Abstract
In this work, the adsorption of copper ions onto activated carbons that oxidatively modified with H2O2 and HNO3 was studied. The prepared sorbents with the surface area of 275–780 m2/g contain 0.53–3.77 mmol/g of oxygen-containing groups. It was found that the oxidized activated carbon effectively adsorbs Cu2+ ions from aqueous solutions. For the most efficient activated carbon–HNO3–30 sorbent, the percentage removal of copper ions from solutions of varying concentration is high. In the concentration range from 1.5 to 6 × 10−4 M of Cu2+, it reaches >55%. At the concentrations lower than 1.5 × 10−4 M, the expected removal is above 80%. The experimental equilibrium adsorption data were modeled with 2- and 3-parameter isotherms. The analysis of adsorption modeling results accounting for the surface heterogeneity effect was provided. The data fit well to the Dubinin–Radushkevich model. Presumably, the complexation of Cu2+ with oxygen-containing groups passes within micropores. We found the correlation between the sorption capacity for Cu2+ and the concentration of surface groups. The presence of the carboxyl, anhydride, and lactone groups make a major impact on the adsorption.
Introduction
The copper that has many applications is an essential trace element supporting the growth of plants and animals. Despite this, the copper ions accumulated by the human body can cause serious health harms. At today’s level of consumption, one can purify heavy metals contaminated wastewater by means of carbon adsorbents. This adsorptive remediation satisfies the diverse demands on sustainability and environmental friendliness. To achieve a high efficiency of ions uptake, it seems to be the most reasonable technique.
Microporous carbons have a large specific surface area and tunable surface layer properties. These carbon materials show a high hydrolytic stability in alkaline and acidic media. As a rule, the presence of different types of pores provides the favorable mass transfer. On this background, the success in the removal of heavy metal ions from waters depends on the chemical behavior of carbon surface. Pyrzyńska (2007) reported that surface chemistry has a significant impact on the adsorption properties of carbons. Obviously, the acid–base behavior of the surface depends on oxygen-containing groups. In most cases, the surface interface regulates the complexation of functional groups with heavy metal ions. The composition of the resulted complexes depends on the type and the concentration of the surface groups (Bhatnagar et al., 2013). Also, one can suggest that these groups (at a high degree of surface coverage) can interplay and have a collective effect on the adsorption behaviour. So the adsorption capacity is influenced by many factors.
Commercial activated carbon (AC), in the most case, has sufficient textural parameters to take an advantage of porous structure for the purification of water (Yin et al., 2007). But the surface functional groups concentration seems insufficient to be used for an effective adsorption toward heavy metals ions. To increase the surface group concentration, the oxidative modification serves to be the most effective and the simplest way. Large concentrations of the oxygen-containing groups can be obtained if one treats the AC with reagents such as H2O2 and HNO3.
Taking all this into account, here we report on the oxidative modification and physicochemical properties of the resulted AC adsorbents. The prepared adsorbents have been characterized by many methods, including N2 adsorption–desorption and Boehm titration. Finally, we carried out batch adsorption experiments to check the feasibility of the removal of Cu2+ ions from water. And, the effect of the initial metal ion concentration on the adsorption capacity was estimated.
Modeling of experimental adsorption isotherm data, which were obtained by the batch tests in a lab, is an essential way for predicting the mechanisms of adsorption (Chen, 2015). Moreover, the adsorption behavior of copper ions may help understand the various (structural and chemical) factors affecting the adsorption capacity in the removal process of other divalent cations on the oxidative-modified ACs. But, one should check the applicability of adsorption models for predicting the properties of carbon adsorbents.
In this paper, we employed the linearized Langmuir, Freundlich, Redlich–Peterson (RP), and Dubinin–Radushkevich (DR) isotherm models. The results of the fitting of a set of the experimental adsorption equilibrium data for the copper ions with the respective theoretical isotherms were compared and discussed.
Materials and methods
The initial AC was derived from a natural source, Prunus Armeniaca fruit stones. This AC is cheap, mechanically stable granular carbon with a particle size of 0.5–1 mm. The initial AC has a large specific surface area of 785 m2/g and a significant pore volume of 0.41 cm3/g determined by water vapor adsorption–desorption. At the microstructure level, this granular carbon contains not only micropores and mesopores but also macropores. Obtained from natural raw materials (fruit stones), the AC maintains a system of microscopic channels. These channels are originated from the rest of the mass transfer capillaries of the fruit. Typically, such developed porous and capillary structure contributes to good kinetic parameters of adsorption.
Before the studies begin, the samples of AC were deashed by repeated washing with HCl, 3 mass% aqueous solutions. The sample was then washed with double distilled water (DDW) to the wash waters pH 5.5–6.0. Resulted washed samples were subjected to the oxidative modification. The oxidative modification was performed using standard, repeatedly described techniques (Jaramillo et al., 2010). Some differences in the ratios of reagents and the variation of the oxidation temperature and time are associated with the different intensity of the oxidative treatment for the different pairs of the initial AC and the oxidant (HNO3 or H2O2). The oxidation with H2O2 solutions does not proceed as intensively, as in the case of conc. HNO3; therefore, for H2O2 the processing time is longer. Despite the details, the purpose of these treatments is the formation of additional oxygen-containing groups on the surface of the AC.
In a typical oxidative modification, AC (2 g) was refluxed with 5 and 30 mass% HNO3 solution (60 ml) on a sand bath at 80℃ for 2 h. Resulted AC-HNO3-5 and AC-H2O2-30 were thoroughly washed with DDW and decanted with a Schott filter. The rest of the acid was washed with DDW to gain a rinsed water pH of 5.5–6.0. The modification products were dried in an air oven at 120℃ overnight. Such modification was also performed with 10 and 30 mass% H2O2 solutions: 2 g of AC was purged in 50 ml of 10 or 30 mass% H2O2 for 4 h. Decanted AC-H2O2-10 and AC-H2O2-30 were washed under pH-metric control and dried as reported above.
The bulk density (γ) was found by measuring the sample weight in a given volume. The water vapor sorption volume (VS) was measured by the static desiccator method at 25℃.
Nitrogen adsorption–desorption isotherms were recorded at −196℃ using an automated gas adsorption analyzer (Micromeritics, TriStar C10900A). Before physisorption measurements, all samples were outgassed at 180℃ for 4 h under flowing pure nitrogen gas (99.995%). Brunauer–Emmett–Teller surface area (SBET) and micropore area (Smicro) were determined from the adsorption data. For pore structure analysis, the isotherms were processed by the adsorption models. These models are derived within the two-dimensional nonlocal density functional theory (NLDFT) (Jagiello and Olivier, 2013). The pore sizes distribution (PSD) was found from the isotherms by means of the Micromeritics SAIEUS program (http://www.nldft.com). The best-fit carbon–N2 model for porous carbons with heterogeneous surfaces was used for the calculation. The proposed NLDFT model can be used in pair with the models available within quenched solid density functional theory. For microporous carbons, the use of both models most commonly gives the same results (Puziy et al., 2016).
The oxygen-containing functional groups on carbons surfaces were characterized by several methods. The total concentration of the oxygen-containing groups was estimated from the data of thermal analysis. The concentration of certain types of the oxygen-containing groups (Cfg) was measured by the Boehm titration. The temperature programmed desorption (TPD) method was used for control and comparison purposes (Salame and Bandosz, 2001). In a typical TPD experiment, dry samples were studied with an instrument reported in Diyuk et al. (2016a). This custom-made instrument combines a thermogravimetric analyzer and an IR spectrometer. The analyzer is operated in an inert gas medium or in vacuum using balances patterned after that of McBain and Bahr. Earlier, the measurements routine was conveyed in Diyuk et al. (2016a, 2016b, 2015). The quantification of certain functional groups was done from the area of the resolved TPD peaks.
The oxygen content of AC samples was investigated by means of electron probe microanalysis. A Cameca Camebax electron microprobe was used for the measurements. Determination of C, H, and N content was carried out on a Perkin Elmer Series II 2400 elemental micro analyzer. To remove physisorbed water, the samples were predried at 127℃ for 5 h and vacuumed up. The estimated error at the elemental analysis for C, H, and N was 0.5 wt%.
Changes in the surface chemistry of AC after the oxidizing treatment were investigated. The surface groups were analyzed by Fourier Transform Infrared (FTIR) spectroscopy. FTIR measurements were performed in attenuated total reflection (ATR) mode, using a spectrophotometer (IRPrestige 21, Shimadzu, Japan). This spectrophotometer was equipped with a ZnSe crystal MIRacle (Pike Technology) accessory. The spectrum of the ZnSe crystal was recorded as a background. The ATR spectra were collected in the wavelength range of 4000–520 cm−1 at a spectral resolution of 2 cm−1, and each spectrum is the average of 1000 scans.
Copper ions adsorption experiments
Batch equilibrium experiments
The capacities of the initial and the oxidized ACs to adsorb Cu2+ ions were determined through a series of experiments. Changes in the concentration of Cu2+ ions were controlled by means of a UV–Visible spectrophotometry. For this purpose, the sensitive method that used Cuprizone as a colorimetric agent was applied (Marczenko and Balcerzak, 2000).
For adsorption measurements, we used the water solutions that differ by the copper ion concentration. It was varied within the range from 6 to 1 × 10−4 M of Cu2+ with a step of 0.5 × 10−4 M. To prepare the solution series, a stock solution was diluted with DDW. This stock solution of 6 × 10−4 M of Cu2+ was obtained by dissolving standard Sigma-Aldrich copper(II) nitrate hemi(pentahydrate) in DDW. Besides, in the same way, we got the most diluted solution of 7 × 10−5 M of Cu2+.
Throughout the course of the experiments, a 50 mg of preliminary dried AC sample was dipped into a 20 ml water solution of copper ions. The resulted suspensions containing known concentration of Cu2+ were shaken in a 25℃ incubator at 150 r/min for a day. Typically, for this type of AC, the adsorption equilibrium is established rather quickly, within 15–18 h. For the convenience of measurements, the time of 24 h was chosen. Adsorption was conducted at fixed pH, which was adjusted to a constant value of 5.5 by using NaOH solution as a pH control reagent. After adsorption, the copper-depleted solutions were filtered through a Whatman 41 filter paper in fresh flasks. The filtered solutions were sampled for the spectrophotometric analysis. All the measurements were performed on a Cary 50 Scan Varian instrument with 1 cm quartz cells.
The analyses of the water solutions containing copper ions, from Cu(II) nitrate,
were conducted as reported below. The sampled aliquots of 5 ml were pipetted out
into 25 ml conical flasks. A 2 ml of a buffer (ammonium acetate) solution was
added into each of them. The studied solutions by default contain no more than
70 mg of Cu. A 3 ml of Cuprizone (Sigma, Bis(cyclohexanone)oxaldihydrazone) was
added to each of the solutions buffered to a pH of 8–9. The absorption of the
highly chromogenic complex with copper ions was measured at ∼600 nm, at the
extinction coefficient 16,000. All the measurements were taken at the optimal
measurement delay time of 10 min. After analysis, the equilibrium adsorption
capacity of Cu2+ on the adsorbent (a, mol/g) was
found by the equation
However, the use of the spectrophotometric technique is usually limited. Typically, it may generate errors when working at a very low concentration of the copper ions. That is why we additionally checked the ions concentration by a flame atomic absorption spectrometry (FAAS). The FAAS measurements of Cu were made on a Carl Zeiss Jena FAAS N1 spectrometer with an air–acetylene flame. The analysis was performed using the procedure described in Brzezicha-Cirocka et al. (2016).
Results
Elemental analysis of ACs.

AC: activated carbon.
Oxygen is calculated by difference from the mass of C, H, and N, and the total mass of the sample.
As can be seen in Table
1, there are changes in the composition. The oxygen content increases
markedly. The review by Jaramillo et al. (2010) indicates that it is the oxygen of the surface
groups of the carbon. Figure
1 presents the ATR spectra of the initial and oxidized AC samples
recorded in the wavenumber range of 2100–800 cm−1. The most intensive
bands in the spectra were addressed as recommended in Barroso-Bogeat et al. (2014). For the
oxidized ACs, the strong bands at 1739 and 1712 cm−1 were attributed to
ν(C = O) stretching bands of carboxyl (Cb), anhydride, and
lactone (A–L) groups. These bands are practically absent in the case of an ATR
spectrum of the initial sample. This means in particular that the concentration of
Cb groups on the surface of the initial AC is negligible. Without any doubts, the
groups Cb and A–L were formed in the course of the oxidation. For the initial AC and
AC-H2O2-30, the ν(C = O) mode of quinolic
(Qu) groups is observed at 1697 cm−1. For the AC-HNO3-30
sample, the band ascribed to this vibration is at 1678 cm−1. Bands
associated with skeletal vibrations of the C = C bond can be clearly seen in Figure 1. The band at about
1640–1680 cm−1 is attributed to vibrations of conjugated C = C bonds.
The bands of aromatic C = C bonds appear at smaller wavenumbers, at about
1510–1550 cm−1. These spectral features of C = C bonds are registered
for all the samples that are studied. For the oxidized ACs, the intensive absorption
bands are observed at 1196 and 1202 cm−1. These two bands correspond to
ν(C–OH) vibrations of phenolic (Ph) groups formed as a result
of oxidation. The absorption bands that could be seen in the range from 981 to
1000 cm − 1 correspond to νs(=C–O–C) of
ether groups. Thus, the ATR spectra imagined the presence of Ph and Cb groups that
formed mainly during the oxidative treatment of the initial AC. Typical ATR spectra of the initial and oxidized
ACs. AC: activated carbon.
Surface characteristics of ACs.

AC: activated carbon.
Boehm titration/TPD data.
According to the TPD data, oxidation of AC with H2O2 caused the significant increase of the content of Ph and A–L groups. The increment of Cb is insignificant. Typically, the concentration of Cb groups does not exceed 0.15 mmol/g. Under conditions of adsorption at pH = 5.5, the aforementioned Ph groups practically do not dissociate. This means that they cannot take part in the formation of surface compounds. The statement is right if one supposes that the adsorption of copper occurs by the mechanism of complexing. Perhaps this complexation involves only certain types of the oxygen-containing groups. All types of groups are intensively formed when the AC is oxidizing with nitric acid. The total concentration of the oxygen-containing groups is very significant. Clearly, this situation can lead to a reaction between neighboring groups upon heating. In contrast to the oxidation with peroxide, the oxidation with HNO3 caused the formation of a significant quantity of Cb groups. From the Boehm titration, the relative content of Cb groups in the samples of AC-HNO3-5 and AC-HNO3-30 is 60 and 50%, correspondingly. In sum, the relative content of the most acidic groups (Cb and A–L groups) is about 70%. The rest are Ph groups, whose content is only 30%. Thus, the total content of acid groups (CA) for the samples of AC-HNO3-30 and AC-HNO3-5 is high. We compared the concentrations of oxygen-containing groups obtained by two methods (Table 2). The data of Boehm titration and TPD method show a certain distinction. Probably, the data obtained by the TPD method are somewhat subjected to systematic errors. The errors are due to the fact that reacting Cb and/or Ph groups can form A, L, and other similar groups. Consequently, the Boehm titration results are more reliable than that of TPD. This is true if one characterizes the oxygen-containing groups as adsorption centers in an aqueous solution.
The nitrogen adsorption isotherms and the rules of PSD are plotted in Figure 2(a) and (b),
correspondingly. N2 adsorption isotherms (a) and PSDs (b) of
the initial and oxidized ACs. AC: activated carbon; PSD: pore sizes
distribution.
Textural parameters of ACs.

AC: activated carbon.
Figure 2(b) presents two types of pores in the AC samples. The small micropores are in the range of widths of 1.06–1.32 nm. The mesopores with a width of about 3.4 nm can also be seen. Upon oxidation, the width of the micropores slightly increases, from ∼1.1 nm in the initial AC to 1.2–1.3 nm in the oxidized ACs. The oxidation with nitric acid leads to a decrease in the number of micropores. The large pores (mesopores) with a width of 3.4 nm are also formed. In the case of the strongly oxidized AC samples, the widening of micropores can convert some micropores into mesopores. For the AC-HNO3-30 sample, the formation of new micropores (pore width ∼1.7 nm) is registered.
In light of these observations, we will summarize the facts below. Actually, macromolecular graphene fragments undergo oxygenolytic degradation. This, without a doubt, has caused the microfracture of the micropores walls. The oxidative destruction is the fast process, especially in the presence of strong oxidants, even at room temperature. The graphene-like carbon matrix is readily destroyed by the oxidation in aqueous solutions. From a structural point of view, the oxygenolysis is one of the reasons of the micropores expansion and the appearance of new mesopores. On this background, one could focus on the adsorption isotherms of Cu2+ that differ significantly.
The experimental data on the equilibrium study for the adsorption of Cu2+
ions onto the adsorbents were plotted in Figure 3(a). The plots show that the most
efficient adsorbents are AC-HNO3-5 and AC-HNO3-30 samples, the
percentage removal R exceeds 50%, as shown in Table S1 of
Supplementary Material. In the range of low concentrations of Cu2+, below
1.5 × 10−4 M, the value of R is above 80%. In our
opinion, this is due to the high concentration of the oxygen-containing groups. 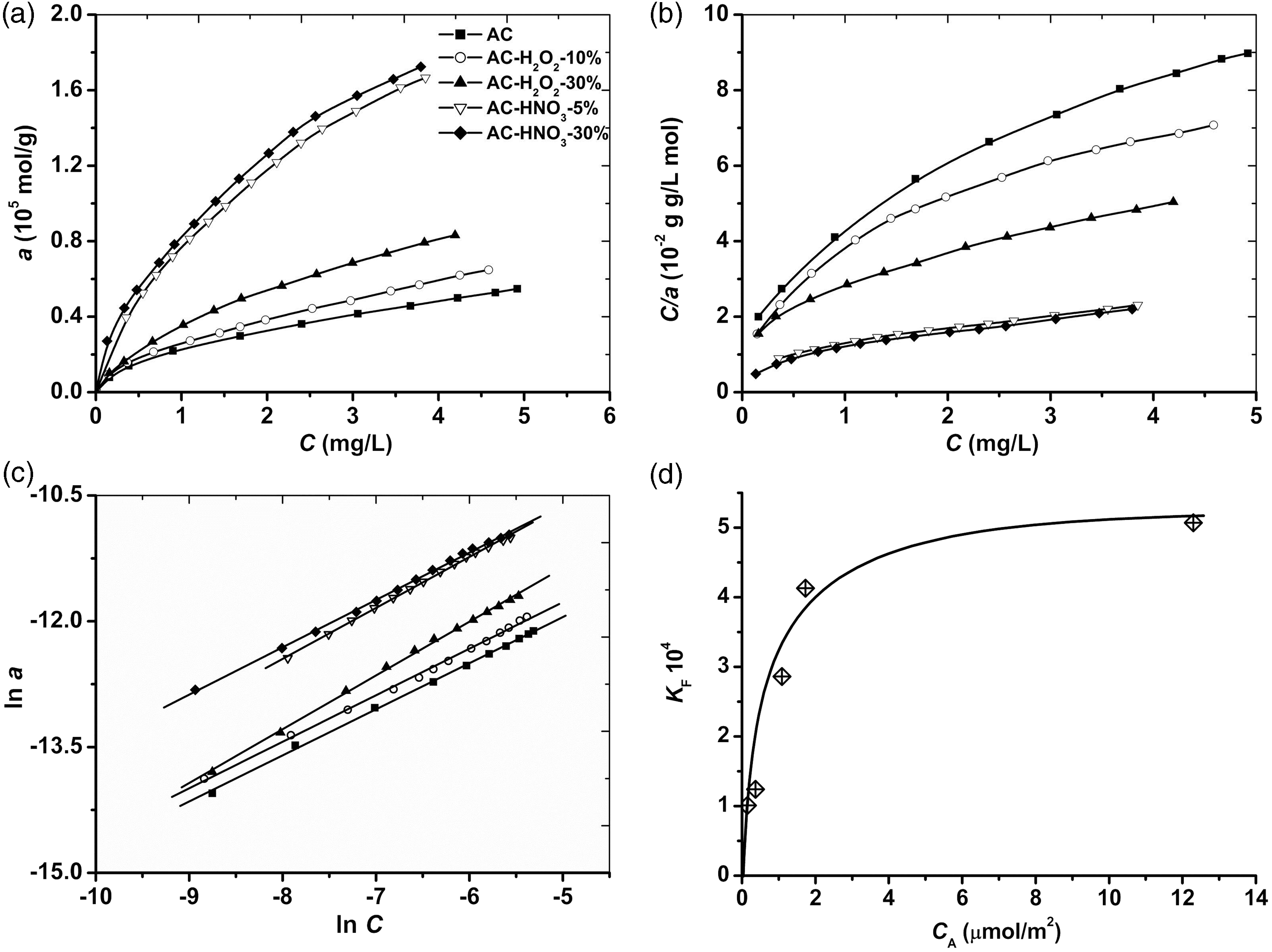
The experimental equilibrium data were fitted to different isotherm models. The linear and nonlinear least squares methods were used to find the best-fitting curve within the framework of the chosen model. The parameters were selected so that the sum of the squared deviations of the theoretical values from the experimental points takes a minimal value. The residual (R) factor was a measure of the agreement between the theoretical models and the equilibrium adsorption data.
At the first, the most widely used Langmuir isotherm is considered for the
description of the experimental data. The use of this isotherm model is reasonable
in the case of the homogeneous surface when adsorption energy is constant over all
sites. The adsorption data were fitted with the most common two-parameter Langmuir
model 

At the second, we have chosen the Freundlich isotherm to describe the adsorption.
This isotherm describes adsorption that passed on the heterogeneous surface with
varied affinity. An empirical Freundlich equation is expressed by 

Constants and correlation coefficients of Freundlich plots.

AC: activated carbon.
The KF increases with the oxygen content O (cf. the data in Tables 3 and 4). This means that the surface oxidation causes an increase of the number of adsorption centers addressed to CA (Figure 3(d)). Consequently, it is not surprising that some newly formed centers have the higher affinity for copper ions. The nF is in the range of 1.56–1.82 and it shows a complex dependence on both O and CA magnitudes.
To describe the adsorption on the heterogeneous surface, one can choose a hybrid
isotherm. It seems reasonable to use the one that involves features of both the
Langmuir and the Freundlich isotherms. This model is the RP isotherm. According to
the model, three parameters are incorporated into an empirical equation



But, to get started, here one has to distinguish between equations (8) and
(9) to
find the suitable form of the RP equation. To fit a set of data with respective
models we used the linear and nonlinear least square regression analyses. The
constants
The adsorption data were also fitted with the two-parameter model of DR, which is
valid for the adsorption in micropores. The DR equation was used in the linear form
Adsorption of copperions on the studied ACs (a)
fitted to RP model, (b) log a against
(log(Cm/C))2,
and (c) am against
CA within DR model. AC: activated
carbon; DR: Dubinin–Radushkevich; RP:
Redlich–Peterson. Constants and correlation coefficients of RP
plots. AC: activated carbon; RP:
Redlich–Peterson. Constants and correlation coefficients of DR
plots. AC: activated carbon; DR:
Dubinin–Radushkevich.



A good mathematical description alike the Freundlich model was obtained for the DR model (Figure 4(b)). This apparently means that the substantial heterogeneity of the surface, which is taken from the Freundlich equation, is associated with the micropores that account the DR isotherm. In other words, if the Freundlich model works well, this means that the surface is extremely heterogeneous. Despite that the description of such heterogeneity having physical meaning only within the DR model of adsorption. Heterogeneity of the surface should, therefore, be understood as a different number of adsorption sites for different samples. As can be seen from the data, the surface oxidation affects the magnitude of am, which can grow by more than five times. A nonlinear function describes the correlation between am and CA (Figure 4(c)). This means that the presence of Cb and A–L groups is necessary for the best adsorption. The mentioned groups should cover the outer surface of the micropores that are involved in the adsorption of Cu2+. We believe that Cb and A–L groups, as the most acidic surface centers, make the major contribution to the process of interest. With nonlinearity, the effect of CA on am depends on the value of CA. Consequently, the reason for the observed correlation is the fact that the most intensive oxidation treatment of the surface suppresses the formation of the functional groups. Alternatively, the concentration of newly formed oxygen-containing groups is markedly limited. This limitation imposed by the relative strength of an oxidizing agent cannot be overcome in the solutions. As discussed earlier, the same type of correlation exists for the Freundlich model (Figure 3(d)). Absorption energy EDR is found to be 22–24 kJ/mol. This value is independent of the degree of surface oxidation. Apparently, it should be, if the adsorption involves similar oxygen-containing groups.
Discussion
The results of intensive studies of the adsorption of metal cations from aqueous solutions were reported in Alvarez-Merino et al. (2005) and Puziy et al. (2004). Previous investigations have shown that the oxygen-containing groups favor the metal ions adsorption on the surface of AC (Alvarez-Merino et al., 2005; Dastgheib and Rockstraw, 2001). The adsorption could realize through the formation of metal complexes (Chen and Lin, 2001; Chen et al., 1996). Also, the adsorption of metal ions could be considered as a kind of ion-exchange reaction (De Mesquita et al., 2006). From the common opinion, the ion exchange takes place with the participation of strong acidic surface groups.
From the literature is known that many factors have an influence on the adsorption. For example, the presence of various oxygen-containing groups and the specific features of the porous structure are among major factors considered. Besides, one should account for pH, the ionic strength of solutions, and the concentration of heavy metal ions in stock solutions. Finally, the adsorption depends on concentrating conditions and the efficiency of complexation. In sum, all the above-mentioned factors will have an impact, directly or indirectly, on the concentration of adsorbed ions.
We should note that the influence of certain factors on the adsorption is not completely clear. For example, it is well known that strong acidic groups are capable of entering into ion-exchange reactions on the surface of AC. But, the presence of the strong acidic groups does not guarantee their participation in the Cu2+ ions adsorption, according to Puziy et al. (2004).
The role of the surface groups, in the case of the oxidized microporous carbons, can be considered as balanced. For easy adsorption, the intermolecular forces and the molecular properties of adsorbate should be at the optimum balance (Terzyk, 2003, 2004a; Terzyk et al., 2011). Moreover, studying different adsorbates, Terzyk (2004a) reported that the interactions between solute and surface groups caused the blocking of micropores. This effect is observed when the primary micropores are filled (Terzyk et al., 2011). It almost vanishes for large micropores at acidic pH. The protonation of carbon surface supports the adsorption by increasing surface heterogeneity (Terzyk, 2003). So, one can conclude that the porous structure and the type of pores are not the major factors determining the adsorption capacity of the samples. To improve the efficiency of adsorption, the optimal dimensions of micropores should combine with the coverage of the surface with specific oxygen-containing groups (Terzyk, 2004b; Terzyk et al., 2011). In our case, very narrow micropores inside the AC can often be blocked. This blocking occurs when the adsorption of water molecules or Cu2+ cations takes place on the functional groups at the entrance to the pore. However, the total geometry of the pore structure changes during oxidation. Moreover, the intensive oxidation caused the micropores widening. These changes reduced some steric hindrances restricting the diffusion.
The present study shows that the AC samples oxidized with nitric acid (AC-HNO3-5 and AC-HNO3-30) have the greatest adsorption of Cu2+ ions. For the oxidized AC samples, compared to the initial AC, the decrease of the SBET can be explained only by the reduction of microporosity. This means that the micropores actively participate in the oxidation process. Besides, it is possible that the required functional groups can form inside the resulted micropores. From the analysis of the PSD rules, it is clearly seen that due to the oxidation, the size of micropores slightly increases. On a background of this increase, new micropores and mesopores are registered. The moderate decrease in VS and a certain increase in γ agree with the marked decrease in the number of micropores that presented in the initial AC. The micropore widening and the formation of many new open micropores compensate that decrease.
Notably, the results of adsorption cannot be explained using only the texture characteristics of adsorbents. Of course, the texture changes and the reducing of the microporosity should have an effect on the adsorption of Cu2+. On the other hand, it should be mentioned that the oxidized carbon matrix has covered with the surface groups that are different in acidity. These groups can be also affecting on the adsorption equilibria. The concentration of Cb groups for AC-HNO3-5 and AC-HNO3-30 compared to AC-H2O2-5 and AC-H2O2-30 is significant. The same holds true for the concentrations of products of the interaction between Cb groups, namely for A and L groups. This led to a conclusion (consistent with the facts) that the adsorption can be associated with the interaction of Cu2+ with the Cb and A–L groups of the oxidized AC samples. These findings are really important. They offer strong evidence that the surface centers of a high acidity take part in the adsorption of metals ions. This statement in somewhat manner is in contrast to findings of Puziy et al. (2004). Besides, we also found a very good agreement between the experimental and the DR theoretical isotherms. This good agreement proves that the adsorption of copper ions is localized in the micropores. The reason of the most effective adsorption could be a better coordination of the copper cation. In fact, for the better ions’ coordination is required the neighboring of several functional groups inside the open porous system. But, it should be recognized that in practice this coordination is difficult to implement beyond the micropores. The sample AC-HNO3-30 is characterized by a decrease in the surface of micropores. The microporosity decrease is accompanied with an increase in the total concentration of Cb and A–L, up to 2.65 mmol/g. In this case, an interaction of Cu2+ with two adjacent Cb groups becomes possible. For oxidative-modified ACs, we suggest the potential mechanism of copper(II) ions adsorption through the formation of a surface complex. The complexing has occurred in the micropores and involves several Cb groups, apparently a pair. Additionally, other groups can be involved in the dynamic adsorption process. For example, hydrolysis of L groups gives the Cb and Ph groups, while A groups transform in two Cb groups. In this respect, we considered the dependence of the maximum adsorption capacity, am, versus CA—the sum of concentrations of the most acidic (Cb, L, and A) surface groups. The obtained correlations for the oxidative-modified AC showed a complex character of dependence.
Conclusions
The evaluation of the applicability of adsorption models for predicting the properties of the oxidative-modified carbons is generally going beyond the focus of the studies reported in the literature. At the considered concentration range, the experimental isotherms can be fitted to many mathematical models. This study demonstrates that equations like Freundlich and DR are modeled reasonably well the adsorption isotherms. Nonetheless, the results of this Cu2+ adsorption modeling have a direct physical meaning only within the DR equation. The very high R-factor values derived for the model-to-data fits, together with visual inspection of the predictions, imply that the DR model adequately describes the surface heterogeneity and suggesting cations adsorption in the micropores of the oxidized AC. We attribute significant adsorption efficiency to a high content of adsorption centers acting simultaneously. Within the hypothesis of the mechanism of adsorption of the copper ions by complexing, these centers were assigned to Cb, A, and L groups that exist on the surface of the oxidative-modified adsorbents.
Footnotes
Acknowledgements
Work “Modeling of copper ions adsorption onto oxidative modified activated carbons” first presented at the “15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: V.V.L. acknowledges a partial financial support from the National Scholarship Program of the Slovak Republic for the Mobility of Students, PhD Students, University Teachers, Researchers and Artists, SAIA Grant, n.o., in 2015 and 2017. This work was partially funded by the Ministry of Education and Sciences of Ukraine, Grants 0111U006261, 0114U003554, and 0116U00258.
Supplementary Material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


