Abstract
The primary goal of the eighth industrial fluid properties simulation challenge was to test the ability of molecular simulation methods to predict the adsorption of organic adsorbates in activated carbon materials. The challenge focused on the adsorption of perfluorohexane in the activated carbon standard BAM-P109. Entrants were challenged to predict the adsorption of perfluorohexane in the activated carbon at a temperature of 273 K and at relative pressures of 0.1, 0.3, and 0.6. The relative pressure (P/Po) is defined as that relative to the bulk saturation pressure predicted by the fluid model at a given temperature (273 K in this case). The predictions were judged by comparison to a set of experimentally determined values, which are published here for the first time and were not disclosed to the entrants prior to the challenge. Benchmark experimental studies, described herein, were also carried out and provided to entrants in order to aid in the development of new force fields and simulation methods to be employed in the challenge. These studies included argon, carbon dioxide, and water adsorption in the BAM-P109 activated carbon as well as X-ray diffraction, X-ray microtomography, photoelectron spectroscopy, and atomic emission spectroscopy studies of BAM-P109. Several concurrent studies were carried out for the BAM-P108 activated carbon. These are included in the current manuscript for comparison.
Introduction
As with the previous seven challenges (http://fluidproperties.org; Case et al., 2004, 2005, 2007, 2008, 2009, 2011; Ross et al., 2014), benchmark data were obtained or measured experimentally to enable the evaluation of entries in the eighth industrial fluid properties simulation challenge. To this end, adsorption isotherm measurements were performed for perfluorohexane in the activated carbon standard BAM-P109 (Panne and Thünemann, 2010) at the conditions specified in the simulation challenge: a temperature of 273 K and at relative pressures of 0.1, 0.3, and 0.6. The relative pressure (P/Po) is defined as that relative to the bulk saturation pressure predicted by the perfluorohexane model at a given temperature (273 K in this case).
Benchmark experimental studies were also carried out and provided to entrants to aid their development of new force fields and simulation methods to be employed in the challenge. These studies included argon, carbon dioxide, and water adsorption in the BAM-P109 activated carbon as well as X-ray diffraction, X-ray microtomography, photoelectron spectroscopy, and atomic emission spectroscopy studies on BAM-P109.
Perfluorohexane was chosen as the adsorbate for the challenge for several reasons. First, it is an example of a class of important commercial perfluorinated solvents. Second, it is computationally tractable to simulate. Third, it is also moderately challenging in terms of developing a force field (compared to simpler compounds such as hexane). Fourth, perfluorohexane is a compound with a potentially strong global warming impact. Finally, until the current publication, there were no previously published studies of the adsorption of perfluorohexane in activated carbons.
Materials and methods
Adsorption studies
The certified reference material BAM-P109 was selected for the benchmark experimental studies. The material consists of activated nanoporous compact spherical carbon beads with diameter ranging from 250 µm to 500 µm. Before running adsorption experiments, the material was degassed under vacuum at 200℃ for a period of 16 h. The temperature was raised slowly at a heating rate of 3℃/min.
High-resolution Ar (87.3 K) isotherm measurements were performed using a manometric (volumetric) technique, i.e. an Autosorb-IQ-MP sorption instrument (Quantachrome Instruments, Boynton Beach, FL) over a relative pressure range from 10−7 to 1. In addition to Ar, CO2 adsorption measurements were performed at 273 K, and water adsorption experiments at 293 K were performed using a dedicated manometric technique (Hydrosorb, Quantachrome).
Perfluorohexane adsorption measurements were performed at 273 K in a relative pressure range from 0.025 to 1 also by a manometric (volumetric) technique (Autosorb 1 Any gas unit).
X-ray diffraction studies
Samples of BAM-P108 and BAM-P109 were milled into fine powders and each sample applied to the surface of a silicon zero background specimen holder. Reflection geometry data were collected in the form of a survey scan by use of a PANalytical vertical diffractometer, copper Kα radiation, and Pixel detector registry of the scattered radiation. The diffractometer was fitted with variable incident beam slits and fixed diffracted beam slits. The survey scan was conducted from 5 to 80° (2θ) using a 0.04° step size and 12-s dwell time. X-ray generator settings of 40 kV and 40 mA were employed.
X-ray microtomography studies
Samples of BAM-P108 and BAM-P109 were placed directly into polymeric tubes of 3 mm diameter. X-ray microtomography data were collected using a Skyscan 1172 (Bruker Micro-CT, Kontich, Belgium) scanner at X-ray generator settings of 40 kV and 240 mA. The scanner employed a 10 megapixel Hammatsu detector operated at a 4 K by 2 K resolution and 16-bit gray scale depth. Projected image data were collected at a resolution of 3.0 microns per detector pixel as the sample was rotated about 180° at a step size of 0.30°. Each projected image was accumulated for 589 ms and 32 averaged images used per collected detector pane. Scanner data collection was accomplished using Skyscan 1172 control software (v1.5.9). The resulting 681 projected images were taken through reconstruction applying minor corrections to remove the effects of ring artifacts to produce a stack of 8-bit gray scale two-dimensional (2D) slice images along the sample data collection rotation axis. Reconstruction was accomplished using Skyscan NRecon (v1.6.5.8) software.
X-ray photoelectron spectroscopy studies
X-ray photoelectron spectroscopy (XPS) measurements were performed with a Physical Electronics Quantera II instrument. The BAM-P109 and BAM-P108 activated carbons were examined in two ways: (1) the powders obtained by crushing pellets were examined with a large area X-ray beam, and (2) the as-received surfaces of individual pellets were probed with a focused X-ray beam. For both methods, samples were secured to the sample holder with adhesive tape. An 85 W X-ray beam rastered over a 1400 µm × 500 µm analysis areas was used for the powder samples. A 100 µm 25 W X-ray beam was used for examination of individual pellets. Neutralization was achieved in both cases by a combination of low energy electron and Ar+ flood sources.
Atomic emission spectroscopy studies
Elemental analysis by inductively coupled plasma-atomic emission spectroscopy (ICP-AES) was performed on samples of BAM-P109 standard activated carbon and BAM-P108 standard activated carbon.
The samples were prepared in duplicate. Samples of 200 mg were weighed to the nearest 0.1 mg into acid-washed quartz beakers; 4 mL of concentrated sulfuric acid were then added to the sample beakers and to two empty control beakers. The beakers were covered with acid-washed quartz watch glasses and heated strongly on a hot plate for 2 h to char the samples; 4 mL of concentrated nitric acid were added dropwise to each heated beaker to complete the sample decomposition. Next, the beakers were partially uncovered to allow the excess acid to evaporate until a solution volume of approximately 0.5 mL was obtained. Deionized water of 10 mL was added, and the solutions were boiled gently for 15 min. Finally, the solutions were cooled, quantitatively transferred into polypropylene centrifuge tubes, and diluted to 25 mL with 18.2 MΩ deionized water.
The instrument used for elemental analysis was a Perkin Elmer Optima 4300DV ICP optical emission spectrophotometer. The samples were analyzed against external calibration curves generated using acid-matched solution standards containing 0, 0.2, 0.5, and 1 µg/g of each analyte. A 0.5 µg/g quality-control standard was used to monitor the accuracy of the calibration curves during the analysis.
A 0.5 µg/g scandium solution was run in-line with the samples and standards to serve as an internal standard. The elements quantified in this analysis were Al, Ca, Cd, Co, Cr, Cu, Fe, K, Li, Mg, Mn, Mo, Na, Ni, P, Sb, Sn, Sr, Ti, V, Zn, and Zr.
Results and discussion
Adsorption studies
A benchmark adsorption isotherm obtained by using argon at 87 K is shown in Figure 1(a) (linear) and (b) (semilogarithmic plot). The advantage of the semilogarithmic display is that this highlights the pore filling region. The tabulated data are included in Table A1 in Appendix A.
(a) Isotherm linear, Ar-87K BAM-P109. (b) Isotherm log scale, Ar-87K BAM-P109.
The adsorption isotherm can be considered as a combination of type I and type IV isotherms (Thommes et al., 2015), i.e. the hysteresis loop in the higher relative pressure range indicates the presence of larger mesopores (see Figure 2(a) and (b) that show the pore size distribution (PSD)).
(a) DFT pore size analysis from CO2-273K and Ar-87K BAM-P109. (b) DFT pore size distribution, Ar-87K BAM-P109.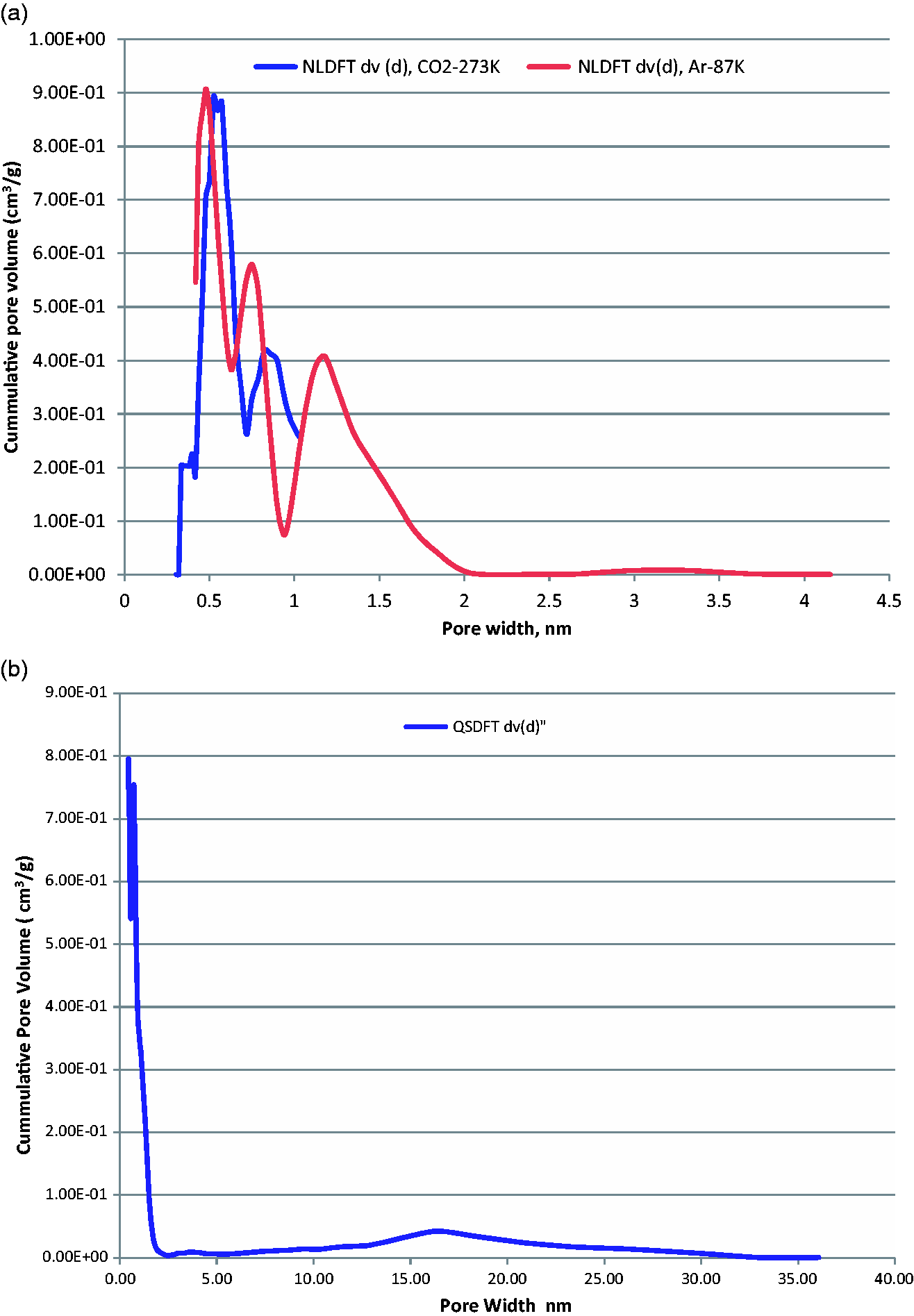
In addition to Ar adsorption at 87 K, CO2 adsorption experiments were carried out at 273 K up to a relative pressure of approximately 3 × 10−2. These data are shown in Figure 3, i.e. Figure 3(a) shows the linear display, whereas Figure 3(b) shows the data in a semilogarithmic scale. The tabulated data are included in Table A2 in Appendix A.
(a) Isotherm linear, CO2-273K BAM-P109. (b) Isotherm log scale, CO2-273K BAM-P109.
CO2 adsorption has become a standard tool for the assessment of microporous carbons with narrow micropores (pore width <0.7 nm) because it eliminates the well-known problem of diffusion limitations into narrow micropores associated with nitrogen and argon (Lowell et al., 2004; Rouquerol et al., 2014; Thommes and Cychosz, 2014; Thommes et al., 2012, 2015), i.e. CO2 molecules are able to easily access the ultramicropores despite the fact that the dimensions of N2, Ar, and CO2 are similar. One of the main advantages for performing CO2 adsorption experiments at 273 K, due to enhanced kinetics, is that a micropore characterization with CO2 can be performed in a few hours, which is much faster than adsorption experiments at cryogenic temperatures.
Non-Local Density-Functional Theory (NLDFT) pore size analysis of BAM-P109 as obtained from CO2 adsorption in the range of narrow micropores up to 1 nm is shown in Figure 2(a) together with a NLDFT PSD curve obtained from argon at 87 K, that extends up to 4 nm. In addition, Figure 2(b) shows the PSD again from argon at 87 K by applying Quenched-Solid Density-Functional Theory over a wide range of micro- and mesopores up to 40 nm. For all Density-Functional Theory calculations, a slit-pore model was used (Landers, 2013; Thommes et al., 2012, 2015). The PSD clearly shows well-pronounced microporosity but also the larger mesopores in the range between 15 and 20 nm.
Figure 4 shows an overlay of the water and argon adsorption isotherms at 293 K and 87 K, respectively. The tabulated data for the water isotherm are included in Table A3 in Appendix A. The shape of the water adsorption isotherm is completely different from that of the argon isotherm, and it can be considered a type V isotherm (Thommes et al., 2015). In the case of argon at 87 K, where the adsorbate completely wets the pore walls, the micropores are already completely filled at a relative pressure <0.1, micropore filling with water occurs at much higher relative pressures (>0.4) which is indicative of the hydrophobic nature of the carbon surface, i.e. up to a relative pressure of about 0.4, almost no adsorption was observed. Furthermore, adsorption/desorption of water into the carbon micropores leads to hysteresis, due to different mechanisms of adsorption/pore filling (via clustering) and desorption (via molecular evaporation), as discussed in the literature (see Thommes et al., 2012 and references therein). The shape of water adsorption data obtained on BAM-P109 demonstrates clearly the hydrophobicity of this carbon surface, which is consistent with the results of other techniques, e.g. the XPS data.
Isotherm overlay linear, Ar-87K & water 293 K.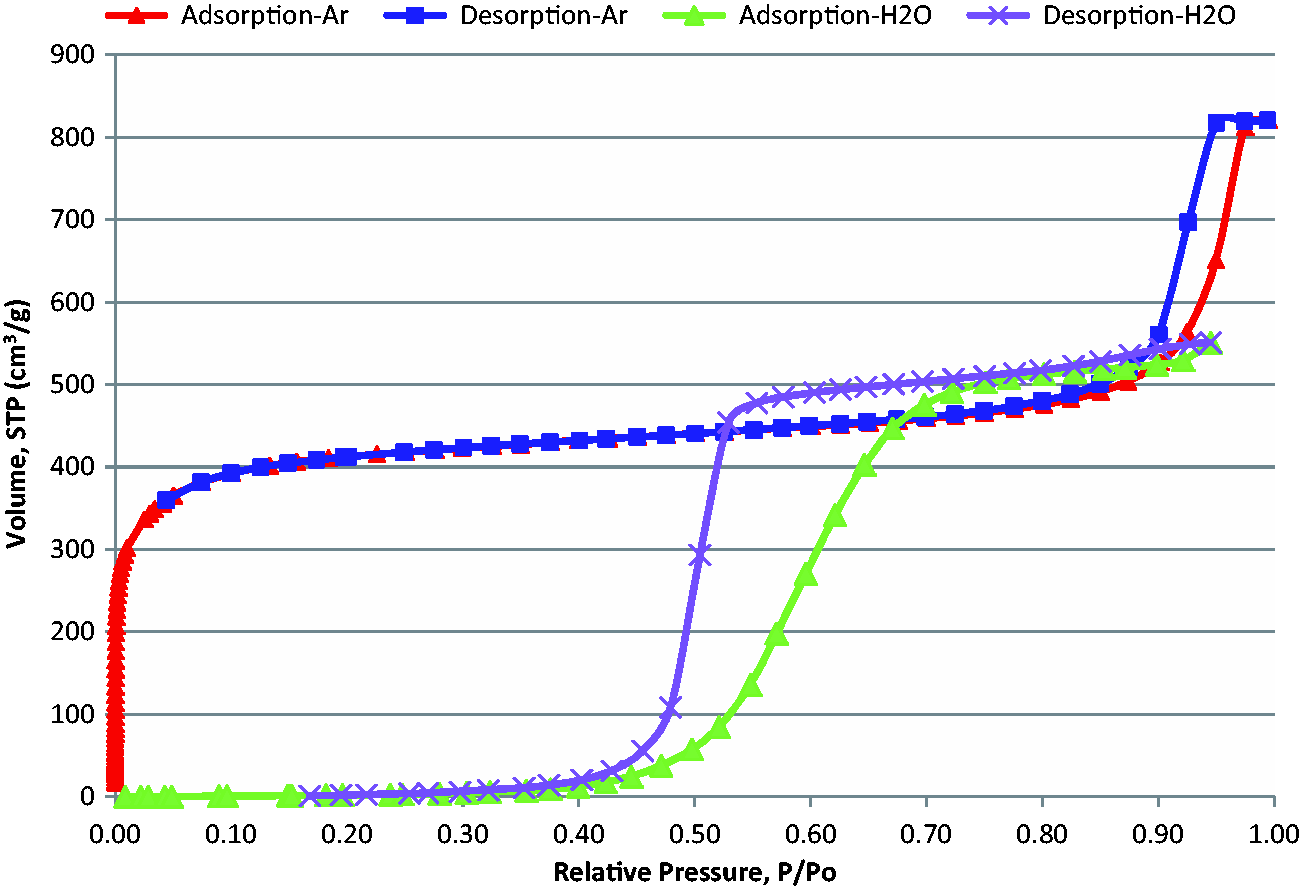
The adsorption isotherms for perfluorohexane on BAM-P109 at 273 K are shown in Figures 5 to 7. The tabulated data for the isotherms are included in Tables A4 and A5 in Appendix A. Figure 7, which shows an overlay of two adsorption experiments, demonstrates very good reproducibility of these experiments. The shape of the adsorption isotherm resembles that of argon at 87 K and can be considered to be a combination of a type I and type IV adsorption isotherm.
Isotherm linear, C6F14-273 K BAM-P109. Isotherm linear, C6F14-273 K BAM-P. Overlay-C6F14 adsorption isotherm 273 K.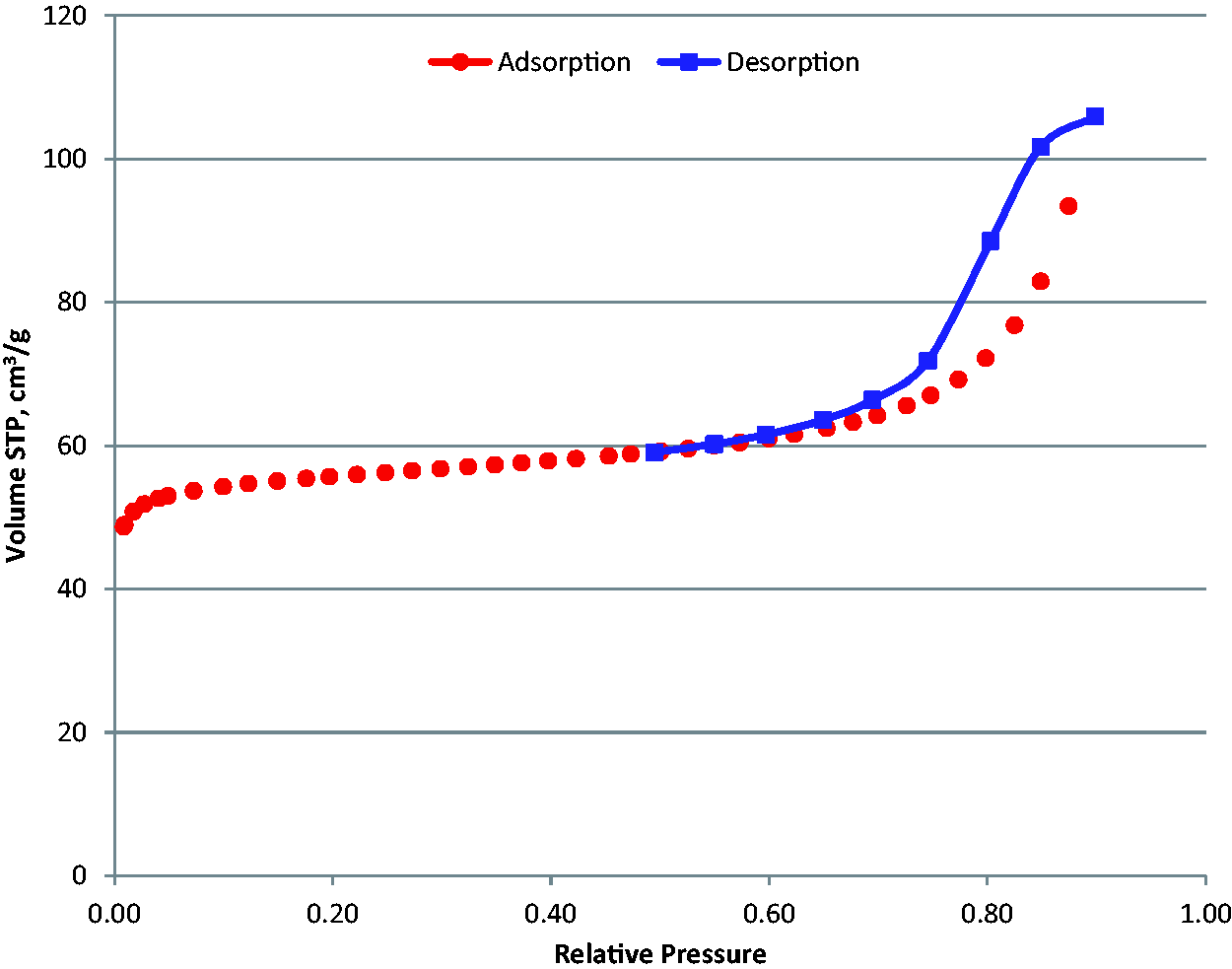


X-ray diffraction studies
The results of the X-ray diffraction studies for BAM-P109 and BAM-P108 are shown in Figure 8.
X-ray diffraction studies.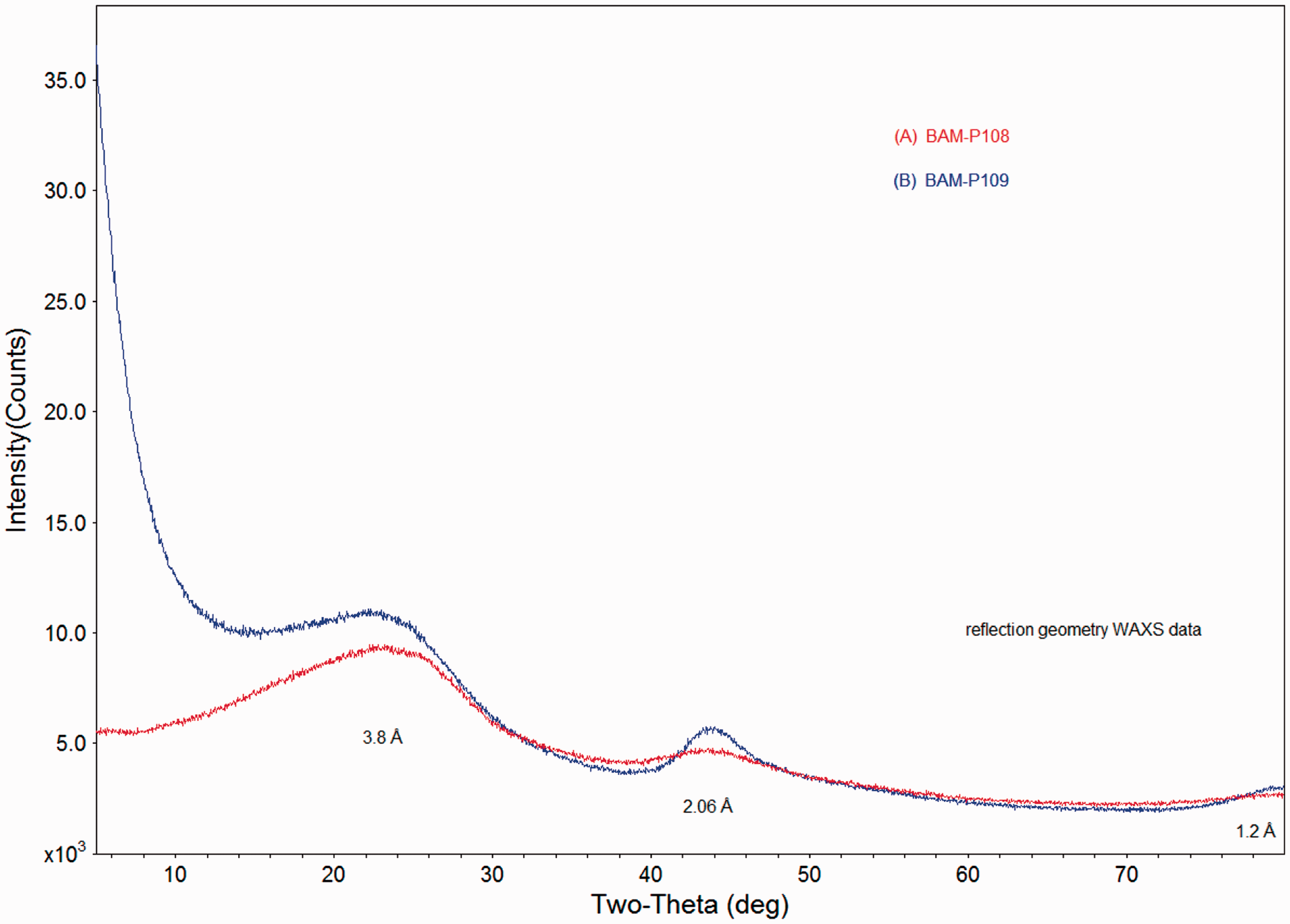
The X-ray diffraction data for BAM-P108 and BAM-P109 are dominated by broad diffuse scattering maxima occurring at 23, 43, and 79° (2θ) and corresponding to effective distances of 3.8, 2.06, and 1.2
X-ray microtomography studies
The results of X-ray microtomography studies are shown in Figures 9 to 14.
X-ray tomography of BAM-P108 (slice 476).
The 2D reconstructed images obtained from the X-ray microtomography data indicate that BAM-P108 and BAM-P109 are nominally spherical in shape. For many of the particles in the BAM-P108 and BAM-P109 samples, the material near the outer surface layer of the particle possesses a higher density than the interior of the particle. BAM-P108 contains a minority, but yet significant, population of particles, where the whole particle possesses a higher density than the average particle in the collection of particles. These higher density particles can be seen as a lighter color in the 2D slice images shown in Figures 9 to 11. By contrast, BAM-P109 contains a minority population of lower density particles. These lower density particles can be seen as a darker color in the slice images shown in Figures 12 to 14. Several of the particles in BAM-P109 are seen to contain fissures that connect the outer particle surface with the particle interior.
X-ray tomography of BAM-P108 (slice 1008). X-ray tomography of BAM-P108 (slice 1548). X-ray tomography of BAM-P109 (slice 569).


XPS studies
The results of the photoelectron studies for %S, %O, %C, and %Na are shown in Table 1 and Figures 15 to 18, respectively.
X-ray tomography of BAM-P109 (slice 1558). X-ray tomography of BAM-P109 (slice 1801). X-ray photoelectron spectroscopy studies for sulphur. X-ray photoelectron spectroscopy studies for oxygen. X-ray photoelectron spectroscopy studies for carbon. X-ray photoelectron spectroscopy studies for sodium. X-ray photoelectron spectroscopy studies. XPS: X-ray photoelectron spectroscopy.



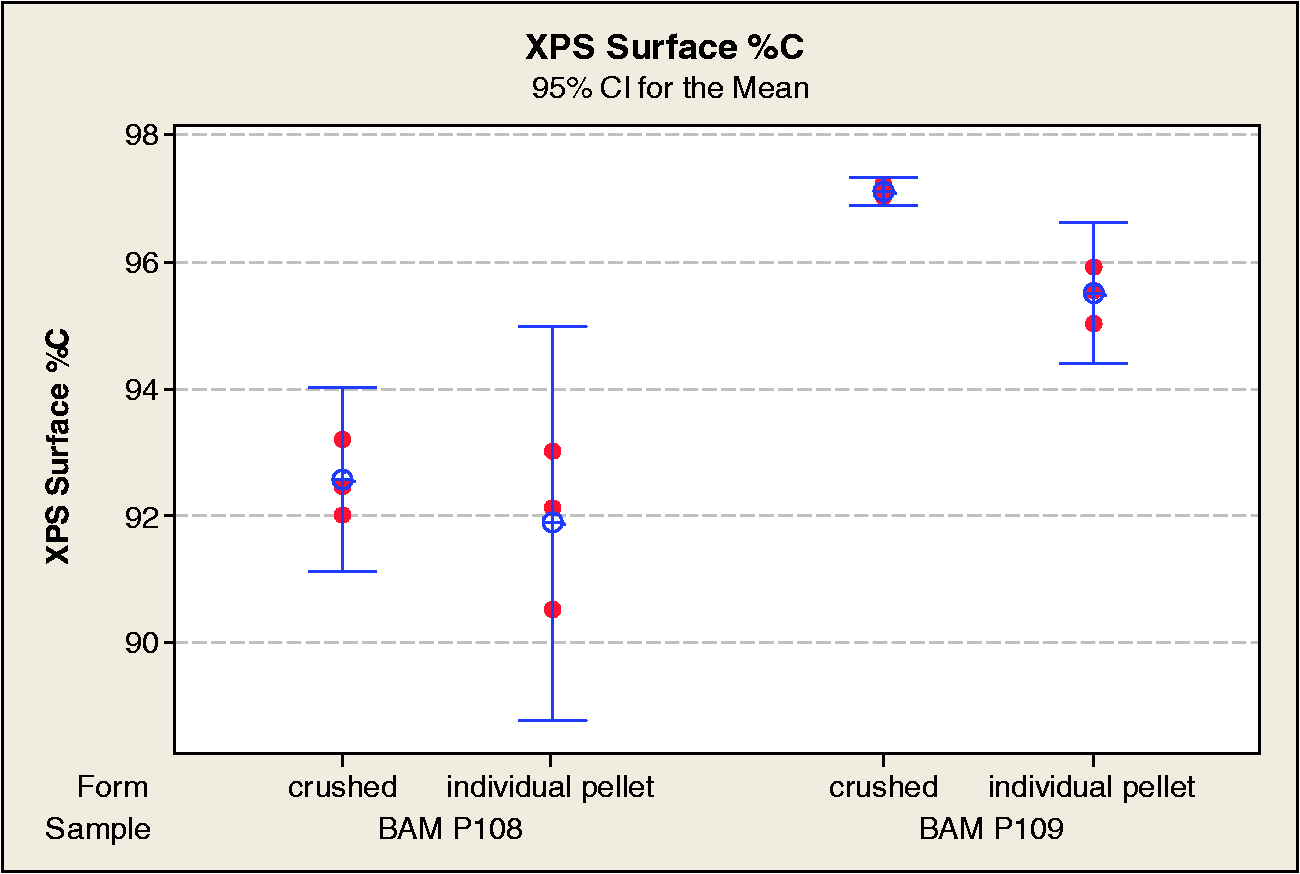
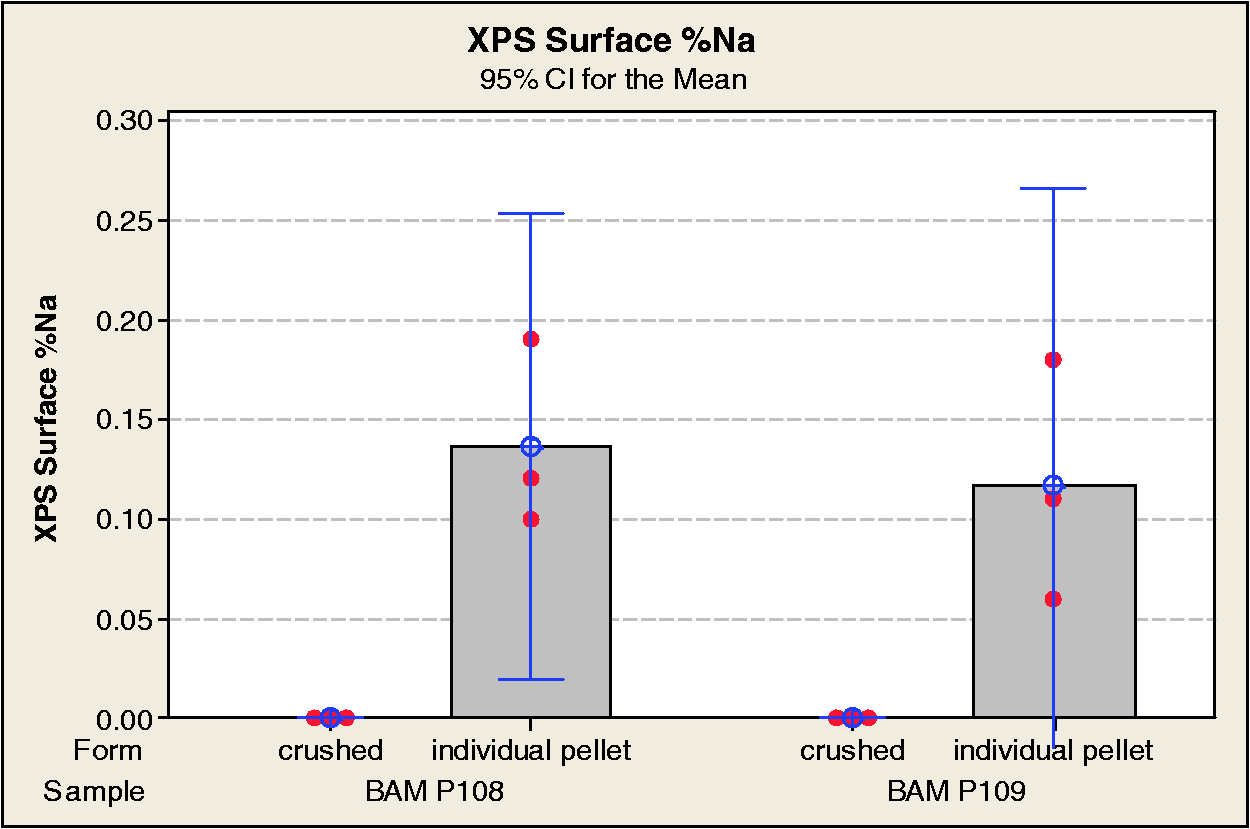

XPS surface compositions were similar for powder and individual pellet measurements, but the pellet surfaces were found to have low level Na present, and also had slightly higher O concentrations than found in the powdered samples. Both the BAM-P108 and BAM-P109 samples had surface compositions dominated by C (>90 atomic %) with low level O. The BAM-P108 was found to have ≈3 atomic % S, primarily in reduced form(s). Only a trace of S (≈0.2 atomic %) was detected on the BAM-P109.
Atomic emission spectroscopy studies
Elemental analysis of BAM-P109 and BAM-P108.

A variety of trace elements were detected by ICP-AES. Of the 22 trace elements determined, the most abundant were Na, Ca, and Fe.
Conclusions
Benchmark experimental studies have been carried out to aid the eighth industrial fluid properties simulation challenge entrants in the development of new force fields and simulation methods to be employed in the challenge. The studies included: Ar, CO2, and H2O adsorption studies in BAM-P109 activated carbon; and X-ray diffraction, XPS, and atomic emission spectroscopy studies of BAM-P109 activated carbon. The results of these studies were provided to entrants. Several concurrent studies for comparison were carried out for the BAM-P108 activated carbon (Panne and Thünemann, 2007).
Experimental adsorption studies of perfluorohexane in BAM-P109 activated carbon at a temperature of 273 K and at relative pressures of 0.1, 0.3, and 0.6 were also carried out in order to judge the challenge entry predictions. The resulting adsorption isotherm data confirmed perfluorohexane adsorbs considerably in the activated carbon and showed consistent behavior with regard to adsorption isotherm shapes and measured pore volume.
Footnotes
Associated Content
Isotherm data reported with this paper are included in the NIST/ARPA-E Database of Novel and Emerging Adsorbent Materials (NIST SRD-205), http://adsorbents.nist.gov, and may be accessed directly at ![]() .
.
Authors' note
Contribution of the National Institute of Standards and Technology, not subject to US copyright.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Certain commercially available items may be identified in this paper. This identification does not imply recommendation by NIST, nor does it imply that it is the best available for the purposes described.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
