Abstract
Chromium (VI) contamination in water presents significant environmental and public health challenges, demanding efficient and sustainable remediation strategies. This study explores the use of biochar (BC) derived from the invasive plant Mimosa pigra, modified with hydroxyapatite (HAp), to form a composite (BC@HAp) for effective CrVI removal. HAp is known for its high adsorption capacity and biocompatibility, and its incorporation into BC enhances heavy metal removal through synergistic effects. BC and BC@HAp were synthesized via the sol–gel method and tested under various conditions including pH, adsorbent dosage, CrVI concentration, contact time, and temperature. Structural and morphological analyses confirmed improved surface characteristics of BC@HAp. The composite exhibited a significantly higher CrVI adsorption capacity (67.68 mg/g) compared to unmodified BC (39.95 mg/g), attributed to increased surface area and new functional moieties. The adsorption mechanism was facilitated by electrostatic attraction between CrVI anions (HCrO4−, Cr2O72−) and positively charged Ca2+ sites, ion exchange with surface carbonate and phosphate groups, and surface complexation involving hydroxyl (–OH) and carboxyl (–COOH) groups. Adsorption followed the Freundlich and Temkin isotherms, indicating heterogeneous surface interactions, and was best described by the Elovich kinetic model. Thermodynamic parameters revealed the process to be spontaneous and endothermic, favoring higher temperatures. The enhanced performance of BC@HAp demonstrates its potential as a cost-effective and eco-friendly solution for both CrVI remediation and invasive species utilization.
Introduction
Heavy metals, particularly chromium (Cr), are harmful to human health and the environment. The rapid development of industrial technology, such as metal plating, tanning, textiles, and nuclear power plants, has worsened heavy metal pollution, threatening ecological balance and human well-being (Kim and Kang, 2016). Chromium exists in two main oxidation states: CrIII and CrVI, with CrVI being much more toxic—100 times more so than CrIII (Barrera-Díaz et al., 2012). Hexavalent chromium (CrVI) therefore is widely recognized as one of the most hazardous water pollutants due to its mutagenic and carcinogenic properties (Chen et al., 2021a; Zhang et al., 2021) (Squadrone et al., 2013). It also poses risks to human and animal health through the food chain and is difficult to degrade, causing long-lasting environmental damage (Roberts and Oris, 2004). Regulatory agencies set stringent limits for its discharge and presence in drinking water, with the World Health Organization (WHO) guideline for total chromium at 50 µg/L and the US EPA standard at 0.1 mg/L (US EPA, 2022; WHO, 2017). CrVI is commonly released from industrial activities such as electroplating, tanning, textile dyeing, and metal finishing, where inadequate effluent treatment leads to its accumulation in natural waters. Elevated chromium contamination has been reported worldwide in soils, sediments, wastewater, and groundwater near industrial zones. For example, sediments in northern Kaohsiung Harbor, Taiwan, contained up to 361.9 mg/kg CrVI due to tannery effluents (Dong et al., 2013), while soils in Chinese industrial regions reached 37,967 mg/kg CrVI, with over 4% of sites exceeding the screening value of 2500 mg/kg (Li et al., 2023a, 2023b). In South Asia, chromite mine soils in Odisha, India, averaged 11,170 mg/kg CrVI (Mohanty et al., 2011), and tannery wastewater in Kasur, Pakistan, contained 2.8–125 mg/L CrVI, far above discharge standards (Younas et al., 2023). Moreover, CrVI-contaminated groundwater in Kanpur, India, has been directly linked to gastrointestinal, dermatological, and hematological disorders in exposed populations (Sharma et al., 2012). These findings highlight the global scale of chromium pollution and the urgent need for low-cost, effective remediation materials.
Conventional methods for removing CrVI from aqueous solutions, such as chemical precipitation (Minas et al., 2017), ion exchange (Dharnaik and Ghosh, 2014), membrane filtration (Kozlowski and Walkowiak, 2002), and electrochemical processes (Jin et al., 2016) have been widely studied and applied. While effective, these techniques often face challenges such as high operational costs, complex procedures, and the production of secondary waste (Mukherjee et al., 2013). For example, chemical precipitation involves adding chemicals to water to facilitate the removal of pollutants, but this often results in the formation of sludge, which is a byproduct that needs to be treated or disposed of separately. The disposal of sludge can be costly and requires additional resources for safe handling, increasing the overall operational cost (Wang et al., 2005). Ion exchange is another effective method, but it relies on specialized resins that can be expensive to purchase and maintain, making the process costly despite its high efficiency (Bhandari et al., 2016). Membrane filtration methods, such as reverse osmosis, can remove contaminants but are prone to membrane fouling, where pollutants accumulate on the membrane surface, reducing its effectiveness and requiring regular cleaning or replacement, further adding to maintenance costs (Men et al., 2023). Electrochemical processes, while effective in certain applications, often consume a significant amount of energy, which can be both costly and environmentally unsustainable over time, limiting their practical application (Nath, 2024).
In contrast, adsorption has emerged as a simpler, more cost-effective, and environmentally friendly method for removing CrVI from industrial wastewater (Islam et al., 2023). This technique works by using adsorbent materials that have the ability to attract and bind pollutants to their surfaces. Adsorption is a highly effective and versatile method for removing CrVI, particularly when integrated with appropriate regeneration techniques (Dakiky et al., 2002). By enabling the regeneration of the adsorbent material, this method addresses the challenges associated with sludge disposal, making the entire system more cost-effective and environmentally sustainable (Baskar et al., 2022). Regeneration also allows for the reuse of the adsorbent, reducing the need for continuous material replacement. This becomes especially advantageous when low-cost adsorbents are used, making the process more economically viable, particularly in resource-limited settings (Gupta et al., 2009).
Various studies have highlighted the effectiveness of biochar (BC) as an adsorbent for chromium removal from soil and water, particularly CrVI (Sun et al., 2022; Xia et al., 2020). BC is typically produced through the pyrolysis of biomass, such as agricultural waste, forestry residues, or other organic materials. This process not only provides a sustainable method for waste management but also results in a high-surface-area material capable of adsorbing heavy metals like CrVI (Sinha et al., 2022). In recent year, there has been a growing shift towards the conversion of invasive plants into BC for pollutant decontamination. The potential of invasive plants for BC production and their effectiveness in water treatment has been reviewed in recent studies (Feng et al., 2021; Nguyen et al., 2022). Several studies have highlighted the potential of BC derived from invasive plants for removing CrVI from water. For example, BC produced from ragweed and horseweed at 450 °C exhibited high adsorption capacities, reaching 139 mg/g for CdII and 358.7 mg/g for PbII (Lian et al., 2020). In another study, Mg-Al/LDH-decorated BC derived from
Pristine BC, however, has limitations as an adsorbent due to its negatively charged surface, low surface area, and lack of acidic functional groups, which hinder its ability to remove anionic pollutants from wastewater effectively (Kavitha et al., 2018). Engineered BCs modified with materials like hydroxyapatite (HAp) can indeed overcome these limitations. HAp, a naturally occurring mineral form of calcium apatite, has a high capacity for adsorbing heavy metals, phosphates, and other contaminants (Balasooriya et al., 2022; Ibrahim et al., 2020). By incorporating HAp into BC, the resulting product can exhibit enhanced adsorption properties, increased surface area, and improved stability (Ahmed et al., 2021; Jung et al., 2019). For instance, tobacco stalk-based BC (TSB) modified with HAp at pyrolysis temperatures of 350 °C and 550 °C exhibited enhanced surface area and changes in active sites (P–O and carboxyl groups). The maximum Cd(II) adsorption capacities of HAp–TSB350 and HAp–TSB550 were 13.17 and 14.50 mg/g, respectively, which were 2.67 and 9.24 times higher than those of TSB350 and TSB550 (Li et al., 2024). Similarly, in a separate study, the sorption capacity of rice straw-derived BC increased from 63.03 to 335.88 mg/g after modification with HAp, marking a fivefold enhancement (Ahmed et al., 2022). This improvement suggests that HAp introduces additional active sites, enhancing the interaction between BC and heavy metals like PbII. Together, these results demonstrate that the BC@HAp composite is a more efficient and cost-effective solution for water purification and heavy metal removal.
Although BC has been widely explored as a low-cost adsorbent for wastewater treatment, studies on its modification with HAp for CrVI removal remain comparatively limited. Previous research has shown that HAp-modified BCs and composites exhibit strong adsorption performance for heavy metals, oxyanions, and dyes, often attributed to mechanisms such as ion exchange with Ca2+, electrostatic attraction, and surface complexation (Ahmed et al., 2022; Li et al., 2024; Shan et al., 2023; Zhu et al., 2022; Zou et al., 2022; Zou et al., 2023). However, many of these studies have demonstrated maximum efficiency under strongly acidic conditions (pH 2–3), which constrains their practical application to real wastewater systems. At the same time, CrVI contamination remains a pressing environmental and public health issue, with field studies reporting concentrations far exceeding safe thresholds in sediments, soils, and wastewaters near industrial zones (Dong et al., 2013; Li et al., 2023a, 2023b; Mohanty et al., 2011; Sharma et al., 2012; Younas et al., 2023).
To address these challenges, the present study utilizes
Materials and methods
Chemical and stock solution preparation
The chemicals used in the experiments were of analytical grade. Chromium (Cr) standard solution, potassium dichromate (K2Cr2O7), hydrochloric acid (HCl), sodium hydroxide (NaOH), calcium chloride (CaCl2), and diammonium phosphate ((NH4)2HPO4) were obtained from Merck. A 1000 ppm CrVI stock solution was prepared by dissolving 0.283 g of K2Cr2O7 (dried at 100 °C for 1 h) in 100 mL of water in a volumetric flask.
Adsorbent material synthesis
Synthesis of Bc material
Preparation of raw material &
Surface cleaning of
Synthesis of BC@HAp material
The dry biomass from the

The synthesis process of BC@HAp from
Adsorbent characterization
The microstructure of BC and BC@HAp was analyzed using Scanning Electron Microscopy (SEM) (JSM-7100, JOEL Ltd, Tokyo, Japan), which provided detailed images of their surface morphology. The elemental composition of BC and BC@HAp surfaces was determined through Energy-dispersive X-ray (EDX) spectroscopy (Hitachi, Japan), offering insights into the distribution of key elements. The BET-specific surface area was determined from low-temperature nitrogen adsorption isotherms, measured with a Nova Station A (Quantachrome Instruments, version 11.0, Miami, FL, USA), providing valuable information about the porosity of BC and BC@HAp. Additionally, Fourier-Transform Infrared (FTIR) spectroscopy (FTIR-PerkinElmer Spectrum 10.5.2, Buckinghamshire, UK) was employed to identify the functional groups present on BC and BC@HAp surfaces, helping to understand their adsorption behavior.
Batch adsorption studies
A stock solution containing 1000 mg/L of CrVI ions was prepared and diluted with double-distilled water to obtain the desired concentrations for the experiments. A known amount (0.01 g) of either BC or BC@HAp was added to 50 mL of CrVI ion solution, with concentrations ranging from 5 to 80 mg/L, in a 125 mL Erlenmeyer flask. To adjust the initial pH within a range of 2–10, 0.1 M HCl or 0.1 M NaOH was used. The mixture was then agitated at a constant speed of 60 rpm at room temperature (298 K) for a specified duration to ensure adequate interaction. Following agitation, the mixture was filtered using Whatman No. 1 filter paper to separate the adsorbent from the CrVI solution. The CrVI ion concentrations in the solution were measured using flame atomic absorption spectroscopy with a Perkin Elmer PinAAcle 900T instrument, ensuring precise and reliable determination of the remaining CrVI concentration after adsorption.
The adsorption capacity (qₑ) and percentage removal (H) were calculated using the following equations:

The adsorption equilibrium data for Cr on BC or BC@HAp were analyzed using the linear forms of three two-parameter kinetic and isotherm models. These models were applied to the experimental values of
Adsorption isotherm and kinetic models.
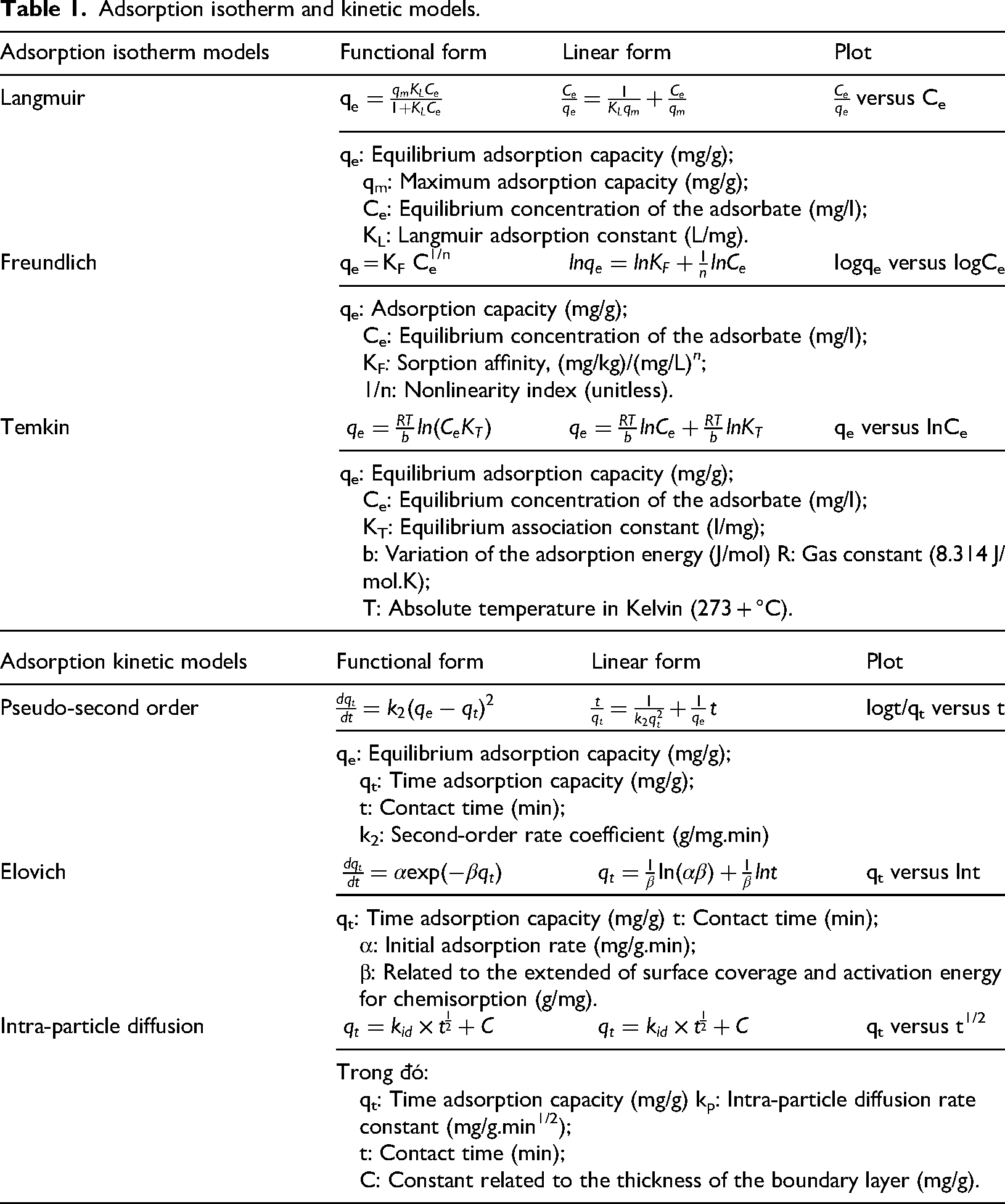
Error analysis
Two error functions, Chi-square (χ²) and the coefficient of determination (R²), were used to identify the best-fitting isotherm and kinetic models for the experimental data. The parameters were obtained by minimizing the respective error functions with the Solver add-in in Excel. The formulas for calculating these error functions are provided in Equations (3) and (4), as referenced from Tchuifon et al. (2018).

Results and discussion
Characterization
BET analysis
The textural properties of BC@HAp and BC are compared in terms of specific surface area, pore volume, pore diameter, and pore type. BC@HAp demonstrates a significantly higher specific surface area (125.13 m2/g) and pore volume (0.096 cm3/g) compared to BC (80.14 m2/g and 0.067 cm3/g, respectively), indicating that the HAp modification enhances the material's adsorptive potential. The pore diameter of BC@HAp (2.32 nm) is slightly larger than BC (2.02 nm), suggesting a more open structure that could better accommodate adsorbates. Both materials are predominantly mesoporous, with BC@HAp having a marginally higher mesopore volume (96.049 vs 95.708 cm³/g), indicating that the modification does not significantly alter the mesopore structure. BC@HAp shows a slightly lower micropore volume (8.57 × 10−27 vs 5.61 × 10−26 cm3/g) and a slightly increased volume of macropores (4.29 vs 3.95 cm3/g), suggesting that the modification creates larger, more uniform pores while reducing smaller pores. This finding aligns with previous studies, for instance, Ahmed et al. (2022) demonstrated that HAp modification increased the specific surface area of rice straw-derived BC and enlarged the average pore size of BC@HAp (Ahmed et al., 2022). Similarly, TSB pyrolyzed at 350 °C and 550 °C had surface areas of 2.52 and 3.63 m2/g, respectively, which increased to 14.07 and 18.36 m2/g after HAp modification, likely due to the dispersion of HAp on TSB surfaces, enhancing its adsorption capacity (Li et al., 2024) (Table 2).
Surface properties and pore type data for BC and BC modified with HAp.

FT-IR analysis
The FT-IR analysis of BC and BC@HAp, shown in Figure 2, reveals key insights into their functional groups and characteristic bond vibrations. In the FT-IR spectrum of BC, a prominent peak at 3423 cm−1 indicates the presence of hydroxyl groups on the BC surface, which is typical for BC (Melanie et al., 2015). Moreover, the FT-IR spectrum also shows peaks at 2836 and 2888 cm−1, which are strongly associated with the asymmetric and symmetric stretching of C-H bonds in alkyl groups (Januszewicz et al., 2023). The peaks observed at 1611 and 1603 cm−1 are indicative of the stretching vibrations of the C = C bonds in aromatic compounds (Ray et al., 2020). Broad bands between 1050 and 1275 cm−1 are assigned to the stretching vibrations of C-O-C bonds, with peaks observed at 1108, 1122, and 1082 cm−1 (Januszewicz et al., 2023). These FT-IR results for BC are consistent with previous studies analyzing BC derived from

The FTIR analysis of BC and BC@HAp.
Meanwhile, the FT-IR spectrum of the synthesized BC@HAp shows the presence of three key functional groups: phosphate (P-O), hydroxyl (OH-), and carbonate (CO32−) groups, which are commonly found in the FT-IR spectra of HAp (Fathi and Hanifi, 2007; Li et al., 2018). Specifically, the peaks at 561 and 961 cm−1 are attributed to the P-O bonds, which are characteristic of phosphate groups in HAp structures (Mehta and George, 2013). Furthermore, the peak at 1042 cm−1 corresponds to the symmetric vibrations within the polyphosphate chain, including the P-O-P and P-O bonds, which are typically found in phosphate esters (Puziy et al., 2006). For the carbonate group, BC exhibits an absorption band in the 1413–1467 cm−1 range, while BC@HAp displays a more pronounced peak at 876 cm−1. This shift and the enhanced intensity suggest a stronger presence of the carbonate group in the modified material, indicating that HAp modification has contributed to the increased incorporation of carbonate species into the BC structure (Li et al., 2018). These observations align with previous research, such as the study by Ahmed et al. (2021), where BC@HAp modified from rice straw using the hydrothermal method showed similar functional group vibrations, including the presence of phosphate, hydroxyl, and carbonate groups (Ahmed et al., 2022).
SEM-EDX analysis
SEM-EDX analysis of BC and BC@HAp (Figure 3) reveals distinct differences in surface morphology and elemental composition. BC has a rough, irregular surface with less developed features, while BC@HAp displays a refined surface with HAp particles forming crystalline clusters, enhancing surface area and providing more active sites. EDX analysis shows that BC is primarily composed of carbon (82.90%) and oxygen (13.60%), reflecting its organic nature. For BC@HAp, carbon content decreases to 46.76%, with oxygen and calcium increasing to 30.31% and 2.10%, respectively, indicating successful HAp incorporation. These changes confirm the synthesis of BC@HAp and suggest an improved surface for adsorption, where calcium and phosphorus play key roles in pollutant binding.
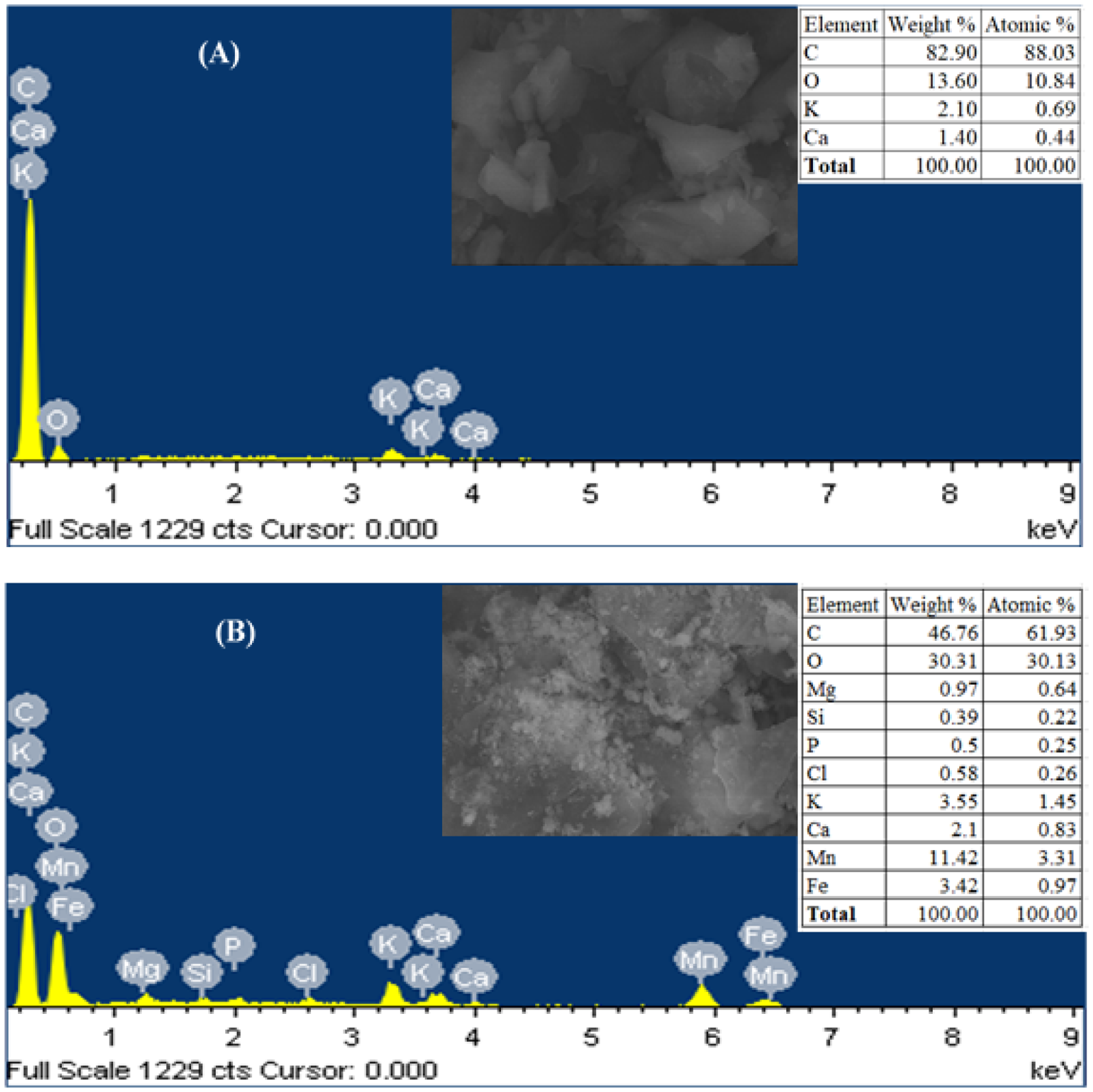
The SEM/EDX analysis of BC and BC@HAp.
While solid-state NMR (SS-NMR) would provide direct structural validation of functional group changes, this technique was not available in the present study. Instead, the combined results of BET, FT-IR, and SEM-EDX offered strong complementary evidence for the successful incorporation of HAp and the associated increase in functional groups. BET analysis revealed a higher surface area (125.13 vs 80.14 m²/g) and pore volume (0.096 vs 0.067 cm³/g), FT-IR confirmed the appearance of new phosphate, hydroxyl, and carbonate groups, and SEM-EDX showed crystalline HAp clusters with elevated oxygen and calcium content. Collectively, these findings provide reliable validation of the enhanced surface functionality and adsorption capacity of BC@HAp, effectively compensating for the absence of SS-NMR.
Point of zero charge
The point of zero charge (pHpzc) represents the pH at which the adsorbent surface has a net neutral charge due to the balance of positive and negative surface charges. When the solution pH is below the pHpzc, the surface is positively charged, favoring the adsorption of anionic species such as CrVI oxyanions, while reducing the uptake of cations. Conversely, when the solution pH exceeds the pHpzc, the surface becomes negatively charged, thereby enhancing cation adsorption (Bagheri et al., 2020).
The pHpzc of BC and BC@HAp was evaluated within a pH range of 2–12 (Figure 4). The pristine BC exhibited a pHpzc of 9.95, whereas the value decreased to 8.09 after HAp modification. This downward shift indicates that HAp loading introduced additional negatively charged functional groups onto the BC surface. Similar observations were reported by Li et al. (2018), where HAp-modified reed BC exhibited a lower pHpzc (7.43) compared to its pristine counterpart (8.5) (Peng et al., 2016). The reduction in pHpzc can be attributed to the incorporation of CO32−, PO43−, and OH− groups from HAp, which increase the density of negative charges on the BC surface.

pHpzc of BC@HAp and BC.
These findings suggest that BC@HAp offers more favorable surface chemistry for interaction with Cr(VI) under near-neutral conditions, supporting its enhanced adsorption performance compared to pristine BC.
Adsorption tests
Effect of pH
pH significantly influences the adsorption of CrVI by BC and BC@HAp by altering both the chemical speciation of CrVI and the surface charge of the materials (Figure 5(A)). In acidic conditions (pH 2–4), HCrO4⁻ is the dominant CrVI species, while as the pH increases (around pH 6–9), Cr2O72− and CrO42− become more prevalent, with CrO42− being significant only at pH ≥ 9 (García-Sosa and Olguín, 2015).

Effect of pH (A), adsorbent mass (B), concentration (C), and time (D) on percent removal (%) and adsorption capacity (qe, mg/g) of CrVI by BC and BC@HAp.
The surface charge of BC and BC@HAp is strongly dependent on pH relative to their point of zero charge (pHpzc). For pristine BC, the pHpzc was determined as 9.95, meaning that the surface is positively charged when pH < 9.95 and negatively charged at pH > 9.95. For BC@HAp, the pHpzc shifted downward to 8.09, indicating that its surface acquires a negative charge at a lower pH compared to BC. This shift reflects the successful incorporation of functional groups such as CO32−, PO43−, and OH− from HAp, which increase the density of negative charges on the adsorbent surface.
At acidic pH values, protonation of surface functional groups occurs. On BC, –COOH groups can form –COOH2+, and –OH groups can protonate to –OH2+, thereby reducing the surface negative charge and limiting electrostatic attraction with Cr(VI) anions. Similarly, BC@HAp also experiences protonation under acidic conditions, as shown in the following equilibria:

However, because BC@HAp has a lower pHpzc (8.09), its surface becomes negatively charged at near-neutral pH (∼6), while pristine BC remains close to neutral or slightly positive under the same conditions. This explains why BC@HAp exhibits superior adsorption of CrVI at pH 6.
In addition to electrostatic interactions, ion exchange between carbonate groups (CO32−) on BC@HAp and Cr(VI) oxyanions plays an important role in the adsorption mechanism, as illustrated below: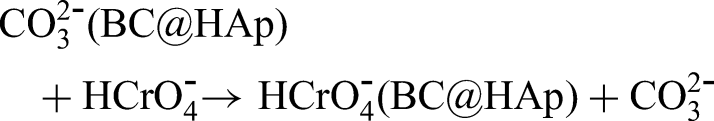

Therefore, the optimal pH for Cr(VI) removal by BC@HAp is approximately pH 6, where its surface is already negatively charged (pHpzc = 8.09, lower than pristine BC at 9.95) and the dominant CrVI species (Cr2O72− and HCrO4−) can effectively interact with the adsorbent through both electrostatic attraction and CO32−/CrVI ion exchange. This observation is consistent with Jung et al. (2019), who reported maximum CuII removal at near-neutral pH using HAp-modified BC (Jung et al., 2019). The ability of BC@HAp to achieve high adsorption efficiency under environmentally relevant near-neutral conditions highlights a significant advantage over many earlier studies, which demonstrated high capacities only under strongly acidic conditions.
Effect of adsorbent dosage
Figure 5(B) shows that as the material mass increases, the amount of CrVI adsorbed per unit mass decreases, while the removal efficiency increases. For BC, the adsorption capacity dropped from 31.94 mg/g at a 0.01 g dose to 4.61 mg/g at a 0.1 g dose, while the removal efficiency increased from 66.15% to 95.52%. Similarly, for modified BC@HAp, the adsorption decreased from 36.43 to 4.45 mg/g, with efficiency rising from 75.44% to 92.05%. This pattern suggests that increasing the adsorbent mass enhances the total surface area, providing more adsorption sites, which improves CrVI removal efficiency (Mane et al., 2024). However, as the adsorbent mass increases, the amount of CrVI adsorbed per unit mass decreases, likely due to the overlap or aggregation of adsorption sites, reducing the effective surface area for interaction with CrVI ions (Li et al., 2022).
These findings align with previous studies, such as those by Li et al. (2018) and Ahmed et al. (2022), which observed similar trends in adsorbent mass affecting both adsorption capacity and efficiency (Ahmed et al., 2022; Li et al., 2018). Notably, the 0.03 g dose achieved CrVI removal efficiencies of 95.1% for BC and 89.9% for BC@HAp, only slightly lower than those at the 0.1 g dose. Based on these results, the 0.03 g dose was selected for subsequent experiments due to its balanced efficiency and manageable adsorption capacity.
Effect of initial CrVI concentration
Figure 5(C) illustrates the effect of solution concentration on CrVI adsorption by both BC and BC@HAp materials. As the initial CrVI concentration increased, the amount of CrVI adsorbed increased, while the adsorption efficiency decreased. For BC, the amount of CrVI adsorbed rose significantly from 8.43 mg/g at 5 mg/L to 40.45 mg/g at 80 mg/L, though the removal efficiency dropped from 99.11% at 5 mg/L to 33.04% at 80 mg/L. A similar trend was observed for BC@HAp, where the amount of CrVI adsorbed increased from 8.44 mg/g at 5 mg/L to 57.87 mg/g at 80 mg/L, with the adsorption efficiency decreasing from 98.27% at 10 mg/L to 47.26% at 80 mg/L.
The increase in CrVI adsorption with higher solution concentration, coupled with the decrease in removal efficiency, can be attributed to the greater availability of CrVI ions at higher concentrations, which enhances contact between the CrVI ions and the adsorbent material (Bayuo et al., 2019). However, as the concentration increases, the adsorption sites become saturated more quickly, leading to a reduction in adsorption efficiency (Zhu et al., 2022).
Effect of adsorption time
The results of CrVI adsorption over time by BC and modified BC@HAp, as shown in Figure 5(D), demonstrate that both the amount of CrVI adsorbed and the adsorption efficiency increase with time. For BC, the CrVI adsorption capacity rose quickly in the first 60 min, increasing from 11.70 mg/g (65.28%) to 17.28 mg/g (98.81%), and continued to gradually rise until 360 min, reaching 17.57 mg/g (98.05%). Similarly, modified BC@HAp also exhibited increased CrVI adsorption over time, with a rapid increase in the first 60 min, reaching 80.95%, and a more gradual rise to 82.71% at 360 min. This indicates that the adsorption rate is initially rapid due to the fast diffusion of CrVI ions to the available active sites on the material surface.
The two-phase adsorption behavior, with an initial rapid adsorption followed by a slower phase, is typical for adsorbents like BC and BC@HAp. The quick initial phase is attributed to the fast diffusion of CrVI ions to the adsorbent's surface, where active sites are abundant. As the adsorption progresses, the rate slows down as fewer sites remain available, and it takes longer for CrVI ions to diffuse deeper into the porous structure of the material. This phase continues until the adsorbent reaches saturation, limiting further adsorption. These findings align with previous studies (Chen et al., 2021b; Zhu et al., 2022), confirming that the adsorption process is controlled by both the availability of active sites and the physical limitations of the adsorbent.
Adsorption kinetics
To understand CrVI adsorption by BC and Fe-Mn@BC, kinetic data were analyzed using three models: Pseudo-second-order, Elovich, and Intra-particle diffusion. These models help identify the adsorption mechanisms and rate-limiting steps. The Pseudo-second-order model assumes chemisorption, where the adsorption rate depends on available active sites (Ho and McKay, 1999). The Elovich model describes a chemisorption process with varying adsorption energies (Aharoni and Tompkins, 1970), and the Intra-particle diffusion model highlights internal diffusion as the rate-limiting step (Lu et al., 2020). Linear kinetic parameters for these models are summarized in Table 3.
Regression parameters of CrVI adsorption kinetics and CrVI adsorption isotherm.

The kinetics data for pseudo second-order mode shows similar behavior for both materials in terms of the rate constant (k2), which is 0.04 g/mg.min for both BC and BC@HAp. However, the coefficient of determination (R2) for BC@HAp is notably higher (0.90) compared to BC (0.44), indicating that the adsorption process for BC@HAp fits the pseudo second-order model better, meaning the adsorption is more likely controlled by a chemisorption process. Additionally, the calculated adsorption capacity (qe,cal) for BC@HAp (14.91 mg/g) is very close to the experimental value (qe,exp = 14.51 mg/g), further supporting this finding. In contrast, the calculated adsorption capacity for BC (qe,cal = 17.66 mg/g) is slightly higher than the experimental value (qe,exp = 17.23 mg/g), suggesting some deviations from the model.
The Elovich model provides a strong fit for both BC and BC@HAp, with high R2 values (>0.94) and low χ2 values (<0.16), indicating an accurate description of the adsorption process. This suggests that CrVI adsorption onto both materials occurs on surfaces with heterogeneous active sites, where the adsorption energy varies. As a result, the adsorption rate for CrVI is initially rapid but decreases over time as high-energy sites become occupied. This pattern is characteristic of chemisorption, where the adsorbate interacts more strongly with specific sites, leading to multilayer adsorption (Elwakeel et al., 2020).
In addition to the Elovich model, the Intra-particle diffusion model is also employed to explore the possible rate-limiting steps of the adsorption process. This model suggests that diffusion within the pores of the adsorbent material could play a critical role in the adsorption of CrVI. Unlike the Elovich model, which emphasizes surface heterogeneity, the intra-particle diffusion model focuses on the transport of adsorbate molecules within the adsorbent's porous structure (Pour et al., 2025).
As shown in Figure 6, CrVI adsorption onto BC and BC@HAp occurs in three distinct stages. The first stage, occurring within the first 20 min, is characterized by rapid adsorption, likely due to the abundance of easily accessible sites on the surface of BC and BC@HAp. The rate constants for each stage (kid1, kid2, and kid3) follow the order of kid1 > kid2 > kid3 for both materials. Specifically, for BC, the rate constants are: kid1 = 1.58 > kid2 = 0.25 > kid3 = 0.005, while for BC@HAp, the values are: kid1 = 1.19 > kid2 = 0.17 > kid3 = 0.008. These results indicate that membrane diffusion (kid1) is the fastest stage, with the rate slowing as CrVI ions move deeper into the material's pores and interact with the adsorbent surface.

Fitting of the intra-particle diffusion for CrVI by BC and BC@HAp.
The second stage, spanning from 20 to 90 min, represents a slower, more gradual adsorption process, influenced by both liquid film diffusion and intraparticle diffusion. During this phase, CrVI ions continue to diffuse into the interior pores of the BC and BC@HAp, with the adsorption rate decreasing as fewer available sites remain on the surface (Dharmarathna and Priyantha, 2024). The third stage, starting after 90 min, reaches adsorption equilibrium, where the rate of CrVI removal diminishes further as both diffusion processes slow. Notably, the values of C1, C2, and C3 increase over time (data not shown), indicating that liquid film diffusion becomes more significant as the process continues (Zhu et al., 2017). This suggests that, over time, the adsorption process increasingly depends on the movement of CrVI ions through the surrounding liquid film and deeper into BC and BC@HAp material. This multi-stage process reflects the complex mechanisms involved in CrVI removal, where diffusion and surface interactions evolve throughout the adsorption process.
Adsorption isotherms
In this study, the equilibrium data for CrVI adsorption by BC and Fe-Mn@BC were modeled using the linearized Langmuir, Freundlich, and Temkin isotherms. The equations for the Langmuir, Freundlich, and Temkin isotherms are provided in Table 1, with their respective model parameters summarized in Table 2. The Langmuir model assumes uniform binding sites with equal affinity for the heavy metal ions, indicating monolayer adsorption on a surface with a finite number of identical sites (Febrianto et al., 2009). In contrast, the Freundlich model suggests that adsorption occurs on heterogeneous surfaces with multilayer adsorption, and the adsorption sites vary in energy, making it suitable for systems with non-uniform surfaces (Foo and Hameed, 2010). The Temkin isotherm, on the other hand, accounts for adsorption that decreases logarithmically with increasing coverage, assuming that adsorbate interactions reduce the overall energy of adsorption sites as the surface becomes more occupied (Kalam et al., 2021).
The comparison between the isotherm data for BC and BC@HAp reveals notable differences in their adsorption capacities and behavior. In the Langmuir isotherm, BC exhibits a maximum adsorption capacity (qm) of 39.95 mg/g with a Langmuir constant (KL) of 0.37 L/mg, showing a moderate fit to the model with an R2 value of 0.51. In contrast, BC@HAp displays a much higher adsorption capacity of 67.68 mg/g and a higher KL value of 0.54 L/mg, with a better fit (R2 = 0.81), suggesting that BC@HAp has a more homogeneous adsorption surface and a higher adsorption efficiency.
For the Freundlich isotherm, BC has a 1/n value of 0.19, indicating favorable adsorption, with a KF value of 17.92 and a strong fit to the model (R2 = 0.95). On the other hand, BC@HAp has a slightly less favorable 1/n value of 0.26 but a higher KF value of 22.59, indicating a greater adsorption capacity. The Freundlich model fits even better for BC@HAp (R2 = 0.99), highlighting its more effective adsorption properties. In the Temkin isotherm, BC shows a higher
Overall, BC@HAp consistently demonstrates superior adsorption performance compared to BC, as evidenced by its higher adsorption capacities, better fits to the Freundlich and Temkin models, and stronger affinities for CrVI. The better fit to the Freundlich model suggests a more favorable adsorption process with heterogeneous sites and multilayer adsorption, while the Temkin model fit indicates a uniform distribution of binding sites with an exponential decay of the adsorption energy. These results highlight the enhanced efficiency of BC@HAp for Cr(VI) removal from aqueous solutions, being approximately 1.7 times more effective than BC. Previous studies also found enhanced adsorption capacity of BC modified with HAp (Ahmed et al. 2022; Li et al. 2024), further demonstrating the beneficial impact of HAp modification on improving BC's adsorption properties.
Adsorption thermodynamics
The spontaneity of CrVI adsorption was assessed using thermodynamic parameters such as Gibbs free energy (ΔG), enthalpy (ΔH), and entropy (ΔS). Adsorption was studied at pH 6.0 and temperatures of 303, 308, and 313, with a CrVI concentration of 50 mg/L. The thermodynamic values were calculated using the following equations (Vasudev et al., 2020):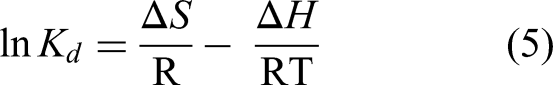


Kd, the distribution coefficient, which represents the equilibrium between the adsorbent and adsorbate concentrations; ΔH, the enthalpy change of the adsorption process (in J/mol); ΔS, the entropy change of the adsorption process (in J/mol.K); R, the universal gas constant (8.314 J/mol.K); and T, the absolute temperature in Kelvin. By plotting
The thermodynamic parameters in Table 4 indicate that both BC and Fe-Mn@BC adsorbents exhibit negative ΔG values, suggesting that the adsorption of CrVI is thermodynamically favorable and spontaneous under the experimental conditions (Barkat et al., 2009). As the temperature increased from 303 to 313 K, ΔG values decreased by 44.3% for BC and 45.2% for BC@HAp, highlighting a rise in spontaneity and efficiency of CrVI removal at higher temperatures (Loc et al., 2024). This trend suggests that higher temperatures enhance the adsorption process, likely due to the increased kinetic energy of CrVI ions, which promotes their interaction with the adsorbent. Consequently, the adsorption efficiency of BC increases from 72.25% to 88.52%, while BC@HAp increases from 83.68% to 96.38% as the temperature rises from 303 to 313 K.
Thermodynamic parameters for CrVI adsorption by BC and BC@HAp.

Furthermore, the ΔG values for CrVI adsorption on BC were consistently higher than those for BC@HAp, with reductions of 1.71, 2.20, and 3.22 kJ/mol at 303, 308, and 313 K, respectively. This suggests that CrVI adsorption on BC@HAp is more spontaneous than on BC (Zhao et al., 2014), explaining the superior adsorption capacity of BC@HAp observed in the isotherm study. The positive ΔH values (ΔHBC = 85.65 kJ/mol and ΔHBC@HAp = 129.83 kJ/mol) further suggest that the CrVI adsorption process is endothermic for both adsorbents, meaning that heat is absorbed from the surroundings during adsorption. Typically, physisorption occurs when ΔH is less than 40 kJ/mol, while chemisorption occurs within the range of 50 to 200 kJ/mol (Cantu et al., 2022). The high ΔH values observed in this study point to the significant role of chemical interactions in the CrVI adsorption process, particularly for BC@HAp. Additionally, the positive ΔS values (ΔSBC = 0.295 kJ/mol.K and ΔSBC@HAp = 0.446 kJ/mol.K) indicate an increase in disorder or randomness at the adsorbent surfaces as CrVI ions are adsorbed (Loc et al., 2024). This suggests that the adsorption process is thermodynamically favorable, endothermic, and results in an increase in the overall entropy of the system. These findings are consistent with previous studies on various adsorbents, such as activated carbon from Fox nutshell (Kumar and Jena, 2017), groundnut, walnut, and almond shells (Das et al., 2019), and inorganic clays modified magnetic chitosan adsorbent (Yuan and Lu, 2024), further supporting the spontaneous and endothermic nature of CrVI adsorption.
Possible adsorption mechanism of CrVI by BC@HAp
The mechanism of CrVI adsorption on both BC and BC@HAp may involve a combination of electrostatic interaction, ion exchange, complexation, and diffusion processes (Figure 7).
1. 2. 3.





Proposed mechanism of Cr(VI) adsorption onto BC and BC@HAp composite.
The carboxyl (−COOH) and hydroxyl (−OH) groups on BC form electrostatic bonds with CrVI ions, leading to surface complexation. 4. 
Comparison with other studies
The BC@HAp composite synthesized in this study achieved a maximum adsorption capacity of 67.68 mg/g for CrVI at near-neutral pH (∼6). This result is particularly significant when compared with other HAp-modified BC and composite systems summarized in Table 5.
Comparison of hydroxyapatite (HAp)-modified biochar and composite adsorbents for pollutant removal.
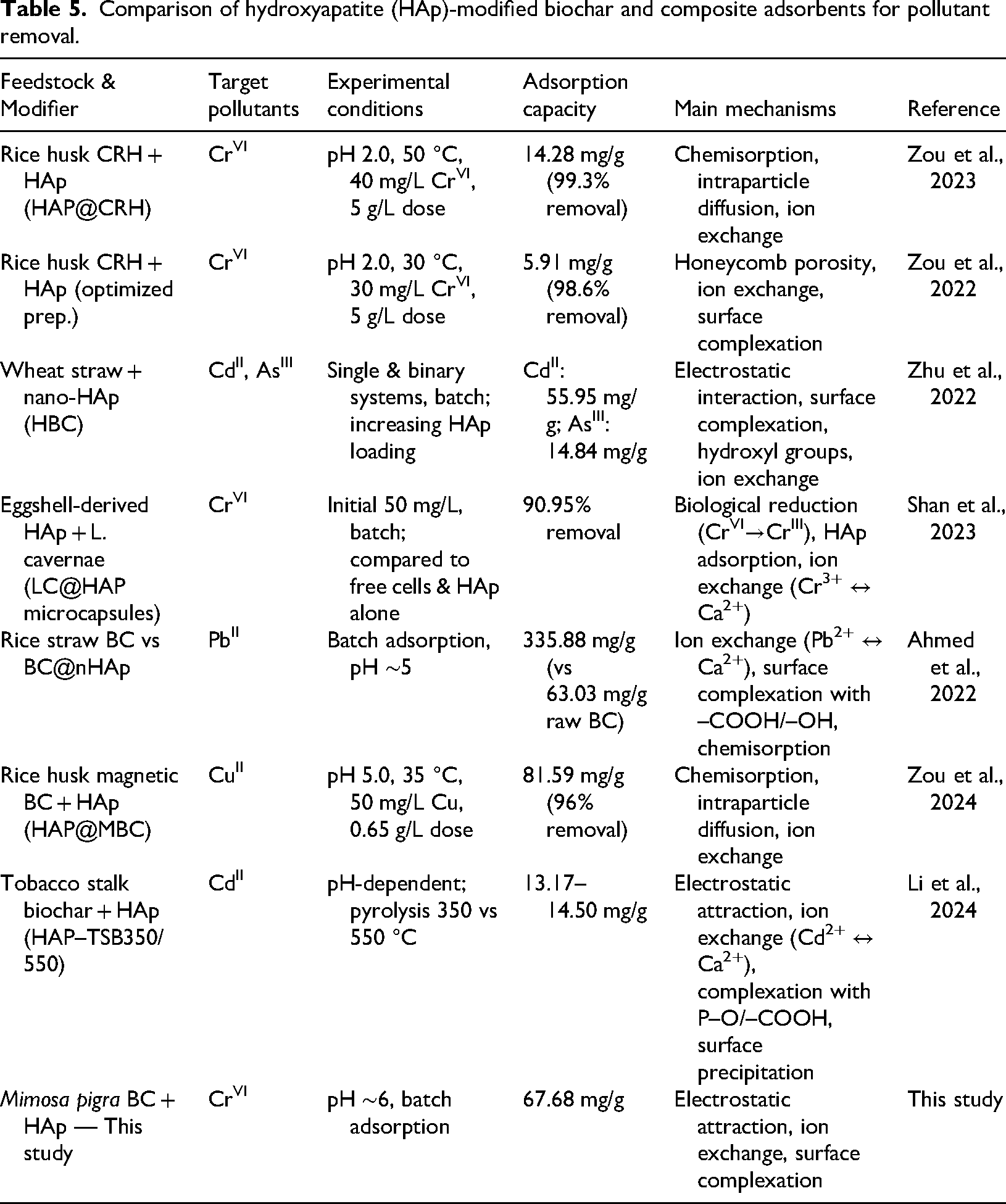
For Cr(VI) adsorption, most HAp-modified BCs perform best under highly acidic conditions (pH 2.0). Zou et al. (2022, 2023) reported adsorption capacities of only 5.91–14.28 mg/g using HAp-modified rice husk BCs, while Shan et al. (2023) achieved 90.95% removal with eggshell-derived HAp microcapsules combined with
For other metals, Ahmed et al. (2022) reported a capacity of 335.88 mg/g for PbII using BC@nHAp, while Zou et al. (2024) observed 81.59 mg/g for CuII with magnetic HAp-BC. Li et al. (2024) achieved only 13.17–14.50 mg/g for CdII with tobacco stalk BC, and Zhu et al. (2022) reported 55.95 mg/g CdII and 14.84 mg/g AsIII with HAp-modified wheat straw BC. These comparisons show that while PbII and CuII generally yield higher adsorption capacities, CrVI remains more challenging; thus, the 67.68 mg/g obtained here is competitive.
Overall, the present BC@HAp composite balances moderately high adsorption capacity with operation under environmentally relevant pH conditions, distinguishing it from many other HAp-modified systems that require extreme acidity. This makes it a practically viable and scalable option for CrVI removal from real wastewater streams.
Conclusions
In the present work, BC derived from the invasive
Despite these promising results, further investigations are necessary to explore the detailed mechanisms behind the adsorption process, including surface interactions and the role of the HAp modification. In addition, to assess the feasibility of scaling up this technology, it is crucial to conduct large-scale removal efficiency studies and techno-economic evaluations. This will help determine the practicality of BC@HAp in real-world applications for wastewater treatment, particularly in large-scale industrial and environmental settings.
Footnotes
Acknowledgement
The authors would like to thank the Environmental Advanced Techniques Lab and Solid Waste Treatment Lab of Can Tho University funded by JICA for conducting experiments and analyzing the CrVI.
Author contributions
Study conception and design: N.X. Loc; data collection, analysis, and interpretation of results: D.T.M. Phuong; draft manuscript preparation: N.X. Loc, D.T.M. Phuong. The results were evaluated by all authors, and the final version of the manuscript was approved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
