Abstract
The production of low-cost biologically activated carbons (BACs) is urgent need of environmental protection and ecological sustainability. Hence, walnut shells were treated by traditional pyrolysis, direct KOH impregnation and combined activation composed of hydrothermal carbonization and two-step H3PO4- and pyrolysis-activation process to obtain porous carbon with high adsorption capacity. It was found that the best adsorption capacity for iodine and organic dye methylene blue (MB) can be obtained using the KOH impregnation at impregnation ratio of 1:1 or combined activation comprising of 2 h H3PO4 activation and 1 h pyrolysis activation at 1000°C. The produced KOH, H3PO4/pyrolysis activated BACs at the optimum conditions are superior to that of commercial ACs, 9.4 and 1.3 times for MB removal, 4 and 4.5 times for iodine number respectively. Characterization results demonstrated their porous structure with very good textural properties such as high BET surface area (1689.1 m2/g, 1545.3 m2/g) and high total pore volume (0.94 cm3/g, 0.96 cm3/g). The N2 adsorption-desorption isotherm of H3PO4/pyrolysis activated hydrochar suggested the co-existence of micro and meso-pores. Moreover, they are more effective for the removal of Fe(III) and Cr(VI) from aqueous solution than the commercial AC, suggesting a promising application in the field of water treatment.
Introduction
Biologically activated carbons are porous biomass carbon material with high aromatization. Biologically activated carbons are promising carbonaceous adsorbent in the field of ecological restoration, agriculture and environmental protection, due to specific surface area, plenty of surface functional groups, good electrochemical performance and environmental friendliness (Gamal et al., 2018; Sawana et al., 2017; Wang et al., 2016; Korottagamage and Sathasivan, 2017). Therefore, the research and application of low-cost BAC is urge for meeting the requirement of the application fields and have attracted much attention.
Specific surface area, pore size and the surface functional groups are critical factors concerning adsorption capacity of activated carbons (AC). Widespread evidence indicates that the adsorption properties of AC depend on raw materials and the preparation conditions, directly relative to their effectiveness in the applications. So far, the procedures of porous biomass carbon synthesis basically divided into pyrolysis, hydrothermal carbonization, and activation including gas activating, and chemical activation involving activating reagents such as phosphoric acid, zinc chloride, and alkali hydroxides (Arami-Niya et al., 2016; Gao et al.,2018; Spagnoli et al., 2017). Traditional pyrolysis requires high temperature (>500°C) and post-treatment of tar byproducts, while hydrothermal carbonization has the advantages of low energy consumption (temperature <300°C) and abundant surface functional groups but produced AC appeared low carbonization degree and aromatization degree (Kambo & Dutta, 2015; Wang et al., 2018). Thus, activation is necessary to the char’s porosity and enlarge the surface area of the activated carbon. Zhang et al. (2019) have developed a H3PO4 activated hydrochar derived from hickory wood and peanut hull and found that chemical modification greatly enhanced the absorption capacity of the hydrochar to acetone and cyclohexane (Zhang et al., 2019). Antero et al. (2019) reported that physical activation can improve the ability of Brazilian Cerrado hydrochar to remove Bisphenol-A from aqueous solutions. Sequence pyrolysis had been also reported to endow pyro-hydrochar with high aromatization degree and strong chemical oxidation stability (Liu et al., 2018). Thus, chemical modified pyro-hydrochar is expected to combine the characteristics of rich functional groups from hydrochar, rich-pore structure from chemical activation and stable carbon from pyrolysis, resulting in good chemical stability and adsorption performance.
Agricultural biomass waste has been becoming a main source of porous BAC for environmental protection and ecological sustainability (Spagnoli et al., 2017; Wong et al., 2018). varieties of biomass materials such as walnut shell, coconut shell, chitosan flakes and rice husks, are employed to the porous carbon materials because of the carbohydrate structure their abundant renewability (González-García, 2018; Rashidi & Yusup, 2017). Walnut shell has been reported to successfully produce into AC in the methods of pyrolysis (Ghasemi et al., 2015), steam activation (González et al., 2009) or chemical activation including potassium carbonate (Alimohammadi et al., 2016; Nazari et al., 2016; Xu et al., 2017), zinc chloride (Ghasemi et al. 2015), potassium hydroxide (KOH) (Liu et al., 2017; Qiu et al., 2017; Yu et al., 2014) and phosphoric acid (H3PO4) (Yang and Qiu, 2010), hydrogen peroxide (Huang et al., 2018), nitricacid (Kang et al., 2018). Among these, H3PO4 activation needs relatively low activation temperature and is facile and more environment-friendly, suitable for mass production. Moreover, H3PO4 activation reveal the meso-porous structures whereas those obtained from pyrolysis show to be predominant microporous structures (Kouotou et al., 2013). Hence, combination of H3PO4 activation and pyrolysis may obtain a hierarchical porous AC.
Regardless of the method, the improvement of adsorption capacity is very important for sustainable production. Hence, to make a comparison with adsorption capacity, walnut shells-based porous carbon was prepared and optimized through traditional pyrolysis, direct KOH activation and combined activation composed of hydrothermal carbonization and sequenced H3PO4 modification and pyrolysis. The adsorptive properties and pore structures were evaluated and prepared using model dyes iodine and methylene blue, compared with the commercial carbons. Specific objectives of this work are to (1) determine the feasibility of H3PO4/pyrolysis-activated hydrochar as a hierarchical porous adsorbent for the complex pollutants; (2) obtain low-cost walnut shells-based porous with high adsorption capacity.
Material and methods
Materials
Potassium hydroxide (KOH), phosphoric acid (H3PO4) and hydrochloric acid (HCl) were supplied from Hangzhou mike chemical instrument (Hangzhou, China). Iodine, tannic acid, methylene blue, sodium thiosulfate and starch were purchased from Sangon biotech (Shanghai, China). All reagents were of analytical grade and were used as received without further purification. The precursor for the activated carbon was from the walnut shells raw material (WS), which was collected from Xinjiang Province (China). WS were thoroughly cleaned by double deionized water to remove the possible dirt, and then dried in electro-thermostatic blast oven at 60°C. Cleaned and dried WS were crushed and ground into powder with size of 2–4 mm using mortar and pestle. The commercial ACs as control was purchased from Green source (Beijing, China) and Lorain.co (Shandong, China).
Synthesis of walnut shell activated carbon
To obtain KOH activated AC, the WS powders were treated with KOH compounds for 1.5 h at impregnation ratio (defined as: a mass ratio of activation agent/precursor) ranging from 1:1 to 2:3, and followed by thermal activation for 1 h at 600–900°C in muffle furnace with a heating rate of 5°C min−1. After cooling, the resulting slurry was removed from the furnace, washed sequentially with 0.1 mol/L HCl, hot water and cold deionized water till neutral PH. Finally, the adsorbents (denoted as AC-K) were filtered and dehydrated overnight in an oven at 120°C further use.
To obtain bio-bar from pyrolysis self-activation, the WS powders were placed in a covered ceramic crucible, then heated to 600–1000°C using muffle furnace under slow rate of 5°C min−1 and maintained. Finally, the produced AC (denoted as AC-P) was dried at 120°C to a constant weight and stored for further analysis.
H3PO4/pyrolysis activated hydrochar (denoted as AC-C) was obtained through hydrothermal carbonization, then followed by activated with H3PO4 and thermal activation. The powders were kept inside and activated at 120°C for 12 hours in the autoclave and soaked into H3PO4 solution (50 wt. %) for 2 h at impregnation ratio of 3:1. Then, the slurry was activated for 1 h under 600–1000°C to modify porous structures. After cooling, the chars were sequentially rinsed with 0.1 mol/L HCl, 0.1 mol/L NaOH and hot water to neutralize excessive H3PO4 compounds. Finally, the produced AC (denoted as AC-C) was dried overnight at 120°C to a constant weight and stored for further analysis.
The yields of produced AC were calculated by equation (1):
Characterization
The surface morphology of the walnut shell and the AC prepared under the optimum conditions was identified by scanning electron microscope (SEM: Hitachi S-4800, Japan) with a 3 kV accelerating voltage.
The surface features of activated carbon prepared were measured by N2 adsorption at –196°C (77 K) with an automatic surface area and porosity analyzer (3H-2000PS1, BeiShiDe Instrument). The specific surface area (SSA) and pore structure were determined according to the Brunauer-Emmet-Teller (BET) model and the Barrett-Joyner-Halenda method (Huang et al., 2014). The surface area (SA) and the porosity of the AC adsorption branches of the isotherms were determined using density functional theory. The surface functional groups were qualitatively determined the Fourier transform infrared (FTIR) spectrum on a Nicolet 5700 FTIR spectrometer (Thermo, Nicolet) in 400–4000 cm−1.
Adsorption studies
The adsorption capacities of Methylene blue (MB) and iodide by prepared walnut shell activated carbon (WSAC) were carried out by batch extraction technique according to Test method of wooden activated carbon—Determination of iodine (GB/T 12496.8–1999) and methylene blue adsorption (GB/T 12496.10–1999).
Iodine number is determined using the sodium thiosulphate volumetric method. The prepared absorbent of 0.50 g was added into10 mL of 5% (v/v) HCl solution and gently boiled for 30 s to remove any sulphur. After cooling to room temperature, 50 mL standardized iodine solution with a concentration of 0.1 mol/L is added and then filtered quickly after string for 15 min. The remaining filtrates were titrated with a standardized Na2S2O3 solution using starch solution as indicator. Iodine numbers of the commercial AC were of 175.5 mg/g and 196.8 mg/g, respectively.
MB adsorption capability is determined using spectrophotometer method. The prepared absorbents of 0.10 g were added into 20 mL of MB solution (600 mg/L) in a 100 mL glass conical beaker and shaken for 2 h in room temperature to reach equilibrium. Then the adsorbent was separated from solution by centrifugation at 3,000 rpm for 10 min and 2 mL of suspension was withdrawn and analyzed by 722 Raster Spectrophotometer (UV-2450, Shimadzu, Japan) at wavelength 665 nm. Then MB content is calculated as the absorbance from a standard curve. The MB removal can be obtained through dividing the mass of the resultant MB by the initial mass of MB in the solution. MB removals of the commercial AC were of 122.9 mg/g and 124.2 mg/g, respectively.
Single-component adsorption
One of prepared adsorbents (stream/H3PO4-activated AC) and commercial AC were used for adsorption of Fe(III) and Cr(VI) from aqueous solution. The standard stock solution of Cr(VI) and Fe(III) (0.02 mol/L) was prepared by dissolution of proper amount of K2Cr2O7 and FeCl3·6H2O in distilled water. Adsorption studies were carried out with 0.5 g sorbent per 25 mL of Cr(VI) or 20 mL Fe(III) solutions under string for 2 h for reaching the equilibrium condition. The contents of the flask were filtered through filter paper. The filtrate was analyzed for remaining Cr(VI) concentration in the sample through titrating a standard solution of ammonium ferrous sulfate with sodium diphenylamine sulfonate as an indicator. For Fe(III) adsorption procedure, EDTA was used as titrate and 10% sulfosalicylic acid solution was selected as indicator.
The amount of metal ions adsorbed per unit mass of the adsorbent (q) was evaluated by equation (2):
Results and discussion
Adsorption studies
Methylene Blue (MB) is a cationic dye widely used in coloring processes and served as a model compound for adsorption studies of organic contaminants. MB removal is closely related to the numbers of mesopores. And the iodine number represents the amount of micropores. Hence, the MB removal and iodine number were employed to determine the adsorption capacity and porous characteristics of the prepared AC.
KOH activation
The iodine number and MB removal of KOH-activated AC were shown in Figure 1. The iodine number of KOH-activated AC was increased gradually as the increase of reaction temperature, but a temperature higher than 800°C resulted in decreasing progressively. The maximum of iodine number was 740.0 mg/g, nearly 4 times than that of commercial AC (186.2 mg/g). In other hand, with temperature ranging 900°C from 800°C, the MB removal shapely increased from 115.7 to 1162.1 mg/g, 9.4 times than that of commercial AC (123.6 mg/g). Initial increased temperature caused carbon molecules depletion from the char network hence developing new pores, but the excessive temperature resulted in the destruction of the microporous structure (Yu et al., 2014). Besides, when KOH/WS ratio decreasing from 3:2 to 1:1, the iodine number reducing to 283.9 from 740.0 mg/g, it indicated the micro-pore deformation. A treatment at 800°C using KOH/WS of 1:1 was the best overall performing KOH-activated AC. All iodine numbers of the obtained AC were found to be larger than the commercial ACs, suggesting well-developed internal porosity of AC.

Iodine number and MB removal of KOH-activated AC (AC-K).
Pyrolysis self-activation
Pyrolysis often induces the precursor microporous AC without chemical reactor. As shown in Figure 2, under the condition of activation temperature of 800°C for 6 min, the iodine number reached the maximum of 317 mg/g, more than 1.7 times compared to the commercial AC. The yield of the sample was 21.46%, and the adsorption capacity of MB was 90 mg/g. With time prolonging, iodine number is obviously decreased, but the adsorption capacity of MB is almost unchanged. Besides, activation temperature has effect on the iodine number by changing the formation rate of micro-porous. It is indicated that the continuous activation of the activated carbon will increase the pore size of the activated carbon, and the collapse of the micro porous structure at high temperature. A treatment of a 6 min pyrolysis at 800°C was supposed to be the best performance for pyrolyzing AC.

Iodine number and MB removal of pyrolysis-activated AC (AC-P).
Combined activation
H3PO4 activation reveal the meso-porous structures whereas those obtained from pyrolysis show to be predominant microporous structures (Kouotou et al., 2013). Sequence H3PO4 and pyrolysis activation was expected to obtain hierarchical porous carbon AC with high absorption capability
As shown in Figure 3, iodine number is rising with the temperature being up, while the yield of AC decreased gradually. In the last preparation step, the slurry with Residual phosphoric acid was pyrolyzed under 600–1000°C for 1 h to modify porous structures. It could be the increased temperature promoting the reaction between phosphoric acid and walnut shell, thus causing the formation of a complex microstructure, and increasing of the specific surface area (Gupta et al., 2013). Notably, pyrolysis played a dominant role in this step, which induce a fivefold IN increases when the temperature is up to 1000°C. When temperature is up to 1000°C, the iodine number and MB removal increase to the maximum of 834.0 mg/g and 155.8 mg/g respectively, 4.5 times and 1.3 times that of commercial AC. A treatment consisting of 2 h activation with H3PO4, followed by a 1 h pyrolysis at 1000°C was the optimum conditions of composite activation. It has been also reported that the typical iodine number range of activated carbons is 500–1200 mg/g, equivalent to surface area between 900 and 1100 m2/g (Code & Provenzano, 2014). All iodine numbers of the obtained AC were found to be larger than the commercial AC, suitable for water purification.

Iodine number and MB removal of H3PO4/pyrolysis activated hydrochar (AC-C).
Characterization of AC under optimum conditions
Textural properties of WS-AC
The adsorption capacity of AC is related to its surface area, internal pore distribution and their structure. The N2 adsorption-desorption isotherm of WS-AC under the optimum condition are shown in Figure 4. According to the IUPAC classification, the isotherm profile could be classified as type I, which representing dense micropore structures. However, the isotherms in term of AC-C show an increasing slope below the relative pressure of 0.4, indicating an appreciable amount of mesopores (Kundu et al., 2012). The H4 hysteresis loop indicates the co-existence of micro and meso-pores.

N2 adsorption (close symbols) and desorption (open symbols) isotherms of WS-AC under the optimum condition.
As displayed in Table 1, the total pore volumes were ranged from 0.23 to 0.96 cm3/g. The average pore diameter was all over 2 nm, indicated highly mesoporous carbons. The total BET surface area ranged from 368.9 m2/g to 1689.1 m2/g. The SBET was in the order: AC-K > AC-C >> AC-P. In comparison, the surface areas of activated carbons derived from other biomaterials are: 200 ∼ 1,300 m2/g from sugar beet pulp activating with carbon dioxide. AC-K and AC-C presented a high SBET value of 1689.1 m2/g and 1545.3 m2/g, remarkably higher than the water standards for the treatment of municipal and industrial wastewater.
Yields and the pore structural parameters of the carbons.

Micropore volume (Vmic), micropore surface area (Smic) and external surface area (Sext) were calculated using the t-plot method. Total pore volume (Vtot) was calculated using DFT method. Average pore diameter (Dp) was calculated as Dp = 4 Vtot/SBET (SBET =mean ± standard deviation).
Surface chemistry analysis
Adsorption characteristics of AC are determined by its porosity and surface chemistry. Surface FTIR analysis provides qualitative information on the functional groups of WSAC. As shown in Figure 5, the spectrum of WSAC displays slightly different characteristic peaks as a result of the carbonization and activation.

The FT-IR spectra of raw walnut shell (a) and prepared activated carbons (b).
The raw walnut shell (RWRM) showed a complex FTIR spectrum (Figure 5(a)), similar to lignocellulosic materials (Abdel-Ghani et al., 2016). The strong broad band at 3500 cm−1 indicates O-H stretching vibration of hydroxyl groups. The band nearly at 2925 cm−1 can be assigned to C-H sp3 stretching bond of Alkane (Azmi et al., 2016). The bands at 2352 cm−1 correspond to ν (C≡C) vibration of alkyne group. The bands at 1742 and 1617 cm−1 at walnut shell spectrum can be attributed to the C = C stretching vibration of aromatic carboxyl groups (Hashemian et al., 2013). The bands in the region between 1370 and 900 cm−1 indicated C–O stretching in alcohol, carboxylic acid, esters and ether (Moreno-Barbosa et al., 2013).The broad intense band between 1100 and 1043 cm−1 at walnut shell spectrum correspond to C–O vibrations in secondary and primary R–OH groups in alcohol, finally the band at 512 cm−1 is described to O–H band (Fan et al., 2013).
In AC spectrum (Figure 5(b)), the peaks at 3000 cm−1, 841 cm−1,933 cm−1are identical to the presence of C-H (aromatic ring), and the peaks at 1510 cm−1 correspond to C-C (aromatic ring) (Ghasemi et al., 2015). The band of alcohol is disappeared, and production of benzene derivatives indicate activation process partially being destroyed the bonds. Especially in pyrolytic AC (AC-P), the strong bands are inferred the high activation temperature being caused substitution aromatic rings (Hesas et al., 2013). Besides, The shoulder at 1080–1070 cm−1 in AC-P spectrum may due to the symmetrical vibration in a chain of P-O-P (polyphosphate) (Kyzas et al., 2016).
Surface morphologies
In order to study its surface texture and pore development, SEM was carried out on the raw WS and its ACs. As shown in Figure 6, raw WS contain few pores and rod-like cellulosic structure which is consists of lignin, cellulose and hemicellulose (Soleimani & Kaghazchi, 2007). The WSAS presents rougher surface with cavities, indicating a well-developed porous structure (Figure 6(b) to (d)). The evaporation of impregnated K metal developed peculiar microspheres features (Figure 6(b)), in agreement with a study on KOH-activated carbon by Bedin et al. (2016). The similar cavities in Figure 6(c) and (d) are resulted from the investigated temperature in the range of 500–800°C, which inducing the destruction of cellulosic structure and the development of a porous surface features. It has been reported that hemicellulose, cellulose and lignin occur decomposition in the temperature ranges of 180 ∼ 240, 230 ∼ 310 and 150 ∼ 750°C respectively (Gucho et al., 2015). Phosphate treatment (Figure 6(c)) did not affect carbon skeleton but induced more cavities with sizes of 2 ∼ 3nm. The cavities on AC surface provide suitable channels for molecules passage into the carbon structure.

SEM image for raw walnut shell (a), AC prepared with KOH activation (b), composite activation (c) and pyrolysis self-activation (d).
High potential of ACs for adsorption of heavy metals
Chromium (Cr) and iron (Fe) are widely used in the industries and usually discharged. Here removal of Cr(VI) and Fe(II) were investigated. As shown in Figure 7, adsorption amount of Cr(VI) lies above the Fe(II) due to the smaller radius of Cr(VI) in hydrated state and also its higher electronegativity (Ghasemi et al., 2015). Moreover, the prepared H3PO4/pyrolysis activated hydrochar and KOH-activated AC could remove (16.62 and 16.66) mg/g of Fe(III), respectively, (92.56 and 90.37) mg/g of Cr(VI), respectively, higher than those of the commercial AC (7.39 mg/g of Fe(III) and 80.01 mg/g of Cr(VI)). These results proven that prepared low-cost AC in the present study offers a promising adsorbent for water treatment in the future.
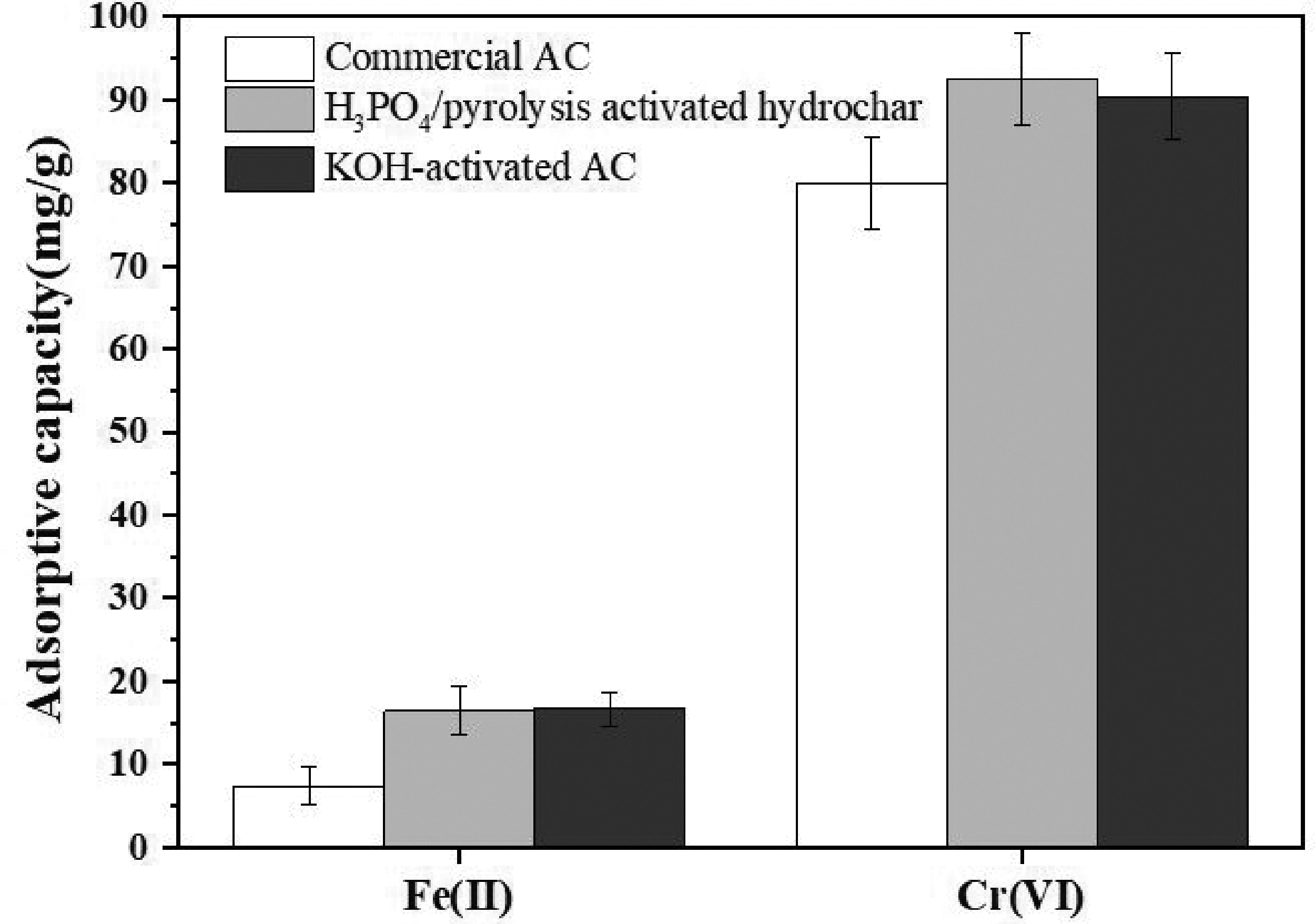
Cr(VI) and Fe(III) adsorption data in the single solution on the optimized H3PO4/pyrolysis activated hydrochar, KOH-activated AC and commercial AC.
Conclusions
In this work, the ACs with the regulated mesopores were successfully prepared from walnut shell waste via chemical activation with KOH at impregnation ratio of 1:1 and combined activation comprising of H3PO4 activation and a 1 h pyrolysis at 1000°C. The adsorption capacities of the produced AC have 9.4 and 1.3 times of MB removal, 4 and 4.5 times of iodine number than commercial ACs, respectively. ACs prepared at the optimum conditions has a BET surface area (1689.1 m2/g, 1545.3 m2/g) and mesoporous percent (50.76%, 99.45%). And, they are could remove Fe(III) and Cr(VI) more effectively in water than commercial ACs. Moreover, the N2 adsorption-desorption isotherm of H3PO4/pyrolysis activated hydrochar suggested the co-existence of micro and meso-pores. Conclusively, the produced AC from walnut shell waste is an economic and promising alternative to the purification of complex pollutants.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Zhejiang Provincial Natural Science Foundation of China (LY18C100002), the Fundamental Research Funds of Zhejiang Sci-Tech University (2019Q051) and Foundation of Education Committee of Zhejiang Province (Y201942241).


