Abstract
MnxOy, CoxOy, and MnxOy-CoxOy supported on activated carbon (AC) adsorbents have been successfully prepared and characterized by specific surface area (BET), X-ray photoelectron spectroscopy, X-ray diffraction analysis, and Fourier-transform infrared spectroscopy. The results show that Mn is present in the form of Mn3+ and Mn4+, and Co is present in the form of Co2+ and Co3+. For all of the modified samples, the functional groups of the AC were well retained and the specific surface areas followed the order: AC > MnxOy-CoxOy/AC > Co3O4/AC > MnxOy/AC. For MnxOy-CoxOy/AC, Mn and Co mainly exist in the forms of MnO2, Co3O4, and Mn3Co2O8. MnxOy-CoxOy/AC was found to exhibit the best H2S desulfurization performance. At 20℃, the breakthrough time and saturated adsorption time were 115 min and 185 min, and the saturated sulfur capacity and desulfurization rate were 347.7 mg/g and 74.2%. The H2S removal capacities of the different adsorbents decreased in the order: MnxOy-CoxOy/AC > MnxOy/AC > CoxOy/AC > AC.
Introduction
Natural gas holds an irreplaceable position among energy resources (Zou et al., 2013). The deep desulfurization of H2S from natural gas is a very important gas clean-up process. Currently, wet and dry desulfurization methods are mainly applied to natural gas. Wet desulfurization is used for high concentrations of hydrogen sulfide. It has the advantage of large capacity, but the disadvantage of a complicated operation process. Dry desulfurization employing solid adsorbents is used for low concentrations of hydrogen sulfide. It has the advantages of less energy consumption and a small footprint. Activated carbon (AC), molecular sieves, and silica are the solid adsorbents usually used for dry desulfurization (Yu et al., 2009).
AC has a developed pore structure and a high specific surface area. However, adsorption on unmodified activated carbon is mainly by physical adsorption, which is poor in desulfurization. Recently, the modification of activated carbon has become a hot topic. As reported by Hui-bin Fang (Fang et al., 2013), metal oxides supported on activated carbon adsorbents showed much higher desulfurization performances for H2S. The desulfurization performances of various adsorbents decreased in the order: Mn/AC > Cu/AC > Fe/AC > Ce/AC > Co/AC > V/AC. It has been shown (Li et al., 2011) that CoxOy/AC has electrocatalytic activity for oxygen reduction reaction under acidic conditions. An adsorbent composed of Mo-Co supported on activated carbon exhibited good carbon dioxide removal performance for flue gas (Wang et al., 2012).
In the present work, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC have been prepared, and their adsorptive desulfurization performances toward a simulated low H2S concentration natural gas have been studied.
Experimental
Preparation of adsorbents
Activated carbon-supported manganese oxide (MnxOy/AC) was synthesized by reduction of KMnO4 using hydrogen peroxide with AC. The solid was dried to constant weight to obtain MnxOy/AC precursor (PMA). The PMA was calcined at 300℃ for 2 h to obtain MnxOy/AC adsorbent with an Mn:AC mass ratio of 1:1. Activated carbon-supported cobalt oxide (CoxOy/AC) was synthesized by impregnating AC with cobalt nitrate solution (8 wt%). After filtration and drying in air, the solid was calcined at 300℃ for 2 h to obtain CoxOy/AC adsorbent with a Co:AC mass ratio of 0.8:1. Activated carbon-supported manganese oxides and cobalt oxides (MnxOy-CoxOy/AC) were synthesized by impregnating PMA with cobalt nitrate solution (8 wt%). After filtration and drying in air, the solid was calcined at 300℃ for 2 h to obtain MnxOy-CoxOy/AC adsorbent with an Mn:AC:Co mass ratio of 1:1:0.8.
Characterization of adsorptions
XPS experiments
X-ray photoelectron spectra (XPS) were obtained by using an Κ-Alpha (Thermofisher Scientific) with Al Ka X-radiation (the resolution of the instrument is 1.15 eV).
X-ray diffraction experiments
X-ray diffraction (XRD) were obtained by using an D/max-2200PC (Rigaku Corporation) at 40 kV and 30 mA using monochromatized Cu-Ka radiation. The patterns were measured over the 2θ range from 10° to 80° at a scan step of 10°/min.
Fourier transform infrared spectroscopy Experiments
Fourier transform infrared spectroscopy (FTIR) were obtained using the KBr disk technique on a TENSOR 27 (Bruker) FTIR spectrophotometer (the resolution of the instrument is 4 cm−1). Background and sample were scanned in a minute by using 24 DigiTect TM detector system and ROCKSOLIDTM interferometer.
BET experiments
BET surface areas were determined by nitrogen adsorption at 77 K by using a Micromeritics ASAP 2020 (USA) instrument. Prior to N2 physisorption, the adsorptions were degassed at 120℃ for 4 h.
Adsorption experiments
Measurements of adsorbent activity were conducted in a fixed-bed reactor. The fixed-bed reactor, which consisted of a quartz glass tube of internal diameter 10 mm and length 300 mm, was positioned vertically in a clam shell furnace. A 0.2 g of the prepared adsorbent was loaded into the center region of the reactor. The up and down sides of the adsorbent were filled with quartz sand. The model fuel gas from a feed tank passed through a reducing valve and entered into the reactor at a flow rate of 20 mL min−1. Any unadsorbed H2S gas in the effluent gas would be absorbed by the liquid in the absorption bottle, and the H2S content of the absorption liquid was determined by iodometry at intervals of 5 min. The breakthrough concentration was defined as that at which the H2S concentration in the effluent gas reached 10% of the initial gas concentration, that is, 2 ppm. The H2S capacity, saturated H2S capacity, and H2S removal rate were calculated according to the concentration of sulfur in the absorption liquid after the experiment.
The H2S breakthrough capacity is defined as the H2S capacity in the adsorbent when the H2S concentration in the effluent gas reaches 10% of the initial concentration (2 mg/L). It can be calculated as follows:
H2S saturation capacity is defined as the amount of adsorbed H2S per gram of adsorbent when the H2S concentrations in the effluent and in the initial gas are the same. It can be calculated as follows:
The H2S removal rate η can be expressed as:
Results and discussion
Adsorbent characterization
XPS analysis
The MnxOy/AC and MnxOy-CoxOy/AC adsorbents were studied by XPS and the Mn 2 p spectra are shown in Figure 1(a). It is well known (Di and Park, 2012; Li et al., 2009) that Mn3+ can be confirmed by an Mn 2p3/2 binding energy within the range 641.3–641.7 eV, and that Mn4+ can be identified by a binding energy of the XPS contribution in the range from 642.2 to 643 eV. The Mn 2p3/2 line for MnxOy-CoxOy/AC was chemically shifted by 0.4 eV and 0.5 eV to lower binding energy compared to that for MnxOy/AC. The Mn3+/Mn4+ mass ratio of MnxOy/AC was 0.19/0.81. Compared with MnxOy/AC, the Mn3+/Mn4+ mass ratio of MnxOy-CoxOy/AC was increased to 0.29/0.71, showing an increase in the amount of Mn3+ by introducing the CoxOy. The Mn 2 p content in MnxOy-CoxOy/AC was decreased from 26.4% to 13.4% by introducing CoxOy (Table 1). The Co 2p3/2 spectra of CoxOy/AC and MnxOy-CoxOy/AC are shown in Figure 1(b). The XPS patterns of CoxOy/AC and MnxOy-CoxOy/AC show two peaks and the shake-up satellite. For CoxOy/AC, the binding energy at 786.1 eV can be attributed to Co2+ species, whereas that at 781 eV can be attributed to Co3+ species (Cheng et al., 2004; Machida et al., 2000). For MnxOy-CoxOy/AC, the Co 2p3/2 line was chemically shifted by 0.2 eV and 0.6 eV to lower binding energy compared to that for CoxOy/AC. The Co2+/Co3+ mass ratio of CoxOy/AC was 0.11/0.89. Compared with CoxOy/AC, the Co2+/Co3+ mass ratio of MnxOy-CoxOy/AC was increased to 0.20/0.80, showing an increase in the amount of Co2+ by introducing MnxOy. The Co 2p content was increased from 2.1% to 3.8% (Table 1).
XPS spectrum of MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC absorbents. Spectral parameters of AC, CoxOy/AC, MnxOy/AC, and MnxOy-CoxOy/AC by XPS analysis.

The electronic absorption abilities of the different species decrease in the order: Co3+ > Mn4+ > Mn3+ > Co2+. For MnxOy-CoxOy/AC, the shift in the peak positions of the Mn and Co species can be attributed to an increase in the electron cloud density, which is caused by sharing electron pairs through covalent bonds because of interaction between these four species.
XRD results
The XRD patterns of AC, CoxOy/AC, MnxOy/AC, and MnxOy-CoxOy/AC are shown in Figure 2. The diffuse peaks at 2θ = 20°–30° (002) and 2θ = 40°–50° (101) can be assigned to activated carbon, and indicate an irregular microcrystalline graphite structure therein. For MnOx/AC, the peak at 2θ = 12.8° (110) can be attributed to birnessite MnO2, and the peak at 2θ = 36.1° can be attributed to birnessite Mn3O4. In addition, the peaks at 2θ = 20°–30° (002) and 2θ = 40°–50° (101) attributable to the microcrystalline graphite structure of activated carbon were no longer seen. For CoxOy/AC, the peaks at 2θ = 36.9° (311), 56° (422), 60.1° (511), and 65.2° (440) can be attributed to birnessite Co3O4, and the peaks at 2θ = 27.9° and 44.7° can be attributed to birnessite carbon, indicating that the regularity of the activated carbon structure was greatly enhanced. For MnxOy-CoxOy/AC, peaks arising from the surface of the microcrystalline graphite structure of the activated carbon were no longer seen, and the peaks at 2θ = 12.8° (101) 18° (200), 28.7° (301), and 49.9° (411) could be attributed to cubic phase MnO2 (JCPDS Card No. 44-1386) (Tan et al., 1991). The peaks at 2θ = 37.4° (311), 41.9° (400), 56° (422), 60.1° (511), and 65.2° (440) could be assigned to cubic phase Co3O4 (JCPDS Card No. 42-1467) (Jiang et al., 2013; Li et al., 1999). The peak at 2θ = 37.5° could be attributed to birnessite Mn3Co2O8, showing the interaction between Mn and Co. In conclusion, Mn and Co mainly exist in the forms of MnO2, Co3O4, and Mn3Co2O8 in MnxOy-CoxOy/AC. These findings are consistent with the XPS results. Compared with MnxOy/AC, the peak at 2θ = 12.8° for MnxOy-CoxOy/AC becomes sharp, showing an increased degree of crystallinity of MnO2.
XRD patterns of AC, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC.
FTIR results
Figure 3 shows the FTIR spectra of the AC, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC adsorbents. For each of the samples, a peak at 3420 cm−1 was observed, which could be attributed to the O–H stretching vibration of hydroxyl, carboxyl, and surface-adsorbed water. The peaks at 2926 and 1629 cm−1 could be attributed to the symmetric and asymmetric stretching vibrations of C–H and C = O of the lactone base, respectively. The peaks at 1383 and 1039 cm−1 could be attributed to asymmetric and symmetric COO− stretching vibrations and skeleton C–O stretching vibrations of the activated carbon, respectively. In conclusion, the functional groups of AC remained unchanged for MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC. Compared with AC, a peak at 601 cm−1 was detected for MnxOy/AC, which could be attributed to the characteristic absorption of Mn-O (Wang et al., 2014). For CoxOy/AC, peaks at 581 and 681 cm−1 could be attributed to the characteristic absorptions of Co–O (Bahaa et al., 2014). For MnxOy-CoxOy/AC, a strong and sharp characteristic peak at 570 cm−1 could be attributed to overlapping absorptions of Mn-O and Co-O, showing the strong interaction between Mn and Co. These findings were in accordance with the results obtained by XRD and XPS.
FTIR spectra of AC, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC.
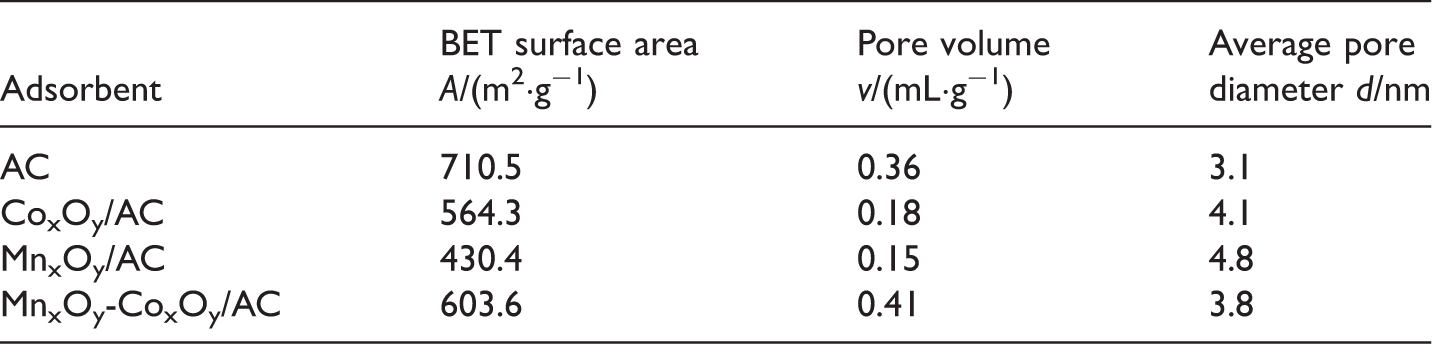
BET results
Pore properties of AC, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC.
Desulfurization performances of the adsorbents
All of the adsorbents were evaluated at room temperature (298 K) with a gas velocity of 20 mL/min using a fixed-bed plug flow reactor. The results are shown in Figure 4 and Table 3. For AC, the desulfurization performance was poor, with a breakthrough time of 20 min and a saturated adsorption time of 70 min. All of the modified adsorbents showed better desulfurization performances compared to that of AC. This was presumably because H2S is mainly physisorbed on AC, whereas it is both physisorbed and chemisorbed on the modified adsorbents. Compared to those of AC, the breakthrough time (115 min) and saturated adsorption time (185 min) for MnxOy-CoxOy/AC were increased five-fold and 2.5-fold, respectively.
Breakthrough curves of AC, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC absorbents. Desulfurization performance of AC, MnxOy/AC, CoxOy/AC, and MnxOy-CoxOy/AC.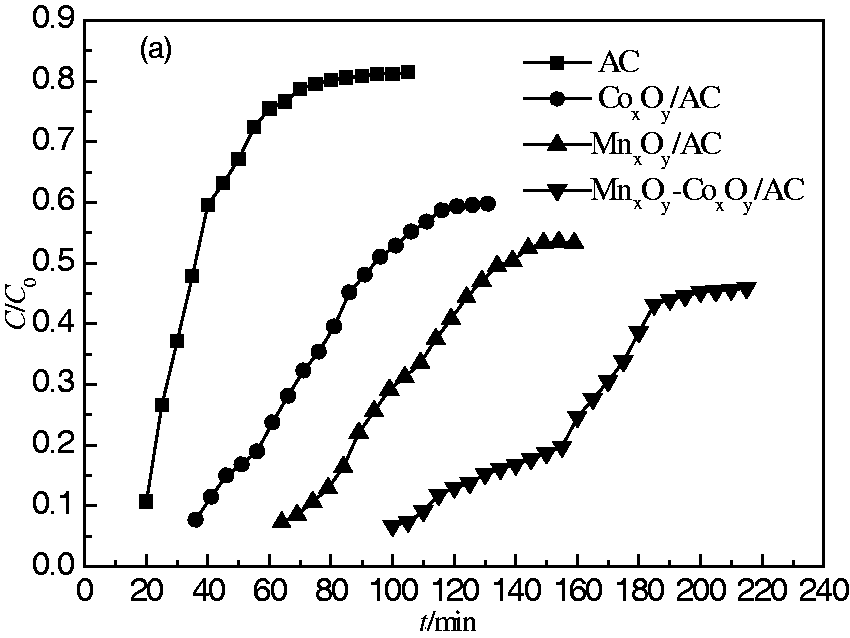

Table 3 shows the saturated sulfur capacities and desulfurization rates for the samples. For AC, the saturated sulfur capacity (37.7 mg/g) and desulfurization rate (25.1%) were poor. Compared to those of AC, the saturated sulfur capacity (347.7 mg/g) and desulfurization rate (74.2%) for MnxOy-CoxOy/AC were increased nine-fold and three-fold, respectively. The highest desulfurization performance of MnxOy-CoxOy may be attributed to the higher surface area (BET result) and greater degrees of crystallinity of MnO2 and Co3O4 (XRD results). The desulfurization performances of the various adsorbents decreased in the order: MnxOy-CoxOy/AC > MnxOy/AC > CoxOy/AC > AC.
Conclusions
Adsorbents consisting of MnxOy, CoxOy and MnxOy-CoxOy supported on activated carbon have been successfully prepared. The results showed that Mn was present in the forms of Mn3+ and Mn4+, and that Co was present in the forms of Co2+ and Co3+. The functional groups of activated carbon were well retained in all of the modified samples. The specific surface area of MnxOy-CoxOy/AC was evaluated as 603.6 m2/g. Compared with the original activated carbon, the specific surface areas of all of the modified samples were decreased, diminishing in the order: AC > MnxOy-CoxOy/AC > Co3O4/AC > MnxOy/AC. For MnxOy-CoxOy/AC, the contents of Mn and Co were 13.6% and 3.9%, respectively, and the mass ratios of Co2+/Co3+ and Mn3+/Mn4+ were increased compared with the monometal-modified AC samples, possibly because of interaction between the active components. The crystal regularities of MnxOy and CoxOy in MnxOy-CoxOy/AC were enhanced. The results of fixed-bed adsorption desulfurization experiments indicated that MnxOy-CoxOy/AC showed the best H2S desulfurization performance. The H2S removal capacities of the various adsorbents decreased in the order: MnxOy-CoxOy/AC > MnxOy/AC > CoxOy/AC > AC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



