinetics of Pb(II) adsorption onto REC/SDBC composites.
Adsorption isotherm parameters of Pb(II) adsorption onto REC/SDBC composites.

The dimensionless separation parameter RL is defined based on the Langmuir isotherm model, and can be calculated as follows:
Adsorption kinetics
The adsorption capacities of Pb(II) by REC/SDBC composites were investigated at varying temperatures of 293.15, 303.15, and 313.15 K in order to determine the effect of temperature on adsorption performance. As shown in Figure 6, the adsorption capacity of Pb(II) increases with higher temperatures, indicating an endothermic process (Wu et al., 2018). The thermodynamic parameters, including enthalpy (ΔH0), Gibbs free energy (ΔG0), and entropy (ΔS0), can be calculated from the temperature-dependent adsorption isotherms using the following equations (equations (12) and (13)):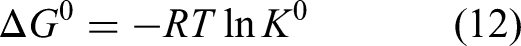

Thermodynamic parameters of Pb(II) adsorption onto REC/SDBC composites.

Adsorption mechanisms
The elemental chemical compositions and chemical states of the REC/SDBC composites were analyzed using XPS before and after adsorption to further support the potential mechanism of Pb(II) adsorption. Figure 7 displays the deconvolution of the high-resolution C 1 s, O 1 s, and Pb 4f peaks of the composites, as well as the XPS scan spectrum. The results of the elemental analysis are consistent with the major elements of the composites, namely C, O, Si, Al, and Na, as shown in Figure 7(a). The element Na, known for its strong cation exchange capabilities, could potentially substitute Pb(II), while surface functional groups containing C and O could also react with Pb(II) (Li et al., 2017b; Wang and Liu, 2018). Furthermore, the spectrum revealed two new Pb 4d and 4f peaks after Pb(II) adsorption, providing evidence for the effective adsorption of Pb(II) by the REC/SDBC composites (Figure 7(b)).
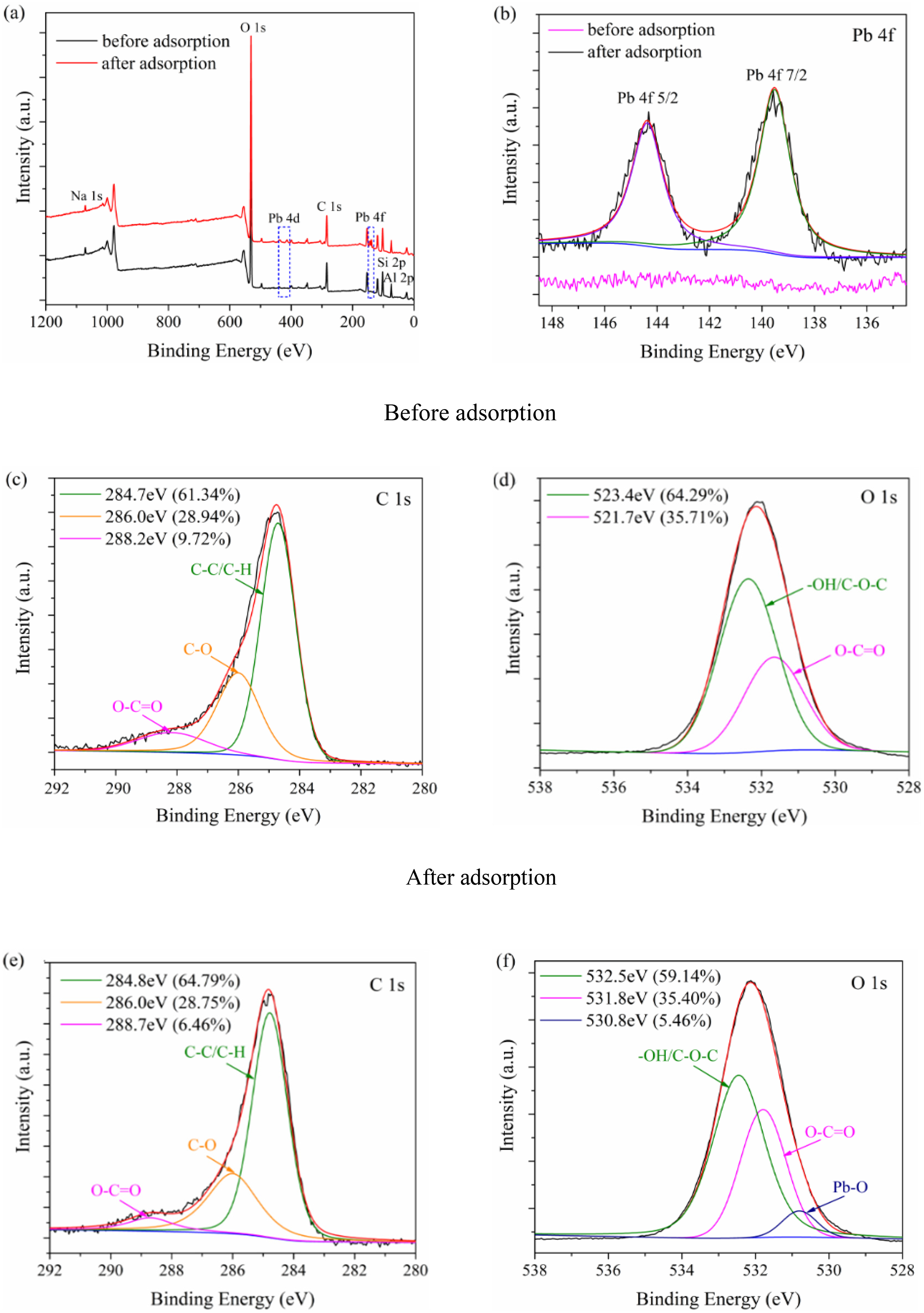
XPS survey spectra (a), high-resolution spectra of Pb 4f (b), C 1 s (c, e), O 1 s (d, f) of REC/SDBC composites before and after adsorption.

Adsorption isotherms for adsorption of Pb(II) by REC/SDBC composites.
The high-resolution XPS spectrum of the composites’ C 1 s peak was fitted into three peaks with binding energies at 284.7, 286.0, and 288.2 eV, corresponding to C-C, C-O, and O-C=O, respectively (Liu et al., 2011; Zhang et al., 2016; Zhou et al., 2017b). After Pb(II) adsorption, the binding energies of C-C and O-C=O shifted to higher values at 284.8 and 288.7 eV, and the peak area ratio of O-C=O decreased from 9.72% to 6.46%. These results confirm the involvement of carboxylic groups in the Pb(II) adsorption process and the formation of carboxyl-Pb complexes, consistent with previous studies (Zhang et al., 2016; Zhou et al., 2017b). In the high-resolution XPS spectrum of O 1 s, a pair of peaks at 531.7 and 532.4 eV, attributed to the oxygen groups of O-C=O and –OH, were observed (Wang and Liu, 2018). The –OH peak can originate from both organic functional groups (such as hydroxyl or ethers) and surface Si-OH groups. After adsorption, the binding energies of O-C=O and –OH shifted to 531.8 and 532.5 eV, and the peak area ratio of –OH decreased from 64.29% to 59.14% due to its involvement in Pb(II) adsorption. Additionally, a new peak at 530.8 eV was observed after adsorption, assigned to the O 1 s in the Pb-O bond (Wang et al., 2007). These results suggest the formation of Pb-O bonds between Pb(II) and the surface functionalities of the composites. The Pb 4f spectrum of the composites before and after the reaction is shown in Figure 9(b). Prior to adsorption, no peaks of the Pb element were evident in the XPS spectrum of the composites. However, after adsorption, a set of peaks with binding energies at 139.5 and 144.4 eV were observed, corresponding to Pb-O and Pb-O-C, consistent with the O 1 s XPS analysis.
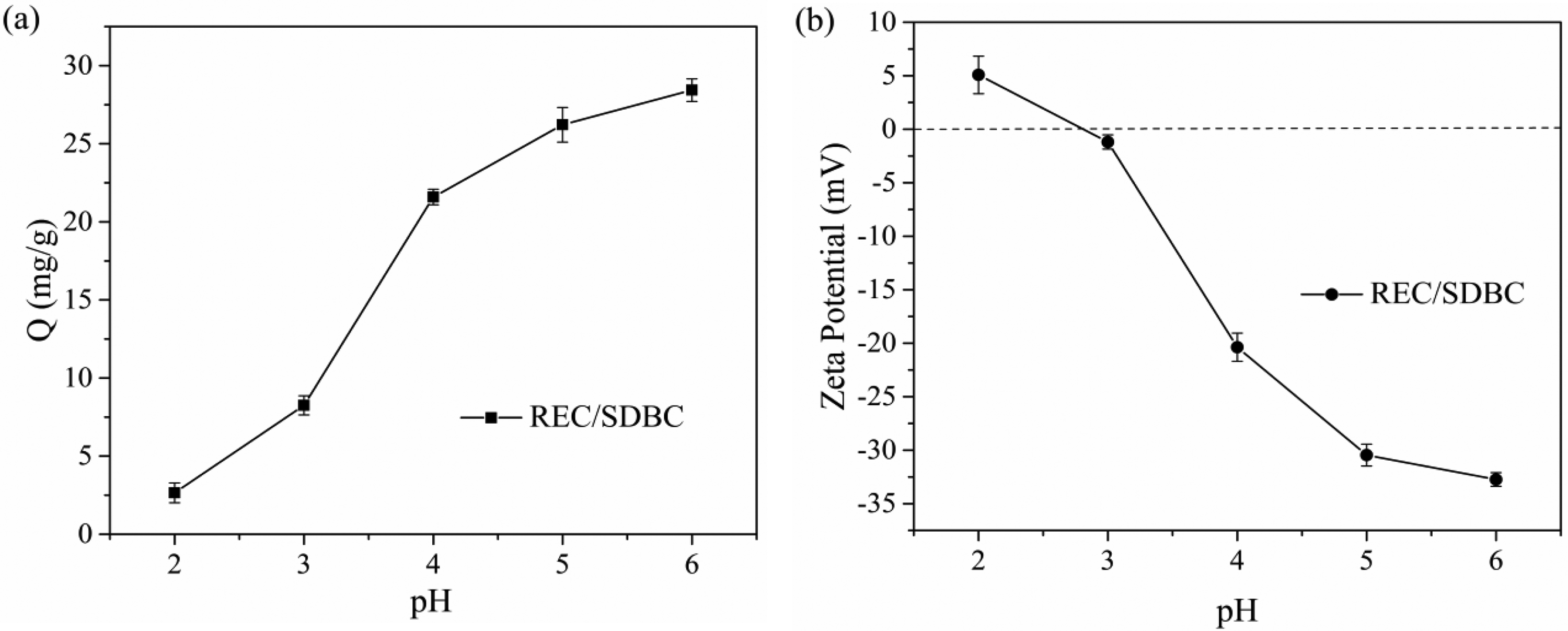
(a) Effects of initial pH on Pb(II) adsorption capacity; (b) Zeta pontentials of REC/SDBC composites at different pH values.
Overall, the XPS analysis shows that Pb(II) adsorption onto the REC/SDBC composites is a combination process involving cation exchange, surface complexation (Figure 10). Previous studies have demonstrated that Pb(II) adsorption by REC primarily involves electrostatic cation exchange and the formation of inner-sphere complexes at the edges of REC particles, facilitated by ≡Si-O- and ≡Al-O- groups (Celis et al., 2000).

Schematic for the proposed mechanisms of Pb(II) adsorption onto REC/SDBC composites.
Chemisorption, which includes surface complexation with free hydroxyl and carboxyl functional groups, surface precipitation, and cation exchange with interlayer Na+ in the REC phase and Ca2+, Mg2+ in the SDBC phase, also controls the adsorption of Pb(II) onto REC/SDBC composites. Nevertheless, the individual contribution to the adsorption was not quantified in the current study.
Conclusions
Novel and cost-effective REC/SDBC composites were successfully synthesized and thoroughly characterized. These composites exhibit a mesoporous structure, with a BET surface area of 16.5 m²/g. The REC component possesses a layered structure, while most SDBC particles are amorphous and granular. In the REC/SDBC composites, REC is primarily dispersed on the surface of the SDBC particles. The composites demonstrated a significantly higher adsorption capacity compared to REC alone and reached equilibrium more rapidly than SDBC. The adsorption process was highly sensitive to pH, with equilibrium achieved within 120 min. Kinetic data were best described by the pseudo-second-order model. The adsorption mechanism for Pb(II) onto the REC/SDBC composites involves cation exchange, surface complexation, and surface precipitation. Additionally, the minimal changes observed in the structure of the REC/SDBC composites indicate their stability. These findings suggest that REC/SDBC composites are promising candidates for effectively treating Pb(II)-contaminated wastewater.
Supplemental Material
sj-doc-1-adt-10.1177_02636174241287698 - Supplemental material for Characterization and adsorption properties of rectorite/sludge-derived biochar composites for Pb(II) removal
Supplemental material, sj-doc-1-adt-10.1177_02636174241287698 for Characterization and adsorption properties of rectorite/sludge-derived biochar composites for Pb(II) removal by Huanhuan Wang, Jing Wang, Zi Wang, Qing Ye and Junye Zhang in Adsorption Science & Technology
Footnotes
Author contributions
Conceptualization, H.W. and J.Z.; methodology, H.W.; software, J.Z., Z.W.; validation, J.W. and Z.W.; formal analysis, J.Z.; data curation, J.Z.; writing—original draft preparation, H.W.; writing—review and editing, J.W. and Q.Y.; project administration, H.W. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Scientific Research Foundation for High-level Talents of West Anhui University (WGKQ2021014) and College Students’ Innovative Entrepreneurial Training Plan Program “Preparation and performance study of rectorite-based phosphorus locking agents” (Grant No. S202110376103 and No. 202310376032).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
