Abstract
Use chitosan (CS) fiber and cellulose acetate (CA) fiber with different mixing ratios as raw materials, the kinetic and thermodynamic characteristics of the adsorption process of chitosan fiber/cellulose acetate fiber (CS/CA) ratio of 90:10 are explored, and the adsorption process conforms to the quasi-second-order kinetic equation. It can be fitted by Langmuir isotherm. The adsorption process is a spontaneous thermal reaction with both physical adsorption and chemical adsorption. The best adsorption time is 30 min, the time to reach adsorption equilibrium is 180 min, and the best adsorption pH is 5, the most suitable temperature is 35°C, and rising the temperature is conducive to the fibers adsorption. The materials before and after the CS/CA blended yarn adsorbed copper ions were characterized by XRD, DSC, FT-IR, and the adsorption mechanism was analyzed. In the orthogonal experiment, the copper ions concentration has the most obvious effect on the adsorption capacity, and the temperature has the most obvious effect on the adsorption efficiency. The optimal adsorption combination is that the mixing ratio of chitosan fiber/cellulose acetate (CS/CA) is 80:20, the pH is 5, the temperature is 35°C, and the copper ion concentration is 100 mg L−1. Comparing the adsorption performance of fibers with different mixing ratios, the adsorption performance of CS/CA ratio of 90:10 is the best. The filtration efficiency of the filter material, made of fibers with different mixing ratios, to copper ions is tested, and the filtration performance with a mixing ratio of 90:10 is the best, and the filtration efficiency is 64.33%.
Introduction
Metals are widely used in various industries, such as mining, energy, metallurgy, aviation, and electrical appliances. 1 Due to the non-degradability and toxicity of metals, they are easily soluble in aqueous environments. Heavy metal ions can bind to biological macromolecules, such as nucleic acids, proteins, and small metabolites in living organisms, and inhibit their activities. Heavy metals in sewage are easily transported and absorbed by passive plants, 2 so the discharge of wastewater containing metal ions in manufacturing is a serious hazard.3,4 During the textile dyeing process, wastewater containing heavy metal ions such as copper, nickel, and lead is generated. Therefore, wastewater containing heavy metals must be treated before being discharged to avoid pollution to the environment and harm to the human body.
Chitosan is widely used in the fields of food, medicine, and wastewater due to its excellent biocompatibility, biodegradability, non-toxicity, adsorption, and antibacterial properties. In the study of the adsorption of heavy metal ions, chitosan derivatives have excellent performance. Liu et al. 5 and Zhang et al. 6 Chemically modified chitosan to make its chitosan derivatives have better antibacterial effects on Escherichia coli. The cotton fabric treated with chitosan by Verma et al. 7 and Bae et al. 8 showed that the growth of Escherichia coli and Staphylococcus aureus were reduced by 97.2% and 98%, respectively. Cellulose acetate fiber is a strictly green environmentally friendly regenerated cellulose fiber, non-toxic, tasteless, soft and skin-friendly, and has certain antibacterial properties. Gao et al. 9 prepared a cellulose acetate/TiO2 film and its antibacterial rate against Escherichia coli was 55.7%. The anti-bacterial rates of reverse osmosis membranes made by Xu and Na 10 using a blend of graphene oxide and cellulose acetate against Escherichia coli and Staphylococcus aureus are 75.93% and 79.98%, respectively. The methods for removing metal ions from wastewater include ion exchange, chemical precipitation, and adsorption. The adsorption method is widely used for its good results. Zhuang et al. 11 made chitosan composite by the wet spinning method has good adsorption of cobalt ions. Liu et al. 12 prepared viscose fibers functionalized with chitosan that can adsorb gold and palladium well. Soylak et al. 13 have found that the acetate membrane filter can detect the removal of metal nickel ions, and the detection limit is 4.87 μg/L. Mokrani et al. 14 electrospinned the acetate cellulose solution, and then subjected to deacetylation and citric acid modification. The functionalized cellulose nanofibers were used as an environmentally friendly biosorbent, and the results showed that the adsorption rate of chromium is very high, and it is an emerging material for wastewater treatment.
The price of chitosan fiber is relatively high, and the price of acetate fiber is relatively low. Hence, the blending of chitosan fiber and acetate fiber can reduce the cost. The addition of acetate fiber can improve the spinnability of chitosan fiber; and the spinning solutions of both are under acidic conditions, which is beneficial to recycling and sustainability. In this paper, chitosan fiber and acetate fiber are used as experimental materials, and the effect of different mixing ratios of chitosan/acetate fiber with copper ions in the contact time, copper ion concentration, temperature, and pH are explored. We also do the kinetic and thermodynamic analysis in its adsorption process and test the antibacterial rate of fiber with different proportions and analyze the antibacterial performance. The chitosan fiber/acetate fiber yarn with different mixing ratios has been prepared and it was wind into a filter element to explore the effect of the filter element on the efficiency of copper ion filtration under different reaction conditions. It is expected to provide the basis for the application and expansion of chitosan/cellulose acetate in water treatment fields such as water purification or sewage treatment.
Experimental and methods
Materials
The cellulose acetate fiber was purchased from Nantong Cellulose Fiber Co., Ltd (China). The chitosan fiber was purchased from Tianjin Zhongsheng Co., Ltd (China). The average length of cellulose acetate fiber is 60 mm, and the linear density is 1.3 D. The average length of Chitosan fiber is 40 mm, and the linear density is 1.3 D.
Copper sulfate pentahydrate, ammonia water, ethyl Alcohol, ditiocarb sodium, sodium hydroxide solution, hydrogen chloride solution was purchased from Aladdin Reagent Co., Ltd (China). Escherichia coli, Staphylococcus aureus was purchased from Nanjing Lezhen Biotechnology Co., Ltd (China). Filter tube was purchased from Kunshan Huangda Plastic Products Co., Ltd (China).
The experimental instruments include UV spectrophotometer (model is SPECORD®210PLUS), ultra-clean table (model is ZHJH-C1109B), oscillator (model is ZWY-100H), constant temperature incubator (model is ZXSD-R1160), autoclaving Pan (model is BOXUN), and filter element winding machine.
Adsorption studies of copper ions
The ratios of CS and CA were set as 90:10, 80:20, 70:30, and the influence of contact time, initial concentration, temperature, pH, and other factors on the absorption of Cu2+ by the fiber was studied.
Stock solution of Cu2+ with concentration of 200 and 300 mg/L in a 250 mL Erlenmeyer flask were prepared 100 mL, adjusted pH = 7, added 0.05 g fiber, and placed it in a constant temperature shaker at 15°C to shake 5, 10, 30, 60, 90, 120, 150, 180, 210, 240 min.
Stock solution of Cu2+ with concentration of 100, 150, 200, 250, 300, 350 mg/L in a 250 mL Erlenmeyer flask were shaked for 180 min. Other conditions were the same as above.
Stock solution of Cu2+ with concentration of 200 mg/L in a 250 mL Erlenmeyer flask were placed in a constant temperature shaking shaker at 15°C, 25°C, 35°C, 45°C, 55°C, and 65°C. Other conditions were the same as above.
Stock solution of Cu2+ with concentration of 200 mg/L in a 250 mL Erlenmeyer flask were adjusted the pH to 1, 3, 5, and 7, and other conditions were the same as above.
Preparation of CS/CA blended yarn
Blended with the three fiber ratios from 2.2, set the yarn fineness to 780 tex, and spin into yarn, the twist coefficient is 260.
Filter experiment
The winding and filtering device of the filter element is shown in Figure 1.
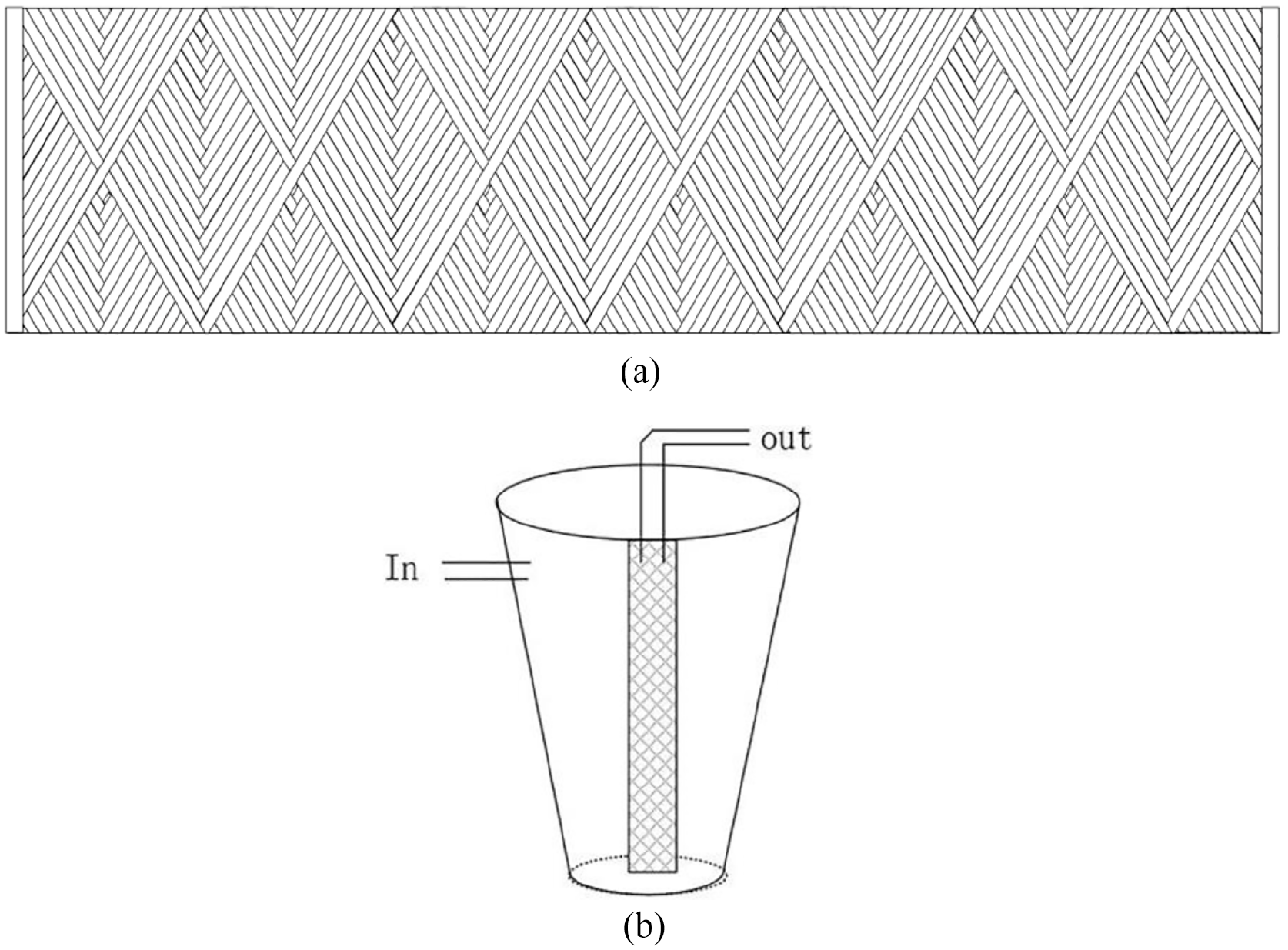
Schematic diagram of filtration: (a) cross-sectional view of filter element winding and (b) filtering device.
Stock solution of Cu2+ with concentration of 100, 150, 200, 250, 300, 350 mg/L were prepared 1000 mL, filtered it in a filter device, the solution flow rate was 200 L/h.
Stock solution of Cu2+ with concentration of 200 mg/L was adjusted pH to 1, 3, 5, and 7, other conditions were the same as above.
Stock solution of Cu2+ with concentration of 200 mg/L, filter element fiber ratios were 90:10, 80:20, 70:30, other conditions were the same as above.
Data analysis
Adsorption efficiency
The adsorption capacity and adsorption efficiency of the yarn to Cu2+ are calculated by formulas (1), (2), and (3) respectively.
Where: Qe – represents the equilibrium adsorption capacity of Cu2+ (mg g−1); Qt – represents Adsorption amount of Cu2+ at time t (mg g−1); Et – represents Adsorption efficiency at time t (%); V – represents Solution volume (mL); Co – represents the mass concentration of Cu2+ in the solution before adsorption (mg L−1); Ce – represents the mass concentration of Cu2+ in the solution at the time of adsorption equilibrium (mg L−1); Ct – represents the mass concentration of Cu2+ in the solution at time t (mg L−1); M – represents dry weight of yarn (g).
Adsorption kinetics, thermodynamics, isothermal fitting
According to the experimental results of the influencing factors of adsorption, the adsorption process was analyzed in kinetics and thermodynamics. Two kinetic equations of quasi-first-order and quasi-second-order were selected, and two adsorption isotherm models of Langmuir and Freundlich were fitted respectively. The relevant fitting equations are carried out with reference to the research of Wang. 1
Inhibition rate
Antibacterial calculation: The formula for calculating the concentration of viable bacteria is shown in (4):
Where: K – represents Concentration of viable bacteria in each flask (CFU/mL); Z – represents Average number of colonies on two plates; m – represents Dilution times.
The formula for calculating the antibacterial rate of the fabric to be tested is displayed in Formula (5):
Where: C is the antibacterial rate of the fabric to be tested; A – represents Concentration (CFU/mL) in the Erlenmeyer flask after shaking and contacting the control sample fabric; B – represents the concentration of the bacterial solution in the Erlenmeyer flask after the fabric is shaken and exposed to the bacterial solution (CFU/mL).
Results and discussion
The influence of experimental conditions on the adsorption of Cu2+ by CS/CA
The effect of the chitosan fiber/acetate fiber ratio of 90:10 and the reaction conditions on the adsorption of copper ions was explored.
Figure 2(a) shows that as the contact time increases, the fiber’s adsorption of Cu2+ increases, and the adsorption rate is faster in the first 30 min. Which means the adsorption amount reaches the maximum at 180 min, and the adsorption capacity of 180 min tends to balance. Cu2+ solutions with different concentrations of 200 and 300 mg/L showed the same adsorption trend.

The effect of reaction conditions on adsorption: (a) contact time, (b) pH, (c) temperature, and (d) the initial concentration.
Figure 2(b) displays that as the pH of the Cu2+ solution increases, the amount of adsorption of Cu2+ by the fiber increases first, and the adsorption amount of the fiber is the largest when the pH is 5, so the pH of 5 can be determined as the solution’s optimal adsorption pH. When the pH of the solution is higher than 7, the absorption of Cu2+ by the fiber begins to decrease. This is because as the concentration of H+ decreases, the adsorption sites can adsorb positively charged Cu2+. When the pH of the solution is higher than 7, the amount of adsorption decreases. This is because too much OH-surrounds the adsorption site, reducing the amount of adsorption.15,16
Figure 2(c) indicated that as the temperature increases, the amount of adsorption of Cu2+ by the fiber increases. After 35°C, the amount of adsorption slows down, and at 55°C, the amount of adsorption shows a constant trend. 35°C can be determined as the optimal adsorption temperature, and heating up will help the adsorption of fibers.
It can be seen from Figure 2(d) that as the initial concentration of Cu2+ increases, the adsorption capacity increases and the adsorption efficiency decreases. Finally, the adsorption will reach saturation and the actual saturated adsorption capacity is 539.6 mg/g.
Fitting analysis of adsorption model
Cu2+ adsorption kinetics
The adsorption kinetics with CS/CA ratio of 90:10 was studied.
The quasi-first-order kinetic equation usually represents physical adsorption, and the quasi-second-order kinetic model represents chemical adsorption. It can be seen from Figure 3 and combined Table 1, R22 > R12 shows that the adsorption of Cu2+ is more in line with the quasi-second-order kinetics. The adsorption in this process is chemical adsorption.
Kinetic model parameters.


Quasi-first-order and quasi-second-order kinetic equations.
Cu2+ adsorption isotherm
Cu2+ adsorption isotherm fitting analysis results and parameters are shown in Figure 4 and Table 2.
Fitting parameters of adsorption isotherms.

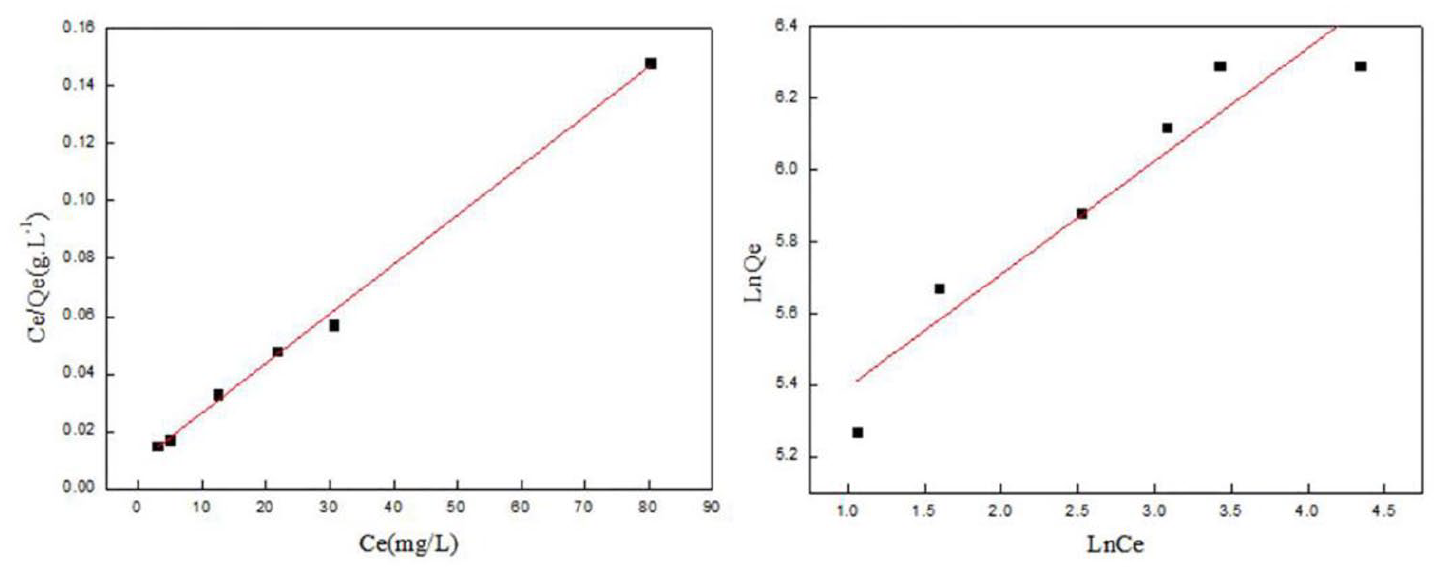
Adsorption isotherms.
From the parameter R12 > R22 in Figure 4 and Table 2, the results show that the adsorption of Cu2+ is more in line with the Langmuir model, and the adsorption process is multi-layer adsorption. At the temperature of 288 K, the maximum theoretical saturated adsorption capacity of Cu2+ is 588.24 mg g−1 for CS/CA, and because 0 < RL < 1, the adsorption process is favorable.17,18
Cu2+ adsorption thermodynamics
CS/CA fiber with different mixing ratios were studied the adsorption of copper ions, the effect of temperature change on adsorption performance was tested. The Calculation formulas are shown in (6), (7), (8):



Where T is the temperature; Kc is the thermodynamic equilibrium constant; ΔG0 is free energy (ΔG0, J/mol); ΔS0 entropy change (ΔS0, J/mol/K); ΔH0 is enthalpy change (ΔH0, J/mol); R is the general gas constant with a value of 8.314 J/mol/K.
With the ratio of CS/CA as 90:10, the Cu2+ adsorption kinetics was explored. The fitting results and parameters of Cu2+ adsorption thermodynamics are shown in Figure 5 and Table 3.
Cu2+ adsorption thermodynamic parameters.


Cu2+ thermodynamic adsorption model.
From Figure 5, the Cu2+ adsorption thermodynamic parameter equation can be obtained. The fitting parameter ΔS value in Table 3 is 35.719, indicating that when the CS/CA adsorbs Cu2+, the disorder and irregularity of the solid-solution interface increases. It seems that due to the CS/CA being wrapped together, the unevenness of the surface has increased. The value of ΔH is 210.15, which reflects the endothermic process of this adsorption. In the temperature range of 15°C–55°C, the value of ΔG is negative, indicating the spontaneity of the Cu2+ adsorption process. In addition, as the temperature increases, the negative value of ΔG continues to rise, which proves that the higher temperature is beneficial to the adsorption of copper ions. 19
Adsorption mechanism
XRD results
Figure 6 shows the XRD patterns of the blended yarn before and after adsorption. In some scholars’ investigations on the properties of cellulose acetate, the positions of the diffraction peaks of cellulose acetate were obtained at 2ϴ at 9.94° and 17.2° and the positions of the diffraction peaks of chitosan fiber at 2ϴ at about 10° and 20° as shown in Figure 6. It can be seen that the blended yarn of chitosan fiber and acetate fiber has a broad diffraction peak at about 15° to 30°, and after the blended yarn absorbs copper ions, the diffraction peak at about 15° to 30° completely disappears. The amorphous area increases, indicating that after the adsorption of the chitosan fiber and acetate fiber blended yarn with copper ions, the regularity of the three-dimensional structure of the molecular chain is weakened, and the intramolecular crystalline area is reduced, so that the copper ions are effectively adsorbed.
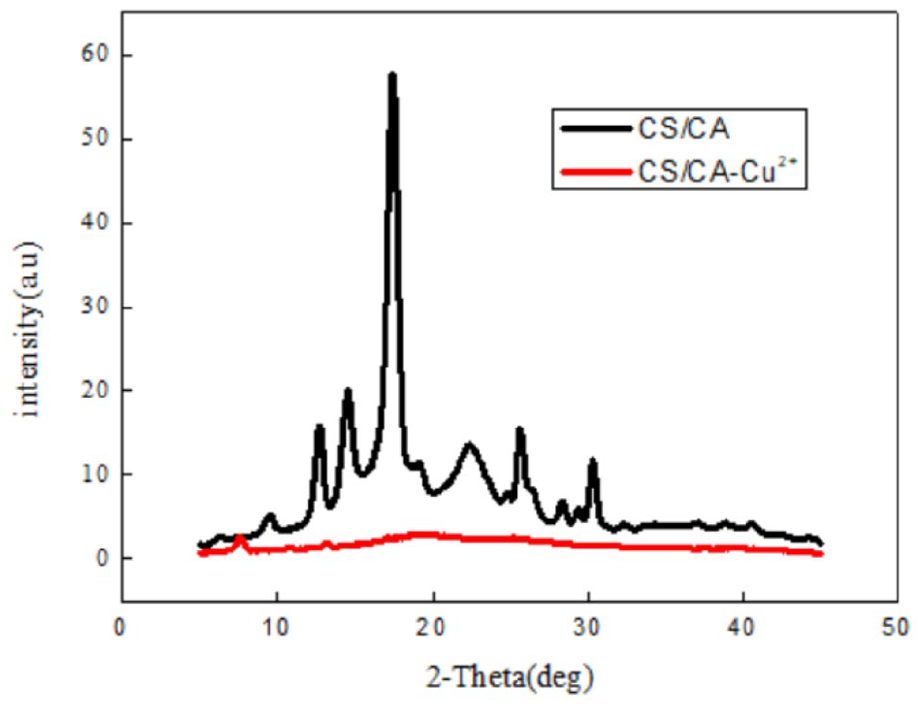
XRD of CS/CA before and after use for Cu2+ adsorption.
DSC pattern
DSC analysis was performed on the CS/CA fiber without adsorption of copper ions and the fiber after adsorption of copper ions, and the test results are shown in Figure 7. It can be seen that there is only one exothermic peak in the DSC curve of CS/CA, and the temperature is 74.6°C. When the CS/CA fiber adsorbed Cu2+, the exothermic peak temperature was 86.3°C. It is inferred that this may be due to the occurrence of thermal cracking reaction.

DSC test analysis.
FTIR pattern
Figure 8 shows that the infrared spectra of CS/CA fiber before and after the adsorption of copper ions are roughly similar, and the position of the characteristic absorption peak is basically unchanged. Compared with the adsorption of copper ions, the polarity of CS/CA is greater because after the formation of coordination bonds between -OH and Cu2+ in the CS fiber, the hydrogen bond between -OH is destroyed, and the Cu2+ steric effect is added, indicating that Cu2+ has been complexed with the CS fiber. It may also be caused by the -COOH of CA fiber.

FTIR of CS/CA before and after use for Cu2+ adsorption.
The broad absorption peak between 3250 and 3500 cm−1 in Figure 8 is a strong peak produced by the stretching vibration of -OH and -NH, and the peak area is obviously wider. Around 3300 cm−1 is the OH stretching vibration peak in the CS/CA fiber. Compared with the Cu-CS/CA fiber, the CS/CA fiber moves from 3291 to 3275 cm−1 in the low frequency direction, and the strength is weakened, indicating that Cu2+ has a new hydrogen bond effect with the -OH of the hydroxyl group, destroying the original Hydrogen bond association.
Inhibition rate
As shown in Table 4, they are the test results of chitosan/cellulose acetate against Escherichia coli and Staphylococcus aureus.
Antibacterial results of yarn.

It can be seen from Table 4 that the ratio of CS/CA is 90:10, the antibacterial rate against Escherichia coli is 97.1%, and the antibacterial rate against Staphylococcus aureus is 95.9%. As the proportion of cellulose acetate increases, inhibition rate reduced.
The performance of three proportions of fibers in adsorption of Cu2+
Performance comparison under different reaction conditions
The difference in the adsorption of copper ions to the fibers of three ratios under different conditions was explored. As shown in Figure 6.
It can be seen from Figure 9 that under different reaction conditions, the CS/CA ratio of 90:10 has the best adsorption performance for copper ions. As the ratio of acetate fiber increases, the adsorption capacity of copper ions becomes relatively poor. In Figure 9(a), it can be seen that as the contact time increases, the adsorption capacity of the three proportions of fibers increases. When the time is 180 min, the adsorption equilibrium is reached. In Figure 9(b) to (d), it is obvious that the adsorption of copper ions by the three proportions of fibers presents the same trend.

The different absorption of copper ions by fibers with different mixing ratios: (a) contact time, (b) pH, (c) temperature, and (d) the initial concentration.
Orthogonal design
Based on the orthogonal experiment design, the following four variables are analyzed: fiber ratio, Cu2+ solution concentration, temperature, pH, and the important influence of these four factors on the adsorption efficiency of copper ions is determined. 20 The best adsorption is that the ratio of CS/CA is 90:10, the pH is 5, the temperature is 35°C, and the concentration is 100 mg g−1. The highest adsorption efficiency of copper ions can reach 93.6%. The results of the orthogonal experiment are shown in Table 5.
Orthogonal experiment.

Range analysis
In the orthogonal experiment, make a range analysis of the three factors on the adsorption efficiency. The resulting parameters are shown in Table 6.
Adsorption efficiency range analysis.

According to the magnitude of the R value in the range analysis, the relationship between the factors can be determined. The primary and secondary factors that can be determined according to the R value are: (primary) C→A(B)→D (secondary), that is, C is the most important factor. The best combination of factors is A1B1C2D1.
In the orthogonal experiment, make a range analysis of the four factors on the adsorption capacity. The resulting parameters are shown in Table 7.
Adsorption efficiency range analysis.
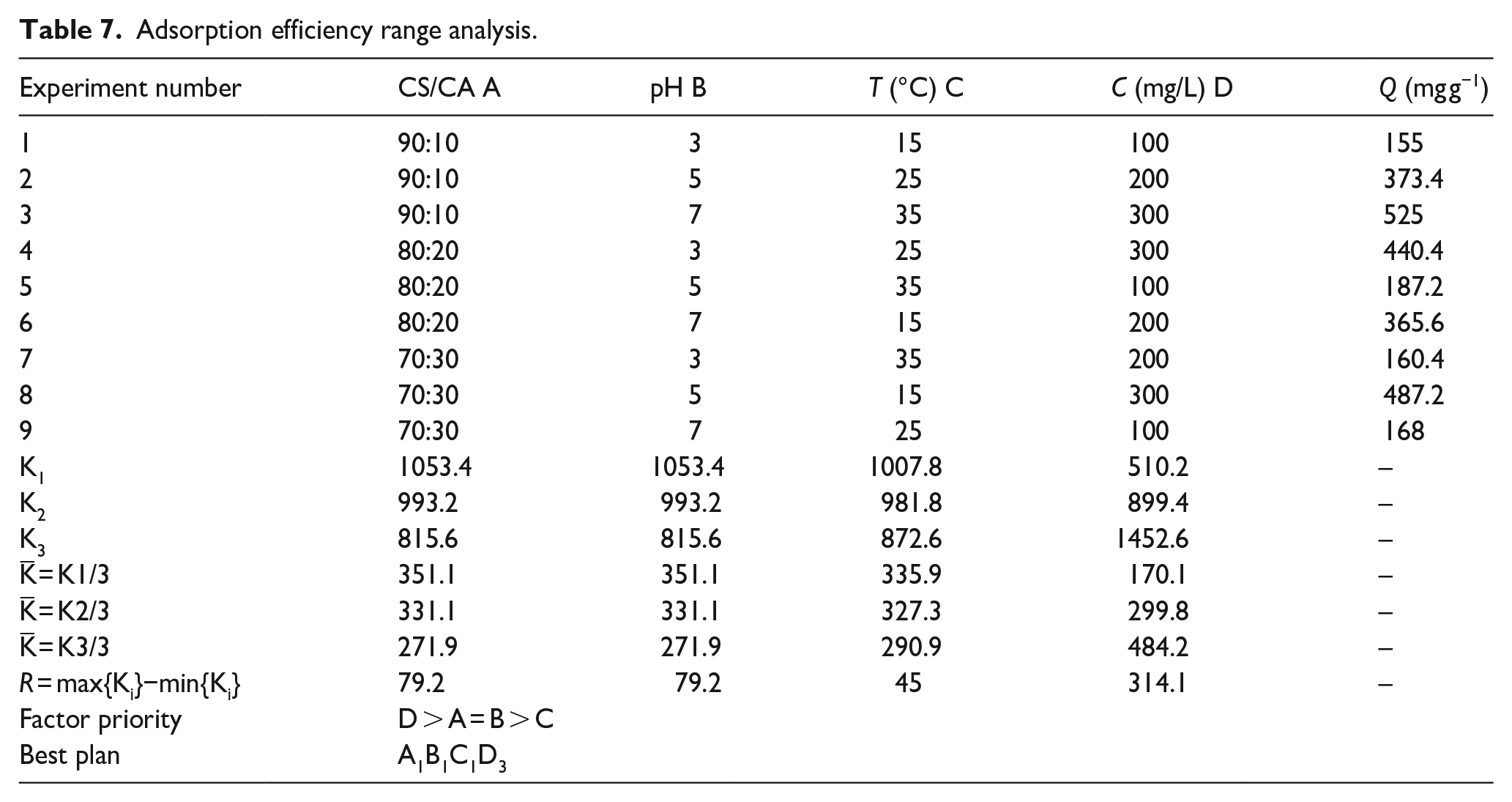
According to the magnitude of the R value in the range analysis, the relationship between the factors can be determined. The primary and secondary factors that can be determined according to the R value are: (primary) D→A(B)→C (secondary), that is, D is the most important factor. The best combination of factors is A1B1C1D3.
Filtration efficiency of the filter element to copper ions
The test results of filter elements made of three kinds of mixed ratio fibers under different reaction conditions are shown in Figure 10.
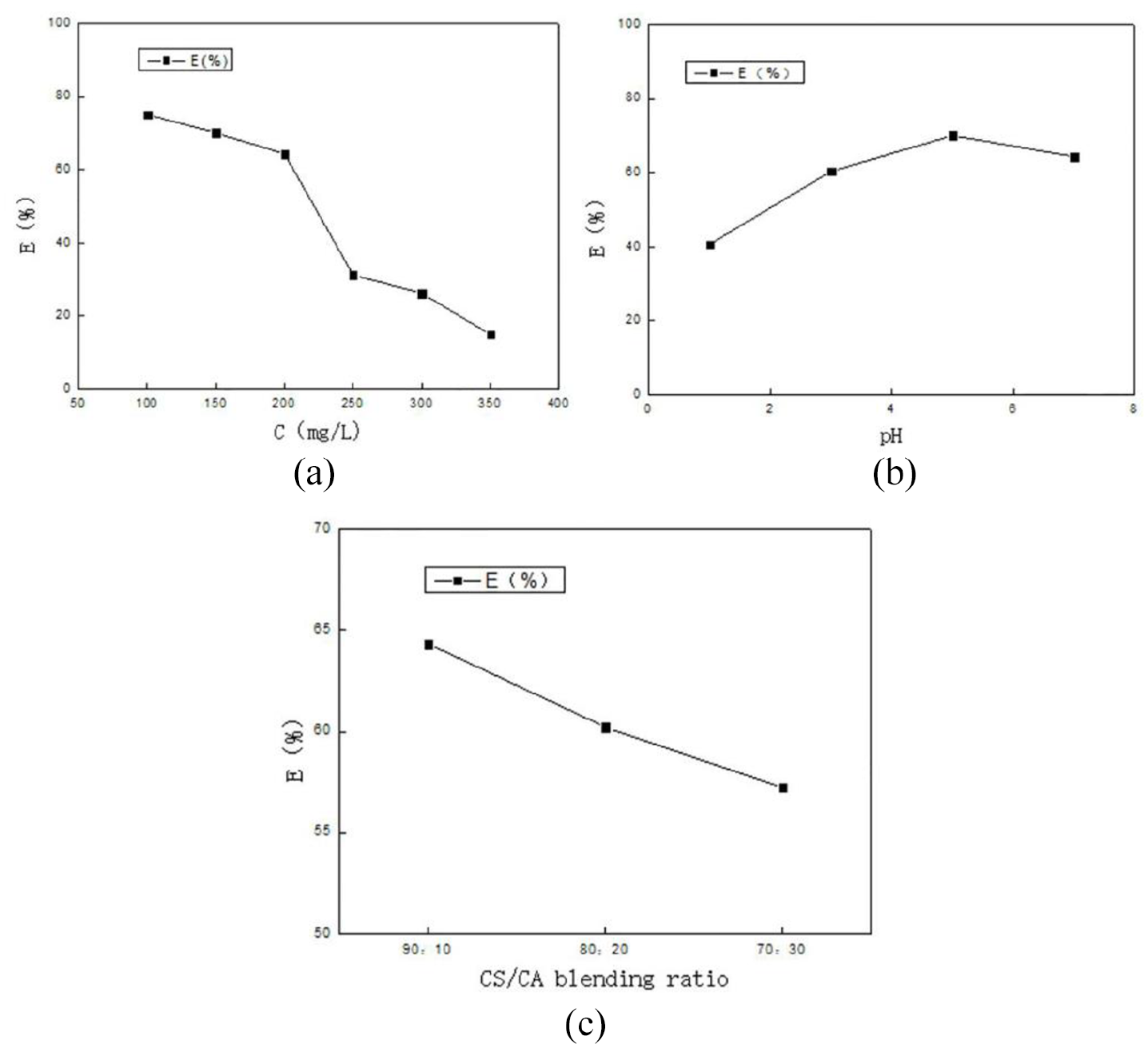
Filtering results: (a) the initial concentration, (b) pH, and (c) CS/CA blending ratio.
Figure 10(a) shows that as the initial concentration increases, the filter element’s filtration efficiency for copper ions decreases. This is because as the concentration of metal ions in the solution increases, the number of coordination bonds of chitosan fibers is fixed. The greater the concentration of metal ions, the less the number of coordination bonds, so the filtration efficiency will be reduced.
Figure 10(b) indicated that as the pH of the solution increases, the filtration efficiency of the filter element for the copper ion solution increases. When the pH of the solution is 5, the filter element has the best filtration efficiency for copper ions. In fiber adsorption, it is known that the concentration of per acid solution is not conducive to the adsorption of copper ions by chitosan fiber.
Figure 10(c) displays that, in the blended yarn, as the proportion of acetate fiber increases, the filtration efficiency of the filter element to the copper ion solution decreases. The filter element with a chitosan/cellulose acetate ratio of 90:10 has a filtration efficiency of 64.33% for copper ions and a fiber adsorption efficiency of 95.7%. It can be seen that as the contact time increases, it is beneficial to the fiber’s absorption of copper ions. When the filter element filters copper ions, the fiber’s contact time with copper ions is shorter, and the filtration efficiency is reduced.
Conclusions
CS yarn adsorbs Cu2+, the adsorption process conforms to the second-order kinetic equation, and electrochemical adsorption plays a leading role. The adsorption isotherm is more in line with the Langmuir model. The adsorption process is multi-layer adsorption, which is a self-heating reaction that combines physical adsorption and chemical adsorption. The adsorption type is effective adsorption. The best adsorption contact time is 30 min, the equilibrium adsorption time is 180 min, and the best adsorption pH is 5. In acidic solution, copper ions mainly exist in the form of divalent ions, so the amino group of chitosan fiber is beneficial to the adsorption of copper ions. The best adsorption temperature is 35°C, and the maximum saturated adsorption capacity can reach 539.6 mg/g.
Through orthogonal experiments, the optimal adsorption combination is that the ratio of CS/CA is 80:20, the pH is 5, the temperature is 35°C, and the copper ions concentration is 100 mg L−1. The range analysis shows that the copper ions concentration has the most obvious influence on the adsorption capacity, and the temperature has the most obvious influence on the adsorption efficiency. Comparing the adsorption of copper ions by three ratios of fibers, the adsorption performance of CS/CA ratio of 90:10 is the best, and the highest adsorption efficiency is 95.7%. The same adsorption trend is shown under different reaction conditions. The chitosan filter element made of fibers with three proportions has the same reaction for the filtration of copper ions and the absorption of copper ions by the fibers under different reaction conditions. The filtration performance with a fiber ratio of 90:10 is the best, and the maximum filtration efficiency is 64.33%. It can be seen that the contact time has the most obvious influence on the filtration efficiency.
Chitosan fiber can be dissolved in acetic acid. The spinning solution of chitosan fiber and acetate fiber are both under acidic conditions. It is an excellent green antibacterial material, which is beneficial to recycling and sustainability. Spinning into chitosan/acetate fiber blended yarn or other textiles, used in water purification and other water treatment fields, can be effective antibacterial and remove harmful metal ions. It is an environmentally friendly material that can be recycled continuously and has broad applications and prospects.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the National Youth Foundation of China (51303128) and the Natural Science Foundation of Tianjin (15JCZDJC38400).





