Abstract
In the present work, a new low-cost activated carbon was prepared from paper mill sludge in order to remove Cr(VI) ions from aqueous solution. The effects of adsorbent dosage, pH, contact time, metal ion concentrations, and temperature on adsorption efficiency were studied by experimental tests. The maximum equilibrium uptake of Cr(VI) by the adsorbent was 23.18 mg g−1 at optimum pH = 4.0, contact time of 180 min, and temperature of 45℃. Analysis of equilibrium adsorption data in terms of several isotherm models revealed that Langmuir isotherm with respect to Freundlich isotherm indicates better agreement with the experimental data. The kinetics of Cr(VI) adsorption onto activated carbon was described with the pseudo-second-order model which indicates the dominance of chemisorption mechanism. Thermodynamic parameters indicated that the Cr(VI) adsorption onto adsorbent was feasible in nature, spontaneous, and endothermic.
Introduction
Adsorption capacity of different low-cost materials for Cr(VI) removal from aqueous systems.

ADAS: Aerobically Digested Activated Sludge.
The literature provides few experimental data for preparation of adsorbent from paper mill sludge as a low-cost carbonaceous material. Herein, the prepared adsorbent was studied as a low-cost activated carbon for Cr(VI) removal from aqueous solutions. The isotherms, kinetics, and thermodynamics of Cr(VI) adsorption were studied to evaluate the effect of several parameters such as adsorbent dosage, pH, initial ion concentration, contact time, and temperature on the adsorption process. The Brunauer–Emmett–Teller (BET), Fourier transform infrared (FTIR), and SEM-energy dispersive X-ray (EDX) analyses were performed to assess the pores volume, chemical form, and surface morphology of the activated carbon, respectively. The equilibrium data have been analyzed using Langmuir and Freundlich isotherms. Moreover, the kinetics and thermodynamics of Cr(VI) adsorption were theoretically interpreted using different models.
Materials and methods
Materials
The paper mill sludge was collected from wastewater treatment plant of Mazandaran Wood and Paper Factory in Sari, Iran. All the primary reagents used in this study were of analytical grade purchased from Merck, Germany and used without further purification. A stock solution of 1000 mg l−1 Cr(VI) ions was made by dissolving 2.8289 g of K2Cr2O7 in distilled water. The working solutions of desired concentrations were made by suitable dilution of the stock solution with distilled water.
Synthesis of activated carbon
Proximate and ultimate analyses of the prepared activated carbon (results are expressed on a dry basis, except for the humidity).

Analysis
Characteristic of produced activated carbon.

Batch adsorption procedure
The batch adsorption experiments were carried out to investigate the influence of major affecting parameters like adsorbent dosage, pH, initial Cr(VI) concentration, contact time, and temperature on the amount of adsorption capacity. For each experimental run, a specified weight of prepared activated carbon was added to 0.1 l of the Cr(VI) solution in a 0.25 l Erlenmeyer flask. The contents in the flask were shaken in incubator shaker with KS 4000i control, IKA model at 180 r/min. During the study, the adsorbent dosage was varied from 1.5 to 7 g l−1, the pH from 2 to 6, the initial Cr(VI) concentration from 10 to 100 mg l−1, the temperature from 25 to 45℃, and the contact time from 0 to 240 min. The pH of solution was adjusted using either 0.1 N HCl or 0.1 N NaOH solutions to the required value. After shaking, the solutions were filtered through a Whatman No. 42 filter paper.
Analysis of Cr(VI) ions
The residual concentration of Cr(VI) ions in the sample was determined by UV–visible spectrophotometer with 2100 SERIES, UNICO model using 1,5 diphenyl-carbazide in acidic solution. The absorbance of the purple colored complex was measured at 540 nm wavelength after 10 min (Association et al., 1915).
The Cr(VI) ion removed by adsorbent was determined according to

The adsorption capacity of prepared activated carbon was calculated by the following equation

Batch desorption procedure
In our previous work (Gorzin and Ghoreyshi, 2013), it was seen that the weakly bonded Cr(VI) ions were removed at acidic or neutral pH, while NaOH solution demonstrated a good desorption efficiency. Hence, in this study the reversibility of Cr(VI) ions adsorption on activated carbon was investigated using NaOH (0.5 M) as desorption agent. Desorption experiments were conducted using the following procedures. At first, 0.01 l of Cr(VI) solution with initial Cr(VI) concentration of 50 mg l−1, pH 4.0, and 3.5 g l−1 of the adsorbent was shaken at 45℃ in a 0.01 l centrifuge tube. After 180 min, solution was centrifuged and the residual Cr(VI) content of the supernatant solution was measured. For desorption purpose, NaOH solution (0.5 N) was added to adsorbent and shaken at 45℃ for 180 min. Finally, the solution was centrifuged and the residual concentration of Cr(VI) ions in the solution was determined. Adsorbent was washed with distilled water until pH reached to neutral. Then Cr(VI) concentration (50 mg l−1, pH 4.0, and 3.5g l−1 of adsorbent) was added to adsorbent for second adsorption–desorption cycle and shaken for 180 min at 45℃. Then, the NaOH solution (0.5N) was added to adsorbent. This procedure was repeated several times and after each cycle, the reused adsorbent was washed with distilled water. Sample was collected after 180 min to evaluate Cr(VI) recovery by the following equation

Results and discussion
Surface characterization
SEM image and EDX spectrum of synthesized activated carbon before and after adsorption of Cr(VI) ions have been shown in Figure 1(a) and (b), respectively. As can be seen in Figure 1(a), the surface of prepared activated carbon has an irregular surface containing pores with different size and shapes. As shown in Figure 1(b), some pores have been closed due to adsorbent surface converge by adsorbate. By comparing the EDX spectrum of Cr(VI) unloaded and loaded adsorbent, it can be concluded that Cr(VI) is adsorbed onto the activated carbon surface.
SEM-EDX images of prepared adsorbent from paper mill sludge (a) before and (b) after adsorption of Cr(VI) ions. EDX: energy dispersive X-ray; SEM: scanning electron microscope.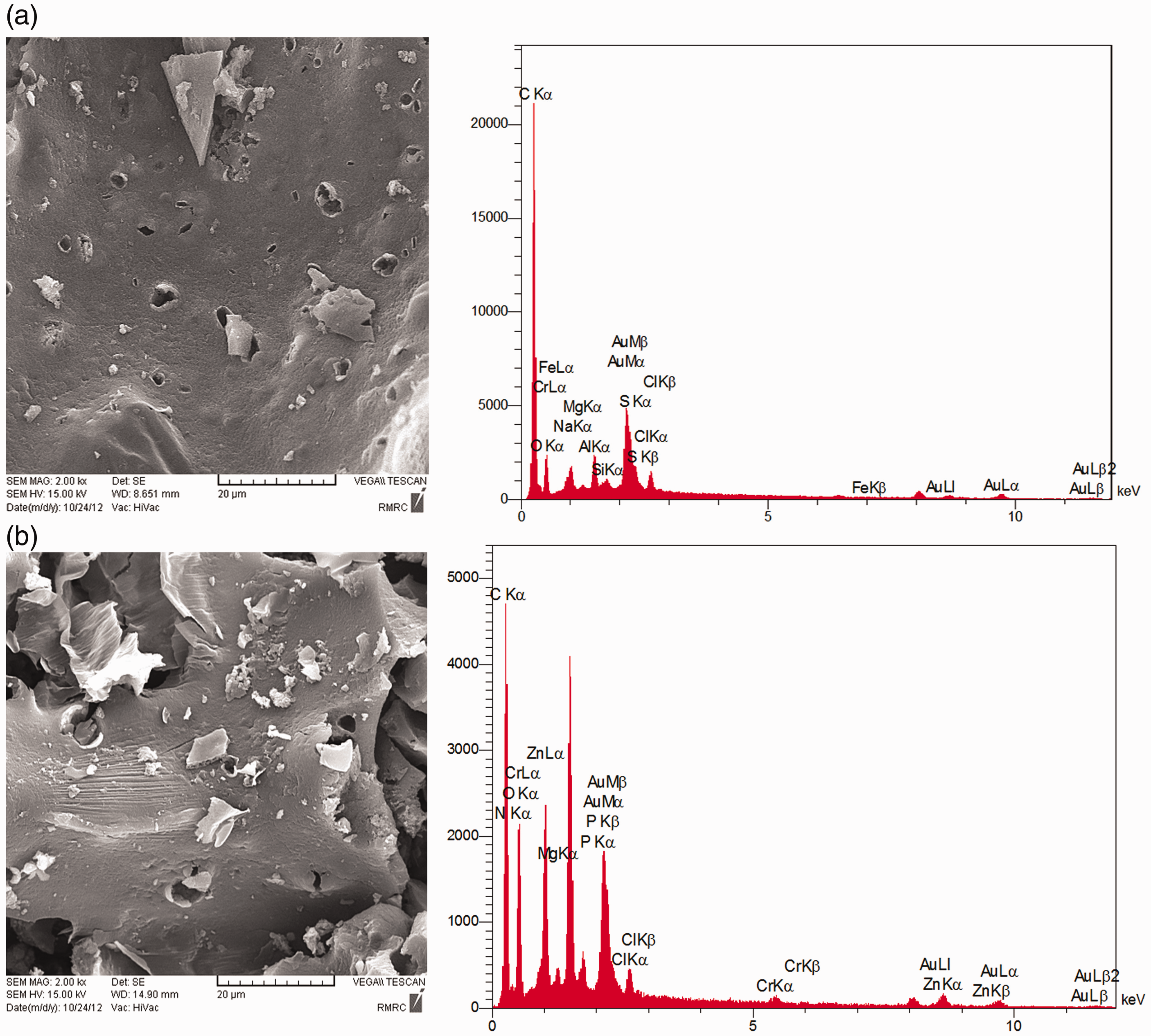
In this work, the FTIR spectra were obtained in order to analyze the mechanism of the Cr(VI) adsorption and identify functional groups on the surface of prepared activated carbon. Figure 2 shows the FTIR spectra of the adsorbent before and after Cr(VI) adsorption. The FTIR spectra bands indicate three major absorption bands including carboxyl, hydroxyl, and amino groups. The differences in the band intensity can be attributed to interaction of Cr(VI) ions with functional groups on the adsorbent surface. The broad absorption band at about 3456 cm−1 is attributed to the complexation between –OH groups which has been shifted to 3476 cm−1 after Cr(VI) adsorption (Gorzin and Ghoreyshi, 2013). The next shift observed at 1618 to 1612 cm−1 may be due to the complexation between Cr(VI) ions with carboxylic group (–C = O) (Levankumar et al., 2009). According to FTIR analysis reported in literature (Bansal et al., 2009; Giri et al., 2012), observed shift from 1004 to 1031 cm−1 is likely due to the interaction of nitrogen from amino group with Cr(VI). Doke and Khan (2012) used treated waste newspaper as a low-cost adsorbent. They revealed that the most abundant functional groups found on the prepared activated carbon include aromatic (C–H), carboxylic acid (C–O, C = O, and O–H), carbonyl (C = O), alkane (C–H), and amine (N–H, C–N). On the other hand, since the paper mill sludge has a significant amount of phosphorus (shown in EDX analysis), so this peak may be referred to the aliphatic P–O stretching (Dehghani et al., 2016).
FTIR spectra of the prepared adsorbent from paper mill sludge (a) without Cr(VI) adsorption and (b) with Cr(VI) adsorption. FTIR: Fourier transform infrared.
Adsorption results
Effect of adsorbent dosage
Adsorbent dose is a mainly important parameter in adsorption which determines the amount of removal as well as the economics of process. To examine the effect of adsorbent dosage on Cr(VI) adsorption, different amounts of activated carbon from 1.5 to 7 g l−1 were used at temperature of 35℃ and contact time of 240 min for fixed initial Cr(VI) concentration of 50 mg l−1 without pH adjustment. Experimental results in Figure 3 showed that the percentage of Cr(VI) removal increased from 47.68 to 76.32% when the adsorbent dosage increased from 1.5 to 7 g l−1. At higher dosage of adsorbent, more adsorption sites are available for Cr(VI) ions. As shown in Figure 3, the adsorption capacity was decreased from 17.63 to 5.52 mgg−1 when the adsorbent dosage increased from 1.5 to 7 g l−1. The decrease in the Cr(VI) adsorption capacity with increase in adsorbent dosage is due to more active sites of adsorbent remained unsaturated during the Cr(VI) adsorption process (Radnia et al., 2012). It is evident that after adsorbent dosage of 3.5 g l−1, extra increase in adsorbent dose did not appreciably change in Cr(VI) removal. Therefore, for all further experiments adsorbent dose was fixed at 3.5 g l−1 solution.
Effect of adsorbent dose on equilibrium adsorption capacity and removal efficiency (C0 = 50 mg l−1, T = 35℃).
Effect of pH
In this study, the effect of pH on adsorption capacity and removal efficiency of Cr(VI) ions by the prepared activated carbon was studied in the pH range of 2.0–6.0 using 0.35 g of adsorbent in 0.1 l of chromium solution with concentration of 50 mg l−1 at 35℃. Figure 4 shows that the removal percentage of Cr(VI) increases from 68.86 to 84.72% while pH increases from 2 to 4. Then, the removal percentage decreases to 79.21% with more increase in pH up to 6.0. The experimental results indicated that the maximum removal percentage of Cr(VI) by the synthesized activated carbon occurred at pH 4.0, and more increase in pH did not illustrate any considerable improvement in Cr(VI) removal efficiency and adsorption capacity. Therefore, the pH value of 4.0 was selected as the optimal pH for more Cr(VI) adsorption experiments to achieve suitable removal efficiency and Cr(VI) uptake capacity. At low pH (2.0–4.0), removal efficiency was increased due to high protonated adsorbent surface. The high protonated adsorbent surface makes a strong electrostatic attraction between oxy-anion (CrO42−, Cr2O72, etc.) and positively charged activated carbon surface (Gupta et al., 2010). The decrease in removal percentage of Cr(VI) with increase in pH (4.0–6.0) may be due to increase in OH− ions on the activated carbon surface which improves a repulsive force between negatively charged adsorbent surface and the oxy-anions of chromium (Selvaraj et al., 2003).
Effect of initial pH on equilibrium adsorption capacity and removal efficiency (C0 = 50 mg l −1, T = 35℃ and adsorbent dose = 3.5 g l −1).
Effect of contact time
The removal efficiency of Cr(VI) was investigated as a function of contact time at two initial concentrations, i.e. 50 and 100 mg l−1 and optimum condition of pH (4.0) and adsorbent dosage (3.5 g l−1). The experimental results were depicted in Figure 5. It is obvious that the removal percentage of Cr(VI) from aqueous solution increases with an increase in contact time and then decreases gradually till the equilibrium conditions after 180 min. At the initial period of adsorption, the rate of hexavalent chromium ions binding with activated carbon surface is great due to the availability of an abundance of adsorption sites, and then metal adsorption becomes slow. This is because of the diffusion of metal ions into the adsorbent pores by intraparticle diffusion (Kumar et al., 2008).
Effect of contact time on removal efficiency (C0 = 50 and 100 mg l−1, T = 45℃ and adsorbent dose = 3.5 g l−1).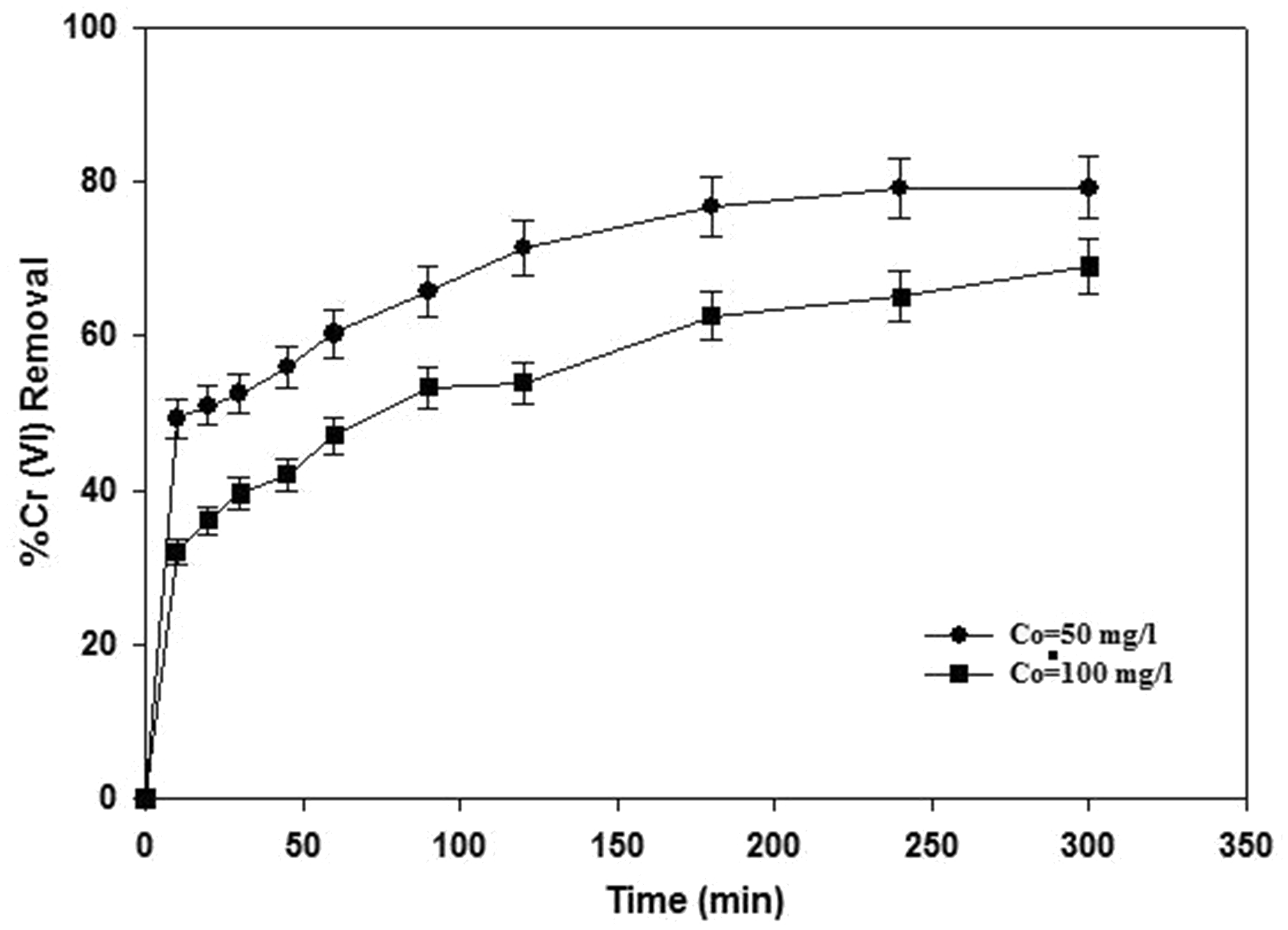
Effect of initial Cr(VI) concentration
Chromium solutions with several initial concentrations in the range of 10–100 mg l−1 at 45℃ and optimal pH 4.0 were equilibrated using 0.35 g adsorbent dose. Figure 6 indicates that the Cr(VI) ion equilibrium adsorption capacity increases from 2.82 to 23.18 mgg−1 and removal efficiency decreases from 98.4 to 76.74% when its initial concentration increases from 10 to 100 mg l−1. At higher initial concentrations, the active sites of prepared adsorbent would be surrounded with more Cr(VI) ions in the solution; hence the equilibrium adsorption capacity of activated carbon increases with increasing the Cr(VI) ion concentration which enhances the adsorption process (Ghorbani et al., 2008). In addition, the removal percentage decreases by an increase in metal initial concentration. At low initial concentrations, the ratio of initial number of chromium ions to the accessible active sites of adsorbent is low; therefore, the removal efficiency of Cr(VI) is higher and at higher concentrations, further residual Cr(VI) ions remain in the aqueous solution (Radnia et al., 2012).
Effect of initial concentration of Cr(VI) on equilibrium adsorption capacity and removal efficiency (C0 = 10–100 mg l−1, T = 45℃, pH = 4, and adsorbent dose = 3.5 g l −1).
Effect of temperature
Temperature had an obvious effect on the extent of hexavalent chromium adsorbed onto activated carbon. To investigate the effect of temperature on the removal percentage of Cr(VI), experiments were carried out at different temperatures, i.e. 25, 35, and 45℃ using optimum adsorbent dose (3.5 g l−1) with initial concentration of 100 mg l−1 and pH equal to 4.0. The effect of temperature on Cr(VI) removal can be concluded from Figure 7. The removal percentage of Cr(VI) from aqueous solution was 65.08% at 25℃ which increased to 76.73% at 45℃. The increase in removal efficiency with the increase in temperature shows that the Cr(VI) adsorption process is endothermic in nature.
Effect of temperature on removal efficiency (C0 = 100 mg l −1, pH = 4, and adsorbent dose = 3.5 g l −1).
Adsorption isotherm
Adsorption isotherms were used to investigate the relationship between the amount of Cr(VI) ions adsorbed onto the activated carbon surface and the concentration of chromium ions in the aqueous phase at equilibrium state. The experimental adsorption data for Cr(VI) ions have been analyzed by Langmuir and Freundlich isotherms in nonlinear form which have been shown in Figure 8.
(a) Langmuir and (b) Freundlich isotherm curves with experimental data at different temperatures (pH = 4, adsorbent dose = 3.5 g l−1).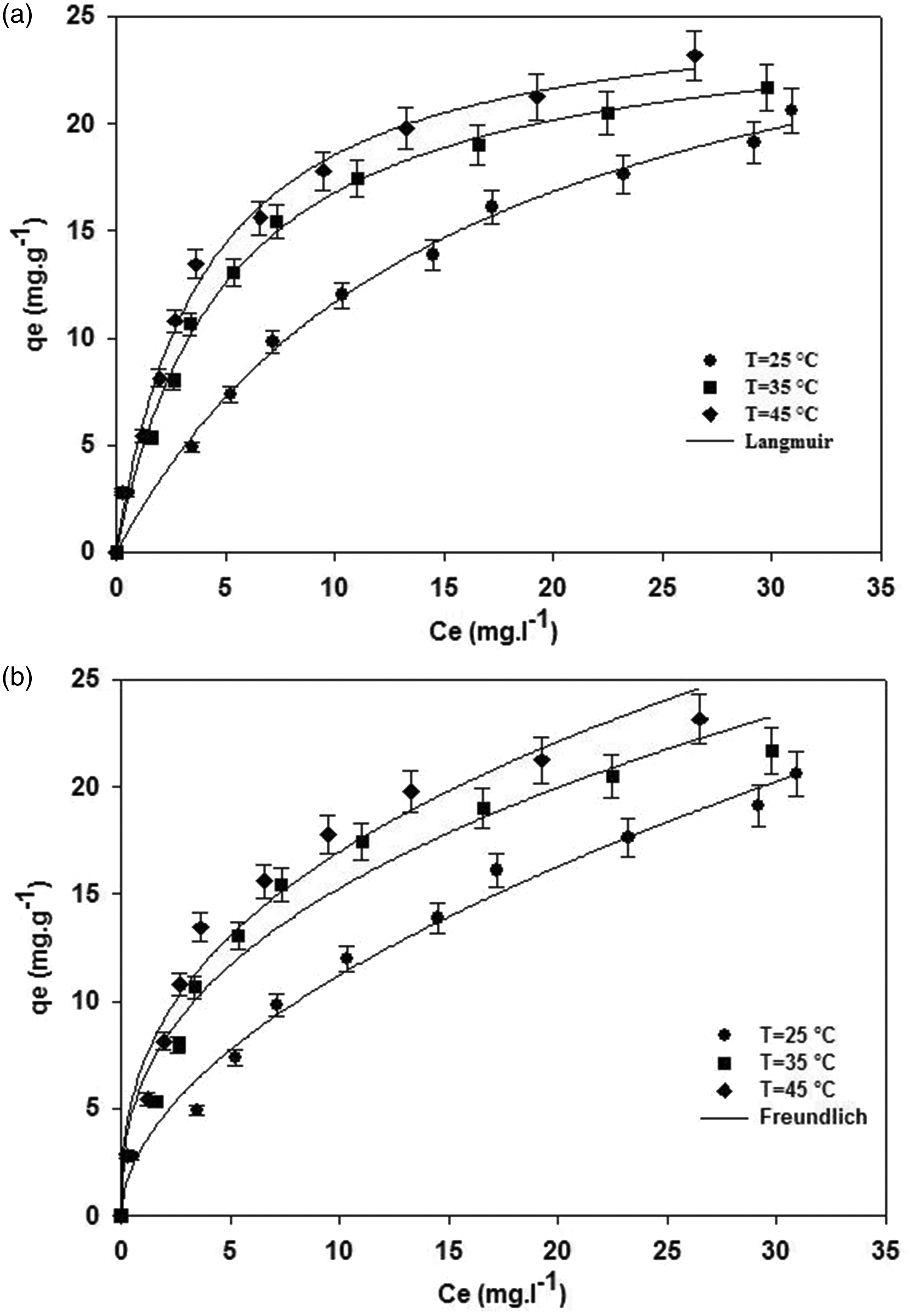
The Langmuir adsorption isotherm is based on the homogeneous surface by identical active sites and the monolayer adsorption of Cr(VI) ions onto the adsorbent surface which was described by Langmuir (1918)

The Freundlich isotherm model is applied based on multilayer adsorption on heterogeneous surface and may be expressed as (Freundlich, 1906)

Constants of Langmuir and Freundlich isotherms for Cr(VI) adsorption of the developed activated carbon.

Adsorption kinetics
Adsorption kinetics studies provide an understanding of adsorption rate and controlling mechanism of the process. In order to elucidate the adsorption kinetics of Cr(VI) ion by prepared adsorbent, the pseudo-first-order and the pseudo-second-order kinetics models were employed to test the experimental data.
Nonlinear forms of the pseudo-first-order (Singh and Tiwari, 1997) and the pseudo-second-order kinetic model (Ho and McKay, 1999) are represented by equations (6) and (7), respectively
(a) Pseudo-first-order and (b) pseudo-second-order kinetics models for Cr(VI) adsorption onto prepared adsorbent at different concentrations (pH = 4,T = 45℃, and adsorbent dose = 3.5 g l −1).


Pseudo-first-order and pseudo-second-order kinetic parameters of Cr(VI) adsorption onto adsorbent.

In this study, in order to determine the diffusion mechanism, intraparticle diffusion model was also used. Such plots may present a multilinearity, indicating that two or more steps are taking place. The rate-determining step of adsorption reaction may be external film diffusion, intraparticle diffusion, or interaction. The first, sharper step is the external surface adsorption or instantaneous adsorption stage. The second step is the gradual adsorption stage, where intraparticle diffusion is rate controlled. The third step is the final equilibrium stage where intraparticle diffusion starts to slow down due to extremely low adsorbate concentrations in the solution. In fact the metal ions are slowly transported via intraparticle diffusion into the particles and are finally retained in micropores (Özacar and Şengil, 2004). This model is presented as
Intraparticle diffusion kinetic model for the adsorption of Cr(VI) ions on activated carbon.

Intraparticle diffusion kinetic model parameters for Cr(VI) ions adsorption by activated carbon.

Adsorption thermodynamics
To investigate thermodynamic behavior of the Cr(VI) adsorption onto prepared activated carbon, thermodynamic parameters of Cr(VI) adsorption including Gibbs free energy change (ΔG0), enthalpy change (ΔH0), and entropy change (ΔS0) were determined using the following equations




Thermodynamic parameters for Cr(VI) ion adsorption onto the activated carbon at different temperatures.

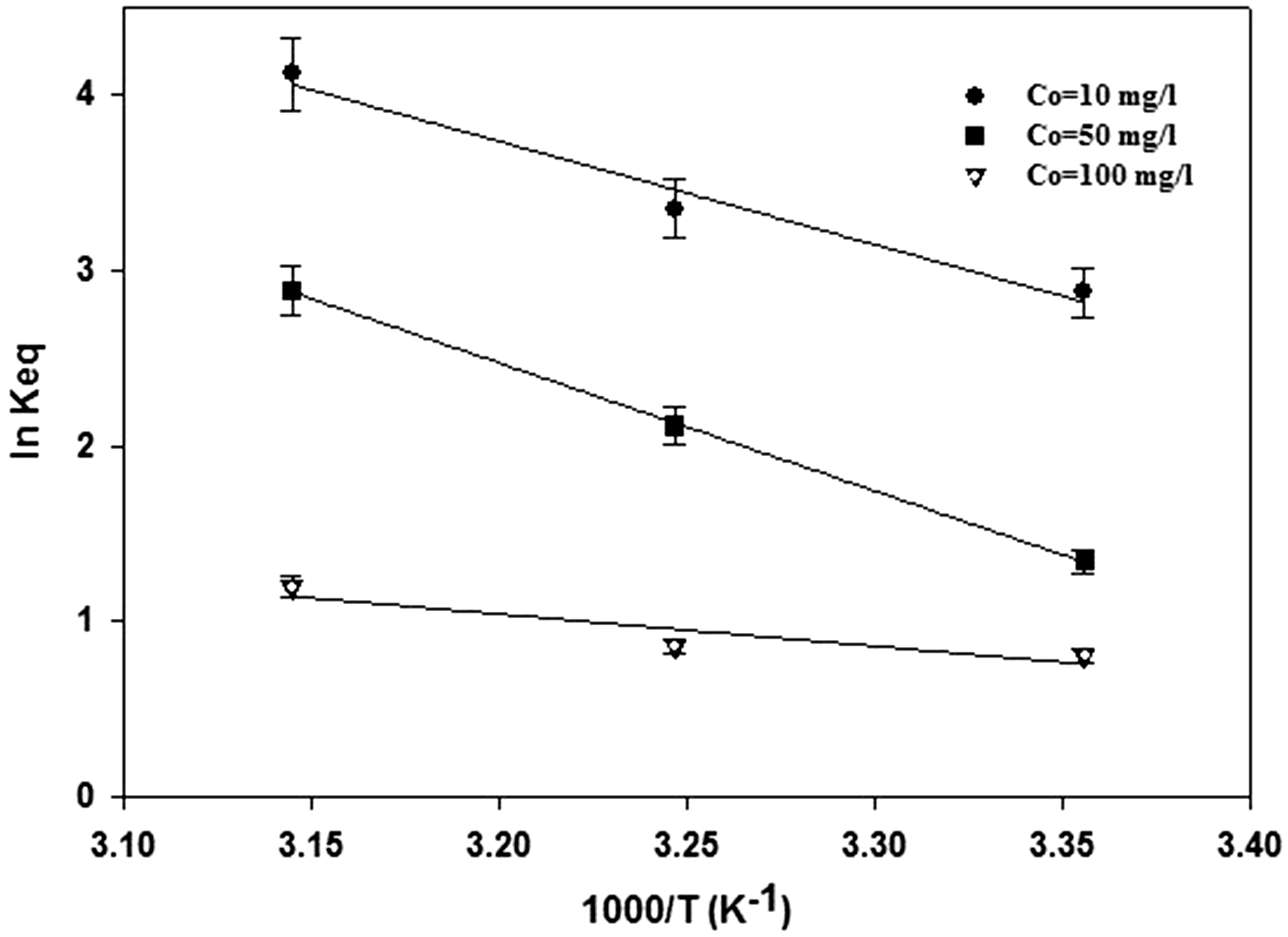
Plot of ln Keq versus 1000 T−1 for adsorption of Cr(VI) ions onto the prepared activated carbon.
Moreover, in order to provide an idea concerning the type of metal ion adsorption, Arrhenius activation energy (Ea) was determined by using linear plot of Arrhenius equation as follows

In physical adsorption, the activation energy is small due to the existence of weak forces between metal ions and adsorbent surface. In this case, the value of activation energy is commonly smaller than 4.184 kJ mol−1. In the current study, the value of activation energy (Ea) (46.7 kJ mol−1) is considerable and this event illustrates that chemisorption or reduction reaction has occurred (Duranoğlu et al., 2012; Dursun, 2006).
Desorption study
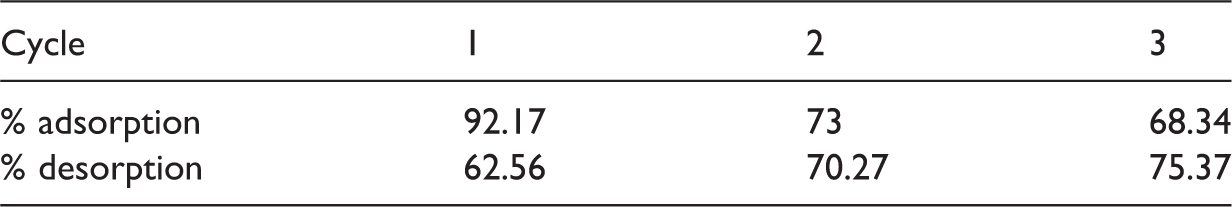
Adsorption and desorption efficiency of Cr(VI) ions after three cycles (50 mg l−1, pH 4.0, 45℃, and 3.5g l−1 of the adsorbent).
Conclusions
A low-cost activated carbon was derived from paper mill sludge by chemical activation with ZnCl2 which characterized and utilized for Cr(VI) removal from aqueous solution in a batch system. It was found that the adsorbent dosage, initial Cr(VI) concentration, pH of solution, temperature, and contact time influenced on the adsorption capacity of the prepared adsorbent significantly. The removal efficiency was increased with increase in temperature and decreased with increase in metal ion concentration. The maximum metal uptake capacity of prepared adsorbent was found to be 23.18 mg g−1 at optimum condition of adsorbent dosage of 3.5 g l−1, pH 4.0, initial Cr(VI) concentration of 100 mg l−1, contact time of 180 min, and temperature of 45℃. The equilibrium adsorption capacity was tested by using Langmuir and Freundlich isotherm models. The Langmuir isotherm indicated better fit compared to Freundlich isotherm model. The Cr(VI) adsorption onto prepared activated carbon followed nonlinear form of pseudo-second-order kinetic model that consists of chemisorption. Moreover, high activation energy (Ea) indicated the chemical interaction among Cr(VI) ions and surface groups on prepared adsorbent. The negative value of ΔG0 confirmed the feasibility of prepared adsorbent and spontaneity of the Cr(VI) adsorption process. The positive value of ΔH0 and ΔS0 indicated endothermic and irreversibility of Cr(VI) adsorption. The results of the present study showed that the prepared adsorbent from paper mill sludge could be appraised as potential adsorbent in order to remove Cr(VI) ions from aqueous solutions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
